Abstract
Hemorrhagic transformation after cerebral ischemia is a well known clinical concern. The frequency of intact basal lamina (BL), identified by laminin antigen, in hemorrhagic and nonhemorrhagic zones after middle cerebral artery occlusion (MCA:O) and 3-h MCA:O with reperfusion in adolescent male baboons was assessed. Parenchymal hemoglobin was not detected prior to 24-h reperfusion. A significant decrease in the density of laminin (BL) in hemorrhagic zones (6.2 ± 2.4) compared with nonhemorrhagic ischemic zones (10.5 ± 2.4) (p < 0.05) and nonischemic basal ganglia (17.0 ± 2.7) (p < 0.01) was observed. Time-dependent changes in BL integrity appear linked to the extravasation of blood components.
Loss of vascular integrity during and after an ischemic stroke underlies experimentally and clinically important changes (Meyer, 1958; Zülch, 1985; del Zoppo et al., 1990; Okada et al., 1994). Together with edema formation, the extravasation of blood cellular elements is a major sign of the disturbed vascular integrity (Pessin, 1991). Hemorrhagic transformation (extravasation of blood cells) may be classified as either hemorrhagic infarction (HI) without clinical deterioration or as parenchymal hemorrhage or hematoma (PH) (del Zoppo et al., 1988, 1995).
The clinical use of plasminogen activators (PAs) has stimulated concerns about the development of secondary hemorrhagic changes after cerebral ischemia (del Zoppo et al., 1990; Levy et al., 1994; Hacke et al., 1995; National Institutes of Neurological Disorders and Stroke rt-PA Stroke Study Group, 1995). Hemorrhagic transformation was detected in up to 37% of patients with ischemic stroke within the initial 24 h of symptom onset (Hacke et al., 1995). Among eight recent angiography-based acute PA intervention studies in thrombotic and thromboembolic stroke, HI occurred in 4.6–42.1% and PH occurred in 0–16.7% (del Zoppo et al., 1988, 1992; Mori et al., 1988; Theron et al., 1989; Matsumoto and Satoh, 1991; Mori et al., 1992; Yamaguchi, 1993; Zeumer et al., 1993). Two recent prospective nonangio-graphic PA trials have reported incidences of 2.9 or 30.3% HI and 0.6 or 6.5% PH in the placebo groups (National Institutes of Neurological Disorders and Stroke rt-PA Stroke Study Group, 1995; Hacke et al., 1995). A local disturbance of cerebral vascular integrity of ischemia may play the key role in the transformation to PH (Meyer, 1958). The nature of the microvascular changes is not known. Furthermore, there is a paucity of adequate animal model data that prospectively follow the evolution of either type of hemorrhage.
Integrity of the cerebral microvasculature is provided mainly by two different anatomical and functional barriers: the blood-brain barrier [provided by the interendothelial tight junctions of capillary and postcapillary venules (Gregoire, 1989)] and the basal lamina (BL). The BL consists of a network of type IV collagen and laminin polymer connected by entactin (Martinez-Hernandez and Amenta, 1983; Yurchenko and Schittny, 1986; Mohan and Schittny, 1990). As a part of the extracellular matrix (ECM), the BL is connected by fibronectin of cellular origin and laminin to the endothelium (Martinez-Hernandez and Amenta, 1983; Bruijn et al., 1988). Cerebrovascular ECM and BL are interdigitated throughout the medial smooth muscle and associated with perivascular astrocytes (Peters et al., 1991). The BL forms an effective barrier to the transmigration of polymorphonuclear leukocytes (Mainardi et al., 1980; Heck et al. 1990; Granger et al., 1995). During experimental focal cerebral ischemia in the baboon, a continuous disappearance of antigens of the major BL components laminin, collagen (IV), and fibronectin occurs (Hamann et al., 1995).
The hypothesis tested by this study is that loss of BL integrity, evidenced by loss of laminin antigen, is at least in part associated with leakage of erythrocytes into the surrounding tissue.
METHODS
All animal procedures were approved by the Animal Research Committee of the Scripps Research Institute and were performed in accordance with standards published by the National Research Council (The Guide for the Care and Use of Laboratory Animals), the National Institutes of Health Policy on Human Care and Use of Laboratory Animals, and the U.S. Department of Agriculture Animal Welfare Act. The principal investigator, veterinarians, and the primate handling staff were present for all procedures.
Thirteen male adolescent baboons were used for the middle cerebral artery occlusion (MCA:O) and MCA:O/reperfusion (MCA:O/R) experiments and three animals served as unoperated controls. The experimental protocol and procedures have been previously described (del Zoppo, et al., 1986b, 1991; Okada et al., 1994). After the surgical implantation procedure, a 7-day interval for observation and recovery was allowed in each animal. Experimental ischemia and reperfusion was performed as 2-h MCA:O (n = 3) and 3-h MCA:O with 1 (n = 3), 4 (n = 3), and 24 (n = 4) h of reperfusion. Neurological outcomes were assessed according to standard scoring instruments (del Zoppo et al., 1986a).
Each experiment was terminated by left ventricular transcardiac perfusion with isosmotic perfusion fluid containing heparin (2,000 IU/L), sodium nitroprusside (6.7 μmol/L), and bovine serum albumin (50 g/L) under Na+ pentothal anesthesia. One to three coronal specimens (1 × 1 × 0.2–0.5 cm) from symmetrically located sites of both basal ganglia and temporal cortex were embedded in Tissue-TEK OCT compound (Miles, Elkhart, IN, U.S.A.), frozen in 2-methyl-butane/dry ice, and stored at −80°C in preparation for sectioning immunohistochemistry.
Antibodies
Hemorrhagic zones were detected with a rabbit anti-human hemoglobin (Hgb) antibody A118 (Dako, Carpinteria, CA, U.S.A.) that cross-reacted with Hgb A, AS, and F. BL laminin was exposed by the murine anti-human laminin monoclonal antibody LAM89 (Sigma, St. Louis, MO, U.S.A.). In immunofluorescence colocalization experiments, the anti-Hgb IgG was compared to LAM89 with appropriate fluorescein isothiocyanate and TRITC secondary antibodies.
Immunohistochemistry
The immunohistochemical procedures and standard controls have been detailed elsewhere (Okada et al., 1994). All frozen sections were fixed with acetone to permeabilize erythrocytes and other cells.
Videoimaging microscopy
The numbers of peroxidase-stained vessels and their minimum transverse diameters were determined with the aid of a computerized videoimaging system (del Zoppo et al., 1991; Okada et al., 1994). The number and relative size classification [internal diameter (del Zoppo, 1994)] of microvessels were determined in whole microscopic fields (area = 0.77 mm2) ischemic and nonischemic basal ganglia, and corresponding cortex with videoimaging microscopy.
Immunofluorescence colocalization studies were assessed by fluorescence microscopy. For the purpose of this study, regions of interest (ROIs) were defined as those areas of tissue displaying fluorescein isothiocyanate fluorescence consistent with the presence of Hgb in the ischemic zones. ROIs varied (21–175 whole microscopic fields or 16.2–134.8 mm2) depending upon the size of the hemorrhage. ROIs for each section were compared with regions of the same size (corresponding number of microscopic fields) without Hgb in the ischemic zone and regions in the nonischemic basal ganglia. The absolute numbers of vascular structures/microscopic fields were corrected for the specific reduction in neuron density in the same fields associated with ischemia. Consistent with the previous report, neuron density decreased to 0.81 ± 0.06 at 24-h reperfusion (Hamann et al., 1995).
Statistics
Data are presented as means ± SD. Wilcoxon morphometric rank sum tests were used to compare the number of microvascular silhouettes in ischemic and nonischemic tissues in relation to hemorrhage. Statistical evaluations were performed using a one-way analysis of variance
RESULTS
Hgb antigen was not detected in control subjects or in the ischemic zone of subjects undergoing 3-h MCA:O until 24-h reperfusion (Fig. 1). At 24-h MCA:O/R, hemoglobin was detected in petechial and confluent petechial patterns.
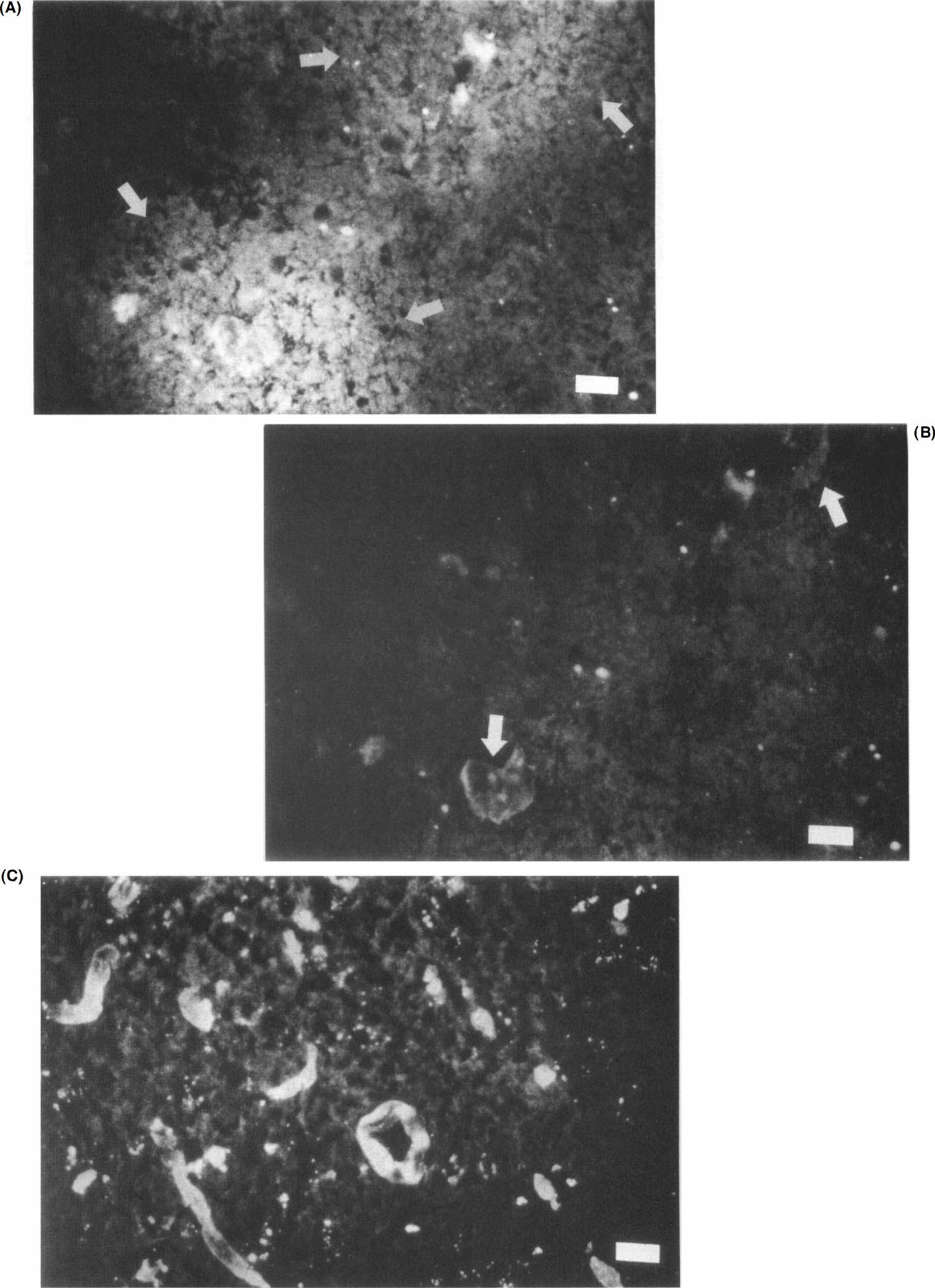
Hemorrhagic transformation in basal ganglia at 24-h reperfusion following 3-h middle cerebral artery occlusion.
At 24-h reperfusion, no difference in neurological impairment between subjects with or without hemorrhage was observed.
The relative number of microvessels per field defined by the laminin antibody LAM89 varied with the presence of Hgb (Table 1). A marked reduction in the normalized density of microvessels between the nonischemic basal ganglia (17.0 ± 2.7) and the contralateral ischemic zones where hemorrhage did not occur (10.5 ± 2.4) (2p = 0.005) was observed. The microvascular density in the hemorrhagic zones (6.2 ± 2.4) was significantly lower than in the nonhemorrhagic zones of the ischemic basal ganglia (10.5 ± 2.4) (2p = 0.013).
Microvessel density
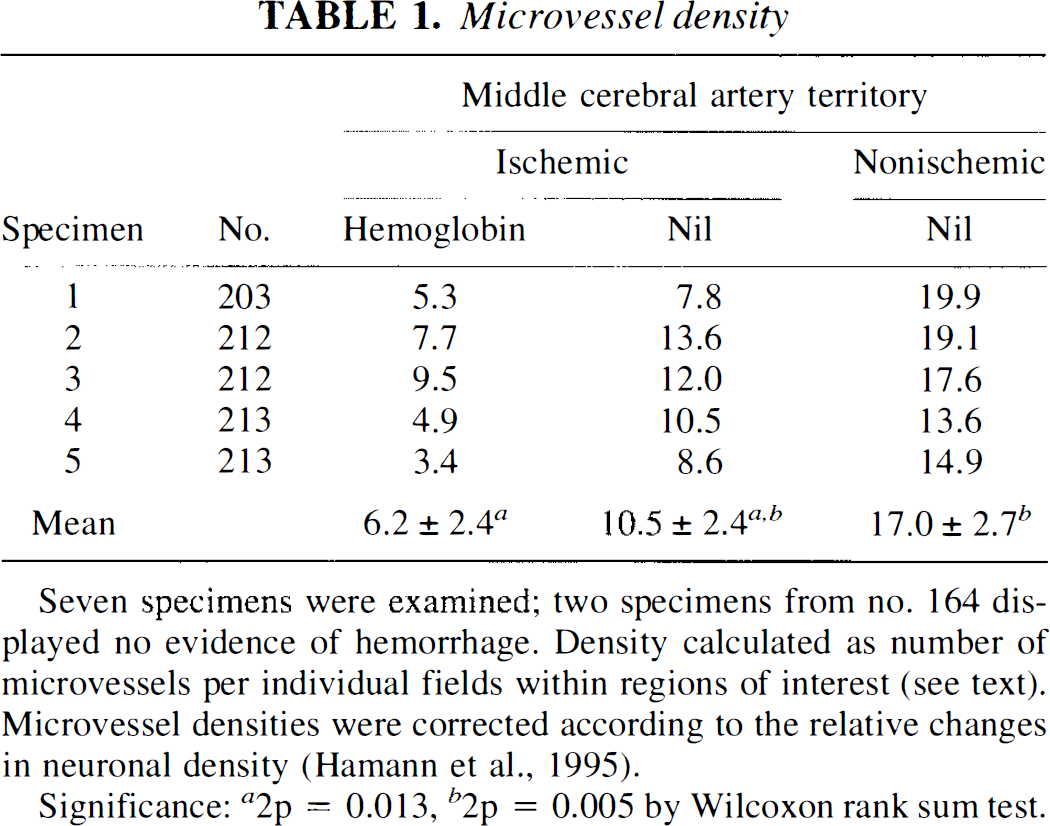
Seven specimens were examined; two specimens from no. 164 displayed no evidence of hemorrhage. Density calculated as number of microvessels per individual fields within regions of interest (see text). Microvessel densities were corrected according to the relative changes in neuronal density (Hamann et al., 1995).
Significance: a2p = 0.013, b2p = 0.005 by Wilcoxon rank sum test.
There was no significant difference in the distribution of microvessel diameters between the hemorrhagic and the nonhemorrhagic areas within respective ROIs. All capillary and postcapillary venule/precapillary arteriole classes were equally affected. Disruption in the microvessel wall in the center of hemorrhagic ROI could be seen (Fig. 2).
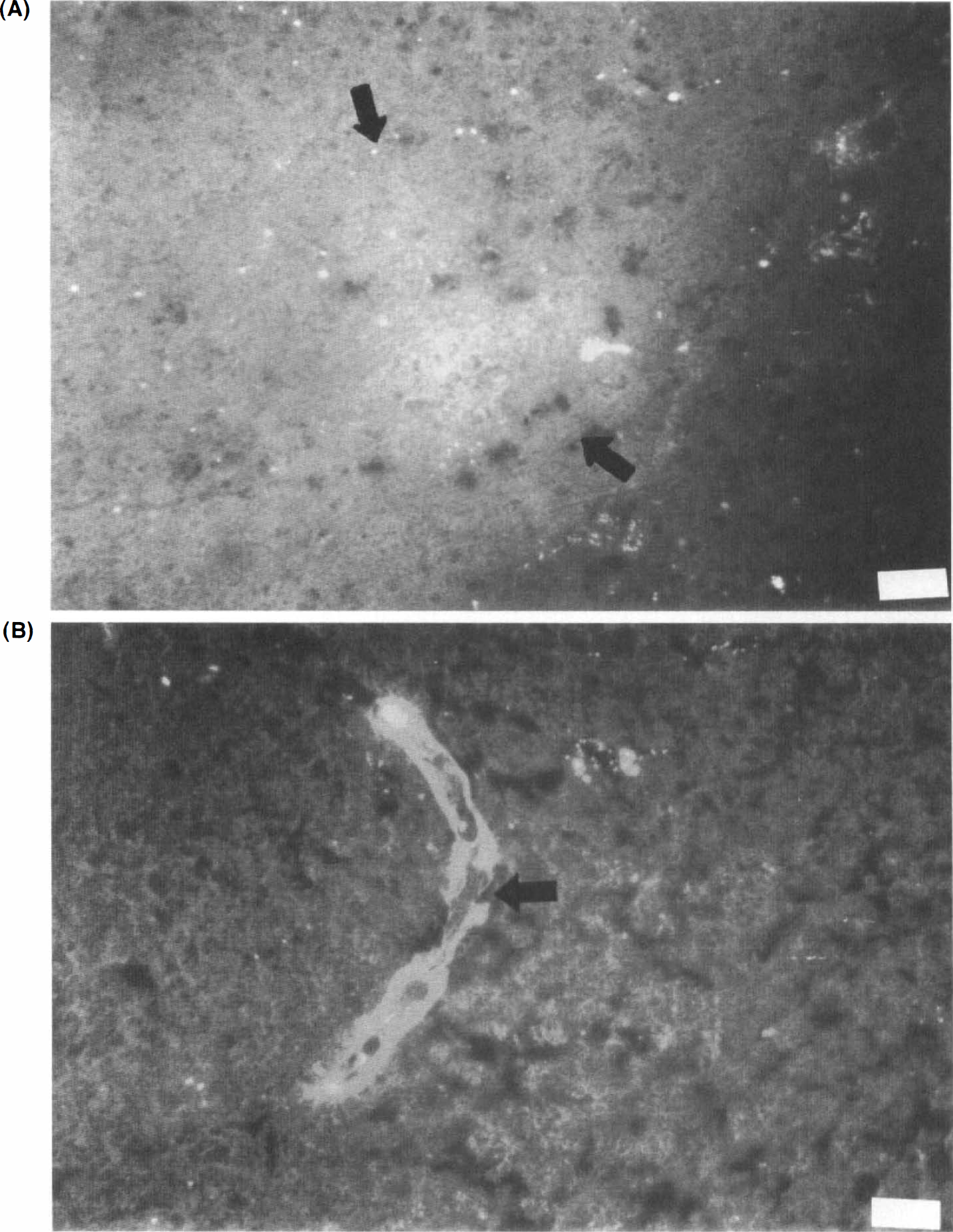
Hemorrhagic transformation in basal ganglia at 24-h reperfusion following 3-h middle cerebral artery occlusion.
DISCUSSION
The finding of petechial hemorrhage in the nonhuman primate only at 24-h reperfusion after 3-h MCA:O is consistent with increasingly impaired vascular integrity in experimental cerebral ischemia (Hamann et al., 1995). This finding is also consistent with the clinical observation of HI observed by 24 h following ischemic stroke in humans (Pessin, 1991). Some of the microvessel-associated hemorrhagic zones found in this study may be the precursors of larger disturbances seen consistently as PH in humans. The significant decrease of microvascular structures defined by laminin provides a direct hint that changes in the BL integrity and ECM after cerebral ischemia/reperfusion may be connected to the extravasation of blood cells and blood components (Okada et al., 1994). The reliability of Hgb deposition as a sign of microvascular hemorrhage was confirmed by preliminary experiments in which anticoagulated whole blood was injected into primate brain tissue ex vivo, with immediate appearance of Hgb antigen with the techniques used here. Given the appearance of Hgb at 24-h reperfusion, it is not certain whether the number and volume of hemorrhages might increase with reperfusion time (del Zoppo et al., 1990). However, the processes appear to begin shortly after the onset of focal ischemia.
Although the molecular bases for these changes were not directly investigated here, leukocyte transmigration and/or the expression or release of proteolytic enzymes may contribute to loss of specific BL components. The latter include gelatinases and the serine protease plasmin, which may digest ECM and alter the blood-brain barrier (Rudd et al., 1991; Rosenberg et al., 1994). This may imply a somewhat different mechanism for hemorrhage from that postulated for the appearance of hemorrhage in postmortem cerebral tissues (Fisher and Adams, 1951, 1987). From those tissues, the migration of emboli in the principal cerebral artery was hypothesized to expose the downstream “ischemic” vessel wall segment to arterial pressure, with consequent rupture. The relative decrease of microvessel density in hemorrhagic areas scattered among nonhemorrhagic ischemic areas suggests that significant loss of BL integrity is focal. These findings, together with evidence of ECM and BL disintegration (Hamann et al., 1995), suggest the alternative hypothesis that hemorrhagic infarction results from compromised microvascular BL integrity, which may involve a number of cellular mechanisms.
Microvascular damage, which progresses in selected ischemic areas, may underlie later, more clinically apparent hemorrhage. These results highlight the potential importance of BL integrity to the pathophysiological cascade leading to the hemorrhagic complications. The specific timing of BL loss and erythrocyte extravasation, important underlying mechanisms of BL degradation, and the possibility that reperfusion may increase erythrocyte extravasation suggest avenues for future investigation.
Footnotes
Abbreviations used
Acknowledgments:
This work was performed with the support of grant NS 26945 of the NINDS and grant Ha 2078/2-1 of the Deutsche Forschungsgemeinschaft (D.F.G.). We thank Pearl Akamine for her expert technical and laboratory expertise and James A. Koziol, Ph.D., for his assistance with statistical analyses.
