Abstract
Microvascular basal lamina damage has been demonstrated after balloon occlusion of the middle cerebral artery in the nonhuman primate and after intravascular filament occlusion in the rat. The aim of the present study was to investigate in the rat whether microvascular damage can be found in the stroke model of intracarotid clot injection as early as 3 hours after clot injection and whether microvascular damage relates to the level of regional cerebral blood flow (rCBF). Microvascular densities and total stained microvascular areas were determined by immunohistochemistry of collagen type IV in cortex and basal ganglia and automatic video-imaging analysis. rCBF was measured by autoradiography in the same brain areas. Compared with the corresponding areas in the nonischemic hemisphere, a significant loss of microvascular density (−16%) and total stained microvascular areas (−10%) was observed in these areas. The reduction of microvascular basal lamina staining was comparable in all animals and was not related to the value of rCBF when measured 3 hours after onset of embolic stroke. In conclusion, microvascular damage occurs as soon as 3 hours after intracarotid clot injection, even in brain areas in which rCBF has returned to normal values.
Keywords
Neuronal and parenchymal damage is a consequence of cerebral ischemia and reperfusion (Dietrich, 1998; Dirnagl et al., 1999). Consequently, therapeutic strategies have focused upon the prevention of neuronal damage (Liebeskind and Kasner, 2000). More recently, it became apparent that morphologic damage is not confined to neurons and parenchyma because microvascular changes were observed after experimental stroke (Del Zoppo and Hallenbeck, 2000; Petty and Wettstein, 2001). The microvascular analysis had different aspects. One aspect concerns inflammatory reactions, including leukocyte adhesion and migration, the expression of endothelial cell adhesion receptors, and the detection of different cytokines (He et al., 2000; Petty and Wettstein, 2001; Ritter et al., 2000). Another aspect is focused upon the perfusion state of capillaries. A progression of microvascular perfusion deficits with time has been found (Vogel et al., 1999a). Finally, microvascular structure and integrity were investigated. The integrity of the basal lamina of the microvessels was found to be essential for the prevention of brain edema and hemorrhagic transformation (del Zoppo et al., 1998; Hamann et al., 1996; Rosenberg, 1999). Petechial hemorrhage was closely related to damaged cerebral microvessels. In addition, extravasation of cellular blood elements was confined to regions of microvascular basal lamina loss (Hamann et al., 1996). Microvascular basal lamina damage was observed as soon as 2 hours after ischemia and was aggravated with time (3 hours ischemia and 24 hours reperfusion) in the model of external balloon occlusion of the middle cerebral artery in the nonhuman primate (Hamann et al., 1995). In this study, a reduction of microvascular structures by approximately 50% was found in the basal ganglia when detected by immunohistochemistry of various basal lamina antigens like laminin, fibronectin, and collagen type IV. Recently, these results could be confirmed in an intravascular filament occlusion model of the rat (Hamann et al., 2002). Western blots showed a severe loss of collagen type IV content in the rat brain after ischemia.
The present study had two aims: to test whether microvascular basal lamina damage in cortex and basal ganglia (1) occurs in an embolic stroke model of the rat and (2) is related to the regional cerebral blood flow (rCBF) 3 hours after onset of embolic stroke.
MATERIALS AND METHODS
Surgical procedure
Animal experiments were approved (Regierungspraesidium Karlsruhe, Germany) and were performed according to the “National Institute of Health – guidelines for the care and use of laboratory animals”. We used male Wistar rats (n = 21) between 280 and 380 g body weight. Animals were kept under diurnal lighting conditions and were allowed access to food and water ad libitum. Animals were anesthetized with 1.7% isofluran in a 70%/30% mixture of N2O/O2. Catheters were placed in the femoral artery and vein for the monitoring of blood pressure, for blood sampling, and for the intravenous application of saline and recombinant tissue plasminogen activator (rt-PA).
After surgical preparation of the right carotid artery, occlusion of the pterygo-palatine artery, and ligation of thyroidal branches of the external carotid artery (ECA), a PE 50 catheter was placed retrograde into the ECA. The tip of the catheter was placed shortly before the bifurcation of the ECA/internal carotid artery (ICA), allowing an unrestricted blood flow through the common carotid artery (CCA) into the ICA. The catheter was continuously perfused (1 mL/h) with 0.9% saline to avoid occlusion of the catheter. After closing the right CCA temporarily, we injected 12 blood clots via the catheter into the ICA to achieve a thromboembolic occlusion of the middle cerebral artery (MCA). An immediate decrease of rCBF in the cortex confirming thromboembolic stroke was monitored in all rats with a laser flow Doppler-system (Perimed, Sweden). A 1-hour treatment with intravenous (n = 7) or intracarotid (n = 7) infusion of human rt-PA (Alteplase, Boehringer Ingelheim, Germany) was started with a 10% bolus 1 h after injection of the clots. A dosage of 10 mg rt-PA per kg body weight in a total volume of 1.0 mL saline was used; control animals (n = 7) received 1.0 mL of saline only. Surgical procedures were similar in the control and in the rt-PA-treated groups.
Clot preparation
At the beginning of the experiment, 0.3 mL of arterial blood was mixed in a syringe with 0.075 mL saline containing 2.25 NIH units of rat thrombin (Sigma, Munich, Germany). After approximately 1 minute, the mixture had coagulated and was injected into a PE50 catheter where it remained for 1 hour at room temperature. The clot string was ejected from the catheter with saline and washed with saline several times for another hour. Part of the clot string lost most of its erythrocytes and thus appeared somewhat white in color. The white part of the string, mostly, was cut into pieces 1.5 mm in length shortly before intracarotid injection. Twelve pieces of mostly white clot material were used in each rat.
Autoradiography
In all animals, rCBF was measured 3 hours after injection of the clot emboli according to the method described by Sakurada et al. (1978). 4-iodo-N-methyl-(14C)antipyrine (125 μCi/kg) (specific activity 50 to 60 mCi/mmol; Biotrend, Cologne, Germany) in 1 mL of saline was continuously infused at a progressively increasing infusion rate for a period of 1 minute. During the 1-minute infusion period, 10 to 16 timed blood samples were collected in drops from the free-flowing arterial catheter directly onto filter paper discs (1.3 cm in diameter) that previously had been placed in small plastic beakers and weighed. The samples were weighed and radioactivity was estimated with a liquid scintillation counter (Packard Tri-Carb 4000 Series) after extraction of the radioactive compound with ethanol. After the 1-minute infusion and sampling period, the animal was decapitated, and the brain was removed as quickly as possible and frozen in 2-methylbutane chilled to −45°C. The frozen brains were coated with chilled embedding medium (Lipshaw Manufactoring Co., Detroit, MI, U.S.A.), stored at −65°C in plastic bags, separated into 20-μm sections at −22°C in a cryostat, and autoradiographed along with precalibrated (14C)methylmetacrylate standards. Local tissue concentrations of 14C were determined from the autoradiographs by densitometric analysis with an image analysis system (MCID, Imaging Research, Brock University, St Catharines, Ontario). Local rates of rCBF were calculated from the local tissue concentrations of 14C and the time course of the blood iodo-(14C)antipyrine concentrations, including corrections for the lag and washout in the arterial catheter as described by Frerichs et al. (1994). Washout correction rate constant was 100/minute. A brain-blood partition coefficient of 0.9 was used for the final calculations.
Silver staining
Cryosections adjacent to the corresponding autoradiography sections were used for silver infarct staining. This staining allows a reliable detection of ischemic brain tissue as soon as 2 hours after MCAO. The staining protocol is described in detail by Vogel et al. (1999b).
Immunohistochemistry
Cryosections adjacent to the corresponding silver staining sections were used for immunohistochemistry. Microvascular damage was localized by immunohistochemical methods using a pooled antiserum against bovine and human collagen type IV from goat (DUNN, Southern Biotechnology Associates, Inc., U.S.A.). This antibody also reacts with collagen type IV from rats (Hamann et al., 2002).
Staining protocol
Sections were fixed with acetone and chloroform (1:1) for 5 minutes at room temperature, immersed in 10 mmol/L glycine in phosphate buffered saline (PBS: 8.3 mmol/L Na2HPO4, 3.2 mmol/L KH2PO4, and 123 mmol/L NaCl, pH 7.4) for 5 minutes, and rinsed several times (3 × 5 minutes) in PBS before being incubated for 20 minutes at 4°C with blotto to reduce unspecific binding (blotto: 50 g nonfat dried milk, 1 mL horse serum, and 0.3 mmol sodium azide in 1 L TRIS saline: 38.5 mmol/L TRIS and 150 mmol/L NaCl, pH 7.4).
The consecutive sections were then incubated with the primary antibodies, either against bovine or human collagen type IV from goat. Antibodies were diluted 1:800 in reagent (400 mg bovine serum albumin and 0.06 mmol thimerosal in 1 L TRIS saline), first for 2 hours at 37°C and then additionally for 12 hours at 4°C. Sections were washed several times in PBS between the different incubation steps. The biotinylated secondary antibodies (antigoat, Jackson ImmunoResearch Laboratories, Inc., U.S.A.) were further incubated for 30 minutes at 37°C. They were diluted 1:200 in a solution of PBS, horse serum, and 10% tween (1000:15:1). For subsequent development with the peroxidase technique, it was necessary to block the activity of endogenous peroxidases by incubating them with H2O2 (1 mL/L PBS of 30% H2O2) for 20 minutes at room temperature. The Vectastain-Elite-Kit (Vector Laboratories, U.S.A.) was used to incubate sections with the avidin-biotin (AB) complex (concentrations according to the manufacturer's instructions) for 30 minutes at 37°C. Subsequently the peroxidase activity was detected with 3-amino, 9 ethyl-carbazole (AEC; concentrations according to the manufacturer's instructions; AEC-Kit, Biomeda Corporation, U.S.A.).
Quantification of microvascular damage by video-imaging microscopy
First an autoradiogram of cortex and basal ganglia was digitized with a CCD-camera, then the grey values were mapped into blood flow data (MCID, Imaging Research, Brock University, St. Catharines, Ontario), and finally the image was imported into OPTIMAS and the regions of interest (ROI) were delineated (basal ganglia and cortical area) (Fig. 1, left panel) using the OPTIMAS 6.5 image analysis system (Media Cybernetics, L.P., Silver Spring, MD, U.S.A.). Basal ganglia and cortex were defined according to the anatomic landmarks (Paxinos and Watson, 1998) as described by Hamann et al. (2002) (Fig. 1, left panel). Within these two regions of interest, measurements of rCBF were confined to those areas in which a maximal decrease in rCBF could be observed in the autoradiograms (Fig. 1, right panel) when compared with the contralateral side. Then the complete section stained for collagen type IV was digitized with a Zeiss Axiophot microscope equipped with a fivefold objective using a Sony Power HAD 3CCD color video camera (Sony, Tokyo, Japan). The image was imported into OPTIMAS. Subsequently, the masks of the most affected areas obtained from the autoradiograms were transferred to the collagen-stained section. The damage of the microvessels was verified from the number of vessels per unit area (vascular density) and the stained vascular area. Both were related to the total area of measurement. These values of the ipsilateral side were divided by the corresponding values obtained from the contralateral control (nonischemic) side. An edema correction factor from each digitized section was used to normalize the vessel density per area of the ipsilateral side (Hamann et al., 2002).
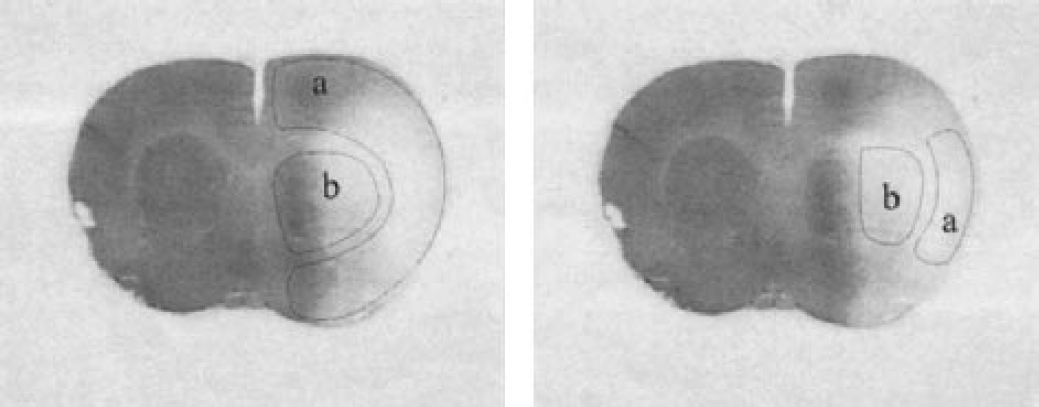
Location of regions of interest in an autoradiogram. Left panel, delineation of cortex
Statistics
Data are expressed as means ± standard deviation (SD). Group comparison (between controls, intravenous rt-PA-, and intraarterial rt-PA-treated animals) was performed by analysis of variance. The vascular density and the vascular fraction of the ipsilateral side was compared with the contralateral side by Student's paired t-test.
RESULTS
The three groups of animals (controls, intravenous rt-PA, and intraarterial rt-PA) did not show significant differences in the measured microvascular parameters and in rCBF values. Therefore, the results of all three groups were pooled.
rCBF in the selected areas within the ipsilateral cortex and basal ganglia showed a wide variation from almost zero to more than 150 mL/100g/min between animals when measured 3 hours after onset of embolic stroke (Fig. 2). Average rCBF in the selected areas within the ipsilateral cortex was 35 ± 47 mL/100g/min (n = 21); within the ipsilateral basal ganglia, it was 43 ± 52 mL/100g/min (n = 21).
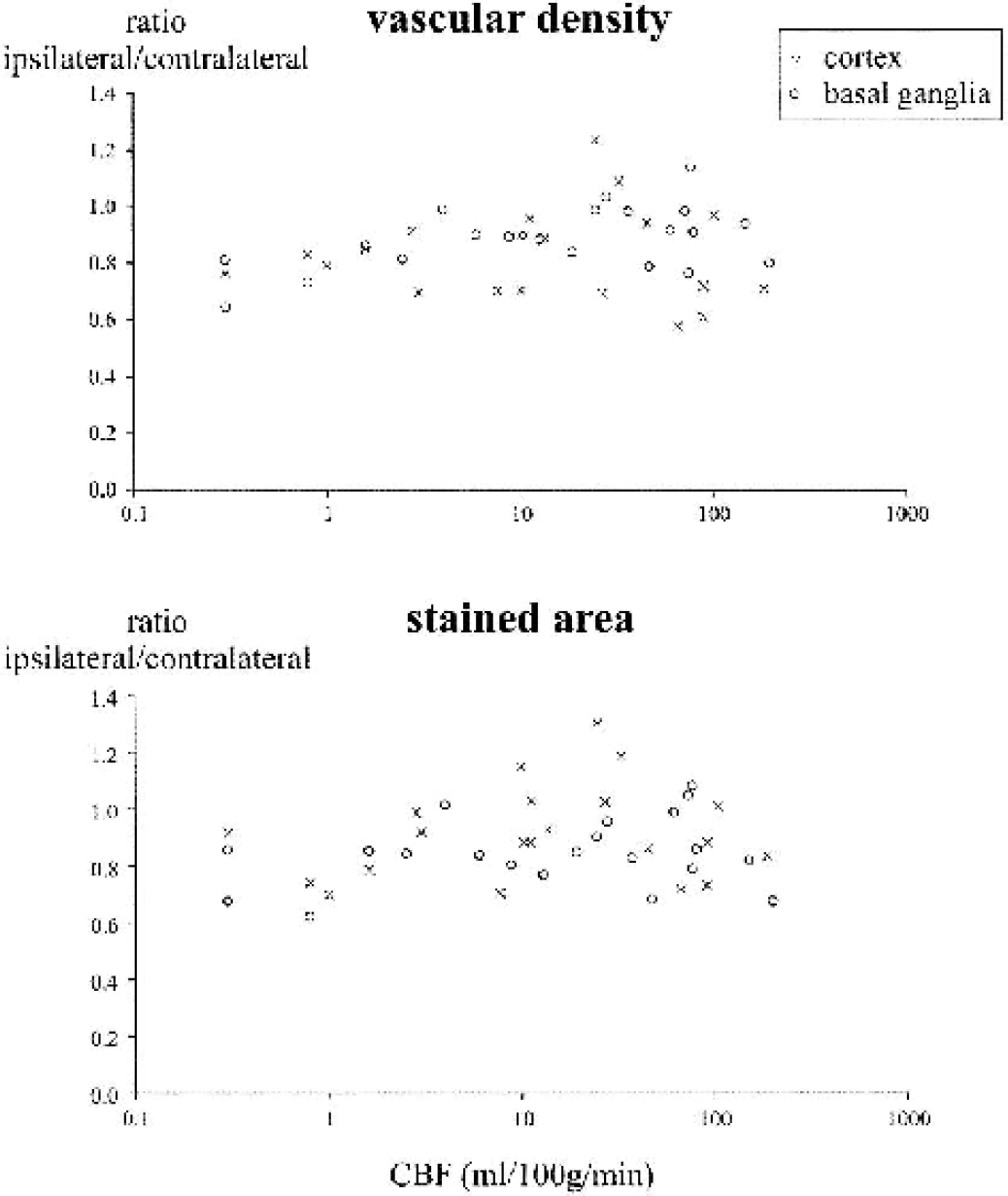
Vascular densities (upper part) and stained vascular areas (lower part) as related to the rCBF in the corresponding brain locations. Decreases in vascular density and total stained area were calculated as the ratio between ipsilateral and contralateral values. The reduction in the vascular density and in the stained area in the analyzed brain regions was not related to a corresponding reduction in rCBF when measured 3 hours after onset of embolic stroke. rCBF, regional cerebral blood flow.
Vascular density in the same areas decreased to a mean of 84 ± 14% (P < 0.01) (83 ± 16% in cortex and 85 ± 12% in basal ganglia). The total stained microvascular area decreased to 90 ± 14% (P < 0.01) (91 ± 16% in cortex and 89 ± 11% in basal ganglia). The values were related to the corresponding contralateral side, which was taken as 100%.
When rCBF was plotted against the vascular density (Fig. 2, upper part) or the total stained microvascular area (Fig. 2, lower part), no correlation was found between these vascular parameters and the values of rCBF.
Silver staining was used to detect ischemic brain tissue. In cortex, a positive silver staining was observed in 20 out of 21 animals. In basal ganglia, 16 out of the 21 animals investigated showed a positive staining. When the anatomic regions that displayed a positive silver staining were relocated in the corresponding autoradiograms, cerebral blood flow was decreased in these regions, compared with the contralateral side in the autoradiogram.
DISCUSSION
The present study has two main results. First, after intracarotid clot injection, a loss of microvascular basal lamina is observed as early as 3 hours in the rat. This loss encompassed both the density of microvessels (number of stained vessels per area) and the total stained area of microvessels (area of specifically stained vessel walls per total area), Second, the value of rCBF as measured 3 hours after onset of embolic stroke is not related to the microvascular damage observed. It appears most likely that microvascular damage was induced by a critical reduction of rCBF after the embolic vessel occlusion. The positive silver infarct staining observed within the areas investigated confirms that embolic vessel occlusion had induced a critical reduction of rCBF, at least in the interval between embolization and the measurement of rCBF. However, rCBF apparently recovered in some of the areas analyzed, but this recovery was not paralleled by a salvage of microvessels. As our data show (Fig. 2), basal lamina damage persists 3 hours after the onset of vessel occlusion, despite an apparent recovery of rCBF to normal values in some of the animals investigated.
In the present study, three experimental groups were investigated. The groups consisted of a control group that was only treated by clot embolization and two other groups treated additionally by intravenous or intraarterial rt-PA, which was administered with the aim of increasing a reduced rCBF pattern with the help of an early recanalization. However, the results showed a comparable pattern of flow reduction in the three experimental groups. Because the microvascular density data as well as the total stained area data were not significantly different between the three groups, the data of the three experimental groups were pooled. The lack of difference between the three experimental groups was unexpected because some previous reports showed reperfusion after the use of thrombolytics in this model (Jiang et al., 1998; Takano et al., 1998). Whereas Takano et al. (1998) used prourokinase intravenously or intraarterially, Jiang et al. (1998) used rt-PA in a similar dosage and time frame as in the present experiments, but they monitored the reperfusion by magnetic resonance imaging. It appears mandatory to monitor the perfusion state in all experimental studies in which reperfusion is investigated. As to the present study, because of the lack of reperfusion, statements can only be given about the level of rCBF and the microvascular damage 3 hours after clot embolism. An incomplete reperfusion has been found by Busch and colleagues (1998) in the rat embolic stroke model even after very early rt-PA administration (15 minutes after embolism).
Rat models of embolic cerebral ischemia using a thromboembolic occlusion of the middle cerebral artery (Busch et al., 1997; Zhang et al., 1997) may be of advantage compared with the thread models because, at least in principle, they allow the use of thrombolytic drugs for recanalization therapy. Microvascular basal lamina damage had not been investigated in clot models of cerebral ischemia. The present results therefore provide evidence for the induction of microvascular damage by thromboembolic occlusion, which is comparable with those results previously reported for the intravascular filament occlusion model in the rat (Hamann, 2002).
The decrease of microvascular basal lamina staining observed after clot embolism in the present study is caused by digestion of basal lamina membranes. The systems involved include the plasminogen-plasmin system (Hamann et al., 1999; Lijnen et al., 1998, 1999; Liotta et al., 1981; Pfefferkorn et al., 2000), the matrix-metalloproteinase system (Rosenberg et al., 1996, 1998, 2001), and the transmigration of leukocytes (del Zoppo, 1997; Ember et al., 1994; Garcia et al., 1994). Whereas it is accepted that these mechanisms are induced by the reduced blood flow to the affected tissue, the present study indicates that even a recovery of rCBF a few hours later does not prevent the induced damage. From these data, it can be hypothesized that the ischemic blood flow triggers early deleterious changes in the basal lamina membranes, which cannot be reversed by a recovery of rCBF.
Footnotes
Acknowledgment
The authors thank Mrs. Judy Benson for editing this manuscript.
