Abstract
The contribution of leukocytes to secondary brain damage after cerebral ischemia is still under discussion. The purpose of the present study was to examine the pial microcirculation after global cerebral ischemia while focusing on leukocyte–endothelium interactions during the early and late reperfusion period of up to 4 days. A closed cranial window technique that leaves the dura mater intact was used. Global cerebral ischemia of 15 minutes' duration was induced in male Mongolian gerbils (n = 91). Pial microcirculation was observed by intravital fluorescence microscopy. Leukocyte–endothelium interactions (LEIs) in pial venules, vessel diameters, capillary density, and regional microvascular blood flow measured by laser Doppler flowmetry were quantified during 3 hours of reperfusion and in intervals up to 4 days after ischemia. Within 3 hours of reperfusion, the number of leukocytes (cells/100 μm × minute) rolling along or adhering to the venular endothelium increased from 0.1 ± 0.2 to 28.4 ± 17.4 (P < 0.01 vs. control) and from 0.2 ± 0.2 to 4.0 ± 3.8 (P < 0.05), respectively. There was no capillary plugging by leukocytes; capillary density remained unchanged. In the late reperfusion period, at 7 hours after ischemia, LEIs had returned to baseline values. Furthermore, from 12 hours to 4 days after ischemia, no LEIs were observed. Changes in regional microvascular blood flow did not correlate with LEIs. Global cerebral ischemia of 15 minutes' duration induces transient LEIs that reach a maximum within 3 hours of reperfusion and return to baseline at 7 hours after ischemia. LEIs are not related to changes in microvascular perfusion, which suggests mainly that the expression of adhesion receptors is necessary to induce LEIs rather than rheologic factors. It seems unlikely that this short-lasting activation of leukocytes can play a role in the development of secondary brain damage.
The activation of leukocytes during postischemic reperfusion is a pathophysiologic phenomenon that occurs in a variety of peripheral organs as well as in brain tissue (del Zoppo et al., 1991; Engler et al., 1983; Hallenbeck et al., 1986; Menger et al., 1992). The attachment of leukocytes to the venular endothelium and subsequent emigration of the cells into the brain parenchyma may contribute to the development of secondary brain damage after the primary ischemic insult (Kochanek and Hallenbeck, 1992). This hypothesis, however, is mainly derived from studies on focal cerebral ischemia and based either on histologic observations (del Zoppo et al., 1991; Garcia et al., 1994) or on antileukocyte treatment by the blockage of adhesion receptors or the induction of neutropenia by antineutrophil serum (Bednar et al., 1991; Chopp et al., 1994; Clark et al., 1991; Mori et al., 1992; Soriano et al., 1996). With regard to global and forebrain ischemia, however, the significance of leukocyte activation in the pathophysiologic process still remains to be defined. Although several studies seem to favor a role of leukocytes in the postischemic reperfusion period (Grögaard et al., 1989; Schürer et al., 1990; Vasthare et al., 1990), a variety of experiments failed to demonstrate a pathologic function of leukocytes in secondary damage from global cerebral ischemia (GCI) (Aspey et al., 1989; Schott et al., 1989; Schürer et al., 1991). Despite that direct observation of leukocyte activation in cerebral venules after experimental cerebral ischemia has already been described by Meyer (1958), only few in vivo studies with quantitative microscopic analysis of leukocyte–endothelium interactions after GCI have been published to date (Abels et al., 1994; Dirnagl et al., 1994; Hudetz et al., 1999). The aim of the present experiments, therefore, was to (1) develop an in vivo model of GCI that would easily allow access to cerebral microcirculation for intravital microscopic observation, and (2) provide solid evidence of whether GCI induces significant leukocyte–endothelium interactions.
MATERIALS AND METHODS
Animals
A total of 91 male barrier–bred Mongolian gerbils (body weight, 60 to 70 g; age, 12 to 16 weeks) were used in this study. Animals were kept single in Macrolon type 3 cages (Ehtet, Emmendingen, Germany) at a room temperature of 22°C. The animals had free access to tap water and pellet food. The experiments, conducted according to institutional guidelines, were approved by the State Government of Bavaria.
Surgical preparation
Anesthesia under spontaneous breathing was induced with 4% halothane and continued during the preparation at a concentration of 2%, with 0.8% during intravital microscopy and 0.4% during ischemia. Oxygen content of inspired air was regulated with an oximeter (Oxydig, Drägerwerke, Lübeck, Germany) and kept at 30%; the flow rate was 1 L/min. The body temperature was maintained at 37.0°C by using a rectal probe connected to a feedback-controlled heating pad. All surgical preparations were performed by using an operating microscope (Zeiss, Oberkochen, Germany). Polyethylene catheters (outer diameter, 0.61 mm; Portex Ltd., Hythe, England) were inserted into the tail artery for measurement of the mean arterial blood pressure and into the femoral vein for application of fluorescent dyes. The blood pressure was monitored continuously (Servomed SMS 308; Hellige GmbH, Freiburg/Breisgau, Germany). After an incision 1.5 cm cranial to the upper margin of the sternum, the common carotid artery was dissected bilaterally while sparing the vagal nerve. Both carotid arteries were encircled with a monofil thread (Prolene 5–0; Ethicon, Norderstedt, Germany) without disturbing the blood flow. The skull was then fixed in a stereotactic frame (Model 51600; Stoelting Co., Wood Dale, IL, U.S.A.) for preparation of the cranial window over the left parietal hemisphere. After a midsagittal skin incision from the forehead to the neck, the calvaria was exposed and a rectangular 4×4 mm window was exposed by trephine over the left parietal hemisphere, with the dura mater left intact. The dura mater was rinsed continuously with normal saline at 37.0°C.
Regional cerebral blood flow
For analysis of regional cerebral blood flow (rCBF) a laser Doppler probe (laser Doppler flowmeter MBF3D; Moor Instruments, Axminster, Devon, England) was placed with a stereotactic micromanipulator on the dura mater at the beginning of the experiment. The regional cerebral blood flow (flux) was measured after each intravital microscopic measurement. Simultaneous measurements were not possible because of partial light interference of fluorescence microscopy with the laser.
Intravital microscopy
For intravital microscopy, the animals were placed on a computer-controlled microscope stage for repeated analysis of identical vessel segments. The intravital fluorescence microscope (Leitz, Wetzlar, Germany) was equipped with a 75 W xenon lamp and a Ploemopak filter block with an N2 and L3 filter block for epiillumination. Enhancement of microvessels was achieved by an intravenous injection of fluorescein isothiocyanate–labeled dextran (FITC-dextran; molecular weight, 150,000; Sigma Chemical, St. Louis, MO, U.S.A.) before the first measurement (bolus of 0.3 mL of a 1% solution). Leukocytes were stained in vivo before each measurement by intravenous injection of 0.05 mL of 0.01% Rhodamine 6G (Merck, Darmstadt, Germany). The microcirculatory parameters were analyzed by using a salt water immersion objective, x25. The intravital microscopic images were recorded by an SIT-video camera (C2400; Hamamatsu Photonics, Herrsching, Germany). Off-line analysis of the video tapes was performed by frame-to-frame analysis with the use of a TV monitor (Trinitron PVM-2130QM; Sony, Tokyo, Japan) at a total magnification of x790. To test the integrity of the blood–brain barrier at the end of the experiment (on-line), 0.2 mL of a 0.1% Na+-fluorescein solution (Sigma) was injected intravenously. For observation of the extravasation of the fluorescent marker, the L3 filter (Leitz) and an objective with a magnification of x10 were used. After the injection of the fluorescent marker, the preparation was observed in short intervals for up to 20 minutes.
Analysis of microcirculatory parameters
Arteriolar and venular diameters (expressed in micrometers), the number of rolling and adherent leukocytes in venules (expressed as number of cells per 100 μm × minute), the capillary density and the integrity of the blood–brain barrier were analyzed. The vessel diameters were measured by using a computer-assisted microcirculation analysis system (CapImage; Ingenieurbüro Dr. Zeintl, Heidelberg, Germany). The white blood cells were classified according to their interaction with the venular endothelium as adherent or rolling leukocytes. Rolling leukocytes were defined by their multiple intermittent contacts with the venular endothelium, thereby advancing definitely slower than the freely moving leukocytes in the center flow axis of a microvessel. Adherent leukocytes were defined by their attachment to the venular wall for more than 20 seconds. Vessel segments of 100 μm in length were studied for 30 seconds. The capillary density, defined as the length of capillaries in each observation field (length/area, cm−1), which are perfused with plasma tracer and blood cells, was quantified by using the CapImage system (Klysz et al., 1997). Occlusion of capillaries could be excluded by observation of moving red blood cells, indicated by a dark gap in the plasma layer, and Rhodamine-stained leukocytes. Because there was no flow during ischemia, leukocyte–endothelium interactions (LEIs) and capillary density could not be assessed during this period.
Experimental design: early reperfusion period
Ten animals, respectively, were randomly assigned to the control or the ischemia group. After surgical preparation the animals were allowed to stabilize during a control period of 60 minutes. During this period, three baseline measurements at 20-minute intervals were performed. Before the first measurement, three or four regions of interest with at least one pial arteriole and one venule, respectively, were selected. Thus, at least 3 or 4 arterioles, and 3 or 4 venules were observed and analyzed per animal. In an additional region of interest, capillaries were visualized for the quantification of the capillary density. Further intravital microscopic measurements were performed at the beginning and 10 minutes after the induction of the global ischemia. Occlusion of both arteries for 15 minutes was achieved by pulling at the Prolene threads with a weight of 15 g. Ischemia was followed by a 3-hour observation period, with intravital microscopic measurements at 5, 20, 40, 60, 90, 120, and 180 minutes of reperfusion. At the end of the experiment, the permeability of the blood-brain barrier to Na+-fluorescein was investigated.
Experimental design: late reperfusion
Six animals, respectively, were randomly assigned to an ischemic and a control group, with microcirculatory analysis at 6 and 7 hours. Five ischemic animals, respectively, were evaluated 12, 24, 48, 72, and 96 hours after ischemia. In addition, at each of these points of time two sham-operated animals were investigated. After temporary ligation of both carotid arteries, the cervical wound was closed again and the animals were allowed to wake up. The cranial window was prepared in a second step before the intravital microscopy observation period. Two measurements were made at a 60-minute interval at each time point.
Histology
Because intravital microscopic analysis of LEIs is limited to the cortical surface, the brains of three ischemic animals of the early reperfusion group and three control animals were studied histologically. For that purpose, they were perfused intracardially with phosphate-buffered paraformaldehyde (2%) at the end of the experiment. The brains were embedded in paraffin, sectioned serially (5 μm), and stained with cresyl violet. Leukocytes were stained red selectively by naphthol AS-D chloracetate-esterase (Sigma) (Li et al., 1973; Yam et al., 1971).
Statistical analysis
Data are presented as mean ± SD values. Because normal distribution was not uniformly found in all data sets, a Kruskal-Wallis test followed by the Mann-Whitney U-test and the Bonferroni-Holm correction were used for analyzing differences between control and treated groups. A statistically significant difference was assumed at P < 0.05.
RESULTS
Exclusion of animals
Eighteen animals were excluded from further evaluation. The most common cause for exclusion was respiratory failure during ischemia (n = 5). Further problems included injury to the dura during the preparation (n = 3), which could have resulted in activation of LEIs before ischemia. Animals with intact dura mater, which showed LEIs during the control period before ischemia, were also excluded from the study (n = 3). Further causes were low quality of the images obtained by intravital microscopy (n = 4) and incomplete ischemia during bilateral occlusion of the common carotid artery (n = 3).
Mean arterial blood pressure
Mean arterial blood pressure was nearly identical in both groups at the beginning of the experiment (control, 76.9 ±1.9 mm Hg; ischemic, 77.0 ± 3.8 mm Hg). During occlusion of the carotid arteries, the blood pressure increased significantly (P < 0.05) but returned to baseline during reperfusion. Mean arterial blood pressure remained constant then until the end of the experiment (Table 1).
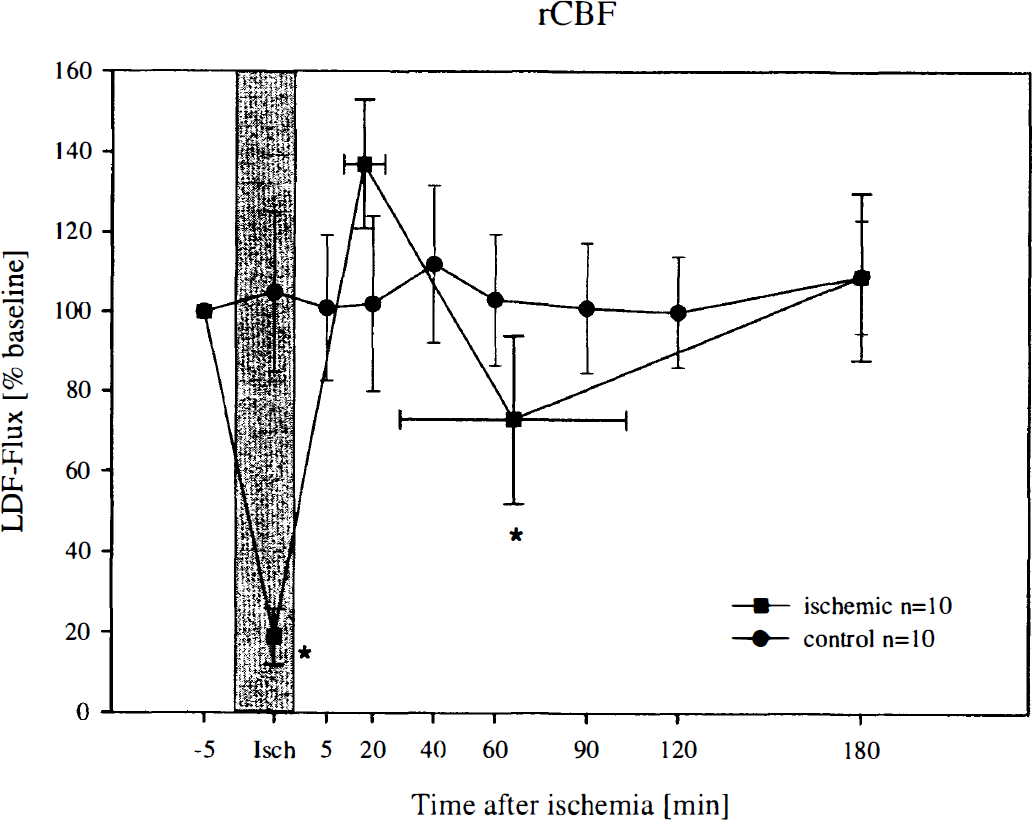
Laser Doppler flowmetry flux indicating regional cerebral blood flow [percentage of baseline value] before and after global cerebral ischemia in gerbils. Mean ± SD. Mann-Whitney U test. *P < 0.05 versus control. During occlusion of both carotid arteries perfusion decreased to 18.7 ± 6.9% of the initial value. Curves in the ischemic group were obtained from 5 values determined for each animal: before ischemia, during ischemia, the highest and lowest value during reperfusion, and final flow at termination of the experiment. Horizontal error bars indicate SD for time. This method encounters the temporal differences in the maximum of postischemic hypo- and hyperperfusion, which would be mitigated by calculation of the average at a fixed point of time.
MABP, arteriolar and venular diameters before, during, and after 15 minutes of global cerebral ischemia in Mongolian gerbils (n = 10)
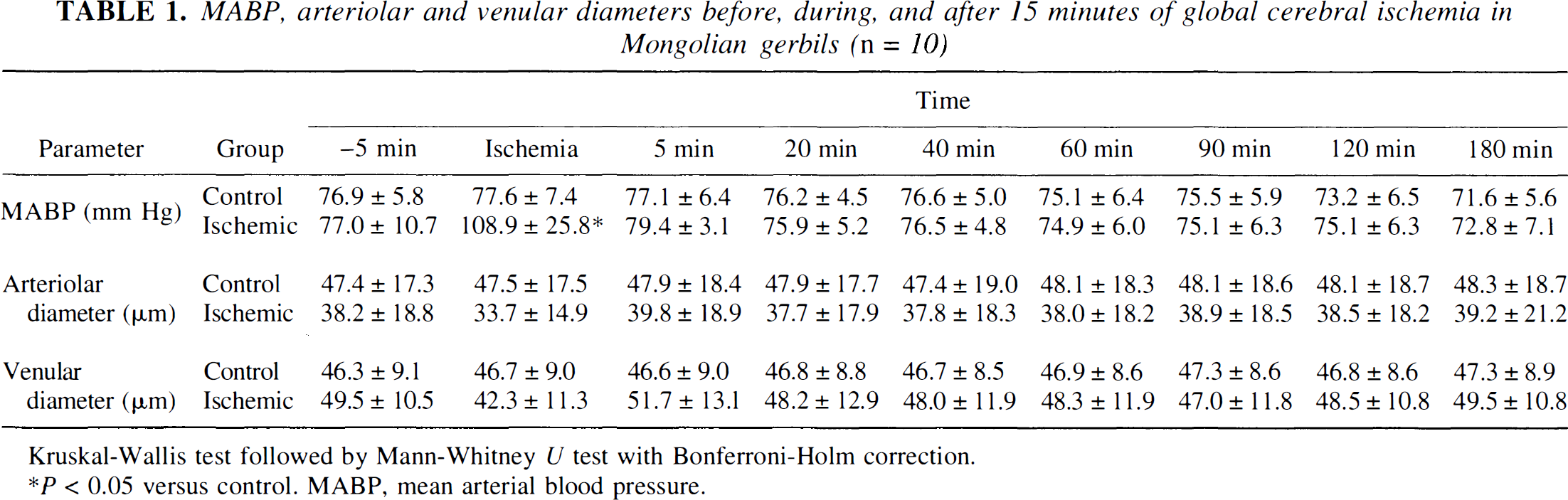
Kruskal-Wallis test followed by Mann-Whitney U test with Bonferroni-Holm correction.
P < 0.05 versus control. MABP, mean arterial blood pressure.
Vessel diameters
Except for a decrease during ischemia, the arteriolar diameters remained unchanged during the whole experiment. No differences were found between control and ischemic animals (Table 1). In contrast, the venules of ischemic animals were dilated at 5 minutes of reperfusion (NS) and then normalized during a period of up to 180 minutes. The venular diameters remained unchanged in control animals (Table 1). During the late reperfusion period, no significant differences were observed between the ischemic and control animals (data not shown).
Regional cerebral perfusion
The results of the laser Doppler flowmetry are presented as percentages of the baseline values (Fig. 1). In control animals, rCBF was not changed significantly throughout the entire observation period. During GCI, perfusion decreased to 18.7 ± 6.9%, which was confirmed by microvascular stasis or low flow state as observed by intravital microscopy. Ischemia was followed by a short hyperemic period, which reached a maximum within 20 minutes of reperfusion. After a period of hypoperfusion, rCBF returned to baseline until termination of the experiment. Because these phenomena occurred with a broad variation with respect to time, the data of the ischemic group are presented as suggested by Stummer et al. (1995), calculating the mean of the postischemic perfusion maxima and minima. By this method, the mitigation of the amplitude encountered in each single animal, by calculation of the average at a given point of time, can be avoided.
Leukocyte–endothelium interactions
LEIs were typically observed in venules, although rarely in capillaries or arterioles (Figs. 2A and 2B). After onset of reperfusion, a continuous increase in the frequency of rolling and adherent leukocytes was observed (Figs. 3 and 4). After 40 minutes of reperfusion, the number of rolling leukocytes (expressed as number of cells per 100 μm × minute) was significantly increased compared with control animals. A significantly higher number of adherent leukocytes was observed after 2 hours of reperfusion. Both parameters reached their maximum at 3 hours of reperfusion (roller, 28.4 ± 17.4 vs. 1.7 ± 1.9, P < 0.01; sticker, 4.0 ± 3.8 vs. 0.0 ± 0.0, P < 0.05). Six hours after ischemia, LEIs were still observed, although the number of rolling and adherent leukocytes had already decreased again (Figs. 5A and B). The number of rolling leukocytes (5.7 ± 6.4) was still significantly higher compared with control animals (0.1 ± 0.2). After 7 hours of reperfusion, no difference was observed between both groups (ischemic, 2.9 ± 3.0; control, 0.3 ± 0.4). Similar results were obtained on leukocyte adherence (Fig. 6A). The number was decreased to 1.2 ± 0.7 (control, 0.0 ± 0.0, P < 0.05) at 6 hours of reperfusion. After 7 hours, the number of adherent leukocytes did not differ any more from the control group (ischemic, 0.2 ± 0.4; control, 0.1 ± 0.2). LEIs were not observed at 12 hours to 4 days after GCI (Figs. 5B and 6B). By use of the esterase stain, leukocytes could also be selectively identified in deep parenchymal vessels of various brain areas. This indicates activation of LEIs not only at the brain surface, as observed by intravital microscopy, but also in blood vessels that supply the brain parenchyma (Fig. 7).
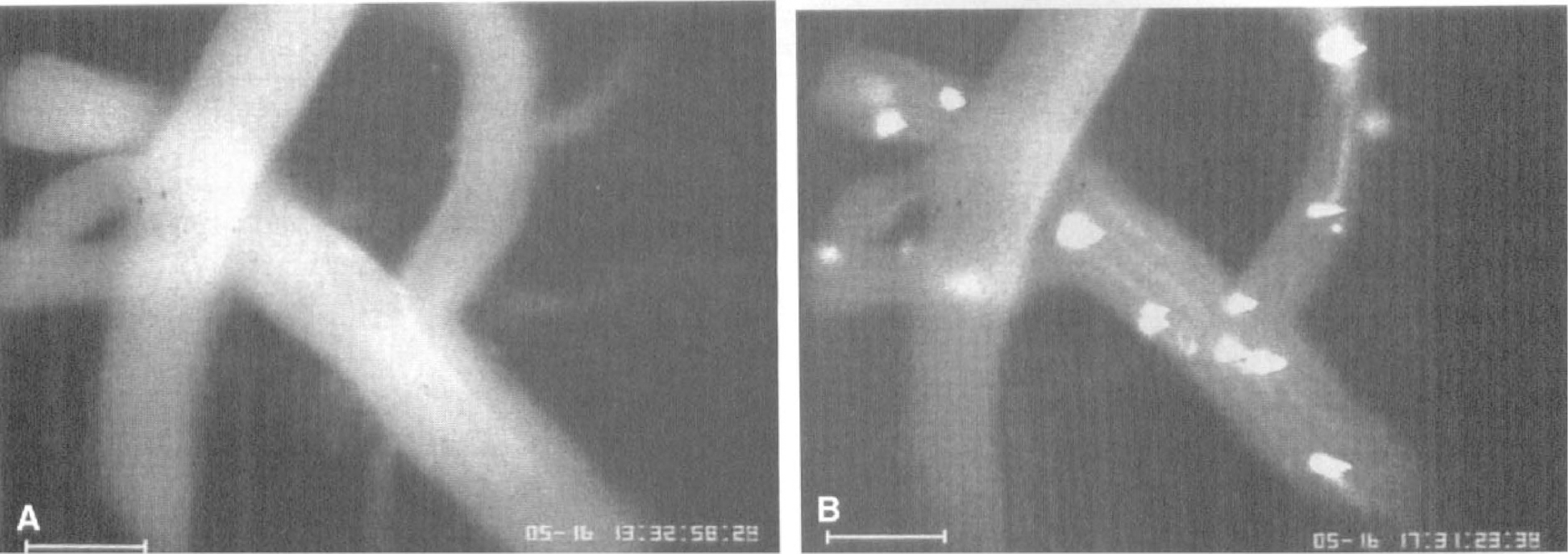
In vivo fluorescence photomicrograph of the brain surface demonstrating pial venules during the control period
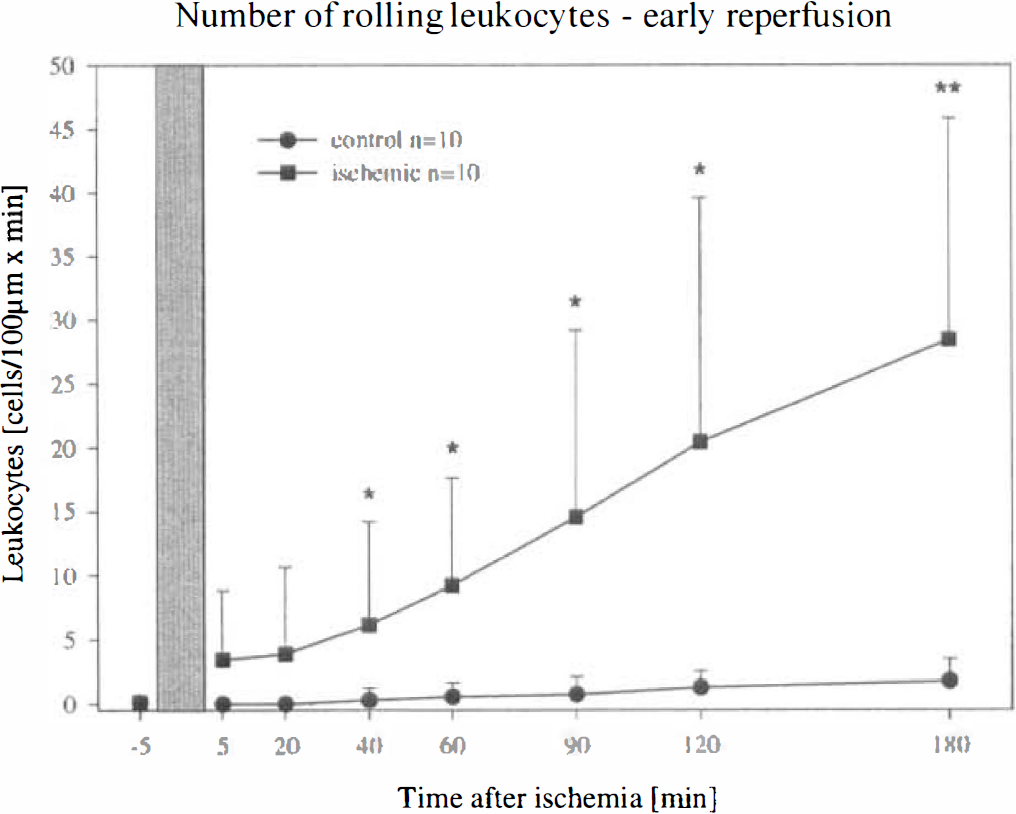
Frequency of leukocytes [cells/100 μm × min] rolling along the endothelium of pial venules before and after global cerebral ischemia in gerbils. Mean ± SD. Kruskal-Wallis test followed by Mann-Whitney t-test and Bonferroni-Holm correction. *P < 0.05 versus control, **P < 0.01. A significant increase in the number of rolling leukocytes was observed after GCI. Bar = time of ischemia.
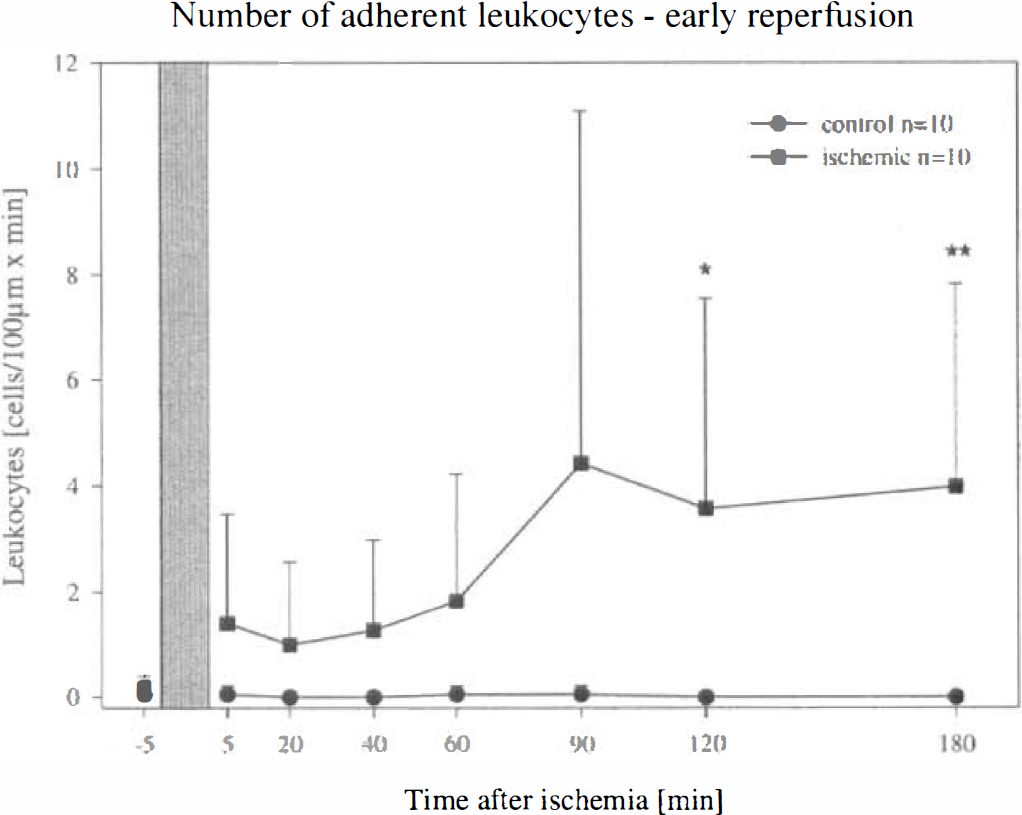
Frequency of leukocytes attached to the endothelium of pial venules before and after 15 minutes of global cerebral ischemia in gerbils. Mean ± SD. Kruskal-Wallis test followed by Mann-Whitney U-test with Bonferroni-Holm correction, *P < 0.05 versus control. After ischemia a significant increase in the number of adherent leukocytes was observed within 3 hours of reperfusion. Bar = time of ischemia.
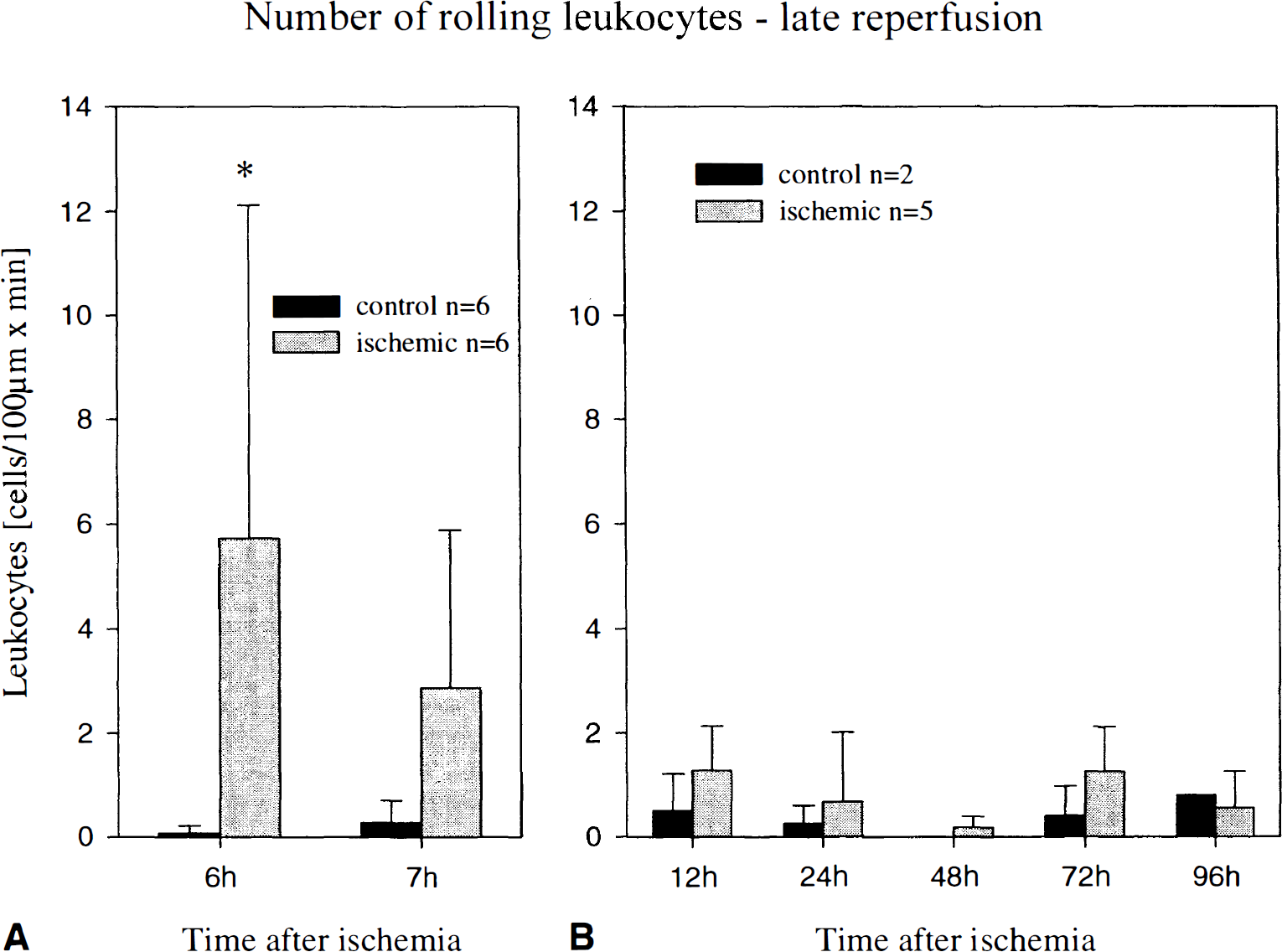
Leukocyte–endothelium interactions during the late phase of reperfusion after 15 minutes of global cerebral ischemia in Mongolian gerbils. Frequency of leukocytes rolling along the venular endothelium 6 to 7 hours after ischemia
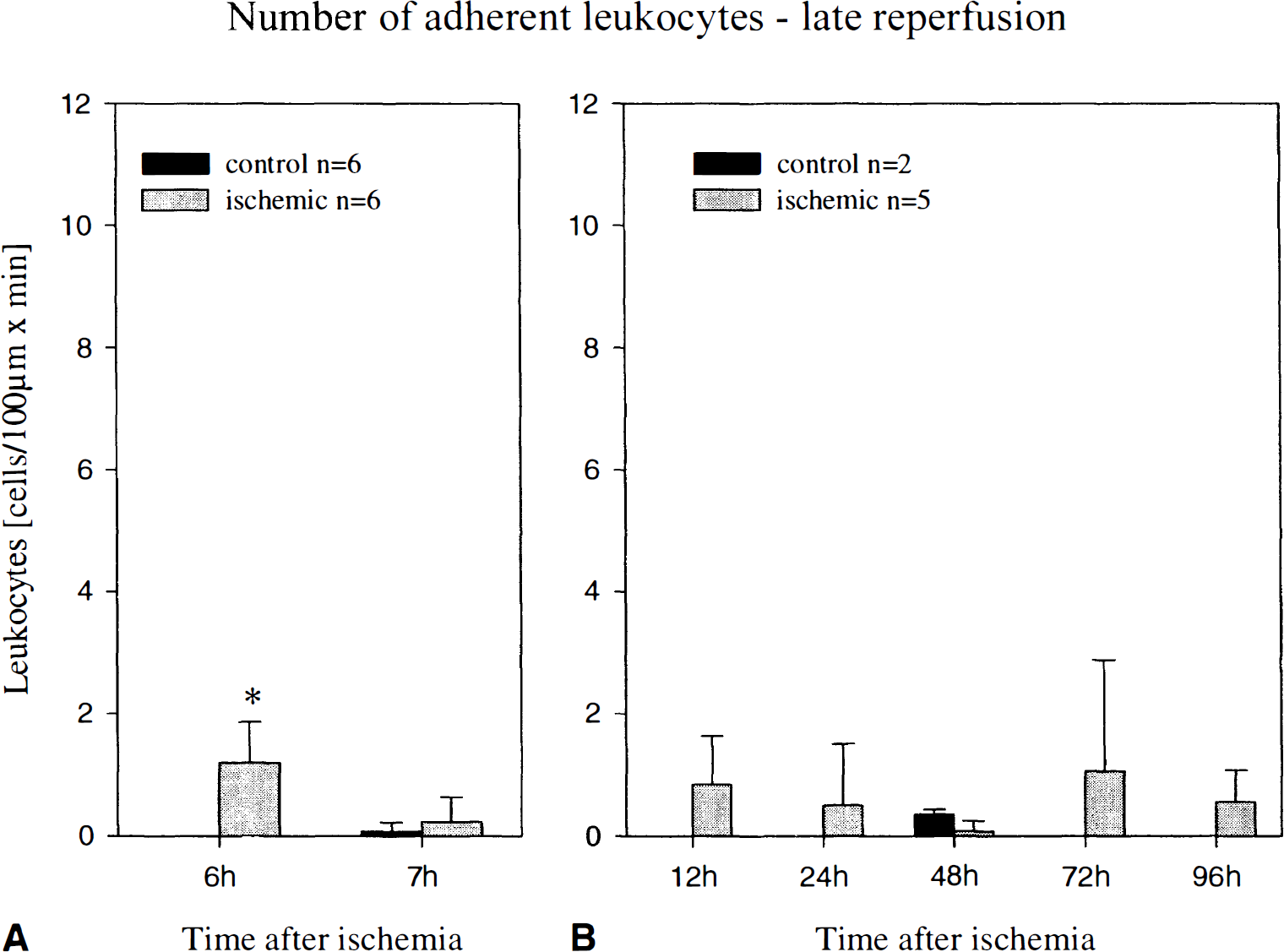
Leukocyte–endothelium interactions during the late phase of reperfusion after 15 minutes of global cerebral ischemia in Mongolian gerbils. Frequency of leukocytes adherent to the venular endothelium 6 to 7 hours
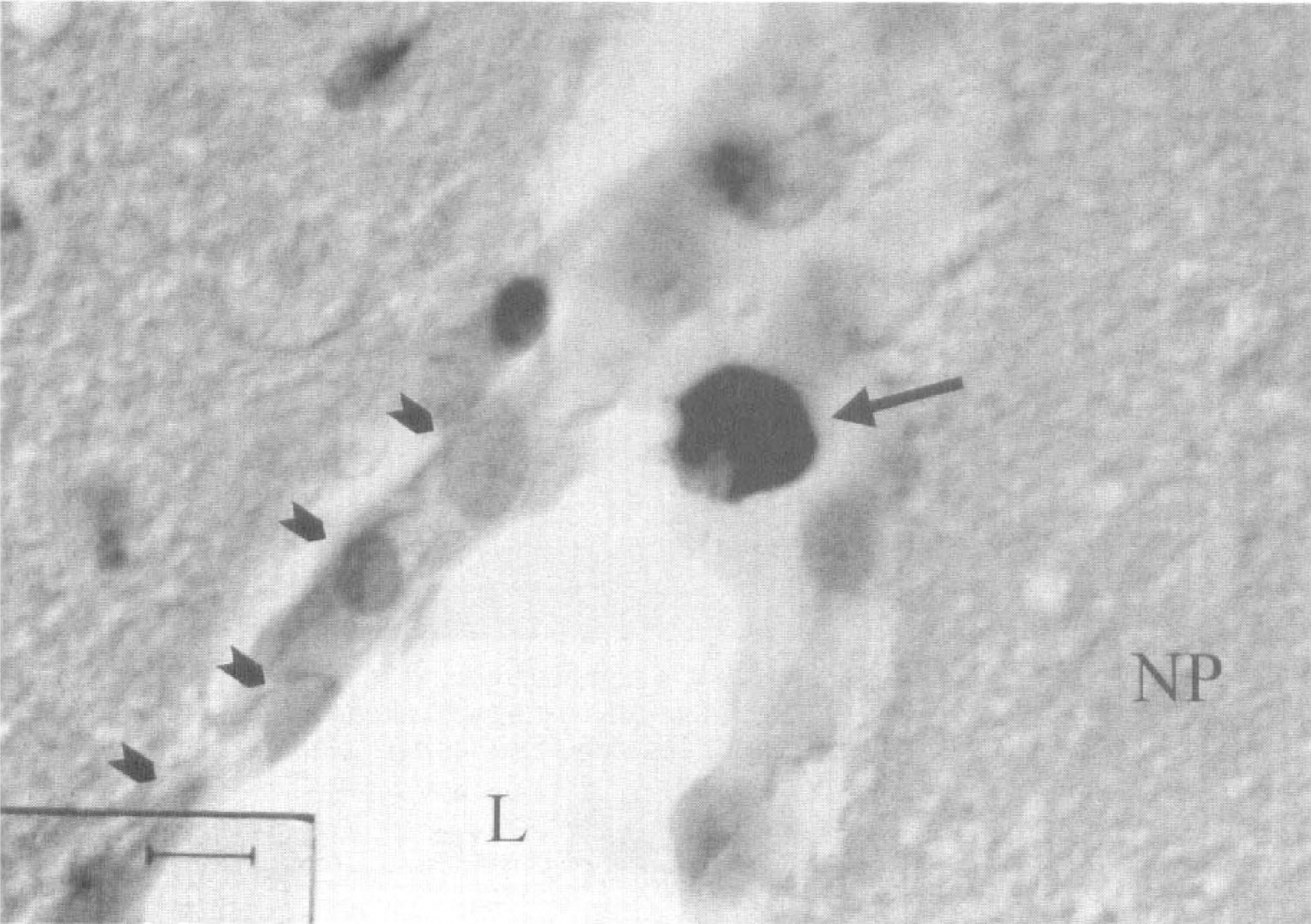
Cross-section of the striatum showing a deep intraparenchymal venule with a leukocyte (arrow) in close contact to the vascular endothelium. No other corpuscular elements can be seen. Short arrows mark endothelial lining. L, vascular lumen; NP, surrounding neuropil. Staining with cresyl violet and naphthol AS-D chloroacetate-esterase for leukocytes. Bar = 10 μm.
Capillary density
Capillary density could be sufficiently assessed in only seven ischemic animals because of impaired visualization of the capillaries after ischemia. This notwithstanding, the capillary density remained unchanged throughout the entire experiment. Capillary density ranged between 136.7 ± 37.3 cm−1 and 159.0 ± 17.0 cm−1 in the control group and between 140.9 ± 34.5 cm−1 and 154.2 ± 16.6 cm−1 in animals with ischemia (Fig. 8).
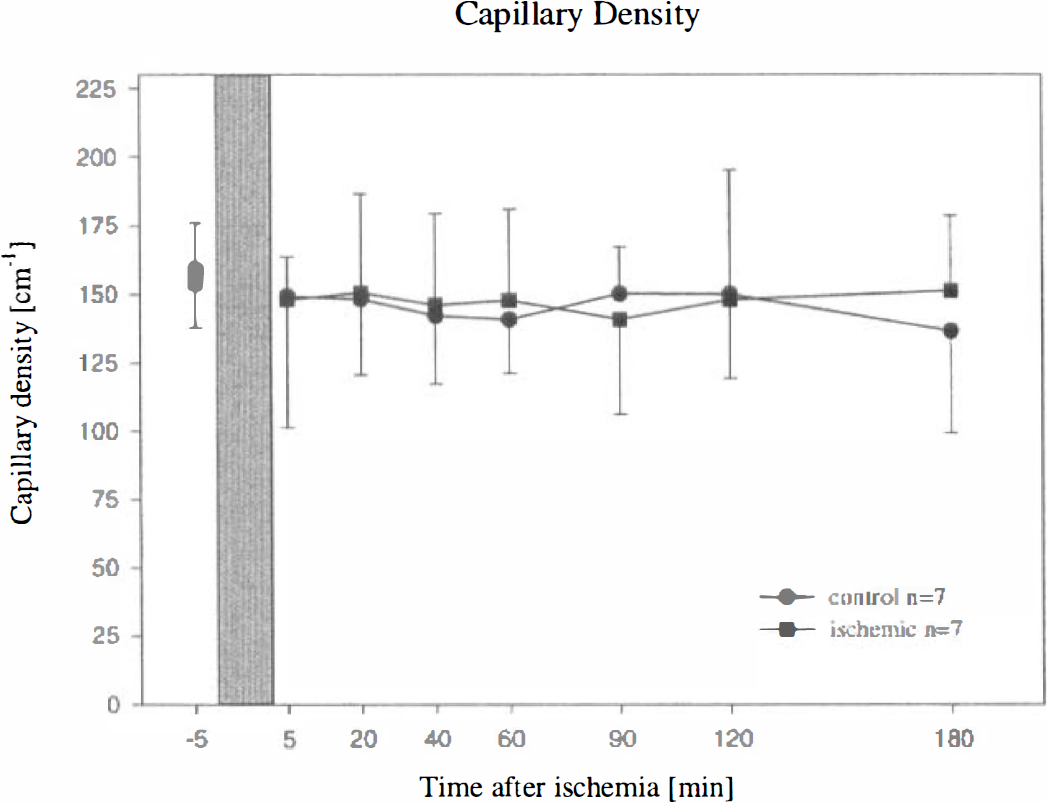
Capillary density before and after 15 minutes of global cerebral ischemia in gerbils. Mean ± SD. No significant difference between control and ischemic animals was observed. Bar = time of ischemia.
Integrity of the blood–brain barrier
Opening of the blood–brain barrier was not observed in the control or the ischemic animals at the end of the experiment. The low molecular weight marker sodium fluorescein remained strictly intravascular during an observation period of 20 minutes after its administration.
DISCUSSION
The model
So far, there exist only a few intravital microscopic studies on LEIs in GCI, and they have been conducted in closed cranial window models that require opening of the dura mater (Dirnagl et al., 1994; Villringer et al., 1994). Compared with these models, the transdural window in gerbils combines three major advantages for studying LEIs in GCI in vivo. First, the dura mater is translucent and does not need to be opened, which omits the requirement for the preparation of a closed cranial window, as has been described, for example, in rats (Kawamura et al., 1990; Uhl et al., 1999). Second, the induction of GCI is simple. It can be performed during intravital microscopy without major manipulations of the animal. There is no need for an additional occlusion of the vertebral arteries or induction of arterial hypotension during ischemia (Dirnagl et al., 1994; Pulsinelli and Briefly, 1979). Third, the model offers the possibility of chronic studies because the dura mater remains intact. In contrast, the currently made observations reveal that complete GCI is not always induced in gerbils by occlusion of the common carotid arteries. However, because intravital microscopy allows direct observation of microvascular perfusion, animals with insufficient ischemia can be excluded. Another disadvantage seems to be the limited possibility to control systemic physiologic parameters during anesthesia. We did not subject animals to mechanical ventilation because this would require repeated blood sampling for adjustment of respiratory parameters, which could result in major hemodynamic changes from hypovolemia. Nevertheless, major changes of systemic parameters were unlikely to have occurred, as, for example, concluded from the stability of the arterial blood pressure throughout the entire experiment. Furthermore, slight changes in systemic parameters seem to have no impact on LEIs in cerebral vessels, which was reported previously by our group using a rat model (Uhl et al., 1999).
GCI and leukocyte–endothelium interactions
Only few in vivo reports have been published that show that GCI induces LEIs (Dirnagl et al., 1994; Hudetz et al., 1999; Park et al., 1999; Villringer et al., 1994). Villringer et al. (1994), using a confocal laser-scanning microscope, described the adherence and emigration of leukocytes in pial venules after 10 minutes of GCI. Dirnagl et al. (1994) also reported a mild induction of leukocytes in the same model. The changes observed, however, did not reach statistical significance. Recently, LEIs were confirmed within 2 hours after 9 minutes of asphyxia in newborn pigs (Park et al., 1999). In contrast, Abels et al. (1994), also using a rat model with 20 minutes of global ischemia, did not detect a significant activation of LEIs during 2 hours of reperfusion.
In the present investigation, it is shown for the first time, that GCI results in a transient activation of LEIs. Activation started already at 5 minutes of reperfusion and reached statistical significance at 60 minutes. The maximum was observed after 3 hours of reperfusion, then LEIs decreased again. In contrast to previous reports (del Zoppo et al., 1991; Dirnagl et al., 1994), postishemic plugging of capillaries by leukocytes was found to be a rare event. This phenomenon has been thought to contribute, at least in part, to the no-reflow phenomenon and hypoperfusion after ischemia in the brain and other organs (Ames et al., 1968; del Zoppo et al., 1991; Engler et al., 1983). It is, however, conceivable that 15 minutes of GCI, as currently studied, may not be sufficient to induce significant leukocyte plugging of capillaries, which results in reduced microvascular perfusion.
This absence of leukocyte plugging was confirmed by the unchanged capillary density throughout the entire experiment. These data support previous studies with a double-staining technique of plasma and capillaries in which a significant reduction of capillary staining was not observed under either physiologic conditions or after 15 minutes of GCI and 4 hours of reperfusion (Göbel et al., 1990; Theilen et al., 1993). Because the authors used a fluorescent plasma marker, they were not able to draw conclusions on red blood cell or leukocyte flow, and a staining of the capillaries by the plasma marker through bypassing a plugging leukocyte or retrograde filling of the microvessels as described by Villringer et al. (1994) could not be excluded. Because we only measured capillaries that were perfused by the plasma tracer and erythrocytes or leukocytes, this problem in the interpretation of capillary density could be eliminated.
As seen, LEIs evolved independently from systemic blood pressure and the microvascular perfusion, which excludes a major influence of mechanical factors. The mechanical forces that act on the leukocytes can be estimated by the calculation of the wall shear rate, which can be calculated from corresponding vessel diameters and the red blood cell velocity (Koller and Kaley, 1991; Ley and Gaethgens, 1991). We were not able to measure the latter either in arterioles or venules, because the velocity was too high for the available microcirculatory analysis system or for a frame-to-frame analysis of the video tapes. As an alternative laser Doppler flowmetry data were used instead as an indirect measure for the red blood cell velocity, to evaluate the dependency of LEI interactions from rheologic factors. No correlation, however, was found between rCBF and activation of LEIs; despite the normalization of the hypoperfusion within 2 to 3 hours after GCI, the number of LEIs increased during this period. This independency of LEIs in cerebral venules has previously been shown in the rat after systemic application of platelet-activating factor (Uhl et al., 1999). In these experiments, we were able to demonstrate that LEIs occur in cerebral venules despite shear rates higher than 400 s−1. This shear rate can be regarded as a threshold value for LEIs induced by rheologic mechanisms (Firrell and Lipowsky, 1989; Ley and Gaethgens, 1991). With regard to the current independency of LEIs from the actual blood flow in our model, we hypothesize that rolling and adherence of leukocyte is essentially based on the expression of adhesion receptors on the leukocytes as well as on the cerebrovascular endothelium. It has been shown that P-selectin (Okada et al., 1994), ICAM–1 (Okada et al., 1994), and E-selectin (Haring et al., 1996; Zhang et al., 1996) are expressed in the brain after ischemia. As also known from other organs, the initial contact of leukocytes with the endothelium, as observed by rolling of leukocytes along the inner lining of the endothelium, is mediated by P-selectin, L-selectin, and E-selectin (Vestweber and Blanks, 1999). ICAM-1 is required for the permanent adherence and the transmigration of leukocytes (Kurose et al., 1994). A significant expression of P-selectin, which is constitutively present in endothelial cells, was found after focal cerebral ischemia in baboons within 1 hour and of ICAM-1 within 4 to 7 hours of reperfusion (Okada et al., 1994). E-selectin is expressed at the beginning of reperfusion and reached a peak at 4 and 10 hours of reperfusion with a subsequent decrease after 72 hours (Haring et al., 1996; Zhang et al., 1996). Although respective data on GCI are not available, the early increase in the number of rolling leukocytes could be explained by the activation of P-selectin followed by the permanent adherence of leukocytes later on mediated by ICAM-1. In contrast to findings obtained in focal cerebral ischemia, in the current experiments, activation of leukocytes was confined to the early reperfusion period, which may be explained by the different duration of ischemia. Whereas leukocyte infiltration of brain tissue was found at up to 72 hours in histologic brain sections of rats after focal cerebral ischemia (Garcia et al., 1994), we were unable to confirm LEIs after 7 hours of reperfusion.
In conclusion, the closed cranial window in gerbils has been found to be a suitable model for investigation of cerebral microcirculation and LEIs in GCI. A major advantage of the model is the preservation of the dura, which excludes the activation of LEIs after opening of the dura. It also offers the possibility of chronic studies. Furthermore, acute effects of GCI on the cerebral microcirculation can be directly studied under the microscope. Animals with an insufficient ischemic blood flow response can be eliminated from further evaluations. The results of the current experiments show that GCI in gerbils results in a prompt, albeit transient, activation of LEIs in the early reperfusion period. These LEIs seem to be independent from rheologic factors and seem to be entirely mediated by adhesion receptor mechanisms. The data also show that LEIs were limited; because, after 7 hours of reperfusion, no further rolling or adherent leukocytes in pial venules were observed. Whether this time-limited manifestation of LEIs can contribute to the development of secondary brain damage after GCI is questionable and requires further evaluation.
