Abstract
In the present study the intrinsic nerve plexuses of the basal cerebral arteries, derived from aged non-Alzheimer's and aged Alzheimer's disease patients were quantified and compared. A previous study described and quantified nerve density on similar arteries from healthy middle-aged patients. Whole-mount preparations of various segments of the basal cerebral arteries were stained for protein gene product 9.5. The deep nerve plexuses, located at the adventitial–medial border, were quantified by image analysis. Transverse cryostat sections were stained for various markers and quantified. Measurements on whole mounts demonstrated that nerve densities were highest in the posterior communicating artery and in the postcommunicating part of the posterior cerebral artery (PCA) for both aged and Alzheimer's groups. Statistical comparison showed a tendency toward decreased nerve density with aging, which was significant for the internal carotid artery, precommunicating part of the PCA, and the anterior choroidal artery in both non-Alzheimer's and Alzheimer's aged groups. In addition, in Alzheimer's patients nerve density was significantly lower in the precommunicating part of the anterior cerebral artery compared with the healthy aged group. Measurements on sections confirmed the tendency to decreased innervation with aging. It is concluded that densities of deep perivascular nerves of human basal cerebral arteries are subject to localized changes caused by aging and Alzheimer's disease.
In our preceding article (this journal) we described and quantified the topographic distribution of the deep perivascular nerve plexuses of the human basal cerebral arteries. Highest nerve densities were found in the posterior communicating artery (PCom), pre- and postcommunicating parts of the posterior cerebral artery (P1 and P2), and the anterior choroidal artery (ChA). Findings on transmission electron microscopy suggested a functional role for the deep nerve plexuses in these vessels.
The specimens studies so far were derived from middle-aged patients (ages 32–53 for whole-mount preparations). From studies on laboratory animals it is known that nerves around cerebral arteries display changes in a temporally specific manner (see Cowen, 1993a for review). Thus, it was found that there is a decline of the overall nerve population in the aging rat (Cowen and Thrasivoulou, 1990), whereas subpopulations of nerves can follow different patterns of age changes (Mione et al., 1988). In humans, age changes in concentrations of neuropeptides in the walls of middle cerebral arteries have been described (Edvinsson et al., 1986, 1987).
Furthermore, it has become clear that certain diseases lead to changes in autonomic nerves, including cerebrovascular nerves. An increase of noradrenaline (NA) and neuropeptide Y (NPY)-containing nerves in cerebral arteries were found in spontaneously hypertensive rats (Dhital et al., 1988). In streptozotocin-diabetic rats a reduction of perivascular nerves containing vasoactive intestinal polypeptide (VIP) and serotonin, but not NPY and NA, has been demonstrated in cerebral arteries (Lagnado et al., 1987).
Quantitative studies of age changes and changes caused by disease, based on the morphology of perivascular nerves of the basal cerebral arteries, are lacking in humans. Basic knowledge of these changes is important, especially because it has been demonstrated in animals that some neurons, supplying cardiovascular tissues, retain capacity for regrowth and plasticity in old age (Gavazzi and Cowen, 1993a, 1993b).
Therefore, the aim of the present study was to investigate whether human cerebrovascular nerves are affected by aging and Alzheimer's disease. To allow optimal comparison with the previously obtained data from middle-aged patients, the same region of interest was studied and the same methodology was used. Thus, we have quantified the deep perivascular nerve plexuses, located at the adventitial-medial border of the human basal cerebral arteries, derived from aged patients (ages 62–85 years) and from age-matched patients who had Alzheimer's disease. Whole-mount preparations, stained for the general neural marker protein gene product (PGP) 9.5, and sections, stained for PGP 9.5, tyrosine hydroxylase (TH) and neurofilament (NF), were quantified using computerized image analysis. These data were compared with those obtained from middle-aged patients (32–53 years) reported in the preceding article.
MATERIALS AND METHODS
Human tissues
Seventeen sets of human major cerebral arteries, obtained from postmortems, were processed. They were classified into two groups: aged non-Alzheimer (ages 62–85 years) and aged Alzheimer (ages 62–85 years). For convenience, the groups will be referred to as the aged group and the Alzheimer group, respectively. Relevant details of the specimens are listed in Table 1. The specimens were fixed by immersion in 4% paraformaldehyde or formaldehyde in phosphate-buffered saline (PBS) at 4°C for 13–24 h. One specimen was fixed for a shorter period and two specimens were perfused with fixative (Table 1). Subsequently, the specimens were rinsed and stored in PBS containing 15% sucrose at 4°C. From each group, seven specimens were used for whole-mount immunohistochemistry. Six aged and two Alzheimer specimens were used for immunohistochemistry on sections.
Relevant details of human specimens
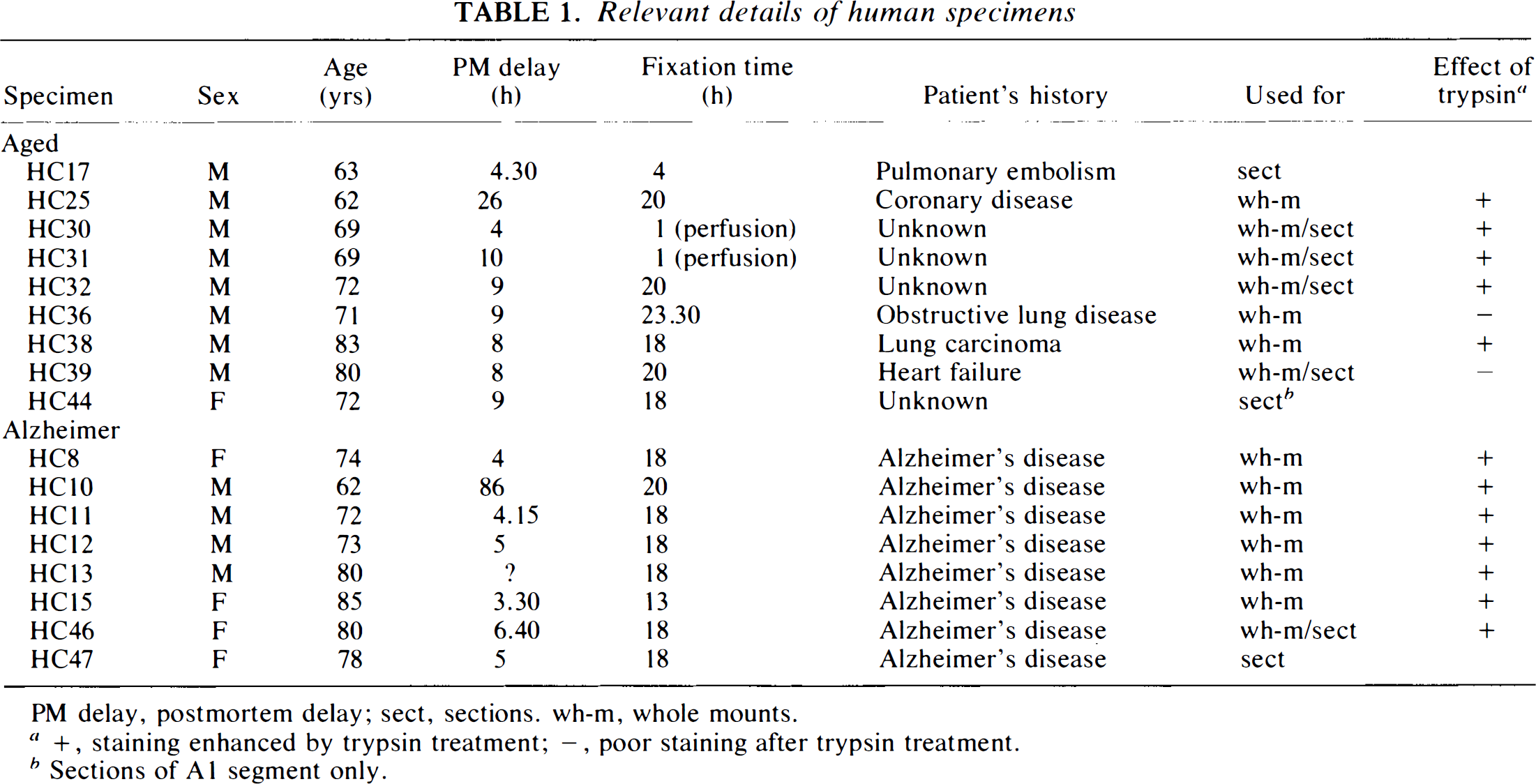
PM delay, postmortem delay; sect, sections, wh-m, whole mounts.
+, staining enhanced by trypsin treatment; –, poor staining after trypsin treatment.
Sections of A1 segment only.
Whole-mount preparations
Whole-mount preparations were processed as described in detail in part I of this article. Briefly, from each set of arteries, 22 segments were taken and any atheroscelerotic plaques were noted. The abbreviations of the arteries used are listed in the legend to Fig.1. The diameters of the segments were measured and a treatment was carried out to increase antibody penetration by freezing and thawing twice, incubation with trypsin in most cases (Table 1), and manually performed delamination. Staining was enhanced in most aged and all Alzheimer specimens by treatment with trypsin. After indirect immunohistochemical staining for PGP 9.5 (primary antibody anti-PGP 9.5, Ultraclone Ltd., Isle of Wight, England; secondary antibody fluorescein isothiocyanate (FITC)-conjugated swine-anti-rabbit, Dakopatts, Glostrup, Denmark), the vessels were mounted as stretch preparations and PGP 9.5-like immunoreactivity of the deep intrinsic nerve plexuses, located at the adventitial–medial border, was quantified by established methods of image analysis and expressed as area percent of the vessel wall and as intercept density (ID) per mm. The data were analyzed by analysis of variance (ANOVA), t tests, and computation of Pearson's correlation coefficients.
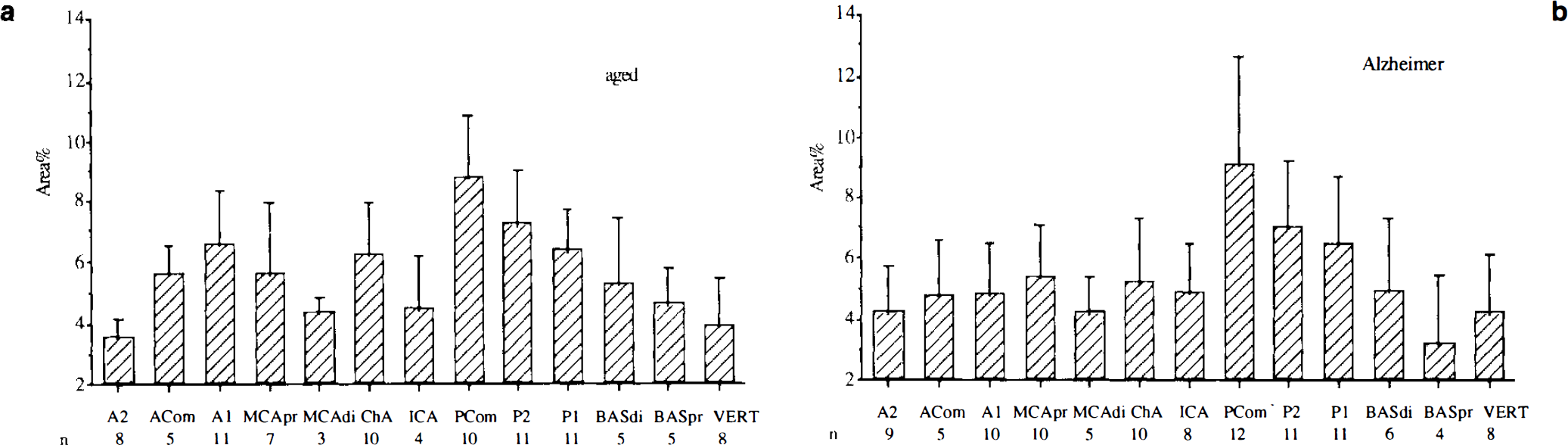
Mean values and standard deviation of nerve densities expressed as area percent for the arteries investigated as whole-mount preparations from aged and Alzheimer groups. Left and right arteries have been put together,
Measurements of stain intensity expressed as gray value was carried out on representative images of A1 segments, PCom, and P2 segments, derived from two specimens of each group, including the middle-aged group (see part I of the article).
Sections
Vessels were selected for sectioning from eight sets of arteries (Table 1). These vessels included the A2 and A1 segments, MCA, P2 segment, and PCom. The sections were processed as described in the preceding article. Thus, immunohistochemical staining was performed for PGP 9.5, TH, NF R39, and NF RT97. Primary antibodies were: anti-PGP 9.5, anti-TH (Eugene Tech International Inc., Ramsey, NJ, U.S.A.), anti-NF (R39), and anti-NF (RT97). Secondary antibodies were: FITC-conjugated sheep-anti-mouse (Dakopatts) for RT97, and FITC-conjugated swine-anti-rabbit for all other primary antibodies. For each marker, except for RT97, the specific fluorescence in the whole adventitial thickness was quantified by image analysis and expressed as percentage of the adventitial cross-section surface area (area percent).
RESULTS
Whole-mount preparations
Visual assessment
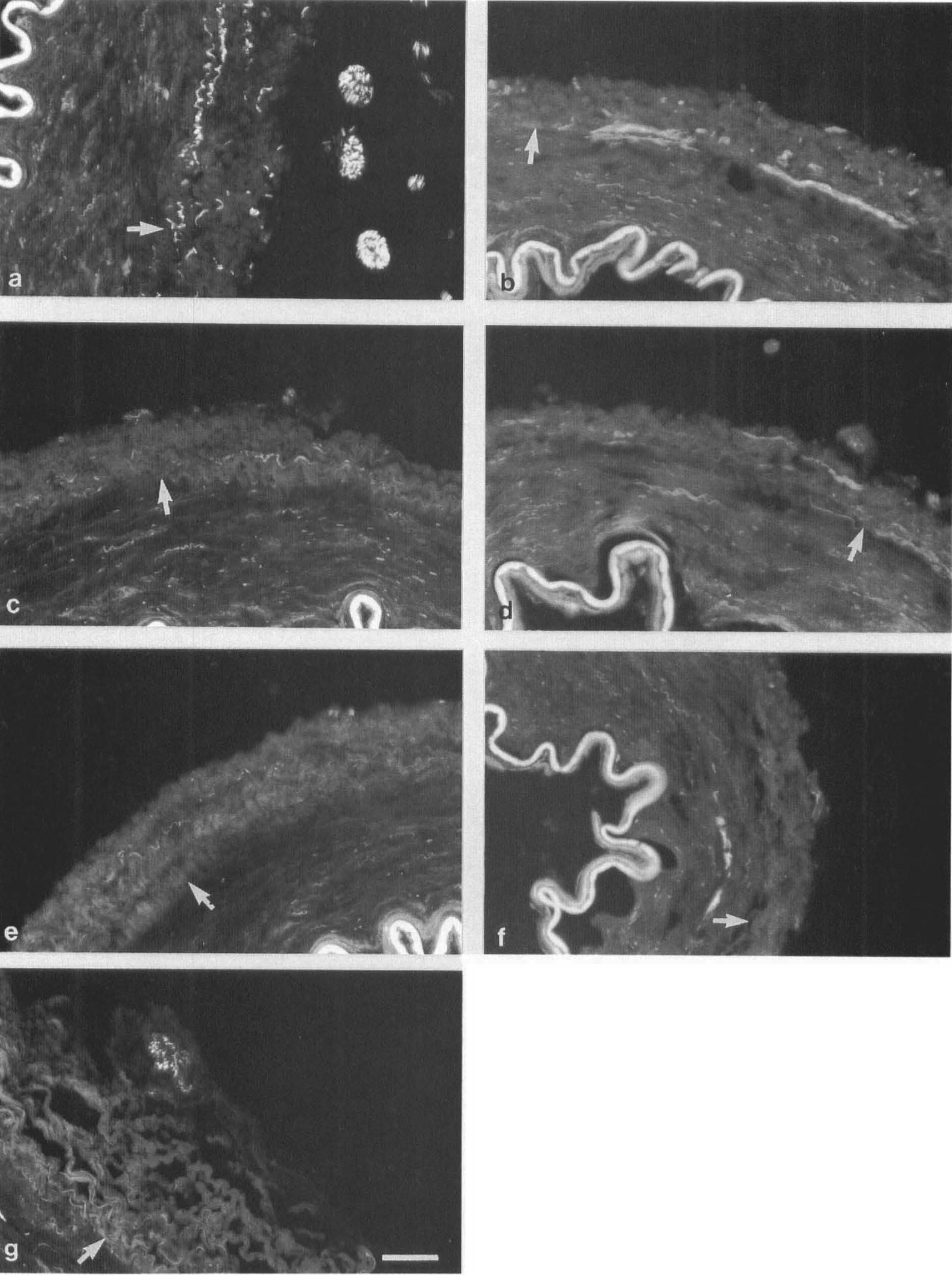
Figure 2 shows staining results for some arteries from the aged and the Alzheimer groups. The results demonstrate that the transversely oriented nerves were usually of a fine caliber and of medium density. The deep plexus of the PCom consisted of very dense bundles of varicose nerve fibers of medium and fine caliber. Many thick, longitudinally oriented, superficial nerve bundles were observed in the ChA.
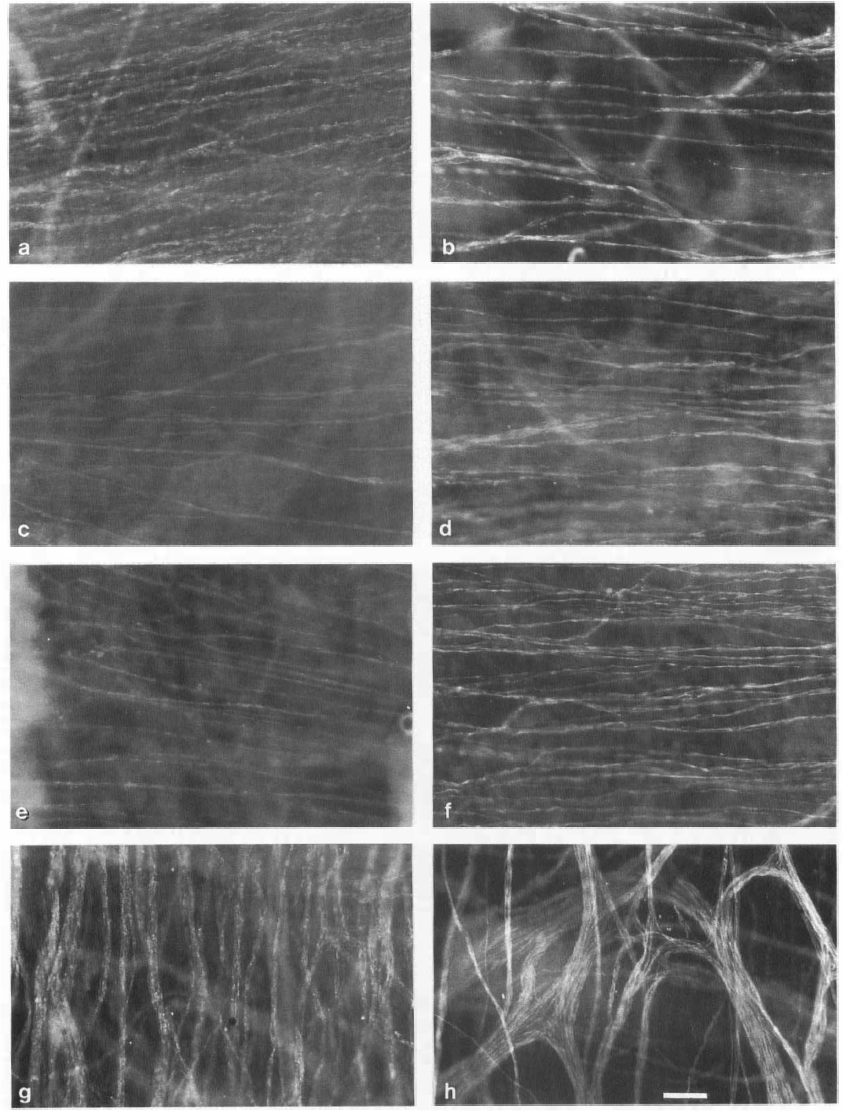
Whole-mount preparations of perivascular nerves of human cerebral arteries, stained for protein gene product 9.5 from aged and Alzheimer groups. The deep plexuses are transversely oriented and located at the adventitial–medial border,
The quality of the staining was variable in a minority of the specimens. When three areas with well-defined nerves could not be found in a segment, the vessel was excluded from quantification.
Image analysis
The number of segments with atherosclerotic plaques was considerably higher than in the middle-aged group (see part I of this article). The ICA, MCA, and the basilar artery especially were affected. Only a few arteries, however, had completely circumferential plaques. For both the aged and the Alzheimer groups, differences in area percent between sclerotic and nonsclerotic segments were tested by t tests. There were no significant differences. Similarly, there were no significant left-right differences. Hence, sclerotic and nonsclerotic arteries and left and right arteries were analyzed together. Furthermore, the data from the two non-trypsin-treated specimens of the aged group were well within the range of the data of the trypsin-treated specimens.
The results from gray value measurements in each group are shown in Table 2. Gray values were lowest in the middle-aged group, but the differences were not statistically significant.
Means ± SD of gray value measurements for the three groups investigated, the mean of each group represents measurements of four arteries (including A1 segment, PCom, P2 segment), derived from two specimens

Aged group
Figure 1a shows the mean values for area percent in the 13 arteries. Nerve density, expressed as area percent, was highest in the PCom (8.84 ± 2.01, mean ± SD) and next highest in the P2 segment (7.24 ± 1.77), A1 segment (6.56 ± 1.77), P1 segment (6.38 ± 1.35), and ChA (6.25 ± 1.69). Analysis of variance (ANOVA) (randomized block design, p < 0.0001) and Fischer protected least significant differences tests (95% significance level) demonstrated that nerve densities in the PCom and the P2 segment were significantly higher than in most other arteries. The randomized block designs (factors were arteries and patients) corrected for variability between patients. Nerve density, expressed as ID/millimeter, correlated well with area percent, showing the highest values for the PCom (57.52 ± 12.58), the P2 segment (47.36 ± 10.65), P1 segment (42.50 ± 7.19), ChA (40.12 ± 13.04), and A1 segment (37.70 ± 14.04). A high correlation between area percent and ID/millimeters was found for all arteries taken together (r = 0.90, n = 98, p < 0.0001), as well as for the individual arteries.
Correlation coefficients between vessel diameter and area percent and ID/millimeter for the individual arteries varied considerably and were generally low. They were not statistically significant (i.e., p < 0.05), showing a lack of correlation between vessel diameter and nerve density.
Comparison for area percent and ID/millimeter between the middle aged and aged groups are shown in Fig. 3. It is clear from the data that nerve density, expressed as area percent, tends to decrease with age. Statistical analysis (one-way ANOVA, assuming that patients in all groups were randomly selected) demonstrated that this decrease was significant for ICA (p < 0.05), P1 (p < 0.05), and ChA (p < 0.05). Nerve density, expressed as ID/millimeter, however, only decreased significantly for the ICA (p < 0.05).
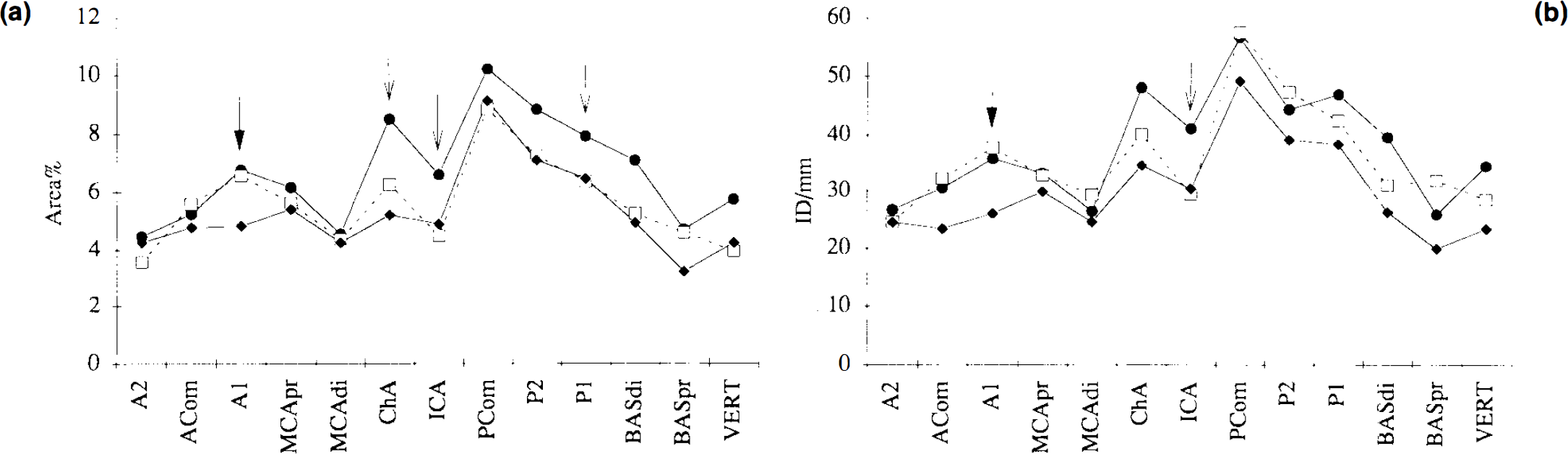
Comparison of the mean values of (
Alzheimer group
Figure 1b shows the mean values for the area percent in the 13 arteries. Nerve density, expressed as area percent, was highest in the PCom (9.12 ± 3.49) and next highest in the P2 segment (7.08 ± 2.17) and P1 segment (6.44 ± 2.30). ANOVA (randomized block design, p < 0.0001) and Fischer PLSD tests (95% significance level) demonstrated that nerve densities in the PCom and the P2 segment were significantly higher than in most other arteries. Again, nerve density, expressed as ID/millimeter, correlated well with area percent, showing the highest values for the PCom (49.20 ± 16.04), the P2 segment (39.15 ± 11.04), and P1 segment (38.40 ± 11.74). A high correlation between area percent and ID/millimeter was found for all arteries taken together (r = 0.88, n = 109, p < 0.0001), as well as for the individual arteries.
As observed in the healthy aged group, correlation coefficients between vessel diameter and area percent and ID/millimeter for the individual arteries were not statistically significant (p < 0.05), showing a lack of correlation between vessel diameter and nerve density.
Comparisons for area percent and ID/millimeter between the three groups studied (middle-aged, aged, and Alzheimer, Fig. 3) showed that nerve density, expressed as both parameters, was significantly lower in the A1 segment of Alzheimer's patients compared with both middle-aged and healthy aged groups (p < 0.05). Furthermore, the comparison showed some similar significant decreases for the Alzheimer group compared with the middle-aged group, as observed in the aged group. Thus, nerve density, expressed as area percent, decreased significantly for the ICA (p < 0.05), P1 (p < 0.05), and ChA (p < 0.01), whereas nerve density, expressed as ID/millimeter, only decreased significantly for the ICA (p < 0.05).
Sections
Visual assessment
Figure 4 shows representative staining results for arteries from both the aged group and the Alzheimer group. Background staining was considerably higher than observed in the middle-aged group. However, PGP 9.5-positive nerves were clearly visible throughout the adventitia. At the adventitial–medial border the nerves were often oriented in a transverse direction. In the PCom these nerve bundles were of a large caliber. The TH staining was less intense than PGP 9.5. Due to the high background, well-defined TH-positive nerves could not be observed in all cases. NF staining by R39 was bright. The number of R39-positive nerves was lower than observed in the middle-aged specimens. Furthermore, they were predominantly of a very fine caliber and mainly confined to the outer zone of the adventitia and to paravascular nerves. R39-positive fibers were found deeper in the adventitia, including the adventitial–medial border, but not consistently. Nerve fibers stained by RT97 were located only in paravascular nerve bundles.
Transverse cryostat of P2 (
Image analysis
Aged group
The results of representative measurements are shown in Fig. 5A. High nerve densities in the PCom and P2 segment on whole-mount preparations were confirmed by these results. Furthermore, results suggest that TH- and R39-positive nerves constitute subpopulations of adventitial nerve plexuses. These results should be considered with care, however, because measurements of nerves stained by different antibodies are compared. Comparisons with measurements on sections in the middle-aged group confirmed the tendency for nerve density to decrease with age. The decrease in PGP 9.5-like immunoreactivity was paralleled by a similar decrease in TH-like immunoreactivity, whereas the decrease for R39 was more dramatic. It must be mentioned that, due to the high background staining and the faintness of the TH staining, TH-like immunoreactivity could not be measured in every vessel. Furthermore, for the same reason, fine TH-positive fibers may have been missed in making measurements. R39 staining, however, was bright, but in some fields there were virtually no labeled nerve fibers, indicating a dramatic decrease for R39. Thus, the results of measurements may be underestimated for TH-positive nerves, but not for the R39-positive nerves.
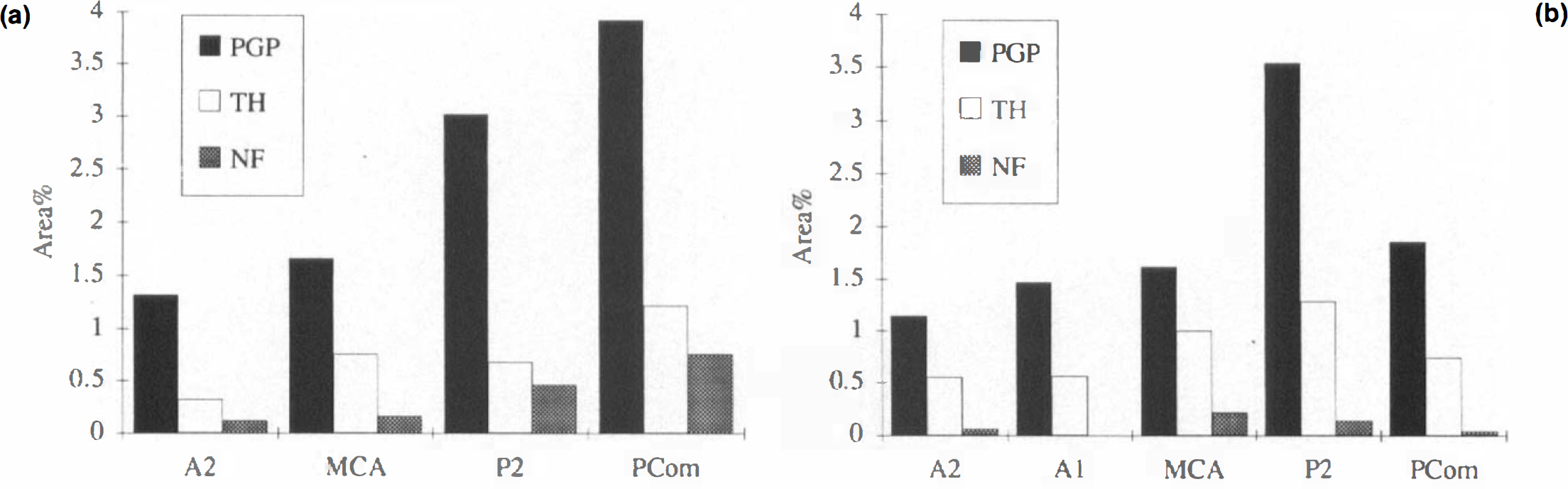
Histograms representing the representative measurements on sections A2, A1 (Alzheimer only), MCA, P2, and PCom of specimens from (
Alzheimer group
The results (Fig. 5B) were in the range of the data of the aged group. PGP 9.5 values for P2, however, were higher than for PCom. One of these specimens was also processed as whole-mount preparations, the image analysis of which, however, showed approximately equal values for PCom and P2. This demonstrates that measurements on sections are less reliable than on whole-mount preparations, as we found before (discussed in part I of this article), and should therefore only be used to establish general trends. Again, the results suggest that TH- and R39-positive nerves constitute subpopulations of adventitial nerve plexuses. As for the healthy aged group, comparisons with measurements on sections in the middle-aged group confirmed the tendency for nerve density to decrease with age, showing parallel decreases in PGP 9.5- and TH-like immunoreactivity, and a more dramatic decrease for R39-stained nerve fibers.
DISCUSSION
In the present study we have quantified densities of deep perivascular nerves of the basal cerebral arteries from aged Alzheimer's and non-Alzheimer's patients, and have compared the results with those obtained from middle-aged patients, as reported in part I of this article. The results demonstrate a significant decrease in nerve density with aging for the ICA, ChA, and P1 segment. In Alzheimer's disease a significant decrease in nerve density was found for the anterior cerebral artery (A1 segment), when compared with the healthy aged group.
Trypsin-treated and non-trypsin-treated specimens were analyzed together in the groups investigated. Morphological changes in aging vessels have been described. Adventitial proliferation of connective tissue fibers occurs in human intracerebral arteries (Ravens, 1978) and accumulation of extracellular material has been observed in the walls of several human arteries (Toda et al., 1980). These changes may explain why most aged and all Alzheimer specimens needed trypsin treatment to increase the penetration of antibodies, and also the fact that nerve staining was not impaired by the trypsin treatment, in contrast to some of the middle-aged specimens (see part I of this article).
Sclerotic arteries were more frequent in the aged specimens than in the middle-aged ones. Comparison of sclerotic and nonsclerotic arteries did not show any differences in nerve density. Thus, atherosclerosis seems not to affect the parameters that we used to establish nerve density. It should be mentioned, however, that the number of circumferential sclerotic plaques in our material was limited. To our knowledge there are no reports that compare perivascular innervation in sclerotic and nonsclerotic arteries. Conversely, it has been suggested that chemical or surgical sympathectomy may be useful in controlling atherosclerosis in specific arterial beds including the carotid arteries (Lichtor et al., 1987). These authors reported that in rhesus monkeys on an atherogenic diet, sympathectomy retarded the development of atherosclerosis. The resulting vasodilation and lowering of the systemic blood pressure may be contributing factors. Other studies have reported a stroke-protective role of sympathetic nerves (Heistad and Marcus, 1979; Hart et al., 1980; Sadoshima et al., 1983; see also discussion in part I of this article). Thus, vascular innervation may have both positive and negative effects in relation to disease.
The high nerve densities found in PCom, P2, and P1 for both the aged and the Alzheimer groups are in accordance with the results of the middle aged group (see part I of this article). The topographical distribution of deep perivascular nerves is to a large extent similar for the three groups investigated (Fig. 5). Furthermore, also in accordance with our previous findings, there appeared to be an absence of correlation between vascular diameter and nerve density. Considering the variability of diameters in segments of the circle of Willis, this emphasizes once more that the density of deep perivascular nerves in the basal cerebral arteries is determined by locality in the arterial network rather than by vessel diameter and suggests close regional differentiation of function.
The gray value measurements indicate comparable expression of the markers used in the three groups investigated. Mean values in the aged and Alzheimer groups were equal, whereas the middle-aged group showed lower values, although the differences were not significant. Increased expression of neurotransmitters has been shown in aging rat cerebral blood vessels (Thrasivoulou and Cowen, 1995).
Age changes
Comparison of the middle-aged and both aged groups showed significant decreases with age in nerve density expressed as area percent in ICA, P1, and ChA. The graphs for ID/millimeter lie closer to each other, indicating that differences for this parameter are not very large and were only significant for ICA. This implies that decrease in nerve density is determined by loss or atrophy of individual nerve fibers rather than by loss of whole nerve bundles, except for ICA. It should be mentioned, however, that, due to coorientation of the transverse direction of the nerves in the deep plexus with the pixel lines across which ID/millimeter was measured, ID/millimeter may be a less reliable parameter than area percent. Age changes in perivascular and other peripheral nerve plexuses occur characteristically in a regionally specific way (Cowen, 1993b). The present observations confirm this view of aging by showing significant differences in only three arteries.
To our knowledge, only a decrease in concentrations of NPY, VIP, calcitonin gene-related peptide (CGRP) and substance P (SP) has been reported in the walls of aging human middle cerebral arteries (MCAs) (Edvinsson et al., 1986, 1987). This was determined, however, by radioimmunoassay, and thus was not able to distinguish the more functionally important nerves at the adventitial–medial border. These findings do not contradict our results, but they do not contribute in drawing valid conclusions on the morphology of aging cerebrovascular nerves either.
In line with our findings are the decreases of PGP 9.5- and TH-like immunoreactive nerve density in the MCA and the posterior cerebral artery in old rats (Andrews and Cowen, 1994; Thrasivoulou and Cowen, 1995). Subpopulations of nerves, however, can follow different patterns of age changes. Whereas noradrenergic and serotonergic nerves supplying cerebral arteries decline markedly in old rats, NPY- and SP-containing nerves remain on the same level compared with young rats (except in the MCA, where SP-positive nerve density decreases), and there is an increase in density of VIP- and CGRP-containing nerves (Mione et al., 1988). Our findings on sections suggest a decrease of TH-positive nerves, consistent with the decline of noradrenergic nerves in old rats.
A dramatic decrease of NF R39-positive nerves in aging was found in sections. As discussed in the preceding article, it has been suggested that NF R39-immunoreactivity represents sensory fibers (Seiger et al., 1984; Dalsgaard et al., 1984; Björklund et al., 1986). The decrease of CGRP- and SP-concentrations in human MCA (Edvinsson et al., 1987) supports this possibility. On the other hand, there is an increase of sensory, CGRP-containing nerves in Wistar rats (Mione et al., 1988), which may be species specific (Cowen, 1993a).
Recent investigations have focused attention on altered nerve-target interactions as an underlying mechanism for age changes in the autonomic nervous system (see Cowen, 1993b for review). There is experimental support for the hypothesis that age changes in target tissues may be responsible for loss of axon collaterals in aging neurons. This has been demonstrated by transplantation experiments in rats, using in oculo transplants of young and old cerebral arteries into young hosts, which showed that the age of the transplants determined the pattern and density of reinnervation (Gavazzi et al., 1992). It was hypothesized that, with aging, altered production of neurotrophic factors by target tissues may be responsible for loss of their innervating nerve fibers. An interesting feature is the fact that sympathetic neurons from the superior cervical ganglion, which supply fibers to cerebral arteries, retain the capacity for regrowth and plasticity in old age (Gavazzi and Cowen, 1993a). Experiments performed to assess the possible involvement of nerve growth factor (NGF) in age-related changes in autonomic neurons have demonstrated that NGF treatment of transplanted old MCAs results in growth of PGP 9.5-immunoreactive nerves, restoring a “young” pattern of innervation (Gavazzi and Cowen, 1993b). Furthermore, miniosmotic pump infusion of exogenous NGF over peripheral nerve plexuses, around cerebral arteries of freely moving old rats induced regrowth of degenerating PGP 9.5-positive nerves (Andrews and Cowen, 1994). If such plasticity of cerebrovascular nerves exists in humans as well, NGF could become important as a therapeutic agent. More insight has to be gained first, however, into the function of cerebrovascular nerves and alterations in function in cerebrovascular diseases. It would be interesting to know whether the decrease in nerve density with aging is related to an increase in the frequency of stroke, because sympathetic nerves have been ascribed a stroke-protective role (Heistad and Marcus, 1979; Hart et al., 1980; Sadoshima et al., 1983).
Changes in Alzheimer's disease
Comparison of the healthy aged and Alzheimer groups revealed almost similar patterns of nerve densities, buta significant decrease in nerve density expressed as area percent and ID/millimeter for the A1 segment was found in Alzheimer's disease. It is known that there is a reduction in regional CBF in Alzheimer's disease, probably due to close coupling of flow and metabolism (Lacombe and Sercombe, 1989). Degeneration of nerves supplying a selective segment of the anterior cerebral artery may be related to a severely decreased activity of neurons in the nucleus basalis of Meynert, as found in Alzheimer's disease (Salehi et al., 1994), because the A1 segments is involved in the vascular supply of the substantia innominata in which this nucleus is located. The exact causes of these region-specific changes in nerve density are unclear.
CONCLUSIONS
In conclusion, deep perivascular nerve plexuses of human basal cerebral arteries are subject to changes caused by aging and Alzheimer's disease. These changes manifest themselves as altered nerve density, which seems to be localized and confined to certain arteries. Recent findings in laboratory animals indicate retained plasticity of the autonomic nervous system in old age and the influence of neurotrophic factors may indicate therapeutic possibilities in the future. However, first, more knowledge of the function of cerebrovascular innervation and its contribution to cerebrovascular disease is necessary.
Footnotes
Acknowledgment:
This project was supported by a travel grant from the Netherlands Organization for Scientific Research (NWO), by the foundation “De Drie Lichten” in the Netherlands, and by a grant to T.C. and B.H. from the British Heart Foundation. We thank the following for generous assistance with the collection of cerebral arteries: Dr. R. Ravid (Coordinator Netherlands Brain Bank, Amsterdam), Mr. W. J. A. van Wolferen (Department of Functional Anatomy, Utrecht University), Dr. N. B. N. Ibrahim (Department of Histopathology, Frenchay Hospital, Bristol), Mr. C. Marriage (Department of Histopathology, Royal Free Hospital School of Medicine, London) and Dr. G. H. Jansen (Department of Pathology, Utrecht University). We thank Dr. D. Dahl (Department of Spinal Cord Injury Research, Harvard Medical School, Boston, MA, U.S.A.) and Professor B. H. Anderton (Department of Neuroscience, Institute of Psychiatry, London) for the generous supply of neurofilament antibodies. We thank Mr. C. Thrasivoulou (Department of Anatomy and Developmental Biology, Royal Free Hospital School of Medicine, London) for expert technical assistance, and Dr. P. Westers (Centre for Biostatistics, Utrecht University) for statistical assistance.
