Abstract
This study was designed to determine age-related changes in autoregulatory responses of the brain stem circulation in vivo. In anesthetized adult (4 to 6 months, n = 8) and aged (24 to 26 months, n = 7) Sprague-Dawley rats, local CBF to the brain stem was determined with laser-Doppler flowmetry and diameters of the basilar artery and its branches were measured through an open cranial window during stepwise hemorrhagic hypotension. In aged rats, the lower limit of CBF auto-regulation shifted upward to 60 to 75 mm Hg from 30 to 45 mm Hg in adult rats. Dilator responses of the basilar artery (baseline diameter: 254 ± 15 μm), large branch (109 ± 23 μm), and small branch (44 ± 10 μm) to hypotension were much smaller in aged rats than in adult rats. The maximum change in diameter of the basilar artery during profound hypotension was significantly smaller in aged rats (11 ± 8%) than that in adult ones (23 ± 12%, P < 0.05); that of the large branch was 12 ± 8% versus 33 ± 17% (P < 0.005); and that of the small branch was 17 ± 7% versus 40 ± 13% (P < 0.0005), suggesting greater attenuation of the responses in the smaller vessels. Thus, this study provides direct evidence that aging diminishes the compensatory dilatation of brain stem arterioles and arteries during hypotension and modifies the autoregulatory plateau of CBF, which seems to increase the risk of the brain stem ischemia during hypotensive conditions.
Autoregulation of CBF, an intrinsic ability to maintain constant cerebral perfusion in the face of blood pressure changes, is modified or disturbed by several pathological conditions such as hypertension, diabetes mellitus, and acute affections of the brain such as ischemia, trauma, and inflammation (Paulson et al., 1990). Aging has also been reported to impair CBF autoregulation in the fore-brain or whole brain by itself (Lartaud et al., 1993) or when accompanied with long-lasting hypertension (Fujishima et al., 1984; Hoffman et al, 1981; Hoffman et al., 1982), and this impairment might lead to the increased incidence of stroke in the elderly during hypotensive conditions. The lower limit of CBF autoregulation in the hindbrain is lower than that in the forebrain in adult animals (Mueller et al., 1977; Sadoshima et al., 1981) as well as infant rabbits (Tuor and Grewal, 1994). Thus, the hindbrain circulation seems to have superior autoregulatory capacity compared with the forebrain. However, few researchers have focused their interest on changes in the hindbrain autoregulation during aging or under other pathological conditions. Laser-Doppler flowmetry permits less invasive and continuous measurement of the relative changes in local CBF to the brain stem (Fujii et al, 1991a; Tuor and Grewal, 1994; Toyoda et al, 1996). Thus, the first goal of this study was to determine the effect of aging on regulation of CBF to the brain stem during hypotension in vivo.
Pial arterioles have been regarded as the primary sites of autoregulatory function by changing their calibers in response to blood pressure changes (Kontos et al., 1978; MacKenzie et al., 1979; Tuor and Farrar, 1984). We have recently shown that large arteries, such as the basilar artery, also actively dilate and contribute to reductions in cerebrovascular resistance around the lower limits of CBF autoregulation, and thereby maintain CBF to the brain stem during severe hypotension (Toyoda et al., 1996). Aging often attenuates cerebral vasodilator responses of both large arteries (Hongo et al., 1988; Hatake et al., 1990; Paternò et al., 1994) and microvessels (Mayhan et al., 1990) to several vasoactive stimuli, and the severity of attenuation was sometimes different among vessels with different sizes (Toyoda et al., 1997a). Thus, if aging altered the brain stem autoregulation, the second goal of this study was to determine whether age-related impairment of dilator responses of the basilar artery and its branches during hypotension varies with different vessel sizes in vivo. For this purpose, we used an open cranial window over the ventral brain stem through which we can observe several vessels with different diameters along with measurement of CBF by laser-Doppler flowmetry (Toyoda et al, 1996).
MATERIALS AND METHODS
Animal preparation
Experiments were performed in male Sprague-Dawley rats, aged 4 to 6 months (adult, n = 8, 538 ± 75 g [mean ± SD]) and 24 to 26 months (aged, n = 7, 637 ± 33 g). Rats were anesthetized with amobarbital sodium, 50 mg/kg intraperitoneally, followed by additional doses of 15 to 20 mg · kg−1 · h−1 intravenously. The depth of anesthesia was examined often by observing the effects of pressure to the paw on heart rate and blood pressure. The trachea was cannulated and each rat was mechanically ventilated with room air and supplemental oxygen after immobilization by 5 to 10 mg/kg d-tubocurarine chloride. Catheters were placed in both femoral arteries: one for the measurement of systemic arterial pressure, and the other for withdrawing blood to change systemic arterial pressure and for sampling arterial blood. A femoral vein was cannulated for the infusion of drugs. Rectal temperature was maintained at 37°C with a heating pad.
A craniotomy was prepared over the ventral brain stem as previously described in detail (Faraci et al., 1987; Fujii et al., 1991b; Toyoda et al., 1996). Portions of the dura and pia mater and the arachnoid membrane were resected. The open cranial window was suffused with artificial cerebrospinal fluid (CSF) (composition in mmol/L: 132 NaCl, 2.95 KCl, 1.71 CaCl2, 0.65 MgCl2, 24.6 NaHCO3, and 3.69 D-glucose), warmed to 37°C, and continuously bubbled with a gas mixture of 5% CO2-95% N2. Suffusion rate of CSF using inlet and outlet ports over the exposed brain stem was 3.0 mL per minute. In CSF sampled from the craniotomies, PCO2 was 39.5 ± 3.3 mm Hg, PO2 was 128 ± 12 mm Hg, and pH was 7.37 ± 0.04. The diameters of the basilar artery, a large branch directly originating from the basilar artery with a baseline diameter ≥70 μm and a small one with a baseline diameter <70 μm were measured in each rat using a microscope equipped with a television camera coupled to a video monitor and a width analyzer (C3161, Hamamatsu Photonics, Hamamatsu, Japan). The images were recorded on videotape for later analyses.
Cerebral blood flow to the ventral brain stem was measured by laser-Doppler flowmetry (ALF 21D Model, Advance Co. Ltd., Tokyo, Japan) equipped with a 550-μm-diameter needle probe as described previously (Fujii et al, 1991a;Toyoda et al, 1996). Briefly, the probe was placed lateral to, but near the basilar artery in the cranial window and advanced into the CSF approximately 0.2 mm above the surface of the brain stem. We regarded laser-Doppler flowmetry outputs as arbitrary units and expressed changes in CBF as a percentage of the baseline CBF. Cerebrovascular resistance, calculated as MABP/CBF, was also described as a percentage of the baseline value.
Experimental protocol
Responses of the basilar artery and branches to acetylcholine (10−6 and 10−5 mol/L) and sodium nitroprusside (10−8 and 10−7 mol/L) were examined in each group. Both drugs were dissolved in artificial CSF and suffused over the craniotomy for 5 minutes. Vessel diameters were measured immediately before application of each drug and during the last minute of application. After application of each drug, 10 minutes of washout periods were allowed before vessel diameters returned to baseline values.
Stepwise hypotension, approximately 15 mm Hg per step, was then induced by controlled withdrawal of blood through the femoral arterial cannula. We maintained MABP at each step for approximately 4 minutes and measured vessel diameter and CBF simultaneously during the last minute, because these parameters were usually stable during the last 2 minutes in each step. Arterial blood gases were determined twice during hypo-tension, at the MABP of 60 mm Hg and 30 mm Hg, as well as before hypotension.
Statistical analysis
Values are expressed as means ± SD. One-way repeated-measures analysis of variance (ANOVA) was used for the comparison of stepwise responses within a group. Two-way repeated-measures ANOVA was used for the comparison of stepwise responses between groups. Unpaired t-test was used to compare differences at each step between groups. P < 0.05 was accepted as statistically significant.
RESULTS
Baseline physiological variables were within normal limits and similar between groups (Table 1). Baseline diameters of the vessels as well as their changes in response to acetylcholine and sodium nitroprusside did not differ between groups.
Physiological variables and vasodilator responses

Values are mean ± SD. Differences between adult and aged rats did not reach statistical significances in any parameters.
In adult rats, CBF was well maintained until MABP was reduced to 45 mm Hg, decreased significantly at 30 mm Hg (P < 0.05, one-way repeated-measures ANOVA), and decreased steeply thereafter (Fig. 1). In aged rats, CBF decreased significantly at 60 mm Hg (P < 0.05), and decreased steeply thereafter. Thus, we regarded 30 to 45 mm Hg as the lower limit of CBF autoregulation in adult rats and 60 to 75 mm Hg as that in aged rats. Changes in CBF through stepwise hypotension were significantly different among the groups (P < 0.02, two-way repeated-measures ANOVA).
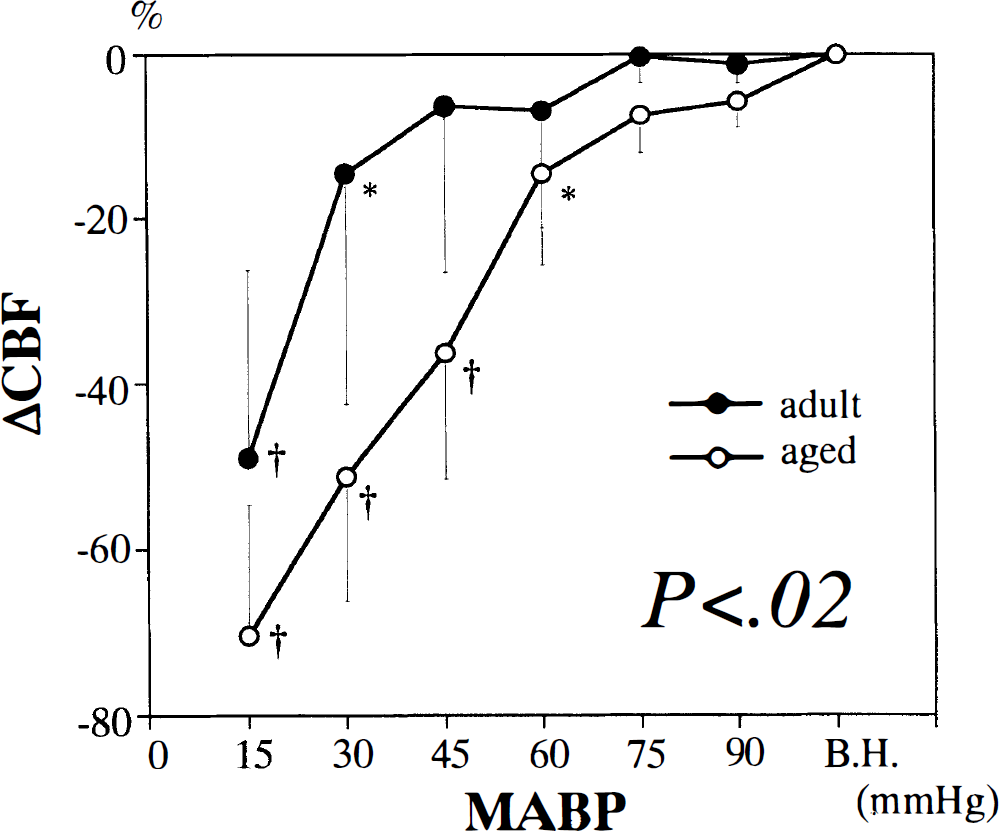
Changes in CBF in adult and aged rats during stepwise hypotension. All values are means ± SD. *P < 0.05. †P < 0.0001 versus before hypotension by one-way repeated-measures analysis of variance (ANOVA). Significant difference (P < 0.02) was shown in MABP-response curves by two-way repeated-measures ANOVA. B.H., before hypotension.
Cerebrovascular resistance decreased constantly during mild and moderate hypotension and reached the minimum level at 30 mm Hg in adult rats (Fig. 2). In aged rats, cerebrovascular resistance decreased linearly until MABP reached 60 mm Hg and changed little thereafter.
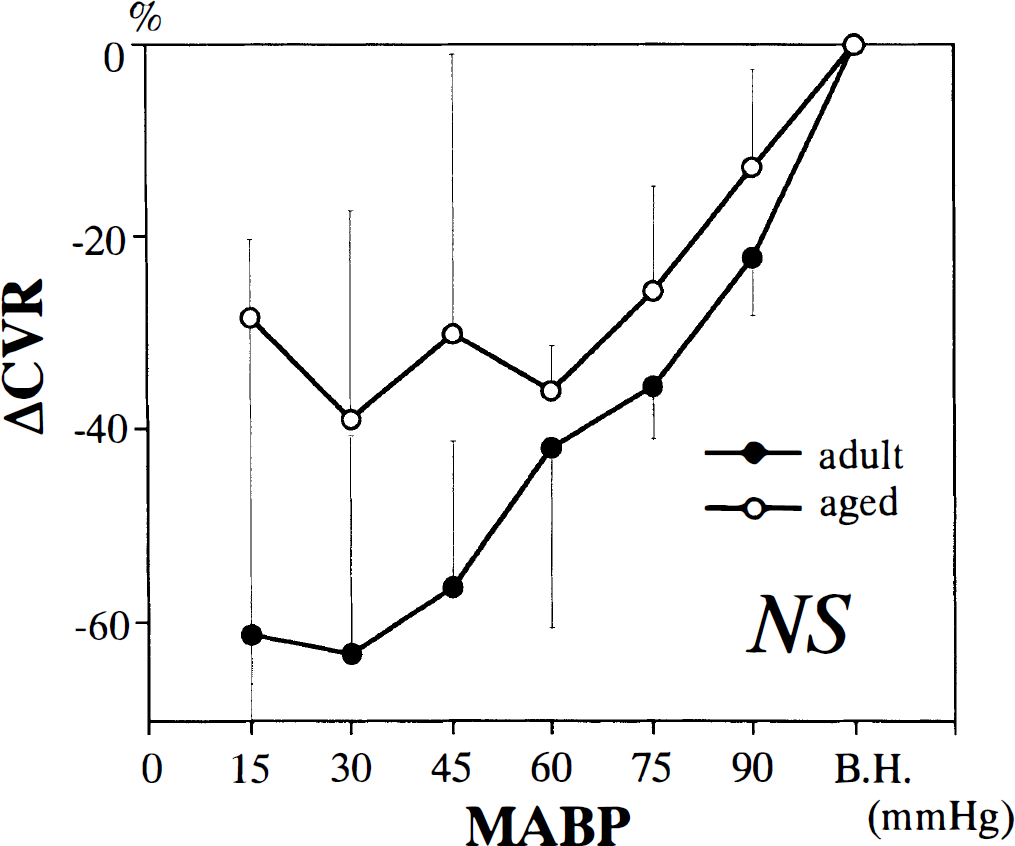
Changes in cerebrovascular resistance in adult and aged rats during stepwise hypotension. All values are means ± SD. Significant difference was not shown in MABP-response curves by two-way repeated-measures ANOVA. B.H., before hypotension. CVR, cerebrovascular resistance; NS, not significant.
Diameter of the basilar artery increased by less than 10% from the baseline value until MABP reached 60 mm Hg, increased steeply thereafter, and reached the maximum level at 30 mm Hg in adult rats (Fig. 3). In aged rats, dilator responses of the basilar artery during moderate and severe hypotension were attenuated compared with adult rats. The maximum change in the diameter (at 30 mm Hg) was 23 ± 12% in adult rats and 11 ± 8% in aged ones (P < 0.05). Changes in the diameter through stepwise hypotension were significantly different between the groups (P < 0.05).
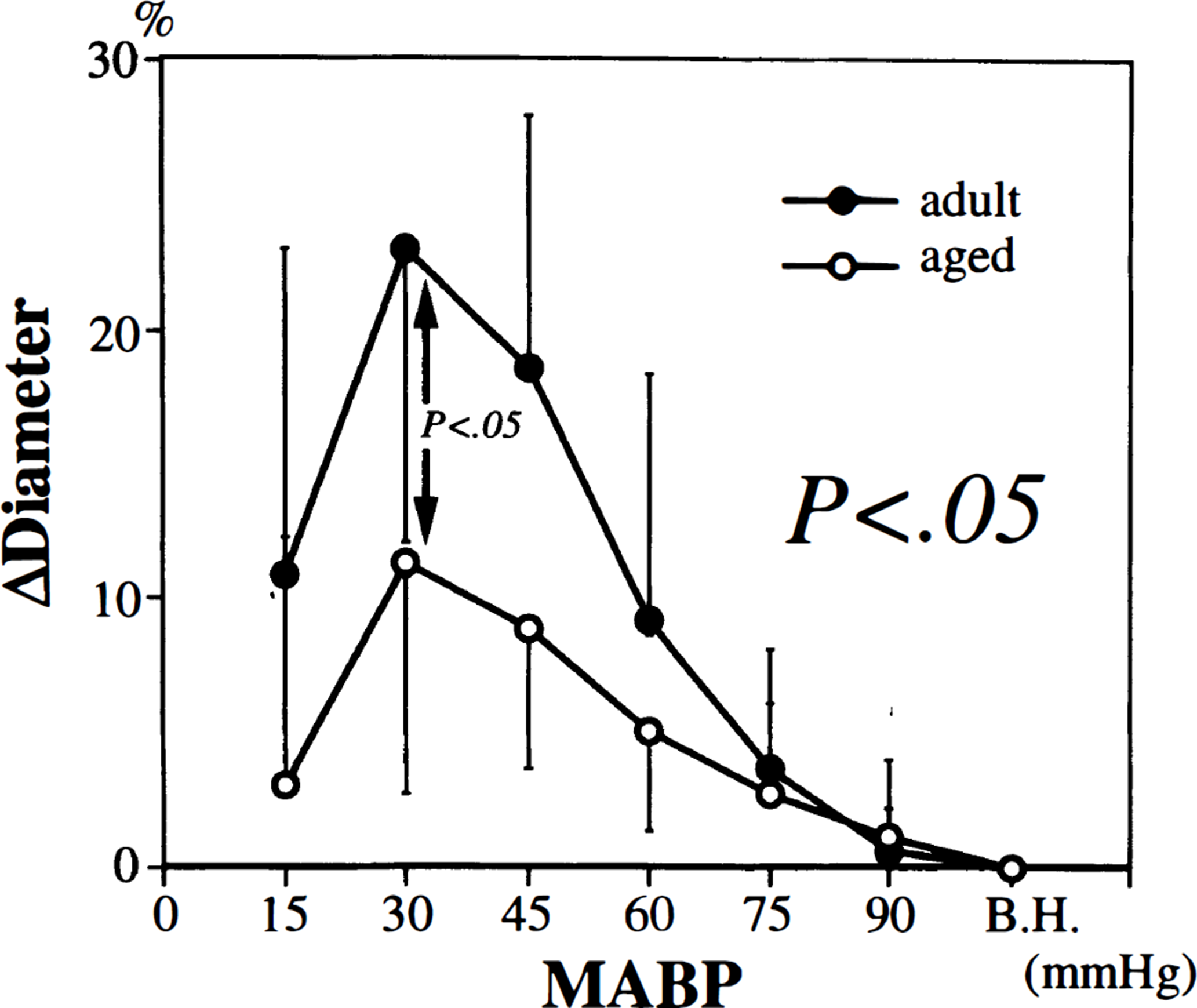
Changes in the diameter of the basilar artery in adult and aged rats during stepwise hypotension. All values are means ± SD. Significant difference (P < 0.05) was shown in MABP-response curves by two-way repeated-measures ANOVA. Significant difference (P < 0.05) was also shown in maximum change in diameter (at 30 mm Hg) by unpaired t-test. B.H., before hypotension.
The dilatations of the large and small branches during moderate and severe hypotension were similarly attenuated in aged rats compared with adult rats (Figs. 4 and 5). The maximum change in diameter of the large branch (at 30 mm Hg) was 33 ± 17% in adult rats and 12 ± 7% in aged ones (P < 0.005); that of the small branch was 40 ± 13% and 17 ± 7%, respectively (P < 0.0005). Changes in the diameter through stepwise hypotension differed to a great extent between the groups for both large (P < 0.005) and small (P < 0.01) branches.
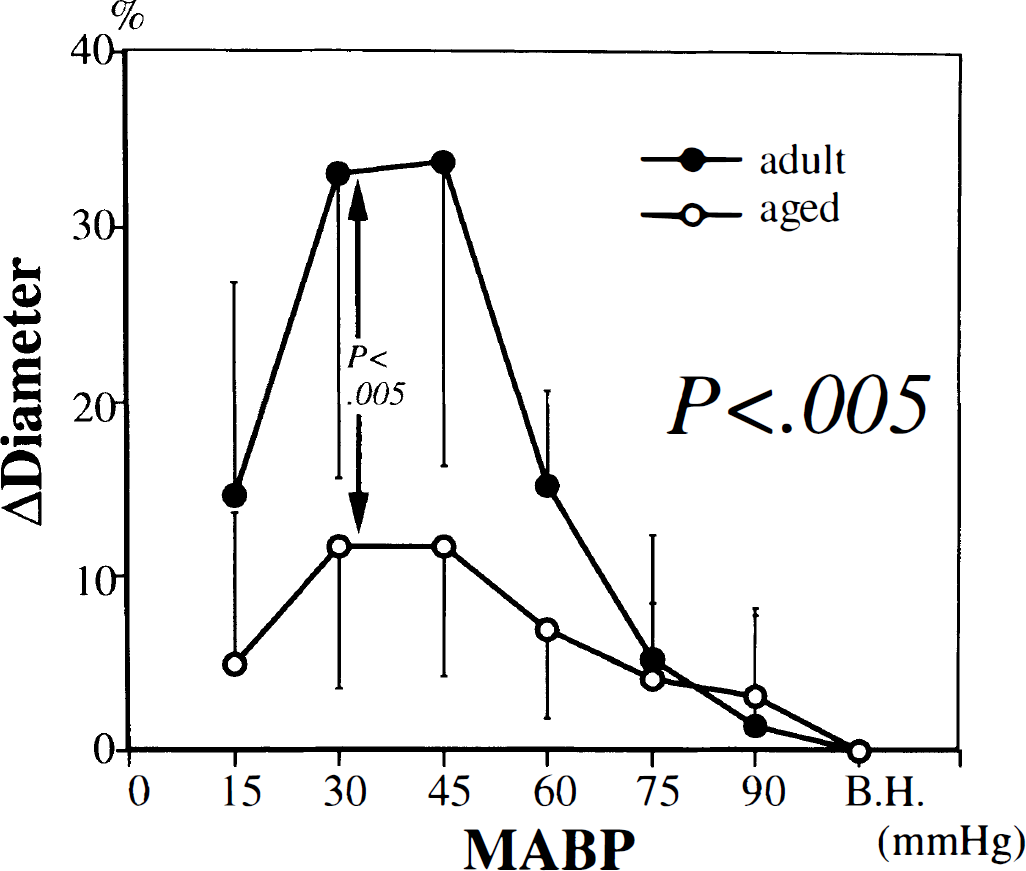
Changes in the diameter of the large branch of the basilar artery in adult and aged rats during stepwise hypotension. All values are mean ± SD. Significant difference (P < 0.005) was shown in MABP-response curves by two-way repeated-measures ANOVA. Significant difference (P < 0.005) was also shown in maximum change in diameter (at 30 mm Hg) by unpaired t-test. B.H., before hypotension.
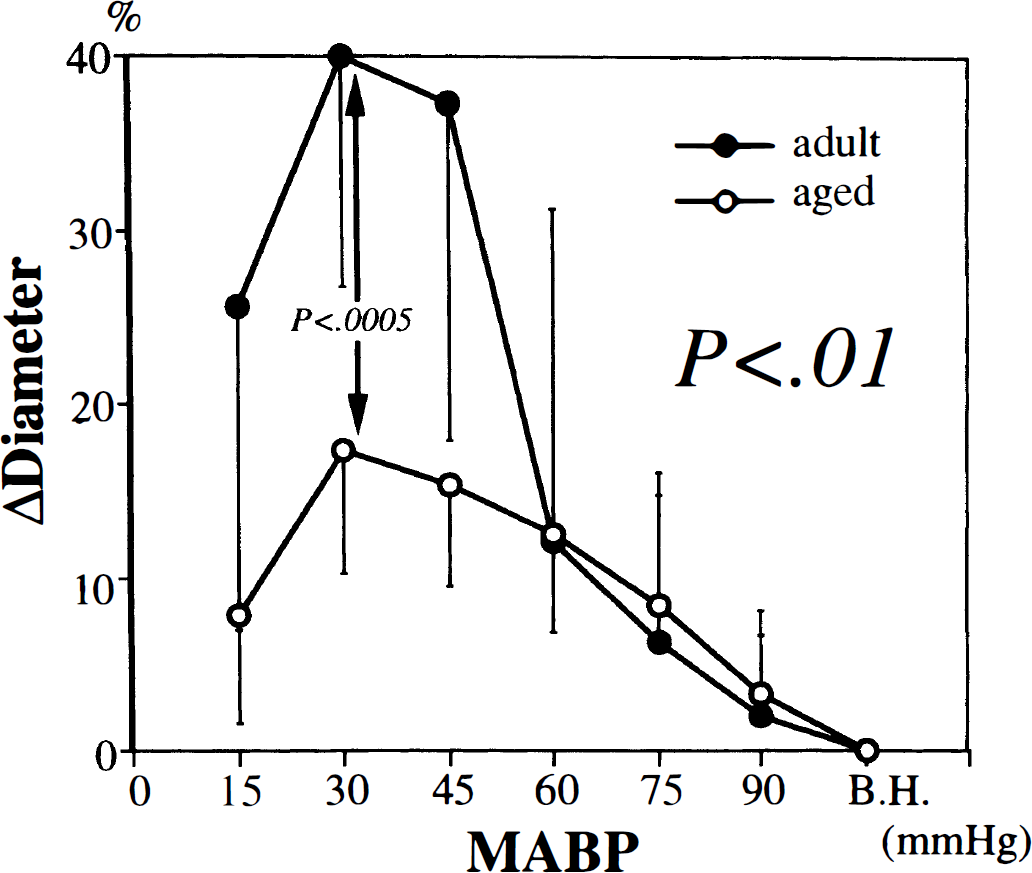
Changes in the diameter of the small branch of the basilar artery in adult and aged rats during stepwise hypotension. All values are mean ± SD. Significant difference (P < 0.01) was shown in MABP-response curves by two-way repeated-measures ANOVA. Significant difference (P < 0.0005) was also shown in maximum change in diameter (at 30 mm Hg) by unpaired t-test. B.H., before hypotension.
DISCUSSION
There are two major new findings in the present study. First, the lower limit of the autoregulatory plateau of CBF to the brain stem was shifted upward in aged rats compared with adult rats. Second, dilator responses of the basilar artery and its branches to hypotension were greatly attenuated in aged rats compared with those in adult rats. In addition, the dilatation of the branches in aged rats appeared to be more severely impaired than that of the basilar artery. Thus, aging seems to impair the autoregulatory dilatation of brain stem arteries and arterioles and attenuate the autoregulatory capacity of CBF to the brain stem.
Consideration of methods
Sprague-Dawley rats are commonly used in research of aging (Masoro, 1980). Male Sprague-Dawley rats have been reported to have a median length of life between 19.5 and 30 months, mostly between 23.5 and 25.5 months (Masoro, 1980). Body weight of the male rats showed rapid increase during the first 4 to 6 months until it reached approximately 80% of its maximum value, and subsequent increments were progressively smaller (Berg, 1960). Thus, rats aged 4 to 6 months in this study belong to mature stage, and rats aged 24 to 26 months belong to senile stage.
The cranial window preparation in the present study has the advantage of simultaneous and continuous observation of changes in CBF and diameters of different-sized vessels. Although this preparation does not allow us to directly observe intraparenchymatous arterioles, presumed major sites for CBF autoregulation, previous researchers postulated that alterations in intraparenchymatous vascular resistance would correspond well to those of the pial arterioles (Tuor and Farrar, 1984).
We used amobarbital for anesthesia in the present study. This anesthesia can alter vessel diameter (Colantuoni et al., 1984) and reduce CBF (Pierce et al., 1962), and may alter autoregulatory responses to hypotension. Constant suffusion with artificial CSF over the open cranial window may also bring the confounding influence on the autoregulatory responses, because this technique washes away locally released vasoactive factors, potent modulators of the vascular tone. The lower limit of CBF autoregulation in our adult rats, 30 to 45 mm Hg, was almost the same as that of the hindbrain circulation in previous studies in which other types of anesthesia and techniques other than the cranial window were used (Mueller et al., 1977: Sadoshima et al., 1981), and thus, we consider that barbiturate anesthesia and the open cranial window technique by themselves do not seem to have a major influence on CBF autoregulation during hypotension. We cannot exclude the possibility, however, that the autoregulatory capacity is somewhat larger or smaller in the absence of anesthesia or under conditions using a closed cranial window.
Aging and vasodilator responses to agonists
Aging does not impair cerebral vasodilator capacity nonspecifically, because the responses of cerebral vessels to nitroglycerin (Mayhan et al., 1990) or sodium nitroprusside (the present study) were preserved in aged animals. In contrast, aging attenuates some responses to endothelium-dependent agonists such as acetylcholine, ATP, and bradykinin (Hongo et al., 1988; Hatake et al., 1990; Mayhan et al., 1990; Paternò et al., 1994) and endothelium-independent agonists such as openers of ATP-sensitive potassium (KATP) channels (Toyoda et al., 1997a). Paternò et al. (1994) reported that endothelium-dependent responses were not significantly impaired in 24 to 26-month-old rats which accords with our findings, but were significantly impaired in 30 to 32-month-old rats. Thus, endothelial dysfunction may have been observed if we used rats much older than those in the present study. Diminished vasodilator capacities during aging may partly result from morphological and functional alterations of the vessel wall of large arteries (Cox et al., 1977; Nagasawa et al., 1979; Haudenschild et al., 1981) and arterioles (Hajdu et al., 1990).
Aging and CBF autoregulation
Decline in basal forebrain CBF and impairment of its autoregulatory response have been reported in aged rats without complications (Lartaud et al, 1993) as well as in aged people (Wollner et al., 1979; Shaw et al., 1984). Latraud et al (1993) showed that the lower limit of CBF autoregulation shifted from 70 to 90 mm Hg during maturation or senility in both Wistar and Fisher 344 rats. In contrast, it shifted from 30–45 to 60–75 mm Hg in our Sprague-Dawley rats, judging from the endpoint of CBF autoregulatory plateau (Fig. 1) or the bottom of cerebrovascular resistance changes (Fig 2). Differences in these values of the two studies seem to mainly result from the difference between forebrain and hindbrain circulations.
In addition, this study has provided for the first time direct evidences that aging diminishes the compensatory dilatation of brain stem arterioles and arteries during hypotension, and modifies autoregulatory plateau of CBF to the brain stem. High blood glucose level and high blood pressure, which are well-known risk factors for cerebral arteriosclerosis, were not observed in aged rats in the present study, and aging per se seems to be responsible to a large extent for the disturbance of auto-regulatory adjustment in these rats.
The dilatation of vessels of all sizes were significantly attenuated in aged rats. Dilator responses of the branches, however, seemed to be more severely impaired than the basilar artery. Changes in vascular tone of the basilar artery and branches seem to be regulated by different mechanisms during hypotension. For example, although topical application of N[omega]-nitro-L-arginine, a non-selective inhibitor of nitric oxide synthase, significantly attenuated the dilator response of the basilar artery during hypotension, it did not affect the response of the branches (Toyoda et al., 1997b). The finding suggests that nitric oxide is not a major regulator of vascular tone of small vessels during hypotensive conditions.
Opening of KATP channels is another potent mechanism of cerebral vasodilation (Kitazono et al., 1995). Topical application of glibenclamide, a selective KATP-channel blocker, disturbed the autoregulatory dilatation of pial arterioles of rats (Lee et al. 1993; Hong et al., 1994), suggesting that KATP channels are important modulators of CBF autoregulation. In our previous study, dilator response of the brain stem arterioles to activation of KATP channels was impaired in aged rats (Toyoda et al., 1997a). Thus, age-related attenuation of autoregulatory capacity of the rat brain stem may partly result from age-related impairment of the dilator response of the brain stem arterioles to activation of KATP channels.
In summary, aging is an independent factor that attenuates autoregulatory responses of the brain stem vessels and may increase the risk of the brain stem ischemia during hypotensive conditions.
Footnotes
Abbreviations used
Acknowledgements
The authors thank Drs. Takanari Kitazono and Tetsuhiko Nagao in the Second Department of Internal Medicine at Kyushu University for their valuable comments, and the staff in the Animal Research Center and Department of Dental Anesthesiology at Kyushu Dental College for technical advice.
