Abstract
In the present study the topographical distribution of the intrinsic nerve plexuses of the basal cerebral arteries in humans was quantified and the relation between vessel diameter and nerve density was investigated. Whole-mount preparations of various segments of the basal cerebral arteries from middle-aged patients were stained for protein gene product (PGP) 9.5. The deep nerve plexuses, located at the adventitial–medial border, were quantified by image analysis. Confocal scanning laser microscopy was used to study nerve plexuses throughout the adventitia. Transverse cryostat sections were stained for PGP 9.5, tyrosine hydroxylase and neurofilament, and quantified. The results showed a three-layered configuration of the adventitial nerves. Measurements on whole-mounts demonstrated that nerve densities were highest in the posterior communicating artery (PCom), and next highest in the proximal parts of the posterior cerebral artery (PCA) and anterior choroidal artery. There appeared to be no clear relation between nerve density and vessel diameter. The measurements on sections confirmed the high nerve densities in the PCom and PCA. Tyrosine hydroxylase- and neurofilament-immunoreactivities appeared to demonstrate separate subpopulations of the overall nerve plexuses, representing sympathetic and, possibly, sensory fibers, respectively. Densities of both subgroups generally followed those of PGP 9.5-immunoreactive nerves. Transmission electron microscopy suggested motor function of the deep nerve plexuses. The results indicate a stronger neuronal influence on this part of the cerebral circulation than hitherto reported. It is concluded that human basal cerebral arteries display a topographical distribution of deep perivascular nerves, and that nerve density is determined by locality rather than by vascular diameter.
Keywords
In the past few decades, knowledge of the nerve supply to the major cerebral arteries in various animals has increased markedly. The perivascular nerves have been classified as sympathetic, parasympathetic, and sensory, with each group being characterized by particular neurotransmitters (Edvinsson et al., 1987a). The functions of cerebrovascular nerves, however, have not yet been completely elucidated. First, there is continuing controversy about the role of the cerebrovascular innervation in the regulation of CBF. It has become clear that it is the pial arteries that are mainly involved in the cerebral autoregulation (see for review Gotoh and Tanaka, 1988) and that there is a well-defined role for sympathetic nerves in shifting the limits of autoregulation to higher pressure levels during hypertension (Heistad and Marcus, 1979; MacKenzie et al., 1979; Edvinsson, 1982). The role of sympathetic nerves during the plateau phase of autoregulation is probably minor (Edvinsson et al., 1993). Studies on the regulation of CBF have focused on the role of the pial arteries, and, more recently, the importance of the internal carotid and vertebral arteries in this respect has been emphasized (Mchedlishvili, 1986; Faraci and Heistad, 1990). Considerably less attention, however, has been paid to the neuronal regulation of the basal cerebral arteries (circle of Willis and major branches), despite the fact that the anastomoses in this part of the cerebral circulation can compensate for flow disturbances. Second, there is evidence that sympathetic nerves exert a trophic influence on the arteries during development (Bevan, 1984) and maturity (Dimitriadou et al., 1988), whilst in maturity the blood vessels provide trophic support for their innervating neurons which may decline in old age (Gavazzi et al., 1992; Co wen, 1993; Gavazzi and Cowen, 1993). Third, there is much debate about the involvement of cerebrovascular nerves in vascular pain syndromes, e.g., migraine and cluster headache (Moskowitz, 1984; Edvinsson, 1991; Hardebo, 1991a, 1991b; Olesen, 1991).
Investigations in humans have been scarce so far, probably due to technical reasons. Perivascular nerve fibers containing noradrenaline, acetylcholinesterase (AChE), serotonin, and neuropeptides have been demonstrated (Edvinsson et al., 1976; Uddman et al., 1981; Edvinsson and Uddman, 1982; Akiguchi et all, 1983; Griffith and Burnstock, 1983; Allen et al., 1984; Edvinsson and Ekman, 1984; Edvinsson et al., 1986; Chertok et al., 1987; Edvinsson et al., 1987b, 1987c; Jansen et al., 1992; Edvinsson et al., 1994). Quantitative studies, based on the morphology (i.e., counting nerve fibers) of the perivascular nerves in the basal cerebral arteries, are few in number. They are limited to data from human fetuses and lack a detailed survey of all parts of the circle of Willis and major branches (Kawamura et al., 1991; Kawamura and Takebayashi, 1994). Furthermore, these reports do not investigate and distinguish between specific parts of the nerve plexuses, e.g., fibers of passage or terminal fibers.
A basic step in revealing the mechanisms of neuronal regulation in human basal cerebral arteries is to provide a detailed description of the nerve fibers in these arteries, based on quantitative data. Considering the variation in arterial diameters at the base of the human brain, it is of interest to investigate the relationship between vessel diameter and nerve density as well. Therefore, the aim of the present study was to quantify the topographical distribution of the overall nerve fiber population supplying arteries at the base of the human brain, and to investigate the relationship between vessel diameter and nerve density. According to various authors (Borodulya and Pletchkova, 1976; Nakakita et al., 1983), nerves around and in the walls of arteries can be classified into several layers, the innermost of which is located at the border between the tunica adventitia and media, and transversely oriented. It is this innermost plexus that is supposed to be functionally important because of the close neuromuscular relationship and the occurrence of varicosities (Nelson and Rennels, 1970; Nielsen et al., 1971; Burnstock, 1975). For this reason, the present study focuses on the deep, intrinsic nerve plexuses. These have been immunohistochemically stained for the general neural marker protein gene product (PGP) 9.5 (Thompson et al., 1983; Gulbenkian et al., 1987; Wang et al., 1990; Cowen and Thrasivoulou, 1990) in whole-mount preparations and quantified using computerized image analysis (Cowen et al., 1986; Cowen and Thrasivoulou, 1992). Care was taken to establish an optimal procedure for whole-mount immunohistochemistry on human arteries. In order to obtain some understanding of subpopulations of adventitial nerves, transverse sections of some of the arteries were stained for PGP 9.5, tyrosine hydroxylase (TH) and neurofilament (NF), and the adventitial nerves quantified. Additional morphologic information was provided by confocal scanning laser microscopy and transmission electron microscopy (TEM).
The quantitative data of cerebrovascular nerves in humans obtained thus far are a conditio sine qua non for further investigations concerning, for instance, age changes and changes caused by disease.
MATERIALS AND METHODS
Human tissues
Eleven sets of human major cerebral arteries, obtained from postmortems, were processed. With the exception of a fetal specimen (32 weeks gestational age), the patients' ages ranged from 24 to 54 years. Relevant details of the specimens are listed in Table 1. The specimens were fixed by immersion in 4% paraformaldehyde in phosphate-buffered saline (PBS) at 4°C for 15–24 h. Two specimens were fixed for a shorter period (Table 1). Subsequently the specimens were rinsed and stored in PBS containing 15% sucrose at 4°C. Six of the 10 middle-aged specimens (ages 32–52 years) were used for whole-mount immunohistochemistry, 3 of them for confocal scanning laser microscopy, four of them for immunohistochemistry on sections and two of them for TEM (Table 1). The vessels used for TEM were differently fixed (see TEM section). The fetal specimen was only studied using whole-mount preparations.
Relevant details of human specimens
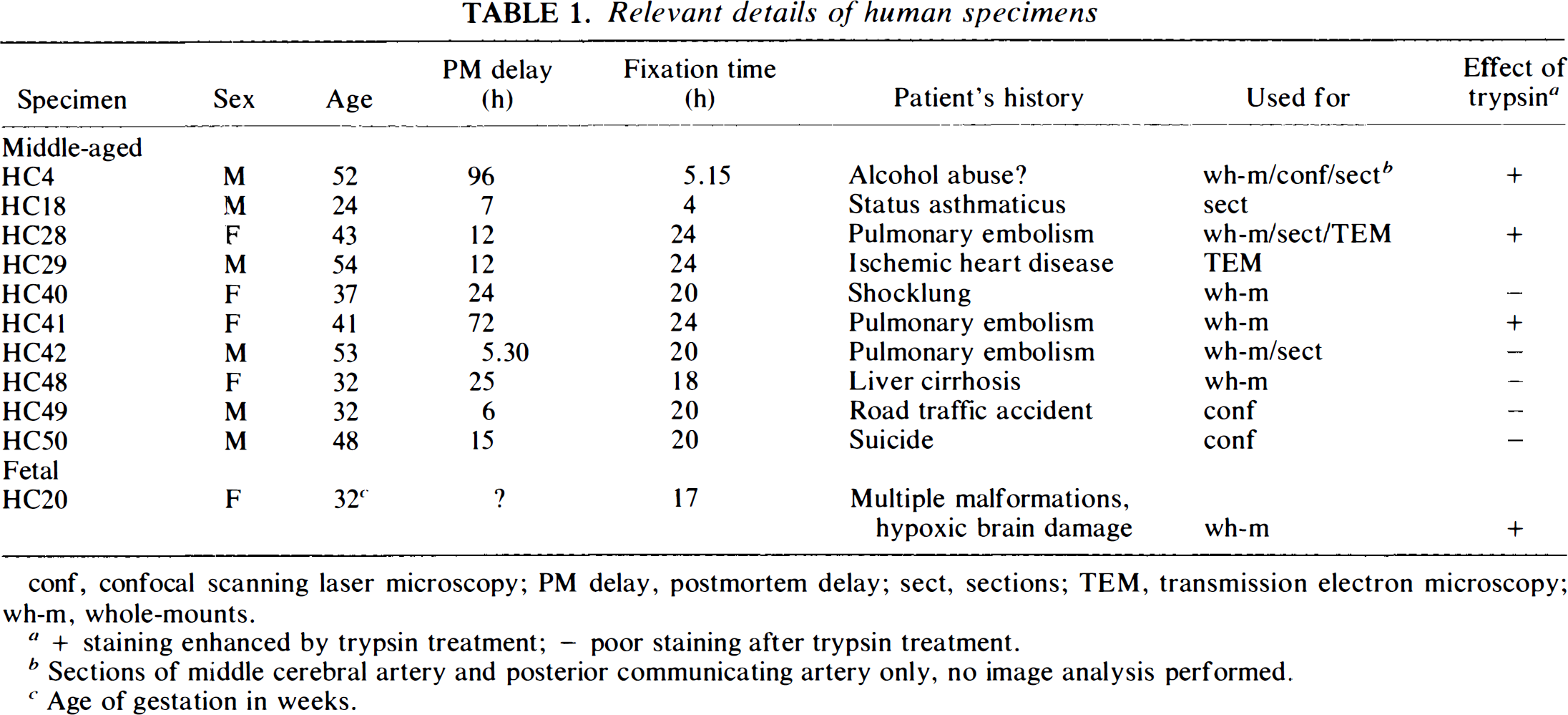
conf, confocal scanning laser microscopy; PM delay, postmortem delay; sect, sections; TEM, transmission electron microscopy; wh-m, whole-mounts.
+ staining enhanced by trypsin treatment; – poor staining after trypsin treatment.
Sections of middle cerebral artery and posterior communicating artery only, no image analysis performed.
Age of gestation in weeks.
Whole-mount preparations
From each set of arteries, 22 segments with a length of 4–5 mm were taken (Fig. 1). The anterior communicating artery (ACom) was short and therefore taken entire. The presence of atherosclerotic plaques was noted. Relative measures of the vessel diameters were obtained by the method described by Hillen (1986). Subsequently the segments were cut open in a longitudinal direction and mounted on pieces of Sylgard 184 (BDH Chemicals, Poole, England) with entomology needles. To improve the penetration of the antibodies, a pretreatment by various steps of freezing and thawing and incubation with 0.125% trypsin type III (Sigma, St. Louis, MO, U.S.A.), in Hanks solution, for 15 min at room temperature (RT), was performed. If trypsin treatment resulted in poor staining, tested on additional segments beforehand, it was omitted for the particular specimen. The effect of trypsin treatment is listed in Table 1. The final part of the pretreatment consisted of manually performed delamination. Under the dissecting microscope the outer zone of the tunica adventitia, including the paravascular nerves, the tunica intima, and most of the tunica media were carefully removed. Immunohistochemistry was performed by the indirect method, at RT. The specimens were washed in Hepes buffer containing 0.1% Triton X-100 for 40 minutes, followed by pre-incubation in 5% swine serum in HEPES buffer for 90 min. Subsequently the specimens were incubated for 40 h (2 nights) in anti-PGP 9.5 antibodies (Ultraclone Ltd., Isle of Wight, England) diluted 1:400 in HEPES buffer, containing 1% swine serum, 0.1% Triton X-100, and 0.1% DL lysine. After washing in PBS, the specimens were incubated in fluorescein isothiocyanate (FITC)-conjugated swine-anti-rabbit antiserum (Dakopatts, Glostrup, Denmark) diluted 1:40 in PBS, containing 1% swine serum, 0.1% Triton X-100, and 0.1% DL lysine, for 90 min. The specimens were washed in PBS, stained with 0.05% pontamine sky blue in PBS, and 1% dimethylsulfoxide (DMSO) for 15 min, to- reduce background autofluorescence (Cowen et al., 1985), and washed again in PBS. The vessels were mounted as stretch preparations on mica in antifade mountant (Citifluor, London, England).
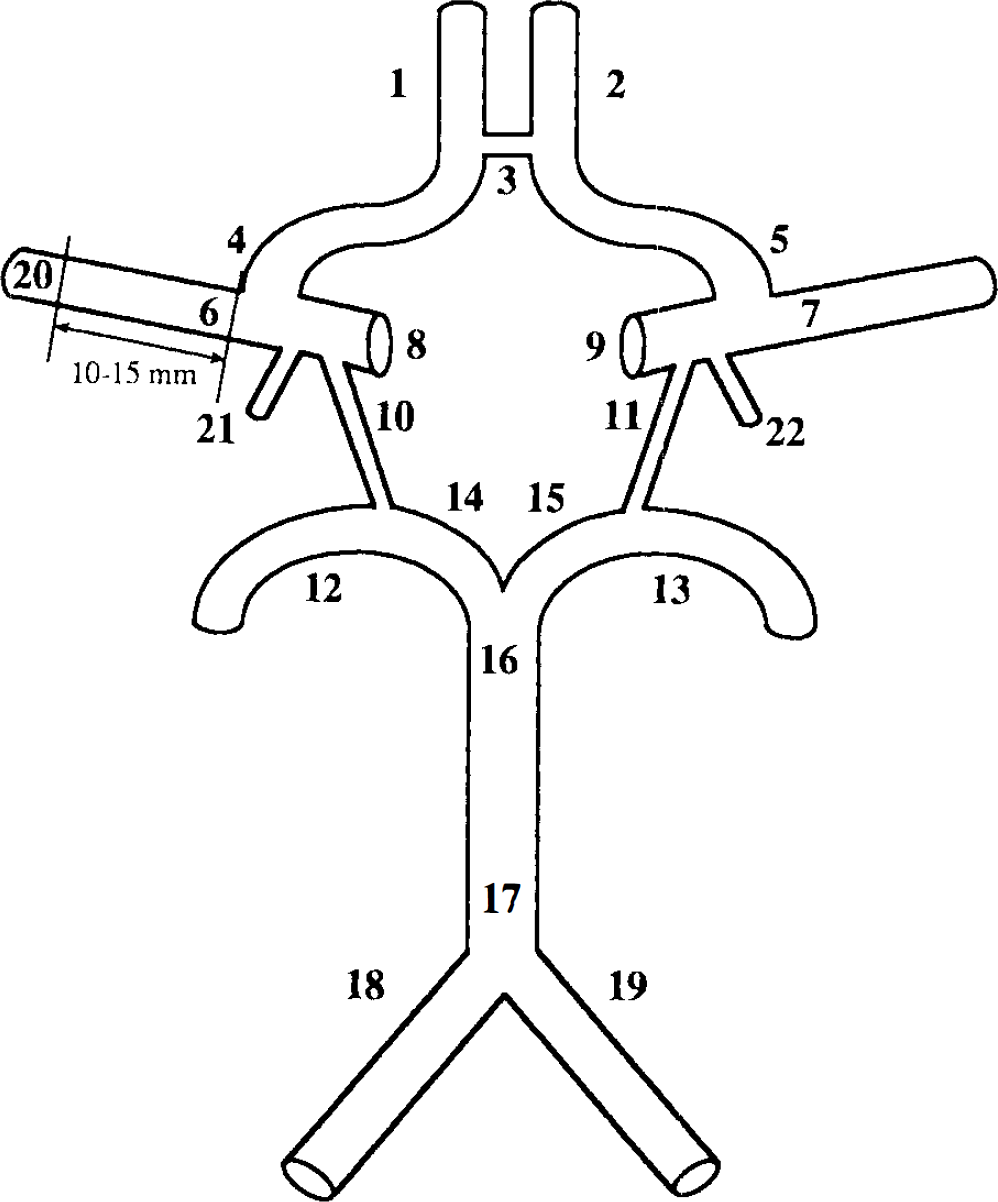
Drawing of the circle of Willis with its afferent and major efferent arteries. The 22 segments investigated as whole-mount preparations are indicated. 1, 2: A2 (postcommunicating part of anterior cerebral artery); 3: ACom (anterior communicating artery); 4, 5: A1 (precommunicating part of anterior cerebral artery); 6, 7: MCApr (proximal part of middle cerebral artery); 8, 9: ICA (internal carotid artery); 10, 11: PCom (posterior communicating artery); 12,13: P2 (postcommunicating part of posterior cerebral artery); 14, 15: P1 (precommunicating part of posterior cerebral artery); 16: BASdi (distal of basilar artery); 17: BASpr (proximal apart of basilar artery); 18, 19: VERT (vertebral artery); 20: MCAdi [distal part of middle cerebral artery (10–15 mm from MCA-A1 bifurcation)]; 21, 22: ChA (anterior choroidal artery).
PGP 9.5-like immunoreactivity was quantified by established methods of image analysis (Cowen et al., 1986; Cowen and Thrasivoulou, 1992). Recognition of the deep plexus was aided by the transverse orientation of its nerves, which were, in addition, the deepest nerves observable when focusing through the tunica adventitia. An image of the nerve plexus was taken into the image analyzer. Images were subject to background subtraction, standard image enhancement, and thresholding routines. The resulting binary image of the nerve plexus was measured automatically using the following parameters: area percent (percentage of measuring frame covered by specific fluorescence), which measured the total number of nerve fibers; intercept density/millimeter (ID/mm; number of nerve bundles per millimeter), which assessed numbers of nerve bundles; and angle (the angles of the nerve bundles with the transverse axis). Of every vessel three areas were measured at random. The mean values were used for statistical analysis. To estimate the scatter of the measurements, on one vessel 10 × 3 additional measurements were made. The data were analyzed by analysis of variance (ANOVA),
The results of ID/millimeters could be underestimations of the true values, resulting from coorientation of the transverse direction of the nerves in the deep plexus with the pixel lines across which ID was measured (Cowen et al., 1982). Data on the orientation of the plexus were therefore required to assess this information.
Confocal scanning laser microscopy was performed on additional whole-mount preparations of middle cerebral artery (MCA) and posterior communicating artery (PCom), partially delaminated (only the intima and part of the media were removed) and stained for PGP 9.5, using a Bio-Rad MRC 600 microscope fitted with a krypton-argon laser. Successive 4-μm optical sections of the adventitial nerves were taken in by the confocal microscope using a 20× objective and stored on optical disc, creating files that were then used to generate images from different depths of the adventitial nerve plexuses.
Sections
Selected vessels from three sets of arteries were used for sections. These vessels included the A2 segment, MCA, P2 segment, and PCom (Fig. 1 for abbreviations). Eight-micron sections were cut in a cryostat. The immunohistochemical staining procedure was as described for whole-mount preparations. Primary antibodies and dilutions were: anti-PGP 9.5 1:800, anti-TH (Eugene Tech International Inc, Ramsey, NJ, U.S.A.) 1:100, anti-NF (R39), 1:1,000, and anti-NF (RT97), 1:50. Secondary antibodies and dilutions were: FITC-conjugated sheep-anti-mouse (Dakopatts) 1:80 for RT97, and FITC-conjugated swine-anti-rabbit 1:80 for all other primary antibodies. Pontamine sky blue staining was carried out without DMSO for 5 min. The sections were mounted in antifade mountant.
For each marker, except for RT97, specific fluorescence was quantified in randomly selected fields as before. Because it was difficult to quantify selectively those nerves at the adventitial–medial border, we quantified the specific fluorescence in the whole adventitial thickness. Large paravascular nerve bundles were excluded from the measurements. The following parameters were measured: field area (the area of specific fluorescence); length of the adventitial–medial border along which the field area was measured; thickness of the adventitia. With these parameters an estimation of the area percent could be computed. This measure corrected for differences in thickness of the arterial wall, because a thick adventitia could produce a high field area, irrespective of the density of the deep nerve plexus. Three fields were measured for each vessel. The mean values were used for comparisons.
TEM
P2 segments and PCom were taken from two sets of freshly dissected arteries after minimum postmortem delay (12 h) and prepared for TEM. These vessels were fixed by overnight immersion in 1% paraformaldehyde and 2% glutaraldehyde in piperazine-
RESULTS
Whole-mount preparations
Visual assessment
The method described for whole-mount preparations resulted in good staining quality in many cases, characterized by a well-defined, transversely oriented deep plexus. Not only deep plexuses, but also superficial plexuses were visible, depending on the degree of adventitial delamination. The superficial plexus formed a meshwork. In specimens that were used in the earlier experiments for establishing the staining procedure and that had not been delaminated, a three-layered plexus of nerves could be seen by focusing through the adventitia: (a) large longitudinally arranged nerve bundles, outside the adventitia; (b) a superficial plexus with large meshes within the adventitia; and (c) a deep, transversely oriented, plexus at the adventitial–medial border. Figure 2 shows some of the staining results of the middle-aged specimens. The transversely oriented nerves of the deep plexus were usually of a fine caliber and medium density. However, in contrast, the deep plexus of the PCom consisted of very thick, dense bundles of nerve fibers with varicosities (Fig. 2c). The anterior choroidal artery (ChA) contained not only a dense deep plexus, but also many longitudinally oriented, superficial nerve bundles (Fig. 2f).
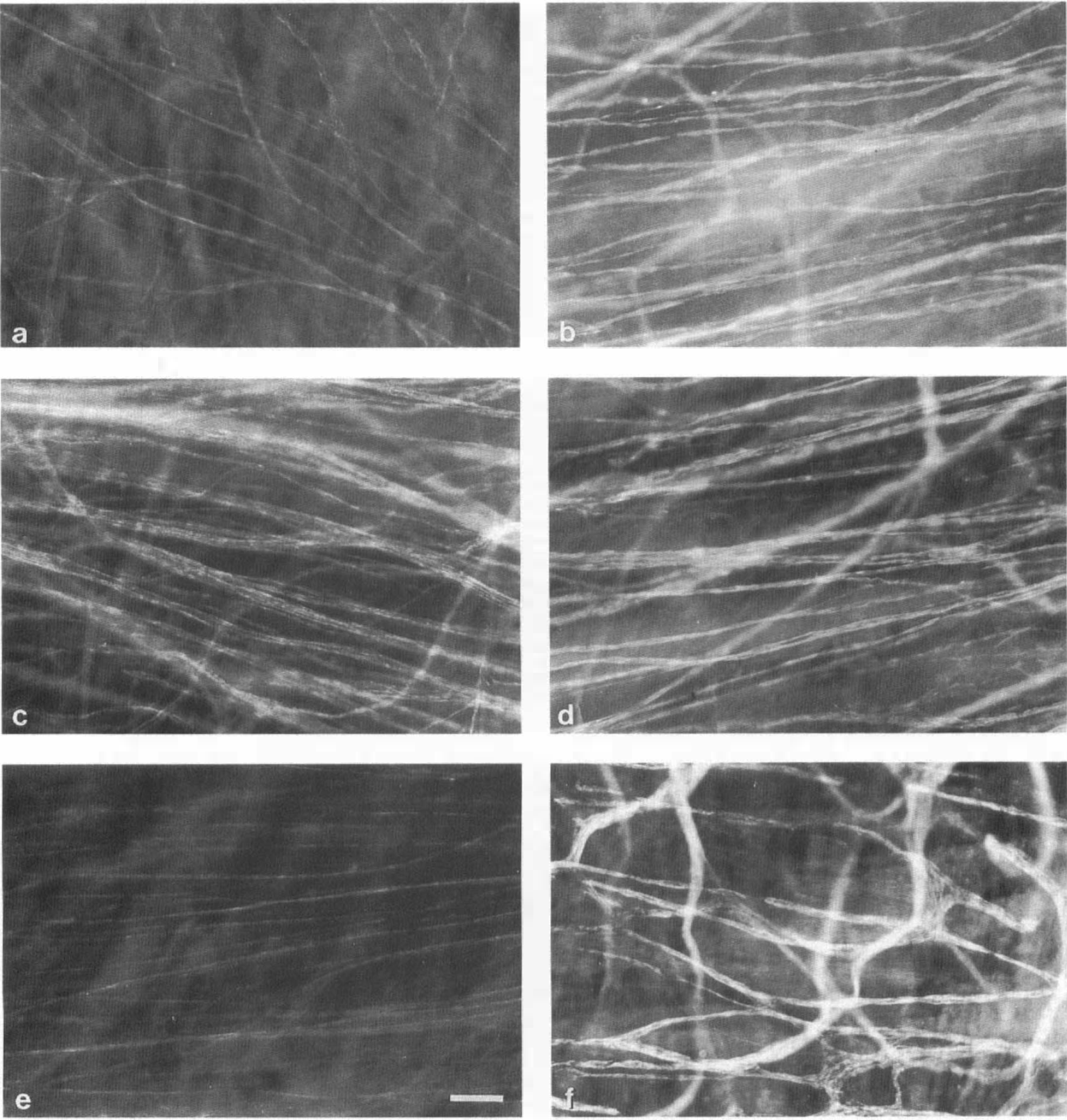
Whole-mount preparations of perivascular nerves of several human cerebral arteries, stained for PGP 9.5. The deep plexuses are transversely oriented and located at the adventitial–medial border,
Confocal microscopy showed two principal layers of nerves within the adventitia of the PCom and MCA (Fig. 3). In some vessels, superficial nerve bundles were relatively sparse and connected via a few branches with a dense, deep terminal plexus.
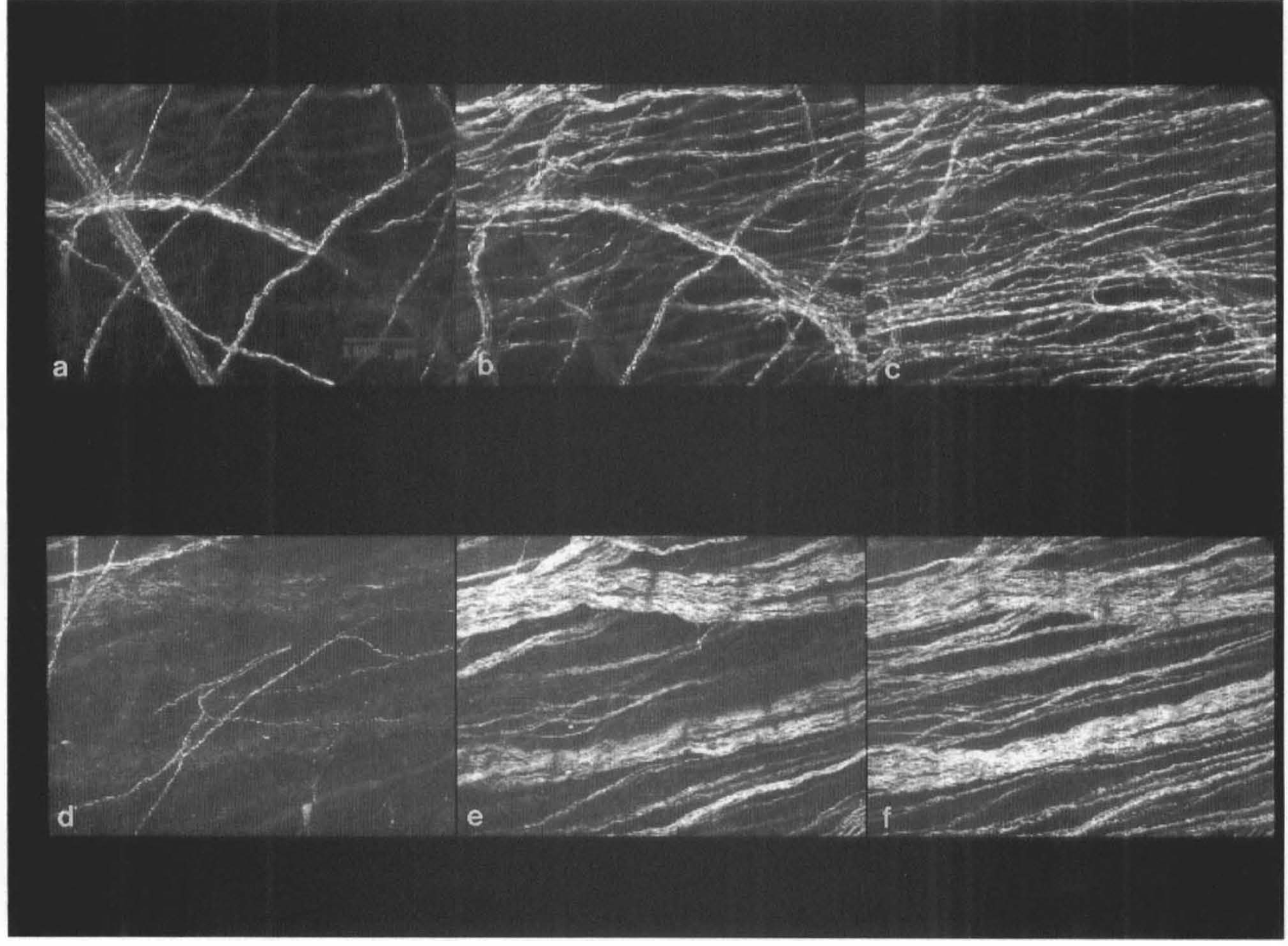
Confocal videomicrographs of MCA (
Staining results of the fetal specimens are shown in Fig. 4. Two plexuses were clearly visible: an extremely regular meshwork-like superficial plexus and a transversely oriented deep plexus of fine fibers and of low density, containing few varicosities.
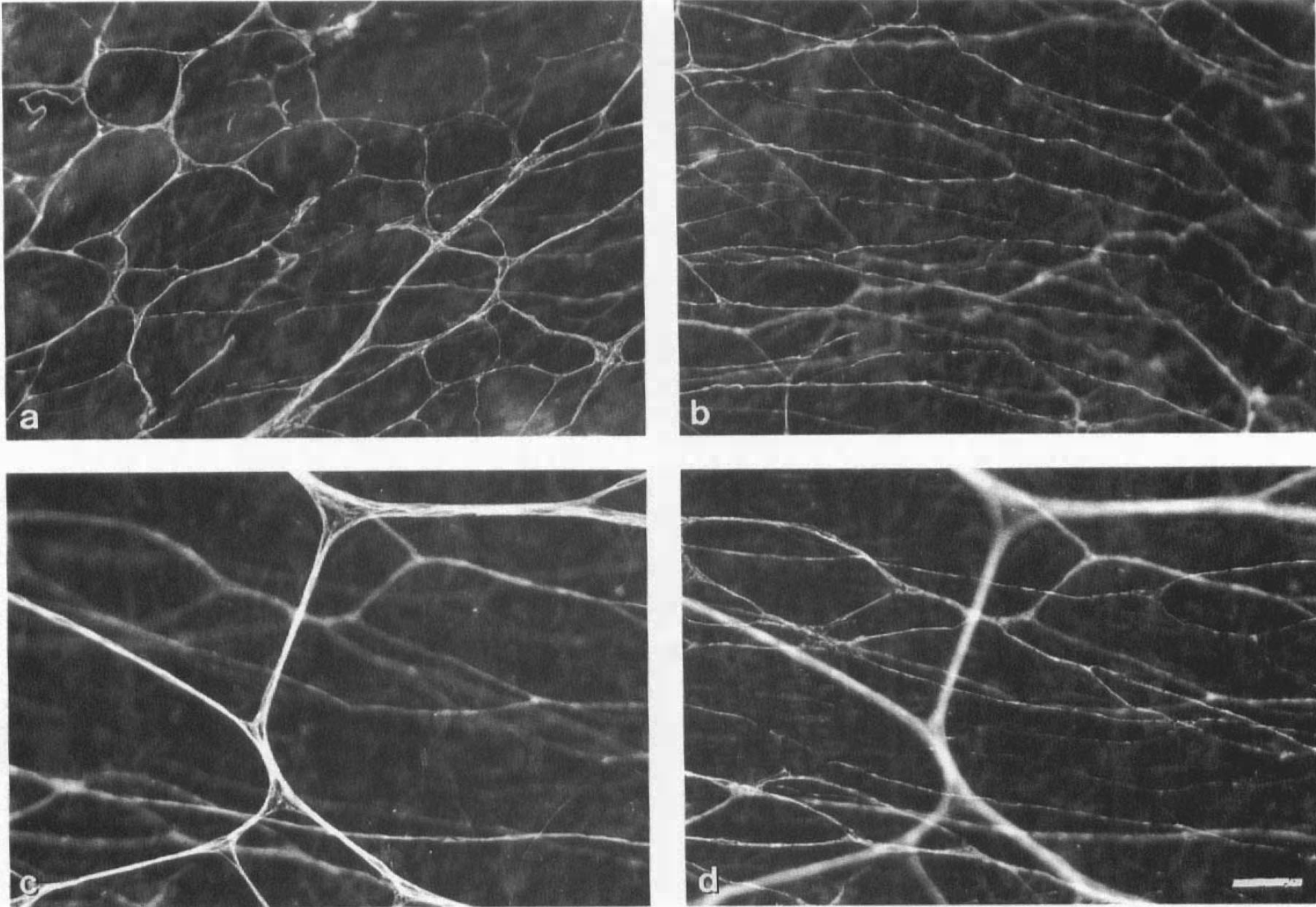
Whole-mount preparations of perivascular nerves of several human cerebral arteries from the fetal specimen, stained for protein gene product 9.5.
Image analysis. Middle aged specimens
The results of image analysis measurements from the few vessels that contained atherosclerotic plaques were well within the range of data from vessels without plaques. Hence, we included the sclerotic vessels in the statistical analysis. Left–right differences in area percent and ID/millimeter were tested by
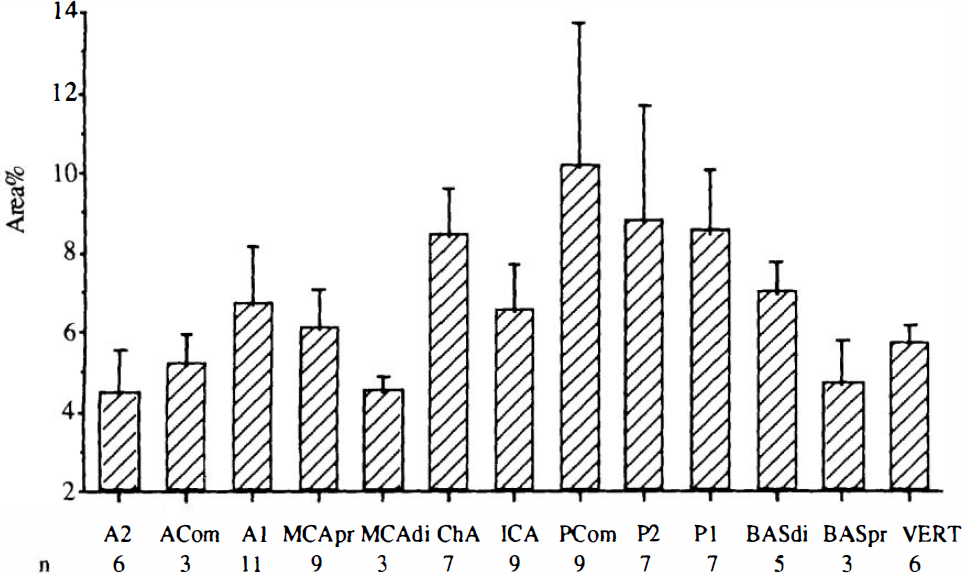
Mean values and standard deviation of nerve densities expressed as area percent for the arteries investigated as whole-mount preparations. Data from left and right arteries have been averaged. See Fig. 1 legend for abbreviations. Area percent was highest in PCom, P2, P1, and ChA. The n number is indicated below the arteries.
The material investigated does not allow reliable comparison between the sexes, but considering the results of the individual measurements, there is no indication of significant differences in nerve density between male and female specimens.
Correlation coefficients between vessel diameter and both parameters for nerve density (area percent and ID/millimeter) were computed for the individual arteries. They varied considerably and were generally low. None of them was significant (i.e., p < 0.05), showing a lack of correlation between vessel diameter and nerve density.
The results from the angle measurements demonstrated that most nerve bundles were in the range from −10 to 10°, in agreement with a transversely oriented nerve plexus. There was however, some variation in the orientation of the nerve bundles, but no major differences between vessels.
Fetal specimen
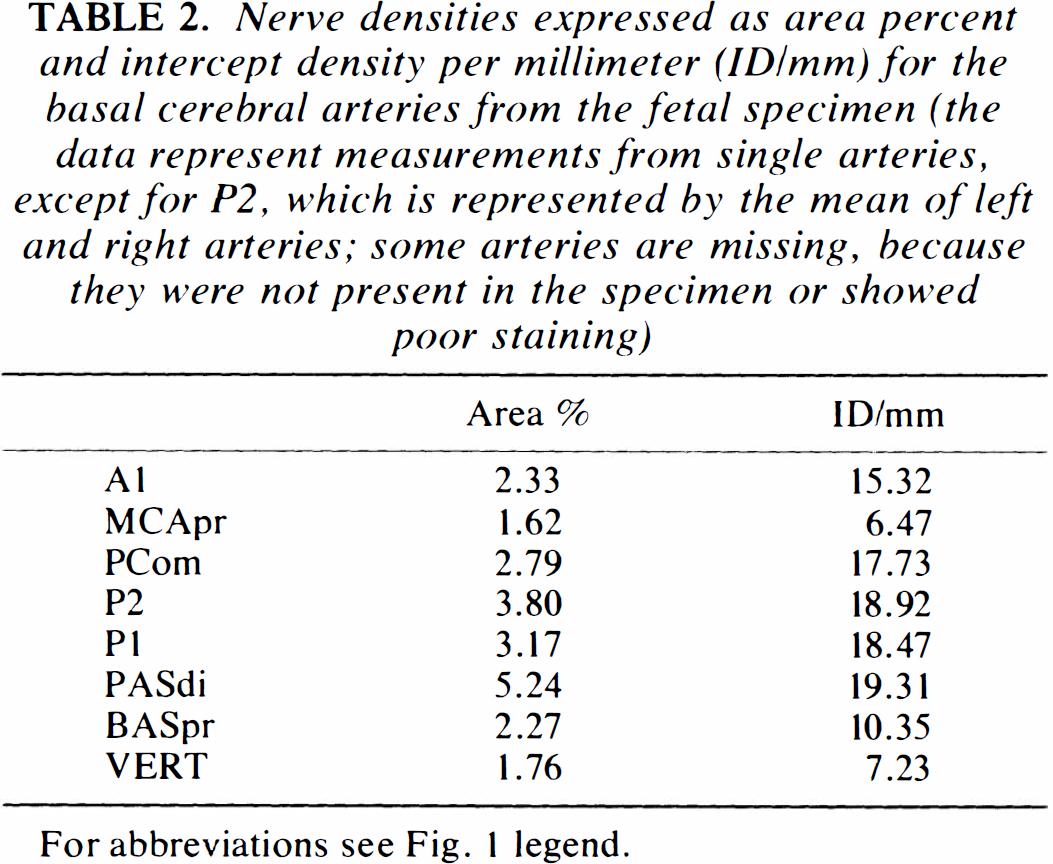
The data obtained from the fetal specimen are listed in Table 2. The values were well below those from the middle-aged group.
Nerve densities expressed as area percent and intercept density per millimeter (ID/mm) for the basal cerebral arteries from the fetal specimen (the data represent measurements from single arteries, except for P2, which is represented by the mean of left and right arteries; some arteries are missing, because they were not present in the specimen or showed poor staining)
For abbreviations see Fig. 1 legend.
Sections
Visual assessment
Figure 6 shows representative staining results. Nerve staining by each antibody was observed in all arteries investigated. The PGP 9.5 staining was intense and showed nerves throughout the adventitia. At the adventitial–medial border the nerves were often oriented in a transverse direction. In the PCom these nerves were of a large caliber. Cross sections through nerves outside the adventitia indicated a longitudinal arrangement. The TH staining was less intense than PGP 9.5. However, nerves could again be seen throughout the adventitia, including at the adventitial–medial border. NF staining by R39 was bright and similarly distributed. In contrast, nerve fibers stained by RT97 were located only in paravascular nerve bundles.
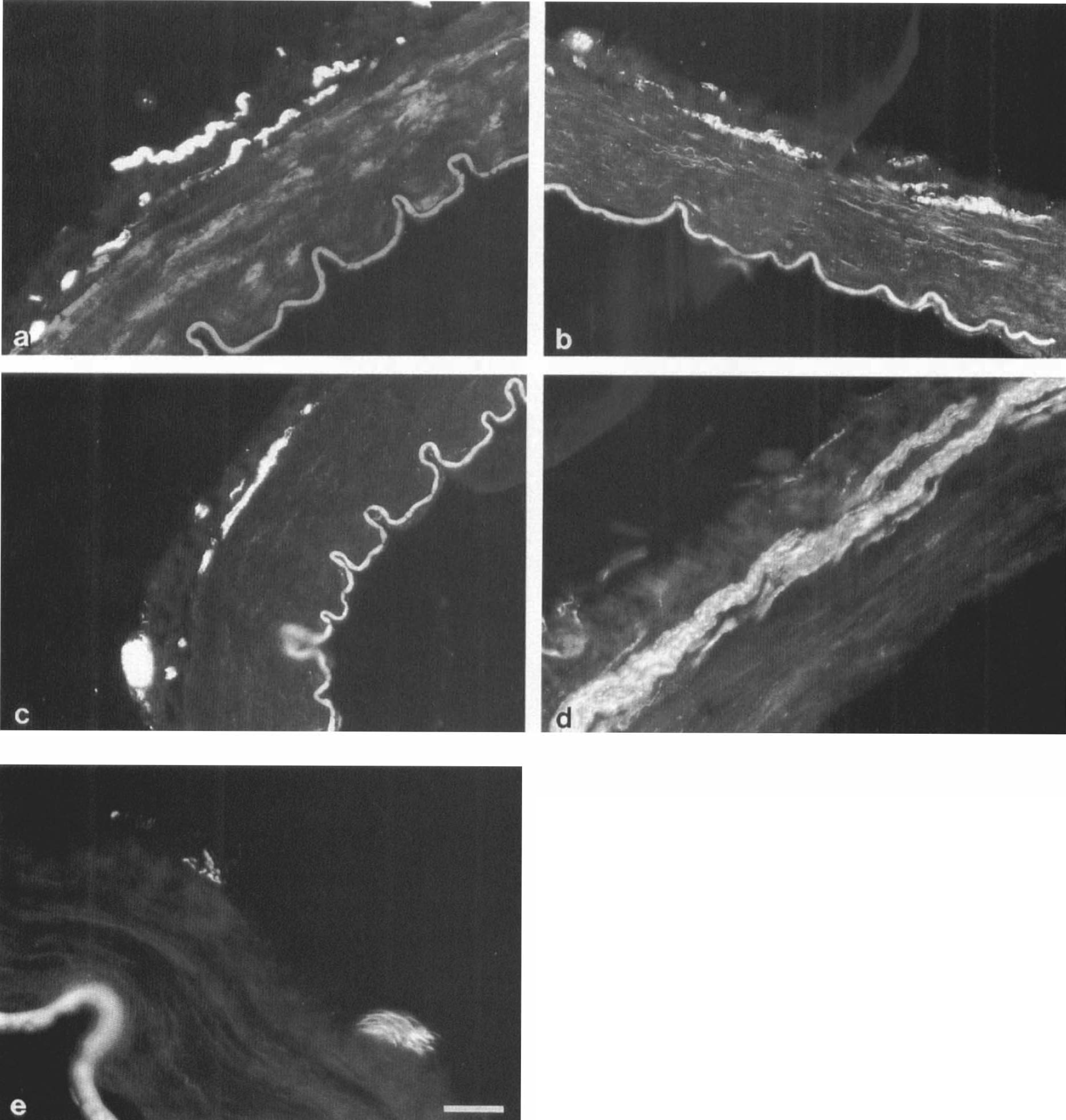
Transverse cryostat sections of several cerebral arteries
Image analysis
The number of sections quantified was too low to perform statistics. However, the results confirm the measurements of high nerve densities in the PCom and P2 segment made on whole-mounts of these specimens. Furthermore, results suggest that TH- and R39-positive nerves constitute subpopulations of PGP 9.5 stained nerve plexuses.
TEM
The sections of PCom and P2 segments studied by TEM revealed a remarkable density of terminal nerve fibers. Close neuromuscular relationships (200–250 nm) and the presence of several varicosities containing neuronal vesicles suggested an important functional role for these nerves (Fig. 7). Some small granular vesicles were observed, indicating the presence of aminergic neurotransmitters in some varicosities.
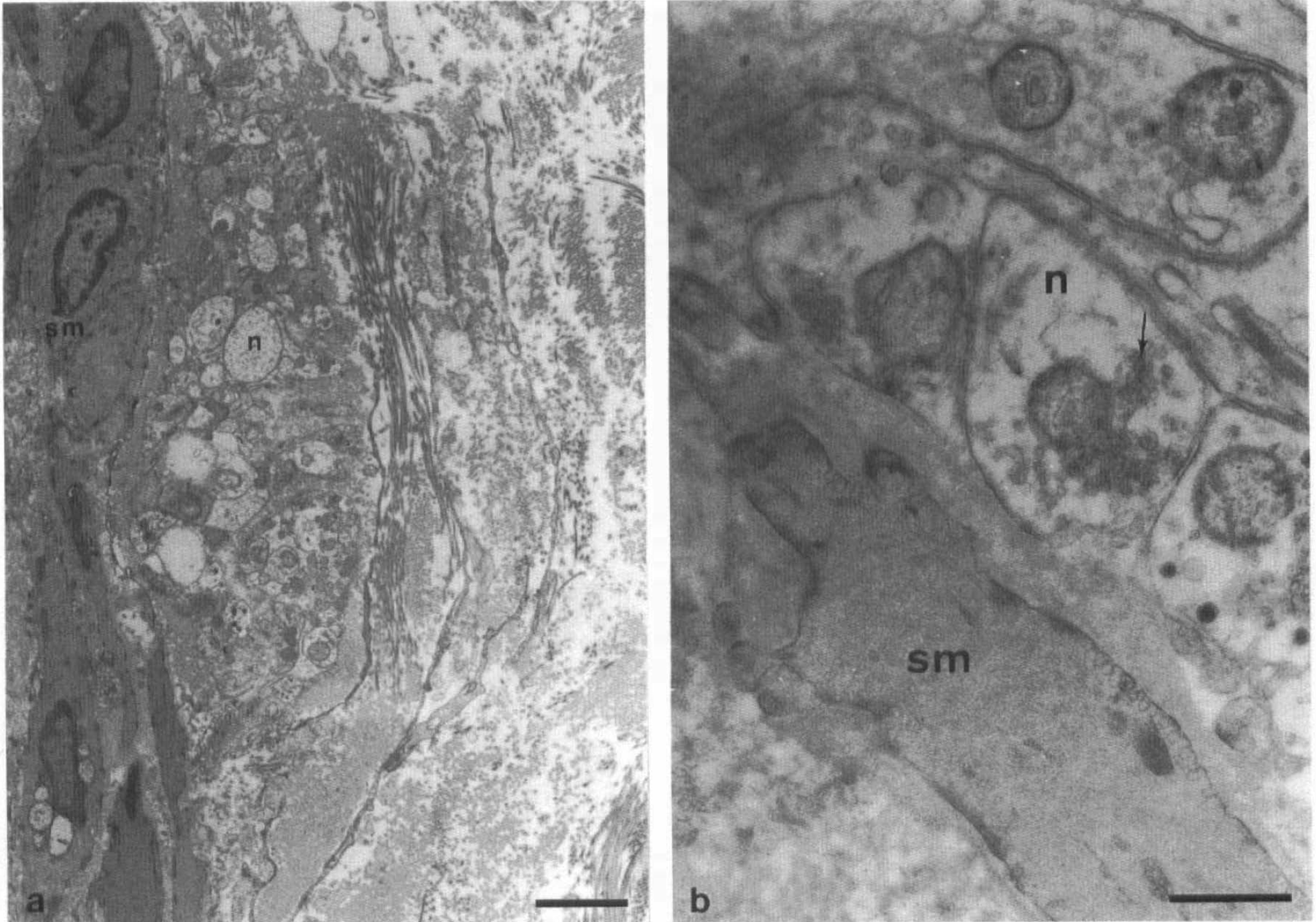
Transmission electron microscopy of posterior communicating artery
DISCUSSION
Methodological considerations
In establishing a reproducible immunohistochemical staining procedure that would allow measurement of nerve densities in
It seems that the most important factor for successful staining is enhancement of the penetration of antibodies. The combination of freezing and thawing, trypsin treatment, and manually performed delamination enabled antibodies to reach the deep nerve plexus. The superficial delamination of the adventitia, and the removal of the highly autofluorescent media and intima proved to be very important to obtain a measurable preparation because of the resulting decrease in background staining. Comparison of trypsin- and nontrypsin-treated segments of the same specimen revealed that trypsin treatment improved the penetration, without impairment of nerve staining. However, nerve plexuses in some specimens proved not to be resistant to trypsin treatment. Omitting the trypsin step in processing these specimens did not seem to influence the results of the measurements, as is shown by comparing the trypsin- and nontrypsin-treated groups. Morphologic changes have been described in aging arteries (Ravens, 1978; Toda et al., 1980). We speculate that these changes, gradually appearing during middle age in varying extents, may explain why some specimens needed trypsin treatment, whereas others appeared to stain well without the treatment.
The staining on
The interpretation of staining by RT97 is difficult. RT97 recognizes the phosphorylated form of the high molecular mass neurofilament subunit (NF-H). Phosphorylation is believed to be a stepwise process, and it has been suggested that RT97 recognizes a high degree of NF-H phosphorylation (Nixon et al., 1994). The same authors demonstrated that in the optic nerve in mice the occurrence of RT97 immunoreactivity coincides with expansion of axon caliber and the site where myelination begins. Considering our results, it is therefore possible, but not certain, that RT97 immunoreactivity, which was only found in paravascular nerve bundles, demonstrates large myelinated axons. Otherwise, the only conclusion is that the phosphorylation state of NF-H is different between those nerve fibers stained by RT97 and those that were unstained, with possibly a high degree of phosphorylation in the RT97-positive fibers.
Morphology of the nerve plexuses
The results on whole-mounts (including confocal scanning laser microscopy) and sections suggest the following arrangement of nerves around and in the walls of human cerebral arteries: (a) paravascular nerve bundles of passage, longitudinally arranged outside the tunica adventitia; (b) a meshwork-like superficial perivascular plexus in the outer or middle zone of the adventitia; and (c) a deep, intrinsic perivascular plexus, located at the adventitial–medial border and transversely oriented. This arrangement is to a large extent in accordance with the arrangements described by several authors for animals, although some differences occur. Most authors mention a two-layered arrangement: periadventitial nerves and an adventitial meshwork for adrenergic nerves in rat (Iwayama et al., 1970); large longitudinal nerves in the outer zone of the adventitia and perivascular nerves reaching the adventitia-medial border for AChE-positive nerves in monkey (Denn and Stone, 1976); a superficial meshwork-like or longitudinally arranged plexus and a deep circular plexus for AChE-positive nerves in dog (Borodulya and Pletchkova, 1976); and superficial longitudinal nerves and a deep circular plexus for adrenergic and AChE-positive nerves in rat (Kobayashi et al., 1981a). The most complete descriptions were by Nakakita et al. (1983). These authors described a three-layered configuration in the rat that closely resembles the configuration observed by us in humans, except for the fact that they ascribed the outermost layer of longitudinal nerves to the outermost zone of the adventitia.
During the development of an AChE staining method for identifying peripheral connections of cerebrovascular nerves (Bleys et al., 1994), we also applied this technique to whole-mount preparations of adult human cerebral arteries. In the peripheral nervous system, AChE is present not only in cholinergic but in most neurons, and can therefore be considered as a general neural marker. The preparations confirmed the three-layered configuration of nerves as currently described for PGP 9.5 staining (Fig. 8).
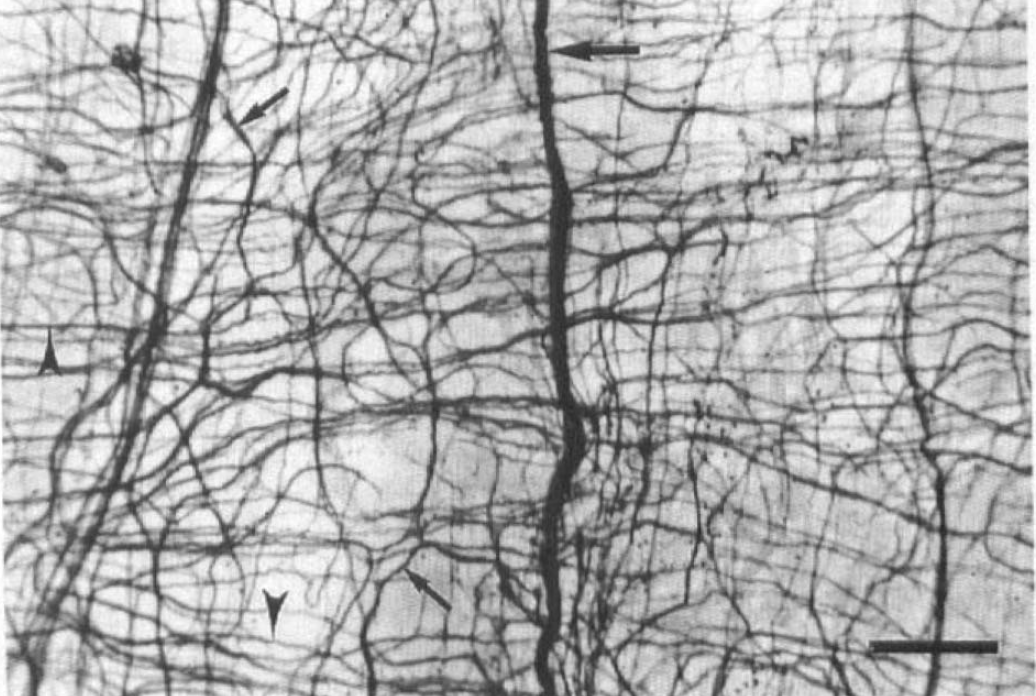
Whole-mount preparation of human posterior communicating artery (age 96 years) stained for acetylcholinesterase demonstrates three layers of nerves around the artery: large paravascular nerve bundles (large arrow), a meshwork-like superficial plexus (small arrows), and a transversely oriented deep plexus (arrowheads), consisting of relatively thick bundles of nerve fibers. (Bar = 250 μm.)
Care has to be taken with the interpretation of our results on the fetal arteries, because they represent a single specimen at 32 weeks' gestational age. A meshwork-like superficial plexus and a transversely oriented deep plexus were observed. Edvinsson et al. (1976), however, only described a two-dimensional meshwork-like plexus in the adventitia, superimposed on the muscular media and large paravascular nerves for adrenergic and AChE-positive fibers in human fetuses at 19–23 weeks' gestational age. The plexus described by Edvinsson et al. (1976) corresponds to the superficial plexus that we found in the fetal specimen. In contrast, a recent developmental study of perivascular nerves of the basilar artery in human fetuses (12–28 weeks' gestational age) recognized circular and longitudinal fibers, with full development at 20 weeks for adrenergic and AChE-positive fibers and at 24 weeks for neuropeptide Y- and vasoactive intestinal polypeptide-positive fibers (Kawamura and Takabayashi, 1994). The method of quantification used by these authors, however, did not distinguish between circular and longitudinal fibers, nor were specimens older than 28 weeks' gestational age studied. Low densities of deep perivascular nerves in comparison with middle-aged specimens, as observed by us, are in line with the descriptions made for developing perivascular nerves in the rabbit (Cowen et al., 1982) and guinea-pig (Dhall et al., 1986), which demonstrated peri- and postnatal development for adrenergic plexuses. Because Kobayashi et al. (1981b) demonstrated that in mice the development of circular nerve fibers occurs even after birth, it seems likely, however, that the development of cerebrovascular nerves in humans starts in an earlier stage of gestation than in laboratory animals. The presence of a deep, transversely oriented nerve plexus with some varicosities in the human fetus suggests that cerebrovascular innervation of humans functions in the prenatal stage and justifies a more detailed investigation of development of human cerebrovascular nerves.
Functional considerations
The main findings from the quantitative data are that human basal cerebral arteries display differences in topographical distribution of the intrinsic nerve plexuses, with the highest nerve densities found in PCom, P2 segment, P1 segment, and ChA, and that there is no clear relation between nerve density and arterial diameter. Hence, it appears that nerve density of arteries at the base of the human brain is determined by locality rather than by vessel diameter. This is best illustrated by the high nerve densities that were consistently found in the PCom and the P1 segment, whereas the diameters of these arteries vary and tend to follow a reciprocal relationship (Hillen, 1986).
To our knowledge, the only quantitative studies based on the morphology of perivascular nerves in the human basal cerebral arteries were performed on fetuses at 12–28 weeks' gestational age (Kawamura et al., 1991). Highest densities of various nerve populations, stained for noradrenaline, AChE, neuropeptide Y, and vasoactive intestinal polypeptide, were found in the anterior cerebral artery, followed by the MCA and basilar artery. However, superficial and deep nerves were not distinguished. Because, in addition, no mention was made of data from the PCom and posterior cerebral arteries, it is not possible to make a comparison between our results and those reported.
Edvinsson et al. (1987c) found relatively high concentrations of neuropeptides by radioimmunoassay in the walls of aged human posterior cerebral artery and PCom. However, because the peptide levels were determined by radioimmunoassay, they do not select the functionally important deep levels of nerve plexuses. Therefore, the peptide levels do not contradict our results, nor do they contribute to the information of deep nerve plexuses.
Our results do not demonstrate significant differences in nerve densities between arteries belonging to the internal carotid system and arteries of the vertebrobasilar system, as is indicated by various authors (Edvinsson, 1975; Kobayashi et al., 1981; Itakura et al., 1984; Dhital et al., 1988). This discrepancy can be due to differences in species, although the difference was found in human fetuses as well (Kawamura et al., 1991). There are, however, differences in field of interest and methodology. We only quantified the deep nerve plexuses at the adventitial–medial border using a general neural marker, whereas other authors assessed superficial and deep nerves of subpopulations of nerves, often in a semiquantitative way, using subjective methods to determine nerve densities. A recent study, using PGP 9.5 staining in some adult human cerebral arteries, suggested that the anterior vessels at the base of the brain receive a denser nerve supply than posterior vessels do (Edvinsson et al., 1994). However, quantification of the nerve plexuses was not performed, and because measures to improve the penetration of the antibodies only included preincubation with Triton X-100, it is not clear whether all deep nerve fibers were visualized.
The high nerve densities observed at the adventitial–medial border in the PCom and the P2 segment suggest a stronger neuronal influence on this part of the cerebral circulation than hitherto reported. We have to be careful in speculating about the functional implications of the results for the following reasons. First, our quantitative data refer to the overall nerve population; we have no reliable data on subpopulations of nerves, which are necessary to gain more insight into function. Second, we are not sure whether high nerve densities indicate a functionally more important vascular innervation, although it has been demonstrated that the innervation is considerably richer in those areas of the cerebral arterial system that have been ascribed a role in the regulation of CBF, such as bifurcation sphincters in the pial arterial system (Baramidze et al., 1981) and the intracranial segment of the ICA (Borodulya and Pletchkova, 1973). With these reservations, we propose the following hypotheses.
Considering the PCom, this artery is important for the hemodynamic balance between the internal carotid and vertebrobasilar circulations. This can be interpreted in two ways: The artery can act as a regulator or as a sensor between the two systems. A third possibility is that it combines both functions. The sections demonstrate that part of the nerves are aminergic, and thus possibly of a sympathetic origin. The close neuromuscular relationship and the presence of vesicles, including small granular ones, as observed by electronmicroscopy, is highly suggestive of motor function. Data by Edvinsson et al. (1994) suggest that the majority of the perivascular nerves in human cerebral arteries are sympathetic.
The high nerve density in the P2 segment, in contrast to other major efferent arteries such as the A2 segment and the MCA, could reflect greater fluctuations in the metabolic need of the vascular territory of this segment (e.g., visual cortex) in comparison to other vascular territories of the brain. This would implicate a regulatory role for the autonomic nerve supply to this major cerebral artery as well.
In the ChA not only a dense deep nerve plexus was observed, but also many superficial, longitudinally, oriented nerve bundles, probably on their way to more distal parts of the artery, were seen. From the whole-mounts it was difficult to assess their position in the adventitia. Although in other arteries superficial nerves were also observed, their number and thickness in the ChA was striking. They may reflect the neuronal role in the production of cerebrospinal fluid (Edvinsson, 1982).
In conclusion, human basal cerebral arteries display a topographical distribution of deep perivascular nerves, irrespective of arterial diameter. The high nerve densities and dense, vesicle-containing varicosities in the PCom and the P2 segment suggest involvement in the regulation of the cerebral circulation. The mechanisms by which these perivascular nerves influence circulation remain unclear.
Footnotes
Acknowledgment:
This project was supported by a travel grant from the Netherlands Organization for Scientific Research (NWO), by the foundation “De Drie Lichten” in the Netherlands, and by a grant to T.C. and B.H. from the British Heart Foundation. We thank Prof. B.H. Anderton (Department of Neuroscience, Institute of Psychiatry, London) for his useful comments on the manuscript and for the supply of RT97 antibody. We thank Dr. D. Dahl (Department of Spinal Cord Injury Research, Harvard Medical School, Boston, MA, U.S.A.) for the supply of R39 antibody. We thank the following for generous assistance with the collection of cerebral arteries: Mr. C. Marriage (Department of Histopathology, Royal Free Hospital School of Medicine, London), Dr. J. Geddes (Institute of Pathology, London Hospital), Dr. R. Ravid (Coordinator Netherlands Brain Bank, Amsterdam) and Mr. W. J. A. van Wolferen (Department of Functional Anatomy, Utrecht University). We thank Mr. C. Thrasivoulou (Department of Anatomy and Developmental Biology, Royal Free Hospital School of Medicine, London) for expert technical assistance and Dr. P. Westers (Centre for Biostatistics, Utrecht University) for statistical assistance.
