Abstract
These investigations characterised the cerebrovascular effects of an endothelin ETA-receptor antagonist PD156707 in normal and ischaemic cat brain. A dose of PD156707 that inhibited the effects of exogenous endothelin-1 was established in nonischaemic cerebral resistance arterioles. Perivascular microapplication of the endothelin–receptor antagonist PD156707 (0.03–3 μM) had a minimal effect on nonischaemic pial resistance arterioles. The perivascular coapplication of PD156707 and ET-1 (10 nM) effected a dose-dependent attenuation of the ET-1 vasoconstrictive response (IC50 = 0.1 μM). Intravenous administration of PD156707 (3 μmol/kg bolus + 5 μmol/kg/h infusion) attenuated the vasoconstriction elicited by perivascular ET-1 (10 nM) in normal pial arterioles (ET-1 vasoconstriction: −37 ± 13% from preinjection baseline; after intravenous PD156707: 6 ± 10% from preinjection baseline). In the focal ischaemia studies, cerebral perfusion was measured in the suprasylvian and ectosylvian gyri (by laser Doppler flowmetry). Occlusion of the middle cerebral artery reduced cerebral perfusion in the suprasylvian and ectosylvian gyri by ∼50%. Intravenous administration of PD156707 (3 μmol/kg bolus + 5 μmol/kg/h infusion), initiated 30 min after middle cerebral artery occlusion, effected a progressive increase in cerebral perfusion up to preocclusion baseline levels, whereas cerebral perfusion in vehicle-treated animals did not vary from its postocclusion level. In these animals, the intravenous administration of PD156707 reduced the hemispheric volume of ischaemic damage by 45% (vehicle: 2,376 ± 1,107 mm3; PD156707: 1,307 ± 548 mm3; p < 0.05). Our investigations indicate that endothelin receptor antagonism may be a new therapeutic strategy for the amelioration of focal ischaemic damage.
Pharmacotherapy of cerebral ischaemia has two general approaches. First, pharmacologic interventions are directed at various points in the cascade of neurochemical events that lead to neuronal damage [e.g., N-methyl-D-aspartate (NMDA) and α-amino-3-hydroxy-5-methylisoxazole propionate (AMPA)-receptor antagonists, glutamate release inhibitors, nitric oxide synthase inhibitors, free radical scavengers and the inhibition of the effects of the cytokines (Rothwell and Relton, 1993; Gill, 1994; Muir and Lees, 1994; Phillis, 1994). Second, pharmacologic interventions are directed at removing the primary intraluminal obstruction [e.g., tissue plasminogen activator (TPA)] or at reducing the vasoconstrictor influences on collateral vessels to the tissue at risk [e.g., L-type calcium channel antagonists (Kazda et al., 1982; Fisher and Grotta, 1993; Overgaard, 1994)].
The endothelins are a family of 21 amino acid peptides and are potent constrictors of vascular smooth muscle. The vascular effects of the endothelins are mediated by two receptor subtypes, endothelin-A (ETA), which are vasoconstrictor, and endothelin-B (ETB), which are vasodilator (Masaki et al., 1994). The application of exogenous ET-1 results in marked constrictions of cerebral vessels in vivo and in vitro (Robinson and McCulloch, 1990; Salom et al., 1993; Feger et al., 1994). Topical or intraparenchymal application of exogenous ET-1 on cerebral blood vessels is associated with sufficient reductions in the CBF to cause irreversible ischaemic brain damage (Robinson et al., 1990; Fuxe et al., 1992; Macrae et al., 1993; Sharkey et al., 1993). Increases in endogenous endothelin levels have been reported after focal cerebral ischaemia in tissue and plasma (Ziv et al., 1992; Viossat et al., 1993; Barone et al., 1994). Our own investigations demonstrated an increase in endothelin-mediated vascular tone in cerebral arterioles in the penumbra after a focal cerebral ischaemic challenge (Patel et al., 1995a).
The recent availability of nonpeptide low-molecular-weight endothelin antagonists, such as Bosentan, that penetrate the blood–brain barrier have enhanced the interest in the physiologic and pathophysiologic role of the endothelins in the cerebral circulation (Clozel et al., 1994; Patel et al., 1994). Recently a new group of nonpeptide endothelin antagonists (butenolides) have been developed, with PD156707 the most potent and selective for the ETA receptor (Doherty et al., 1995).
The aims of these investigations were threefold. First, the cerebrovascular effects of the endothelin–receptor antagonist PD156707 were characterised in the nonischaemic brain to identify a dose of the antagonist, administered intravenously, that would inhibit the vasoconstrictor effect of exogenous ET-1. Second, the ability of the validated dose of PD156707 to alter cerebral perfusion (measured by laser Doppler flowmetry) in the ischaemic penumbra was assessed after permanent middle cerebral artery (MCA) occlusion in the cat. Third, the ability of the validated dose of PD156707 to reduce the volume of ischaemic damage after permanent focal cerebral ischaemia in the cat was assessed in the animals in which cerebral perfusion had been measured.
MATERIALS AND METHODS
General surgical preparation
The experiments were performed on 14 adult female cats weighing 2.5 to 3 kg. Anaesthesia was induced by using alphaxolone/alphadolone administered into the radial vein. The animals were intubated, and positive pressure ventilation with N2O/O2 (70%:30%) was initiated. The femoral arteries and veins were cannulated for the monitoring of arterial blood pressure and arterial blood gas status and for the administration of fluids and drugs, respectively. Anaesthesia was maintained by using α-chloralose (60 mg/kg), and further supplements of 1–2 ml of α-chloralose (1% solution) were administered to prevent the return of the corneal reflex. Four cats were used to examine vasomotor reactivity under physiologic conditions, and 10 cats were used in the MCA-occlusion study. Arterial blood samples were taken at regular intervals, and the animals were maintained normocapnic by adjusting the stroke volume; metabolic acidosis was controlled by the administration of sodium bicarbonate (8.4% solution). Core temperature was measured by a rectal thermometer, and the temperature was maintained at 37°C by means of a heating blanket controlled by the rectal thermometer.
Microapplication studies
Four animals were placed in a stereotactic frame. After a midline incision, the scalp was retracted and sutured onto a metal ring to form a well over the calvaria. For the microapplication studies, the temporalis muscle was retracted, and a rectangular craniectomy (2.5 × 1.5 cm) was made over the parietal cortex by using a saline-cooled dental drill. The exposed dura was bathed with mineral oil at 38°C. There was a continuous flow of oil at the rate of ∼1 ml/min. With the aid of a stereomicroscope, the dura was excised and then reflected laterally. Bipolar diathermy was used to control bleeding from the dural vessels. The inspired gas mixture was then altered to O2-supplemented air (∼25% O2).
Measurement of arteriolar calibre. Pial arteriolar calibre was measured by the method of Baez (1966) by using an image splitter linked to a closed-circuit video display system. Individual pial arterioles were viewed through a stereomicroscope, and arteriolar diameter was measured from the degree of shear applied to the image splitter to tangentially appose the two images. The system is calibrated at ×40 and ×70 against threads of known diameter, and this allows the direct measurement of vascular diameter in absolute units (micrometers). The calibres of cerebral arterioles examined were in the range from 46 to 303 μm (138 ± 62 μm). The vessel diameter was typically measured before insertion of micropipettes, before injection, and after injection of substances; the vessel diameter was monitored over a period of 1–2 min. A detailed description of the surgical preparation and measurement technique appeared previously (Robinson and McCulloch, 1990).
Administration of drugs
Sharp glass micropipettes (tip diameter, 10–12 μm) were vacuum filled with either artificial CSF or drugs dissolved in CSF. The composition of the CSF was Na+, 156 mM; K+, 3 mM; Ca2+, 2.5 mM; HCO3–, 12 mM; Cl−, 152 mM; with the pH adjusted to 7.2 by aeration with 5% CO2/95% 02. All solutions were prepared freshly for each study. The filled pipettes were stored under mineral oil until required. The substances were applied by subarachnoid microapplication by using a micromanipulator to position the pipettes in the perivascular space around the blood vessel. Small volumes (5 μl) were delivered by a pressure-ejection system. Individual arterioles were studied on only a single occasion. The perivascular microapplication of CSF elicited a small constrictor response (2.3 ± 11.2% of preinjection baseline). Perivascular microapplication of ET-1 (10 nM) was used as the challenge dose, as this is the median effective concentration (EC50) in this preparation (Robinson and McCulloch, 1990).
To investigate the ability of intravenously administered PD156707 to gain access to cerebrovascular smooth muscle, PD156707 was dissolved in saline and administered initially as an intravenous bolus (3 μmol/kg) and then by continuous infusion (5 μmol/kg/h) for 120 min. Perivascular microapplications of ET-1 were carried out 15–75 min after the intravenous administration of PD156707. Arterial blood samples were taken at 30-min intervals to determine drug plasma levels by HPLC.
Focal ischaemia studies
The MCA was exposed in 10 cats by a transorbital approach by a neurosurgeon as described previously (Chen et al., 1991). Briefly, the contents of the left orbit were exenterated and the optic foramen enlarged to expose the dura mater and the MCA underlying it. The dura was excised, the trunk of the MCA and its lenticulostriate branches were occluded by using bipolar diathermy, and the artery was divided with microscissors.
Laser Doppler flowmetry
Two small craniotomies (3 mm diameter) were drilled overlying the rostral suprasylvian and ectosylvian gyri. Laser Doppler flow probes were placed on the intact dura and attached to the stereotactic frame. Baseline levels of CBF were determined for ≥15 min before the occlusion of the MCA. Thereafter, cerebral perfusion was assessed continuously for the duration of the study without repositioning of the laser Doppler probes, and the data were stored in a computer (Moor Instruments Ltd., Axminster, U.K.). In two animals, the suprasylvian laser Doppler probes yielded unstable flow values, particularly after MCA occlusion, and the probes were removed in both animals and data not included in any of the analyses.
Volume of ischaemic damage
Six hours after the occlusion of the MCA, the animals were killed by transcardiac perfusion fixation with FAM (40% formaldehyde, glacial acetic acid, and methanol in a ratio of 1:1:8), as described previously (Bullock et al., 1994). Briefly, the animals were placed in a supine position and given heparin. A thoracotomy was performed, and a cannula was introduced into the ascending aorta via the left ventricle. Physiologic saline was infused, at a pressure equivalent to mean arterial pressure of the animals, for 20 s. This was followed immediately by 1.5 L of FAM fixative at the same pressure. After perfusion, the animals were decapitated, and the heads were stored in the same fixative at 4°C for ≥12 h. The brains were removed and, after marking the left side of the forebrain with India ink, the hindbrain was detached by a cut through the midbrain, and the cerebral hemispheres were cut into five equally spaced coronal slices. These slices were embedded in paraffin wax, and sections 7- to 8-μm thick were cut at 200-μm intervals throughout the specimens (∼150–200 sections from each forebrain). The sections were stained by haematoxylin and eosin and by a method combining cresyl-violet and Luxol fast blue. Those sections that corresponded most closely to 16 stereotactically predetermined coronal planes of the cat brain from the atlas of Reinoso-Suarez (1961) were examined by conventional light microscopy by one of us (D.I.G.) without prior knowledge of the animal's history. Any abnormalities were charted on scale diagrams, and the area of ischaemic damage in the cerebral hemisphere, cerebral cortex, and caudate nucleus was determined by using a computer-based image-analysis system (Quantimet 970, Cambridge Instruments, Cambridge, U.K.). The volumes of ischaemic damage were calculated from the areas of damage at the different coronal planes and their anteroposterior coordinates.
In the MCA-occlusion studies, PD156707 (3 μmol/kg) was dissolved in isotonic saline and administered in five animals as a slow intravenous bolus injection 30 min after occlusion of the MCA. Thereafter, PD156707 was administered as a continuous intravenous infusion (5 μmol/kg/h) for the succeeding 330 min. Five additional animals received saline. Samples of arterial blood were taken before administration of the drug, within 5 min of the bolus, and at 60 min intervals thereafter for analysis of drug-plasma levels.
Statistical analysis
Data are presented as mean ± SD. Data from the microapplication studies were analysed by using analysis of variance (ANOVA) followed by Student's t test with Bonferroni correction for multiple comparisons. Cardiovascular and respiratory parameters were analysed using two-tailed Student's t test. The laser Doppler flowmetry and pial arteriolar calibre data from the MCA-occlusion studies was analysed by two-way ANOVA with covariance. The quantitative histopathology data were analysed by one-tailed Student's t test because an a priori decision was made to examine the ability of the drug to reduce the volume of hemispheric ischaemic damage.
RESULTS
Microapplication studies
The mean arterial blood pressure at the outset of the study was 92 ± 9 mm Hg and did not vary significantly from this value. The intravenous injection of PD156707 had minimal effect on mean arterial blood pressure. The arterial Pco2 was maintained between 26 and 30 mm Hg, and arterial Po2 was maintained at >100 mm Hg.
The subarachnoid perivascular microapplication of CSF minimally altered pial arteriolar calibre. PD156707 (0.03–3 μM) had minimal effect on pial arteriolar calibre (Fig. 1). The perivascular microapplication of ET-1 (10 μM) produced a marked constriction of the pial arterioles examined (36.7 ± 12.6% change from preinjection baseline). The coapplication of PD156707 (0.03–3 μM) with ET-1 (10 μM) effected a dose-dependent attenuation of the ET-1-induced vasoconstriction (IC50 ∼ 0.1 μM; Fig. 2).
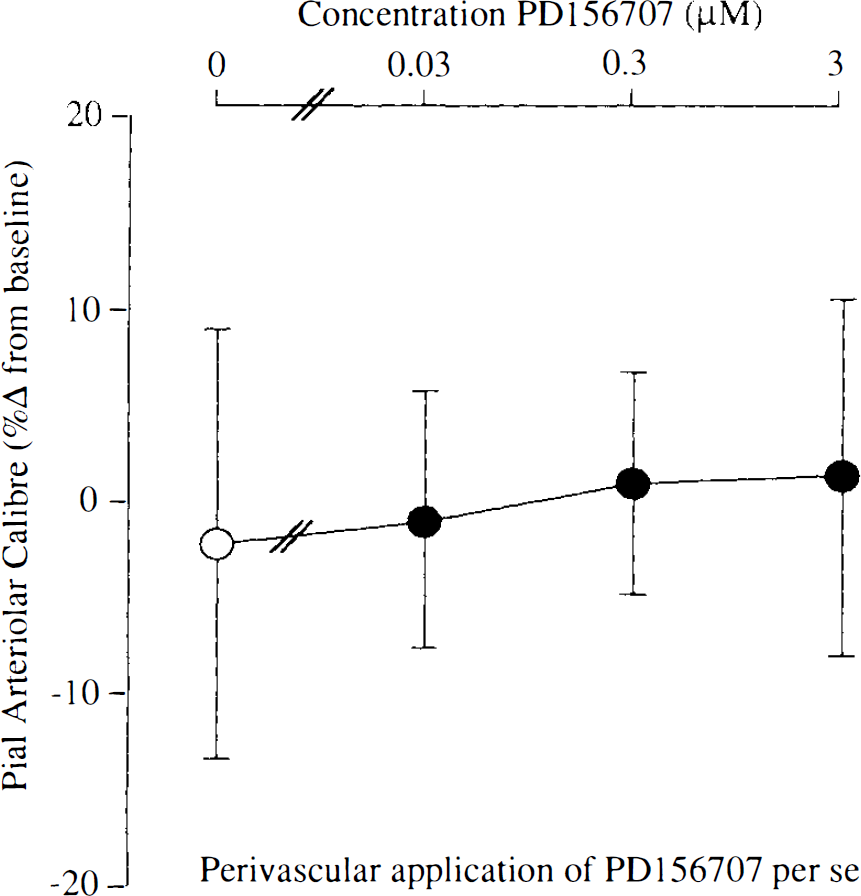
Vasomotor responses of pial arterioles to perivascular microapplication of PD156707 per se. There are no significant alterations in pial arteriolar calibre at any concentration of PD156707. Data are the percentage alteration from preinjection baseline in pial arteriolar calibre. Data are presented as mean ± SD (n, number of arterioles examined = nine or ten for each concentration).
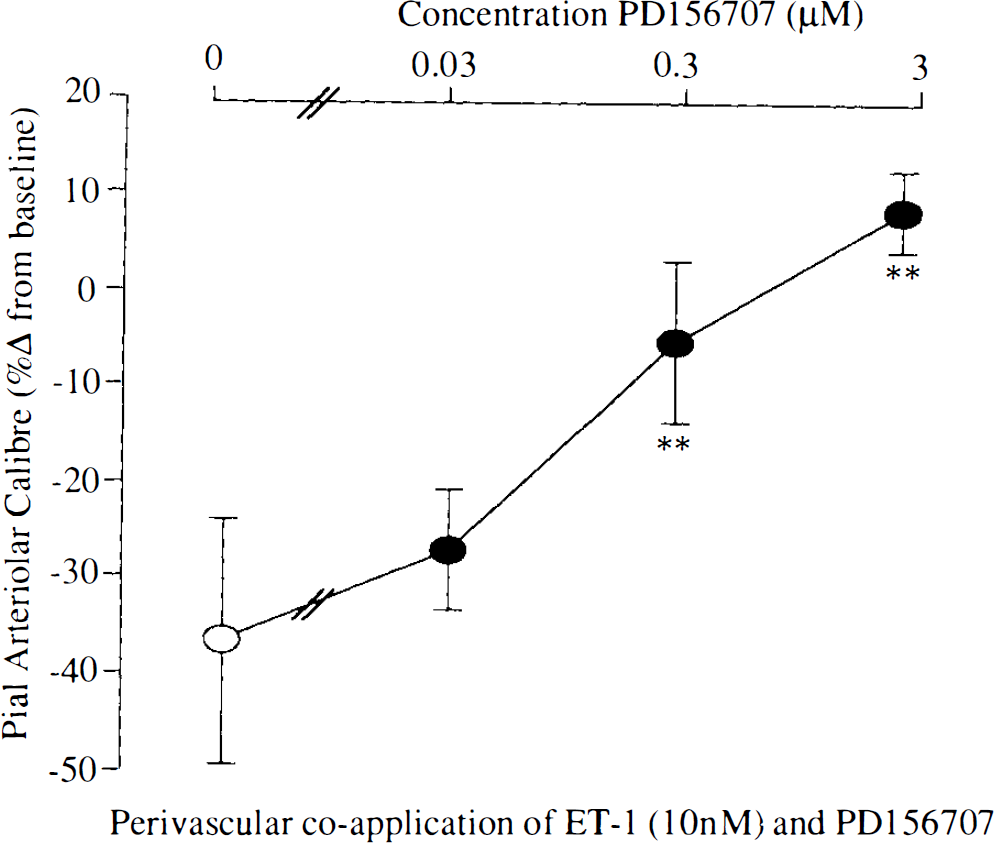
Vasomotor responses of pial arterioles to perivascular coapplication of endothelin-1 (10 nM) and PD156707. PD156707 significantly reduces the constrictions induced by endothelin-1. (**p < 0.01 for the comparison with endothelin alone). Data are the percentage alteration from preinjection baseline in pial arteriolar calibre. Data are presented as mean ± SD (n, number of arterioles examined = nine for each concentration).
The intravenous administration of PD156707 (3 μmol/kg bolus + 5 μmol/kg/h infusion) had minimal effect on mean arterial blood pressure. The perivascular microapplication of ET-1 (10 μM) was carried out 15–75 min after the intravenous administration of PD156707. The vasoconstrictive response induced by the perivascular microapplication of ET-1 (10 μM) was markedly attenuated (Fig. 3).
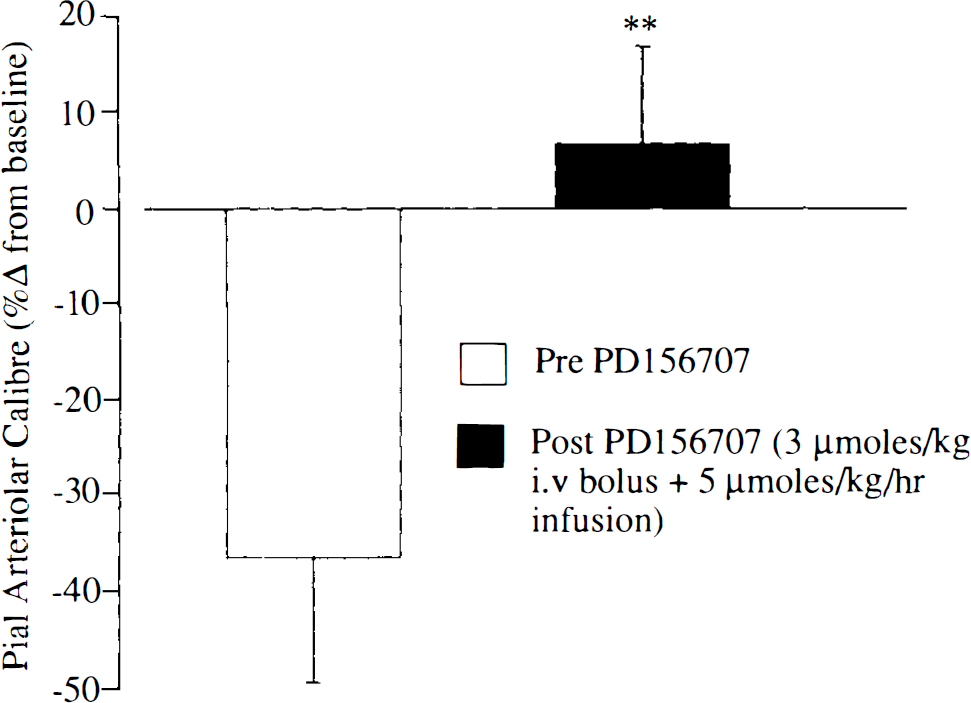
Vasomotor responses of pial arterioles to endothelin-1 (10 nM) after intravenous injection of PD156707 (3 μmol/kg bolus + 5 μmol/kg/h). PD156707 significantly reduces the constrictions induced by endothelin-1 (**p < 0.01 for the comparison with endothelin alone). Data are the percentage alteration from preinjection baseline in pial arteriolar calibre. Data are presented as mean ± SD (n, number of arterioles examined = nine to 13 in each group).
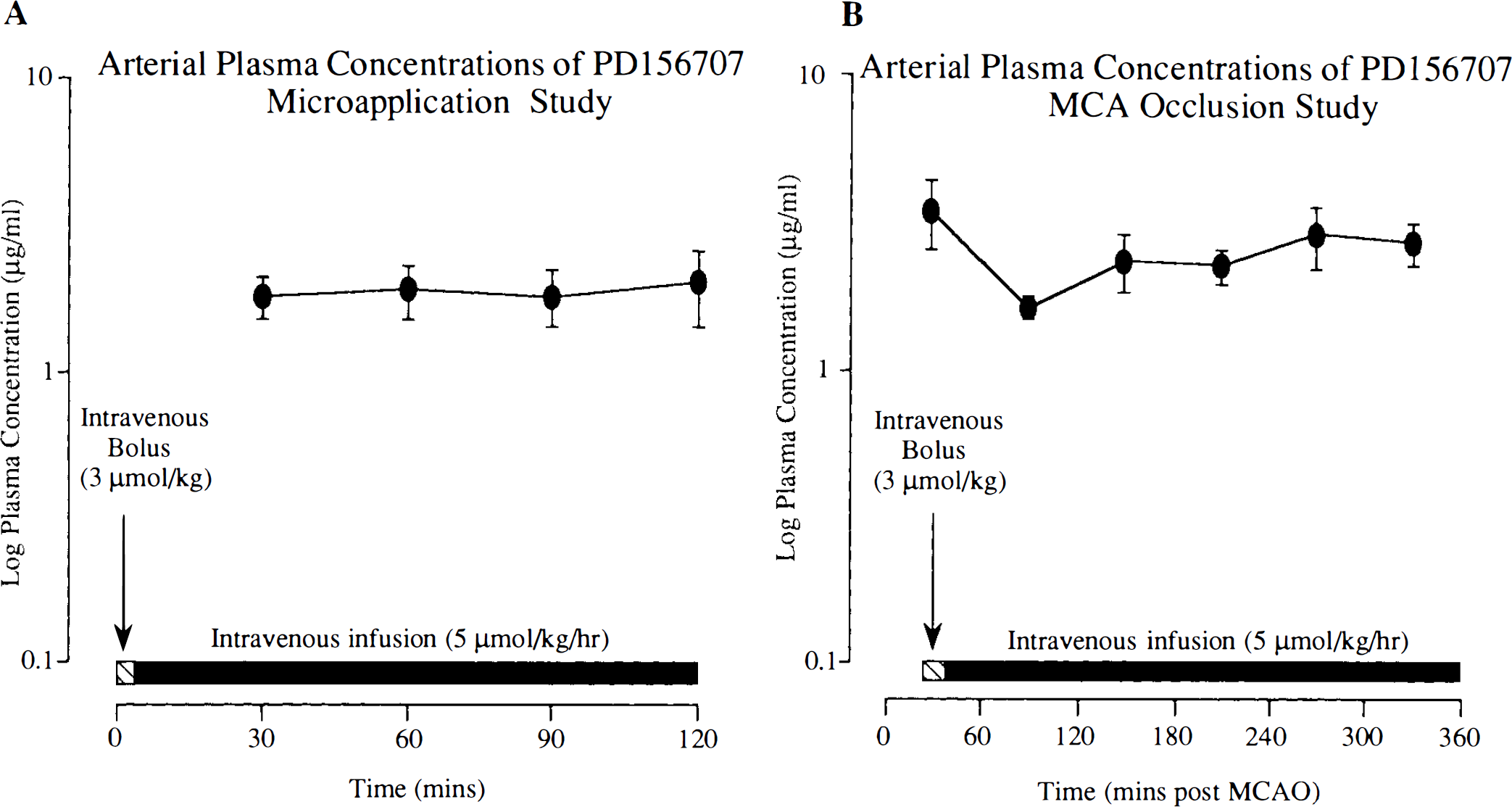
In the microapplication studies, intravenous administration of PD156707 (3 μmol/kg bolus + 5 μmol/kg/h infusion) resulted in drug-plasma levels of 1.9 ± 0.8 μg/ml at 60 min after administration and were maintained at these levels for the duration of the experiment (120 min) (Fig. 4A).
Focal-ischaemia study
Mean arterial blood pressure at the outset of the study was 95 ± 18 mm Hg in the vehicle-treated group and 93 ± 14 mm Hg in the PD156707-treated group. The blood pressure did not vary significantly from these values. Arterial Pco2 was maintained between 28 and 38 mm Hg, and arterial Po2 was maintained at >100 mm Hg for the duration of the experiment. Core temperature was maintained between 36 and 37°C through the course of the experiment.
In the vehicle-treated animals, cerebral perfusion (measured by laser Doppler flowmetry) after MCA occlusion was reduced to 60% of preocclusion baseline levels. The vehicle was administered 30 min after MCA occlusion, and the reduction in cerebral perfusion persisted at 60% of preocclusion baseline levels for the experimental period (Fig. 5). In PD156707-treated animals, MCA occlusion reduced cerebral perfusion to 40% of preocclusion baseline levels. After the intravenous administration of PD156707 (3 μmol/kg bolus + 5 μmol/kg/h infusion) 30 min after MCA occlusion, there was progressive increase in cerebral perfusion, with the restoration of flow to preocclusion baseline levels at the end of the experimental period (Fig. 5).
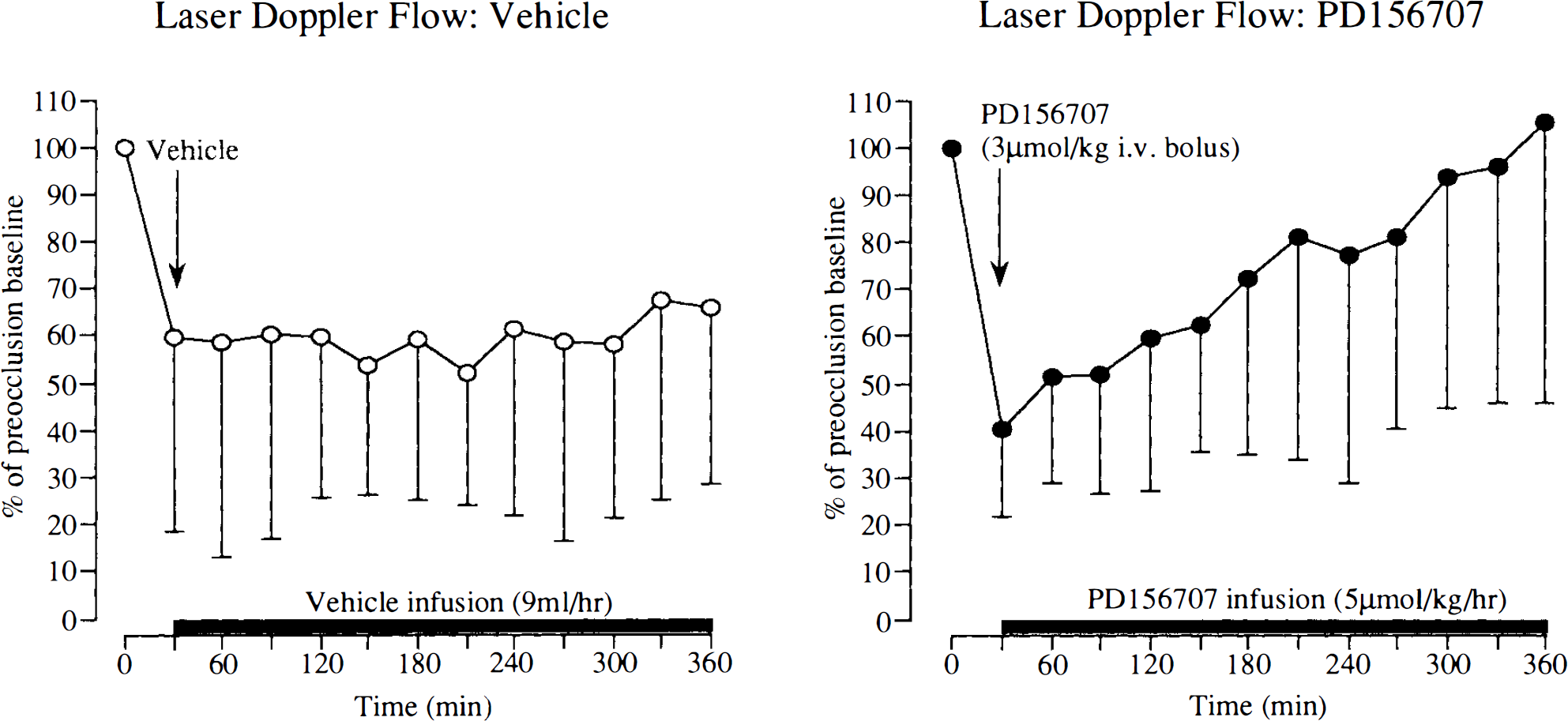
Cerebral perfusion (determined by laser Doppler flowmetry) in vehicle- and PD156707-treated animals. Data are mean ± SD from eight to 10 probes overlying the ectosylvian and suprasylvian gyri in five cats (in each group). The vehicle (saline) was administered 30 min after middle cerebral artery (MCA) occlusion as an intravenous bolus injection, followed by a continuous infusion. PD156707 (3 μmol/kg) was administered as an intravenous bolus injection 30 min after MCA occlusion, followed by a continuous intravenous infusion (5 μmol/kg/h).
Intravenous administration of PD156707 (3 μmol/kg bolus + 5 μmol/kg/h infusion) administered 30 min after MCA occlusion reduced the volume of ischaemic damage in the cerebral hemisphere by 45% (Fig. 6). PD156707 reduced the volume of ischaemic damage in the cerebral cortex by 59% (Fig. 6). The volume of ischaemic damage within the caudate nucleus remained unaffected by PD156707.
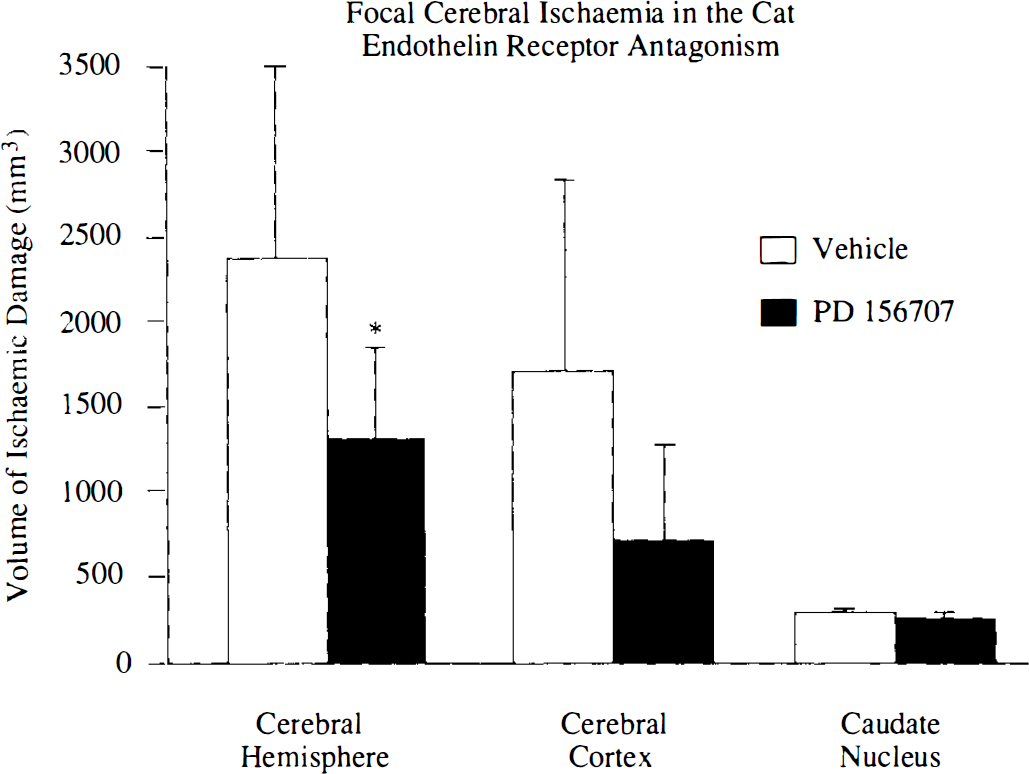
Effect of PD156707 (3 μmol/kg bolus + 5 μmol/kg/h infusion) on volume of ischaemic damage after permanent MCA occlusion. PD156707 was administered 30 min after middle cerebral artery (MCA) occlusion. Data are expressed as mean ± SD (n = 5 in each group). *p < 0.05 compared with vehicle-treated animals (one-tailed Student's t test).
In the MCA-occlusion studies, intravenous administration of PD156707 resulted in drug-plasma levels of 1.7 ± 0.3 μg/ml at 60 min after administration and did not vary significantly from these levels (Fig. 4B).
DISCUSSION
A persuasive case is developing for the involvement of endothelins in the pathophysiology of focal cerebral ischaemia and for the therapeutic potential of endothelin antagonists (Ziv et al., 1992; Rubanyi and Polokoff, 1994). The ability of exogenous ET-1 to overwhelm homeostatic mechanisms that maintain CBF, even in the normal CNS, has attracted interest in its role in influencing postischaemic CBF (Robinson et al., 1990; Fuxe et al., 1992; Macrae et al., 1993; Sharkey et al., 1993). Endothelin receptor antagonists have no significant effect on cerebral arterial or pial arteriolar calibre in nonpathologic conditions in vivo nor do they alter CBF in normal animals (Foley et al., 1994; McAuley et al., 1994; Patel et al., 1994; Patel et al., 1995b). However, topical application of endothelin antagonists increases the calibre of cortical arterioles in vivo in the ischaemic penumbra after a focal cerebral ischaemic challenge (Patel et al., 1995a). The present study demonstrated that the ETA-receptor antagonist PD156707, administered intravenously, increases CBF to periischaemic areas and reduces the extent of ischaemic damage.
Despite the existence of multiple homeostatic mechanisms to protect blood flow to the brain from compromise, constrictor mechanisms that limit tissue perfusion to the ischaemic penumbra after cerebral ischaemia are well recognised (Hossmann et al., 1973). A variety of mechanisms for reductions in CBF after focal ischaemia have been proposed. These include vasoconstriction, increased tissue pressure, and luminal obstruction (Teasdale et al., 1981; Hatashita and Hoff, 1986; Wahl and Schilling, 1993). However, the precise mechanism for the maintenance of reduced postischaemic CBF is unknown. The ability of the endothelin–receptor antagonist PD156707 to increase cerebral perfusion indicates a role for endothelin in the regulation of vascular tone and CBF after MCA occlusion. The sources and mechanisms that underlie the increases in endothelin-mediated vascular tone are unclear, but increases in tissue and CSF endothelin immunoreactivities after focal cerebral ischaemia have been reported by a number of groups (Ziv et al., 1992; Viossat et al., 1993; Barone et al., 1994).
The cellular source of endothelins after a cerebrovascular insult is unclear. The presence of endothelin messenger RNA (mRNA) and release of ET-1 from cerebrovascular endothelial cells have been demonstrated (Yoshimoto et al., 1990; Saito et al., 1991; Spatz et al., 1994). In brain tissue, both astrocytes and neurons have been reported to possess endothelin mRNA (MacCumber et al., 1990; Ehrenreich et al., 1991). The release of endothelin from endothelial cells or from neurons and astrocytes may account for the rapid perturbations in blood vessel calibre after MCA occlusion (MacArthur et al., 1994). The presence of stores of endothelins in some cells indicates that de novo synthesis of endothelins may not be necessary for the development of the acute effects. There are a number of possible triggers for the release and synthesis of endothelins, including tissue hypoxia (Elton et al., 1992). The reduction in CBF and the possibility of blood pooling in the ischaemic region could trigger the release or synthesis of endothelins (Kurihara et al., 1989; Ohlstein and Storer, 1992). Products from platelets such as transforming growth factor (TGF)-β and platelet-derived growth factor (PDGF) are known inducers of endothelin synthesis and may also provide a trigger for the release/synthesis of endothelins after an ischaemic insult (Kurihara et al., 1989).
PD156707 per se failed to alter the calibre of nonischaemic feline pial arterioles, indicating that under our experimental conditions, there is minimal endothelin-mediated tone in cerebral resistance vessels. The immediate response after MCA occlusion is a decrease in cerebrovascular resistance to compensate for the reduction in blood flow. A number of investigations have been performed in which vasodilator substances, such as papaverine and nimodipine, increase CBF provided systemic hypotension is avoided (Kazda et al., 1982; Brandt et al., 1983; Date and Hossmann, 1984; Mohammed et al., 1985). The perivascular microapplication of the calcium channel antagonist nifedipine demonstrated an increase in calibre of postischaemic constricted arterioles, indicating an active vasoconstriction of these blood vessels (Brandt et al., 1983). The perivascular microapplication of endothelin–receptor antagonists on ischaemic arterioles demonstrated an increase in endothelin-mediated vascular tone after focal ischaemia (Patel et al., 1995a).
Recent reports demonstrated the efficacy of endothelin–receptor antagonists in experimental models of global ischaemia (Feuerstein et al., 1994; Ohlstein et al., 1994). Although the role of the endothelins as neurotransmitters or neuromodulators has been suggested, the effects of endothelin–receptor antagonists per se on neuronal function have not been examined (Gross et al., 1993; Berrino et al., 1994). The mechanism of action of the endothelin–receptor antagonists in global cerebral ischaemia is unclear. Few studies have examined the efficacy of endothelin–receptor antagonists in focal cerebral ischaemia (McAuley et al., 1994; Patel et al., 1995a). PD156707 (at a single dose validated for efficacy) reduces the volume of focal ischaemic damage; however, the mechanism of action for this reduction in unclear. It can be speculated that the observed effect may be the result of an enhancement of periischaemic cerebral perfusion.
The endothelin–receptor antagonists targeted at cerebral resistance blood vessels must penetrate the blood–brain barrier (Edvinsson et al., 1993; Patel et al., 1995b). The intravenous dose of PD156707 examined in this investigation attenuates exogenous ET-1-induced vasoconstriction of nonischaemic arterioles, demonstrating that bioactive amounts of PD156707 gain access to the adventitial surface of cerebral resistance arterioles. Our unpublished observations have indicated that ∼12% of the plasma-drug level is detectable in the CSF (T. R. Patel, unpublished observations). This validated dose of PD156707 was subsequently used in the focal-ischaemia investigations and demonstrated its ability to increase cerebral perfusion and reduce ischaemic damage.
PD156707 is a highly selective antagonist for endothelin receptors. PD156707 in concentrations ≤l0 μM did not inhibit the binding of 25 ligands to various neurotransmitter systems, including angiotensin II, neuropeptide Y, and vasoactive intestinal peptide (Reynolds et al., 1995). In vitro pharmacologic data for PD156707 indicate that this compound is selective for the ETA-receptor subtype with an IC50 of 0.3 μM and 420 μM at ETA and ETB receptors, respectively (Doherty et al., 1995). In in vitro functional studies, PD156707 demonstrated a 100-fold selectivity for the ETA receptor compared with the ETB receptor (Reynolds et al., 1995). The dose of PD156707 used in our investigations markedly attenuated the vasoconstrictor response to ET-1 (1 nmol/kg, intravenous bolus) and did not affect the vasodilator response (ETB receptor mediated) elicited by this dose of ET-1, indicating the selectivity of PD156707 for the ETA receptor (T. R. Patel, unpublished observations).
Previous investigations to characterise the endothelin–receptor subtypes in feline cerebral arterioles demonstrated that ETA receptors mediate vasoconstriction and ETB receptors mediate vasodilatation (Kobari et al., 1994; Patel et al., 1995c). The increase in cerebral perfusion in periischaemic areas induced by PD156707 is probably due to antagonism of ETA receptor-mediated vasoconstrictions and, perhaps, the unmasking of ETB receptor-mediated vasodilatation (Kobari et al., 1994; Patel et al., 1995c). The contribution of ETB receptors to the regulation of perfusion in cerebral ischaemia could be assessed by the selective ETB receptor antagonists and agonists. The efficacy of PD156707 in increasing cerebral perfusion in periischaemic areas and reducing hemispheric ischaemic damage after experimental focal cerebral ischaemia indicates the therapeutic potential of endothelin–receptor antagonists in the amelioration of ischaemic damage.
Footnotes
Acknowledgment:
T.R.P. is supported by an MRC/CASE award. We thank the technical staff at the Wellcome Surgical Institute for their support.
