Abstract
The actions of Bosentan and PD155080, non-peptide endothelin receptor antagonists, were examined in feline pial arterioles in situ following middle cerebral artery (MCA) occlusion to gain insight into the cerebrovascular influence of endogenous endothelins in focal cerebral ischaemia. Immediately following permanent MCA occlusion, all pial arterioles overlying the suprasylvian and ectosylvian gyri displayed marked dilatations, which were maintained in a population of vessels but differentiated into sustained constrictions in others. Perivascular subarachnoid microinjections of Bosentan (30 μM), PD155080 (30 μM), and artificial CSF (pH 7.2) were performed between 30 and 210 min following MCA occlusion. The perivascular microapplication of Bosentan (30 μM) and PD155080 (30 μM) around pial vessels overlying the suprasylvian and ectosylvian gyri, which are within the territory of the occluded MCA, elicited an increase in the calibre of postocclusion dilated and constricted pial arterioles. The perivascular microapplication of PD155080 (30 μ/M) around postocclusion constricted arterioles overlying the ectosylvian and suprasylvian gyri elicited an increase in the calibre of arterioles (69 ± 49% from preinjection baseline; n = 8). The perivascular microapplication of Bosentan (30 μM) around postocclusion constricted arterioles overlying the ectosylvian and suprasylvian gyri also elicited an increase in the calibre of arterioles (68 ± 60% from preinjection baseline; n = 13). In contrast, the microapplication of CSF (pH 7.2) elicited small reductions in pial arteriolar calibre of postocclusion constricted arterioles (—8 ± 13% from preinjection baseline; n = 8). The perivascular microapplication of PD155080 (30 μM) around postocclusion dilated pial arterioles overlying the ectosylvian and suprasylvian gyri elicited an increase in the calibre of arterioles (11 ± 10% from preinjection baseline; n = 38). The perivascular microapplication of Bosentan (30 μM) around postocclusion dilated arterioles elicited an increase in the calibre of arterioles (16 ± 15% from preinjection baseline; n = 36). In contrast, the microapplication of CSF (pH 7.2) elicited small reductions in pial arteriolar calibre of postocclusion dilated arterioles (—9 ± 6% from preinjection baseline; n = 44). Perivascular microapplication of Bosentan or PD155080 had minimal effect on the calibre of pial arterioles on the parasagittal gyrus (anterior cerebral artery territory), although these arterioles had also displayed sustained dilatation following MCA occlusion. These results indicate that contractile factors (whose effects can be reversed with endothelin receptor antagonists) constrict or impair dilatation of cortical resistance arterioles in an acute cerebral ischaemic episode.
Keywords
The survival of cerebral tissue is dependent on the continuous delivery of substrates by the blood. Multiple homeostatic mechanisms exist to protect blood flow to the brain from compromise (Paulson et al., 1990). Despite these physiologic mechanisms, constrictor mechanisms that limit tissue perfusion to the ischaemic and periischaemic regions are well-recognized in cerebral ischaemia. Although a variety of mechanisms have been proposed to account for submaximal dilatation in the penumbra of a focal ischaemic episode, such as vasoconstriction, increased tissue pressure, and luminal obstruction, the precise cause of impairment of blood flow to the ischaemic penumbra is unknown (Teasdale et al., 1981; Hatashita and Hoff, 1986; Wahl and Schilling, 1993).
The endothelin family of peptides, endothelin-1 (ET-1), endothelin-2 (ET-2), and endothelin-3 (ET-3), are profound vasoconstrictors of cerebrovascular smooth muscle in vitro and in vivo (Robinson and McCulloch, 1990; Adner et al., 1993; Salom et al., 1993; Feger et al., 1994). Circumstantial evidence has implicated endogenous endothelins in a variety of cerebrovascular pathologies (Suzuki et al., 1992; Ziv et al., 1992). Exogenous endothelin-1 can overwhelm homeostatic mechanisms in the normal CNS and reduce CBF to pathologically low levels (Robinson et al., 1990; Fuxe et al., 1992; Macrae et al., 1993; Sharkey et al., 1993). ET-1 also exacerbate focal ischaemic damage (Nikolov et al., 1993). Elevations of endothelin immunoreactivity in brain tissue have been described following permanent focal ischaemia in the rat, and endothelin levels are increased in the CSF following ischaemic stroke (Duverger et al., 1992; Ziv et al., 1992; Gang-Zhi et al., 1993; Barone et al., 1994).
The effects of the endothelins are mediated via two receptor subtypes, endothelin-A (ETA) and endothelin-B (ETB) (Masaki et al., 1994). The predominant effect of ETA receptors is to mediate vasoconstriction, while ETB receptors mediate vasodilatation. However, vasoconstrictor effects mediated by ETB receptors have been reported in some vascular beds (Masaki et al., 1994). Bosentan is an antagonist of both ETA and ETB receptor subtypes, whereas PD155080 is a selective antagonist for the ETA receptor subtype (Clozel et al., 1994; Doherty et al., 1995). In feline cortical pial arterioles in situ, perivascular coadministration of ET-1 with either Bosentan or PD155080 prevents the vasoconstrictor effect of ET-1 with half maximal inhibition at a concentration of 1 μM of each antagonist (Patel et al., 1994, 1995b). Under normal physiologic conditions, Bosentan per se (in concentrations up to 300 μM) and PD155080 per se (in concentrations up to 30 μM) have no demonstrable effect on pial arteriolar diameter (Patel et al., 1994, 1995b). In the study reported here, we employed perivascular microapplication of Bosentan and PD155080 around feline pial arterioles to gain insight into the pathophysiologic role of endogenous endothelins in vasomotor disturbances associated with acute cerebral ischaemia.
MATERIALS AND METHODS
Surgical preparation
The studies were carried out using 10 adult female cats, weighing between 2 and 3 kg. Anaesthesia was induced using alphaxolone/alphadolone (Saffau, Glaxo) administered into the radial vein. The animals were then intubated, and positive pressure ventilation with N2O/O2 (70/30%) was initiated. The femoral arteries and veins were cannulated for the monitoring of arterial blood pressure and arterial blood gas status and also for administration of fluids and drugs. Anaesthesia was maintained using a-chloralose (60 mg/kg), and supplements of 1–2 ml (1% solution) were administered i.v. during the experiment. The inspired gas mixture was altered to oxygen-supplemented air (25% O2) prior to the start of the microapplication studies.
The animals were placed in a stereotactic frame. After a midline incision, the scalp was retracted and sutured onto a metal ring to form a well over the calvarium. The temporalis muscle was retracted, and a rectangular craniectomy (3 ×2 cm) was made over the parietal cortex, using a saline-cooled dental drill, exposing the parasagittal, suprasylvian, and ectosylvian gyri. The exposed dura was bathed with mineral oil at 38°C to form a pool, and a continuous flow of oil at a rate of ∼1 ml/min was maintained. With the aid of a Bausch & Lomb stereomicroscope the dura was excised and reflected laterally, and the preparation was allowed to equilibrate for ∼30 min. Bipolar diathermy was used to prevent bleeding from the dural vessels.
Arterial blood samples were taken at regular intervals, and the animals were kept normocapnic by adjusting the stroke volume. Metabolic acidosis was controlled by the administration of sodium bicarbonate (8.4% solution). Core temperature was measured by rectal thermometer and the temperature was maintained at 37°C by a heating blanket. At the outset of the study, the mean arterial blood pressure was 86 ± 5 mm Hg, arterial pH was 7.42 ± 0.05, arterial Pco2 was 31 ± 4 mm Hg, and arterial Po2 was 201 ± 25 mm Hg; these parameters were maintained within these ranges for the duration of the experiment.
Measurement of pial arteriolar calibre
Pial arteriolar calibre was measured by the method of Baez (1966) using an image splitter linked through a closed-circuit video display system. A detailed description of the measurement techniques has appeared previously (Robinson and McCulloch, 1990). Briefly, the individual pial vessels are viewed through a stereomicroscope, and the arteriolar diameter is measured from the degree of shear applied to the image splitter to tangentially appose the two images. The system is calibrated at ×40 and ×70 against threads of known diameter, which allows the direct measurement of vascular diameter in absolute units (micrometers).
Sharp glass micropipettes (tip diameter, 10–12 μm) were vacuum-filled with either artificial CSF or drugs dissolved in CSF. The composition of the CSF was as follows: Na+, 156 mM; K+, 3 mM; Ca2+, 2.5 mM; HCO3–, 12 mM; Cl−, 152 mM. The pH was adjusted to 7.2 by aeration with 95% O2 and 5% CO2. Bosentan (30 μM), PD155080 (30 μM), or endothelin (10 nM) was dissolved in CSF and applied by perivascular microapplication using a micromanipulator to position the pipettes in the perivascular space surrounding the vessel. Small volumes (∼5 μl) were delivered by a pressure ejection system.
In the first study, the ability of Bosentan or PD155080 to reverse the constriction induced by the perivascular microapplication of ET-1 was investigated in three cats with no exposure of the middle cerebral artery (MCA). The calibre of the vessels was recorded for 5–15 min following the perivascular microapplication of ET-1 (10 nM). Bosentan (30 μM), PD155080 (30 μM), or CSF (pH 7.2) were applied to the ET-1-constricted vessels 5–15 min subsequent to the first microapplication of ET-1. The vessel diameter was measured before insertion of micropipettes and before and after injection of substances; the vessel diameter was monitored for 1–2 min.
The MCA was exposed in seven cats by a neurosurgeon via a transorbital approach as previously described (Chen et al., 1991). Briefly, the contents of the left orbit were exenterated, and the optic foramen was enlarged to expose the dura and the MCA underlying it. The dura was excised, the MCA and its branches were occluded using bipolar diathermy, and the artery was sectioned. In each cat, the diameter of three to eight pial arterioles was monitored for 10–20 min prior to MCA occlusion and for 30 min after MCA occlusion. Perivascular microinjections of Bosentan (30 μM), PD155080 (30 μM), and CSF (pH 7.2) were made 30–210 min following MCA occlusion.
Statistical analysis
The ability of Bosentan or PD155080 to reverse exogenous ET-1-induced vasoconstriction was assessed by unpaired Student t test. The data from the vessels overlying the parasagittal gyrus and from the postocclusion dilated and constricted vessels overlying the suprasylvian and ectosylvian gyri were analyzed using one-way analysis of variance (ANOVA) and the Student t test with Bonferroni correction for multiple group comparisons (Bosentan or PD155080 vs. CSF). All results are expressed as mean ± SD of the percentage of change of pial arteriolar calibre from preinjection baseline (n = the number of arterioles examined).
RESULTS
Reversal of vasoconstriction induced by exogenous endothelin-1
The perivascular microapplication of Bosentan per se (0.3–300 μM) or PD155080 per se (0.3–30 μM) on nonischaemic pial arterioles has no demonstrable effect on arteriolar calibre (Patel et al., 1994, 1995b). Pial arterioles were constricted by the perivascular microapplication of ET-1 (10 nM); constriction was −29.1 ± 1.9% from preinjection baseline (n = 24). CSF applied 5–15 min following microapplication of ET-1 had minimal effect on the calibre of vessels constricted by ET-1 (−0.6 ± 4.1% from ET-1-constricted baseline; n = 12). Bosentan (30 μM) applied 5–15 min after the first microapplication of ET-1 caused a marked increase in vessel calibre from the ET-1-constricted baseline (22.4 ± 9.6% from ET-1-constricted baseline; n = 7; p < 0.01; Fig. 1).
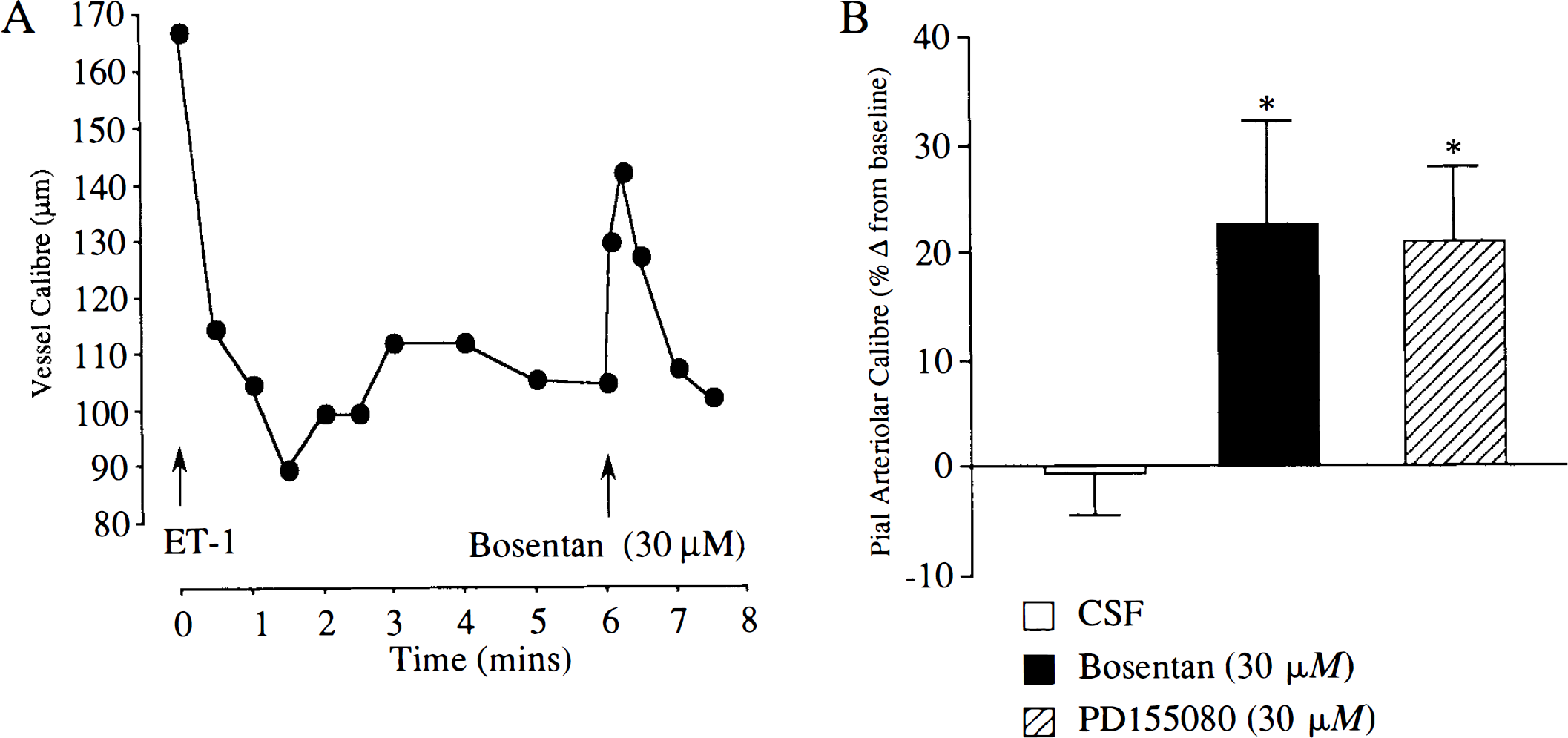
The perivascular administration of PD155080 (30 μM) to a pial arteriole constricted with ET-1 resulted in a significant increase in calibre from the level immediately prior to PD155080 injection (mean change in calibre, 21 ± 6.9% from ET-1-constricted baseline; n = 8; p < 0.01; Fig. 1). The reversal or dilatation was transient with a typical duration of ∼30–60 s before the vessel returned to the calibre it had prior to the application of the antagonist.
Alterations in vascular diameter after MCA occlusion
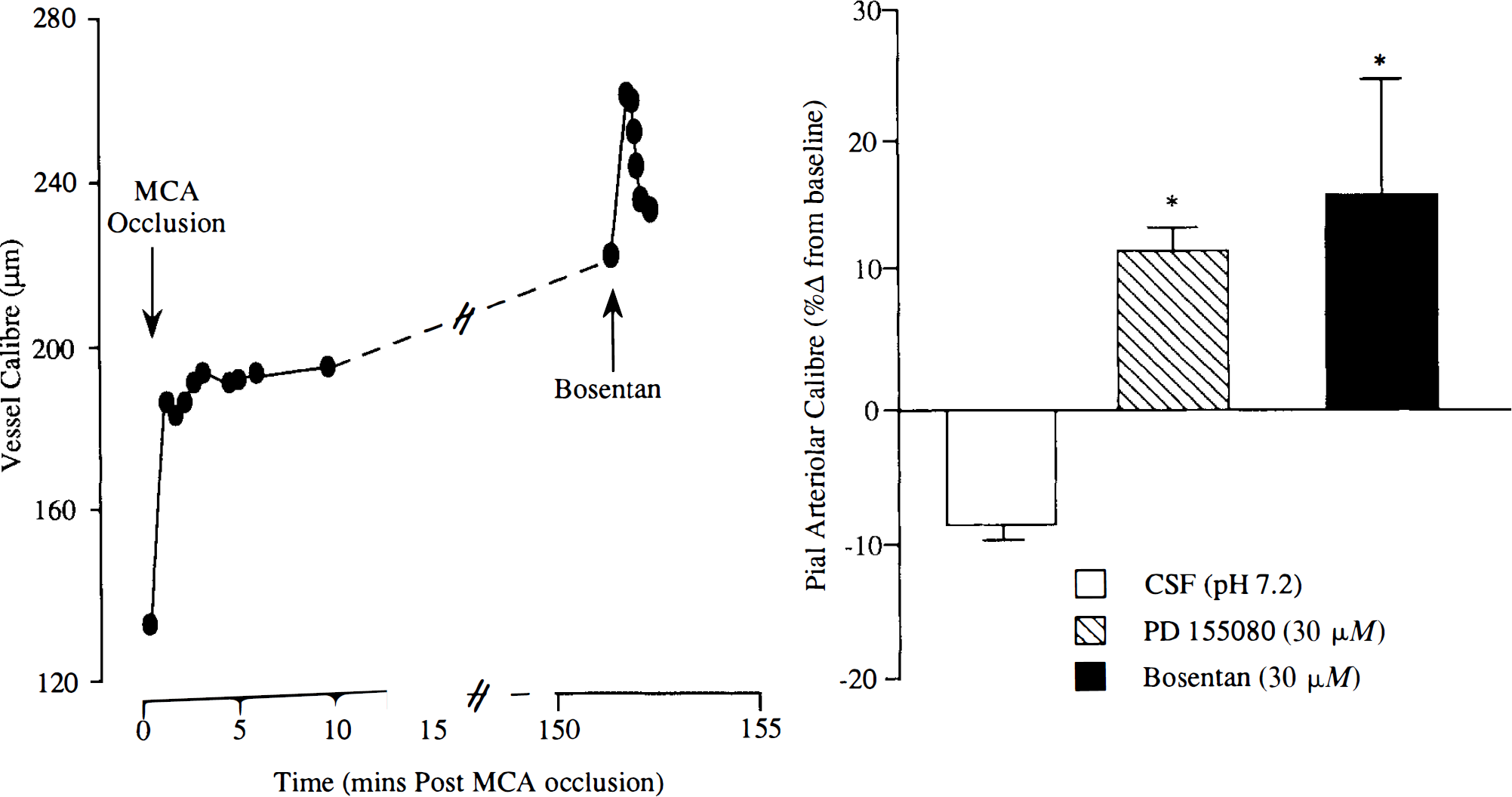
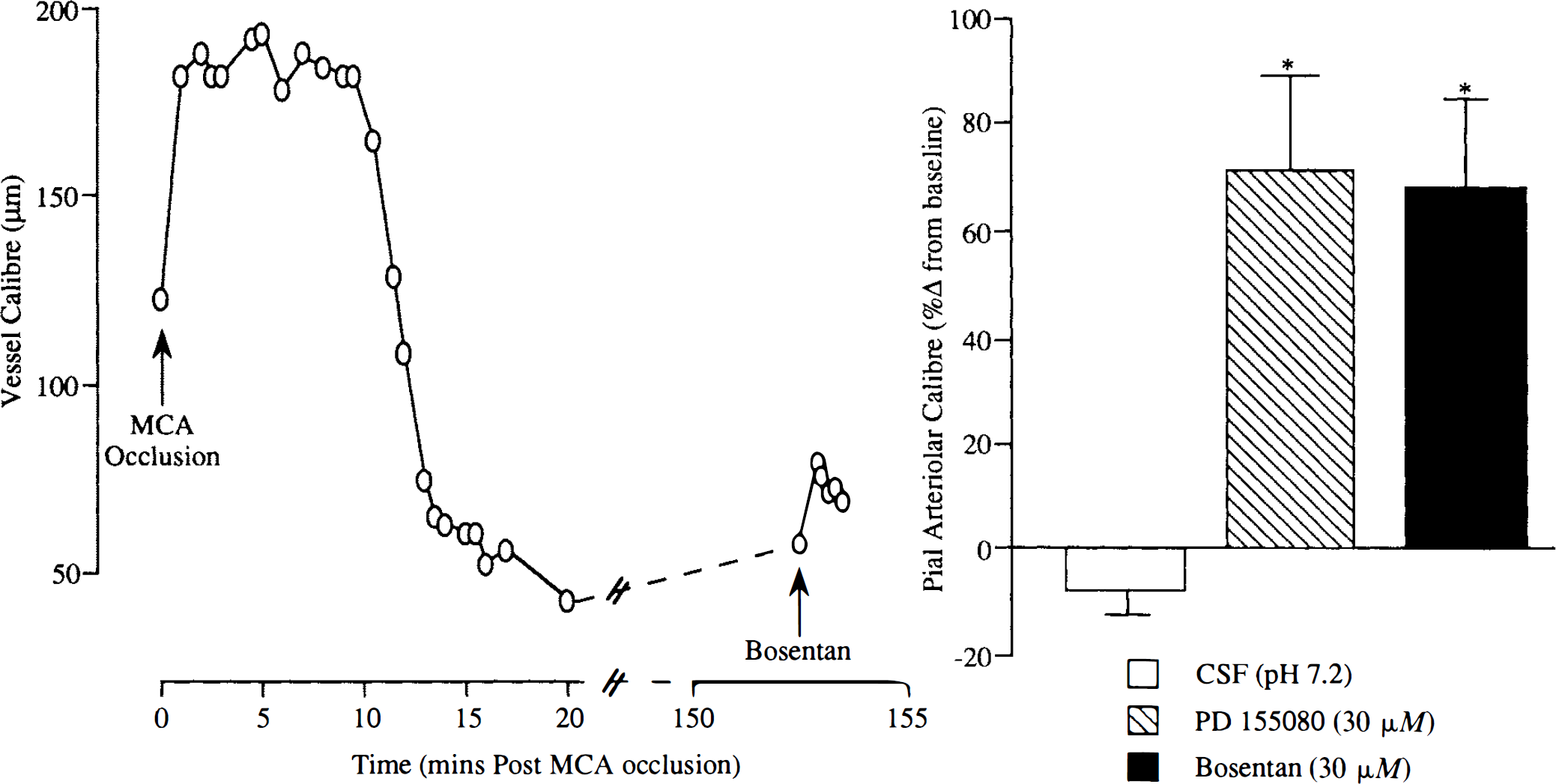
The cranial window exposed cortical pial arterioles overlying the ectosylvian, suprasylvian, and parasagittal gyri. The permanent occlusion of the MCA produced an immediate increase (∼50% from preocclusion levels) in pial arteriolar calibre irrespective of the neuroanatomical site of the vessels. Over the course of the succeeding 30 min, all of the vessels on the parasagittal gyrus (anterior cerebral artery territory) displayed sustained dilatations. Pial arterioles located on the suprasylvian and ectosylvian gyri (the region supplied by the MCA and representing ischaemic penumbra) displayed a mixture of vasoconstriction and vasodilatation by 30 min after MCA occlusion. In four cats, pial arterioles on the ectosylvian and suprasylvian gyri displayed sustained vasodilatation. In two cats, there were widespread zones of sustained vasoconstriction (with reductions in arteriolar calibre >50% from preocclusion levels) within the ectosylvian and suprasylvian gyri. In one cat, there was a circumscribed zone of vasoconstriction confined to the ectosylvian gyrus, which was labile over time; elsewhere in the ectosylvian and suprasylvian gyri there was marked vasodilatation. The onset of vasoconstriction after MCA occlusion varied considerably among the vessels. In a few, it was well-developed by 10 min after MCA occlusion; in others it began to develop 20 min after MCA occlusion. Once vasoconstriction had developed, it was generally sustained. Of the 25 vessels whose calibre was followed sequentially for 30 min postocclusion, 15 displayed marked sustained dilatation and 10 displayed marked sustained vasoconstriction. The frequency of constriction or dilatation reflects anatomical bias in the selection of vessels for sequential assessment in the preocclusion and immediate postocclusion period.
Vasomotor effect of CSF after MCA occlusion
In vessels overlying the ectosylvian and suprasylvian gyri, the perivascular microapplication of CSF resulted in small reductions in pial arteriolar diameter in the period 30–210 min after occlusion. The magnitude of the vascular responses to perivascular microapplication of CSF was similar in vessels that displayed sustained vasodilatation after MCA occlusion and in vessels that displayed sustained vasoconstriction (Figs. 2 and 3). The response of pial arterioles to perivascular microapplication of CSF did not alter significantly in the period 30–210 min after MCA occlusion (Fig. 4).
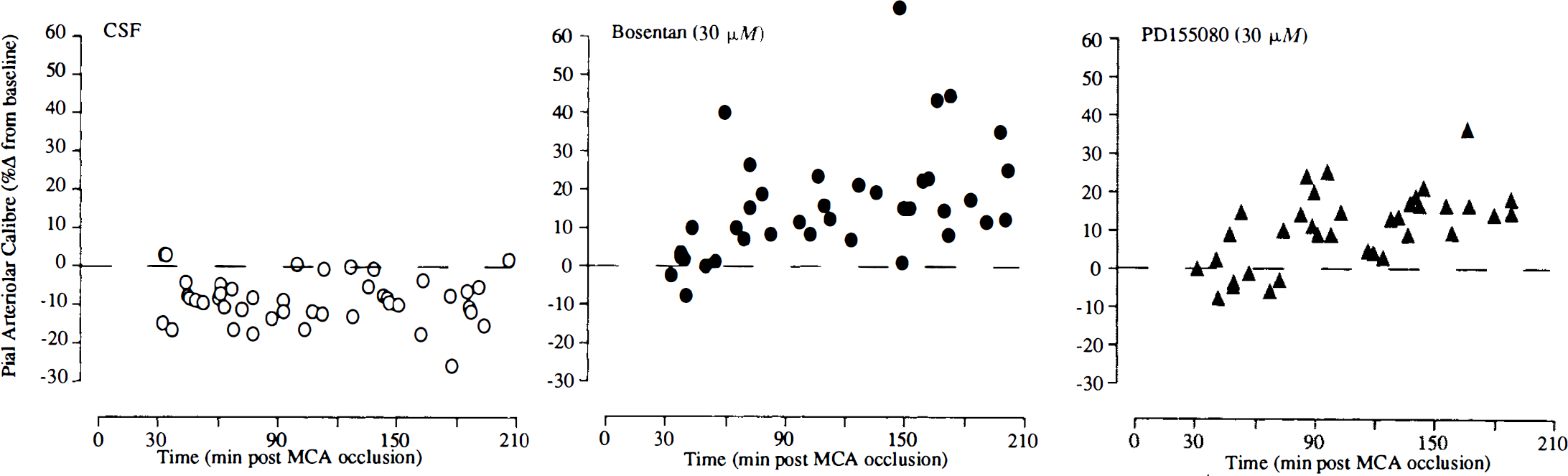
Responses of individual dilated pial arterioles to CSF, Bosentan (30 μM), and PD155080 (30 μM) at varying times after MCA occlusion. All arterioles had displayed sustained vasodilatation after MCA occlusion. Each point represents the response of a single pial arteriole. The MCA was occluded at time zero. Data are expressed as percentage change from preinjection baseline.
Vasomotor effect of Bosentan after MCA occlusion
In vessels overlying the ectosylvian and suprasylvian gyri, the perivascular microapplication of Bosentan (30 μM) resulted in marked increases in pial arteriolar diameter in the period 30–210 min after MCA occlusion in all vessels studied, irrespective of the nature of their response following MCA occlusion. The diameter of pial arterioles displaying sustained dilatation after MCA occlusion increased from 175 ± 67 to 202 ± 80 μm (n = 38) following perivascular microapplication of Bosentan (Figs. 2 and 4). The response to microapplication of Bosentan on postocclusion dilated arterioles was significantly time-dependent; the response was significantly greater later after MCA occlusion (two-way ANOVA, p < 0.05). The diameter of pial arterioles displaying sustained constriction after MCA occlusion increased from 97 ± 52 to 153 ± 75 μm (n = 13) following perivascular application of Bosentan (Figs. 3, and 5).
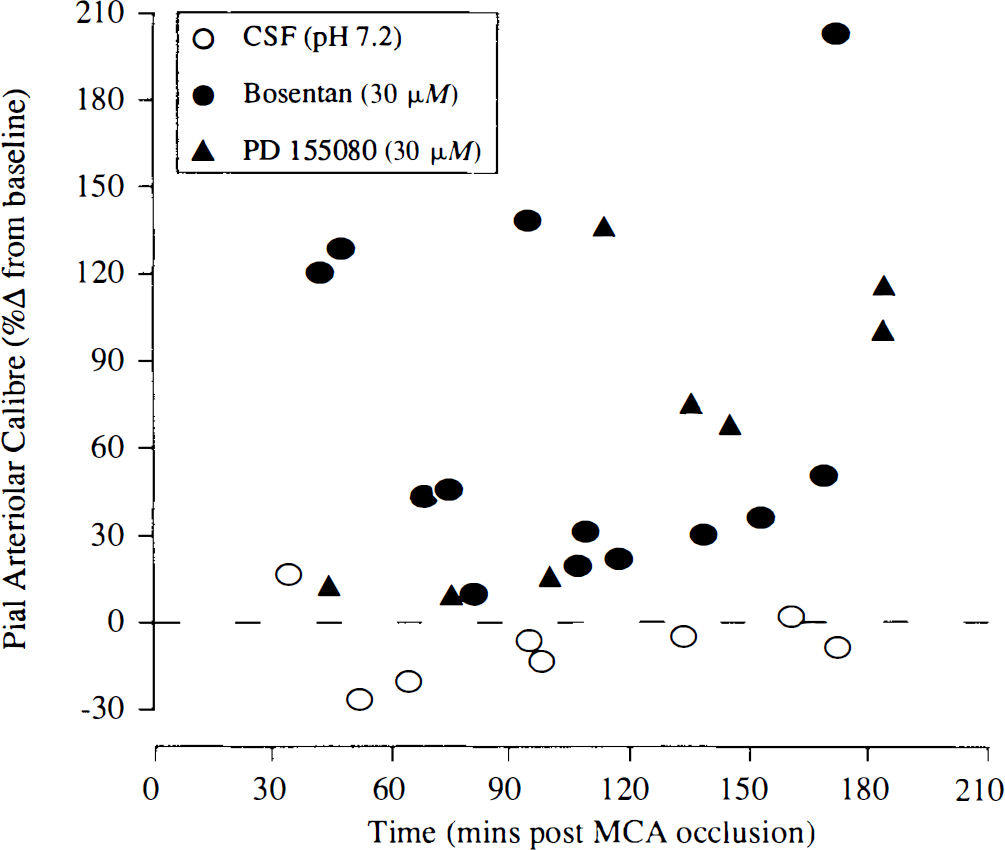
Responses of individual constricted pial arterioles to CSF, Bosentan (30 μM), and PD155080 (30 μM) at varying times after MCA occlusion. All arterioles had displayed sustained vasoconstriction after MCA occlusion. Each point represents the response of a single pial arteriole. The MCA was occluded at time zero. Data are expressed as percentage change from preinjection baseline.
In vessels overlying the parasagittal gyrus, there was no significant difference between the response of pial arterioles to perivascular microapplication of Bosentan (−2.1 ± 5.5% from preinjection baseline; n = 7) and the response to CSF (−7.1 ± 6.7% from preinjection baseline; n = 5); these investigations were made at 131 ± 9 and 114 ± 13 min after MCA occlusion, respectively.
Vasomotor effect of PD155080 after MCA occlusion
The perivascular microapplication of PD155080 in standard CSF resulted in increases in arteriolar calibre of postocclusion dilated and constricted arterioles (Figs. 2–5). The diameter of pial arterioles displaying sustained dilatation after MCA occlusion increased from 161 ± 57 to 173 ± 59 μm (n = 36) following perivascular microapplication of PD155080 (30 μM). The diameter of pial arterioles displaying sustained constriction after MCA occlusion increased from 92 ± 44 to 150 ± 62 μm (n = 8) following perivascular application of PD155080. The magnitude of the increase in calibre after microapplication of PD155080 was numerically greater in vessels displaying postischaemic constriction than in vessels displaying postischaemic dilatation (Figs. 2 and 3). The response to microapplication of PD155080 on postocclusion dilated arterioles became significantly greater as the postocclusion time period increased (two-way ANOVA, p < 0.05; Fig. 4).
In vessels overlying the parasagittal gyrus, there was no significant difference between the response of pial arterioles to perivascular microapplication of PD155080 (−1.5 ± 6.9% from preinjection baseline; n = 5) and the response to CSF (−7.1 ± 6.7% from preinjection baseline; n = 5); these investigations were made at 141 ± 41 and 114 ± 13 min after MCA occlusion, respectively.
DISCUSSION
The potent vasoconstriction effects of the endothelins has attracted much attention for their involvement in almost every vascular pathology, although the precise definition of their pathophysiologic role has proven elusive (Brunner et al., 1992; Ziv et al., 1992; Gellai et al., 1994; Rubanyi and Polokoff, 1994). The perivascular microapplication of Bosentan or PD155080 on nonischaemic feline pial arterioles had minimal effect on arteriolar calibre (Patel et al., 1994, 1995b); these results indicate that under normal physiologic conditions there is minimal endothelin-mediated vascular tone. The present data suggest that following focal cerebral ischaemia, endogenous endothelins constrict and restrict the dilatation of cortical arterioles in the ischaemic penumbra. The constrictor effects of endogenous endothelins (defined by reversal with the endothelin antagonists Bosentan and PD155080) are demonstrable early in the evolution of ischaemic injury, at times when neuroprotective drugs, such as NMDA receptor antagonists, can still salvage tissue from irreversible damage (Ozyurt et al., 1988). Our findings provide insight into a possible pathophysiologic role for endothelins in cerebral ischaemia and also provide a basis for the use of endothelin antagonists in the treatment of occlusive stroke.
A marked reduction in cerebrovascular resistance and the dilatation of the cerebral vasculature occur at the onset of cerebral ischaemia to try to maintain tissue oxygenation. The mechanism for these events has long been attributed to the release of “chemical products of cerebral metabolism,” with hydrogen ions, potassium ions, and adenosine among the most favoured candidates. On the other hand, there is evidence that endogenous constrictor mechanisms evolve that effectively limit tissue perfusion to ischaemic and periischaemic brain regions (Teasdale et al., 1981). In focal ischaemia, vasodilator agents (calcium entry blockers, papaverine, etc.) have been utilised to produce further decreases in vascular resistance, to increase the calibre of arterioles, and, if systemic hypotension is avoided, to increase tissue blood flow to the ischaemic penumbra (Brandt et al., 1983; Date and Hossman, 1984; Mohammed et al., 1985).
Several mechanisms have been proposed (and disputed) to restrict tissue perfusion in cerebral ischaemia, including intraluminal obstruction, mechanical compression of the intraparenchymal vessels (consequent to astrocytic swelling and ultimately tissue oedema), and active vasoconstriction (Teasdale et al., 1981; Hatashita and Hoff, 1986; Wahl and Schilling, 1993). The demonstration that arteriolar calibre in the ischaemic penumbra is increased after microapplication of the endothelin receptor antagonists Bosentan and PD155080 indicates that endogenous endothelins may be involved in limiting blood flow to this zone. While our findings clearly demonstrate that there is an endothelin-mediated component to alterations in vessel calibre following a focal ischaemic insult, endothelin antagonists (including Bosentan) have been shown to markedly reverse the vasospasm of arteries associated with subarachnoid hemorrhage in several species (Rubanyi and Polokoff, 1994) with subarachnoid hemorrhage. The stimulus for the increased levels of endothelins is likely to be blood products and agents present at the site of injury (Yanagiswa et al., 1988; Kurihara et al., 1989; Ohlstein and Storer, 1992).
The trigger for and sources of the putative increased perivascular endothelins in focal ischaemia are presently unclear. Intracellular concentrations of ET-1, as determined by radioimmunoassay, are increased 24 h after a focal (MCA occlusion) or global (two-vessel occlusion plus hypotension) ischaemic insult (Duverger et al., 1992; Barone et al., 1994). Endothelial cells and neurones from the brain and spinal cord express ET mRNA and ET immunoreactivity, implicating these cells as potential sources of endothelins following an ischaemic result (Giaid et al., 1989; Lee et al., 1990; MacCumber et al., 1990). Although secretory granules are not generally found in endothelial cells, in contrast to neurones, it is known that endothelial cells do contain some ET-like peptide, which may be released under certain conditions (MacArthur et al., 1994). This fact supports evidence that de novo synthesis of this peptide (which may take hours) is not always necessary and may explain the ability of Bosentan and PD155080 to alter or reverse ischaemically induced perturbations in pial vessel calibre relatively acutely after the insult in the present study.
In a gerbil global ischaemia model, protection of the CA1 neurones was achieved by intervention with the endothelin antagonists BQ-123 and SB209670 (Feuerstein et al., 1994; Ohlstein et al., 1994). Increased perfusion of the area following administration of the ET antagonist cannot be ruled out as a mechanism of this therapeutic effect in global ischaemia; however, there is controversial evidence that endothelins may have a direct action on neuronal viability (Hokfelt et al., 1989; Lustig et al., 1992; Tence et al., 1992).
Hypoxia, following an occlusive insult, it is the most likely stimulus which likely elicits an increase in endothelins to vasoactive concentrations. Several studies in cell culture support this idea (Elton et al., 1992). However, evidence in the present study indicates that rheologic factors (or reactive alterations in vessel calibre) per se are insufficient to elicit vasoactive amounts of endothelins, since there was no vasomotor response to Bosentan or PD155080 in the postocclusion dilated vessels overlying the parasagittal gyrus (with blood flow derived from the anterior cerebral artery). Evidence from cell culture studies indicates that shear stress may induce an increase in ET-1 mRNA or ET-1 release (Kuchan and Frangos, 1993; Morita et al., 1993; MacArthur et al., 1994). Differences in the source and target of endothelin may occur, depending on the brain injury (Kurihara et al., 1989; Barone et al., 1994).
Interpretation of our results depends on the selectivity of action of Bosentan and PD155080 on endothelin receptors. Bosentan inhibits the binding of radiolabeled ET-1 to ETA and ETB receptors with k; values of 5 and 95 nM, respectively (Clozel et al., 1994). Bosentan has no demonstrable effects on the binding of other peptides with the exception of neurokinin A, the binding of which was reduced at very high Bosentan concentrations (10 μM) (Clozel et al., 1994). In contrast, PD155080 inhibits binding of radiolabeled ET-1 to ETA and ETB receptors with IC50 values of 7.8 nM and 3.5 μM, respectively (Doherty et al., 1995). In feline pial arterioles in situ, perivascular microapplication of Bosentan or PD155080 have no effect on arteriolar calibre under normoxic, normocapnic, and normotensive conditions (Patel et al., 1994, 1995b). Coadministration of Bosentan or PD155080 with ET-1 (10 nM) inhibits the ET-1-induced constriction of pial arterioles (but not that produced by other vasoconstrictors) with an IC50 of 1 μM for both antagonists; complete blockade was achieved with coapplication of higher concentrations of the antagonists (micropipette concentration, 30 μM) (Patel et al., 1994, 1995b). Despite the slow dissociation of endothelins from their receptors (Marsault et al., 1993), the present data in normal cats indicate that partial reversal of ET-1-induced cerebral vasoconstriction can be achieved by delayed treatment with either Bosentan or PD155080.
We have examined the effects of combined ETA-ETB receptor antagonist (Bosentan) and an ETA-selective receptor antagonist (PD155080) on ischaemic arterioles. Bosentan and PD155080 elicited similar magnitude of effects on ischaemic dilated and constricted arterioles. Activation of ETA or ETB can mediate vascular contractions depending on the vessel examined. In feline pial arterioles, however, the ET-1-induced vasoconstriction appears to be mediated via ETA receptors, whereas ETB activation produces vasodilatation (Kobari et al., 1994; Patel et al., unpublished observations). Receptor characterisation of ET-1 cerebrovascular actions in other species generally supports such a view of the function of ET receptor subtypes (Kitazono et al., 1993; Feger et al., 1994). The use of a mixed antagonist such as Bosentan does not allow elucidation of the endothelin receptor subtype that mediates the observed cerebrovascular constriction. However, the ability of the endothelin ETA receptor-selective antagonist PD155080 to increase the calibre of both postocclusion dilated and constricted arterioles indicates that ETA receptor activation is probably the dominant mediator of the cerebrovascular effects of endogenous ET-1 following focal ischaemia. The ability of an ETA receptor antagonist to increase CBF to the ischaemic penumbra and reduce ischaemic neuronal damage has been demonstrated (Patel et al., 1995a). An important aspect of the pharmacology of Bosentan and PD155080 is the ability to gain access to the vascular smooth muscle of cerebral resistance vessels after i.v. administration (Patel et al., 1994, 1995b). These observations and the finding that the calibre of vessels in the ischaemic penumbra can be increased by Bosentan and PD155080 provide a basis for assessing the therapeutic utility of endothelin receptor antagonists in focal cerebral ischaemia.
Footnotes
Acknowledgment:
T.R.P. is supported by an MRC/CASE studentship. The authors thank Dr. M. Clozel (Hoffmann-La Roche) for the gift of Bosentan, Dr. A. Doherty (Parke-Davis Pharmaceuticals) for the gift of PD155080, and technical staff at the Wellcome Surgical Institute.
