Abstract
Cocaine- and amphetamine-regulated transcript (CART) peptides are known to be involved in the stress response and have been implicated in the regulation of the cardiovascular system. We evaluated the direct vasoactive properties of CART in the cerebral circulation and its potential mechanisms of action. Penetrating cerebral arterioles, isolated from male Sprague—Dawley rats, were cannulated using a concentric micropipette setup, pressurized and perfused. The vascular response to intraluminal and extraluminal CART peptide was characterized. The endothelium dependence of this response was assessed by means of the endothelial light—dye injury model. The nonspecific endothelin receptor antagonist PD-145065, the ETA-specific antagonist BQ-123, the ETB-specific antagonist BQ-788, and the inhibitor of endothelin-converting enzyme phosphoramidon were used to characterize the involvement of the endothelin pathway in the vascular response to CART peptide. Extraluminal and intraluminal application of CART peptide (0.1 nm to 1 μmol/L) evoked a long-lasting dose-dependent constriction of isolated penetrating cerebral arterioles to ~80% of resting myogenic tone. Disruption of the endothelium by the endothelial light/dye injury model resulted in the abolition of this response (P<0.05). Extraluminal administration of PD-145065, BQ-123, and phosphoramidon blocked the constriction response to CART peptide (P<0.01). The ETB antagonist, BQ-788, did not alter the constriction response to CART peptide. Cocaine- and amphetamine-regulated transcript peptide is a potent vasoconstrictor in the cerebral circulation. Its direct vasoactive properties are endothelium-dependent and are mediated by ETA, not ETB, endothelin receptors.
Introduction
Since initial identification in 1995 (Douglass et al, 1995) as a transcript linked to acute psychostimulant use, the physiologic relevance of cocaine- and amphetamine-regulated transcript (CART) and its associated peptides have been the subject of considerable investigation. Association with cocaine and amphetamine administration and abundance in the hypothalamus (Couceyro et al, 1997; Gautvik et al, 1996; Koylu et al, 1998) has led to much attention in the role of CART peptides in the regulation of feeding behavior (Kristensen et al, 1998; Kuhar and Dall Vechia, 1999; Lambert et al, 1998), in reinforcement and reward (Jaworski et al, 2003; Kuhar and Dall Vechia, 1999), and in meditating the locomotor effects of psychostimulants (Kimmel et al, 2000). An increasing body of evidence, however, indicates a broader physiologic role for CART.
There is substantial support for CART's role in the regulation of the hypothalamo—pituitary—adrenal (HPA) axis. In addition to being one of the most abundant hypothalamic transcripts (Gautvik et al, 1996), CART mRNA expression and CART immunoreactivity (CART-IR) have been observed in the pituitary and adrenal glands (Couceyro et al, 1997; Koylu et al, 1997). Within the paraventricular nucleus (PVN), CART-IR is present in oxytocinergic neurons (Vrang et al, 1999). Cocaine- and amphetamine-regulated transcript IR is also found in regions known to regulate corticotropin releasing factor (CRF) including the hippocampus, amygdala, arcuate, and the PVN (Koylu et al, 1998). Central administration of CART peptide results in the activation of CRF-neurons in the PVN in addition to an increase in circulating adrenocorticotropic hormone (ACTH) and corticosterone levels (Smith et al, 2004; Vrang et al, 2000). The regulation of CART mRNA levels in the pituitary and PVN and of circulating CART peptide by glucocorticoids (Balkan et al, 2003; Stanley et al, 2004; Vicentic et al, 2004) further suggests that CART is involved in the stress response.
Consistent with this role, CART has been implicated in the brain's regulation of the cardiovascular system. Within the medulla, CART-immunoreactive cells have been localized to the nucleus of the solitary tract and the area postrema (Koylu et al, 1998)–major cardiovascular centers. Central administration of CART peptide evoked an increase in arterial blood pressure in conscious rabbits (Matsumura et al, 2001) and blocked phenylephrine-induced bradycardia in the rat (Scruggs et al, 2003). Further, CART peptide is released into the hypothalamic—pituitary portal circulation in response to NO-induced hypotension (Larsen et al, 2003). Based on these cardiovascular effects, we sought to investigate whether CART peptide may possess vasoactive properties. The effect of CART directly on cerebrovascular tissue is not known. Thus, the objective of the present study was to determine whether rat CART peptide (55-102) is vasoactive in isolated rat cerebral arterioles and if so, by what mechanisms these effects were mediated.
Materials and methods
This study was conducted in accordance with the National Institutes of Health guidelines for the care and use of animals in research and under protocols approved by the Oregon Health & Science University Institutional Animal Care and Use Committee.
Vessel Isolation and Cannulation
The techniques for the dissection and isolation of intracerebral arterioles were adapted from previously published methods (Dacey and Duling, 1982; Ngai and Winn, 1993; Ngai and Winn, 1995). Briefly, 52 male Sprague—Dawley rats (350 to 400 g) were anesthetized with sodium pentobarbital (50 mg/kg intraperitoneally) and decapitated. An unbranched segment of the intracerebral arteriole, 100 to 500 μm in length and 40 to 80 μm in diameter, was isolated and placed in a temperature-controlled bath. The isolated segment was cannulated proximally and distally with a concentric micropipette system that consisted of an inner perfusion pipette and an outer holding pipette. The vessel was visualized and the diameter measured with a video dimension analyzer attached to a monochrome video camera and monitor.
After cannulation, the vessel was pressurized to 60 mm Hg and the passive diameter was measured. It was superfused extraluminally with a MOPS-buffered saline solution (pH = 7.3) at a rate of 1 mL/min while intraluminal flow of a 1% albumin saline solution (pH = 7.4) was maintained at a rate of 0.2 μL/min. The vessel bath temperature was increased to 37°C, and over a period of 30 to 45 mins viable vessels developed vasomotion and spontaneous myogenic tone characterized by a stable baseline diameter. Vessel reactivity was assessed with an extraluminal acidic buffer (pH = 6.8) challenge. Vessels failing to achieve spontaneous myogenic tone (<75% of passive diameter) or lacking a robust pH response (>15% dilation) were excluded from data collection.
Cocaine- and Amphetamine-Regulated Transcript Peptide Dose—Response
In all studies ‘CART peptide’ refers to rat biologically active CART 55-102 fragment of the pro-CART peptide. A cumulative concentration—response curve was constructed for CART peptide by the stepwise replacement of extraluminally perfused MOPS buffer with increasing (0.1 nmol/L to 1.0 μmol/L) CART peptide concentrations. In initial studies, the CART response had an onset within 1 to 2 mins, achieved maximal effect within 5 to 7 mins and remained stable during the duration of CART perfusion up to 15 to 20 mins. Beginning with 0.1 nmol/L, CART peptide was perfused for 10 mins and the resulting steady-state vessel diameter was recorded. The CART peptide concentration was increased in 10-fold increments and the responses were observed through the maximal CART peptide concentration of 1 μmol/L. Extraluminal perfusion of neutral MOPS buffer was restored and the vessel was allowed to regain baseline diameter. Vessels that failed to achieve resting tone (±10% of baseline diameter) after CART washout were excluded from the present study.
The effects of intraluminal versus extraluminal CART peptide administration on cerebral arterioles were compared. We intraluminally perfused CART peptide (1 nmol/L) for 10 mins at 0.2 μL/min and immediately followed with the perfusion of 100 nmol/L CART peptide. Based on a statistical analysis of these data, we determined that for further trials, intraluminal and extraluminal routes of CART peptide administration were equivalent. Further, we concluded that 100 nmol/L was the optimum concentration against which to measure the effects of other compounds on the vascular response to CART peptide.
Specificity of Cocaine- and Amphetamine-Regulated Transcript Peptide-Mediated Vasoconstriction
We determined whether the observed CART peptide-evoked vasoconstriction affected the actions of other, presumably independent, vasoactive agents. Before CART peptide administration, each vessel was exposed to extraluminally applied acidic MOPS buffer (pH = 6.8), extraluminal sodium nitroprusside (SNP, 10 μmol/L), and extraluminal adenosine (ADO, 10 μmol/L). The dilation responses to these three stimuli were determined and the vessels were allowed to regain resting tone. Cocaine- and amphetamine-regulated transcript peptide (100 nmol/L) was then perfused intraluminally and while still in the presence of intraluminal CART peptide, acidic buffer, SNP, and ADO were applied extraluminally in turn and their respective effects recorded. Cocaine- and amphetamine-regulated transcript peptide was washed out for 20 mins with intraluminally perfused 1% albumin solution and the vessel was allowed to regain baseline diameter. After CART peptide washout, the vascular responses to extraluminally applied acidic buffer, SNP, and ADO were again determined.
Endothelial Light—Dye Injury
The endothelium dependence of CART peptide-evoked constriction of intracerebral arterioles was determined by the use of the endothelial light—dye injury (LDI) model adapted from Emerson and Segal (Emerson and Segal, 2000) for use within our preparation. Sodium fluoresceine (0.5%) was perfused intraluminally and the vessel exposed to light from a mercury lamp filtered to transmit between 450 and 490 nm. Preliminary experiments indicated that a single 60 secs period of activation was necessary for a selective disruption of endothelium function.
Before endothelial LDI, the arteriolar response to extraluminal acidic and alkaline buffer and intraluminal ATP (10 μmol/L) was assessed to obtain basline dilatation and constriction responses. The effects of extraluminal pH on cerebral vessel diameter are VSM-mediated events (West et al, 1992), while the dilation response to intraluminal ATP is endothelium-dependent (Janigro et al, 1997). The baseline response to intraluminally applied CART peptide (100 nmol/L) was determined. After 60 secs of LDI, vessel damage was evaluated with intraluminal and extraluminal propidium iodide (PI, 1 μmol/L), which illuminates only damaged cells (Geeraerts et al, 1991). Vessels were then perfused with intraluminal ATP (10 μmol/L) to confirm disruption of the endothelium. Extraluminal acidic and alkaline buffer was applied to assess vascular smooth muscle function after LDI. Once successful endothelial LDI had been confirmed, CART peptide (100 nmol/L) was applied intraluminally for 10 mins and the resulting vascular response measured.
Involvement of Endothelin System in Cocaine- and Amphetamine-Regulated Transcript Peptide Response
The involvement of endothelin receptors in the constriction response to CART peptide was assessed. The baseline vascular response to intraluminal CART peptide (1 nmol/L) was first determined, followed by a 20 mins washout with 1% albumin solution. Once the vessel had regained resting tone (±10% of baseline diameter), the dilation response to acidic buffer was observed to ensure continued health of the vessel. The vessel was then perfused extraluminally for 10 mins with its respective inhibitor of the endothelin system: the nonselective antagonist PD-145065 (10 μmol/L, PD), the ETA-specific antagonist BQ-123 (10 μmol/L), the ETB-specific antagonist BQ-788 (10 μmol/L), or the inhibitor of endothelin-converting enzyme (ECE) phosphoramidon (PHO, 100 nmol/L). After the vascular responses to the antagonists alone were measured, CART peptide and the endothelin antagonists or PHO were coadministered and changes in vessel diameter were recorded. To ensure that PD, BQ-123, BQ-788, and PHO were acting selectively, we determined their effect on vasoconstriction evoked by extraluminal perfusion of alkaline buffer (pH = 7.6).
Involvement of ETB Receptors in the Maintenance of Spontaneous Myogenic Tone
We characterized the observed role of endothelin ETB receptors in maintaining spontaneous myogenic tone in cerebral arterioles. A cumulative concentration—response curve for the ETB-specific antagonist BQ-788 was constructed by its stepwise extraluminal perfusion from 1 nmol/L to 10 μmol/L. BQ-788 was perfused for 10 mins at each concentration, after which the perfusion of MOPS buffer was restored and the vessel was allowed to regain baseline diameter. The effect of intraluminal versus extraluminal administration on the vascular response to BQ-788 was determined. BQ-788 was cumulatively and intraluminally perfused for 10 mins at 1 nmol/L, 100 nmol/L, and 10 μmol/L and the resulting changes in vascular diameter were noted. The vessel was flushed with 1% albumin saline solution and allowed to regain baseline diameter. Vessels that failed to achieve resting tone (±10% of baseline diameter) after exposure to BQ-788 were excluded from the study.
Drugs and Solutions
The composition of the MOPS-buffered saline solution was as follows (in mmol/L): 144.0 NaCl, 3.0 KCl, 2.5 CaCl2, 1.5 MgSO4, 5.0 glucose, 2.0 pyruvate, 0.02 EDTA, 2.0 MOPS, and 1.2 NaH2PO4. Bovine serum albumin, adenosine, SNP, ATP, sodium fluoresceine, and PI were purchased from Sigma (St Louis, MO, USA). Cocaine- and amphetamine-regulated transcript peptide (rat CART 55-102), PD-145065, BQ-123, and BQ-788 were purchased from American Peptide Company (Sunnyvale, CA, USA). ADO, SNP, PI, CART peptide, PD-145065, BQ-123, and BQ-788 were dissolved directly into the MOPS-buffered saline solution. ATP, PI, and CART peptide were dissolved directly into the 1% albumin saline solution.
Data Analysis
All data are expressed as means±standard deviation (s.d.). Only one vessel was studied from each animal. For comparison of responses to vasoactive agents, internal vessel diameters were normalized as a percentage of control diameters (diameters measured after the development of steady tone at 60 mm Hg of intraluminal pressure and 37°C). Data and statistical analyses were performed with GraphPad Prism® 4 (GraphPad Software Inc., San Diego, CA, USA). Comparisons between concentration—response curves were made using two-way analysis of variance (ANOVA) and mean values were compared using paired and nonpaired t-tests. A value of P<0.05 was considered significant.
Results
Effect of Cocaine- and Amphetamine-Regulated Transcript Peptide on Cerebral Arteriolar Diameter
The extraluminal perfusion of CART peptide evoked a long lasting, dose-dependent, and reversible constriction of cerebral penetrating arterioles (Figure 1A, n = 6). This response presented a considerably different profile than that observed with alkaline buffer perfusion. The onset of the constriction response was slower, taking 1 to 2 mins with CART peptide versus ~30 secs with alkaline buffer. Rate of response was also markedly smaller, CART peptide taking 5 to 7 mins to achieve maximal response compared with 1 to 2 mins for alkaline buffer. Vessel diameter was reduced to 93.1%±3.0%, 90.2%±5.0%, 87.4%±4.7%, 81.3%±4.4%, and 79.8%±1.4% of baseline diameter in response to 0.1 nmol/L, 1.0 nmol/L, 10 nmol/L, 100 nmol/L, 1 μmol/L CART peptide, respectively. This response had an EC50 of 1.2 × 10−9 mol/L. Intraluminal perfusion of 1 and 100 nmol/L CART peptide resulted in the constriction of arterioles to 89.2%±2.3% and 82.0%±3.4%, respectively (Figure 1B, n = 6). These values are not significantly different from those seen with extraluminally applied CART peptide of the same concentrations.
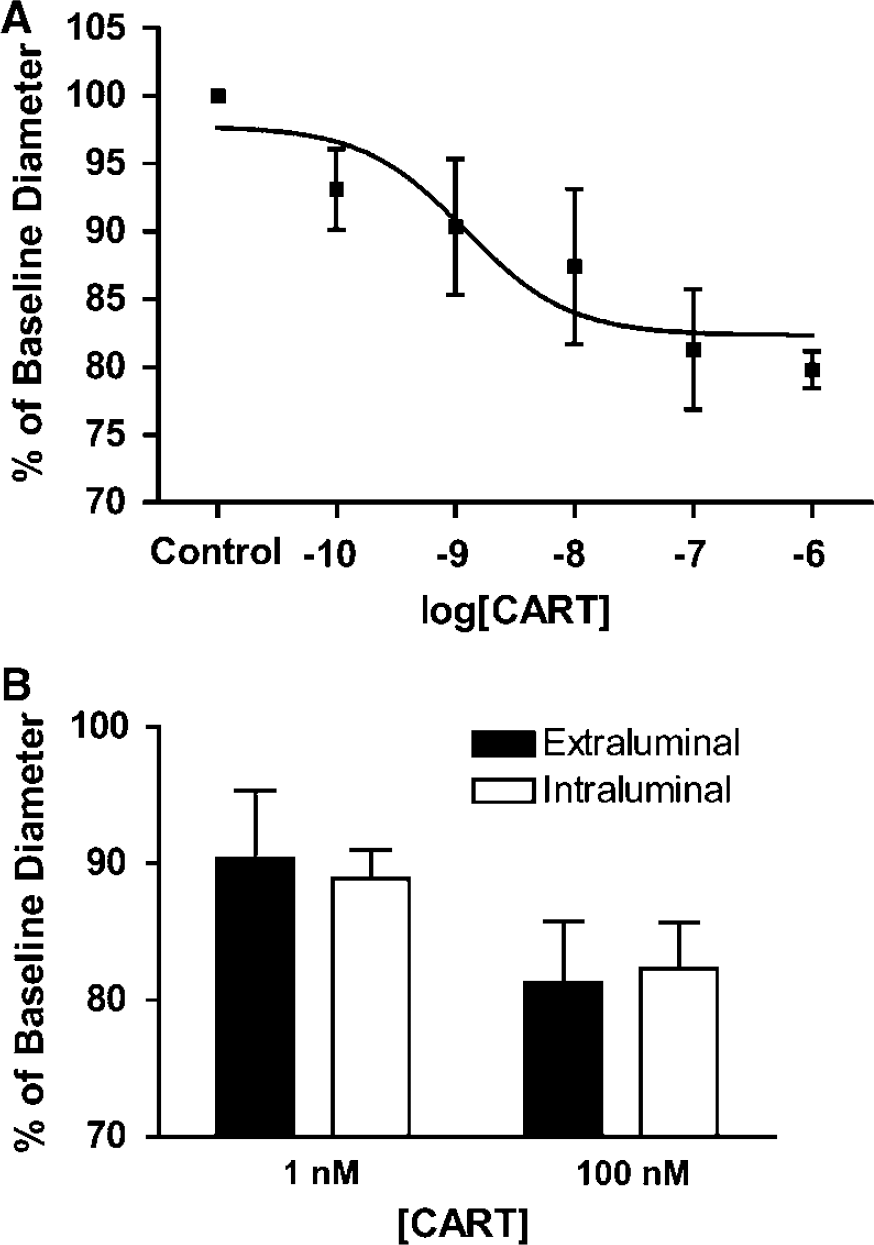
Changes in cerebral arteriolar diameter after cocaine- and amphetamine-regulated transcript (CART) peptide administration. (
Influence of Cocaine- and Amphetamine-Regulated Transcript Peptide on Vascular Response to Other Vasoactive Agents
The presence of CART peptide did not alter the dilation response to extraluminally applied acidic buffer, SNP (10 μmol/L), or ADO (10 μmol/L) (Figure 2, n = 4). The dilation response to acidic buffer before, during, and after the application of CART peptide was 125.7%±8.7%, 125.3%±9.8%, and 127.7%±9.4%, respectively. The dilation response to SNP before, during, and after the application of CART peptide was 120.1%±5.3%, 124.6%±12.9%, and 117.8%±1.9%, respectively. The dilation response to ADO before, during, and after the application of CART peptide was 119.0%±0.2%, 128.1%±7.1%, and 125.5%±3.0%, respectively.
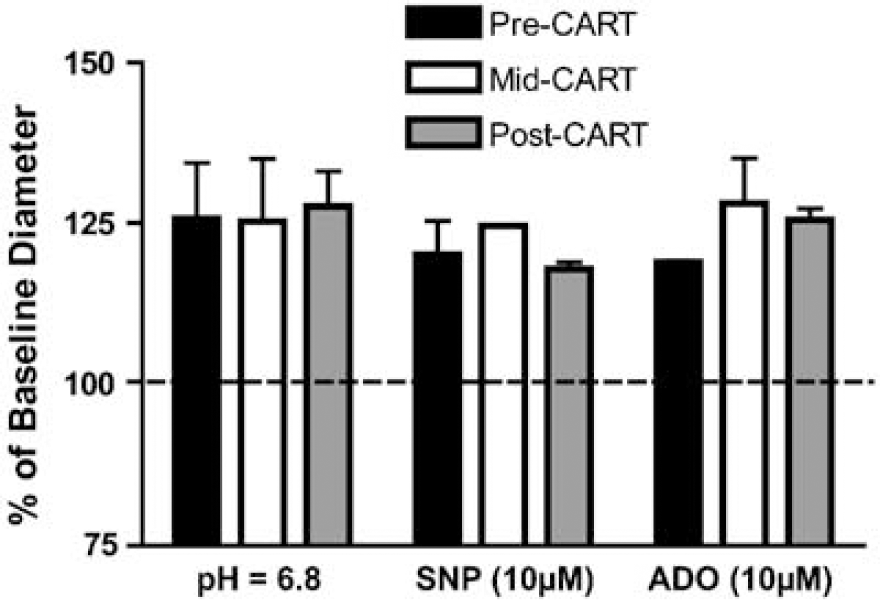
Effect of intraluminal cocaine- and amphetamine-regulated transcript (CART) peptide (100 nmol/L) on dilation response to extraluminally perfused acidic buffer (pH = 6.8), sodium nitroprusside (SNP, 10 μmol/L), and adenosine (ADO, 10 μmol/L). The dilation responses to acidic buffer, SNP, and ADO in the presence of CART peptide and after CART peptide washout were not significantly different than those preceding CART peptide administration.
Effect of Endothelial Light/Dye Injury on Cocaine- and Amphetamine-Regulated Transcript Peptide-Evoked Vasoconstriction
Selective disruption of the endothelium completely abolished the constriction response to 100 nmol/L CART peptide (Figure 3B, n = 5). Before LDI, CART peptide evoked an 81.7%±1.6% constriction from baseline tone. After endothelial disruption, the response was abolished (101.1%±3.7%, P<0.01). Selective disruption of the endothelium was visually assessed by means of PI staining, and pharmacologically by comparing the pre- and post-LDI vascular responses to intraluminal ATP (10 μmol/L) and extraluminal acidic and alkaline buffer. Sixty seconds of LDI caused the PI labeling of longitudinally oriented endothelial cells, while circumferential vascular smooth muscle cells remained unlabeled (Figure 3A). Before LDI, ATP, acidic buffer, and alkaline buffer evoked vascular responses to 138.7%±18.8%, 132.1%±5.3%, and 76.1%±10.6% of baseline diameter, respectively. After LDI, the dilation response to ATP had been completely abolished (103.3%±4.2%, P<0.01) while the responses to acidic and alkaline buffer remained unchanged (123.6%±6.3%, 81.7%±2.9%). Thus, LDI caused a selective effect on endothelial-dependent responses, but did not affect VSM-dependent dilation or contraction.
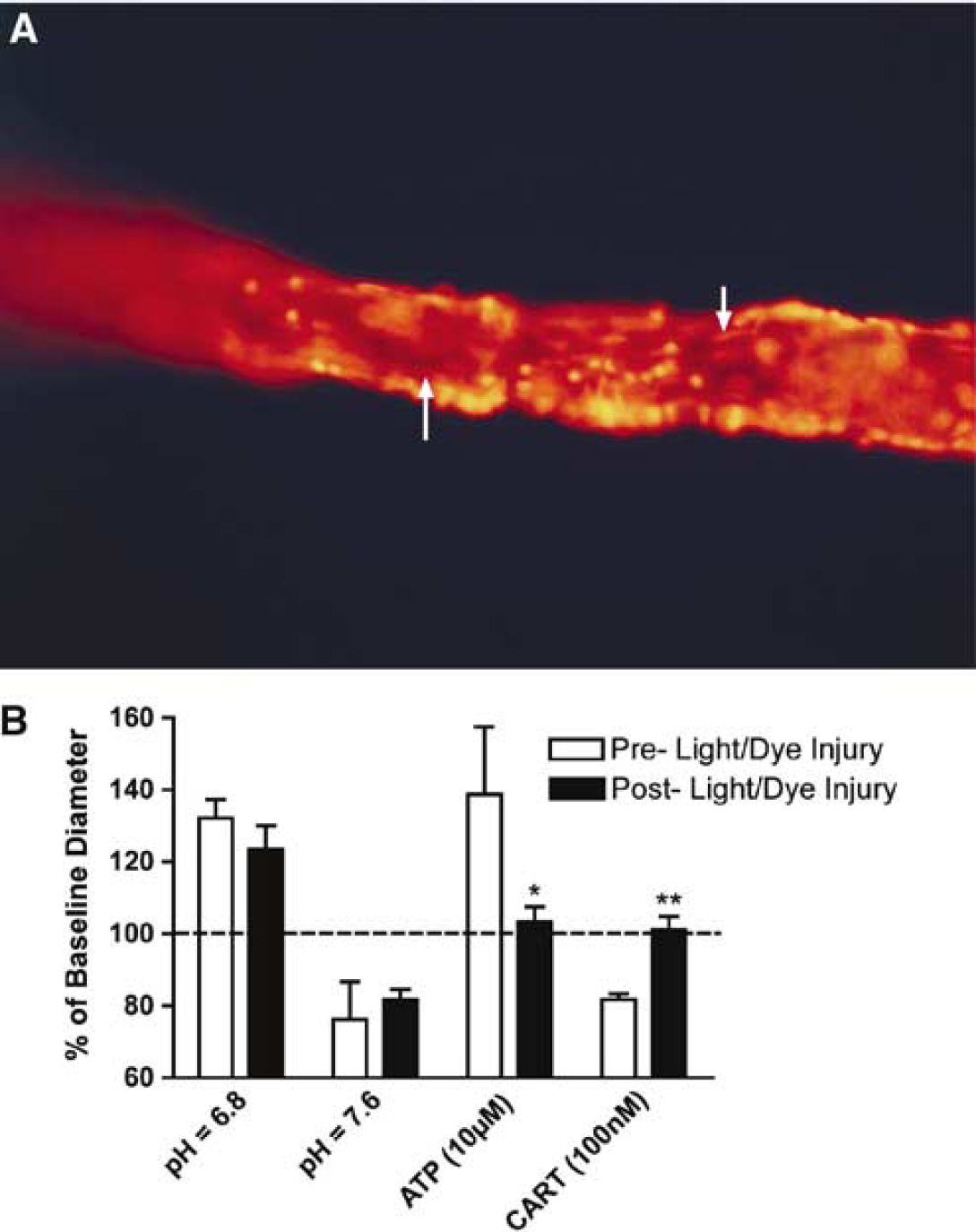
Disruption of endothelium by endothelial light/dye injury (LDI) blocked the constriction response to cocaine- and amphetamine-regulated transcript (CART) peptide (100 nmol/L). (
Effect of Endothelin Receptor Antagonists on Cocaine- and Amphetamine-Regulated Transcript Peptide-Evoked Vasoconstriction
Coadminsitration of the nonspecific endothelin receptor antagonist PD (10 μmol/L) or the ETA-specific antagonist BQ-123 (10 μmol/L) blocked the vasoconstrictor response to 100 nmol/L CART peptide from a baseline of 81.9%±1.3% to 99.6%±11.2% and 96.4%±1.2% (P<0.01), respectively (Figure 4A, n = 4). The ETB-specific antagonist BQ-788 (100 nmol/L) failed to affect the vascular response to CART peptide (82.7%±2.3%, n = 4). Neither PD nor BQ-123 altered baseline vessel diameter when administered alone (98.2%±9.1% and 104.5%±4.2%, respectively). BQ-788 alone, however, significantly (P<0.01) increased the myogenic tone of the isolated vessels, constricting them to 87.4%±7.1% of baseline diameter (Figure 4B, n = 4).
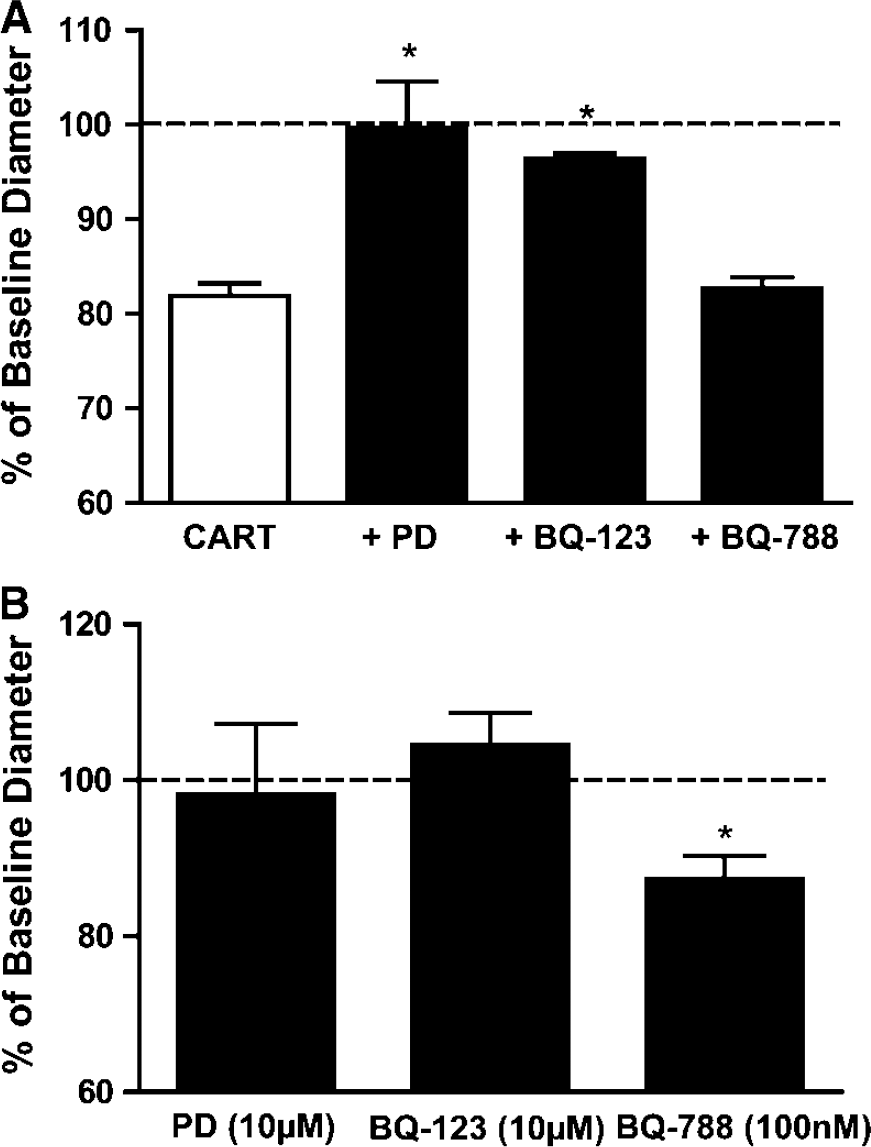
Endothelin ETA receptor antagonists block the constriction response to cocaine- and amphetamine-regulated transcript (CART)-peptide (100 nmol/L). (
Effect of Inhibitor of Endothelin Synthesis on Constriction Response to Cocaine- and Amphetamine-Regulated Transcript Peptide
The extraluminal coadministration of the endothelin-converting enzyme inhibitor phosphoramidon (100 nmol/L) attenuated the constriction response to intraluminal CART peptide (Figure 5, n = 5). In the absence of PHO, CART peptide constricted vessels to 81.9%±1.3% of baseline diameter. After PHO administration, the constriction response to CART peptide was blunted to 91.6%±8.1% of baseline diameter (P<0.01). Extraluminal administration of PHO alone caused a mild yet significant dilation of isolated vessels that ranged between 108.1%±2.9% at 10 nmol/L and 112.7%±16.0% at 100 μmol/L. At PHO concentrations of 1 μmol/L and above, a sudden and complete loss of spontaneous myogenic tone was intermittently observed (n = 3). Vessels that exhibited this response were excluded from further study.
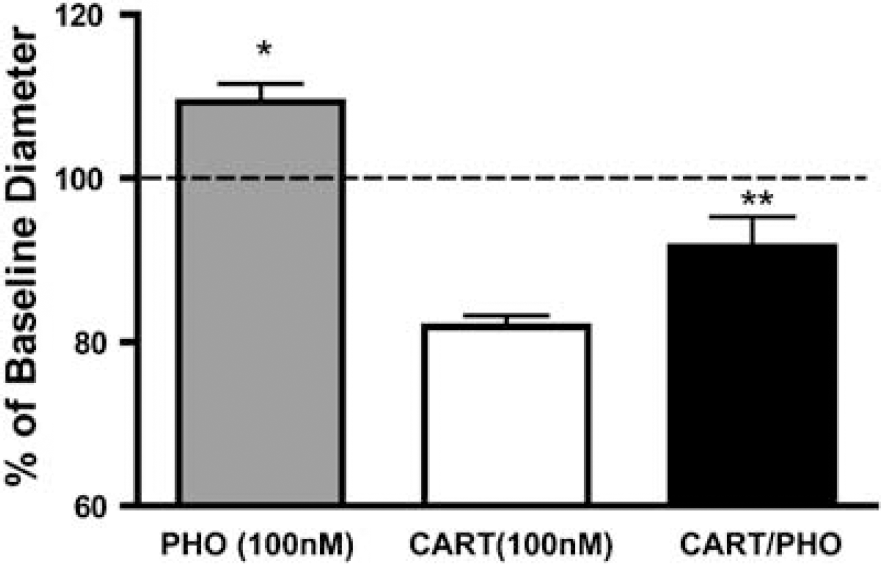
Attenuation of the cocaine- and amphetamine-regulated transcript (CART) peptide (100 nmol/L) constriction response by inhibitor of endothelin-converting enzyme. Extraluminal administration of the ECE inhibitor phosphoramidon (100 nmol/L) significantly dilated isolated cerebral arterioles from baseline diameter (*P<0.01, one sample t-test). Coadministration with extraluminal PHO markedly attenuated the constriction response to intraluminal CART peptide (**P<0.01, t-test CART versus CART/PHO).
Effect of Endothelin ETB-Specific Antagonist and on Spontaneous Myogenic Tone
The extraluminal perfusion of BQ-788 evoked a dose-dependent constriction of cerebral penetrating arterioles (Figure 6A, n = 6). Vessel diameter was reduced to 95.7%±3.2%, 91.7%±3.3%, 89.4%±1.6%, 85.2%±3.1% and 95.4%±2.2% of baseline diameter in response to 1 nmol/L, 10 nmol/L, 100 nmol/L, 1 μmol/L, and 10 μmol/L BQ-788, respectively. This response had an EC50 of 6.2 × 10−9 mol/L. Intraluminal perfusion of 1 nmol/L, 100 nmol/L, and 10 μmol/L BQ-788 was significantly more potent (P<0.01) than extraluminal perfusion, resulting in the constriction of arterioles to 91.4%±4.6%, 85.4%±3.6%, and 81.5%±4.9%, respectively (Figure 6B, n = 5).
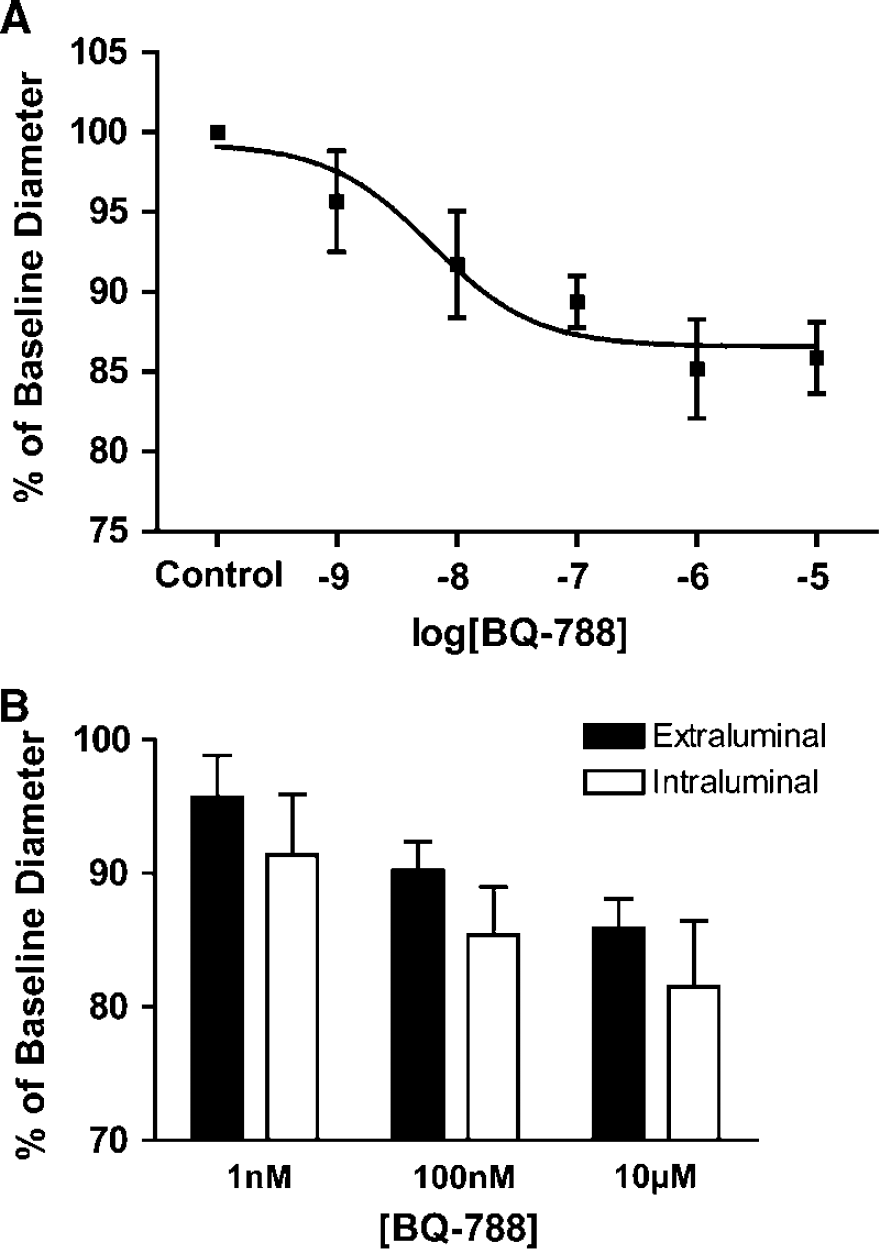
Changes in arteriolar diameter resulting from endothelin ETB receptor antagonist administration. (
Discussion
The present results represent the first demonstration of a potential role for CART peptide in the direct regulation of vascular tone. Cocaine- and amphetamine-regulated transcript caused a dose-dependent constriction of cerebral arterioles with potency in the nanomolar range. The constriction that was observed in response to either intraluminal or extraluminal administration of CART peptide was substantial (~80% of baseline tone, Figure 1). According to the fourth power relationship between vessel caliber and blood flow, this would amount to a 60% reduction in flow. The time course of the constriction response to CART peptide was protracted compared with the response to alkaline buffer (qualitative observation). To achieve maximal constriction, CART peptide required several minutes whereas alkaline buffer (pH = 7.6) required only seconds to achieve maximal effect. Similarly, the constrictor effect of CART-peptide was difficult to wash out, requiring 15 mins or more to return to baseline tone. Vessels treated with alkaline buffer returned to baseline diameter within minutes of the exchange of extraluminal perfusate.
To address initial concerns regarding the specificity of CART peptide's action, we observed its effect on the vascular responses to other vasoactive agents with known different mechanisms of dilations. The response of cerebral arterioles to changes in extracellular pH (i.e., acidic/alkaline buffer) involves the direct modulation of Ca2+ channels by H+ ions (West et al, 1992) on VSM cells. The vasodilator effect of NO is mediated through the activation soluble guanylyl cyclase while ADO evokes vasodilation via the membrane-bound adenosine A2A receptor (Ngai et al, 2001) and several second messenger pathways (Murphy et al, 2003; West et al, 2003). Cocaine- and amphetamine-regulated transcript peptide, either in its presence or after its washout, did not alter the dilation responses to these three agents (Figure 2). These observations argue against a nonspecific effect of CART peptide on the vessel or its contractile machinery.
Seeking to investigate its specific site of action within the microvasculature, we used the endothelial LDI model (Emerson and Segal, 2000) to determine the effect of selective endothelial disruption on CART peptide-evoked vasoconstriction. Successful disruption of the endothelium by the LDI was confirmed both by the PI staining of endothelial cells (Figure 3A) and by the abolition of the vascular response to intraluminal administration of the endothelium-dependent vasodilator ATP (Janigro et al, 1997). The specificity of the LDI injury was confirmed by the preservation of the smooth muscle-dependent (West et al, 1992) vascular responses to acidic (dilatation) and alkaline (constriction) buffer (Figure 3B). The elimination of the constriction response to CART peptide by selective endothelial LDI indicated that this effect was indeed endothelium-dependent.
Within the cerebral circulation, endothelin is known as an endothelium-derived modulator of vascular tone. It is synthesized from its precursor proendothelin by endothelin-converting enzyme and acts primarily through the smooth muscle-bound ETA receptor to cause potent and long-lasting vasoconstriction (Inoue et al, 1989; Yanagisawa et al, 1988). Nonspecific endothelin receptor and ETA-specific blockade by PD and BQ-123, respectively, blocked the constrictor effect of CART peptide (Figure 4A). Endothelin is also known to act as a vasodilator via the endothelium-bound ETB receptor (Kobari et al, 1994; Touzani et al, 1997). The ETB-specific antagonist BQ-788 did not alter the constriction response to CART peptide, but caused constriction independent of any agonist being present. The concentrations at which PD, BQ-123, and BQ-788 were administered were within the range for which they are known to act specifically (Ishikawa et al, 1994). These findings indicate that the CART peptide-evoked vasoconstriction is specifically mediated through the stimulation of the ETA receptor. Moreover, the attenuation of the constriction response to CART peptide by PHO, an inhibitor of ECE (Figure 5), suggests that CART peptide acts in the cerebral circulation to stimulate the de novo synthesis of bioactive endothelin from proendothelin. Newly synthesized endothelin then appears to activate smooth muscle-bound ETA receptors, evoking the dramatic vasoconstriction observed in the present study. The concentration of phosphoramidon used in the present study is within the range for which it is believed to act specifically to inhibit ECE activity within the cerebral circulation (Vatter et al, 2002; Vatter et al, 1998). In terms of the observed dynamics of the constriction response to CART peptide, it appears plausible that the protracted timeline may be a result of the involvement of the endothelin pathway—the stimulation of smooth muscle ETA receptors by endothelium-synthesized endothelin accounting for the longer time to peak response and prolonged constriction compared with that of perfused alkaline buffer.
The physiologic significance of the vasoactive properties of CART peptide is not immediately clear. As of yet, no CART peptide receptor has been identified and its mode of action remains to be elucidated. Cocaine- and amphetamine-regulated transcript peptide has been implicated in the mediation of the HPA axis and in the modulation of sympathetic outflow (Balkan et al, 2003; Koylu et al, 1997; Smith et al, 2004; Stanley et al, 2004; Vicentic et al, 2004; Vrang et al, 1999, 2000). Central administration of CART peptide increases systemic blood pressure (Matsumura et al, 2001) and blocks phenylephrine-induced bradycardia (Scruggs et al, 2003). Cocaine- and amphetamine-regulated transcript peptide has been shown to be released into the hypothalamic—pituitary portal circulation during NO-induced hypotension (Larsen et al, 2003). The role of CART peptide in promoting the stress response coupled with these observed cardiovascular effects suggests (albeit indirectly) that it may play a role in the autoregulation of blood flow within the brain under normal physiologic conditions. Cocaine- and amphetamine-regulated transcript peptide levels in the blood are also known to exhibit a glucocorticoid-mediated diurnal rhythm, varying within the pico-molar range (Vicentic et al, 2004). No studies have observed vasoactive levels of CART peptide in the serum or CSF, nor have any measured CART mRNA or CART peptide in the vasculature. Thus, it is unknown whether local CART peptide levels in the brain reach higher vasoactive levels in vivo. Future studies will seek to investigate this possibility and to elucidate the physiologic relevance of direct vascular effects of CART in the brain.
In contrast, the pathophysiologic relevance of CART peptide's role as a vasoconstrictor is considerably more provocative. Cocaine- and amphetamine-regulated transcript was initially identified as a transcript upregulated by psychostimulant use (Douglass et al, 1995). Despite more recent evidence questioning the role of amphetamines in the regulation of CART expression (Vrang et al, 2002), cocaine use continues to be implicated in the regulation of brain CART levels (Albertson et al, 2004; Hurd et al, 1999). Cocaine is a known vasoconstrictor in the cerebral circulation (Kaufman et al, 1998; Madden et al, 1995; Salom et al, 1996) and is associated with cerebrovasospasm (Fandino et al, 2003; He et al, 1994; Kaufman et al, 1998). Cocaine induces the release of endothelin from the endothelium (Hendricks-Munoz et al, 1996; Wilbert-Lampen et al, 1998) and endothelin receptor blockade attenuates vasospasm after acute cocaine administration and subarachnoid hemorrhage (Fandino et al, 2003; Kikkawa et al, 1999; Zuccarello et al, 1998b). Thus, our findings that CART peptide is a potent endothelin-mediated vasoconstrictor within the cerebral circulation is in agreement with the literature and suggests that CART peptide may be involved in the pathogenesis of cocaine- and amphetamine-related stroke. Future studies will seek to investigate this hypothesis.
In addition to CART peptide's vasoactive role in the cerebral circulation, the findings that PHO, an inhibitor of ECE, and the endothelin ETB-specific antagonist BQ-788 alter resting myogenic tone in the cerebral microcirculation are novel. When perfused extraluminally, PHO alone dilated arterioles by ~10% while BQ-788 evoked a dose-dependent constriction of isolated penetrating cerebral arterioles (Figure 6A). The dilator effect of PHO is consistent with the inhibition of a tonic constrictor influence of endothelin in maintaining resting tone within the cerebral circulation. This effect is likely mediated via the smooth-muscle-bound ETA receptor, although a role of smooth-muscle-bound ETB receptors (contributing to vasoconstriction) cannot be ruled out. The absence of an effect by the ETA-specific antagonist BQ-123 may be because of an incomplete blockade of these receptors at 10 μmol/L—this dose being sufficient to abolish the CART peptide-evoked constriction yet insufficient to antagonize low-level tonic stimulation. The constrictor effect of BQ-788 was more pronounced during intraluminal perfusion compared with extraluminal perfusion (Figure 6B), suggesting that access to the endothelial cell layer influenced this response. Within the cerebral vasculature, ETB receptors bound to the endothelium are thought to mediate vasodilation via a NO-mediated pathway (Kitazono et al, 1995; Schilling et al, 1995; Szok et al, 2001) whereas smooth-muscle-bound ETB receptors appear to mediate a vasoconstrictor effect (Zuccarello et al, 1998a). Thus, the observed constrictor effect of BQ-788 is consistent with the blockade of endothelial ETB receptors and the associated NO-mediated vasodilatory influence on resting vascular tone. The roles of ETB receptors and ECE in modulating basal vascular tone in cerebral penetrating arterioles noted in the present study are different from previous studies in the cerebral circulation. Within the basilar artery, both in vitro and in vivo, no such effect of ETB antagonism (Schilling et al, 1995; Zuccarello et al, 1998a, b) or ECE inhibition (Vatter et al, 1998) on baseline vascular tone has previously been oberved. A possible explanation for the differences are likely attributable to the vasoactive heterogeneity along the vascular network. Arteries are composed of several vascular smooth muscle layers and a single endothelial layer, while the arterioles observed in the present study consist of a single layer of each—architecture reflective of their varying roles in the local regulation of blood flow. Such differences coupled with the differential expression of ETA and ETB receptors within the vascular smooth muscle and endothelium, respectively, may serve to reconcile the present observations to previous reports in larger vessels.
Our findings are in further agreement with an emerging body of evidence supporting the interdependent contributions of the endothelin and NO signaling pathways in the maintenance of vasomotor tone in the peripheral circulation (Cardillo et al, 2000; Hocher et al, 2004; Verhaar et al, 1998; Wiley and Davenport, 2001). It has recently been posited that endothelin and NO contribute interrelated vasoconstrictor and vasodilator effects in the regulation of tone within the cerebral microcirculation (Chen et al, 2003). The potential involvement of the endothelin system, specifically the ETB receptor, and its relationship to the NO system in the regulation of cerebral blood flow deserves further study.
In the present study, we have evaluated the direct vasoactive effects of CART peptide on isolated, pressurized, and perfused intraparenchymal arterioles. To the best of our knowledge, this is the first assessment of CART peptide's direct effects within the vasculature, cerebral or otherwise. We found that CART peptide evoked a long-lasting and dose-dependent constriction of cerebral arterioles—an effect that was not dependent on intraluminal versus extraluminal administration. This constriction appears to be a specific effect in that the responses of the vessels to other vasoactive agents remained intact even in the presence of CART peptide. Based on the sensitivity of the CART peptide-evoked vasoconstriction to endothelial disruption, we conclude that the mechanism of action is endothelium-dependent. Furthermore, the attenuation of the constriction response to CART peptide by the inhibition of ECE phosphoramidon, the nonspecific endothelin antagonist PD, and the ETA-specific antagonist BQ-123, but not the ETB-specific antagonist BQ-788 suggests that this response is mediated by the de novo synthesis of endothelin and the subsequent stimulation of ETA, but not ETB, receptors.
Footnotes
Acknowledgements
The authors thank Addie Peretz for laboratory support and Beth Fee for administrative support.
