Abstract
In a prospective study of the natural development of total cerebral blood flow volume (CBFV), the common, external and internal carotid and vertebral arteries were examined in 94 healthy children and adolescents between 3 and 18 years of age (sex and age evenly distributed) using a 7.0-MHz transducer of a computed sonography system. Intravascular flow volumes were calculated with the product of angle-corrected time-averaged flow velocity and the cross-sectional area of the vessel. CBFV was determined as the sum of flow volumes in the internal carotid and vertebral arteries of both sides. CBFV increased significantly between 3 and 6.5 years of age (from 687 ± 85 to 896 ± 110 ml/min; age correlation, p ≤ 0.01) and declined thereafter (p ≤ 0.001) to a constant level of ∼700 ml/min at 15 years of age. There was no difference in CBFV between sexes. The proportion of bilateral vertebral artery flow volume in total CBFV decreased significantly between the ages of 3 and 18 years (p ≤ 0.001). As the flow volumes of the external carotid arteries increased markedly from childhood to adulthood, flow volumes of the common carotid arteries were not representative of CBFV. Intrasession test-retest correlation of CBFV was high (r = 0.89, p ≤ 0.0001). Reference data for the childhood years presented here and previously described results from healthy adults allow us to outline the natural evolution of CBFV in humans. The reliability of the method has already been demonstrated. Thus, it may now be introduced into clinical application.
Keywords
Little information is available on the development of cerebral blood flow (CBF, measured in ml/100 g brain tissue/min), in childhood, since measurement techniques used until now have been based on invasive procedures and/or use of radionuclides (Kennedy and Sokoloff, 1957; Chiron et al., 1992). We recently reported on the noninvasive determination of total cerebral blood flow volume (CBFV, measured in ml/min) by color duplex sonography of the internal carotid (ICA) and vertebral arteries (VA) in healthy adults (Schöning et al., 1994). In the present study, we apply this method to children and adolescents in order to trace the natural development of CBFV from childhood to adulthood.
METHODS
Ninety-four healthy children and adolescents (45 girls, 49 boys) who had no history of cerebral or cerebrovascular diseases participated in a prospective study on color duplex sonography of the intra- and extracranial arteries. In each age group of 3–18 years, we planned to examine at least three girls and three boys. This was possible with one exception; in the group of 3-year-olds there were only two girls and three boys. As the test persons had to lie quietly for ∼1 h, children <3 years of age were excluded when the study was designed. The mean age of the subjects was 10.5 ± 4.3 years. Written informed consent was obtained prior to the examination from the parents of all of the children and from all test persons >10 years of age. The study was approved by the Ethical Committee of the University of Tübingen.
During the first 30 min, transcranial color duplex sonography of the basal cerebral arteries was performed while the subjects were lying in a supine position (results of transcranial examination are not included in this report). Subsequently, the extracranial arteries were explored, i.e., the common (CCA) external (ECA) and internal carotid (ICA) arteries and VA.
A 7.0 MHz linear array transducer of a computed sonography system (Acuson 128 XP10, Mountain View, CA, U.S.A.) was used. The subjects continued to lie supine with their heads slightly elevated and turned to the contralateral side by ∼10° during examination of the CCA and VA, and by ∼25–40° during examination of the ICA and ECA. Measurements were taken at a location 1.5–2.0 cm below the carotid bulb in the CCA and 1.5–2.0 cm above the bifurcation in the ICA and ECA. The VA was examined most frequently in the C4–C5 intertransverse segment.
The examination technique has been described in detail previously (Schöning et al., 1994). Briefly, the luminal diameter of the vessels, d, was measured on a magnified, high-resolution B-mode image between the bright internal layers of the parallel vessel walls at the exact site of color duplex measurement and perpendicular to the artery's longitudinal axis. (For technical reasons, the second diameter of the vessel could not be measured, as it was very difficult to properly apply a linear transducer of 38 mm aperture size after turning it for exact 90° behind the mandibular angle. In this way, it could not be guaranteed that an accurate cross-section of the vessel, perpendicular to its course, was obtained at the very same point as in the longitudinal axis. Therefore, only one luminal diameter was taken, and the area of the circular vessel was calculated with d/2 × π) The calipers could be adjusted in 0.1 mm increments. For each vessel, the mean of duplicate luminal measurements was evaluated.
The color duplex sonography procedure consisted of color Doppler imaging of a long vessel segment plus a time-frequency analysis of the intravascular flow in each artery. At a site of undisturbed color Doppler flow, the pulsed Doppler sample volume was positioned over the entire width of the vessel. The electronic steering feature of the linear probe was used to keep the angle of insonation as low as possible, most frequently at ∼60°. A spectral curve was then recorded with the color Doppler image “frozen.” The Doppler signal was evaluated only if it was stable over at least 5 s and did not contain bidirectional frequencies (which would indicate disturbed flow). The angle of insonation was adjusted in 1° increments by aligning an angle indicator parallel to the walls of the vessels and along the intravascular color Doppler stream. Finally, angle-corrected time-averaged flow velocity (TAV), i.e., the integral of the mean frequencies over time, was measured over at least three complete cardiac cycles.
The intravascular flow volume, FV, can be determined by calculating the product of the angle-corrected TAV and the cross-sectional area of the circular vessel, A, with the formula FV = TAV × A = TAV × [(d/2)2 × π]. Each measurement was recorded with a video printer. Flow volumes of the respective bilateral pairs of arteries were recorded as “bilateral flow volumes.” CBFV was determined by adding the bilateral ICA and VA flow volumes, i.e., the sum of flow passing through the anterior and posterior brain circulation.
Examination of all the extracranial arteries required ∼20 min. The ECA and CCA were not examined in the first five and eight children of the study, respectively. In the last 53 subjects of the whole group (mean age 10.4 ± 4.6 years), an immediate retest examination of CBFV was performed by repeating flow volume measurements in the ICA and VA on both sides.
Statistical analysis was done using the SAS-program (version 6.04, SAS-Institute, Cary, NC, U.S.A.). All values are shown as mean ± SD. Differences between the various age groups were evaluated with the Wilcoxon test and differences between the sexes were tested with Student's t-test. The correlation of flow volume to age was determined using the Spearman correlation coefficient, while test-retest correlations were tested with the Pearson correlation coefficient. The coefficient of variance (CV) was determined by comparing SD of the test differences as percentages of the average in both series. The level of statistical significance was set at p ≤ 0.01 for all tests.
RESULTS
Examination of the extracranial arteries could be completed in all of the children except for one 3-year-old girl who interrupted the procedure.
There was no significant difference between girls and boys in CBFV (760 ± 129 and 781 ± 112 ml/min, respectively), or in bilateral ICA (556 ± 98 and 568 ± 87 ml/min, respectively) and VA flow volumes (203 ± 50 and 213 ± 51 ml/min, respectively). CBFV and bilateral flow volumes in the ICA and VA were significantly higher in children <10 years of age than in older children and adults (Table 1).
Bilateral flow volumes in extracranial arteries and total CBFV in different age groups
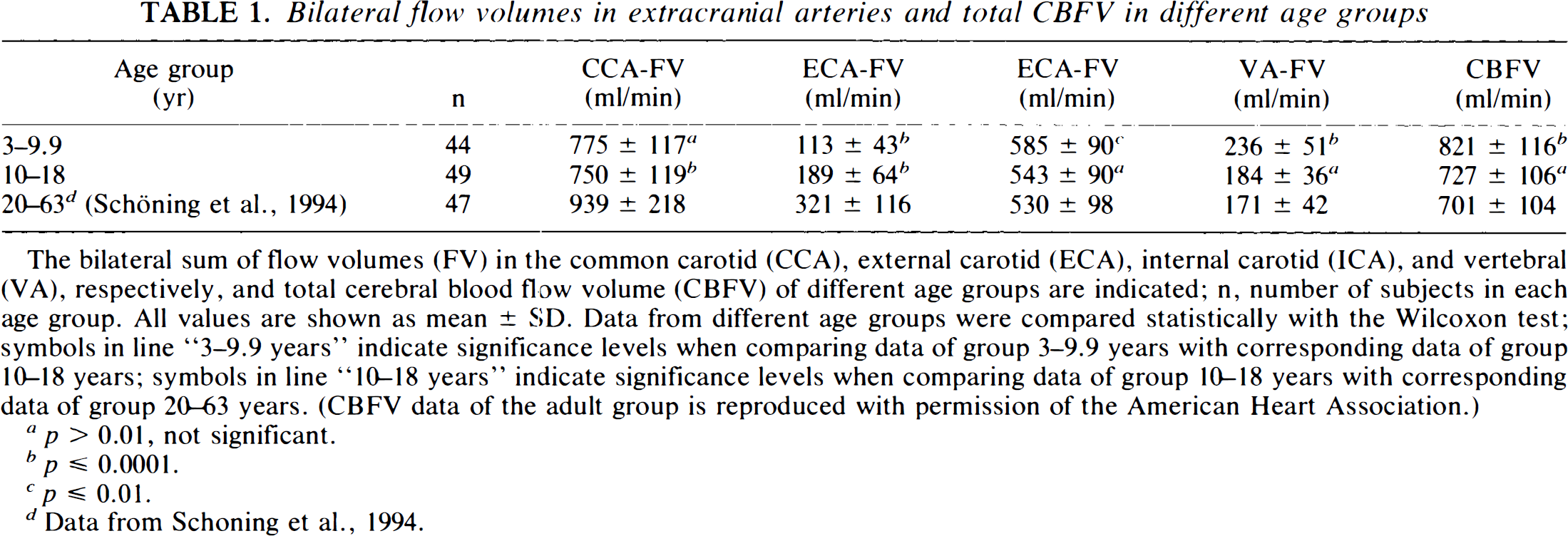
The bilateral sum of flow volumes (FV) in the common carotid (CCA), external carotid (ECA), internal carotid (ICA), and vertebral (VA), respectively, and total cerebral blood flow volume (CBFV) of different age groups are indicated; n, number of subjects in each age group. All values are shown as mean ± SD. Data from different age groups were compared statistically with the Wilcoxon test; symbols in line “3–9.9 years” indicate significance levels when comparing data of group 3–9.9 years with corresponding data of group 10–18 years; symbols in line “10–18 years” indicate significance levels when comparing data of group 10–18 years with corresponding data of group 20–63 years. (CBFV data of the adult group is reproduced with permission of the American Heart Association.)
p > 0.01, not significant.
p ≤ 0.0001.
p ≤ 0.01.
Data from Schoning et al., 1994.
The age dependence of bilateral flow volumes in the extracranial arteries and CBFV is shown in Figs. 1A–D and 2A–B. Mean CBFV increased markedly from 687 ± 85 ml/min at 3 years of age to a peak of 896 ± 110 ml/min at 6.5 years of age, after which it declined slowly to reach a constant level of ∼700 ml/min at 15 years of age (Spearman correlation coefficient for ages 3–6.5 years, r = 0.59, p ≤ 0.01, n = 23 subjects; for ages 6.5–18 years, r = −0.38, p ≤ 0.001, n = 70). From age 3 to 6.5 years, there was a significant rise in bilateral ICA flow volumes (r = 0.59, p ≤ 0.01), while the subsequent slight decrease until the age of 18 years was not significant (r = −0.22). After a slight increase in bilateral VA flow volumes between the ages of 3 and 6.5 years (r = 0.34; p, not significant), there was a marked decrease until the age of 18 years (r = −0.49; p ≤ 0.0001).
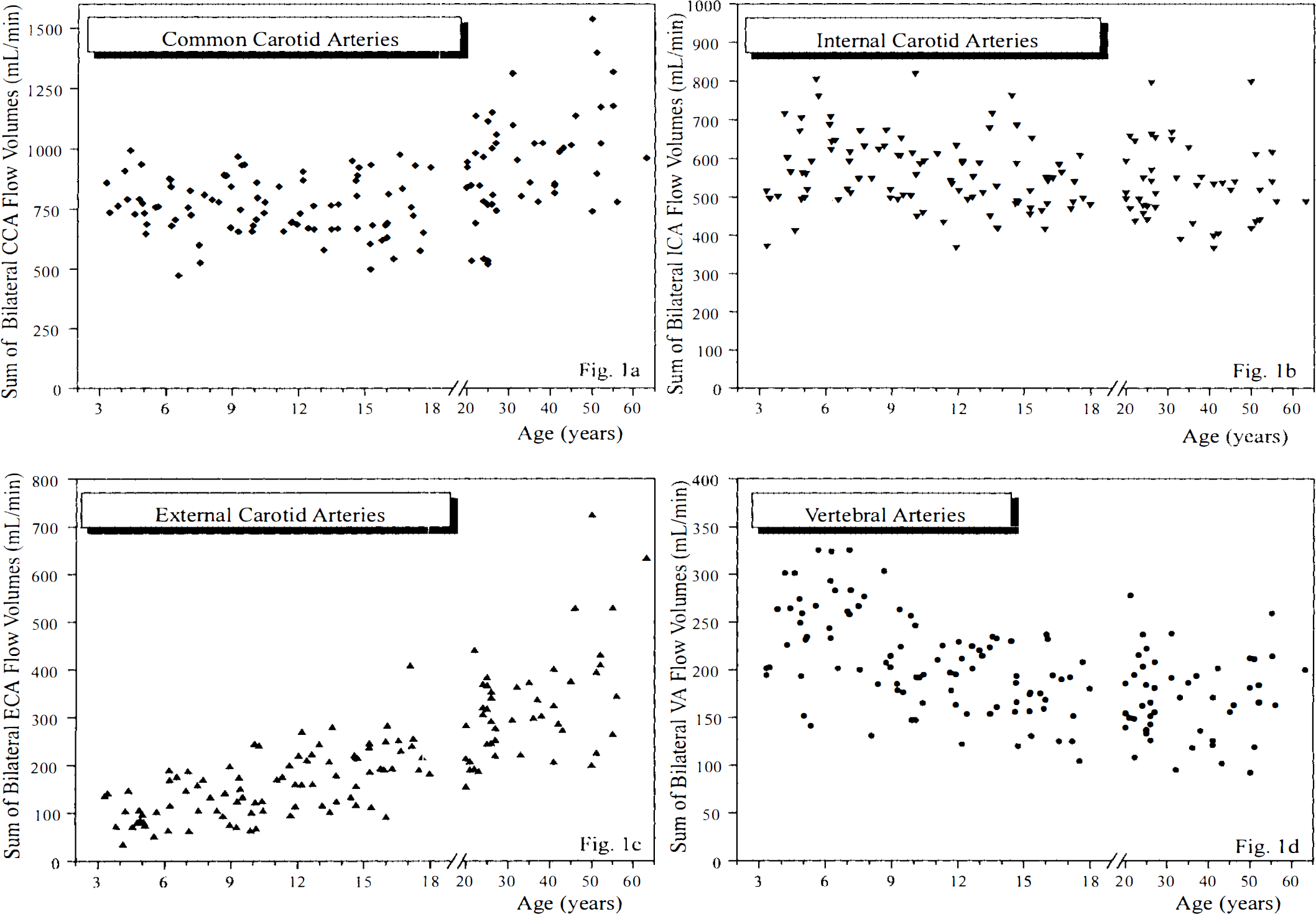
Development of flow volumes in the common, internal and external carotid arteries and vertebral arteries from childhood to adulthood. Scatterplots show the sum of bilateral flow volumes in the common carotid arteries (CCA)
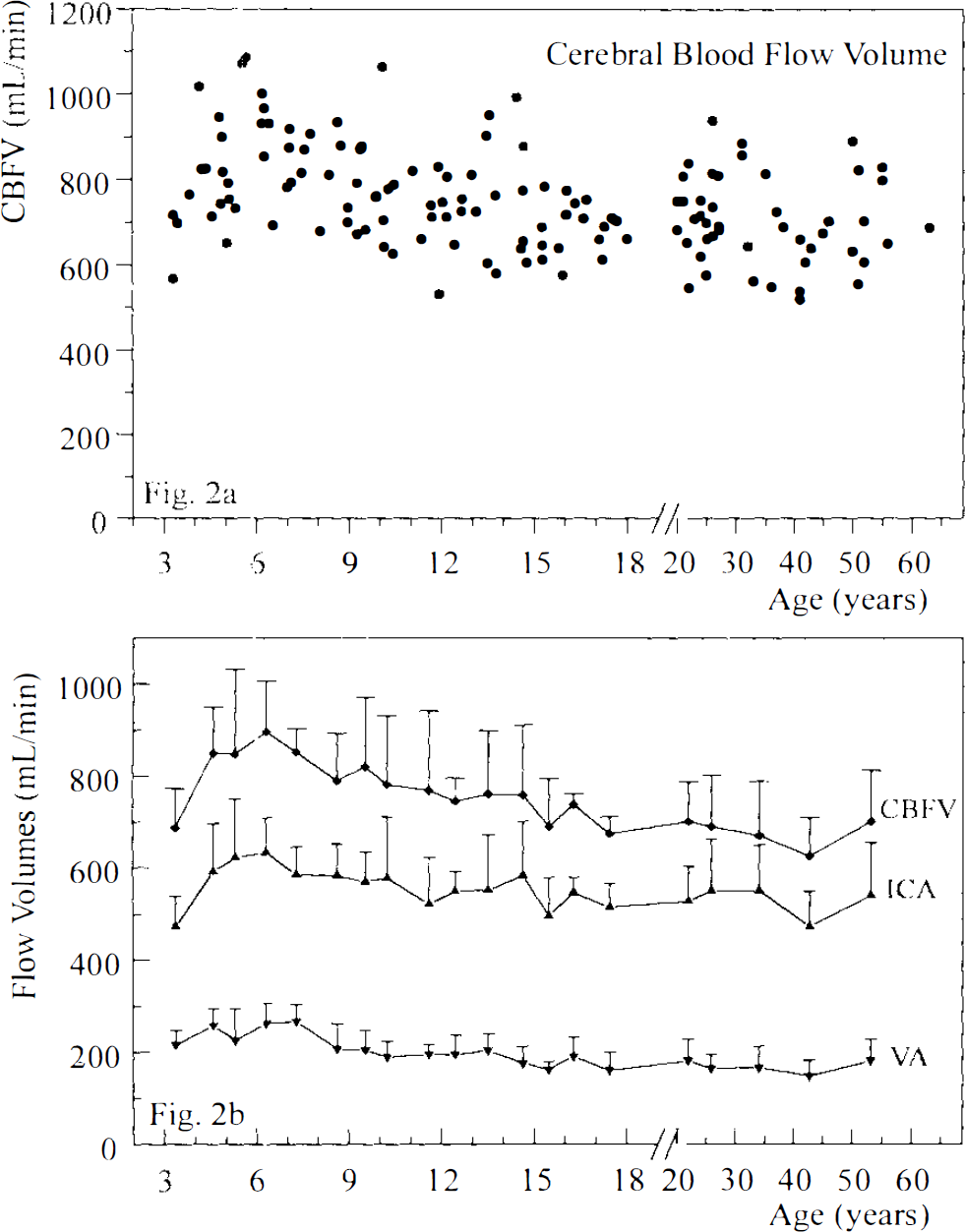
Development of total cerebral blood flow volume from childhood to adulthood. Scatterplot
Bilateral ECA flow volumes increased continuously from early childhood until adulthood (age groups 3–18 years, r = 0.62, p = ≤ 0.0001). CCA flow volumes remained constant throughout childhood.
Although the absolute reduction in the bilateral ICA and VA flow volumes from childhood to adulthood was equal (see Table 1), the proportion of VA flow volume in total CBFV decreased from 31% at the age of 4 years to 24% at the age of 18 years (ages 3–18 years: r = −0.38, p ≤ 0.001) (Fig. 3).
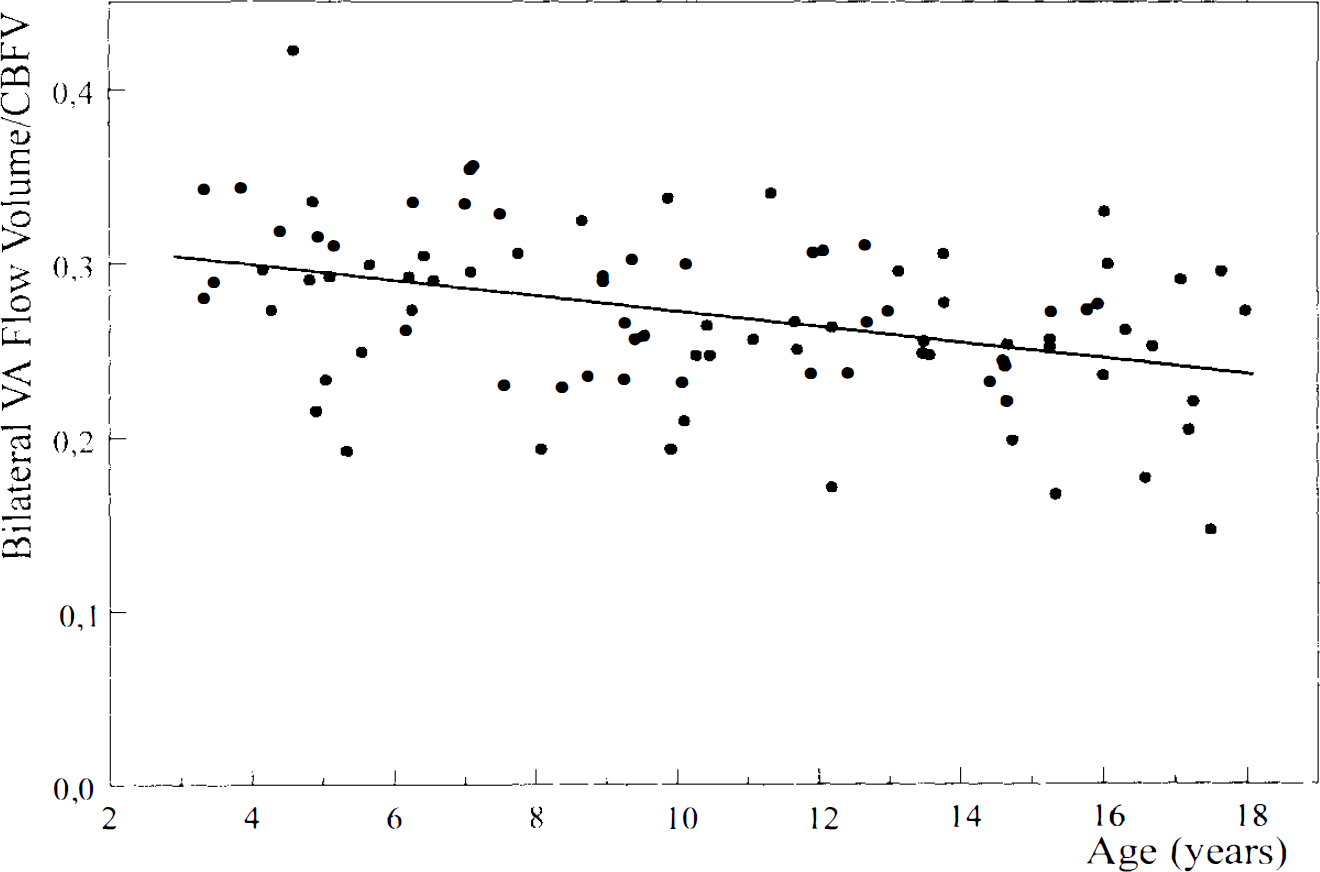
Contribution of the posterior cerebral circulation to total CBFV from childhood to adulthood. Scatterplot shows the proportion of the sum of bilateral vertebral artery flow volumes to total CBFV in each subject 3–18 years of age.
Retest measurements of CBFV differed only minimally (2 ± 58 ml/min, not significant) from the first examination. Test-retest correlation was high (Pearson correlation coefficient, r = 0.89. p ≤ 0.0001, n = 53); CV = 7.3%.
DISCUSSION
Since the first study published by Kennedy and Sokoloff (1957) ∼40 years ago, no prospective investigation of normal CBF in childhood has been performed. For ethical reasons, it is not possible to subject healthy children to invasive procedures based on the cannulation of the jugular bulb and arterial blood sampling or to expose them to radioactive isotopes. This is why “normal” data on CBF in childhood are based on a total of < 150 examination nations performed in patients with neurological problems, which were found, in retrospect, to be transient and not accompanied by cerebral lesions (Tzourio et al., 1988; Ogawa et al., 1989; Chiron et al., 1992; Kuroda et al., 1993).
Color duplex flowmetry of the extracranial arteries is a noninvasive and innocuous method of estimating CBFV. Reference data on adult CBFV were established in a first prospective study on 48 healthy subjects of 20–63 years of age (Schöning et al., 1994). The results were compatible with CBF data from studies using nitrous oxide or the [133Xe] single photon emission computed tomography (SPECT) technique (Kety and Schmidt, 1948; Shirahata et al., 1985; Waldemar et al., 1991). However, Doppler flowmetry is an operator-bound method and, thus, sensitive even to minor errors (Smith 1984; Gill 1985; Hoskins 1990): deviations of 0.1 mm in ICA diameter measurement result in errors in flow volume calculations of ∼4% (Schöning et al., 1994). The reliability of this technique, therefore, still had to be confirmed.
In the present study, a high reproducibility in CBFV measurements could be demonstrated by intrasession retest measurements performed by the same examiner. In a recent intra- and interobserver test-retest investigation on CBFV in 32 healthy children and adults, results from two “blinded” examiners were strikingly similar (Pearson correlation coefficient, r = 0.90, p ≤ 0.0001; mean percent interobserver difference, 1.7 ± 9.2%) (Schöning and Scheel, 1996). These data were similar to those of a reproducibility study on mean CBF by H215O positron emission tomography (PET) performed in 25 normal male adults by Matthew et al. (1993) (r = 0.83 ;p ≤ 0.001; mean percent test-retest difference, 2.3 ± 8.7%). We can conclude that the reliability of color duplex flowmetry of CBFV is comparable to that of mean CBF measurement by H215O-PET.
Using the nitrous oxide method, Kennedy and Sokoloff (1957) observed a mean CBF in nine children, aged 3–11 years, that was ∼1.8 times as high as that in a control group of healthy students (106 versus 60 ml/min/100 g). A steady decline in mean CBF from birth to adulthood was described by several authors using 133Xe methods in patient groups (Ogawa et al., 1989; Kuroda et al., 1993). Chiron et al. (1992) observed a biphasic development of CBF using the [133Xe]-SPECT technique in 42 patients without cerebral lesions, aged 2 days to 19 years. Mean CBF started at ∼50 ml/100 g/min at birth, rose to a maximum of 70 ml/100 g/min between 4 and 8 years, and then decreased slowly to reach the adult CBF level of ∼50 ml/min/100 g at 15–19 years of age. In the same study, a characteristic developmental pattern of regional CBF in different brain regions was observed that was very similar to that of the local glucose metabolism rate by Chugani et al. (1987) with PET.
An exact comparison of CBFV (measured in ml/min) and CBF (measured in ml/min/100 g brain tissue) is not yet possible because individual brain weight cannot be reliably measured in vivo. According to anatomical studies, human brain weight increases from 400 g at birth to 1,000 g during the second year of life, reaching 92% of the mean adult brain weight by the age of 6 years, with an average maximum of 1,340 g in women and 1,450 g in men at ∼18 years of age (Dobbingand Sands, 1973; Dekaban and Sadowsky, 1978; Ho et al., 1980). By dividing the present CBFV data by the mean brain weight for different age groups [as reported by Dekaban and Sadowsky (1978)], mean CBF would work out to ∼60, 70, and 50 ml/min/100 g at the ages of 3, 6, and 18 years, respectively. Hence, parallel development of CBF and CBFV is shown in the study done by Chiron et al. (1992) and the present investigation.
In most studies performed in normal adults, the mean CBF was higher in women than in men (Gur et al., 1982; Davis et al., 1983; Shaw et al., 1984; Mathew et al., 1986; Rootwelt et al., 1986; Gur et al., 1987; Rodriguez et al., 1988), while some others found no sex difference (Melamed et al., 1980; Shirahata et al., 1985; Waldemar et al., 1991). Corresponding data on CBF in girls and boys are still missing. The higher CBF rate in women has been attributed, in part, to lower hematocrit values before menopause (Shaw et al., 1984; Rootwelt et al., 1986). Hatazawa et al. (1987) observed that the 9% higher local glucose metabolism rate compensated for the 9% smaller brain size in women compared with men. Rodriguez et al. (1988), therefore, hypothesized that global CBFV—when measured in ml/min and not ml/100 g brain tissue/min—may be perfectly equal in both sexes. In fact, we could find no difference in total CBFV between boys and girls in the present study nor between men and women in an earlier investigation (Schöning et al., 1994). Since most anatomical studies on brain weight reported it to be ∼10% higher in men than in women at any particular age (Dekaban and Sadowsky, 1978; Ho et al., 1980; Marshall 1892; Muehlmann 1927; Pakkenberg and Voigt, 1964), our results indirectly confirm the findings of a higher flow rate per weight of brain tissue in women versus men.
Unlike the SPECT and PET methods, color duplex flowmetry of CBFV cannot be used to determine regional CBF in specific parts of the brain. However, with this method, the relative contribution of the anterior and posterior brain circulation to total CBFV may now be measured, for the first time, in humans. In the macaque monkey, ∼30% of the total cerebral blood passes through the vertebrobasilar system (Dumke and Schmidt, 1943), while in humans, that proportion has been estimated to be 20% (Edvinsson et al., 1993). We could observe a continuous reduction of bilateral VA flow volumes in relation to CBFV from early childhood (31%) to adulthood (24%). Even in the study by Chiron et al. (1992), regional CBF of the cerebellar hemispheres was found to be at its greatest between 3 and 8 years of age. These findings may point to a prominent role of the brain regions supplied by the posterior circulation, e.g., the cerebellum and the visual cortex, in early human development.
In most previous studies on Doppler flowmetry of the extracranial arteries, global CBFV has been estimated using the sum of the flow volumes in the CCAs (Keller et al., 1976; Fitzgerald et al., 1982; Payen et al., 1982; Uematsu et al., 1983; Müller et al., 1984; Müller et al., 1987; Wada et al., 1991). Yet, we can show that CCA flow volumes are not representative of CBFV, since development of CCA flow volumes and CBFV from early childhood to adulthood are fundamentally different. Up to the sixth decade of life a continuously increasing proportion of CCA flow feeds the EC A territory. The almost threefold increase in ECA flow volumes from early childhood to adulthood has not yet been described, and we do not know the underlying physiological mechanism. Biphasic development of ICA flow volume, which is nearly parallel to that of CBFV, and the changing pattern of VA flow clearly show that flowmetry of both of the real brain-feeding arteries is imperative for accurate CBFV measurement throughout life.
CBF is one of the most important parameters to know in the critically ill patient, and monitoring of CBF is essential in the intensive care unit. To date, CBF could be only estimated indirectly by measuring flow velocities in the basal cerebral arteries using transcranial Doppler and color duplex techniques (Aaslid et al., 1982; Tsuchiya et al., 1991; Schöning and Walter, 1992; Schöning et al., 1993a,b). Color duplex flowmetry of the internal carotid and the vertebral arteries now offers us an additional tool for investigation of brain circulation by directly measuring flow volumes in the brain-feeding arteries and, hence, CBFV. In contrast to PET, the procedure may be performed at any time and repeated for serial bedside measurements. The method has proven to be highly reliable when meticulous examination technique is observed. In our opinion, there are vast potential applications for this method in the monitoring of pathological states of low and high cerebral perfusion, investigation of normal variability in CBFV, and research on psychophysiological and pharmacological factors affecting CBF.
