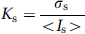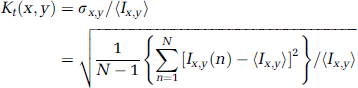Abstract
Mini-ischemia localized into a specific brain area has promoted understanding of the mechanisms underlying brain recovery in stroke. However, the conventional mini-stroke model adopted permanent arterial ligations but lacked controllable reperfusion, which is crucial for the outcome of delayed functional recovery. In this study, we devised a new rat mini-stroke model in which the vascular ligations can be easily reversed to induce targeted reperfusion. Specifically, a flexible ring was incorporated into the conventional small arterial ligations to tighten the ligating loops and facilitate cutting the ligatures for sufficient reperfusion afterwards. The distribution of cerebral blood flow was explored directly through a cranial window using laser speckle contrast imaging. A distinct ischemic core, which well fits the profile of the ligated ring, was bordered by a penumbral zone and then together surrounded by nonischemic tissue immediately after the arterial ligations involving the ring. After cutting the ligatures, post-recanalization hyperperfusion occurred in the previous ischemic core and to a greater extent at 24 h after reperfusion. In contrast, recirculation of common carotid artery in the conventional mini-stroke model hardly altered hypoperfusion status within the ischemic core. Evidence from two kinds of control groups indicated that the ring might produce a compression effect on the underlying cortex and then contribute to the more highly localized infarct that was identified by triphenyltetrazolium chloride staining. Our data suggest that this model provides opportunities for investigating the neurovascular dynamics in acute stroke and rehabilitation, especially with emerging optical imaging techniques.
Keywords
Introduction
Stroke is the leading cause of adult disability. It induces acute neurological and functional deterioration, the recovery from which is delayed and long-term process (Carmichael, 2003). Developing effective therapeutic strategies to improve functional outcome after stroke relies on a better understanding of functional recovery and neuronal plasticity (Carmichael, 2003; Heiss and Teasel, 2006; Ward and Cohen, 2004; Zhao et al, 2006). Many recent studies have focused on the neurovascular unit, suggesting promising targets for the repair processes in stroke therapy (Iadecola, 2004; Lo et al, 2003; Ward and Cohen, 2004). However, further details of the mechanisms underlying functional recovery are poorly understood, in part because of the lack of stroke models relevant to humans, in which one can trace the neurovascular dynamics and characterize the restoration of lost brain function in stroke (Carmichael, 2005). Strokes in humans prone to functional recovery are mostly small in size, ranging from 4.5% to 14% of the ipsilateral brain hemisphere (Carmichael, 2005), whereas most previous stroke models produced relatively large infarcts involving a number of extensive neuronal circuits, the anatomical and physiological connection of which is not well understood (Belayev et al, 1996; Dirnagl et al, 2003; Traystman, 2003). Therefore, a mini-stroke model with a small infarct in a well-defined neuronal circuit, such as the barrel somatosensory cortex, is essential for understanding human neuronal repair and functional recovery after stroke (Carmichael, 2005; Watanabe et al, 2001; Wei et al, 1995).
The current mini-stroke model in rats, primarily introduced by Woolsey and colleagues (Wei et al, 1995), is produced by permanently ligating three to six branches of the middle cerebral artery (MCA) (as shown in Figure 1, left and middle) and temporarily occluding the common carotid artery (CCA). Although this procedure successfully produced a small ischemic infarct within the barrel somatosensory cortex, the extent of reperfusion in the ischemic core was hard to determine because of the presence of sutures left after surgery. Early reperfusion has been recognized as one of the necessary aims of the treatments for human ischemia, whereas delayed reperfusion leads to a progressive destruction of the initial ischemic tissue (Schaller and Graf, 2004). Apart from the size or severity of initial ischemia, the time and extent of reperfusion are also critical factors in the degree of delayed functional recovery after stroke. Therefore, compared with the current mini-stroke model, a more controlled region of initial ischemia and reperfusion within a small brain area would lead to a better model for understanding neural plasticity and repair in stroke.
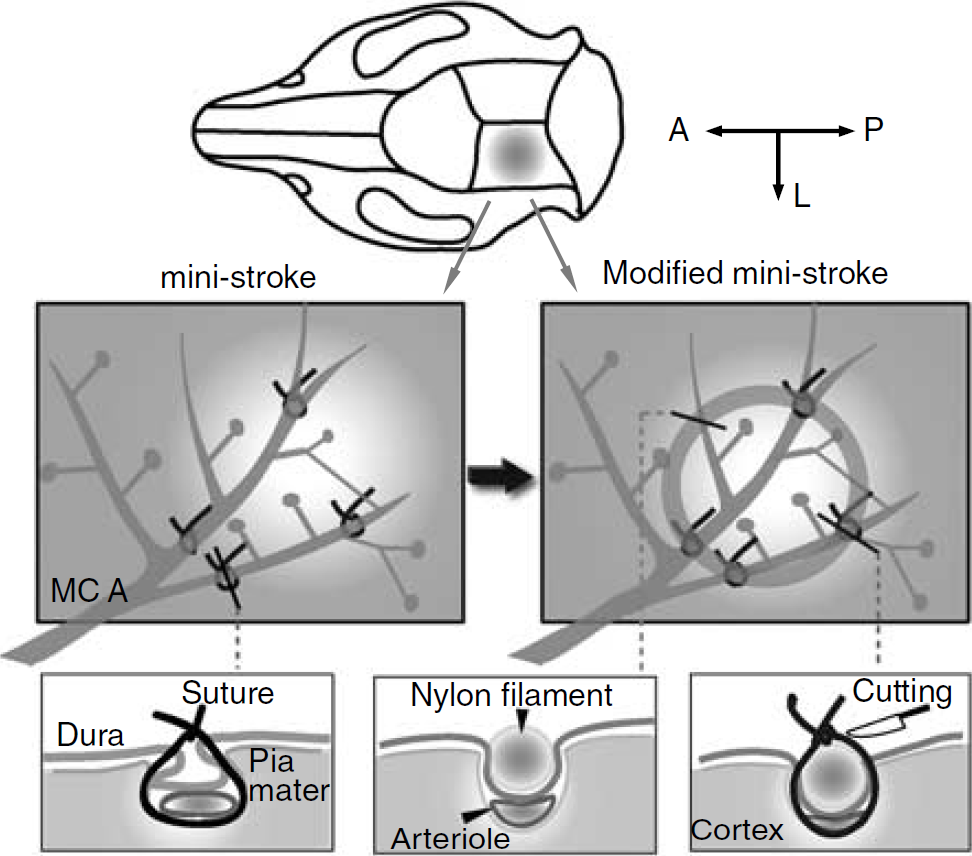
Schematic drawing of the ligating procedure in the conventional mini-stroke model and the modified model. First, the targeted cortex was exposed. A, anterior; P, posterior; L, lateral. In the mini-stroke model (left one in the middle row), several branches of the MCA that surrounded the targeted region were selected and then tied with the sutures. In the modified mini-stroke model (right one in the middle row), a flexible ring was incorporated into the ligations and facilitated tightening and cutting the sutures. Furthermore, the ring could depress into the cortex and compress the underlying vessels. The profiles of the three representative sites are illustrated in the bottom row.
Direct and full reperfusion requires recanalization of the previously occluded vessels, which is impossible in the conventional mini-stroke model because of the risk of damaging the vessels or the underlying brain tissue (Wei et al, 1995). Moreover, inserting ‘snare’ ligatures (Shigeno et al, 1985) or microclips (Buchan et al, 1992; Traystman, 2003) in such a confined region and at several sites is technically demanding because of the restricted space; it also needs the incision of the dura. Cortical compression model (Watanabe et al, 2001) can readily induce a localized ischemia/reperfusion in the barrel field of the rat somatosensory cortex, by compressing the targeted cortex with a brass cylinder. However, this cylinder-shaped device prevents high spatial resolution imaging techniques from obtaining direct and full field observations of the ischemic core and the outer regions.
A simple modification of the current mini-stroke model (Wei et al, 1995), which shares some of the advantageous features with the cortical compression model (Watanabe et al, 2001), was used here to form a focal infarct from ischemia/reperfusion in the rat cortex. Specifically, a flexible ring was incorporated into the conventional small arterial ligations (Wei et al, 1995) to separate the ligatures from the artery on the outside surface of the ligature and allow easy cutting of the suture to provide local reflow. Moreover, it can be directly observed with high-resolution optical imaging techniques. Therefore, laser speckle imaging (LSI), which has recently been proven capable of sensitive detection of changes in blood flow with high spatiotemporal resolution (Ayata et al, 2004a; Briers and Webster, 1996; Dunn et al, 2001; Durduran et al, 2004; Paul et al, 2006; Strong et al, 2006; Wang et al, 2007), was used here to assess the perfusion status of the rat cortex before, during, and after mini-ischemia.
Materials and methods
Animal Preparation
Male Sprague-Dawley rats weighing between 250 and 310 g, obtained from Hubei Center of Disease Control and Prevention, were used in the experiments. All experimental procedures were conducted in accordance with the Institutional Animal Care and Use committee of Hubei Province. Rats were intraperitoneally anesthetized with the mixture of α-chloralose (50 mg/kg) and urethane (600 mg/kg) and maintained in a state unresponsive to toe pinching by supplements of one-third of the initial dose. Body temperature was kept constant at 37.0°C ± 0.5°C with a heating regulator pad. The left femoral artery was cannulated for continuous blood pressure monitoring and to discontinuously obtain arterial pH, pCO2, and pO2 samples. These physiological parameters remained within normal limits throughout the experiments.
Each rat was placed in a stereotaxic apparatus; a longitudinal incision of 15 mm length between the lateral canthus and the external auditory meatus was made under an operating microscope (SZ12, Olympus, Tokyo, Japan), and the temporalis was incised and retracted. A craniotomy over the primary somatosensory cortex (the center of which was located 5 mm lateral to the sagittal suture and 3 mm caudal to the coronal suture) was made with a high-speed dental drill (Fine Science Tools, Foster City, CA) under cooled saline. The skull bone was removed with care to keep the dura intact. Cranial windows of 7 mm × 9 mm size were then formed and continually bathed with artificial cerebrospinal fluid at 37°C.
To functionally localize the barrel field that represents the rat contralateral whisker, the exposed cortex was imaged by optical imaging of intrinsic signal at a wavelength of 550 ± 10 nm while the contralateral whiskers were stimulated (Li et al, 2003; Luo et al, 2007; Wei et al, 1995). Therefore, the branches of the MCA that perfuse the defined barrel field (also termed the ‘targeted cortex’ in the following text) were determined by the spatial extent of the intrinsic optical signal in response to the contralateral whisker movement according to the procedures described in the conventional mini-stroke model (Wei et al, 1995; also see Supplementary Figure 1).
Ring Preparation
An ˜10mm length nylon thread (0.168 or 0.2 mm in diameter) was bent and then glued with plastic adhesive to form a circle measuring approximately 3 mm in diameter. The ring was then placed into a ventilated environment for 24 h to dry and fix the glue. Or, the material for individual vascular occlusion at one or the other site (see below, the control experiment in group 4) was made by cutting the nylon thread into small pieces (˜ 1 mm long).
Focal Ischemia/Reperfusion in the Rat Somatosensory Cortex
The rats in this experiment were divided into four groups. In group 1 (the ring involving the mini-stroke model), eight rats were subjected to focal ischemia/reperfusion according to the following procedure (Figure 1, right and middle): First, the short pieces of 11-0 suture thread were passed through the dura and under the selected branches of the MCA, where each branch crossed into or out of the targeted area, and left to complete the ligation (Wei et al, 1995). Then a homemade nylon ring was positioned on the surface of the defined barrel cortex and between every pair of the open ligature ends. Finally, mini-ischemia was produced by tying all ligature loops to include the nylon ring, the dura, and the selected branches of MCA. Because of the flexibility of the nylon ring, its presence hardly limited the choice of arteries available to tie off or the sites suitable for ligation. The sites of ligation (the total numbers: median 4 and range 3 to 6) were distributed around the targeted cortex. In some cases, the angle between the adjacent ligatures was more than 90°. To fix the ring and consequently enhance the compression of the vessel, some additional ligatures that did not contain the vessel (median 1, range 0 to 2; see Figure 5C) were inserted between two widely separated ligatures. After 2 h of ischemia, the sutures over the nylon ring were cut by a surgical scalpel. Subsequently, the broken sutures and the ring were picked up as carefully as possible so as to promote recirculation in the arteries.
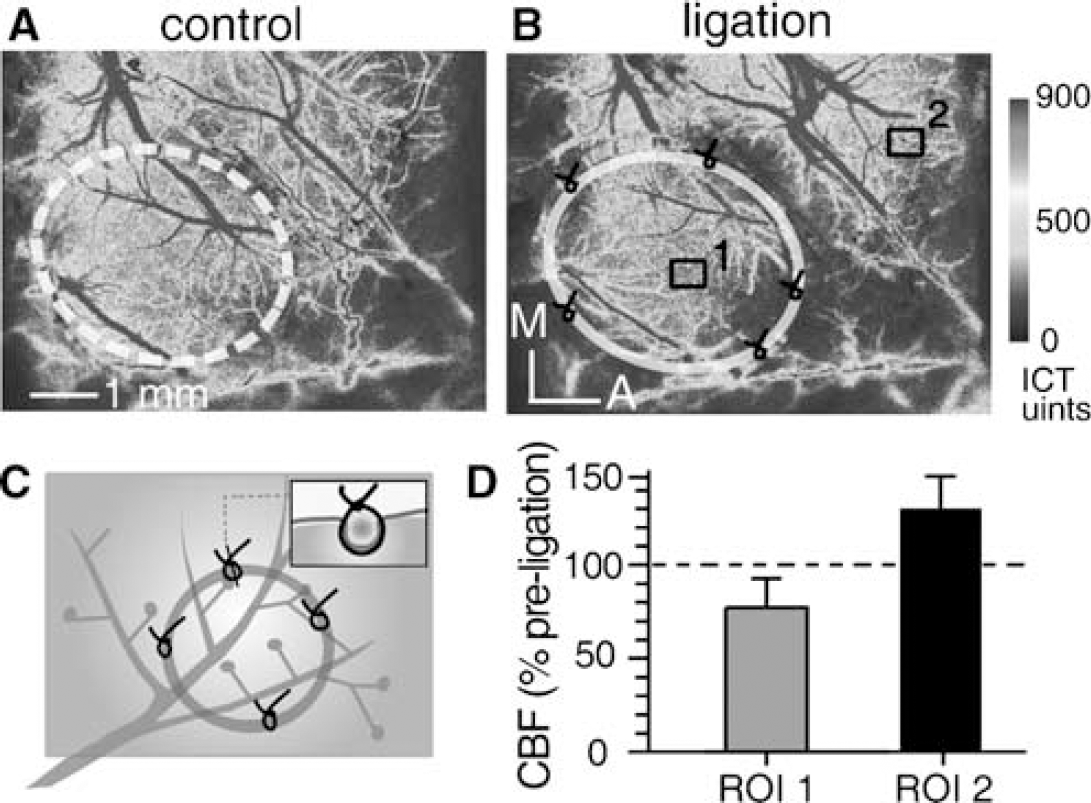
The compression effect induced by the ligated ring. (
In group 2, five rats were used according to the procedure of the conventional mini-stoke model (Wei et al, 1995). The selected branches of the MCA (median 4, range 3 to 6), which serve the barrel cortex, were ligated by passing short pieces of 11-0 suture through the dura and under the vessels (Figure 1, left and middle). Subsequently, the bilateral common carotid arteries (biCCA) were further occluded by a remotely controlled snare. Similarly, after 2 h of ischemia, the occlusion of biCCA was released to allow reperfusion.
Groups 3 and 4 were used to validate the effectiveness of the nylon ring in achieving compression of the vessels. In group 3 (n = 5), the ligations were performed by carefully passing the suture through the dura, and then touching the superficial layer of the cortex, while excluding the branches of the MCA. As a consequence, only the ring was ligated and depressed into the imaged cortex (see Figure 5C).
The alternative mini-ischemia/reperfusion model in group 4 (n = 5) was performed by integrating a small piece of nylon thread (approximately 1 mm in length, the same material as used for making the ring) within each individual vascular ligation on site (Figure 6D). In this case, the incorporation of the nylon thread can also facilitate the tightening and cutting of the sutures at each ligating site, while lacking the compressing effect caused by the ring on the collateral vessels.
The dynamic changes of CBF in another modified mini-stoke model incorporated with pieces of nylon thread. (
The rats (approximately 20% of the animals) whose cranial window showed significant bleeding or inflammation after surgery were excluded from the subsequent imaging process. After the end of the optical imaging experiment, the skull bone was replaced into the cranial window and all the wounds were closed with 6-0 sutures. Erythromycin ointment was topically applied to the wounds to prevent infection. The animals were returned to their cages; the temperature was maintained with a heating lamp, and water and food were allowed ad libitum. In group 1, 24 h after reperfusion onset, the rats were reanesthetized and the cranial window was reopened to be further observed by LSI.
Laser Speckle Imaging
The distribution of cerebral blood flow (CBF) in the rat cortex was determined by LSI (Briers and Webster, 1996; Dunn et al, 2001; Durduran et al, 2004). The procedure of LSI has been described in detail elsewhere (Cheng et al, 2003; Li et al, 2006). Briefly, a He-Ne laser beam (Λ = 632.8 nm, Melles Griot, Carlsbad, CA) was used to illuminate the observed cortex in a diffuse and uniform manner. The aperture was adjusted to keep the speckle size comparable to the area of a single pixel in the CCD chip. Raw speckle images were recorded by a 12-bit CCD camera (480 × 640 pixels, Pixelfly, PCO, Kelheim, Germany), which was attached to a stereoscopic microscope (SZ6045TRCTV, Olympus, Tokyo, Japan). In the traditional laser speckle contrast analysis (LASCA) method (Briers and Webster, 1996; Dunn et al, 2001), the blurring of the speckles was quantified by local spatial speckle contrast (Ks), which is defined as the ratio of the standard deviation (δs) to the mean intensity (〈Is〉) in a small region of the image:
This method uses the spatial intensity variations in a speckle pattern to obtain the laser speckle contrast map. In practice, a spatially scanning window of 5 × 5 or 7 × 7 pixels through the whole image is used.
In our previous works, a new analysis method for producing laser speckle contrast map based on temporal statistics of time-integrated speckle patterns was developed and termed ‘laser speckle temporal contrast analysis’ (LSTCA) (Cheng et al, 2003; Li et al, 2006). In this method, the speckle temporal contrast image was constructed by calculating the speckle temporal contrast of each image pixel successively over time. The value of the speckle temporal contrast Kt at pixel (x, y) was calculated as
where Ix,y (n) is the CCD count at pixel (x, y) in the nth laser speckle image, AT is the total number of images, and 〉Ix,y〉 is the mean value of CCD counts at pixel (x, y) over the N images. For a comparison, the same data set (the images 10mins after ligation in group 1, 30 frames) was used to construct a speckle contrast image using the LASCA and the LATCA methods (Figure 2).
(
Assuming to be proportional to the regional CBF (Briers and Webster, 1996; Dunn et al, 2001), the images of inverse correlation time values (ICT, 1/τ, arbitrary units) were calculated from the speckle contrast images with in-house written code based on Matlab 7.0 (The Mathworks Inc., Natick, USA). This ICT image was then used as an indicator of the CBF parameter to visualize the evolution and distribution of ischemia in the rat somatosensory cortex. Approximately 10 mins before applying focal cerebral ischemia, image acquisition commenced with a set of baseline images and continued at the following times: about 10 mins after ischemia, about 10 mins after reperfusion, and 24 h after reperfusion in group 1. At each time point of acquiring images, five to ten trials (each including 30 frames, 40 fps) were accomplished at an interval of 1 min for further averaging.
Measurements of infarct volume
The procedures for assessment of infarct volume have been reported in detail previously (Joshi et al, 2004). In brief, the animals were allowed to survive after ischemia for 3 days and then deeply reanesthetized. The whole brains were quickly removed from the cranial cavity and incubated in 0.5% 2,3,5-triphenyltetrazolium chloride (TTC) solution at 37°C for assessment of mitochondrial dehydrogenase activity. Alternatively, the removed rat brains were sectioned into 2-mm-thick fresh slices in the coronal plane and then incubated in 0.5% TTC solution. After 30 mins, the coronal sections were viewed with a digital camera and the infarct volumes were segmented and then calculated using Matlab 7.0 (The Mathworks Inc.).
Data Analysis
The spatial distribution of severe ischemia was calculated by determining the pixels in the imaged cortex where ICT value was below 40% (Yao et al, 2003) of the mean control value (the average value of several randomly selected areas of the cortical parenchyma before the ligations; see Supplementary Figure 2 in detail), whereas the penumbral area was identified by determining the pixels in the imaged cortex where the ICT units were between 40% and 70% of the baseline level (Heiss, 2000). For analysis of the distribution and changes in perfusion level in the imaged cortex, a number of small size (˜15 × 15 pixels) regions —of interest (ROIs) were drawn freehand so as to avoid surface blood vessels (Strong et al, 2007) (e.g., one ROI was selected in the ischemic core, one in the penumbral zone, and one in the normal perfused tissue). The relative CBF in each ROI after ischemia or reperfusion was represented as the percent change from the baseline value in the same area. The targeted cortex was outlined by the ligation sites that were first determined according to the OIS response pattern (also see Supplementary Figure 1). The border of the targeted cortex in group 1 was equivalent to the rim of the ligated ring (denoted by the dashed circles in Figure 3E), whereas the targeted cortex in groups 2 and 4 was presumed to be a round region with a rim fitting most of the ligating sites (denoted by the dashed circles in Figures 3F and E). Statistical values are expressed as mean ± s.d. The statistical differences in the residual CBF in the targeted cortex (denoted by the yellow dashed circles in Figures 3C and 3F) between groups 1 and 2 were determined by Student's t-test. Statistical significance was accepted at a probability of P < 0.05.
(
Results
Physiologic Monitoring
All the physiologic parameters monitored were within the normal physiologic range during the experiments. The mean arterial blood pressure and blood gas pressure were as follows: mean arterial blood pressure = 98 ± 11 mmHg, pO2 = 145.3 ± 14 mmHg, pCO2 = 37.2 ± 4.1 mmHg and pH = 7.4 ± 0.1.
Laser Speckle Temporal Contrast Analysis
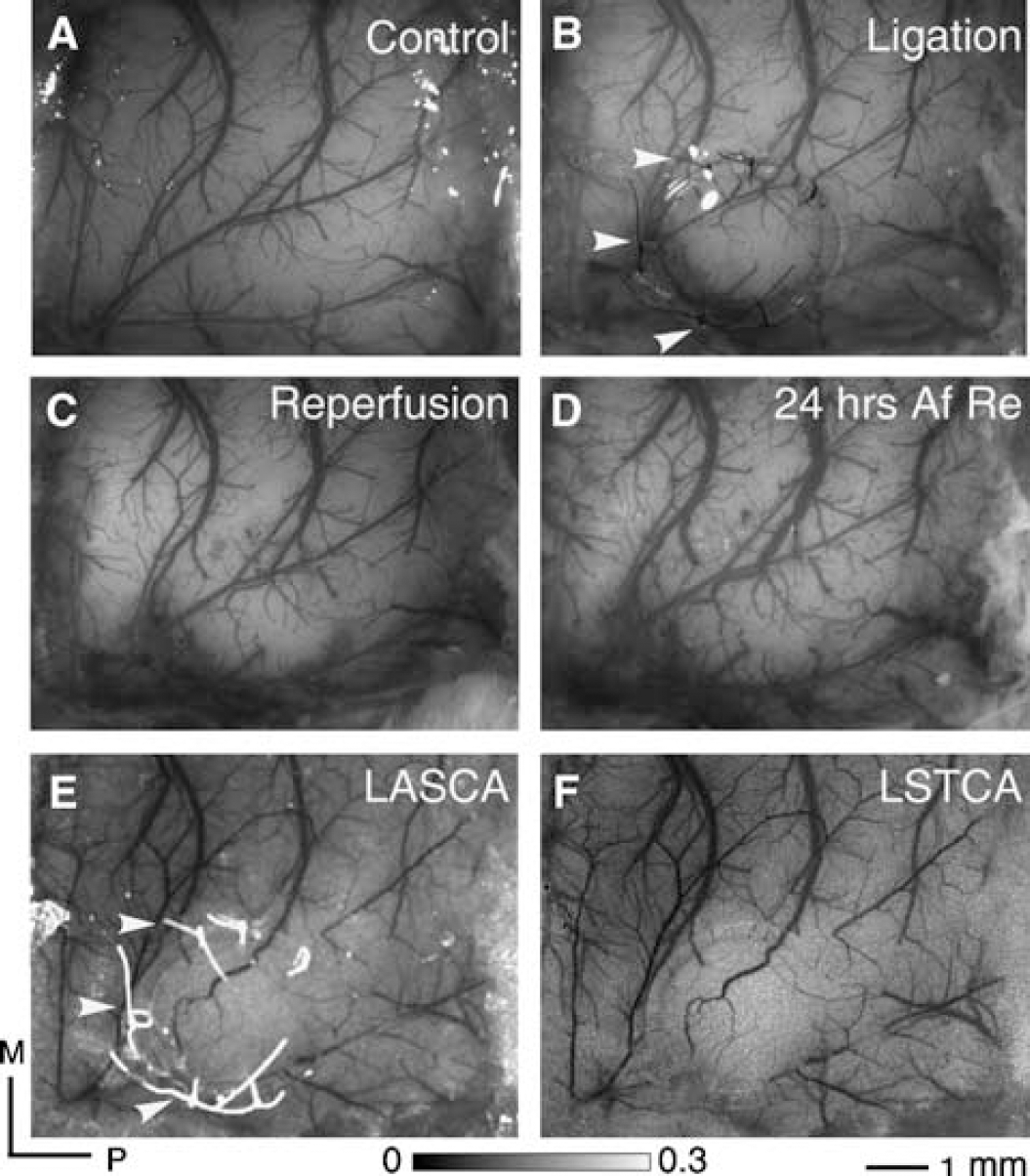
The speckle contrast images calculated with the methods of LASCA (Briers and Webster, 1996; Dunn et al, 2001) and LSTCA (Cheng et al, 2003; Li et al, 2006) from the same data sequence (10 mins after the ligation involving the ring in group 1) are illustrated in Figures 2E and 2F. The contaminations from the ligating knots and nylon ring within the imaged cortex are obvious in the LASCA image (denoted by the white arrows in Figures 2B and 2E), whereas the LSTCA image provided higher spatial resolution and a clear view (Figure 2F). Therefore, in the following results, the ICT data that are proportional to the regional CBF were acquired from the laser speckle contrast image based on the calculation of the LSTCA method.
Distribution of CBF during and after Ischemia
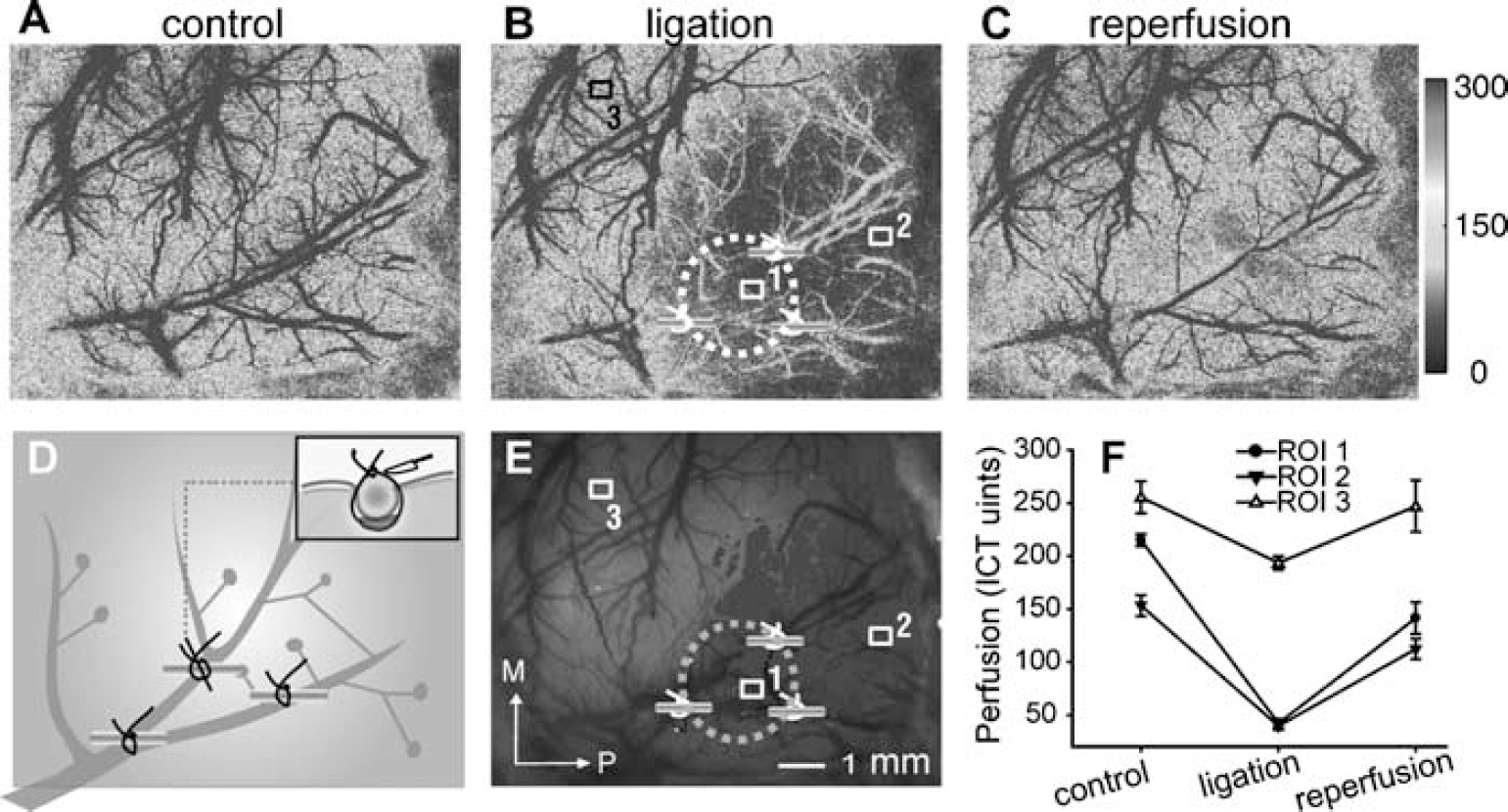
As shown in Figure 3, the LSI was used to trace the distribution of CBF before, at, and after ischemia over rat somatosensory cortex in two representative animals from groups 1 and 2. In the new mini-stroke model group (group 1), the nylon ring adopted here was 3.5 mm in diameter. Three ROIs (Figure 3E) were drawn within (ROI 1, control ICT units were 103.1 ± 10.4) or distal (ROI 2, CBF, 120.5 ± 15.7 ICT units) to the targeted cortex, or in the normally perfused cortex (ROI 3, CBF, 182.1 ± 7.4 ICT units). Immediately after the ring was bonded with the branches of MCA by the suture, the imaged cortex presented a distinct CBF gradient from the ischemic core (dark-highlight area around ROI 1 in Figure 3E, 25.9 ± 2.5 ICT units) to the penumbral zone (light-highlight area around ROI 2, 46.9 ± 6.1 ICT units) to normal tissue (around ROI 3, 120.5 ± 4.7 ICT units), recovering 10 mins after the sutures were cut, to 116.1 ± 6.5, 84.8 ± 11.5, and 112.1 ± 9.1 ICT units, respectively. At 24 h after reperfusion, the perfusion level in the previous ischemic core (ROI 1, 249.8 ± 15.1 ICT units) was further significantly higher than that in the surrounding cortex (ROI 2, 144.1 ± 13.1 ICT units; ROI 3, 157.6 ± 9.8 ICT units). In the presumed penumbral zone, the change of blood flow in the collaterals was apparent (Figure 3E, ROI 2), whereas the blood flow of the collaterals in the ischemic core was relatively rarely observed (Figure 3E, ROI 1). In group 1, the mean residual CBF of the severely ischemic core, whose distribution was perfectly indicated by the profile of the ligated ring (Figure 3E), was decreased to 23.2% ± 5.8% (n = 8) of the preligation perfusion level (Table 1). The white light images captured at the corresponding time points are also illustrated in Figures 2A to D. The ligatures and the flexible ring did not hinder the direct microscopical observations.
The rCBF changes in the targeted cortex

rCBF, the residual CBF that is represented as percentage of the baseline value in the targeted cortex. The ischemia and reperfusion values in group 1 were acquired 10 mins after the ring involved MCA's branches' ligations and 10 mins after the MCA's branches' recanalization, whereas the data in group 2 were traced 10 mins after the MCA's branches' ligation plus further occlusion of the bilateral CCA and 10 mins after the release of bilateral CCA.
Significant difference compared to the value in group 1, P < 0.05. Values are expressed as means ± s.d.
In the conventional mini-stroke model group (group 2), three ROIs were also drawn at the sites (1) in the targeted cortex (ROI 1 in Figure 3F), (2) in the distal region to the targeted cortex (ROI 2 in Figure 3F), and (3) in the normally perfused cortex (ROI 3 in Figure 3F). The baseline perfusion values in the three ROIs were 121.9 ± 10.1,108.4 ± 10.8, and 143.2 ± 20.7 ICT units, respectively. There was an immediate decrease in perfusion, as the ligatures were tightened around the branches of MCA in the targeted cortex, to 44.8 ± 1.7 and 47.8 ± 1.4 on ROI 1 and distal ROI 2, respectively. A further biCCA occlusion aggravated the severity of ischemia within the whole imaged cortex (blood flow decreased to 36.0 ± 1.5 and 43.8 ± 1.1 ICT units in ROIs 1 and 2) and also caused a larger effect in the surrounding normal cortex (ROI 3, decreased from 177.1 ± 8.7 to 112.8 ± 4.8 ICT units). The reperfusion after the release of CCA occlusion largely reversed this effect. In the outer area of the imaged cortex (ROI 3) blood flow recovered to 182.2 ± 24.4 ICT units, and also perfusion in the targeted cortex showed a slight increase (46.1 ± 5.5 and 51.1 ± 4.1 ICT units in ROI 1 and ROI 2, respectively). The extent of severe ischemia in the imaged cortex was larger than that observed in group 1 and shifted a little in relation to the targeted cortex (ligating sites surrounding region, indicated by a dashed circle in Figure 3F). Collateral vessels within this ischemic core as well as the distal region were easily identified. Among five rats that were subjected to small arterial ligations in group 2, the relative perfusion level over the targeted cortex was 25.5 ± 4.9% from the baseline level.
Infarct Volume and Location
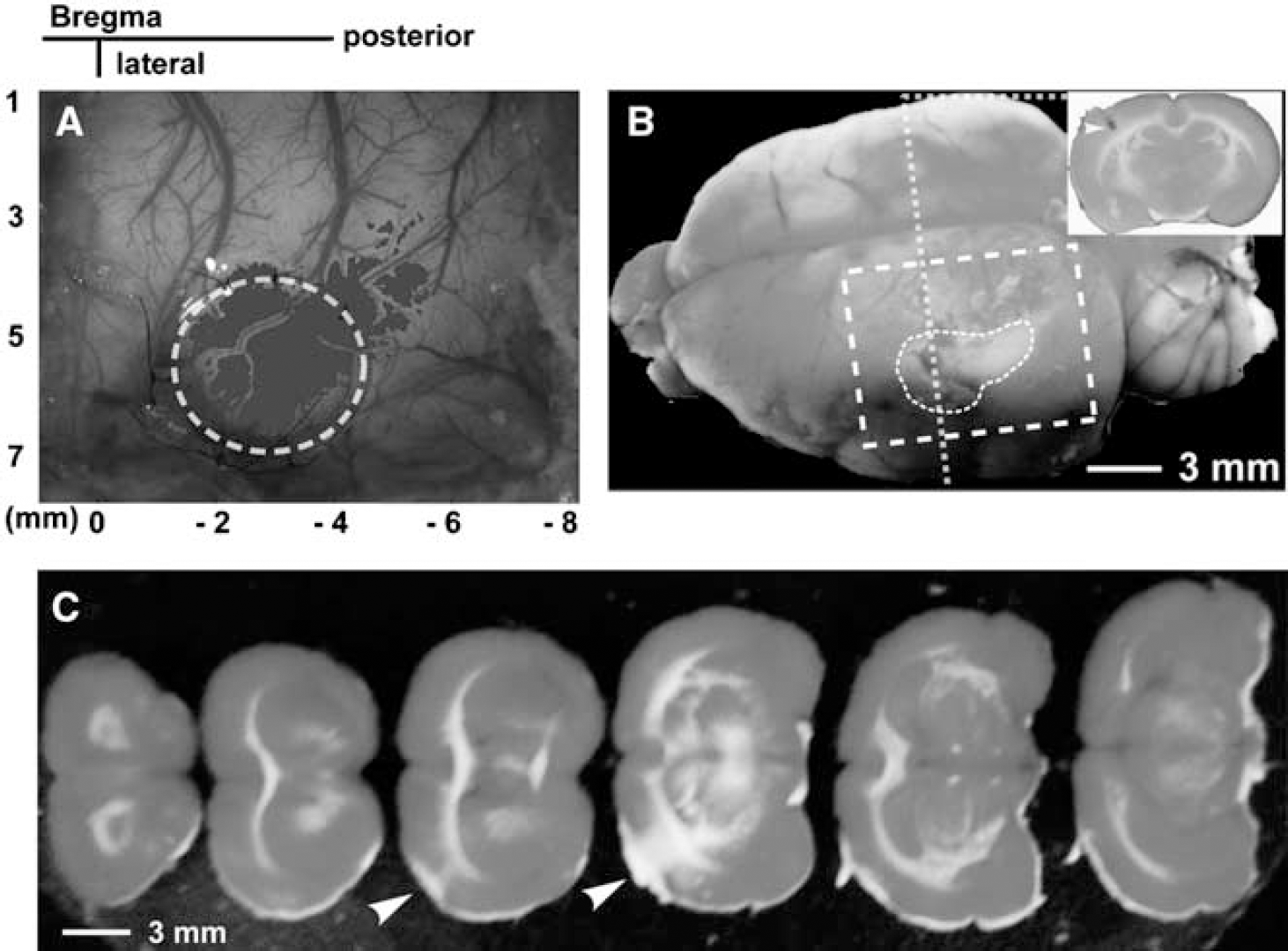
The postmortem results from the brain tissue stained with TTC showed that the reproducible infarcts were induced in both groups 1 and 2. A representative TTC staining from a rat 3 days after reperfusion in group 1 indicated that the infarct area was essentially in accordance with the targeted cortex and the ischemic threshold map (<40% of the mean baseline value at the time of preischemia) (Figures 4A and 4B). Furthermore, in three of eight rats in group 1, a small intracortical hemorrhage was observed through the brain slice (Figure 4B, inset box). Small focal infarcts were typically 3 to 4 mm in diameter and mainly confined to cortical gray matter (Figure 4C), and the rats in group 1 had a cortical ischemic lesion volume that was 5.1% ± 2.8% of the contralateral hemisphere.
Ischemic infarcts indicated by TTC staining. (
Effect of Compression on Cerebral Blood Flow
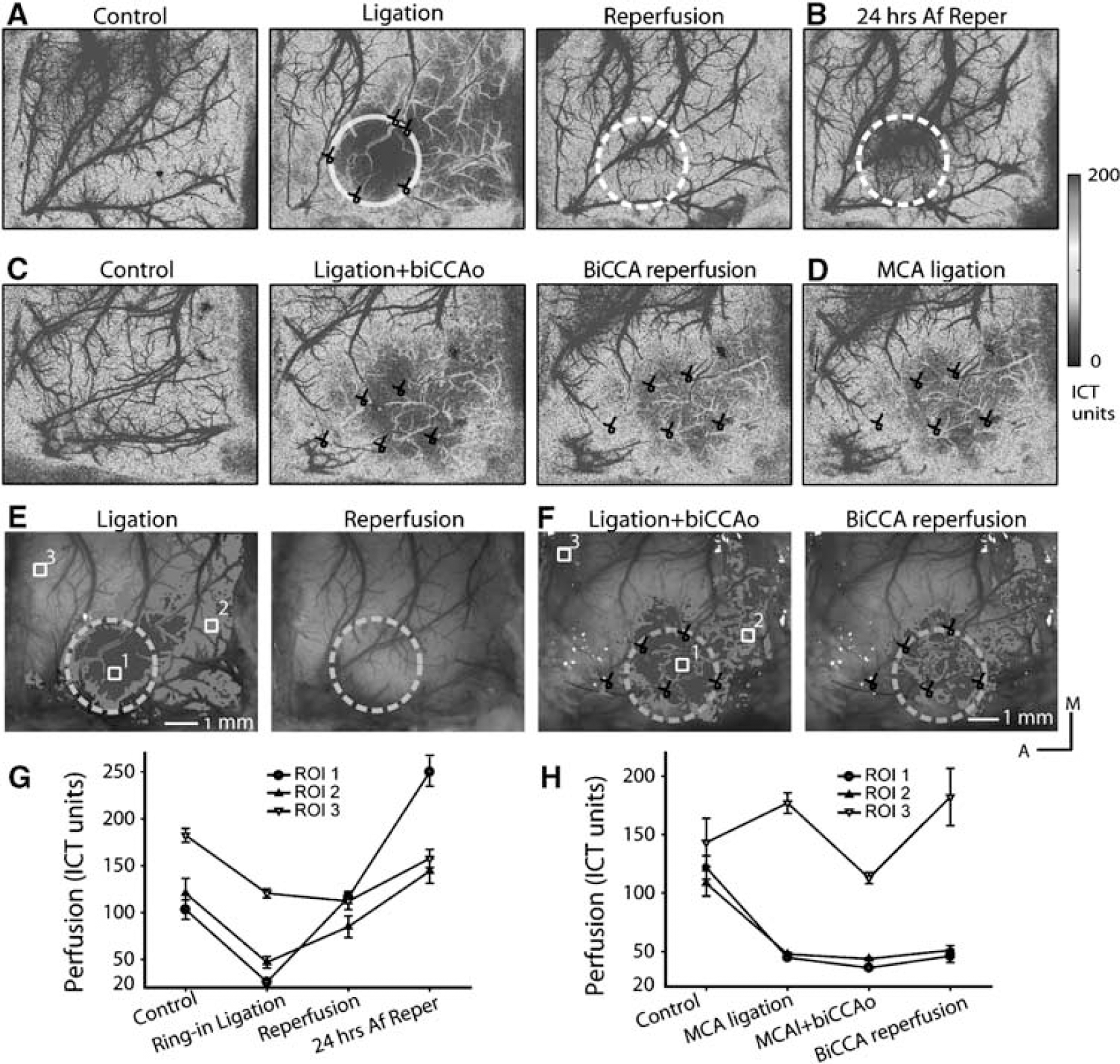
As the nylon ring was ligated into the cortical tissue, without ligating the branches of MCA in group 3 (Figure 5C), the distribution of CBF in the ring interior and surrounding cortical regions was also disturbed. The residual CBF within the ring and the branches of MCA compressed by the rim of the ring all showed a slight reduction (to 77.3 ± 14.1% of the baseline level), whereas CBF in the regions outside the ring tended to increase (Figures 5B and 5D). The vascular ligations in group 4 were performed with a short length of nylon thread on each site (Figure 6D). This model showed a larger and shifted ischemic core that is identical to that in group 2 (Figures 6B, 6E and 6F), while also allowing for vascular recanalization after cutting the sutures, which was possible because of the incorporation of nylon threads into the ligations (Figures 6C and 6F).
Discussion
In this study, we have devised a modified mini-stroke model that shares features of MCA branches (Wei et al, 1995) or cortical compression with previous mini-stroke models (Watanabe et al, 2001). This new model incorporates a flexible ring into the conventional vascular ligation, and is characterized by the possibility of complete recirculation after highly localized ischemia in the rat somatosensory cortex. An advanced technique of laser speckle imaging, which can monitor the CBF with high spatiotemporal resolution (Briers and Webster, 1996; Cheng et al, 2003; Li et al, 2006), was used here for tracing the full-field distribution and dynamic changes in perfusion in the exposed cortex.
Laser speckle imaging based on LASCA was first introduced by Briers and Webster (1996), and was successfully applied to monitor the evolution of CBF under normal (Ayata et al, 2004a; Dunn et al, 2001; Durduran et al, 2004) and pathological conditions, such as cortical spreading depression (Ayata et al, 2004a, b ) and ischemia (Atochin et al, 2004; Ayata et al, 2004a; Paul et al, 2006; Strong et al, 2006), because of its simplicity and high spatiotemporal resolution. The LSTCA method that advanced our previous works can provide even higher spatial resolution (Cheng et al, 2003) and can suppress the influence of the speckle contrast from a stationary structure (Li et al, 2006), such as the skull, nylon ring, or ligating knots. Therefore, these new features, shown in our results (Figures 2 and 3), could afford facilities for improved monitoring of CBF distribution around the ischemic core.
Our modified model offers several advantages over current mini-stroke models to induce localized ischemia/reperfusion in rat somatosensory cortex. First, our model allows for a suture's ligating procedure to be more controllable and safe. Small branches of MCA selected in any mini-stroke model are less than 100 μm in diameter so it is crucial to apply an appropriate force when tightening the ligating loops to avoid breaking the delicate vessels. The dura, which is relatively tougher than the underlying brain tissue, was kept intact in the conventional mini-stroke model, partially for the sake of reducing the risk of vascular fracture during the ligating procedure (Wei et al, 1995). Our model could further almost eliminate the risk of damage to vessels by incorporating an extremely tough material (a flexible ring or piece of nylon thread) into the vascular ligating loops. Based on the stability conferred by the inner nylon thread, the extent of tightening is more controllable. Second, our model presents a simple, safe, and practical strategy to accomplish vascular occlusion and subsequent recirculation. In previous studies, Shigeno et al (1985) obtained vascular occlusion and reperfusion by pulling and releasing a snare ligature that had been placed around the MCA. However, this technique is not pragmatic in experiments with small animals (Traystman, 2003). Subsequently, a neurosurgical clip instead of the snare ligature was often included in the studies focusing on reperfusion injury (Buchan et al, 1992). Likewise, this technique is also faced with space constraints and the necessity for incising the dura. In our model, the nylon thread was inserted into the ligating loops and then acted as a chopping block to facilitate cutting the ligating sutures around it, as well as a barrier to prevent damaging the inferior vessels and cortical tissue. After all the ligating loops were cut and the ring was removed from the cortex, full recirculation could easily be reached in the previously ischemic area.
With high-resolution optical imaging techniques, this modified mini-stroke model can also provide the possibility of long-term assessment of hemodynamic responses in detail over a territory including the whole ischemic core, the penumbral zone, and portions of the normal cortex (Figure 3). Accordingly, a significant post-recanalization hyperperfusion over 24 h after reperfusion was identified to occur in the previous ischemic core (Figure 3A). This result, firstly obtained through in vivo optical imaging, is in line with previous studies using the traditional autoradiographic technique (Tsuchidate et al, 1997). Post-ischemia hyperperfusion may contribute to secondary cellular injury and therefore be a specific target for neuroprotective therapy (Kidwell et al, 2001). So, our mini-stroke model with region-directed reperfusion is ideally suited for investigating the mechanisms underlying reperfusion injury or neuroprotection in the case of postischemic hyperperfusion (Marchal et al, 1999). Furthermore, owing to the global view on/beyond the ischemic territory, this model will encourage more investigators to trace the spatiotemporal pattern of unpredictable occurrence and evolution of ischemia-associated events, such as periinfarct depolarization (Strong et al, 2007).
Another potential advantage offered by this modified mini-stroke model is to restrict the collateral circulation around the ischemic core. Collateral circulation is crucial to the degree and distribution of reduction in CBF over the ischemic tissue (Brozici et al, 2003; Liebeskind, 2003). Accordingly, the constraint of the collateral supply will help investigators to achieve a more consistent ischemic infarction (Carmichael, 2005; Traystman, 2003). Temporary or permanent CCA occlusion was routinely used in focal and global ischemic models as a highly effective method to restrict collateral perfusion (Traystman, 2003). Conversely, Watanabe et al (2001) have also shown that a brass cylinder depressing into the cortex can confine the underlying blood flow to form a highly localized ischemic infarct. Likewise, our result from group 3 suggests that there is an effect of ring compression on the unligated branches of MCA. Furthermore, a comparison of the distribution of perfusion between the modified mini-stroke model (group 1; see Figure 3A) and the control model from group 4 (see Figure 6) implied that probably the more localized ischemic core in this modified mini-stroke model may partially be attributed to the confinement of collateral flow into the targeted cortex by the ligated ring.
There are also some limitations to this described model implying the need for caution in its application. First, as in the cortical compression model (Watanabe et al, 2001), the possibility of traumatic brain injury induced by compression of the ring should be excluded (Marmarou et al, 1994). Watanabe et al (2001) have shown that the low rates of compression of the cortex (less than 15 mm/s) or limited range in depth (less than 0.2 mm) would not cause any obvious trauma. In our model, the ring depresses into the cortex by less than 0.15 mm and the rate of depression is also limited, owing to careful experimental handling. Thus, such traumatic injury because of compression is highly unlikely in this modified mini-stroke model. Second, it is possible that the compression effect using the ring is not completely stable. In contrast to the external pressure applied to the barrel cortex in the cortical compression model (Watanabe et al, 2001), the compression effect on the underlying cortex by the ring in our model is attributed to the inherent tension or pressure that derived from the suture-fixation of the ring to the underlying brain tissue. The controlled manner of compression effect in the cortical compression model was accomplished by real time adjusting the depressing depth plus further temporary CCA occlusion (Watanabe et al, 2001). Therefore, further study is needed to seek similar methods or devices to improve the long-term performance of compression in the mini-stroke model described here and to test its efficacy. Finally, the manipulations, especially the ligating procedures in the exposed cortex, in the traditional and this new mini-stroke model need to be performed carefully. Inappropriate puncturing depth or sites of the suture needle and excess force of tightening the loops will lead to uncontrollable bleeding that had an appearance probability of ˜20% of all animals in this study. However, there are few obstacles in the way of adopting this new mini-stroke model for investigators who are already proficient in preparing the traditional mini-stroke model. The selection of stroke models also largely depends on the targeted interests of the researchers. For example, those studies in which the mechanism of reperfusion is of major interest and the focal ischemic territory is not strictly required to outline, the method of providing mini-ischemia/reperfusion described in group 4 may be a more convenient choice rather than the one involving the ring in group 1.
The important role that the neurovascular unit (the integration of neurons, glia, and cerebral blood vessels) plays in the defense mechanisms that maintain brain perfusion, and its importance in stroke pathophysiology, has been given more attention recently (Iadecola, 2004; Lo et al, 2003). Our mini-stroke model is able to show distinct phases of stroke, providing the opportunity for direct observation of structural and functional changes in the progressive development of stroke if combined with optical imaging methods (Dunn et al, 2001; Zhang et al, 2005). Optical imaging techniques with high spatial resolution, such as intrinsic optical signal imaging, laser speckle imaging, and optical confocal imaging, are capable of tracing cortical reorganization and the neurovascular response at the functional columnar level (Zepeda et al, 2004) and the cellular level (Zhang et al, 2005). In conclusion, the use of this simple, modified mini-stroke model as a basic platform should open new avenues for addressing many fundamental questions in stroke research.
Footnotes
Acknowledgements
The authors would like to thank Professor Ling Wei for technical guidance in preparation of the mini-stroke model, Dr Zheng Wang for helping in the earlier version of this manuscript, and Professor William John Cram for helping in English editing.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.