Abstract
Hypertonia and postural deficits are observed in cerebral palsy and similar abnormalities are observed in postnatal rabbits after antenatal hypoxia–ischemia. To explain why some kits become hypertonic, we hypothesized that white matter injury was responsible for the hypertonia. We compared newborn kits at postnatal day 1 (P1) with and without hypertonia after in vivo global fetal hypoxia–ischemia in pregnant rabbits at 70% gestation. The aim was to examine white matter injury by diffusion tensor magnetic resonance imaging indices, including fractional anisotropy (FA). At P1, FA and area of white matter were significantly lower in corpus callosum, internal capsule, and corona radiata of the hypertonic kits (n = 32) than that of controls (n = 19) while nonhypertonic kits (n = 20) were not different from controls. The decrease in FA correlated with decrease in area only in hypertonia. A threshold of FA combined with area identified only hypertonic kits. A reduction in volume and loss of phosphorylated neurofilaments in corpus callosum and internal capsule were observed on immunostaining. Concomitant hypertonia with ventriculomegaly resulted in a further decrease of FA from P1 to P5 while those without ventriculomegaly had a similar increase of FA as controls. Thus, hypertonia is associated with white matter injury, and a population of hypertonia can be identified by magnetic resonance imaging variables. The white matter injury manifests as a decrease in the number and density of fiber tracts causing the decrease in FA and volume. Furthermore, the dynamic response of FA may be a good indicator of the plasticity and repair of the postnatal developing brain.
Introduction
Cerebral palsy (CP) is a common cause of disability in children (Koman et al, 2004). Cerebral palsy occurs in both term and preterm infants after hypoxia–ischemia (H–I), but disproportionately so in preterm infants (Cummins et al, 1993). A central role for white matter (WM) involvement has been postulated in the pathogenesis of CP (Volpe, 2001) as WM lesions are often observed by ultrasound in preterm babies, including the cystic lesions of periventricular leukomalacia (PVL).
In the perinatal period, WM tracts develop from a disorganized state to a more organized, streamlined bundle of fiber tracts. This makes it especially amenable to investigation by diffusion tensor imaging (DTI) by Magnetic Resonance Imaging (MRI) that estimates the diffusion of water in various axes. One DTI index, fractional anisotropy (FA), reflects a ratio of the diffusivity along fiber tracts to that across fiber tracts. Disorganization with WM injury by H–I would affect the diffusivity in various axes. Thus, abnormalities in FA and similar DTI indices have been provided as evidence of WM injury in sick and even normal preterm babies in numerous studies (Huppi et al, 1998; Inder et al, 1999a; Miller et al, 2002). White matter injury results in abnormalities in the development of critical fiber tracts involved in motor development as in the periventricular WM and internal capsule in preterm infants (Huppi et al, 2001), but to the best of our knowledge, the direct association between motor deficits of CP and previous WM injury, caused by H–I, has not been addressed before.
Our hypothesis was that WM injury was responsible for the hypertonia. To test this hypothesis, we utilized the rabbit model developed in our laboratory (Derrick et al, 2004) that allows us to investigate specific animals with hypertonia compared with those without hypertonia. Our aim was to dynamically examine WM injury by DTI indices that had been previously shown to be reflective of the functional and cellular state of WM (Drobyshevsky et al, 2005). The secondary goal of this study was to establish the basis of developing noninvasive indices of WM injury that could be applicable to humans. Most of the work in animals, mimicking H–I insult in the perinatal period, has been carried out on stroke models (Follett et al, 2000; Liu et al, 2002; Poggi et al, 2005; Vannucci and Vannucci, 1997). Average overall diffusivity, using apparent diffusion coefficient (ADC), in brain tissue was abnormal during an acute H–I event in neonatal rat (Nedelcu et al, 1999; Qiao et al, 2002) and rabbit (D'Arceuil et al, 1998), and in a chronic in utero rat hypoxia model (Baud et al, 2004). However, in all these animal models, significant motor deficits have been hard to elicit (Strata et al, 2004; Wright and Rang, 1990), despite the brain damage on histopathology. This makes it difficult to discover any in vivo MR correlates of motor deficits in such models. Thus, the present study was undertaken to investigate whether hypertonia could be explained by WM injury and whether any clinically relevant MRI indices specific to hypertonia could be determined.
Materials and Methods
Animal Surgery
In vivo global H–I of fetuses was induced by uterine ischemia in timed pregnant New Zealand white rabbits (Myrtle's Rabbits, Thompson Station, TN, USA) at 22 days gestation (70% gestation; E22) as described previously (Derrick et al, 2004). Briefly, the dams were anesthetized with intravenous fentanyl (75 μg/kg h) and droperidol (3.75 mg/kgh), followed by spinal anesthesia by the administration of 0.75% bupivicaine. An arterial balloon catheter was introduced into the left femoral artery; advanced 10 cm into the descending aorta and the balloon was inflated for 40 min inducing continuous uterine ischemia, which resulted in fetal H–I. At the end of the procedure, the balloon was deflated and the catheter was removed. The dams were allowed to deliver in a nest box at term (31.5 days). If the dam did not give birth by 32 days, the fetuses (n = 12) were delivered by Cesarean section under anesthesia as described above. Delivered rabbit kits were resuscitated with bag and mask ventilation if needed. Surviving rabbit kits were subjected to neurobehavioral testing as described before (Derrick et al, 2004) and underwent an MRI examination.
Groups
Control: Naive animals not subjected to antenatal H–I (n = 19).
Hypoxia, non hypertonia: Apparently healthy kits selected from litters born after antenatal H–I (n = 20).
Hypoxia, hypertonia: Kits with severe hypertonia and postural deficits (n = 32).
Longitudinal Studies
A subset of kits after H–I and control kits were allowed to grow till P5. Kits that were not severely affected were returned to the dam. Severely affected kits that could not suck and swallow were gavage fed, via a soft silicone catheter three times a day, with 200 to 250 cc/kg day of rabbit milk, and reared in a temperature-regulated neo-natal incubator. Rabbit milk was obtained from a separate set of lactating rabbit mothers that had been allowed to deliver spontaneously without any interventions. The rabbit mothers were given 3 units intramuscular Oxytocin 1 h before obtaining milk using a suction device. The rabbit milk was then filtered through gauze to remove extraneous hair and then refrigerated. At P5, the rabbit kits underwent a second MRI examination (10 per group).
Magnetic Resonance Imaging Methods
Rabbit kits were sedated with intramuscular injection of a mixture of ketamine (35 mg/kg), xylazine (5 mg/kg), and acepromazine (1.0 mg/kg). Animals were placed supine in a cradle heated with water blanket at 35°C. Rectal temperature and respiratory rate were monitored using physiologic monitoring system MP150 (BioPack Systems, CA, USA). Magnetic resonance imaging was performed on a 4.7 T Bruker Biospec system (Bruker, Billerica, MA, USA) using surface coils.
Diffusion tensor imaging experiments consisted of six images with non-collinear diffusion weighting (DWI), using spin-echo diffusion weighted sequence with δ = 5 ms, Δ = 15 ms, TR/TE 2000/35 ms, b = 0, 780 s/mm2 eight averages. If motion artefacts were detected then these images were discarded a priori. Ten oblique coronal brain slices were acquired; matrix size was 128 × 64, zero-filled to 128 × 128. Slice thickness/in-plane resolution after interpolation were 1/0.156 mm for P1, 1.2/0.179 mm for P5 kits. Voxel dimensions were similar to DTI protocol used in rodents (Sun et al, 2003). Slice thickness was chosen to keep number of slices constant and cover approximately the same area of cerebrum from the posterior edge of olfactory bulbs to anterior edge of superior colliculus for all age groups. To ensure reproducible oblique imaging sections, slices were placed perpendicular to the plane connecting the most inferior point of cerebrum, as determined with the aid of multislice sagittal localizer scan.
Diffusion tensor was calculated using multivariant linear fitting of signal attenuation from the acquired diffusion weighted images (Basser and Jones, 2002). Principal eigenvectors and eigenvalues were obtained after diffusion tensor diagonalization. Apparent diffusion coefficient, axial (first eigenvalue of the diffusion tensor, λǁ) and radial (average of the second and third eigenvalues, λ┴) diffusivities (Song et al, 2002), and FA maps were calculated (Basser et al, 1994) using in-house software written on Matlab 5.6 (MathWorks, Natick, MA, USA). Trace DWI were generated for anatomical reference, as they provided superior tissue contrast to T2-weighted images in neonatal rabbits (Drobyshevsky et al, 2005).
Region of Interest Analysis
Regions of interest (ROIs) were placed on the slice by a masked observer. All measurements were made on a single slice in a reproducible location on the level of hippocampus and anterior thalamic nuclei, passing through corpus callosum and anterior commissure approximately 1 mm posterior bregma. White and gray matter structures were outlined manually by placing polygon ROIs on the anatomical images using in-house Matlab software. Directionally encoded FA maps, with pixel color defined by the direction of its primary eigenvector and the intensity proportional to the FA (Pajevic and Pierpaoli, 1999), were used to identify major fiber tracts and selected ROIs. Detected WM tracts served as references in the placement of ROI for gray matter in the cortex, hippocampus, putamen, and caudate nucleus. Circular ROI (5 mm in diameter) were placed on the thalamus and red nucleus avoiding cerebral peduncle and optic tract as these were difficult to separate from adjacent structures. Full brain coverage and volumetric analysis was not possible owing to time limitations for the sick kits in the hypoxic group and hardware limitations of our scanner, which restricted the number of DWI slices. Areas on the same slices were used as representative of the volume of brain structures and reported on the selected ROIs placed bilaterally, except cerebral cortex, which was outlined on left hemisphere.
Immunohistochemistry for Phosphorylated Neurofilament
To estimate the amount of neuropil axonal network, we immunostained a separate set of brains for phosphorylated neurofilaments. The panaxonal neurofilament marker SMI 312 allows us to track specific areas of the perinatal brain. SMI 312 identifies the large class of phosphorylated neurofilaments, which give structure to axons and are found in normally developing axons but rarely in normal neuronal cell bodies. A delay in appearance or loss of maturation-dependent phosphorylated axonal epitopes in neurofilaments are more sensitive indicators of injury to immature brain than staining against total neurofilaments (Back et al, 2001; Ulfig et al, 1998). Coronal sections of immersion-fixed neonatal rabbit brains (30 μm) were sectioned with a cryostat, pretreated (30 min) in 50% methanol in 0.3% H2O2 and TBS, permeabilized (30 min) in 0.4% Triton X-100/TBS and pre-incubated (1 h) in 4% normal goat serum diluted in 0.1%TX/TBS. Sections were incubated (12 h at room temperature, covered with parafilm) in SMI-312 (1:50 dilution) (Sternberger Monoclonals, Baltimore, MD, USA), Triton 0.1%, 2% normal goat serum in TBS. After incubation, sections were rinsed (3 × 10 min) in TBS, incubated (1 h) with goat anti-mouse IgG F(ab')2 fragment (1:20 dilution) (Cappel, West Chester, PA, USA), Triton 0.1%, 2% normal goat serum in TBS, rinsed (3 × 10 min) in TBS, and incubated (1 h) with mouse ClonoPap (1:100 dilution) (Sternberger Monoclonals, Baltimore, MD, USA), 2% normal goat serum in TBS. After final incubation, sections (3 × 10 μm) were rinsed in TBS and developed using a standard diaminobenzidine reaction. Omission of the primary antibody eliminated all immunoreactivity (data not shown). Slides were photographed, scanned and converted to grayscale. Optical densitometry (OD) measurements were obtained with IP Lab Gel for equal size regions within the cortex, corona radiata, corpus callosum, and internal capsule from 2 to 3 sections/animal at the level of the anterior dentate gyrus. As very little phosphorylated neurofilament is found in the soma of cortical neurons, this measurement was used as background. Optical densitometry of the corona radiata, corpus callosum, and internal capsule was corrected for background, measurements from each section averaged to give one number per region per animal and compared between hypoxic brains with hypertonia (n = 3) and without hypertonia (n = 2), and controls (n = 5).
Statistical Analysis
Values are expressed as mean ± s.e.m. Between-group comparisons were carried out by analysis of variance with post-hoc comparison of means by Bonferroni or Tukey test. Alpha error was equal to 0.05. Group comparisons of gross lesions were performed with Kendall's Taub and by calculating confidence intervals. Bivariate correlation (α = 0.001) and multiple regression (α = 0.05) were carried out to tease out the effect of ventriculomegaly on area of both WM and gray matter ROIs. Group comparison of optical density of neurofilament staining was carried out with nonparametric Mann-Whitney test.
Results
Hypertonia was observed in ~ 80% of the newborn survivors at P1 after antenatal H–I as found previously (Derrick et al, 2004). Fixed involuntary postures were almost always associated with hypertonia and were also observed in stillbirths. In the hypoxia group, apparently healthy kits were chosen and compared with kits with severe hypertonia and postural deficits and naive controls.
Persistence of Motor Deficits
One of the diagnostic criteria for cerebral palsy in humans is persistent motor deficits. To assess persistence of hypertonia in the rabbits, we kept P1 kits alive by artificial feeding and housing them in a thermoregulated incubator. The hypertonic kits had 100% mortality if left to the care of the rabbit dam. Because of intensive care, the mortality of the hypertonic kits at P5 was 47%. Hypertonia and postural deficits did not change from P1 to P11 in all hypertonic survivors (see accompanying video).
White Matter Injury is Associated with Hypertonia
We asked if WM injury demonstrated by changes in FA was associated with hypertonia and motor deficits after H-I. Fractional anisotropy was significantly lower in internal capsule, corona radiata, and corpus callosum in the hypertonic group compared with controls or the nonhypertonic group (Table 1). Values of FA in WM tracts of nonhypertonic kits were not significantly different from controls. However, 40% of nonhypertonic kits had FA values higher than 99 percentile of controls in the internal capsule and corpus callosum. This suggests that some kits recover from the H–I and overcompensate with even higher FA levels.
Parameters of white matter tracts in control and hypoxia groups with and without hypertonia at P1
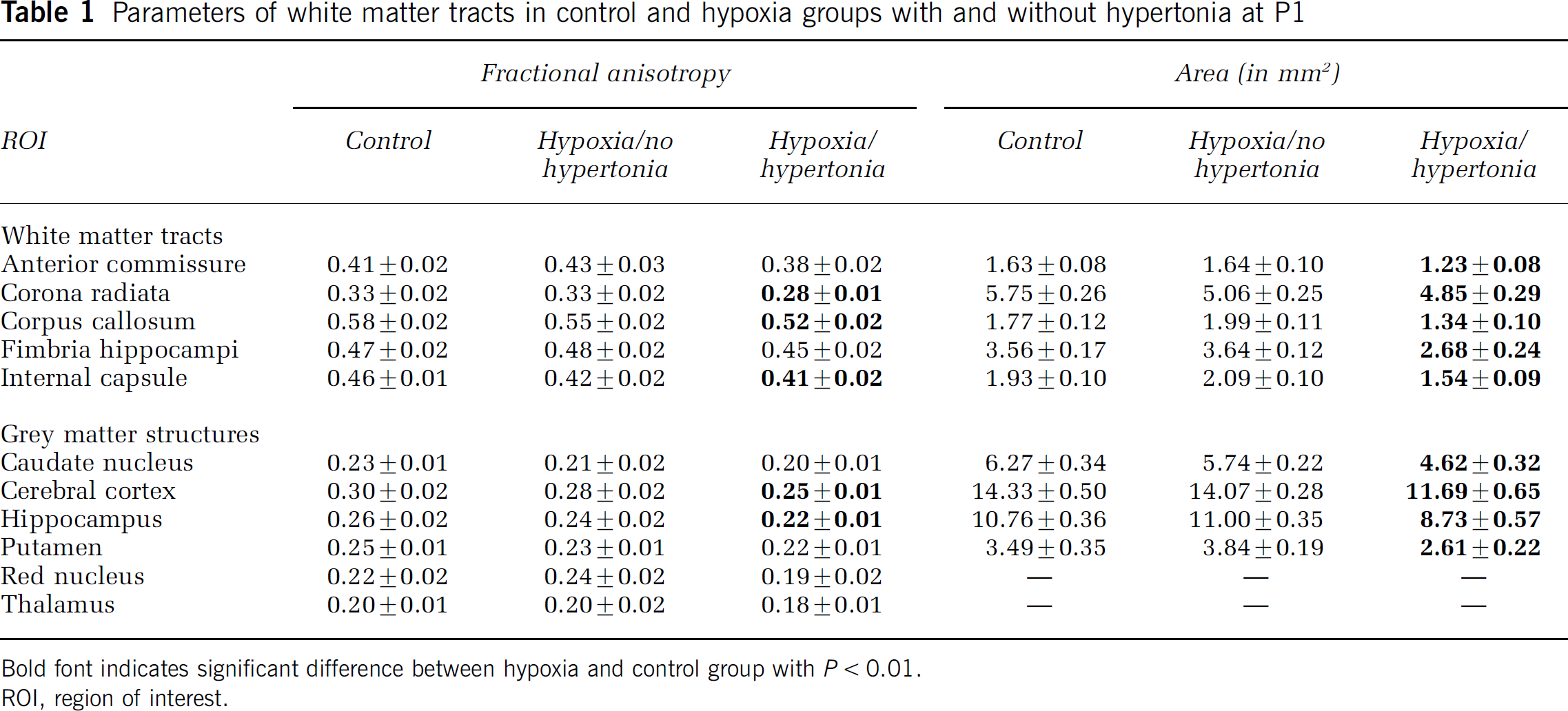
Bold font indicates significant difference between hypoxia and control group with P < 0.01.
ROI, region of interest.
Microstructural Changes in Injured White Matter Revealed by Diffusion Tensor Imaging Component Analysis
Fractional anisotropy is a directionally invariant index of the diffusion anisotropy and reflects the variance among the three eigenvalues of the diffusion tensor. The first eigenvalue of the diffusion tensor is presumed to measure the diffusion rate along WM fibers; thus, a measure of axial diffusivity. An estimate of radial diffusivity was obtained from the average of the second and third eigenvalues, which reflects diffusion across the fibers in a cylindrical shape.
Analysis of the separate contribution of the directional diffusivities reveals that decrease of WM FA in hypertonic kits occurs because of the increase of the radial diffusivity (Figure 1, P < 0.03, one-way ANOVA). Increase in radial diffusivity was significant in corpus callosum, corona radiata, and internal capsule.
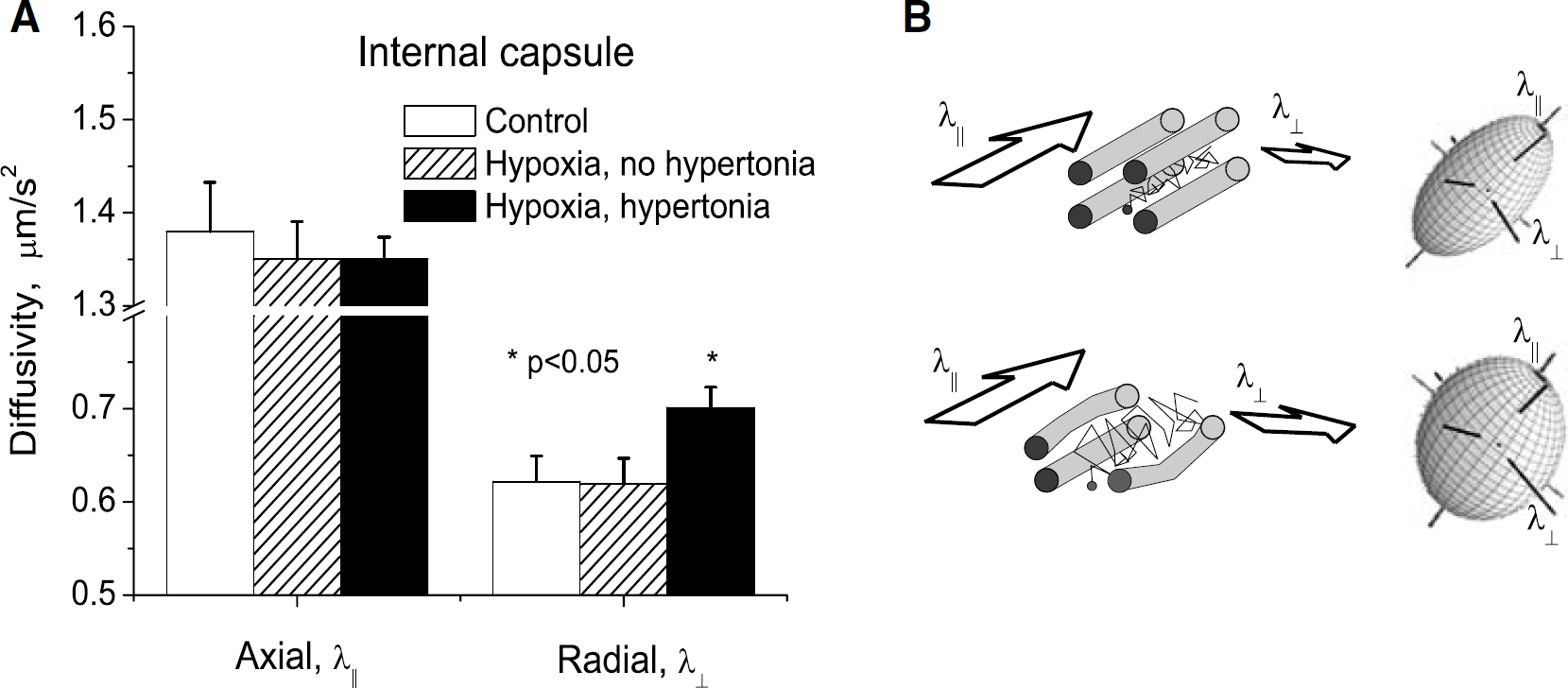
Fractional anisotropy components in internal capsule. (
Brain Injury is Increased in Hypertonic Rabbit Kits with Ventriculomegaly
We observed several cases of rabbit kits with unilateral lateral ventricle enlargement and corresponding postural deficit on the contralateral side (Figure 2). We therefore analyzed whether the dynamic change of FA is affected by the presence or absence of ventriculomegaly.
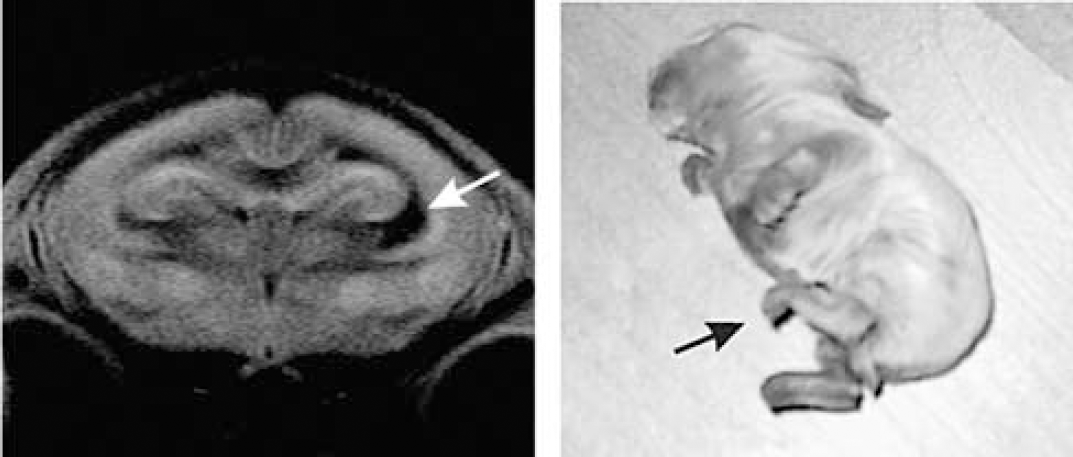
Unilateral lesion. Note enlarged ventricle near right internal capsule (arrow). Left hindlimb is hypertonic (arrow), right hindlimb has normal tone.
The decrease in FA at P1 was more pronounced in corona radiata, corpus callosum, and internal capsule in the ventriculomegaly group compared with rabbits without ventriculomegaly (Table 2). Apparent diffusion coefficient was significantly higher only in corona radiata (Table 3).
Parameters of white and gray matter in Hypoxia group with hypertonia, subdivided by presence of ventriculomegaly
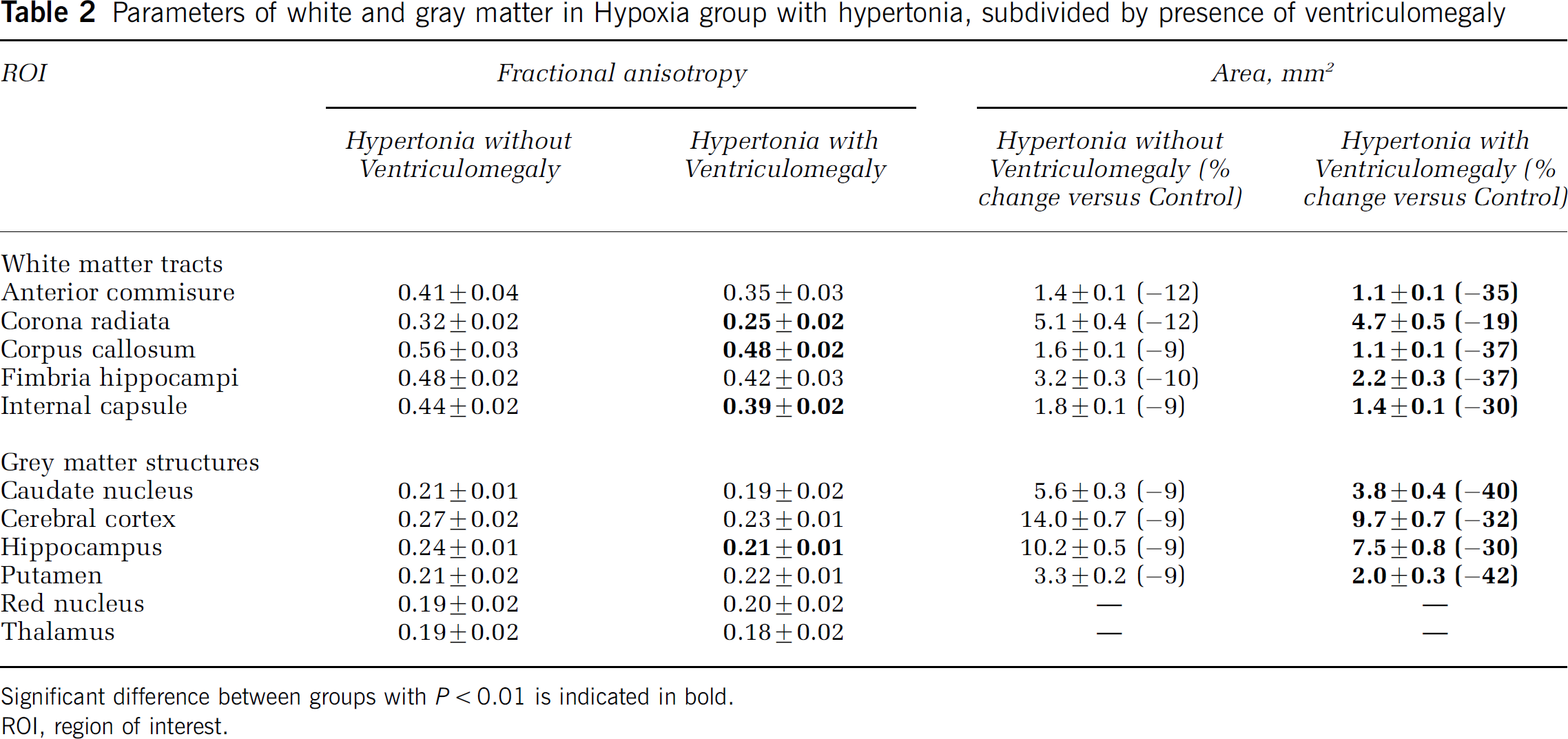
Significant difference between groups with P < 0.01 is indicated in bold.
ROI, region of interest.
Apparent diffusion coefficient (μm2/s) of white and gray matter in control and hypoxia groups at P1
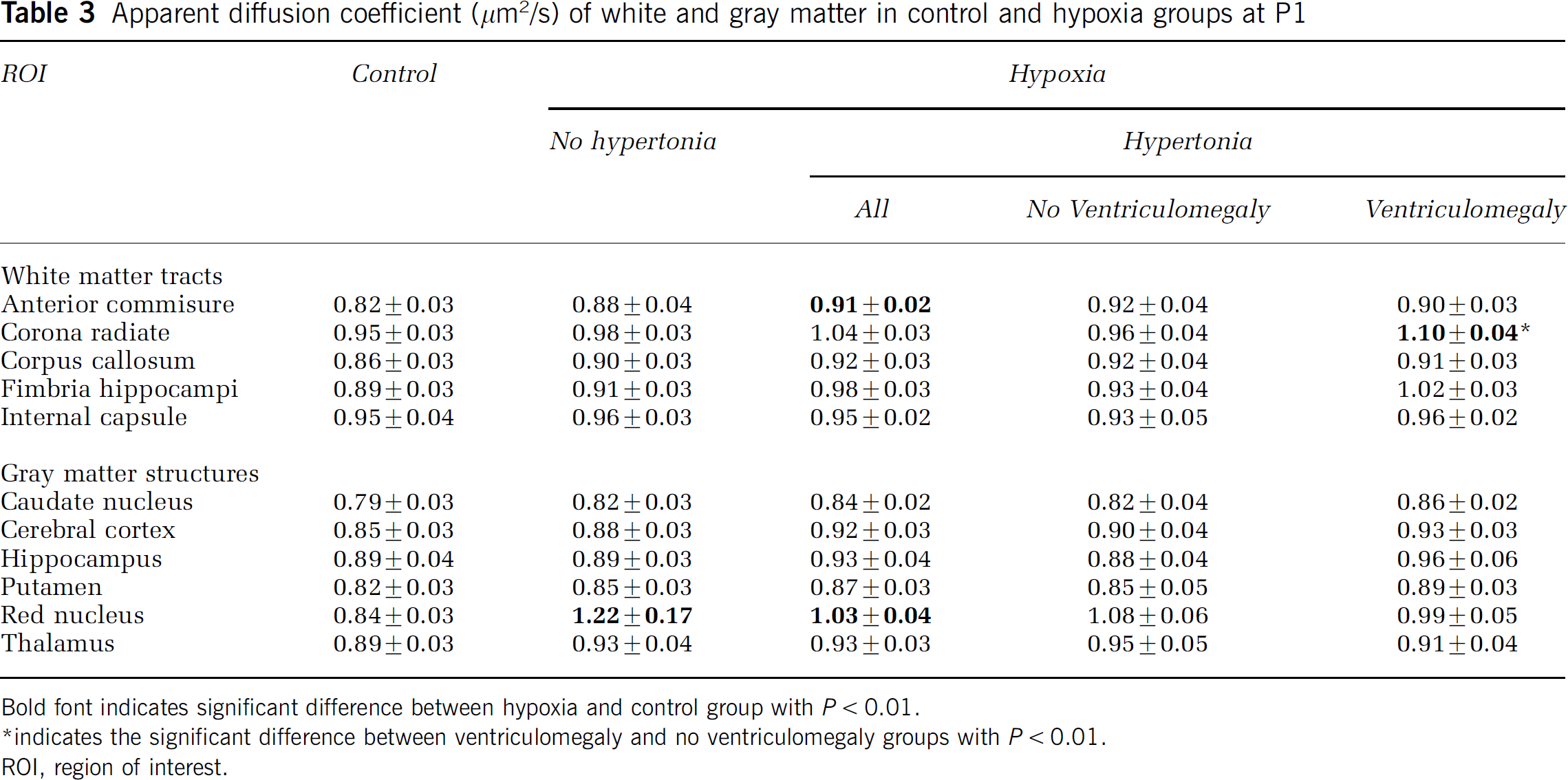
Bold font indicates significant difference between hypoxia and control group with P < 0.01.
indicates the significant difference between ventriculomegaly and no ventriculomegaly groups with P < 0.01.
ROI, region of interest.
Ventriculomegaly is Associated with Further Decrease of Fractional Anisotropy from P1 to P5
Figure 3 shows the dynamic change of FA from P1 to P5 in corpus callosum, internal capsule, and fimbria hippocampi in a subset of animals. The normal development is that FA increases from P1 to P5 significantly in all WM fibers by 7% to 10% (Figure 3). In non-hypertonic rabbits, FA showed a similar increase as in controls, except for internal capsule where the increase (16%) was even higher than controls (not shown for brevity). Fractional anisotropy in the hypertonic group without ventriculomegaly was initially lower than controls at P1 and the rate of increase from P1 to P5 did not reach control levels (Figure 3). In contrast, with added ventriculomegaly and hypertonia, FA decreased significantly from P1 to P5 in the WM fiber tracts (Figure 3). Thus, the change of FA correlated with the severity of the injury, with the best rabbits similar to controls, the hypertonic rabbits without ventriculomegaly showing initial injury and some recovery (but not to control levels), and the hypertonic rabbits with ventriculomegaly showing the same degree of initial injury but worsening of injury with time.
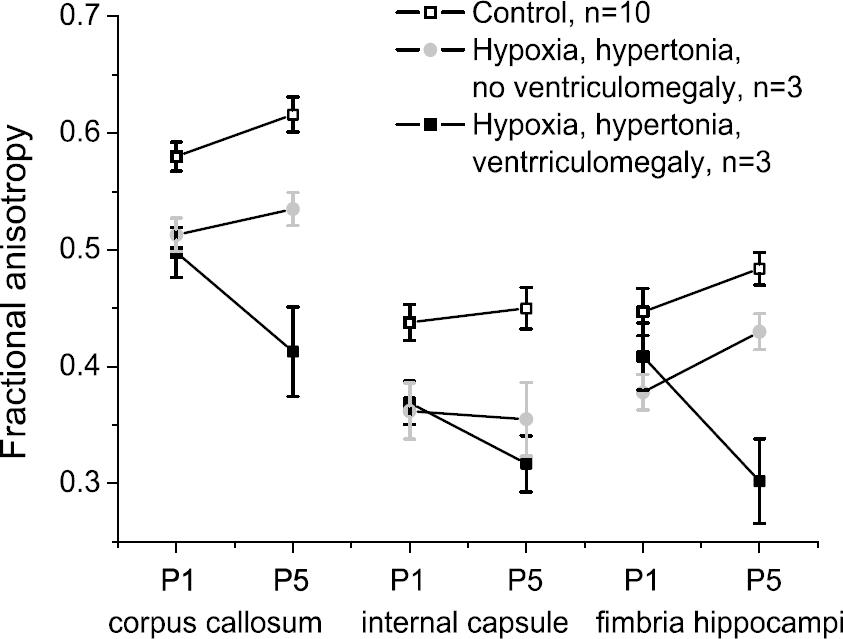
Dynamics of Fractional anisotropy (FA) from P1 to P5 in rabbits after hypoxia-ischemia. Subset of P1 rabbits were reared to P5 and scanned serially. In control animals, FA significantly increased in all white matter fibers by 7% to 10%. Fractional anisotropy in hypertonic groups 2 and 3 were lower than controls at P1. In group 2, FA increased by P5, but did not reach values of controls. In contrast, FA in group 3 failed to increase and even significantly decreased (P < 0.01) in corpus callosum and internal capsule.
White Matter Size was Decreased in Hypertonic Rabbits with Concomitant Decrease in Diffusion Anisotropy
The size of WM of hypertonic rabbit kits was decreased compared with controls (Table 1). There was a gradation observed in the hypoxic groups with the size not different from controls in the nonhypertonic group, a slightly less, albeit nonsignificant, decrease in area in the hypertonic group without ventriculomegaly, and the greatest decrease in the hypertonic group with ventriculomegaly.
The effect of FA decrease, ventriculomegaly, and incidence of hypertonia on WM loss in post-hypoxic kits was analyzed using both bivariate correlation and multiple regression. For the internal capsule, FA and presence of hypertonia significantly correlated with area of fiber tract at a priori significance of P < 0.001, with Pearsons correlation coefficients (r) of 0.446 and —0.489, respectively. Ventriculomegaly had r of −0.434, P =0.0012. For corpus callosum, results were similar: only FA and group significantly correlated with the area at P < 0.001 (r = 0.477 and —0.499, respectively); ventriculomegaly had r —0.436, P =0.011. Linear combination of FA, hypertonia, and ventriculomegaly in multiple regression accounted for 31% and 35% of the variance in internal capsule and corpus callosum (P < 0.0005 and P < 0.0001, respectively). Beta weights (multiple regression coefficients) and uniqueness indices were reviewed to assess the relative importance of the three variables in the prediction of WM size. In the internal capsule, none of the variables displayed significant beta weights or uniqueness indices. In the corpus callosum, FA and hypertonia accounted for 7.5% and 6.8% of the variance in WM size, P < 0.05 while ventriculomegaly had no effect. This analysis suggests that H-I-mediated injury caused the decrease in size of the WM fiber tracts and the decrease was not due to pressure from increased cerebrospinal fluid in cases of ventriculomegaly.
This is supported by examining the scatter plot of FA and the area of internal capsule measured on the same MRI slice (Figure 4). There was no significant correlation found between FA and area in controls (Figure 4A) and non-hypertonic animals (Figure 4B), indicating that the decrease in WM area does not cause a decrease in FA. Only in hypertonia, was a significant correlation found (Pearson r =0.51, P =0.006) (Figure 4C). Predominantly low values of FA and area in hypertonic kits (Figure 4C) indicate that the loss of the WM is accompanied by the injury of the remaining tissue. The findings in the three groups thus indicate that primary H–I injury to WM is the common origin of processes leading to loss of FA and area. The two hypertonic kits with hydrocephalus (black squares in Figure 4C) have relatively little WM loss and high FA values, suggesting that tissue compression actually raises FA. Values of FA less than 0.35 and area less than 2.2 mm (hatched area in Figure 4D) predict 100% hypertonia. Most of the correlation of FA with area is explained by the changes with radial diffusivity (λ┴) as the radial diffusivity was also significantly correlated with area (r = —0.35, P =0.005) (Figure 5).
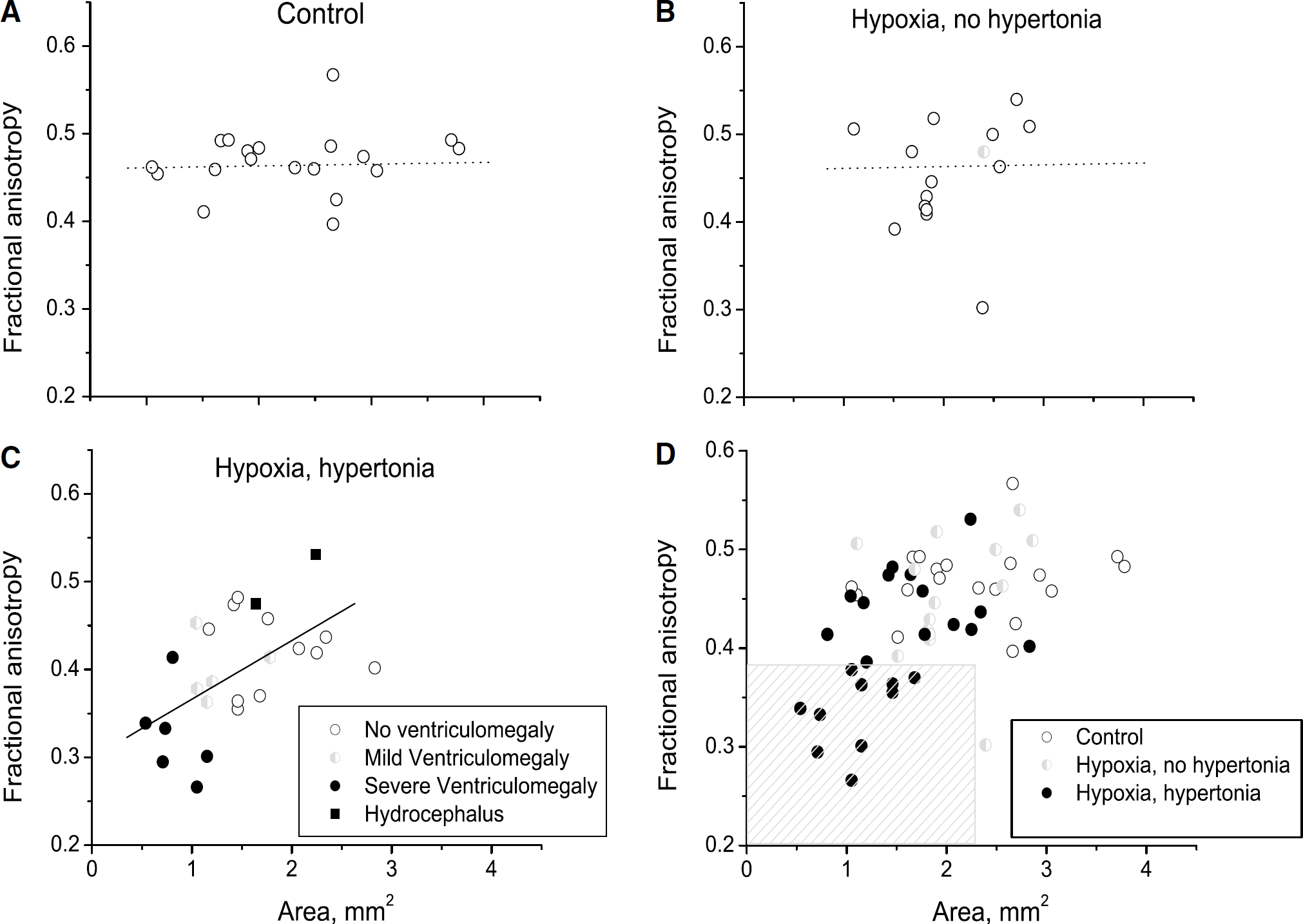
Scatter plot of FA (ordinate) versus area of internal capsule (abscissa), measured on the same MRI slice. Area is not related to FA in controls (
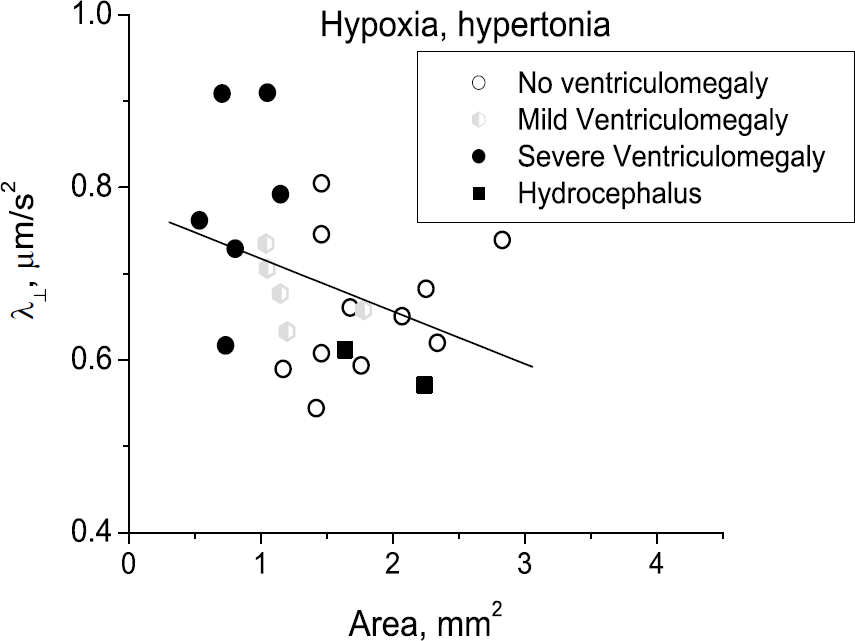
Decrease in Fractional anisotropy in hypertonic group can be explained by increased radial diffusivity (λ┴) that correlated with area of white matter. Note weak relationship with ventriculomegaly. Low values of radial diffusivity in two hypertonic kits with hydrocephalus (black squares) indicate compression of fibers.
Area and Density of White Matter is Decreased on Immunostaining
Staining for phosphorylated neurofilaments on Figure 6A–D shows dramatic reduction in area and density of fiber tracts in the corpus callosum, and the internal capsule. The loss of immunoreactivity for phosphorylated neurofilaments in both the corpus callosum and internal capsule may explain the loss of FA and area on MRI. White matter fibers were less dense, less compact and less streamlined in the hypertonia group (Figure 6F) compared with controls (Figure 6E), explaining the increase in radial diffusivity and the reduction of FA in WM observed in hypertonia. Hypertonic animals showed a significant loss of phosphorylated neurofilaments (Figure 6G) in the corpus callosum (OD 17.3±5.5 versus 44.8±6.7 in controls) and internal capsule (24.2±3.4 versus 37.7±3.4 in controls) (Mann–Whitney test, P < 0.05). The hypoxic but nonhypertonic group were not analyzed statistically because of n = 2, but this group had values that tended to be higher than that in controls, in the corpus callosum (50.8±3.8), and internal capsule (60.6±1.2).
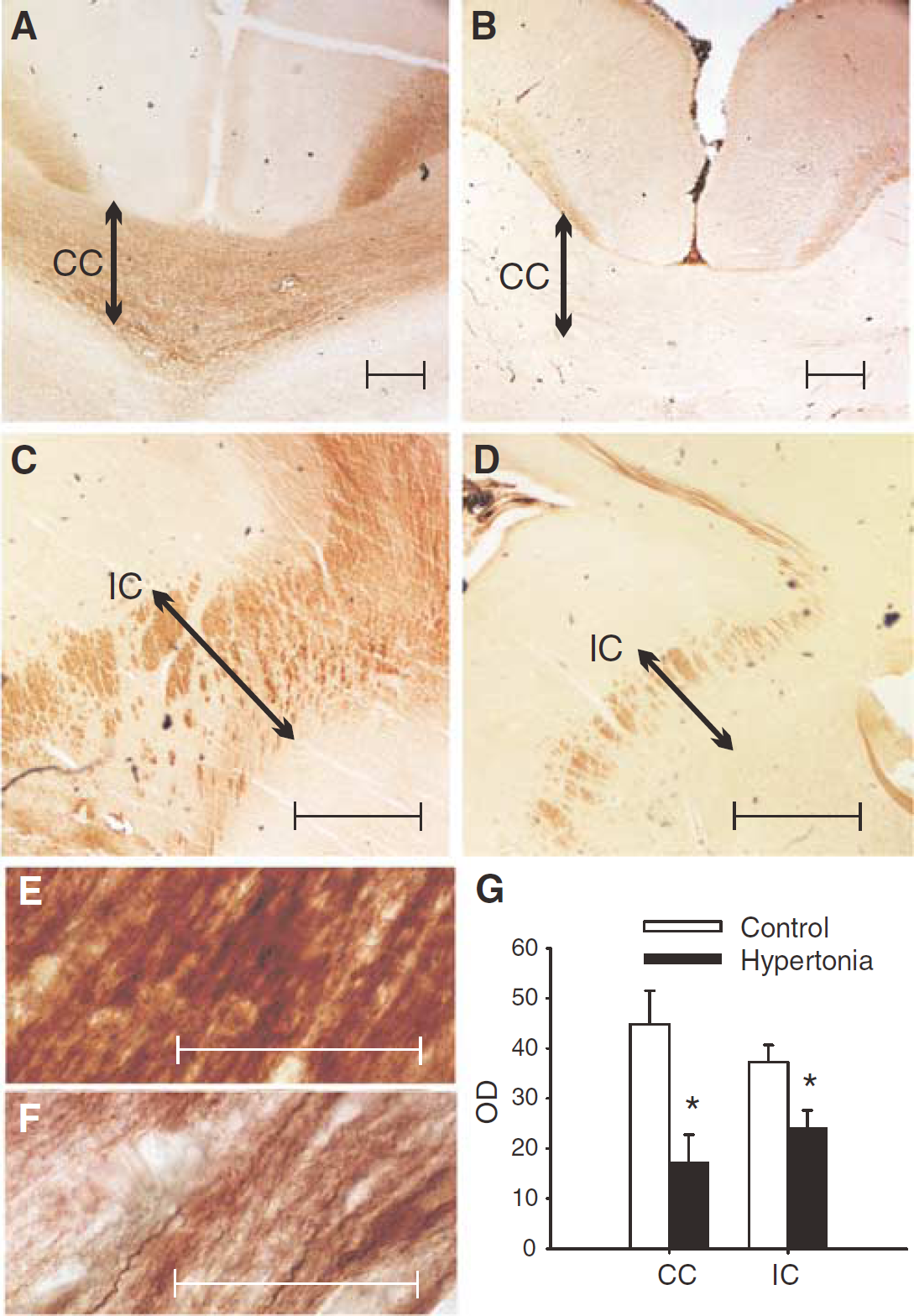
Staining for phosphorylated neurofilaments SMI312 at the level of the anterior thalamus shows dramatic reduction in hypoxic animals with hypertonia (right sided panels) in the corpus callosum (CC, panel
Selective Motor Injury is Associated with Localized Decrease of Fractional Anisotropy in Internal Capsule
Some newborn rabbits exhibited selective motor injury including unilateral left/right (Figure 2) or asymmetrical front/hindlimb impairment. Previous studies in the rat have delineated the internal capsule topography (Coleman et al, 1997) with the fibers serving the hindlimb distinctly separate from that of the forelimb. In a case of selective hindlimb hypertonia, the decrease of FA was more pronounced in the area corresponding to the fibers from hindlimb motor cortex, extrapolated from rat topography.
Fractional Anisotropy in Gray Matter Shows a Larger Decrease
We have previously shown that the normal trend for FA in gray matter is to decrease with increasing gestational age (Drobyshevsky et al, 2005), similar to rodents and in humans (McKinstry et al, 2002). Interestingly, FA in the hippocampus and cortex was lower in groups after H–I and significantly lower in hypertonic kits (Table 1). In contrast to FA, ADC values showed a greater change in H–I rabbits only in anterior commissure and red nucleus (Table 3).
Gray Matter Size was Decreased in Hypertonic Rabbits
Similar to WM, the size of gray matter decreased in the hypertonia group compared with controls or the non-hypertonic group (Table 1). The loss of gray matter was greatest in hypertonic rabbits with ventriculomegaly. However, using similar analysis to the WM fiber tracts, neither FA nor hypertonia nor ventriculomegaly alone accounted significantly for the variance in size of cerebral cortex, although the three variables together significantly accounted for 21% of the variance of the size (P < 0.01). In hippocampus, ventriculomegaly accounted for 12.1% of the variance of the size, P < 0.01, and FA and group did not have significant affect on size. This again suggests that the decrease of gray matter can be explained only to a small extent by ventriculomegaly.
Discussion
This is the first study to show that fetal H–I causes WM injury that has a strong association with hypertonia and postural deficits. This study also shows that WM injury defined by MRI indices of diffusion anisotropy can even predict a subpopulation of hypertonia. We found disruption of WM microstructural organization, that is, WM injury, as reflected by lower FA and area in internal capsule and corpus callosum in the hypoxic-ischemic kits with hypertonia and postural deficits.
White matter damage is considered to be the predominant form of perinatal hypoxic brain injury in human neonates (Huppi, 2004; Iida et al, 1995; Neil et al, 2002). White matter injury is observed more often in preterm babies, the most vulnerable population for cerebral palsy, and is exemplified by periventricular leukomalacia with cystic lesions and diffuse changes, detected by conventional imaging and DWI (Counsell et al, 2003; Volpe, 2003). Changes in FA are detectable not only near the site of primary injury (the periventricular WM), but also in the posterior limb of the internal capsule, indicating a disturbance of developing fibers that project through this area (Huppi et al, 2001). In nearterm to term infants with hypoxic-ischemic encephalopathy, cerebral cortical damage is mainly restricted to areas of primary myelination, such as internal capsule and adjacent subcortical WM (Azzarelli et al, 1980, 1996). The development of CP occurs after the perinatal period, usually after a year. As it is impossible to get histologic evidence of WM injury (Volpe, 2001) in CP patients, noninvasive markers like MRI are useful to investigate dynamically perinatal WM injury. Fractional anisotropy of internal capsule at near-term or term was decreased in three preterm infants that developed CP later (Arzoumanian et al, 2003). Fractional anisotropy has been shown to be decrease in two CP patients at 6 years (Hoon et al, 2002) and five CP patients at 12 to 16 years (Thomas et al, 2005).
Our rabbit model is the first animal H–I model to mimic the motor deficits in CP. This allowed the investigation of both hypertonic and non-hypertonic rabbit kits, with the non-hypertonic kits serving as a hypoxic control. For H–I to cause hypertonia, evidently a significant WM injury needs to be present causing a decrease in both the number and density of fiber tracts. This is supported by the findings of decreased FA, increased radial diffusivity and decreased area on MRI, as well as neurofilament immunostaining. Diffusion tensor imaging becomes predictive of motor deficits only if a combination of FA and area is used. Also of note is that hypertonia is observed as early as E30 in rabbits while myelination does not start till P5. This suggests that axonal degeneration may be a more likely initial event than primary WM injury. Further evidence of axonal/neuronal injury are cases of selective motor deficits (left versus right or fore limb versus hindlimb deficits) when portions of the fibers in the internal capsule are selectively affected and have a lower FA.
Although FA in the non-hypertonic group was not significantly different from controls, there was a subpopulation in the non-hypertonic group that had even larger FA than controls. This could be because of a preconditioning response of the fetal hypoxic event before the stress of labor at term. It is possible that disorganized fibers are pruned, and new regenerating axons are better organized to explain the greater FA. The regeneration could be a result of stimulation of neurogenesis that is observed after moderate H–I (Ferriero, 2005).
The dynamic changes of FA reveal the response of the WM to injury. The rabbit findings confirm the clinical suspicion of ventriculomegaly being ominous indicator of morbidity (Gaglioti et al, 2005), The maturation pattern of FA of WM in hypertonic kits without ventriculomegaly from P1 to P5 were not different from controls. However, FA in this group did not recover to the level of controls by P5. It is unknown whether FA reaches a nadir before P1 (E32) some time after the H–I insult. In the hypertonic kits with ventriculomegaly, FA did not increase with age and even decreased sometimes. This indicates that FA can reflect repair mechanisms in WM, and serial measurements of DTI parameters give a more complete picture of injury and recovery. This corroborates with a serial diffusion MRI study in newborns, where ADC and FA of selected regions with WM injury failed to follow normal maturation trends (Miller et al, 2002). Of note is the fact that we could do MRI only on the survivors at P5. A percentage of very severely affected rabbit kits died before P5, which implies that the FA results at P5 could be an underestimation of the actual brain injury and an overestimation of recovery.
As to the cellular basis of WM maturation, it is known that oligodendrocytes play an important role, facilitating fiber tract alignment and packing that subsequently affects FA (Drobyshevsky et al, 2005; Rasband et al, 1999). The maturation of diffusion anisotropy in fetal and perinatal rabbit development coincides with increased immature oligodendrocytes as well as compound action potential area (Drobyshevsky et al, 2005). Thus, FA could be considered as a dynamic, in vivo MRI surrogate of WM fiber organization and function. Increase of axial diffusivity in pre-myelinated WM is believed to be associated with decreased cell and membrane density, reduced cohesiveness and compactness of the fiber tracts, increased extra-axonal space (i.e., less packing), changes in extracellular and intracellular matrix (Beaulieu, 2002). One of the possible explanations for disruption of structural organization of WM in hypertonic kits could be initial injury to oligodendroglia after H-I. Depletion of immature oligodendrocytes has been suggested as an important pathophysiologic event leading to WM injury of periventricular leukomalacia (Back et al, 1998). Fractional anisotropy of the WM can also be affected by retrograde axonal degeneration because of cortical neuronal death after H-I. There may be direct injury to neurons and axons, or combination of both ‘oligopathy’ and ‘axonopathy’ as proposed for periventricular leukomalacia (Dammann et al, 2001). Although technically challenging, future fiber-oriented approach and detailed histology will help elucidate the contributions of ‘oligopathy’ and ‘axonopathy’ to the development of hypertonia.
Previous studies have investigated cell death or apoptosis by histopathology examination and tried to characterize injury to gray or WM. The majority of rodent neonatal H–I models show both gray and WM injury on histopathology examination. Despite the massive cortical lesions in the Rice-Vannucci model, observable motor deficits are most often absent (Liu et al, 2004; McQuillen et al, 2003). Subtle motor deficits can be picked up by sophisticated neurobehavioral testing (Bona et al, 1997; Liu et al, 2004). Selective WM injury by histopathology has been described in a postnatal rodent model (Follett et al, 2004; Follett et al, 2000). Subtle motor deficits are observed in this model (Follett et al, 2004), unlike the clear hypertonia and postural deficits in our model. Other studies showing a more selective WM injury and relative sparing of gray matter have been observed in a bilateral carotid occlusion model (Uehara et al, 1999), in a model with mild injury with a short duration of H–I at a lower body temperature (Qiao et al, 2004), and in a model of global prenatal hypoxia (Baud et al, 2004). Again, motor deficits have not been reported in these models.
The significant reduction of cortical and subcortical gray matter suggests a mixed nature of the brain injury in our model, with involvement of WM and gray matter. Selective WM injury probably does not occur in premature infants as newer studies show concomitant decrease in cortical gray matter by MRI (Inder et al, 1999b) and cortical dysplasia in overlying areas by neuropathology (Huppi, 2004). Although, gray matter injury, indicated by cystic lesions and tissue loss, correlated with degree of brain injury and hypertonia in our model (Tan et al, 2005), it was unclear if gray matter injury was the cause of hypertonia, as 55% of hypertonic animals did not have appreciable changes in cortex and striatum volumes. Although there was an increase in the incidence of gross lesions such as ventriculomegaly (lateral and III/IV), hydrocephalus, and cystic lesions in the hypertonic group (Tan et al, 2005), there were many cases of hypertonia that did not have any gross lesions and some non-hypertonic kits also exhibited gross lesions. This would imply that identification of gross lesions on structural images would not necessarily predict motor deficits and possibly are a manifestation of a downstream event that caused the motor deficits. This is suggested by the lack of correlation between area and FA between control and non-hypertonic hypoxic groups. Furthermore, increasing ventriculomegaly correlated with decreased FA, but hydrocephalus had high FA (Figure 4C).
In summary, WM injury that causes a concomitant fall in FA and volume of WM is probably responsible for hypertonia. The number of fiber tracts is less dense and decreased in hypertonic animals. Below a threshold, the combination of FA and area can identify a population with hypertonia. Plasticity of the brain reaction to the prenatal hypoxic insult can be assessed by dynamics of FA changes in early postnatal development.
Footnotes
Acknowledgements
The authors thank Wei Hsueh, MD, Professor of Pathology, Northwestern University for the helpful comments. The study was funded by Grants NIH NS 43285, NS41476, 1 S10 RR15685-01.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
