Abstract
Hypoxic-ischemic (HI) encephalopathy is a cerebrovascular injury caused by oxygen deprivation to the brain and remains a major cause of neonatal mortality and morbidity worldwide. Therapeutic hypothermia is the current standard of care but it does not provide complete neuroprotection. Our aim was to investigate the neuroprotective effect of oleuropein (Ole) in a neonatal (seven-day-old) mouse model of HI. Ole, a secoiridoid found in olive leaves, has previously shown to reduce damage against cerebral and other ischemia/reperfusion injuries. Here, we administered Ole as a pretreatment prior to HI induction at 20 or 100 mg/kg. A week after HI, Ole significantly reduced the infarct area and the histological damage as well as white matter injury, by preserving myelination, microglial activation and the astroglial reactive response. Twenty-four hours after HI, Ole reduced the overexpression of caspase-3 and the proinflammatory cytokines IL-6 and TNF-α. Moreover, using UPLC-MS/MS we found that maternal supplementation with Ole during pregnancy and/or lactation led to the accumulation of its metabolite hydroxytyrosol in the brains of the offspring. Overall, our results indicate that pretreatment with Ole confers neuroprotection and can prevent HI-induced brain damage by modulating apoptosis and neuroinflammation.
Introduction
Hypoxic-ischemic (HI) encephalopathy is a neonatal cerebrovascular injury caused by insufficient blood flow and oxygen supply to the brain. 1 This pathology affects 1-8/1000 live births1 –3 and remains a major cause of death and disability in the neonatal population, with most deaths arising in low- and middle-income countries.3,4 Common neurological deficits associated with neonatal HI include cerebral palsy, seizures, visual and hearing impairment, learning and behavioral disabilities, and motor deficits. 5 The pattern and extent of brain injury can vary from mild to severe damage, 3 depending on the severity and duration of the hypoxic insult, the gestational age, and the degree of brain maturation. 6
Inflammation, oxidative stress, and apoptosis contribute to the pathophysiology of HI, which involves three phases.7,8 The primary energy failure phase results from the reduced cerebral blood flow, which causes mitochondrial dysfunction, overproduction of reactive oxidative species (ROS) and excitotoxicity, initiating the ischemic cascade. Minutes after the HI there is a strong inflammatory response that includes glial activation, proinflammatory cytokine release and blood-brain barrier (BBB) breakdown, resulting in brain edema and triggering apoptosis and necrosis.9,10 In the latent phase, cerebral circulation is restored, and this constitutes the optimal time window for therapeutic interventions. The secondary energy failure phase starts 6–48 hours after the HI insult and can last several days. 8 During this phase oxidative stress, excitotoxicity, and inflammation processes are exacerbated, resulting in seizures, cytotoxic edema, impaired cerebral energy metabolism, and neuronal cell death.7,8
Therapeutic hypothermia has become the standard treatment for term or near-term (≥36 weeks of gestation) newborns with moderate to severe HI encephalopathy 11 , significantly reducing in mortality and neurodevelopmental disability. 12 However, 40–50% of neonates treated with hypothermia still die or develop chronic neurological sequels. 13 As such, it is imperative to develop novel neuroprotective strategies for the prevention and treatment of HI, to ideally cover a greater percentage of affected newborns. There is a growing interest in investigating natural compounds whose bioactive properties can target the ischemic cascade, potentially reducing HI injury.14 –16 Indeed, in vivo studies have shown that several phenolic compounds such as resveratrol,17 –22 pterostilbene,23,24 quercetin,25 –27 icariin,28,29 or cannabidiol30 –32 can exert neuroprotection against neonatal HI when administered prophylactically or after HI injury, reducing brain damage and associated cognitive impairments (reviewed in 16 ).
Oleuropein (Ole) is a glycosylated secoiridoid found in olive trees (Olea europaea L.) and constitutes the main bioactive compound in the leaves and unprocessed olives. Ole has shown neuroprotective effects against different neurological disorders such as Alzheimer’s disease, traumatic brain injury, spinal cord injury, and stroke (reviewed in33,34). In regard to cerebral ischemia/reperfusion injury, Ole has shown antiapoptotic, antioxidant, anti-inflammatory, and antithrombotic activities in vivo, reducing brain injury and neurological deficits when administered as a pretreatment, 35 post-treatment,36,37 and as a dietary supplementation of Ole-enriched olive leaf extract.38 –40 Ole pretreatment has also shown similar biological activities in vivo against other ischemia/reperfusion injuries, including cardiac41 –46 and kidney. 47
To date, however, little is known about the potential neuroprotective benefits of Ole in the context of neonatal HI, with the exception of a recent study which showed that post-treatment with Ole in combination with therapeutic hypothermia protected against white matter loss and preserved myelination in a swine model of HI. 48 Given the properties of Ole in different neurological and ischemia/reperfusion disorders, we aimed to elucidate whether pretreatment with Ole could protect the neonatal brain and reduce HI-induced histological and cellular damage. To this end, we employed the widely used Rice-Vannucci model of neonatal HI on postnatal day seven (P7) mice, a postnatal stage at which the maturity of the central nervous system in rodents is similar to that of human term neonates (34–36 weeks of gestation).49,50 We administered Ole before HI induction and examined the effects on infarct size, white matter integrity, microglia, astrocytes, and apoptotic and inflammatory markers. Our results show for the first time that Ole pretreatment confers neuroprotection in a dose-dependent manner in a mouse model of neonatal HI encephalopathy.
Materials and methods
Animals
This study was approved by the Animal Ethics Committee of “Consejería de Agricultura, Ganadería, Pesca y Desarrollo Sostenible; Junta de Andalucía” (ref. 17/05/2021/0078). All procedures complied with European Directive 2010/63/EU and Spanish Royal Decree 53/2013, and were reported following ARRIVE guidelines. Mice litters at postnatal day (P7) were obtained from the Animal Production and Experimentation Service at the Institute of Biomedicine of Seville, and kept in a climate-controlled environment on a 12-hour light/dark cycle with food and water ad libitum.
Neonatal hypoxia-ischemia mouse model
Hypoxic-ischemic brain injury was induced in seven-day-old CD1 mice from both sexes following the Rice-Vannucci method, 51 with some modifications (Figure 1(a)). Briefly, pups weighting ≥4 g (mean weight 5.0 ± 0.5 g) were anesthetized with inhaled isoflurane and the left common carotid artery (LCCA) was isolated and ligated. Pups were returned to their mother for approximately 2 h and then exposed to 90 min of hypoxia with 9% O2 in a hypoxic chamber (O2 Control InVivo Glove Box, Coy Laboratory Products Inc.). Animals were placed on a heating pad during surgery, recovery, and hypoxia. Sham group pups underwent anesthesia and LCCA isolation, without ligation or hypoxia. The mortality rate among HI pups was 17% (15/87), all surviving HI pups were included in the study (see Supplementary Methods for details).
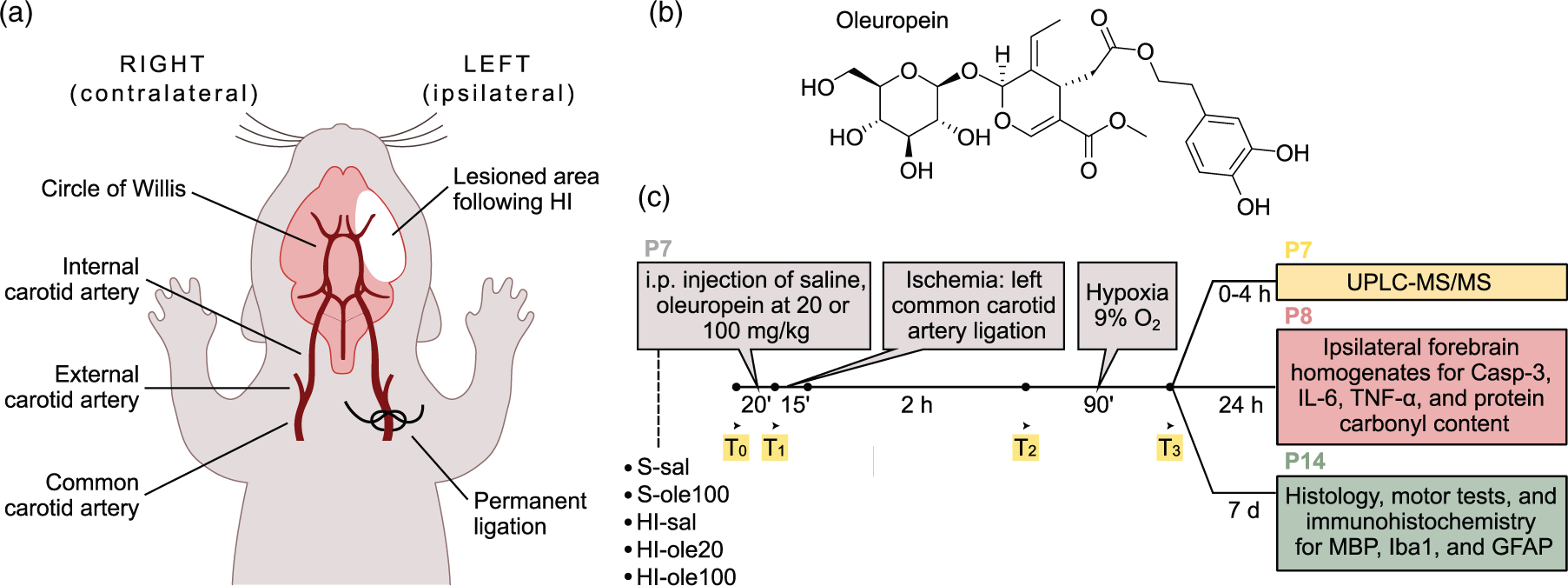
Summary of the experimental design. (a) Schematic representation of the modified Rice-Vannucci protocol for the induction of neonatal HI to postnatal day 7 (P7) mouse pups by unilateral ligation of the left common carotid artery followed by hypoxia (90 min with 9% O2). (b) Molecular structure of Ole and (c) timeline of the study. Five groups were included: sham pups, which only suffered the exposition of the left common carotid artery but no hypoxia and received either saline (S-sal) or Ole at 100 mg/kg (S-ole100), and HI pups which received saline (HI-sal), Ole at 20 mg/kg (HI-ole20) or Ole at 100 mg/kg (HI-ole100). Brain and plasma samples were collected at several timepoints (T0–T3) at P7 for ultra-performance liquid chromatography-mass spectrometry (UPLC-MS/MS), brain homogenates were prepared at P8 for biochemical assays, whereas histology, immunohistochemistry, and motor tests were performed at P14. All the samples used in the study were coded and all the experiments were performed by blinded researchers.
Experimental groups and oleuropein administration
Oleuropein (Ole; Figure 1(b)), purchased from TCI Chemicals (O0420; CAS 32619-42-4, >98% purity) was dissolved in ethanol, stored at –20°C, and diluted 1:10 in saline (0.9% NaCl) prior to use. Ole was administered by intraperitoneal (IP) injection 20 min before LCCA ligation at a dose of 20 or 100 mg/kg, as done in similar studies.17,19,23,28 Equal volumes of saline (10% ethanol; 10 ml/kg) were used as vehicle. Ole concentrations were based on previous studies of cerebral and cardiac ischemia/reperfusion where Ole was administered as IP pretreatment.35,52
Pups were randomly allocated into five experimental groups (Figure 1(c)); GraphPad was used to randomly assign subjects to treatment groups (https://www.graphpad.com/quickcalcs/randomize1/). Sample size was based on similar studies.17,19,53 –55 Sham group pups received saline vehicle (S-sal) or Ole at 100 mg/kg (S-ole100). HI group pups received saline vehicle (HI-sal), Ole at 20 mg/kg (HI-ole20), or Ole at 100 mg/kg (HI-ole100). Seventy-five pups from seven litters were used for histology and immunohistochemistry studies at P14 (n = 10 S-sal, n = 10 S-ole100, n = 18 HI-sal, n = 19 HI-ole20, and n = 18 HI-ole100). Since no apparent differences were observed between S-sal and S-ole100 groups and only partial neuroprotection was seen in HI-ole20 group, we opted against subjecting these groups to biochemical assays to avoid unnecessary animal testing. Thus, n = 40 pups from five litters were used for biochemical assays at P8 (n = 8 S-sal, n = 17 HI-sal, and n = 15 HI-ole100). Average litter size (13 ± 2 pups) and mothers age (16 ± 3 weeks) were consistent across all litters.
Neonatal motor tests
Grip strength 56 and locomotor aptitude were measured at P14 by a blinded researcher to assess motor deficiencies. The pups underwent three trials for each test, and the mean output of the test performance was calculated (Supplementary Methods).
Histology and immunohistochemistry studies
At P14, pups were deeply anesthetized with inhaled isoflurane and perfused intracardially with ice-cold saline followed by 4% paraformaldehyde (PFA) dissolved in phosphate-buffered saline (PBS; pH 7.3). Brains were dissected and fixed in 4% PFA for 2 h at 4°C, dehydrated with graded ethanol and xylene, embedded in paraffin, and cut into 7 µm-thick coronal sections using a microtome, from Bregma + 1.10 to –2.80 mm, according to the Franklin and Paxinos atlas. 57 Sections were kept on coded slides, with the code not being broken until all measurements were made. Coronal sections were used for histology and immunohistochemistry studies, all measurements were performed by a researcher who was blind to the experimental group of the samples.
Sections stained with methylene green were used to quantify the extent of tissue loss, as a readout of brain infarct, and to assess the severity of tissue damage using a semi-quantitative histopathological scoring system developed in our laboratory (Fig. S1), and modified from similar scales.17,51,58 –61 Besides, sections were immunolabelled with antibodies against myelin basic protein (MBP), ionized calcium binding adaptor molecule 1 (Iba1), and glial fibrillary acidic protein (GFAP). Further details are provided in the Supplementary Methods, Fig. S1 and Table S1.
Biochemical studies in brain homogenates
Twenty-four hours post-HI insult, at P8, pups were anesthetized, and forebrains were dissected and homogenized. Brain homogenates were used for biochemical assays including caspase-3 (Casp-3), interleukin 6 (IL-6), tumor necrosis factor α (TNF-α), protein carbonyl content, and 8-hydroxydeoxyguanosine (8-OHdG). All protocols were conducted by a blinded researcher. See Supplementary Methods for details.
Quantification of ole and related metabolites in brain and plasma by UPLC-MS/MS
Thirty-five P7 pups from four litters were used to quantify Ole and its metabolites (hydroxytyrosol, tyrosol, and oleacin) by ultra-performance liquid chromatography-mass spectrometry (UPLC-MS/MS)62,63 after IP administration of Ole. Fifteen pups received saline and fifteen received Ole at 100 mg/kg. Both groups were sacrificed at three timepoints post-IP injection: 20 min (T1), before LCCA ligation; 2:20 h (T2), post-LCCA ligation, pre-hypoxia; and 3:50 min (T3), post-LCCA ligation and 90 min of hypoxia. Five pups without IP injection served as baseline controls (T0; Figure 1(c), highlighted in yellow). Pups were anesthetized with isoflurane, blood was collected from the right auricle and centrifuged to obtain plasma (2,500 g, 15 min at 4°C). Pups were perfused with ice-cold saline and brains were dissected. Brain and plasma samples were frozen in liquid nitrogen and stored at –80°C before UPLC-MS/MS analysis by a blinded researcher.
Besides, we conducted a pilot study to explore whether Ole or its metabolites reached the pups after maternal supplementation with Ole during pregnancy and/or lactation. A pregnant mouse received Ole at 100 mg/kg/day from E12.5 to P7 via oral gavage, similar to other maternal supplementation studies.53,64 At P0, five age-matched pups from a non-supplemented dam were transferred to the Ole-supplemented dam. Brain and plasma samples were collected at P7 for UPLC-MS/MS; five P7 pups from a non-supplemented dam served as baseline controls. See Supplementary Methods for details.
Statistical analysis
Plots and statistical analyses were performed using GraphPad Prism 9.0. Data are reported as the median with the interquartile range (IQR) shown in brackets and are plotted as box plots. Normality was analyzed using Shapiro-Wilk test. Kruskal-Wallis test followed by Dunn's multiple comparisons test was used for the comparisons between of three or more groups, including histological studies, immunohistochemical studies (comparisons between groups), biochemical studies, and motor tests. All groups were compared against S-sal, and HI-ole20 and HI-ole100 were compared against HI-sal. Wilcoxon matched-pairs signed rank test was used for the comparisons between hemispheres in the immunohistochemical studies. Chi-square test was used for comparison of the histological damage score in different brain regions. Linear regression fit was used to study the correlation between MBP staining and tissue loss. P < 0.05 was considered statistically significant.
Results
Ole ameliorates HI-induced brain damage and tissue loss
At P14, one-week post-HI, weight gain, walking ability, and grip strength were measured. HI-sal and HI-ole20 groups showed less weight gain than S-sal, but no differences in motor abilities were observed (Table S2). Mice were then euthanized, and brains collected for histology (Figures 2, S1 and S2). Whole-brain coronal sections revealed that the hippocampus, cortex, and striatum were the most susceptible regions to HI, with the severity of brain damage ranging from mild tissue damage to extensive infarct of the ipsilateral hemisphere, as shown before.51,61,65 Subfield images of the CA2/3 region of the hippocampus and parietal cortex revealed significant cell loss and damage to neurons in HI-sal, as observed fewer intact neurons and pyknotic-like nuclei (Figure 2(c′) to (c″)). The HI-ole20 group showed mild hippocampal cell loss but not in the cortex (Figure 2(d′) to (d″)), whereas over half of HI-ole100 samples resembled controls (Figure 2(e′) to (e″)).
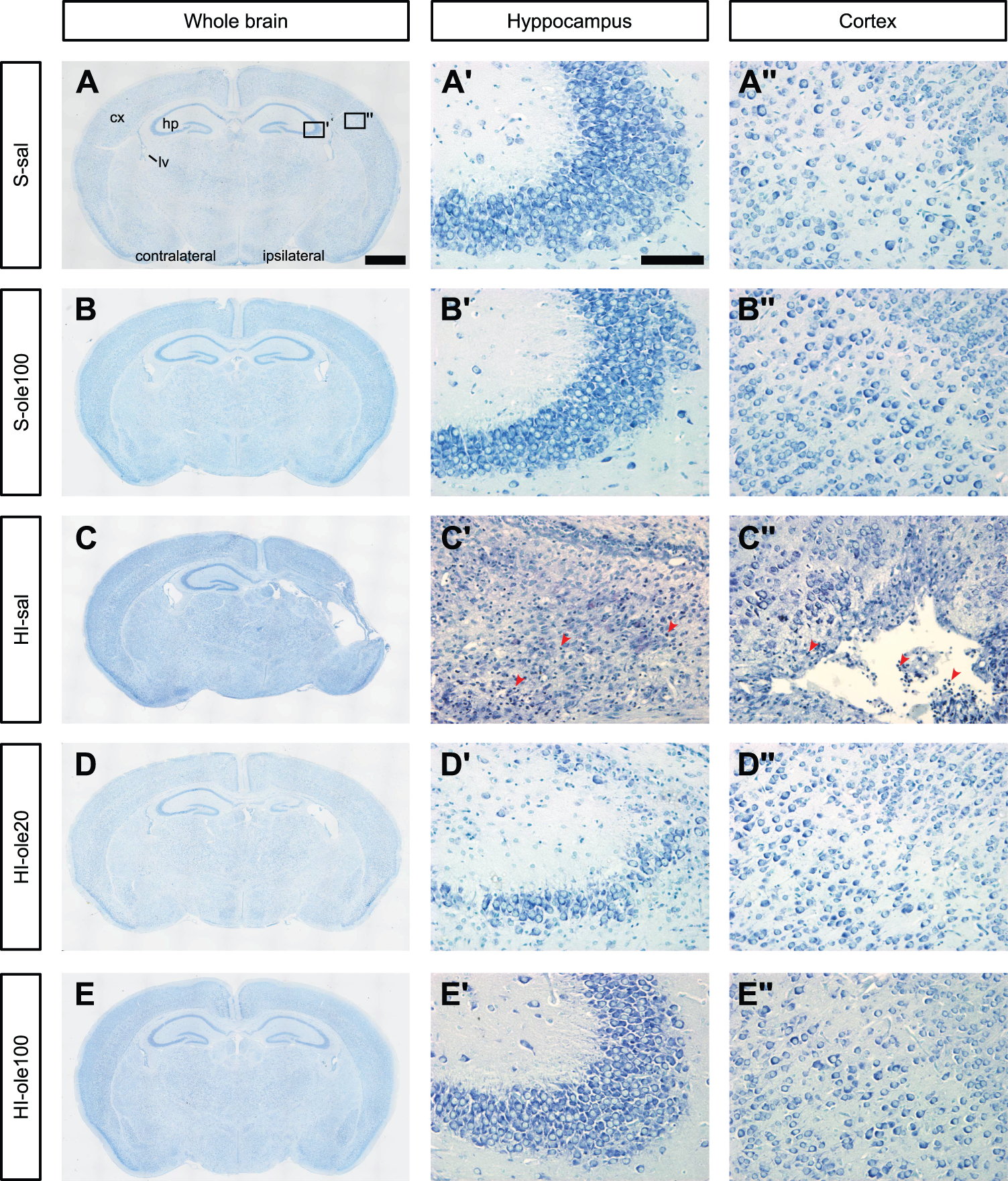
Neonatal HI causes ipsilateral brain damage and cell loss which are reduced by Ole pretreatment. (a–e) Representative whole-brain micrographs of methylene green coronal sections at the hippocampal level of the different experimental groups at P14, a week after the HI insult. (a) S-sal and (b) S-ole100, with normal brain morphology; n = 10 each. (c) HI-sal, showing brain infarct accompanied by hippocampal (hp) and cortical (cx) tissue loss, dilation of the lateral ventricle (lv), and ipsilateral hemisphere asymmetry; n = 18. (d) HI-ole20, showing hippocampal hypotrophy and lateral ventricle dilation but no cortical damage; n = 19 and (e) HI-ole100, with a macroscopic morphology similar to the control group; n = 18. The median histological damage score of each group was used to select a representative image. (a′–e′) Magnification of the CA2/3 region of the hippocampus and (a″–e″) the parietal cortex, as shown in A. HI-sal presented cell loss and neuronal damage in both brain regions, as observed by the lower number of morphologically intact neurons and the presence of pyknotic-like nuclei (red arrowheads). Scale bars: 1000 µm (a–e), 100 µm (a′–e″).
Histology images were used to calculate the percentage of tissue loss in the ipsilateral hemisphere as a readout of the infarct area (Figures 3(a), S2F). HI-sal group had a median tissue loss of 42.8% (IQR 25.1–69.3%, n = 18), while Ole pretreatment reduced tissue loss dose-dependently: HI-ole20 had a median tissue loss of 13.6% (6.8–42.6%, n = 19) and HI-ole100 of 8.6% (4.5–36.1%, n = 18). Macroscopic brain damage was evaluated using a histology damage scoring system (Fig. S1). As for tissue loss, Ole pretreatment ameliorated global histological damage dose-dependently (Figure 3(b)), with median histological damage scores of 20 points in HI-sal (IQR 16–26), 14 points in HI-ole20 (IQR 4–21), and 1 point in HI-ole100 (IQR 0–19). Tissue loss and histology scores reflected the variability in HI-induced brain damage both in the untreated and pretreated HI groups. Although there was a slight trend towards more severe damage in males than females, there were no statistically significant differences between sexes or litters (Fig. S3).
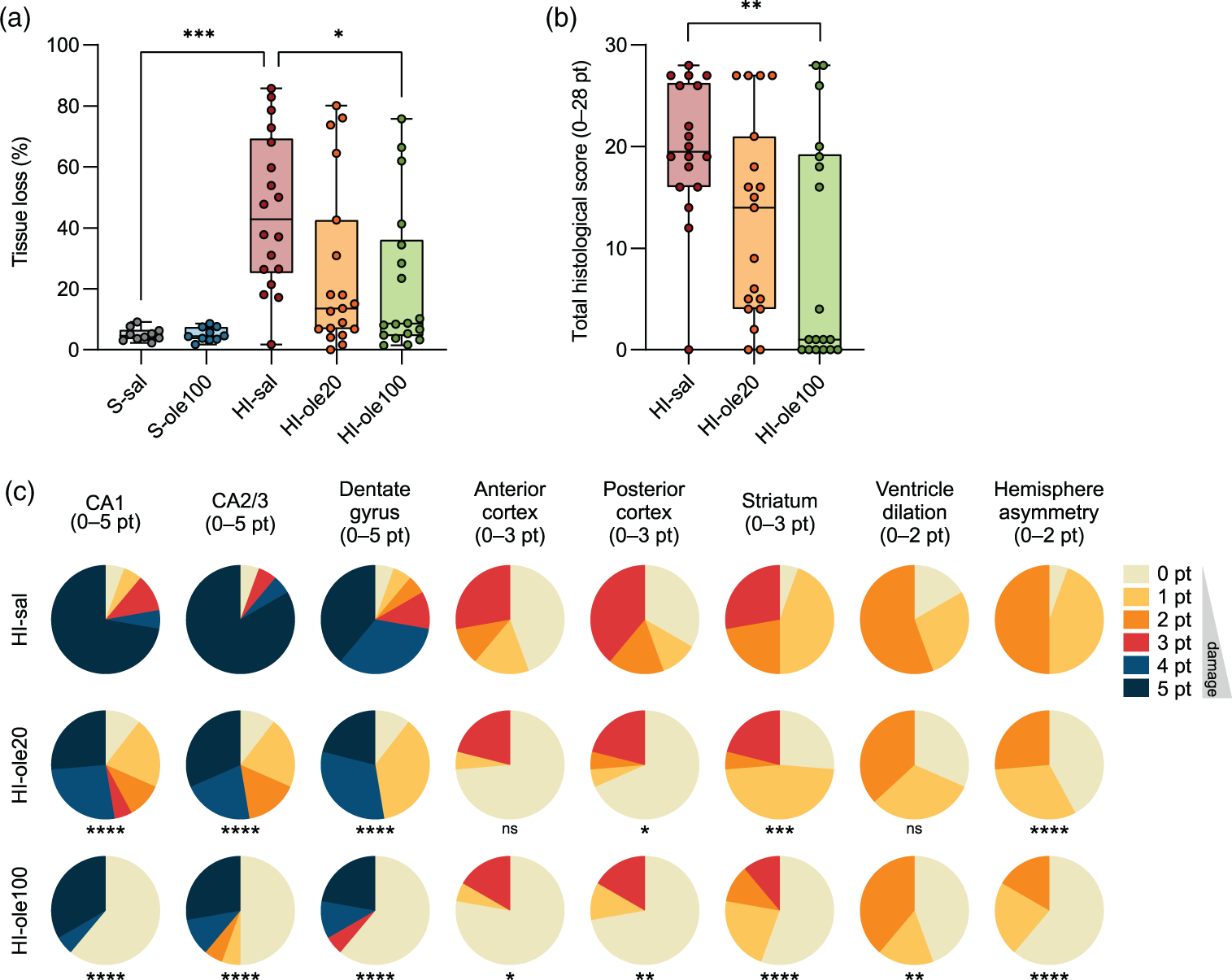
Ole pretreatment reduces tissue loss and ameliorates HI-induced brain damage in a dose-dependent manner. (a) Percentage of tissue loss in the ipsilateral hemisphere at the hippocampal level, measured from coronal brain sections stained with methylene green at P14, a week after the HI insult. (b) Total histological damage score, from 0 to 28 points, evaluated from histology images. (c) Histological damage score in different brain regions, pie charts represent the proportion of samples with a given score. The total histological damage score was the sum of all the evaluations in C. Histology images were evaluated blindly and independently by two researchers; all S-sal and S-ole100 samples received a score of 0 points and thus are not shown in B–C. Statistical significance was calculated using Kruskal-Wallis test followed by Dunn's multiple comparisons test (a–b) or Chi-squared test (c); *P < 0.5, **P < 0.01, ***P < 0.001; n = 10–19 per group.
The histological damage score was also used to categorize the damage in the most susceptible regions to HI: the hippocampus, cortex, and striatum (Figure 3(c)). In the HI-sal group, the hippocampus and striatum showed hypotrophy or tissue loss in 94% (17/18) of pups. Cortical tissue loss (67%, 12/18), ventriculomegaly, observed as the dilation of the ipsilateral lateral ventricle (83%, 15/18), and ipsilateral hemisphere asymmetry (94%, 17/18) were common histological features. In severe cases, the infarct comprised almost the entire ipsilateral hemisphere (28%, 5/18). In the HI-ole20 group, a smaller proportion of brains showed cortical tissue loss (32%, 6/19) and although most samples showed some degree of hippocampal hypotrophy or tissue loss (89%, 17/19), the histological damage was milder than in the HI-sal group. Ventriculomegaly was common (63%, 12/19), but ipsilateral hemisphere asymmetry was less frequent (58%, 11/19). Over half of HI-ole100 samples resembled sham (56%, 10/18), but some showed hippocampal (39%, 7/18) or striatal (44%, 8/18) damage, accompanied by cortical tissue loss (28%, 5/18) in the most severe cases. Ventriculomegaly (56%, 10/18) and ipsilateral hemisphere asymmetry (39%, 7/18) were less frequent than in HI-sal. Overall, the results summarized in Figure 3 suggest Ole pretreatment ameliorate HI-induced tissue loss and histological damage dose-dependently, although there is a proportion of severe HI cases that cannot been prevented by Ole.
Ole reduces white matter injury and preserves myelination
Immunohistochemistry studies at P14 revealed white matter injury in the HI-sal group, shown by a substantial loss of MBP immunostaining in the ipsilateral hemisphere affecting the cortex, striatum, and external capsule. HI-ole20 brains also exhibited some MBP loss, especially in the striatum, whereas most HI-ole100 brains were similar to sham (Figure 4(a)). White matter integrity was analyzed by measuring the area of MBP staining in each hemisphere normalized to the contralateral hemisphere and the absolute MBP coverage in the striatum, confirming the macroscopic observations: there was a significant decrease in ipsilateral MBP coverage in HI-sal and, less strongly, in HI-ole20, but not in HI-ole100 (Figure 4(b) and Table S3). This suggests that Ole can preserve white matter integrity and protect against HI-induced demyelination dose-dependently.
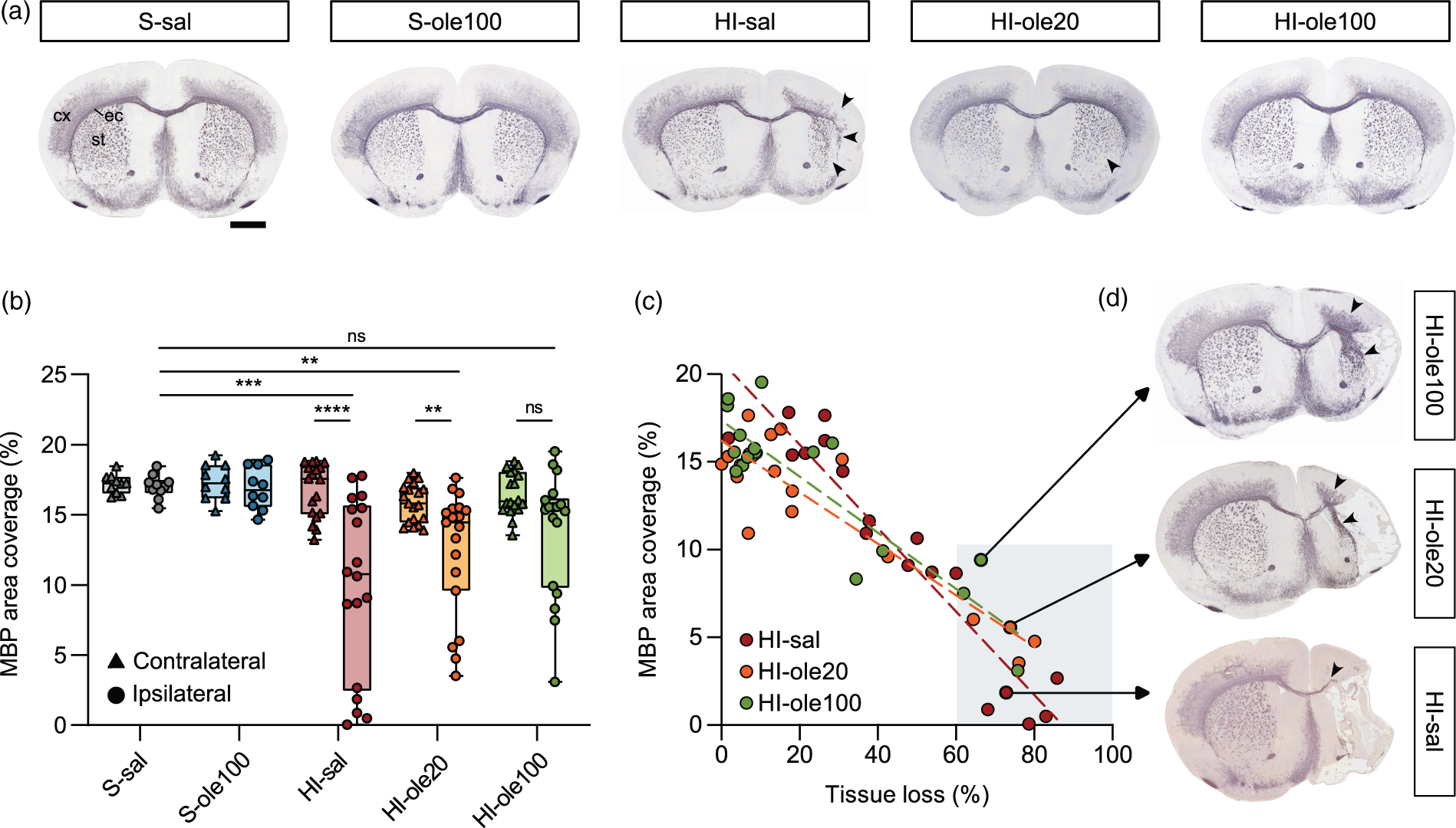
Ole pretreatment reduces white matter injury and preserves myelination in a dose-dependent manner. (a) Representative whole-brain micrographs immunolabelled for myelin basic protein (MBP) at P14, a week after the HI insult. From left to right: S-sal (n = 10); S-ole100 (n = 10); HI-sal (n = 18), showing MBP loss (marked with black arrowheads) in the external capsule (ec), cortex (cx), and striatum (st); HI-ole20 (n = 19), showing some hypotrophy in the striatum accompanied by a reduction in MBP; and HI-ole100 (n = 18), similar to control groups. The median MBP area coverage of each group was used to select a representative image. Scale bar: 1 mm. (b) Analysis of the MBP area coverage per hemisphere in the different experimental groups. Wilcoxon matched-pairs signed rank test was used for the comparisons between hemispheres and Kruskal-Wallis test followed by Dunn's multiple comparisons test was used for the comparisons between groups; **P < 0.01, ***P < 0.001, ****P < 0.0001, n = 10–19 per group. (c) Correlation between MBP area coverage and tissue loss in the ipsilateral hemispheres; linear regression fits are shown as dashed lines. (d) Even in those brains with severe damage and tissue loss >60% (marked with a shaded grey box in c), samples pretreated with Ole showed more MBP coverage than untreated ones, as apparent from the illustrative HI-ole100, HI-ole20, and HI-sal micrographs.
Ipsilateral tissue loss was typically accompanied by reduced MBP coverage, though this correlation was weaker in pups that received Ole pretreatment (R-squared 0.90 in HI-sal, 0.83 in HI-ole20, and 0.79 in HI-ole100; Figure 4(c)). In samples with severe brain damage (tissue loss >60%; shaded area in Figure 4(c)), untreated pups showed less MBP coverage than treated ones: HI-sal group had a median ipsilateral MBP coverage of 0.9% (n = 5), HI-ole20 of 5.2% (n = 4) and HI-ole100 of 7.5% (n = 3; Figure 4(d)). This suggests that even if it does not confer complete neuroprotection, in the cases of severe tissue loss Ole can preserve myelination in the non-infarcted areas.
Ole inhibits microglial activation
We next investigated whether Ole could modulate microglia using immunohistochemistry against Iba1 at P14. We measured the number of Iba1+ cells in different brain areas that have previously shown microglial activation upon HI damage (Figure 5(a) and (b)). 66 The number of microglia was significantly increased in the ipsilateral hemisphere of HI-samples, in the cortex, the midline corpus callosum, and most strongly in the striatum. In comparison to HI-sal, Ole pretreatment at both doses significantly reduced microglia cell density in these regions (Figure 5(c) and Table S4). In the striatum, however, there was a subset of HI-ole20 (5/19) and HI-ole100 samples (5/18) that had an elevated number of microglia. These samples also showed severe brain damage in the histology images (Figure 3(c)), which suggests that there may be some severe HI cases that cannot be rescued by Ole pretreatment. Overall, these results indicate that Ole pretreatment can reduce microglial activation with a similar neuroprotective effect at 20 and 100 mg/kg.
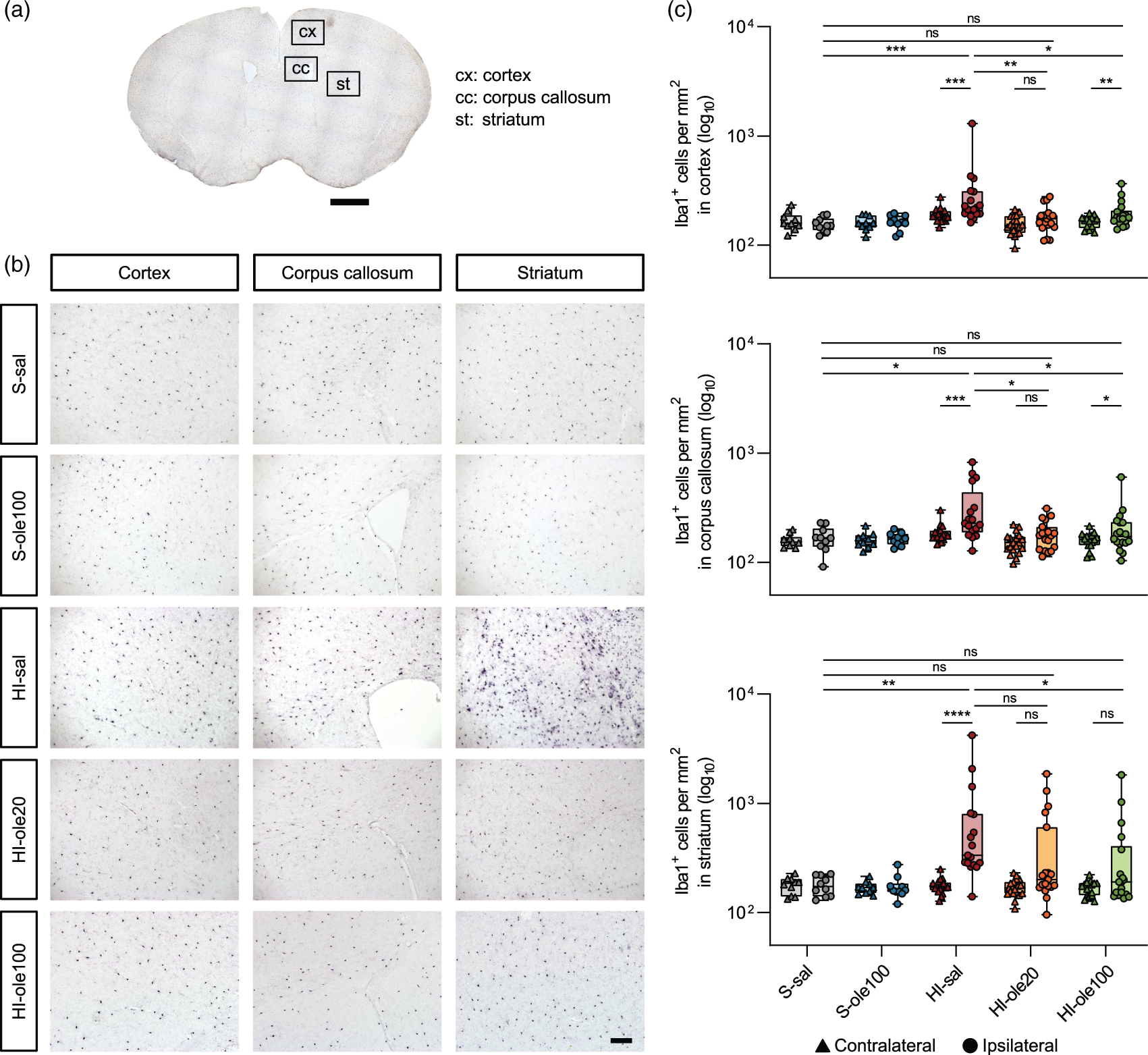
Ole pretreatment abolishes HI-induced microglial activation. (a) Microglia were visualized using Iba1 immunohistochemistry at P14, a week after the HI insult. Iba1 cell density was analyzed in the regions highlighted in boxes: the M2 supplementary motor cortex (cx), the midline corpus callosum (cc), and the striatum (st). (b) Representative coronal sections at the striatal level immunolabelled for Iba1. HI-sal presented higher number of microglia in the cortex, corpus callosum, and especially in the striatum, whereas HI-ole20 and HI-ole100 were similar to sham controls. The median number of Iba1+ cells per mm2 was used to select a representative image for each group and (c) quantification of microglial cell density, expressed as the number of Iba1+ cells per mm2 in the aforementioned brain regions. Wilcoxon matched-pairs signed rank test was used for the comparisons between hemispheres and Kruskal-Wallis test followed by Dunn's multiple comparisons test was used for the comparisons between groups; *P < 0.5, **P < 0.01, *** P < 0.001, ****P < 0.0001, n = 10–19 per group. Scale bars: 1000 µm (a), 100 µm (B).
Ole partially reduces astroglial reactive response
We next studied the effect of Ole pretreatment on astroglial reactive response using immunohistochemistry at P14 to examine GFAP expression in different brain regions. There was a significant increase in the abundance of astrocytes, measured as the percentage of area covered by GFAP staining, in the ipsilateral hemispheres of all the HI groups in the three regions studied: frontal neocortex,the parietal neocortex, and most prominently in the thalamus (Figure 6(a) and (b)). The increase in astrocyte abundance was more pronounced in the untreated HI-sal samples than in the HI-ole20 and HI-ole100 groups (Figure 6(c) and Table S5), suggesting that Ole pretreatment can partially revert the HI-induced astroglial reactive response.
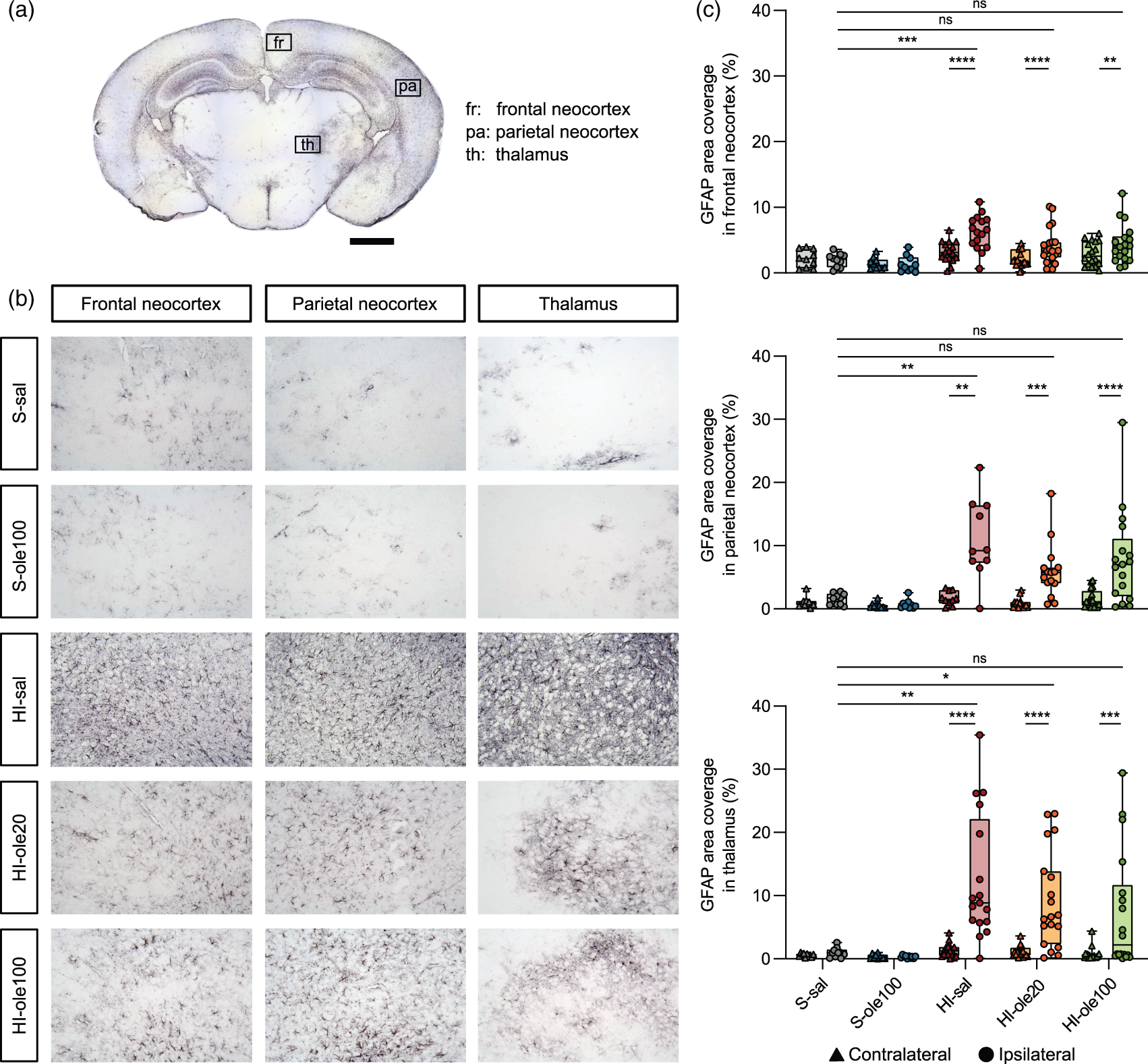
The HI-induced astroglial reactive response can be partially reduced by Ole pretreatment. (a) astrocytes were visualized using GFAP immunohistochemistry at P14, a week after HI induction. GFAP expression was analyzed in the regions highlighted in boxes: the frontal neocortex (fr), parietal neocortex (pa), and thalamus (th). (b) Representative coronal sections at the hippocampal level immunolabelled for GFAP. Astrocytes were overexpressed in the three brain regions in HI-sal group and less strongly in HI-ole20 and HI-ole100 groups. The median GFAP area coverage was used to select a representative image for each group and (c) analysis of astroglial reactive response, expressed as the percentage of area covered by GFAP staining in the aforementioned brain regions. Wilcoxon matched-pairs signed rank test was used for the comparisons between hemispheres and Kruskal-Wallis test followed by Dunn's multiple comparisons test was used for the comparisons between groups; *P < 0.5, **P < 0.01, ***P < 0.001, ****P < 0.0001, n = 10–19 per group. Scale bars: 1000 µm (a), 100 µm (b).
Ole decreases the expression of apoptotic and inflammatory markers 24 h after HI
To investigate the early effects of Ole pretreatment on HI damage, we analyzed several biochemical markers at P8, 24 h post-HI, in ipsilateral forebrain homogenates (Figure 7 and Table S6). Given that the strongest neuroprotection was seen with 100 mg/kg Ole and that there were no apparent differences between S-sal and S-ole100 groups, we focused the biochemical analyses on S-sal, HI-sal and HI-ole100 groups to avoid unnecessary animal testing.
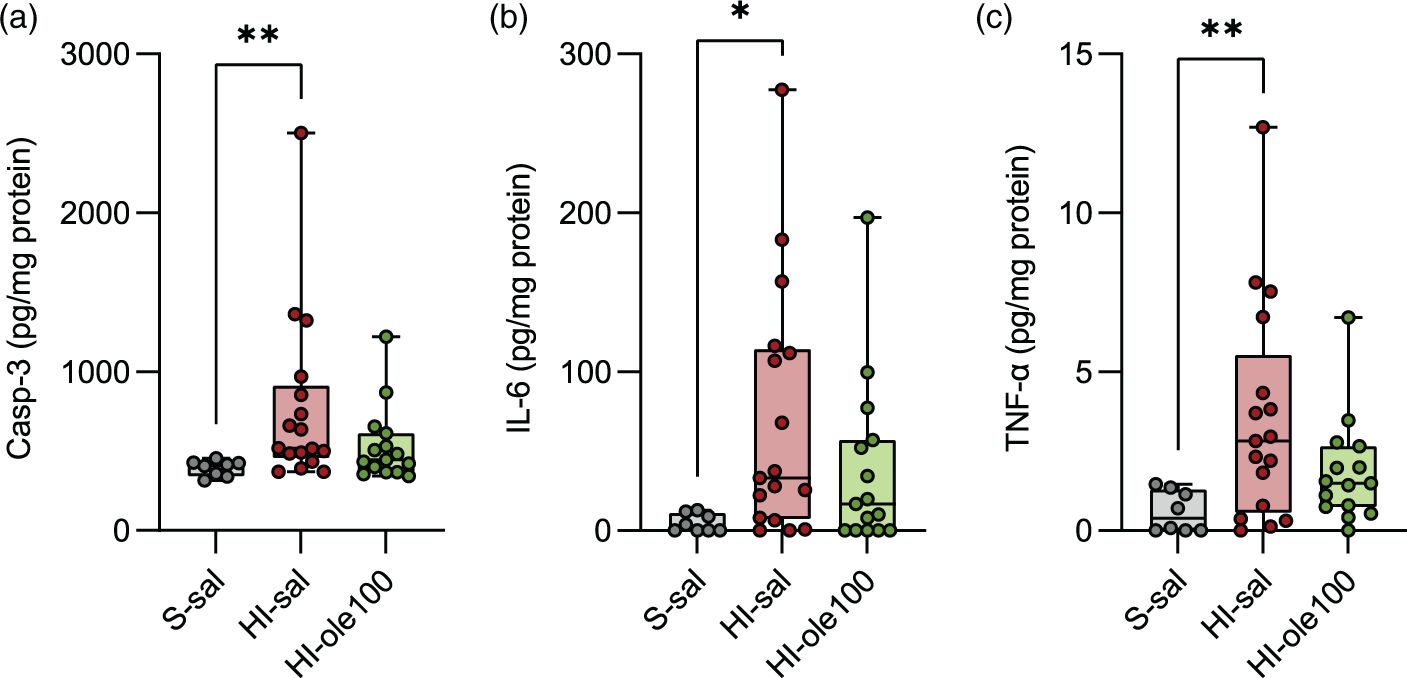
Ole pretreatment can partially abrogate the brain levels of apoptotic and proinflammatory markers 24 h after the HI insult. The levels of the apoptotic marker caspase-3 (Casp-3) and the proinflammatory cytokines IL-6 and TNF-α were studied at P8 in ipsilateral forebrain homogenates and were normalized to the total protein content. (a) Quantification of Casp-3, (b) IL-6, and (c) TNF-α by sandwich ELISA. Statistical significance was calculated using Kruskal-Wallis test followed by Dunn's multiple comparisons test, only significant changes are indicated. *P < 0.5, **P < 0.01, n = 8–17 per group.
We speculated that the reduction in infarct size observed with Ole pretreatment could be linked to the inhibition of apoptosis, which plays a prominent role in HI-induced neurodegeneration,67,68 and we examined the levels of the apoptotic marker Casp-3. Compared to S-sal, Casp-3 levels were significantly elevated in HI-sal but not in HI-ole100 (Figure 7(a)). Next, since Ole pretreatment reduced microglial activation, which contributes to the production of proinflammatory cytokines, 9 we evaluated the levels of IL-6 and TNF-α. Compared to S-sal, IL-6 and TNF-α levels were significantly upregulated in HI-sal but this overexpression was reduced in HI-ole100 (Figure 7(b) and (c)). Finally, given that oxidative stress plays a key role in HI physiopathology,7,8 we measured the protein carbonyl content and 8-OHdG levels, an oxidized nucleoside of DNA, as readouts of oxidative damage to evaluate the antioxidant activity of Ole. However, our results did not reveal any significant differences among the groups (Table S6). Overall, these results suggest that as early as 24 h after the HI, Ole pretreatment can partially reduce the levels of apoptosis and inflammation induced by HI, which may be related to the neuroprotection observed at the histological and cellular level at later stages.
Hydroxytyrosol accumulates in the neonatal brain after maternal supplementation with ole
After evaluating Ole's neuroprotective properties against HI, we quantified Ole and related metabolites in plasma and brain after its intraperitoneal (post-IP) administration. IP injection of Ole at 100 mg/kg caused a ∼10,000-fold increase in Ole plasma levels at T1 (20 min post-IP), reaching 3.8 ± 1.2 × 106 ng/ml (mean ±SD, n = 5), and returning to baseline at T2 (2:20 h post-IP; Fig. S4A). In the same timeframe, hydroxytyrosol (Htyr) plasma levels increased ∼10-fold to 15 ± 5 ng/ml (n = 5) at T1, returning to baseline at T2 (Fig. S4B). A ∼7-fold increase in Ole levels was observed in brain samples, although the concentration reached (2.1 ± 1.1 ng/g brain, n = 5) was much lower than in plasma. This increase was maintained at T2 and returned to baseline at T3 (3:50 h post-IP; Fig. S4C). There were some basal levels of oleacin in plasma (32 ± 9 ng/ml plasma, n = 5; not shown) that did not vary between groups or timepoints, suggesting they were not related to Ole administration. Tyrosol was not detected.
We next explored maternal supplementation as a route for Ole administration. Htry accumulated in the brains of P7 pups exposed to maternal supplementation with Ole during pregnancy and lactation, from E12.5 to P7 (24 ± 16 ng/g brain, n = 5) or lactation only, from P0 to P7 (27 ± 12 ng/g brain, n = 5; Fig. S4D). Neither Ole or Htyr were detected in plasma and only low levels of Ole (0.2–0.4 ng/g brain) were detected in some brain samples (3/5 from E12.5-P7 group and 2/5 from P0-P7 group; not shown). Overall, this exploratory study suggests that maternal supplementation with Ole during pregnancy and/or lactation can lead to some accumulation of its metabolite hydroxytyrosol in the brains of the offspring.
Discussion
HI encephalopathy remains one of the major causes of death and disability in the neonatal population1 –4 and despite hypothermia12,13 new therapeutic interventions are needed, with an emphasis in preventing rather than rescuing brain damage. In the present study, we showed that Ole pretreatment can protect the neonatal brain from HI at multiple levels: reducing brain infarct and tissue damage, preserving myelination, as well as decreasing microglial and astroglial cell density and several apoptotic and proinflammatory markers. Previous in vivo studies have shown that Ole exhibits antiapoptotic, antioxidant, and anti-inflammatory activities under different pathological conditions, including stroke (intracerebral hemorrhage 69 and cerebral ischemia/reperfusion35 –40) and other ischemia/reperfusion injuries.41 –44,47 Moreover, results from several clinical trials support that Ole has a safe toxicological profile, as no side effects were observed after the acute or prolonged consumption of Ole as Ole-rich olive leaf extract.70 –74
The neuroprotective effect of Ole in reducing HI-induced tissue loss and brain damage was dose-dependent, similar to previous findings in a murine cerebral ischemia/reperfusion model where Ole pretreatment (IP) reduced infarct size at 50 or 100 mg/kg, but not at 10 mg/kg. 35 In our study, although both Ole concentrations (20 and 100 mg/kg) showed a similar reduction in brain infarct area, the HI-ole20 group often showed hippocampal hypotrophy whereas over half of HI-ole100 samples did not show histological damage and were similar to sham (Figures 2(d) and (e) and 3). Some HI cases could not be rescued by Ole pretreatment (Figure 2(b) and (c)), similar to results with the polyphenol piceatannol. 53 This suggests that Ole can revert mild to moderate HI cases but not the most severe ones. Given the variability of HI brain damage and neurological deficits in humans3,6,75 and murine models of HI,51,60,61 as research in this fields evolves future studies should mention the proportion of treated samples showing recovery, as done here and by others.53,54 This would help identify which natural compounds, as well as the timing, dosage, and administration routes, are most effective against severe HI cases and aid in designing neuroprotective strategies for resistant ones. Combining Ole with therapeutic hypothermia improves post-HI subcortical white matter protection in a swine model of neonatal HI. 48 Future experiments could explore Ole as an adjuvant to other treatments or in combination with other bioactive compounds as their synergistic effects may provide greater neuroprotection.76,77
There is strong evidence that male infants are at increased risk for HI encephalopathy78,79, a pattern also seen in animal models.80 –82 Sex-specific cell death mechanisms (i.e., the proportional activation of caspase-dependent and independent apoptotic pathways)81,83,84 and gonadal hormones during the perinatal period82,85,86 may make males more vulnerable to early brain damage. In this study, we observed a slight but not significant tendency towards greater histological brain damage in males (Fig. S3A-B) and no significant sex differences in Casp-3 levels (not shown). Sexual dimorphism emphasizes the need for HI studies to include both sexes, 81 as we have done here.
Besides protecting against tissue loss, Ole pretreatment also protected the glial cell populations of the brain as myelin was preserved and both the microglial and astroglial cell density were reduced. Pre-myelinating oligodendrocyte progenitors —precursors of the oligodendrocytes responsible for the formation of myelin in the developing brain— are particularly susceptible to HI insults. 87 HI leads to myelination deficits and white matter injury that manifest as periventricular leukomalacia (in preterm neonates) or diffuse hypomyelination. 88 Here, in the untreated HI group, white matter injury was observed as loss of MBP staining in cortical and subcortical white matter, especially in the external capsule and the striatum, whereas Ole pretreatment preserved myelination in a dose dependent manner. These results are in agreement with a previous study in which Ole (administered after HI in combination with hypothermia) protected subcortical white matter from HI. 48 Our study was limited to observation of Ole clinical effect in an animal model of neonatal HI and did not explore the mechanism of action of the observed benefits. In regards to Ole mode of action, two studies by Lee et al.48,89 in a swine model of neonatal HI showed that Ole preserved MOG (myelinating oligodendrocyte glycoprotein), a component of the myelin sheath that contributes to myelin maturation, organization, and maintenance; 90 BIN1 (bridging integrator-1), a marker of mature, myelinating oligodendrocytes; 91 and Olig2, a basic helix-loop-helix transcription factor of the oligodendrocyte lineage typically overexpressed during oligodendrocyte progenitor cell proliferation and remyelination. 92 The authors concluded that Ole contributes to subcortical white matter protection by preserving myelinating oligodendrocytes, myelin density, and oligodendrocyte markers.48,89
It is noteworthy that even in those severe HI cases with >60% tissue loss, it appears that Ole preserved white matter integrity in the non-infarcted ipsilateral areas, since the MBP coverage was larger than in the untreated group (Figure 4(c) and (d)). This myelin preservation may enhance brain connectivity in those areas, the supplementary motor cortex, external capsule, and parts of the striatum, preserving their function. In this regard, pretreatment with several nutraceuticals that preserved HI-induced myelin loss—including the polyphenols resveratrol 17 or quercetin 26 and the omega-3 fatty acid DHA58, also improved long-term behavioral and cognitive deficits, such as the spatial and non-spatial working memory, anxiety, or neophobia. Our study focused on the short- and medium-term effects of Ole pretreatment on HI, with biochemical and histological analyses performed 24 h and 7 days post-HI, respectively. Moreover, motor tests performed at P14, a week after HI, showed no significant impact on gross motor abilities, aligning with previous findings.17,58,93,94 Future research should explore the longer-term effects of Ole pretreatment to determine if its neuroprotective actions translate to better neurological outcomes. There is some evidence that suggests Ole can ameliorate neurological deficits secondary to ischemic brain injury, as seen in middle cerebral artery occlusion models.35,36,40 Administration of Ole before 35 or after 36 injury (at 100 mg/kg), or as a dietary pretreatment of Ole-enriched olive leaf extract 40 improved sensorimotor and cognitive impairments. These studies also showed that Ole alleviated neuronal cell death and reduced apoptotic markers like Casp-3, Bax, or Bcl-2.35,36 Consistently, in the HI-ole100 group we observed a reduction in Casp-3 levels 24 h post-HI (Figure 7(a)) and a reduction in cell loss and neuronal damage, with more intact neurons and fewer pyknotic-like nuclei observed histologically at P14 (Figure 2(c) to (e)). These observations suggest Ole may reduce apoptosis, contributing to less brain damage and smaller infarct size. Altogether, the preservation of brain structures and white matter integrity in neocortical and subcortical brain areas (sensorimotor cortex, hippocampus, and striatum) suggests that Ole may protect the neuronal networks responsible for learning and memory, potentially improving HI-induced neurological deficits, though further research is needed.
Common pathological features of HI include neuroinflammation, oxidative stress, and increased blood-brain barrier (BBB) permeability.9,10 Our results suggest that Ole has anti-inflammatory properties as it reduced the cell density of microglia and astrocytes (Figures 4 and 5). Concomitantly, brain levels of proinflammatory cytokines IL-6 and TNF-α were lower in HI pups pretreated with Ole (Figure 7(b) and (c)). The effects of Ole on neuroinflammation in the context of ischemic brain injury remain poorly understood, but our results align with in vivo studies on myocardial ischemia/reperfusion 42 or post-traumatic stress disorder 95 where Ole suppressed TNF-α, IL-1β, and IL-6 upregulation. In vitro, Ole suppressed the LPS-induced increase in nitric oxide and proinflammatory cytokines in microglia cultures, likely by inhibiting ROS production and the ERK/p38/NF-κB pathway. 96 We showed that Ole seems to have a potent inhibitory effect on microglial activation, since a similar reduction microglia cell density was observed with both 20 and 100 mg/kg doses. However, Ole only partially reverted the astroglial reactive response at either concentration, suggesting different glial populations that contribute to neuroinflammation may respond differently to Ole. In response to cerebral ischemia, microglia generate ROS, initiate antigen presentation and phagocytosis, and secrete inflammatory mediators and metalloproteinases, contributing to BBB breakdown. 9 Although we did not examine BBB permeability in this study, previous research has shown that Ole preserves BBB integrity in animal models of stroke,40,69 which could be partially attributed to its inhibitory effect on microglia. Microglia are also important contributors to both demyelination (pro-inflammatory microglia) and remyelination (pro-regenerative microglia) following brain ischemia. 97 Although future studies are warranted to identify specific microglia subtypes affected by Ole, 98 the observed decrease in microglia cell density suggests reduced microgliosis, which may help preserve white matter by decreasing demyelination. 99
In addition to its anti-inflammatory activity, Ole has known antioxidant properties. It acts as a direct free radical scavenger due to its ortho-diphenolic group100,101 and has demostrated in vivo antioxidant activity in several models of cerebral, 37 cardiac,42,44 –46 and kidney 47 ischemia/reperfusion. Among its activities, Ole preserved the activity of antioxidant enzymes (including superoxide dismutase, glutathione peroxidase, catalase, and heme oxygenase-1), enhanced the level of non-enzymatic antioxidants like glutathione, and reduced the levels of thiobarbituric acid-reactive substances, lipid peroxidation, and protein carbonylation.37,42,44 –47 It has also been suggested that Ole can reduce oxidative stress and improve mitochondrial function by modulating the Nrf2 (nuclear factor erythroid 2-related factor 2) pathway,44,102,103 which controls the expression of many antioxidant response elements (AREs), responsible for cellular defense mechanisms against oxidative damage. 104 Furthermore, a relationship between Nrf2 and NF-κB, responsible for inflammatory response, has been indicated. 105 Ole increased Nrf2 and heme oxygenase-1 (HO-1) levels in angiotensin-induced senescent endothelial progenitor cells, involved in the neovascularization of ischemic tissue. 103 In another study, Ole increased antioxidant defenses in the paraventricular nucleus of spontaneously hypertensive rats by increasing Nrf2 and the Nrf2-mediated phase II enzymes HO-1 and NQO1 (NAD(P)H quinone oxidoreductase-1), leading to increased expression of the renin-angiotensin system components, higher mitochondrial biogenesis and more regulated mitochondrial dynamics. 102 Similar upregulation of Nrf2 and its targets (HO-1 and superoxide dismutase-2) by Ole was observed in cardiac ischemia/reperfusion injury. 44 Ole’s metabolite, hydroxytyrosol, was also shown to increase cellular antioxidant defense in neuronal systems via Nrf2 activation.106 –108
Despite Ole's known antioxidant properties, we did not observe significant changes in protein carbonyl or 8-OHdG levels among the studied groups (Table S6). This could be explained by the lack of sensitivity of the detection technique given the limited sample source. Working with neonatal mouse brain samples, we typically extracted less than 100 mg of ipsilateral brain tissue per mouse. Hence, most studies of neonatal HI are conducted in rats, in which the Rice-Vannucci method was initially described, 51 as their larger size may provide a better sample source for brain homogenates. Additionally, other studies have found that 24 hours after transient focal cerebral ischemia, changes in the oxidative stress markers 8-OHdG and 4-hydroxynonenal (4-HNE, a lipid peroxidant product) could be detected in plasma but not in brain homogenates. 109 Future studies on Ole's antioxidant properties against HI should consider these factors.
In addition to investigating Ole neuroprotective activities, we examined the distribution of Ole and some of its metabolites after IP administration. We administered the IP pretreatment 20 min prior to HI induction, consistent with similar studies.17,19,23,28,58 Ole concentration peaked in plasma 20 min after the IP, reaching mg/ml levels, while Htyr, a bioactive metabolite of Ole, peaked in the same timeframe in the ng/ml range (Fig. S2A-C). As expected, the IP injection resulted in a high bioavailability and a shorter time to peak plasma concentration compared to oral administration 110 . For example, in adult rats unmodified Ole peaked in plasma 2 hours after a single oral dose of 100 mg/kg, reaching 200 ng/ml, while no Htyr was detected. 111 We also detected small levels of Ole in the brain after IP administration (in the ng/g range). This aligns with previous reports of Ole accumulation in the brain (in the nanomolar range) after an acute oral dose of 3 g/kg of phenolic extract from olive cake 112 , altogether suggesting that Ole can cross the BBB.
Finally, we conducted a pilot study where Ole was orally administered to a pregnant mouse to explore maternal supplementation as a potential route for Ole administration. Our results showed that maternal supplementation with Ole during pregnancy and lactation (E12.5-P7) or lactation only (P0-P7) led to Htyr accumulation in pups’ brains (Fig. S2D). Htyr was not detected in brain samples after a single IP dose of Ole, suggesting that its presence may result from prolonged maternal exposure. Htyr is a dietary phenolic compound found in olive oil and red wine and can be produced from the de-glycosylation and de-esterification of Ole and as a byproduct of dopamine metabolism.113,114 Our findings, supported by other studies, suggest that Htyr can cross the BBB and accumulate in the brain.115,116 For instance, oral supplementation with Ole at 5 mg/kg/day for 21 days in rats led to the accumulation of Htyr and its phase II metabolites in the brain at low nanomolar concentrations. 115 Due to the exploratory nature of our study, we only quantified the unmodified forms of Ole and Htyr. However, Htyr undergoes extensive metabolism during and after absorption, resulting in significant conjugation, 113 whereas oleuropein aglycone (a derivate from Ole de-glycosylation) is thought to be more permeable than the glycated metabolites and be able to cross the BBB by passive diffusion.33,34 Thus, our measurements of the free forms of Htyr and Ole may be an underestimate of their intake to the neonatal brain after maternal supplementation. Maternal supplementation has been a successful strategy in reverting HI brain damage and associated neurological deficits for other nutraceuticals, including omega-3 fatty acids117 –120 and several phenolic compounds such as resveratrol, piceatannol, or pomegranate juice polyphenols.19,20,53,64
Here, we used a pretreatment strategy to model the clinical scenario in which a preventive treatment could be administered to fetuses at increased risk for adverse central nervous system outcomes due to intrauterine or postnatal HI, such as premature labor, preterm rupture of membranes, cord compression, or placental disruption.121,122 Although further research is needed to determine if maternal supplementation with Ole can reduce HI brain damage, our finding that maternal diet supplementation during pregnancy and lactation leads to Htyr accumulation in offspring brains is promising. This opens up the possibility of a nutritional and transgenerational approach for preventing HI in humans. Ole could be included in dietary supplements for pregnancies at higher risk for neonatal HI, such as those with intrauterine growth restriction or placenta previa.
In summary, we have shown that the olive leaf bioactive constituent Ole can reduce HI brain damage in vivo. Our results indicate that Ole can protect the brain at multiple levels: by protecting different cell populations (neurons, oligodendrocytes, microglia, and astrocytes) and by interfering with inflammation and apoptosis, two key mechanisms of the ischemic cascade. With the growing interest in nutraceuticals as preventive agents for neonatal HI, the functional properties of Ole described here support its potential as a prophylactic for HI.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241270237 - Supplemental material for Pretreatment with oleuropein protects the neonatal brain from hypoxia-ischemia by inhibiting apoptosis and neuroinflammation
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241270237 for Pretreatment with oleuropein protects the neonatal brain from hypoxia-ischemia by inhibiting apoptosis and neuroinflammation by Marta Reyes-Corral, Laura Gil-González, Ángela González-Díaz, Javier Tovar-Luzón, María Irene Ayuso, Miguel Lao-Pérez, Joan Montaner, Rocío de la Puerta, Rut Fernández-Torres and Patricia Ybot-González in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the following funding bodies: (1) Consejería de Transformación Económica, Industria, Conocimiento y Universidades; Junta de Andalucía [DOC_01701 to M.R.-C., P20_01267 to P.Y.-G.]. (2) Consejería de Salud y Bienestar Social; Junta de Andalucía [RH-0033-2020 to M.R.-C., C-0025-2018 to P.Y.-G.]. (3) Instituto de Salud Carlos III (ISCIII), co-funded by the European Union [PI20/00769 to P.Y.-G.]. (4) Fundación Alicia Koplowitz [“Ayudas a proyectos de investigación en neuropediatría 2018” to J.M.].
Acknowledgements
The authors warmly thank Eduardo Ponce-España, Carmen del Rıo-Mercado, Noelia Sola-Idıgora, and Rafael Gonzalo-Gobernado for technical advice, as well as the core facilities from the Institute of Biomedicine of Seville. We would also like to thank Eloisa Rubio Perez, Statistical Researcher at FISEVI, for her help with the statistical tests. We would also like to thank the CSIC for covering the costs associated with open access to this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
The authors confirm contribution to the paper as follows: M.R.-C., P.Y.-G., R.P. and R.F.-T. conceptualized and designed the study; M.R.-C., L.G.-G., R.F.-T., A.G.-D., J.T.-L. and M.L.-P. collected the data and interpreted the results; P.Y.-G., J.M. and M.R.-C. secured funding for the project with the collaboration of R.P. and M.I.A; finally, M.R.-C. prepared the manuscript with support of P.Y.-G. and M.I.A. All authors reviewed and approved the final version of the manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
