Abstract
We employed an in vitro hypoxia cell culture model system and gene transfer technology to examine the effect of the decorin gene on cell survival against oxygen and glucose deprivation (OGD). Ectopic expression of decorin in subventricular zone (SVZ) cells from adult male mouse brain and human glioblastoma U-87 cells kept the cells viable against 24 h of OGD. Fewer than 1% of decorin-synthesizing cells were apoptotic after 12 h of OGD. In contrast, 100% of the control cells were apoptotic even after 4h of OGD. De novo decorin synthesis in SVZ and U-87 cells induced expression of p21, p27 and Ras, AKT (acutely transforming retrovirus AKT8 in rodent T-cell lymphoma), and phosphorylated AKT. Blocking of phosphoinositide 3-kinase (PI-3K), Ras, and the epidermal growth factor receptor with specific inhibitors had no effect on induction of Ras, p21, and p27 at the messenger RNA level in decorin-synthesizing SVZ and U-87 cells. PI-3K inhibitors significantly increased apoptosis in decorin-expressing cells. Our data indicate that induction of p21, p27, Ras, AKT, and phosphorylated AKT by decorin inhibits apoptosis and protects U-87 and SVZ cells against OGD. Therefore, our data suggest that decorin is a potent trophic factor that protects neuronal progenitor cells and glioma cells from OGD.
Introduction
Decorin is a small leucine-rich proteoglycan (Iozzo, 1998) implicated in controlling matrix construction (Danielson et al, 1997), and cell differentiation and cell survival (Comalada et al, 2004) of macrophages. Decorin reduces proliferation in most transformed cells derived from primary malignant tumors (Santra et al, 1995, 1997, 2000, 2002). Decorin protects cells from programmed cell death through the PI-3K/AKT (acutely transforming retrovirus AKT8 in rodent T-cell lymphoma)/p21Waf1/p27kip1 pathway (Schonherr et al, 2001, 2005). Although decorin is expressed in the brain and the central nervous system (CNS) (Kallmann et al, 2002; Higaki et al, 2004), the function of this proteoglycan in the brain and the CNS is unknown. The ability of decorin to promote survival of decorin expressing glioma cells implanted in rat (Biglari et al, 2004), the association of decorin with the beta-amyloidal protein of Alzheimer's disease (Snow et al, 1995), and the induction of decorin expression in the CNS by injury (Russell et al, 2002) suggest neuroprotective and regenerative roles for decorin in the CNS.
We therefore investigated the effect of decorin on neuroprogenitor and stem cells derived from the subventicular zone (SVZ). These cells play an important role in the recovery from stroke and trauma (Zhang R et al, 2004a, b; Qu et al, 2005). They proliferate in the SVZ and migrate to the site of injury, where they enter a hostile environment of cell damage (Zhang R et al, 2001; Zhang J et al, 2004; Arvidsson et al, 2002). Enhancing the survival of these cells by transfection of decorin may benefit restoration of neurologic function. To test the cell specificity of decorin as a protective agent, we also transfected the U-87 glioma cells with decorin and measured survival under an adverse condition of oxygen and glucose deprivation (OGD). Our data showed that OGD-induced apoptosis in both SVZ cells and in human U-87 glioma was significantly inhibited by decorin synthesis. This protection was mediated by decorin via induction of cyclin kinase inhibitors, p21 and p27, G-protein, Ras and the cell survival signaling proteins, AKT, and phosphorylated AKT.
Materials and methods
All experimental protocols have been approved by the Henry Ford Hospital Institutional Animal Care and Use Committee.
Cell Cultures
Human glioblastoma U-87 cells and human 293T cells obtained from American Type Culture Collection (Rockville, MD, USA) were maintained in Dulbecco's modified Eagle's supplemented with 5% fetal bovine serum (FBS), 2 mmol/L glutamine, 100 U/mL penicillin, and 50 g/mL streptomycin. The SVZ of the adult male mouse brain was examined under a microscope (Olympus BX40; Olympus Optical, Tokyo, Japan) and was surgically resected according to the method described previously (Wang et al, 2004). Subventricular zone cells were dissociated in the medium containing 20 ng/mL of epidermal growth factor (EGF; R&D system, USA) and basic fibroblast growth factor. Passage 8 cells were used in this study.
Construction of Cytomegalovirus Expression Vector and Stable Transfection
Full-length decorin complementary DNA (cDNA) (Vetter et al, 1993) was subcloned into pcDNA3 (Invitrogen Corp., San Diego, CA, USA), as described previously (Santra et al, 1995). The orientation of the insert was verified by restriction endonuclease digestion and DNA sequencing from Applied Genomics Technology Center, Wayne State University, Detroit, MI, USA. The U-87 cells (106) were transfected with 10 mg of purified DNA by Lipofectamine™ 2000 Reagent (Invitrogen) according to the manufacturer's protocol, and incubated at 37°C in a humidified incubator for 12 to 16 h. Stably transfected cells were isolated and cultured routinely, as described previously (Santra et al, 1995). Five highly decorin-expressing U-87 clones were pooled on the basis of real-time polymerase chain reaction (PCR) data (data not shown). The pooled clones were used as decorin-synthesizing U-87 cells.
Cell Treatment and Induction of Hypoxia
The cells were rinsed twice with serum- and glucose-free media. The cells were incubated for different times with minimum volume of serum- and glucose-free medium in hypoxia without oxygen in an airtight Plexiglas-humidified chamber (Anaerobic Environment, Sheldon, USA), which was maintained at 37°C and continuously gassed with a mixture of 99% N2/1% CO2/0% O2. Cells were placed into this Sheldon hypoxia chamber on culture day 4 and remained there for 4 to 24 h.
Construction of Lentivirus Vector and Virus Infection
The lentivirus vector LV-TH (Wiznerowicz and Trono, 2003) containing green fluorescence protein (GFP) expression cassette was digested with BamH1 and EcoR1 and ligated with the insert containing decorin cDNA fused to the 3′ end of the human cytomegalovirus early gene promoter/enhancer from the above-mentioned pcDNA-3 and the decorin cDNA construct, digested with BglII and EcoRI. The decorin expression vector and LV-TH were digested with Sa/I and NdeI to delete the GFP cassette. Digested vectors were purified by 0.8% agarose gel electrophoresis, filled with Klenow enzyme to make blunt ends, and ligated to make decorin-expressing lentivirus and LV-TH without GFP. All recombinant lentiviruses were produced by transient transfection of 293T cells and infected in SVZ cells, according to the method of Wiznerowicz and Trono (2003).
Real-Time Semiquantitative Polymerase Chain Reaction
The cDNA was synthesized by using Superscript II RT from total RNA, according to the manufacturer's protocol (Invitrogen). All DNA primers used are shown in Table 1. Real-time PCR was performed in ABI Prism 7700 Sequence Detection System (Applied Biosystem, USA) by using SYBR Green PCR Master Mix (Applied Biosystem) for 2 mins at 93°C, followed by 28 to 36 cycles of 7 secs at 93°C, 30 secs at 65°C, and 30 secs at 70°C, as described previously (Zhang J et ai, 2004). Dissociation curves and agarose gel electrophoresis were used to verify the quality of the PCR products. Each sample was tested in triplicate using quantitative real-time PCR. All values were normalized to β-actin. Samples obtained from five independent experiments were used for analysis of relative gene expression data using the 2−ΔΔT method (Livak and Schmittgen, 2001).
Sequence of DNA primers used in a real-time semiquantitative PCR

PCR=polymerase chain reaction.
TUNEL Assay, Western Blot Analysis, and Cell Proliferation Assays
Terminal deoxynucleotide transferase dUTP nick-end labeling (TUNEL) stains (Chen et ai, 2003) were performed using the ApopTag Fluorescein Kits (Intergen, USA), according to the manufacturer's protocol. For immunoblotting of decorin, media conditioned by the various cells for 24 h in 0.1% serum were separated in 10% sodium dodecylsulfate-polyacrylamide gel electrophoresis (SDS-PAGE), Immunodetection was performed by using rabbit polyclonal antibodies to decorin (Vetter et ai, 1993). For Western blot analysis, cells were incubated in serum-free medium for 1.5 h, rinsed twice with phosphate-buffered saline and harvested in ice-cold lysis buffer containing 50 mmol/L 4-(2-hydroxyethyl)piperazine-1-ethanesulfonic acid, pH 7.4, 1% Triton X-100, 10% glycerol, 150 mmol/L NaCl, 2 mmol/L ethylenediaminetetraacetic acid, 10 mmol/L NaF, 1 mmol/L Na3VO4, 10 mg/mL leupeptin, 10 mg/mL aprotinin, and 1 mmol/L phenylmethylsulfonyl fluoride. Proteins (typically 40 μg) were directly subjected to SDS-PAGE, separated by SDS-10% PAGE, transferred onto nitrocellulose membranes, blocked with 5% bovine serum albumin for 18 h, and analyzed by immunoblotting with the indicated specific antibodies using the ECL detection system (Santra et al, 1995). Proteins were transferred onto a nitrocellulose membrane and incubated with anti-rabbit AKT antibodies (1:1000; recognizing total human, rat and mouse AKT, and anti-rabbit phospho-specific Ser473, New England BioLabs, Beverly, MA, USA), anti-rabbit β-actin antibody (1:1,000; Santa Cruz Biotechnology, Santa Cruz, CA, USA) as primary antibody, and anti-rabbit horseradish peroxidase antibody (1:1,000; Santa Cruz Biotechnology, Santa Cruz, CA, USA) as secondary antibody.
The CellTiter 96 Aqueous Non-Radioactive Cell Proliferation Assay (Promega Corp., Madison, WI, USA) was used to determine the number of viable cells in the proliferative phase, according to the manufacturer's protocol. This nonradioactive cell proliferation assay is a colorimetric method based on the use of a modified tetrazolium/formazan (MTS/PMS) assay (Santra et al, 1995). In this method, the amount of formazan product is time dependent and proportional to the number of viable cells.
Statistical Analysis
One-way analysis of variance followed by Student–Newman–Keuls test was used. The values were the mean of 5 to 10 independent experiments for real-time PCR data and three independent experiments in the case of Western blot analysis. The data are presented as mean±s.d. The P-values of less than 0.05 are considered as significant.
Results
Decorin Gene Transfer Promotes Cell Survival after Serum, Oxygen, and Glucose Deprivation
Subventricular zone cells, infected with control lentivirus and decorin gene-containing lentivirus (+/− GFP) as well as transfected vector control and decorin human glioblastoma U-87 cells were subjected to OGD. Both transfected U-87 cells and infected SVZ cells overexpressed decorin at the protein level (Figure 1). Infection was monitored by GFP expression in SVZ cells (Figure 1C). The decorin-expressing lentiviruses without GFP were used in all experiments to infect SVZ cells. The decorin-infected SVZ cells and decorin-transfected U-87 cells remained viable against OGD up to 24 h, based on MTS/PMS assays from five individual experiments (Figure 2). During OGD, the decorin-synthesizing U-87 cells remained attached to the plate for 24 h and changed their morphology to spindle structure (Figure 3). In contrast, the control U-87 cells detached after 8 h of OGD without altering their morphology (Figure 3). Control SVZ cells were diffusely scattered in the plate after OGD. However, the decorin synthesizing SVZ cells turned into spheres after 4 h of OGD (Figure 3). Our data show that decorin-synthesizing SVZ and U-87 cells were remarkably viable after 24 h of OGD with alteration of their morphology. To test whether decorin synthesis reduces apoptosis after OGD, TUNEL assay was performed.
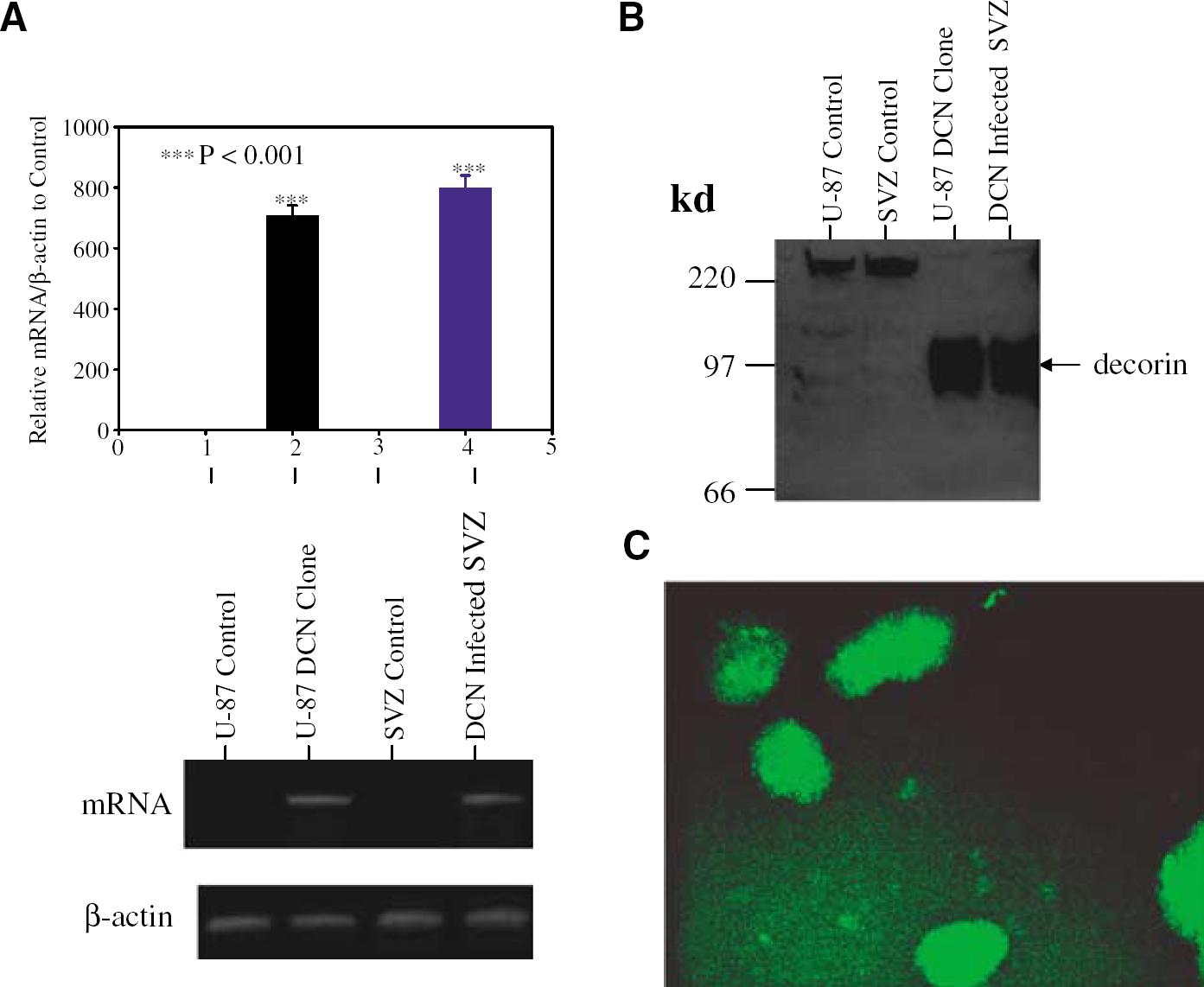
Ectopic expression of decorin (DCN) in U-87 cells, decorin-transfected U-87 (U-87 clone), mouse subventricular zone (SVZ) cells, and decorin containing lentivirus-infected SVZ cells (SVZ infection). The expression of DCN was analyzed by semiquantitative real-time polymerase chain reaction (PCR) shown in top panel of (
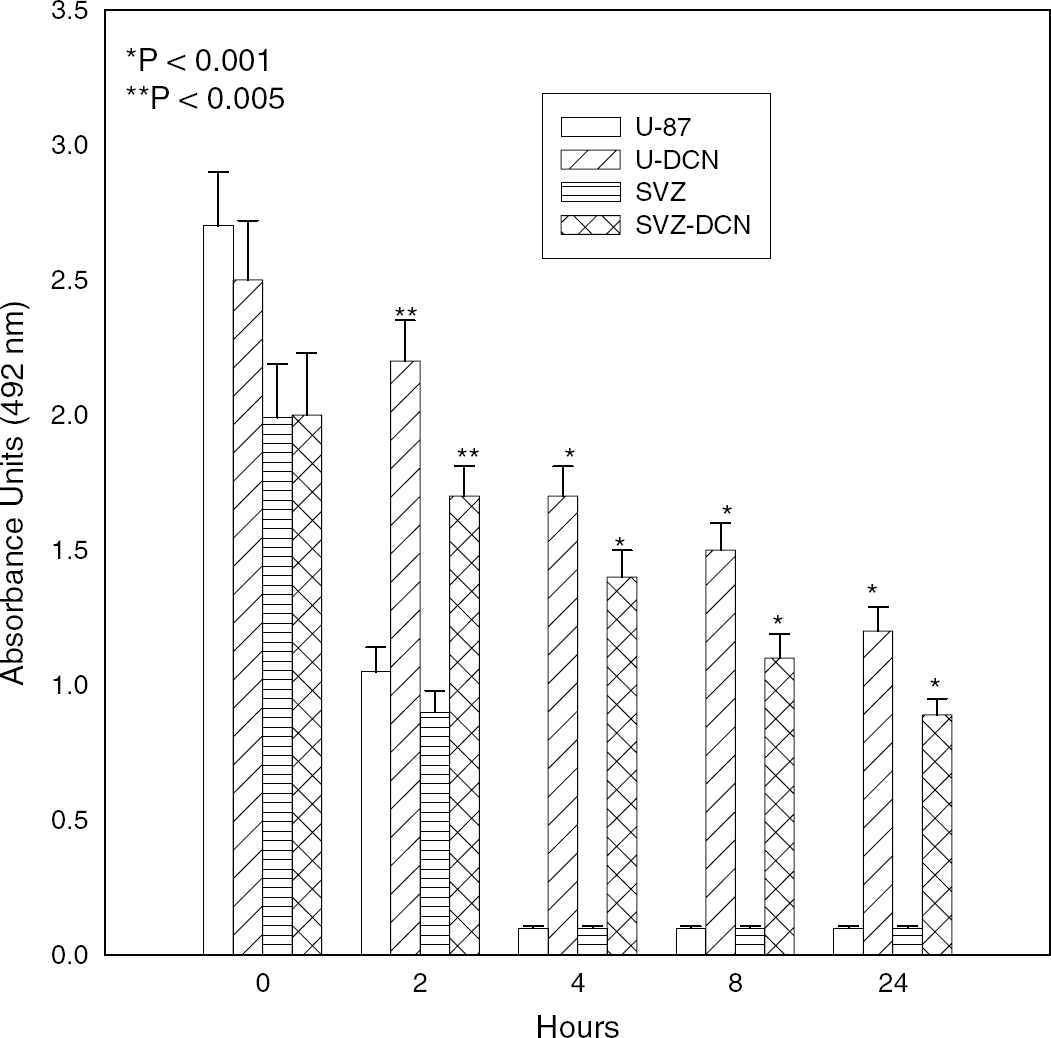
De novo decorin expression induces cell survival against hypoxia in U-87 and subventricular zone (SVZ) cells based on a nonradioactive tetrazolium/formazan assay. The U-87 cells transfected with empty pcDNA-3 vector as U-87 control, decorin expression vector as U-87-DCN, SVZ cells infected with empty lentivirus as SVZ Control, and SVZ cells infected with decorin-expressing lentivirus as SVZ-DCN are subjected to oxygen and glucose deprivation (OGD) for 2 to 24 h. Asterisks indicate significant different between decorin-expressing samples and vector control.
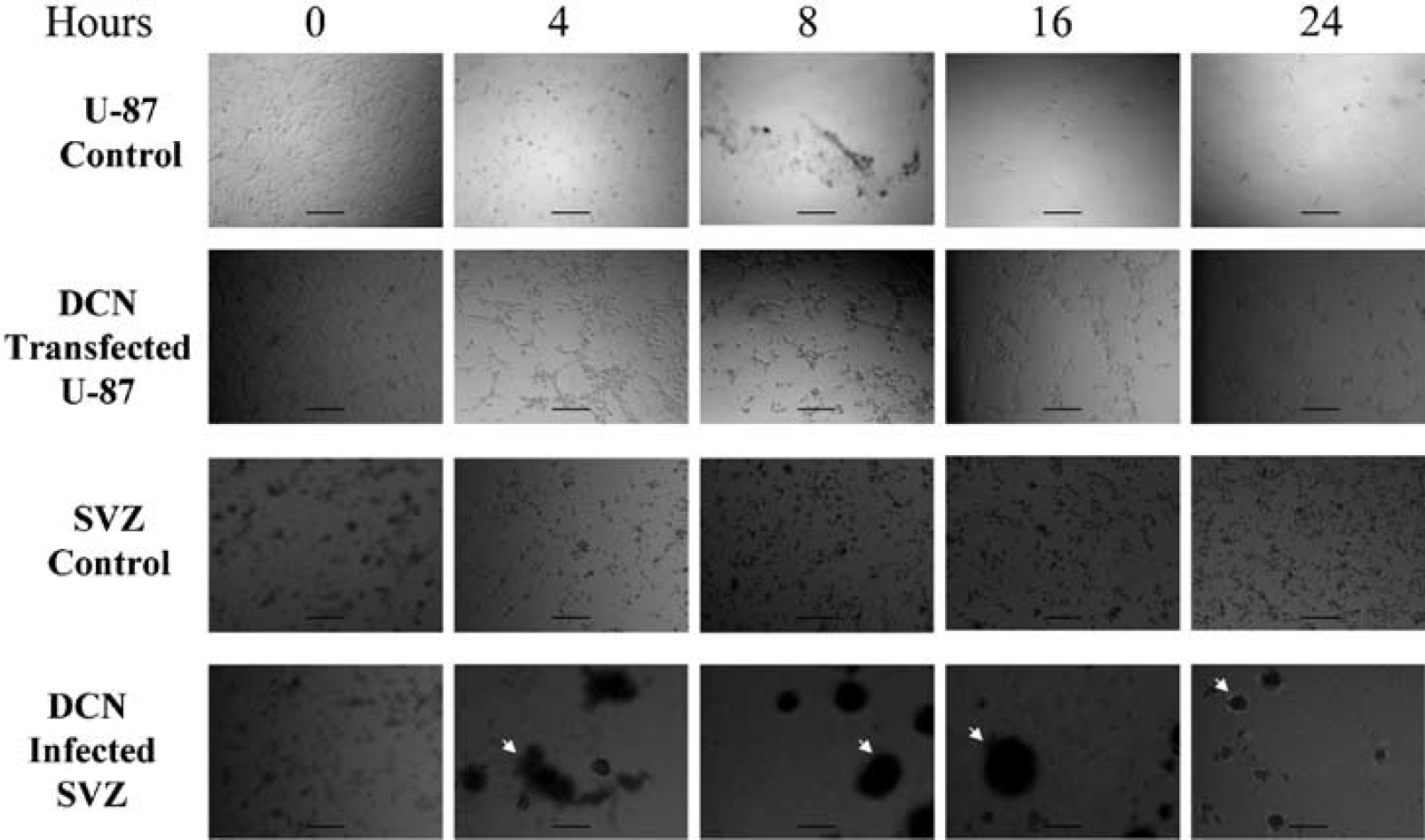
Ectopic decorin (DCN) expression reveals morphologic diversity of the cells during oxygen and glucose deprivation (OGD). The U-87 cells transfected with control vector as U-87 control, decorin expression vector-transfected U-87 cells as DCN-transfected U-87, subventricular zone (SVZ) cells infected with control lentivirus as SVZ Control, and SVZ cells infected with decorin-expressing lentivirus as DCN-infected SVZ cells were subjected to OGD from 0 to 24 h. Pictures were taken using a phase-contrast microscope. Arrows indicate aggregation of DCN-infected SVZ cells in panels 4, 8, 16, and 24h. Calibration bar = 200 μm. Note that DCN-transfected U-87 cells turn into endothelial-like during OGD. DCN-infected SVZ cells turn into spherical shape after 4 h of OGD.
Antiapoptotic Effect of Decorin
Nearly 100% of control U-87 cells and SVZ cells underwent apoptosis after OGD. Small DNA fragments diffused from the nucleus of both cells. TUNEL staining was confined to the nucleus or what appeared to be diffuse nuclear debris. In contrast, decorin-expressing cells had very faint fluorescent intensity, indicating less apoptosis (∼ 1%) based on cell count under Fluorescent Illumination Microscope (Olympus IX71/IX51, Japan) in five independent experiments, and large DNA fragments remained in the nucleus, as shown in Figures 4 and 5. These data indicate that decorin-synthesizing cells were resistant to apoptosis after 12 h of OGD. To investigate the mechanisms underlying the antiapoptotic effect of decorin, we utilized specific pharmacological inhibitors for signaling molecules. Synthetic wortmannin and LY294002 interact directly with the ATP binding pocket of phosphoinositide 3-kinase (PI-3K) and induce conformational change in their catalytic domains and specifically inhibit PI-3K (Walker et al, 2000). We utilized these inhibitors to elucidate the role of PI-3K/AKT in signal transduction pathways that may be involved in cell survival and reduction of apoptosis. Cell counts under the Fluorescent Illumination Microscope in five independent experiments showed that the PI-3K inhibitors, LY29402 and wortmannin, significantly enhanced the number of fluorescent cells in decorin-synthesizing cells depending on the time of incubation under OGD (Figures 4 and 5). Therefore, induction of apoptosis by PI-3K inhibitors in decorin-synthesizing cells (40% to 60%) indicates that decorin-mediated cell survival was dependent on the PI-3K/AKT pathways after 12 h of OGD. In contrast, nearly 100% of control cells (decorin-deficient cells) underwent apoptosis without PI-3K inhibitors after only 4 h of OGD. These data show that decorin-induced cell survival is regulated not only by PI-3K/AKT pathways but also by other factors. To identify the other pathways, we studied the effect of decorin on downstream and upstream signaling pathways. Based on previous reports of decorin-induced upregulation of the cyclin kinase inhibitor, p21 (De Luca et al, 1996), we investigated the effect of the cyclin kinase inhibitors, p21 and p27, on decorin-mediated brain cell protection against OGD. In addition, using cDNA library (made from decorin ± A-431 cells) screening by subtraction hybridization (data not shown), we investigated the Ras gene as an upstream signal of decorin-mediated brain cell protection against OGD.
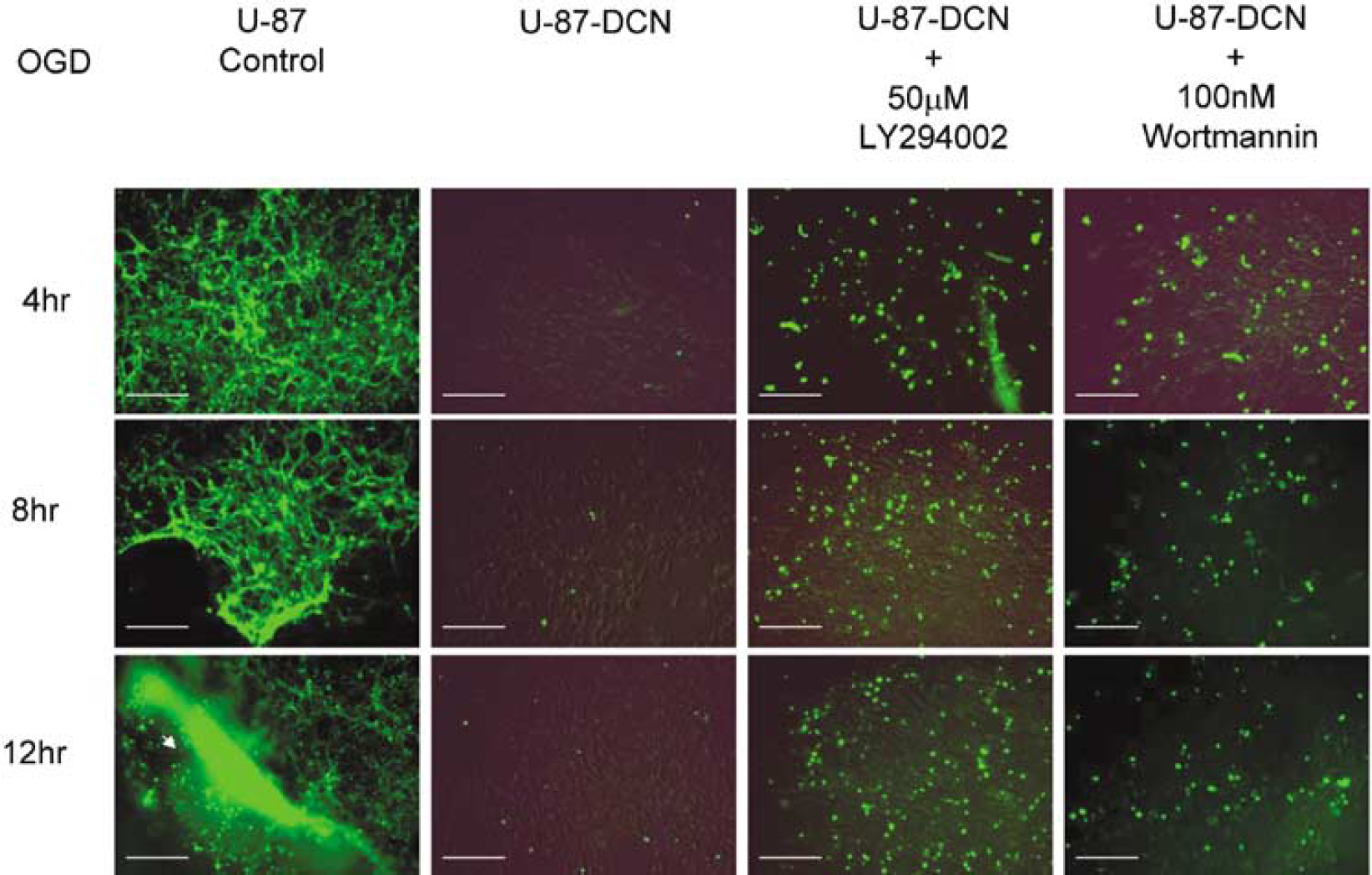
De novo decorin (DCN) expression protects U-87 cells from apoptosis. Cell death was detected by TUNEL staining. The U-87 cells transfected with control vector as U-87 control, and decorin expression vector-transfected U-87 cells as DCN-transfected U-87 were subjected to oxygen and glucose deprivation (OGD) from 4 to 12 h with and without phosphoinositide 3-kinase (PI-3K) inhibitors, LY294002, and wortmannin. Calibration bar = 400 μm. Note that 100% of control cells are apoptotic at the 4 h OGD incubation point; in contrast, less than 1% of DCN-expressing cells are apoptotic after 24 h of OGD. Pretreatment with inhibitors, LY294002 and wortmanin, increases apoptosis with time of incubation under OGD condition.
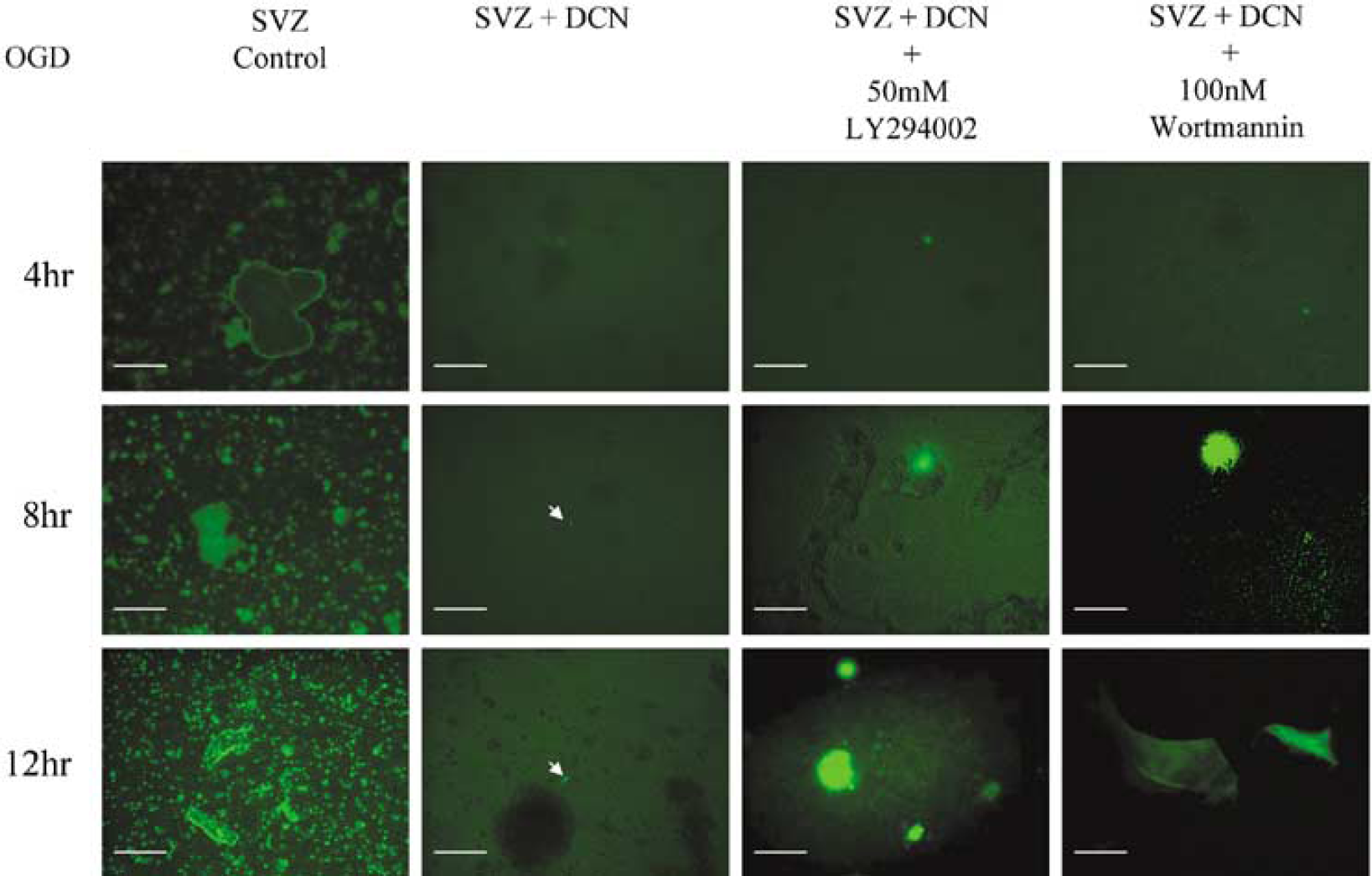
Mouse subventricular zone (SVZ) cells are protected from apoptosis by ectopic decorin expression. Detection of cell death was performed by TUNEL staining. Mouse SVZ cells infected with control lentivirus without green fluorescence protein (GFP) as SVZ Control and SVZ cells infected with decorin-expressing lentivirus without GFP as DCN-infected SVZ were subjected to oxygen and glucose deprivation (OGD) for 4, 8, and 12 h with and without phosphoinositide 3-kinase (PI-3K) inhibitors, LY294002 and wortmannin. Arrows indicate very faint bright spots as apoptotic nuclei at 8 and 12 h in DCN-treated SVZ cells. Calibration bar = 375 μm. Note that 100% of control cells are apoptotic at 4 h of OGD; in contrast, less than 1% of DCN-expressing cells are apoptotic after 24 h of OGD. Pretreatment with inhibitors, LY294002 and wortmanin, increases apoptosis with time of incubation under OGD condition.
De Novo Expression of Decorin is Associated with Marked Induction of p21 and p27 as Cyclin Kinase Inhibitors and Ras as G-Protein
As a control before OGD, the messenger RNA (mRNA) levels of p21, p27, and Ras in decorin-synthesizing cells (SVZ and U-87) and decorin-nonsynthesizing cells (SVZ and U-87) were measured by semiquantitative real-time PCR in 10 independent experiments when the cells were growing in the exponential stage. DNA bands of PCR products separated by 1.5% agarose gel electrophoresis, normalized with β-actin, showed a marked upregulation of p21, p27, and Ras genes in both U-87 and SVZ decorin-synthesizing cells (Figure 6). Normalization on β-actin gave similar results with a 150- to 215-fold increase in p21, p27, and Ras mRNA in decorin-synthesizing U-87 and SVZ cells. Decorin-induced upregulation of p21, p27, and Ras correlated with decorin-mediated cell survival and the antiapoptotic properties of decorin against OGD. These data indicate that p21, p27, and Ras are involved in cell survival via an antiapoptotic pathway in decorin-synthesizing cells. In contrast, ectopic expression of decorin had no significant effect on BCL-2 gene expression at the mRNA level in both SVZ and U-87 cells based on semiquantitative real-time PCR analysis in five independent experiments, normalized with β-actin, although there was a trend towards an increase in the DCN-induced cells (Figure 6). These data indicate that the involvement of BCL-2 in decorin-mediated antiapoptotic pathway was marginal in decorin-synthesizing cells. However, we are aware that BCL-2 mRNA regulation may not reflect BCL-2 function and further experiments are warranted. To identify the link between downstream regulation of the cyclin kinase inhibitors, p21 and p27, and upstream signaling pathways via Ras, PI-3K/AKT, and EGF receptors (EGF-R), reported as cell survival factors (Bock-Marquette et al, 2004, Xue et al, 2000; Koumenis et al, 2001), we employed specific inhibitors to EGF-R, PI-3K, and Ras.
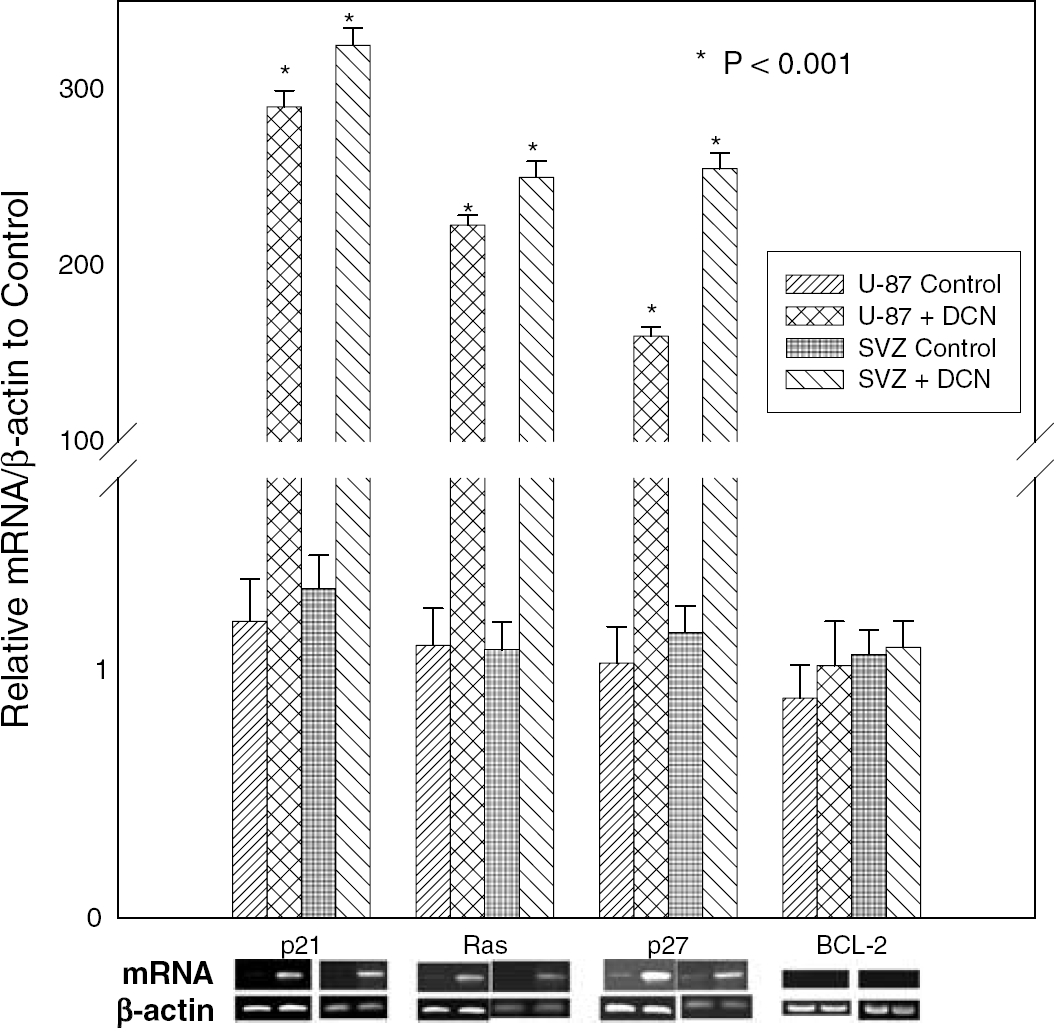
Effect of decorin on expression of p27kip1, p21cip1, C-H-Ras, and BCL-2 based on analysis by semiquantitative real-time polymerase chain reaction (PCR) in exponential stage of the cells before OGD as control. The U-87 cells transfected with control vector as U-87control, decorin expression vector-transfected U-87 cells as U-87 + DCN, SVZ cells infected with control lentivirus as SVZ Control, and SVZ cells infected with decorin-expressing lentivirus as SVZ+ DCN. In the bottom panel, the corresponding PCR products are analyzed by 1.5% agarose gel.
Effect of Inhibitors: EGF-R-Specific Inhibitor Tyrphostin AG-1478, PI-3K-Specific Inhibitor LY294002, and Ras-Specific Farnesyltransferase Inhibitor FTI-276 on Decorin-Induced Upregulation of p21, p27, and Ras
The decorin-synthesizing SVZ cells and U-87 were subjected to OGD for 12 h with and without the EGF-R-specific inhibitor tyrphostin AG-1478 (Moscatello et al, 1998), the PI-3K-specific inhibitor LY294002 (Walker et al, 2000), and the Ras-specific farnesyltransferase (FTase) inhibitor FTI-276 (Eberlein et al, 2001). The level of mRNA of decorin-induced p21 and p27 were analyzed by semiquantitative realtime PCR. One of the tyrphostin analogs, AG-1478, specifically blocks autophosphorylation of EGF-R, as reported previously (Moscatello et al, 1998), but not platelet-derived growth factor receptor (Rice et al, 1999). Therefore, this tyrphostin analog, AG-1478, was used to block the decorin-induced mitogen-activated protein kinase kinase pathway via EGF-R. The PI-3K-specific inhibitor LY294002 was employed to block the PI-3K/AKT pathway, and the FTase inhibitor FTI-276 to block the Rasmediated signaling pathway. FTI-276, a peptide mimetic of the COOH-terminal Cys-Val-Ile-Met of K-Ras 4B potently inhibits FTase in vitro (IC50 = 500 pmol/L) and is highly selective for FTase over geranylgeranyltransferase I (Livak and Schmittgen, 2001; Eberlein et al, 2001; Zhang and Casey, 1996). Our data from semiquantitative real-time PCR analysis of five independent experiments show that the cyclin kinase inhibitors, p21 and p27, remained unaffected at the mRNA level even with 50 μmol/L of EGF-R-specific inhibitor AG-1478, 50 μmol/L of PI-3K inhibitor LY294002, and 50 μmol/L of Ras inhibitor FTI-276 for 12 h against OGD in both SVZ cells and U-87 cells (Figure 7A). Decorin-nonexpressing control U-87 and SVZ cells did not exhibit altered mRNA expression ofp21, p27, and Ras when exposed to 50 μmol/L LY294002, AG-1478, and FTI-276 (data not shown). These data suggest that induction of p21, p27, and Ras by decorin is not mediated by the EGF-R/MAPK pathway, not by the PI-3K/AKT pathway, and not by the Ras-mediated signal pathway. These data also indicate that decorin may provide a unique pathway that regulates p21, p27, and Ras expression in decorin-synthesizing cells. AKT and phosphorylated AKT are major cell survival factors (Bock-Marquette et al, 2004). Therefore, we investigated the effect of decorin on AKT and activation of AKT.
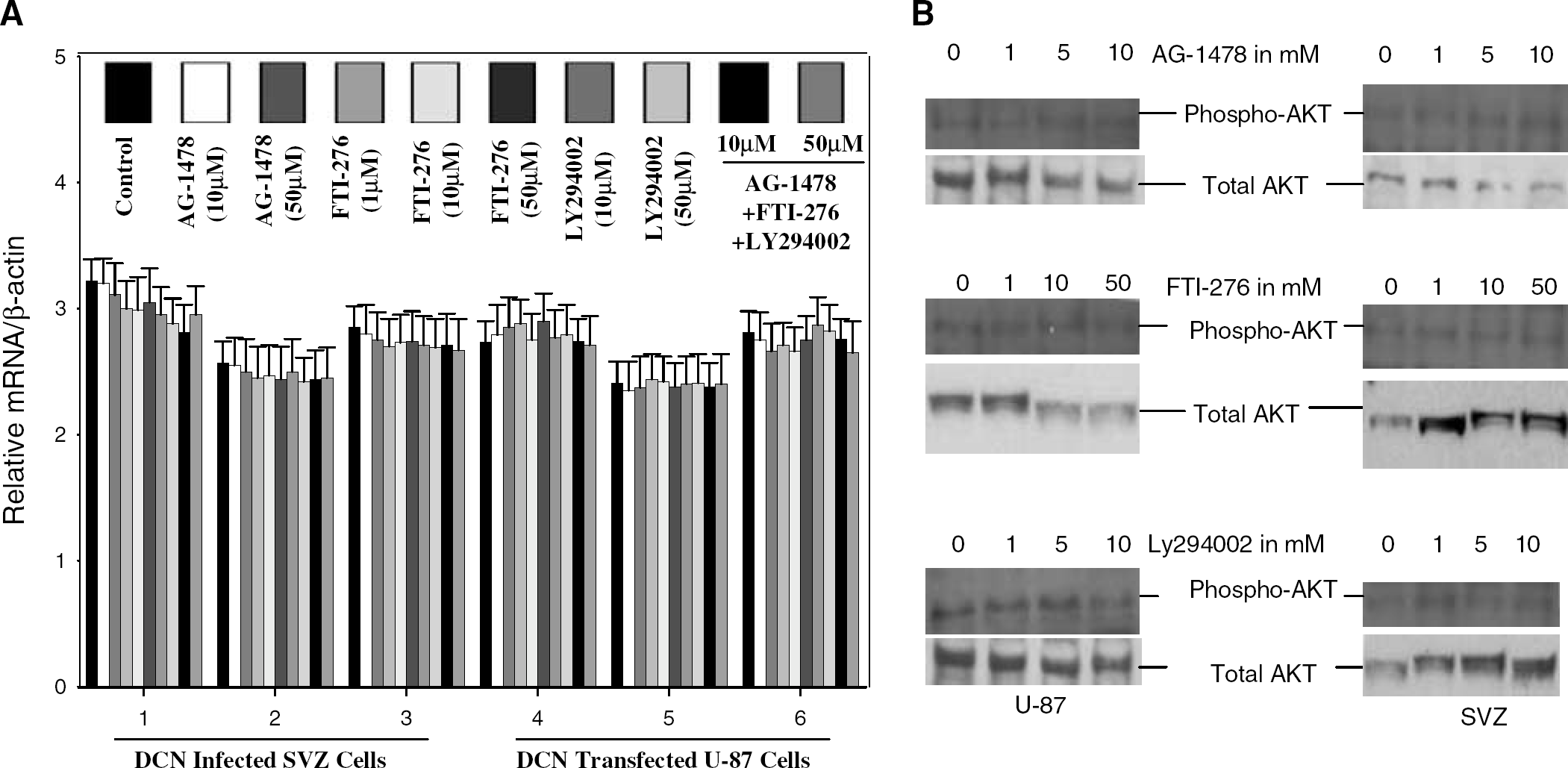
Effect of decorin on messenger RNA (mRNA) expression (left panel,
Upregulation of AKT and Phosphorylation of AKT by Decorin
Quantitative histogram analysis of protein expression of AKT and phospho-AKT protein bands on Western blots was performed in three independent experiments. These data were normalized to β-actin. For quantitative analysis of phosphorylation of AKT, histogram data were normalized to total AKT. Phosphorylation of AKT remained unaffected in decorin-deficient control U-87 and SVZ cells when these cells were treated with different concentrations of LY294002, AG-1478, and FTI-276 (Figure 7B). AKT expression at the protein level was significantly induced by decorin in SVZ and U-87 cells (Figure 8A). Phosphorylation of AKT was robust in the decorin-synthesizing SVZ and U-87 cells (Figure 8B). A low dose, for example, 1 μmol/L of the PI-3K inhibitor LY294002, EGF-R-specific inhibitor AG-1478, and Ras inhibitor FTI-276 caused a down-regulation of AKT phosphorylation (Figure 8B). This effect was enhanced when AG-1478 and FTI-276 were combined (Figure 9). Inhibition of AKT phosphorylation by the Ras inhibitor FTI-276 occurred in a dose-dependent manner (Figure 9), indicating specific inhibition of Ras activation. The dose-dependent inhibition of AKT phosphorylation by both PI-3K inhibitors, wortmannin (data not shown) and LY294002 (Figure 9), showed that decorin-induced AKT phosphorylation was mediated specifically through PI-3K (Figure 9). Our data show that decorin induced substantial upregulation of Ras (Figure 6) and both total and phosphorylated AKT in SVZ cells and U-87 cells (Figures 8A and 8B). The Ras inhibitor FTI-276 suppressed AKT phosphorylation even at a low dose of 1 μmol/L and also in a dose-dependent manner with higher doses in both SVZ and U-87 cells (Figures 8B and 9). These data suggest that Ras is directly involved in AKT phosphorylation. As a result of upregulation of both Ras and AKT, activation of AKT was strong. FTI-276 and AG-1478 both inhibited AKT phosphorylation in mouse SVZ cells and human U-87 cells treated with 10 μmol/L concentrations of each. When FTI-276 and AG-1478 both were combined at the same concentration of 10 μmol/L, AKT phosphorylation was strongly reduced with no visible band (Figure 9). Our data indicate that decorin-induced AKT activation was mediated via two independent pathways, Ras and EGF-R. As the activation of EGF-R, Ras, and AKT were reported to be involved in cell survival (Koumenis et al, 2001), we next investigated the involvement of activation of EGF-R, Ras, and AKT, in decorin-induced cell survival against OGD by using their respective specific inhibitors.
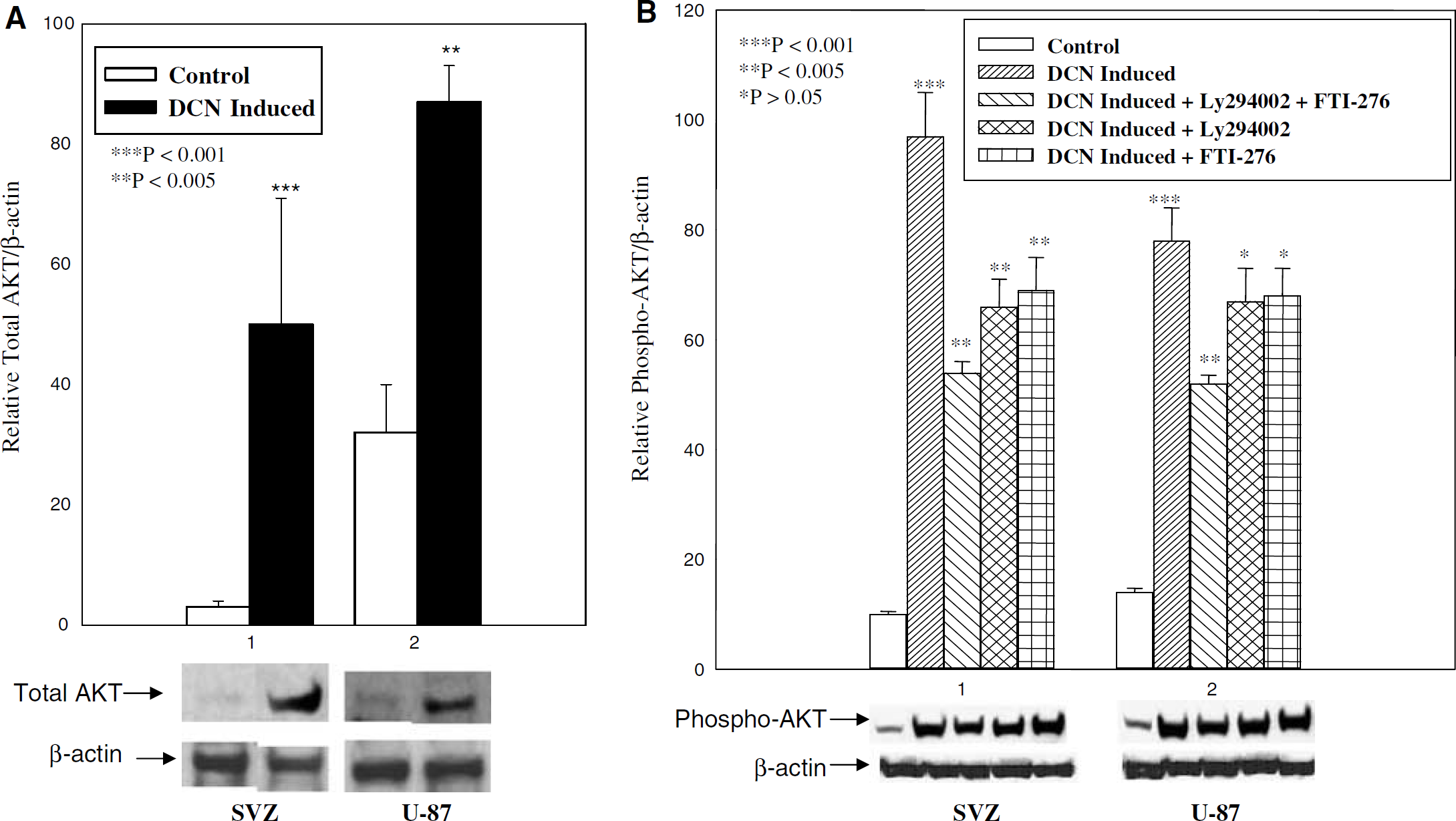
Induction of total AKT and phospho-AKT in decorin-synthesizing mouse SVZ and U-87 cells were analyzed by Western blot analysis. Total AKT, phospho-AKT, and β-actin are marked on the left in the bottom panels of (
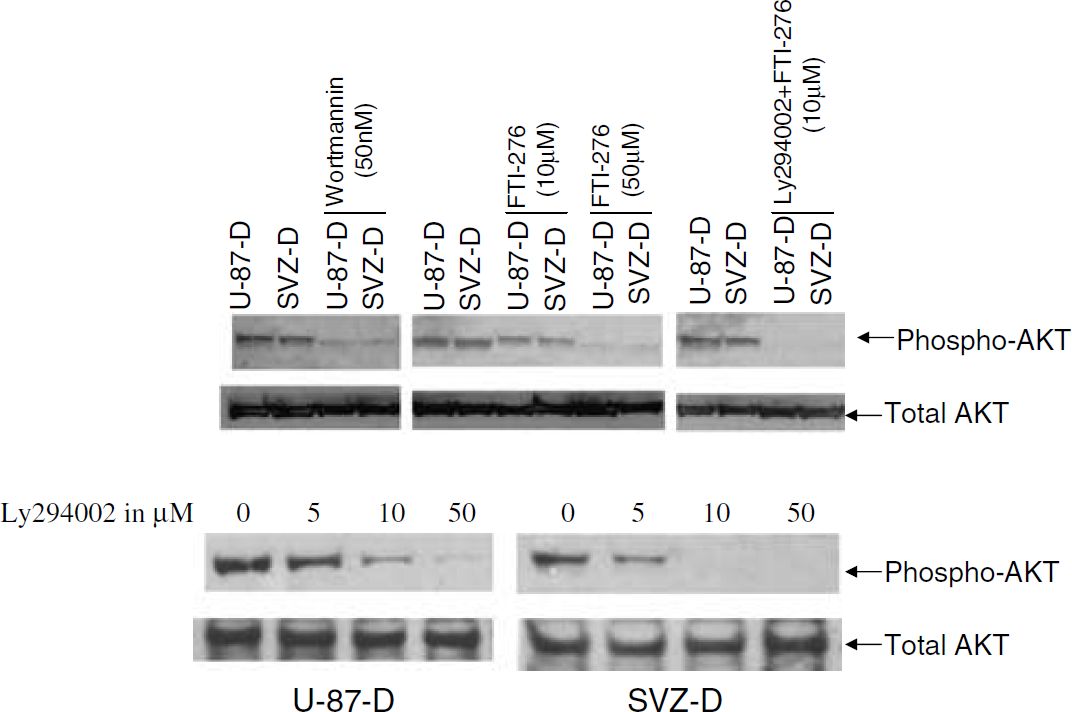
Pretreatment with wortmannin, LY24002, and FTI-276 inhibits decorin-induced AKT phosphorylation in different concentrations. Decorin-expressing cells subventricular zone (SVZ) (SVZ-D) and U-87 (U-87-D) were pretreated with different inhibitors at different concentrations for 1.5 h without serum. Phospho-AKT and total AKT are marked on the right. Bottom panel indicates inhibition of phospho-AKT in a dose-dependent manner by LY294002.
Effect of AG-1478, LY294002, and FTI-276 Inhibitors on Decorin-Induced Cell Survival
Decorin-synthesizing SVZ and U-87 cells were subjected to OGD and treated with different concentrations of AG-1478, FTI-276, LY294002, and also a mixture of all three inhibitors for 8 h. Cell viability was measured by the MTS/PMS assay (Santra et al, 1995). Epidermal growth factor receptor inhibitor AG-1478, PI-3K inhibitor LY294002, and Ras inhibitor FTI-276 inhibited growth of decorin-expressing SVZ and U-87 cells in a dose-dependent manner under OGD for 8 h (Figure 10). These data suggest that EGF-R, Ras and AKT are involved in decorin-induced cell survival. When these decorin-synthesizing SVZ and U-87cells were treated with a mixture of all three inhibitors AG-1478, FTI-276 and LY294002, growth of cells was significantly suppressed (P < 0.05) in a dose-dependent way, but growth was not completely reduced (Figure 10). The decorin-expressing SVZ and U-87 cells survived after 8 h of OGD with high dose (50 μmol/L) of all three inhibitors (Figure 10). In contrast, after 8 h of OGD, the control decorin-deficient SVZ and U-87 cells showed significantly reduced values in the MTS/PMS assay (Figure 2). These data show that other factors are involved in decorin-induced cell survival.
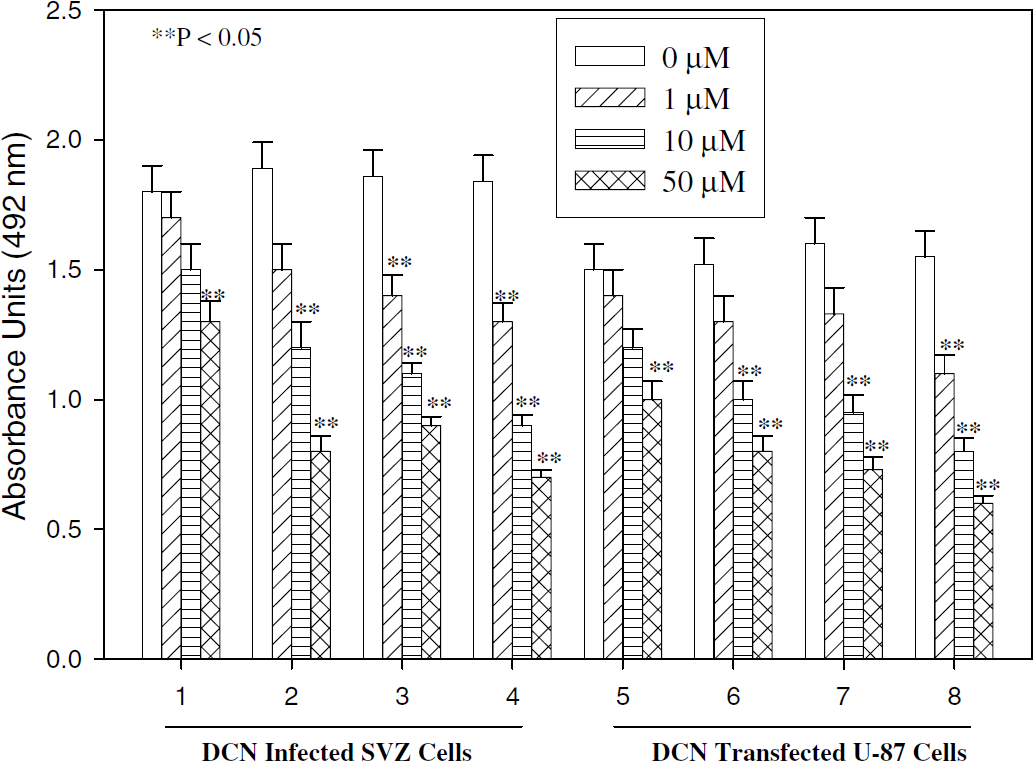
Treatment with inhibitors, AG-1478, LY294002, and FTI-276, inhibits cell survival induced by decorin against 8 h OGD. Decorin-synthesizing subventricular zone (SVZ) cells (1 to 4) and U-87 cells (5 to 8) were subjected to OGD for 8 h with and without 1 to 50 μmol/L AG-1478 (1, 5); FTI-276 (2, 6); AG-1478 + LY294002 + FTI-276 (3, 7), and LY294002 (4, 8). The number of viable cells is established by using a nonradioactive tetrazolium/formazan assay.
Discussion
Brain cells in culture exhibit low survival after 6 h of exposure to combined hypoxia and hypoglycemia in the presence of glucose substitute, 2-deoxyglucose (Lyons and Kettenmann, 1998). We showed here that the decorin-synthesizing SVZ and U-87 cells remained viable after 24 h of OGD. In contrast, control SVZ and U-87 cells survived less than 4 h of OGD (Figures 2–4). Decorin is a secretory protein and may act as a trophic factor to protect both neuroprogenitor and tumor cells against ischemia. The decorin-synthesizing SVZ and U-87 cells upregulated p21, p27, and Ras at the mRNA level and AKT and phosphorylated AKT at the protein level. The two major factors that protected the decorin-synthesizing SVZ and U-87 cells against severe OGD deprivation were the antiapoptotic properties of decorin and activation of AKT (Bock-Marquette et al, 2004). Ectopic decorin expression in both SVZ and U-87 cells also induced Ras, AKT, and phosphorylated AKT. Ras interacts directly with PI-3K and activates AKT, which are involved in cell survival (Pacold et al, 2000). Decorin-induced Ras is antiapoptotic and activates AKT and promotes neuroprotection against severe OGD in decorin-synthesizing cells. AKT, a major cell survival factor (Bock-Marquette et al, 2004), is not only upregulated by decorin but is also activated. This activated form of AKT leads to neuroprotection in decorin-synthesizing SVZ and U-87 cells.
Expression of decorin in mouse SVZ cells and human U-87 cells is concomitant with induction of p21cip1, p27kip1, and c-Ha-ras, suggesting the involvement of these genes in the protection of SVZ and U-87 cells against OGD. Mesangial cell survival depends on p21 upregulation by insulin via the PI-3K/AKT pathway (Hiromura et al, 2002). Our data showed that decorin-induced upregulation of p21 and p27 is not affected by the PI-3K inhibitor, LY294002. Therefore, decorin-induced upregulation of p21 and p27 was not regulated by the PI-3K/AKT pathway. Mouse oligodendrocyte progenitor cells from p21- and p27-deficient mice fail to grow into mature cells and fail to differentiate with wild-type kinetics. Therefore, both p21 and p27 play a major role in the maturation and differentiation of mouse oligodendrocyte progenitor cells (Zezula et al, 2001). Decorin may function as a survival factor by inducing the two cyclin kinase inhibitors, p21 and p27. Morphology of decorin-synthesizing U-87 cells changed from oval to spindly shape during OGD. This may be because of overexpression of Ras and AKT, which promote morphologic changes of Madin—Darby canine kidney (MDCK) cells to dispersed spindly from wild-type MDCK cells, which are a typical epithelioid morphology, growing in compact islands (Khwaja et al, 1997). Morphology of decorin-synthesizing mouse SVZ cells also changed, possibly, for the same reason because of a high level of activated Ras and AKT during OGD.
Decorin promotes robust survival of SVZ and U-87 cells against OGD by activating the PI-3K/AKT pathway. Phosphoinositide 3-kinase is strongly and directly activated by H-Ras G12V in vivo or by GTPgS-loaded H-Ras in vitro (Pacold et al, 2000). From our data, decorin induced upregulation of Ras, total AKT, and phosphorylated AKT. Inhibition of AKT phosphorylation by the Ras inhibitor, FTI-276, in a dose-dependent manner confirmed the involvement of Ras in PI-3K activation and cell survival. Our present data show that decorin induced Ras, AKT, and activation of AKT by two independent mechanisms via the Ras/PI-3K/AKT and EGF-R/Ras/AKT pathways. We, however, do not exclude the possibility that other pathways may contribute to the cell survival, and other pathways downstream of AKT, for example, bad and bax should also be examined. The power of decorin as a protective signal generator in decorin-synthesizing SVZ and U-87 cells resides in its ability to upregulate cyclin kinase inhibitors p21 and p27, and to overexpress Ras, AKT, and phospho-AKT. By blocking the signal pathways mediated by them with their specific inhibitors, FTI-276, AG-1478, and LY294002, respectively, induction of p21, p27, and Ras by decorin remained unaffected. The mechanisms by which decorin promotes upregulation of p21, p27, and Ras, and ultimately protects cells in brain against OGD, are unknown.
Subventricular zone cells play a role in the recovery of function after neuronal injury and stroke (Zhang R et al, 2001; Zhang J et al, 2004; Arvidsson et al, 2002). These cells proliferate early after the injury and migrate to damaged areas of the brain. Furthermore, they survive over considerable periods and differentiate into mature cells. In the striatum, only 20% of the newly generated cells survive, resulting in an estimated replacement of only 0.2% of the cells that are lost as a result of ischemia (Arvidsson et al, 2002; Zhang et al, 2004). This response can be potentially increased by infecting SVZ cells with decorin-expressing lentivirus, which protects the cells under adverse conditions, and thereby decorin, by increasing the number of surviving neuronal stem cells, may enhance functional recovery after neuronal injury. We also speculate that transfection of decorin into parenchrymal cells may increase their resistance to hypoxic and ischemic injury. The proteoglycan decorin is a potent trophic factor for neural stem cells in vitro, which could give an important suggestion for survival of stem cells in injured brain tissue in the future. Increased decorin expression in tumor cells may promote the survival of these cells under potentially lethal insults, such as OGD. Overexpression of decorin in glioma cells may resist toxic agents designed to destroy them. Therefore, decorin, if expressed in tumor cells, should be blocked to increase the sensitivity of these cells to anticancer treatment.
Footnotes
Acknowledgements
We thank Dr Didier Trono for providing us lentivirus vectors and Dr Larry Fisher for decorin cDNA and antibodies.
