Abstract
Angiogenesis is regulated by concerted actions of angiogenic and angiostatic factors. Homeobox D3 gene (HOXD3) is a potent proangiogenic transcription factor that promotes angiogenesis by modulating the expression of matrix-degrading proteinases, integrins, and extracellular matrix components. Application of HOXD3 can promote angiogenesis in the skin, but its role in other vascular beds has not been examined. The authors examined HOXD3 expression in human brain vessels by in situ hybridization. Although little or no HOXD3 mRNA was detected in normal brain vessels, increased levels of HOXD3 and its target gene, αVβ3, were found in angiogenic vessels in human brain arteriovenous malformations. The authors further investigated whether HOXD3 plays a role in cerebral angiogenesis in a murine model. Expression of HOXD3 in mouse brain was achieved through retroviral vector—mediated HOXD3 gene transfer. HOXD3 expression lead to a significant induction of cerebral angiogenesis as shown by quantitative microvessel counting (HOXD3: 241 ± 19 vessels/mm2 vs. saline: 150 ± 14 vessels/mm2, P < 0.05). The data also showed that focal cerebral blood flow was increased in the angiogenic region with less vascular leakage. Moreover, expression of HOXD3 led to an increase in the expression of a direct downstream target gene αVβ3 integrin. The data suggest that HOXD3 may play an important role in regulating cerebral angiogenesis, and that gene transfer of HOXD3 may provide a novel and potent means to stimulate angiogenesis.
Angiogenesis, the sprouting of new capillaries from preexisting blood vessels, involves coordinated endothelial cell proliferation, migration, and reestablishment of patent tubular structures (Carmeliet and Jain, 2000;Jain, 2003). The proper development of new microvessels involves coordinated changes in the transcription of a number of genes, including those that mediate interactions between the endothelial cell and the extracellular matrix. The expression of matrix-degrading proteinases, integrin receptors, and extracellular matrix components is altered during the process of angiogenesis, and thus requires coordinate regulation to ensure proper vessel production (Brooke et al., 2003;Ingber, 2002). To this end, we have been investigating a role for the homeobox (HOX) gene family of master transcription factors in coordinately regulating the expression of genes involved in extracellular remodeling. The HOX genes were initially discovered for their roles in morphogenesis and organogenesis during development and, more recently, have been shown to be involved in adult tissue remodeling (Gorski and Walsh, 2000;Prince, 2002). Work from our laboratory and others has shown that HOX genes can profoundly influence endothelial cell behavior and angiogenesis (Boudreau et al., 1997). Moreover, HOX genes can directly influence the transcription of a number of extracellular matrix—remodeling genes including matrix-degrading proteinases, integrins, extracellular matrix components, growth factors, and other transcription factors (Gorski and Walsh, 2000;Myers et al., 2002;Wu et al., 2003). Specifically, we showed that HOXD3 increased the expression of several proangiogenic molecules such as urokinase plasminogen activator, integrin αVβ3, and type I collagen in microvascular endothelial scells in culture (Boudreau et al., 1997), and led to increases in angiogenesis in vivo (Hansen et al., 2003). Moreover, we observed that the promoters for both the β 3- and α5-integrin subunits were direct targets of HOXD3 transcriptional activity (Boudreau and Varner, 2004). HOXD3 can be induced by angiogenic factors including bFGF and developmentally regulated endothelial cell locus-1 (Del-1), and antisense against HOXD3 blocks both bFGF- and Del-1–induced angiogenesis in vivo (Zhong et al., 2003). Although a role for HOXD3 in inducing angiogenesis in the chick chorioallantoic membranes and in mouse skin has been established (Hansen et al., 2003), it is not clear whether HOXD3 can induce angiogenesis in other vascular beds such as the adult brain. Cerebral microvessels express the HOXD3 target gene, αVβ3 integrin, which undergoes a rapid up-regulation after focal ischemia, suggesting a functional role in cerebral angiogenesis (del Zoppo and Mabuchi, 2003). In this study, we tested whether retrovirally mediated HOXD3 expression could induce angiogenesis in adult mouse brain and lead to increased expression of its downstream target gene, αVβ3. We also investigated the expression pattern of HOXD3 in normal, nonangiogenic brain tissues and in brain arteriovenous malformations that exhibit excessive angiogenesis and vascular remodeling. Our data show that elevated HOXD3 expression led to significant increases in cerebral angiogenesis with increased functional blood flow and increased expression of αVβ3 integrin. Our findings suggest that HOXD3 activity may play an important role in regulating cerebral angiogenesis, and further suggest a means to therapeutically induce angiogenesis after cerebral ischemia.
MATERIALS AND METHODS
In situ hybridization of HOXD3
Human brain tissue was harvested at the time of surgery as previously described (Hashimoto et al., 2001, 2003). Tissues were fixed and embedded in paraffin, and in situ hybridization was performed on 5-μmol/L sections of tissue. Briefly, 415-bp sense and antisense digoxigenin-labeled Hox D3 RNA probes were generated as described elsewhere (Boudreau et al., 1997) using T7 or SP6 RNA polymerase and digoxigenin-conjugated dUTP (Genius; Roche Molecular, Indianapolis, IN, U.S.A.). In situ hybridization was performed after deparaffinization, re-hydration and digestion for 10 minutes with proteinase K. The sections were then hybridized overnight with 800 ng/mL of riboprobe at 50°C. After washing, the sections were incubated overnight with a 1:500 dilution of HRP-conjugated antibodies against digoxigenin and developed using NBT/BCIP (DIG nucleic acid detection Kit; Roche Molecular). Nonspecific hybridization was assessed using the sense riboprobe.
Construction of retroviral-expression vectors
Retroviral HOXD3 vectors were constructed as follows. Briefly, total RNA was isolated from primary human umbilical vein endothelial cells (HMVECs) using the Qiagen RNEasy kit (Qiagen, Valencia, CA, U.S.A.), and the entire HOXD3 cDNA was amplified for 40 cycles at 95/58/72°C using the following primers:forward primer: 5′-AGGGTCAGCAGGCCCTGG AGC-3′, corresponding to nucleotides 1370 to 1391 of the Human HOXD3 genes (Genbank accession number D11117), reverse primer: 5′-AGAGCGGGGAAGGGGGTTCCC GAACT-3′ corresponding to nucleotides 4598 to 4572 of the human genomic sequence. The resulting 1.3-kb HOXD3 cDNA was cloned into the TOPOII PCR cloning vector (Invitrogen, Carlsbad, CA, U.S.A.) and the sequence was confirmed using the Big Dye Terminator Sequencing (University of California, San Francisco, Biomolecular Resource Facility, San Francisco, CA, U.S.A.). To create the HOXD3 retrovirus, the entire 1.3-kb HOXD3 cDNA was excised from the TOPOII vector using Hind III and Xho I and a blunted Hind III/ Xho I fragment was ligated into the Hpa and Xho I sites of the viral vector pLXSN (BD Biosciences, Palo Alto, CA, U.S.A.). Virus was produced by CaPO4 transfection of the Phonix packaging cells and viral supernatants were collected 48 hours later and either used immediately or stored at −70°C. The viral vector pLXSN lacking any inserted sequence or with EGFP cDNA was used as a control. EGFP, the entire coding region of the EGFP cDNA, was excised with from the EGFPC2 vector (BD Biosciences) using Eco RI and Xho I and inserted into the pLXSN viral vector. In addition, adenoviral vector containing VEGF (AdVEGF) or lacZ genes (AdlacZ as control) was obtained from Dr. Rong Wen's lab at the University of Pennsylvania.
Retrovirus-mediated HOXD3 gene transfer in brain
The Institutional Animal Care and Use Committee approved procedures for the use of laboratory animals. Adult male CD-1 mice (Charles River Laboratories, Wilmington, MA, U.S.A.) weighing 30 to 35 g were anesthetized with 50-mg/kg ketamine and 10-mg/kg xylazine intraperitoneally. Animals were given a 30% O2 gas mixture via inhalation mask to maintain a PaO2 of 100 mm Hg or higher (Mao et al., 1999). After induction of anesthesia, the mice were placed in a frame with a mouse hold (Kopf Model 921; David Kopf Instruments, Tujunga, CA, U.S.A.), and a burr hole was drilled to the pericranium 2 mm lateral to the sagittal suture and 0.6 mm anterior to the coronal suture. A 10-μL Hamilton syringe was inserted into the lateral caudate about 3.0 mm under the cortex. Two microliters of supernatant containing 1 × 107 particles (pHOXD3 or pEGFP) was stereotactically injected into the right caudate putamen at a rate of 0.1 μL/min using an UltraMicroPump system (WPI, Sarasota, FL, U.S.A.). The needle was then withdrawn over the course of 20 minutes, the bone hole was sealed with bone wax, and the wounds were closed with a suture. The control animals received the same amount of saline injection. Three groups of mice (pHOXD3, pEGFP, and saline) with six mice per group underwent 1 to 5 weeks of treatment. A total of 90 mice were reanesthetized and killed, and the brain tissues were processed at different time points after the treatment.
Microvessel staining using lectin and microvessel counting
Microvessel staining and counting were performed as described (Yang et al., 2003). Briefly, 5 minutes before the animals were killed, fluorescein-lycopersicon esculentum (tomato) lectin (100 μg/100 μL, Vector Laboratories, Burlingame, CA, U.S.A.) was injected intravenously. Animals were exsanguinated while perfused intracardially with a solution of 4% para-formaldehyde in phosphate-buffered saline (pH 7.4). The brains were removed and postfixed in the same solution for 4 hours, and then immersed in 30% sucrose solution until they sunk. The brains were then embedded in Tissue-Tek O.C.T. (Sakura Finetek Inc., Torrance, CA, U.S.A.). Twenty-micrometer-thick coronal sections were cut using a cryostat.
Two brain coronal sections from the lectin fluorescein staining brain, 1 mm anterior and 1 mm posterior from the needle track, were chosen. Three areas of microvessel, immediately to the left, right, and bottom of the needle track, were chosen, and photographed using a 10× objective. Microvessel counting was performed on these photographs. Vessels with a diameter between 3 and 8 μm were counted. The microvessel counting was conducted in a blinded fashion. The numbers of microvessel were calculated as the mean of the blood vessel counts obtained from three pictures.
Immunohistochemistry
As described previously (Yang et al., 2001), frozen sections were dried for 30 minutes and fixed in acetone for 15 minutes at −20°C. The sections were incubated with Laszlo's blocking solution (10 mmol/L Trizma, 500 mmol/L NaCl, and 0.05% Tween-20; pH7.6) for 30 minutes in a moisture chamber to block nonspecific binding and then incubated with rat anti-MEC antibody (1:00 dilution; Pharmingen, Palo Alto, CA, U.S.A.) overnight at room temperature. The sections were incubated with biotin rat immunoglobulin G (1:200 dilution; Bio Care Inc., Walnut Creek, CA, U.S.A.) for 1 hour at room temperature and followed by an ABC process (Vector Laboratories). The sections were then developed with DAB (Zymed Inc., South San Francisco, CA, U.S.A.) and counter-stained with hematoxylin (Sigma, St. Louis, MO, U.S.A.). Immunostaining of frozen sections of human brain AVM tissue was performed as described previously using monoclonal mouse anti—human αVβ3 antibody (1:50 dilution; Chemicon, Temecula, CA, U.S.A.) as a primary antibody and horse anti—mouse immunoglobulin G as a secondary antibody. Negative controls were performed by omission of primary antibody.
Blood—brain-barrier permeability was determined using albumin staining as described by Yang and Betz (1994). The brain coronal sections were incubated with rabbit anti—mouse albumin antibody (1:400 dilution, Vector Laboratories) and biotinylated goat anti—rabbit secondary antibody followed by an ABC process. Albumin-positive staining areas showed a dark-brown color. Negative controls were performed by omission of primary antibody.
Integrin-β3 mRNA expression and reverse-transcription PCR
Left and right sides of mouse brains were excised and placed into Trizol reagent (Gibco Life Technologies) and immediately frozen in liquid nitrogen and stored at −70°C. Tissues were subsequently homogenized using a 22-G syringe and total RNA was extracted using the Trizol reagent according to the manufacturer's instruction. One microgram of total RNA was reverse transcribed using MMuLV RT (Life Technologies), and 1/25 of this reaction was linearly amplified for 25 cycles at 95°C, 50°C and 72°C using Taq polymerase (Qiagen) with the following primers:forward: 5′-ACTACTTGATGGACCTGTCTT-3′, reverse: 5′-CATTTTCAGTCACTGCAAAGATC-3′, corresponding to base pairs 438 to 458 and 1012 to 1034 of the human 3′ integrin gene (Genbank accession number NM 000212.1). Total RNA was normalized using alternative 18s internal standards at a 2:8 ratio (Ambion, Austin, TX, U.S.A.). PCR products were resolved by electrophoresis through 0.8% agarose gels and visualized by staining with ethidium bromide. The 596-bp PCR product corresponding to 3′ integrin was ligated into the TOPO II TA vector (Invitrogen) and the identity was confirmed by sequencing (Biomolecular Resource Center, University of California, San Francisco, CA, U.S.A.).
Cerebral blood flow measurement
For surface CBF measurement, a laser-Doppler flowmetry monitor (Model BPM2 System; Vasamedics Inc., St. Paul, MN, U.S.A.) equipped with a small-caliber probe 0.7 mm in diameter (P-433, Vasamedics) was used. Two points were measured: the injection region 2.5 mm lateral to the sagittal suture and 0.1 mm anterior to the coronal suture on the right side, and points on the noninjected left side. Baseline blood flow recordings of the two regions were made 5 minutes before viral vector injection. Changes in CBF were measured 15 minutes before the animals were killed at different time points after vector transduction. Blood flow values were calculated and expressed as a percentage of baseline values.
Statistical analysis
Parametric data among the p HOXD3, pEGFP-transduced, and saline-treated groups were compared using a one-way analysis of variance followed by the Scheffé test. All data are presented as means and standard deviations. A probability value of less than 5% was considered to represent statistical significance.
RESULTS
Expression of HOXD3 and integrin αVβ3 in normal and angiogenic brain tissue
To begin to investigate whether HOXD3 may play a role in cerebral angiogenesis, we performed in situ hybridization of normal, nonangiogenic brain vasculature and of vessels in brain AVMs, which are characterized by excessive angiogenesis and vascular remodeling (Fleetwood and Steinberg, 2002). Whereas the expression of HOXD3 was almost undetectable in normal brain tissue (Fig. 1A), there was a marked increase in the expression of this proangiogenic gene in endothelial cells and in adjacent stromal tissue in brain AVMs (Figs. 1B and 1C). The HOXD3 target gene, integrin αVβ3, was also expressed in the vessels of brain AVMs (Figs. 1D and 1E). These results suggest that HOXD3 plays a role in cerebral angiogenesis.
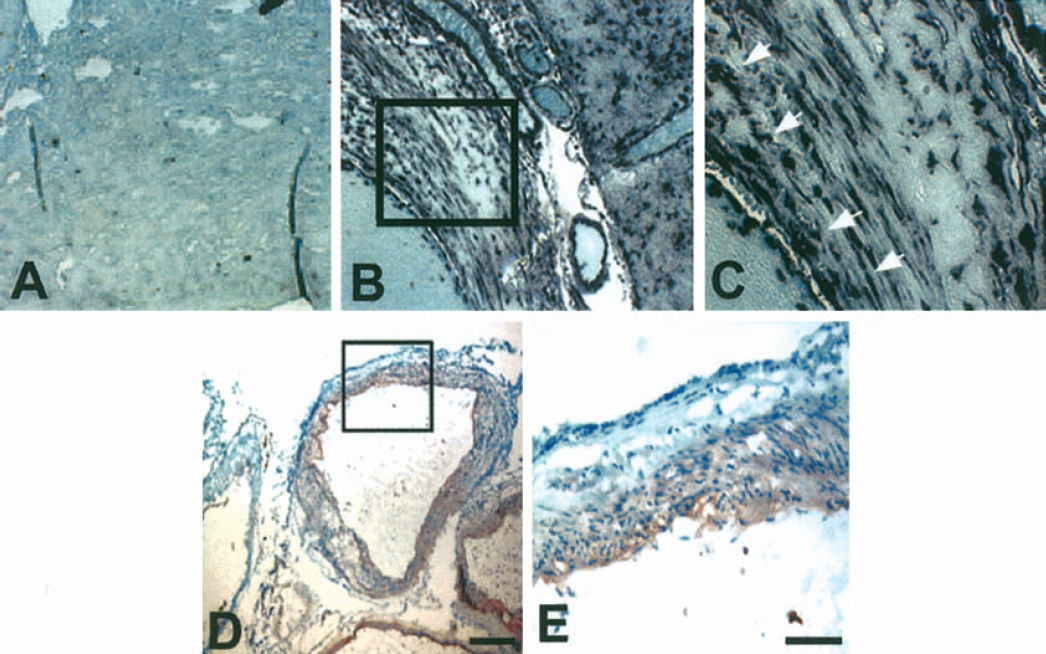
Expression of HOXD3 and aVβ3 integrin in normal brain and brain AVM tissue. (
Induction of cerebral angiogenesis after retroviral vector delivery of HOXD3
The extent of the virus spread was quantified using image analysis to determine the brain area that was positive for green fluorescence in serial sections. We found that the spread of the virus was approximately 3 mm away from the needle track, and green fluorescence could be detected up to 2 weeks after pEGFP injection. To directly determine whether HOXD3 could induce angiogenesis in the brain, we constructed retroviral vectors to express HOXD3. Control or HOXD3 expressing retrovirus (1 × 107 particles/2 μL) or normal saline was stereotactically injected into the right caudate putamen of the mice brain. The mice showed no visible neurologic deficits such as hemiparesis, epilepsy, or abnormal behavior up to 5 weeks after the injections of pHOXD3, pEGFP, or saline. After 1 or 2 weeks of HOXD3 transduction, we observed dilated vessels. During 2 or 3 weeks of HOXD3 transduction, angiogenic sprouts arose from enlarged microvessels. Angiogenic sprouting is a consequence of the angiogenic response of preexisting vessels to angiogenic stimuli. Sprouts developed by endothelial cell proliferation and migration. As shown in Fig. 2, angiogenic sprouts (circles) arose from enlarged vessels. To determine whether HOXD3 transduction induces brain angiogenesis, we assessed microvessel density after perfusion with a FITC-labeled vascular lectin. Fig. 3A shows that lectin specifically stains blood vessels as determined by colocalization with CD-31. Compared with saline-treated or control EGFP virus—treated mice, those transduced with HOXD3 showed significantly increased vascular labeling with the lectin as early as 2 weeks after the injection (Figs. 3B and 3C). Assessment of microvessel density indicated that HOXD3-treated mice showed significantly elevated vascular densities for up to 5 weeks after HOXD3 gene delivery. Moreover, despite the sustained increase in vascular density, we did not observe evidence of abnormal vessel morphology for up to 5 weeks after administration of HOXD3 (data not shown).
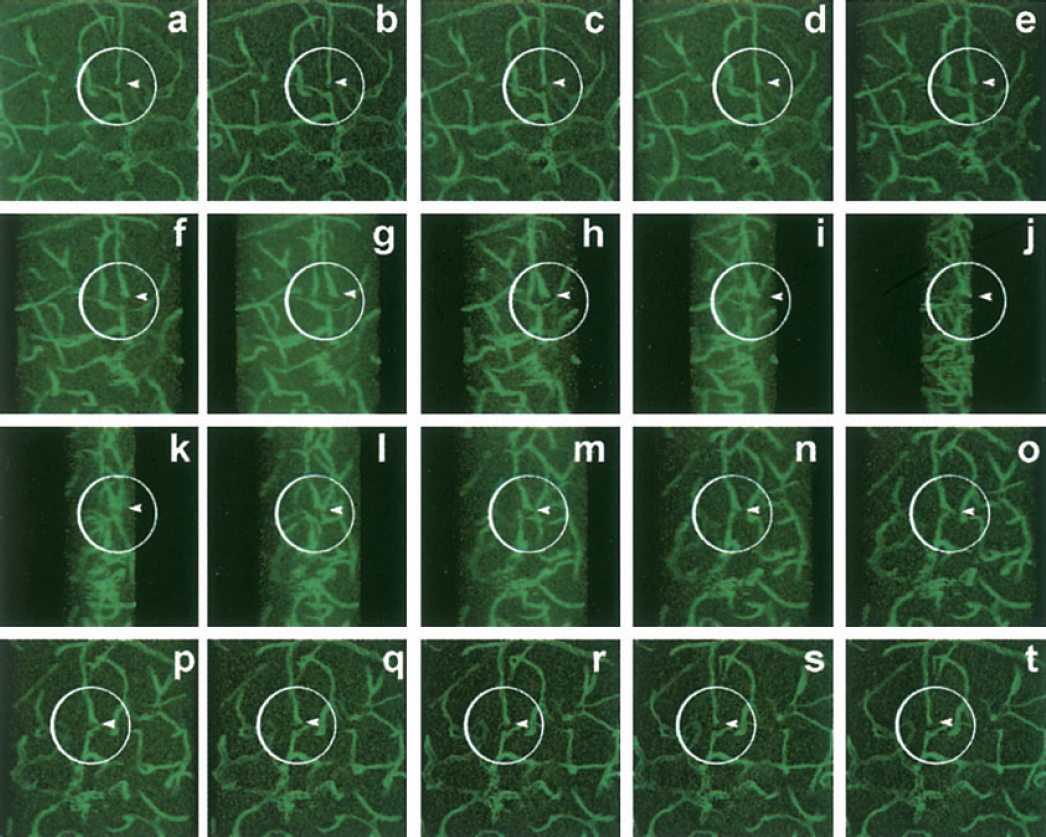
Confocal microscopy and three-dimensional reconstruction images showed angiogenic sprout induced by HOXD3 gene transfer. A series of 5-degree-rotation pictures are reconstructed from 20 consecutive 1-μm-thick sections to confirm the newly formed microvessels after 2 weeks of HOXD3 gene transfer. Arrowhead indicates the formation of angiogenic sprout (circles) from enlarged vessels.
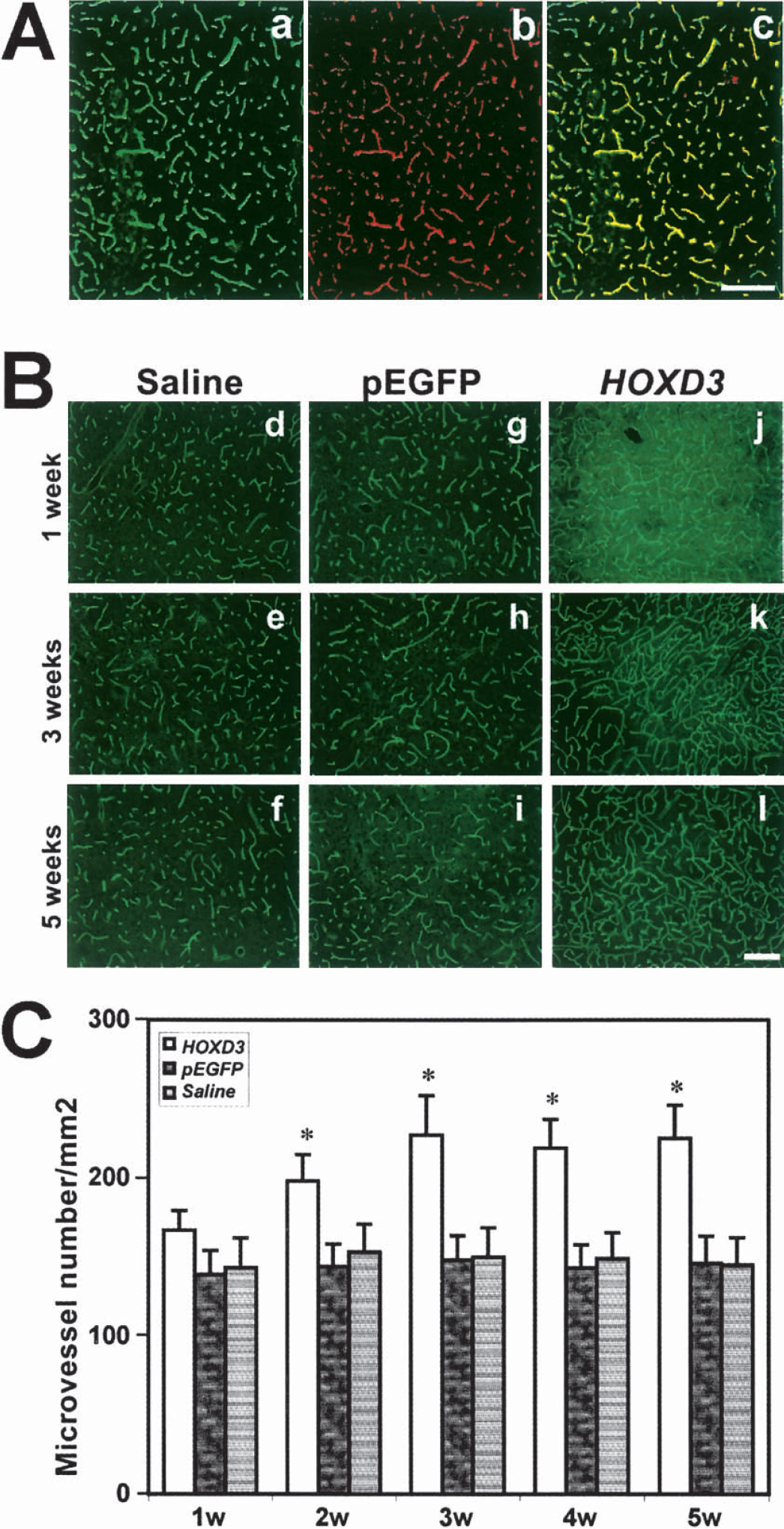
HOXD3 gene transfer increases the number of cerebral microvessels. (
We further examined cell proliferation by immunostaining with PCNA, an intranuclear acidic protein highly expressed in proliferating cells. As shown in Fig. 4, PCNA immunoreactivity was increased in the HOXD3-transduced tissue as compared with the pEGFP-injected brain tissue, indicating that increased angiogenesis was accompanied by enhanced cell proliferation. Colocalization of PCNA signal with MEC/CD31 staining (Fig. 4A) indicated that HOXD3 promotes the proliferation of endothelial cells. Images of the newly formed vessels at high magnification (Fig. 4B) suggested that the majority of vessels were in the capillary range (3 to 8 μm in diameter).
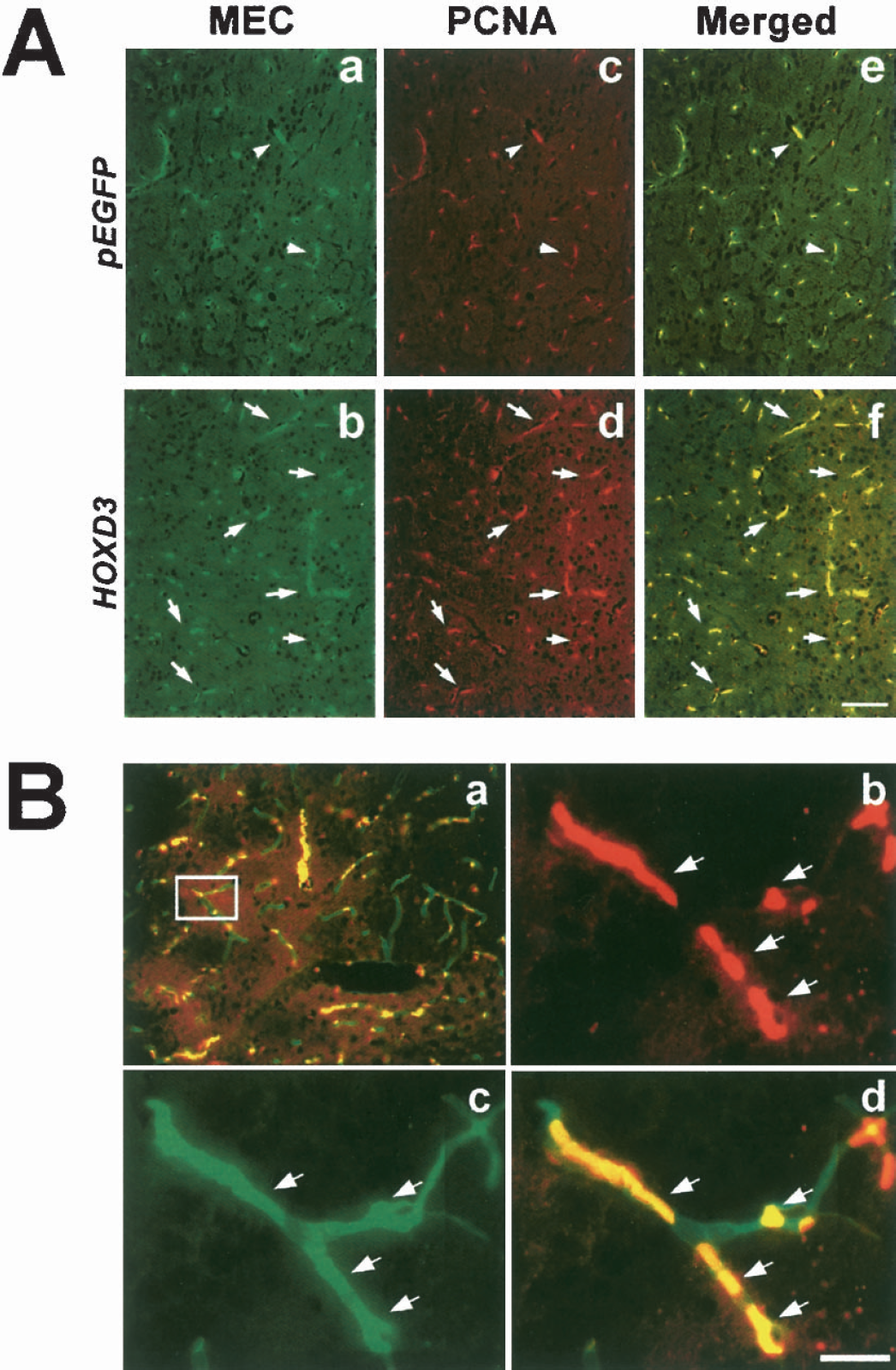
Endothelial cell proliferation is increased in the HOXD3 transduced mice. (
To determine whether gene transfer of HOXD3 also resulted in increased expression of the HOXD3 target gene, αVβ3 integrin, we performed semiquantitative reverse-transcription PCR on brain tissues infected with control virus or HOXD3 expressing retrovirus. In brain tissue treated with control pLXSN virus, no differences in the mRNA levels of the β3-integrin subunit of αVβ3 integrin were observed between the right side of the brain, which received the control virus, and the sham-injected left side of the brain (Fig. 5A). In contrast, increased expression of β3 integrin mRNA was detected in tissues injected with HOXD3 retrovirus as compared with the contralateral sham-injected tissue (Fig. 5B). Thus, the HOXD3-mediated increase in angiogenesis in brain tissue correlated with increased expression of its target gene, αVβ3 integrin.
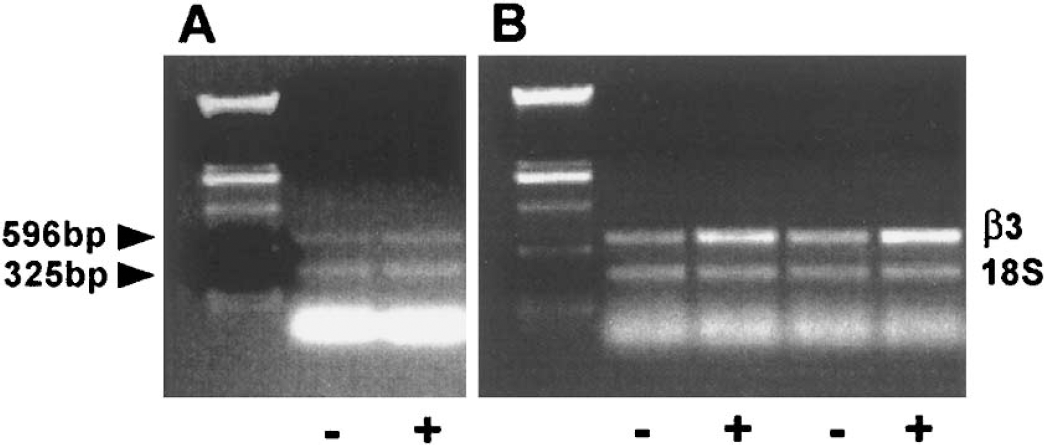
HOXD3-induced angiogenesis is associated with increased expression of β3 integrin. (
HOXD3 treatment enhances focal cerebral blood flow
To assess whether the increase in microvascular density induced by HOXD3 resulted in an increase in cerebral microvascular blood flow, we performed laser-Doppler flowmetry at different time points after the injection. The advantages of laser-Doppler flowmetry measurement are that it is noninvasive, simple, fast, and the CBF changes can be continuously monitored in animals that can be further used to study other endpoints. Laser-Doppler flowmetry can detect blood flow approximately 2 mm deep from the surface of the brain. Therefore, changes in striatal blood flow, at least in part, could be detected by the laser-Doppler flowmeter. To support the laser-Doppler measurements, magnetic resonance imaging or radioisotope assay might be used in the future to quantify regional CBF. HOXD3 gene transduction, but not pEGFP or saline transduction, substantially increased CBF as measured by laser-Doppler flowmetry. Increased CBF was detected by 3 weeks after HOXD3 gene delivery, and remained elevated for 5 weeks after injection (Fig. 6A) corresponding with the increases in vascular density observed with HOXD3 gene treatment. As shown in Figs. 3C and 6A, the increased microvessel density correlated with increased CBF after HOXD3 transduction from week 3 to 5. The microvessel number, but not the CBF, was increased at 2 weeks after injection. This result suggested that newly formed vessels were not mature enough at early stages to establish functional collateral flow.
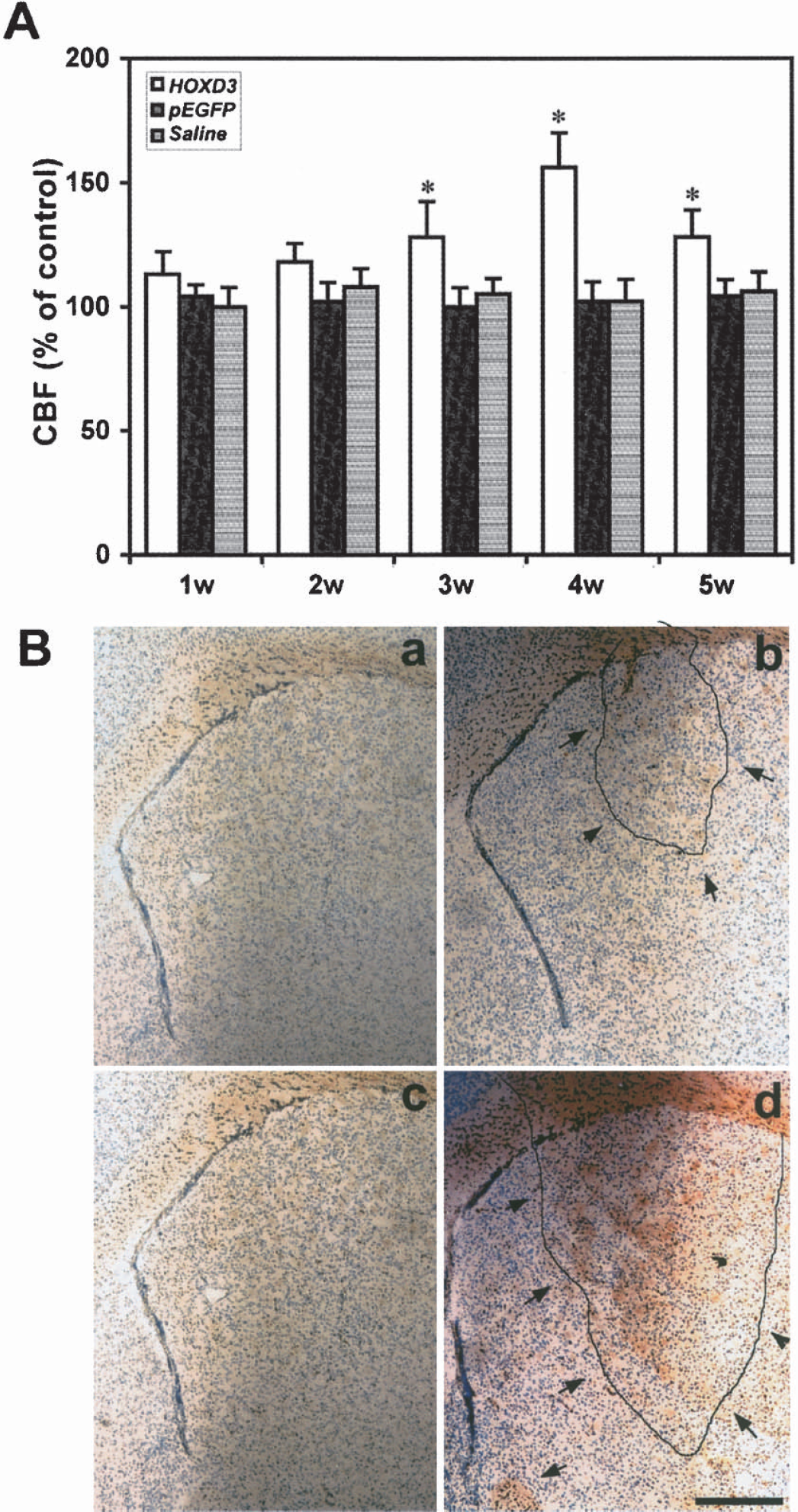
HOXD3 gene transduction increases cerebral blood flow without disrupting blood—brain-barrier integrity. (
To determine whether newly formed microvessels were mature and stable, we assessed blood—brain-barrier integrity using albumin immunohistochemistry. Because VEGF is a well-established inducer of vascular permeability (Wang et al., 1996;Zhang et al., 2000), we injected mice with an adenoviral vector—expressing VEGF as a positive control. Fig. 6B shows that whereas infection with VEGF induces strong albumin immunoreactivity indicative of increased vascular permeability, HOXD3 treatment did not lead to significant albumin staining. Brain tissues injected with saline or pEGFP did not show any positive albumin signal. This indicates that not only does HOXD3 increase vascular density and cerebral flow, but also that the vessels HOXD3 induced were stable and mature.
DISCUSSION
In the present study, we show that gene transfer of HOXD3 using a retroviral vector induced impressive cerebral angiogenesis and increased cerebral flow. Moreover, the newly formed microvessels were stable and did not display excessive permeability. The majority of the newly formed vessels were in the capillary range because the vascular diameter was between 3 and 8 μm. These results suggest that HOXD3 mainly promoted angiogenesis. Arteriogenesis may also occur after initial angiogenesis and contributes to increased CBF.
Angiogenesis is essential for development, normal growth, and repair. It is complete around postnatal day 20, and is suppressed in mature tissues. However, angiogenesis is triggered in many pathologic processes, including tumor progression and ischemic diseases (Bikfalvi and Bicknell, 2002;Hayashi et al., 2003;Zhang and Chopp, 2002). The role of angiogenesis in pathophysiology is different in each disorder, and manipulation of new vessel production could be effective treatments for some diseases. Whereas suppression of angiogenesis can block tumor growth (Bikfalvi and Bicknell, 2002;Folkman, 1995), potentiation of new vessel formation may represent an effective means to reduce ischemic injury because ischemic tissue is dependent on restoration of microvascular circulation. Brain ischemia induces angiogenesis (Carmeliet, 2003;Hayashi et al., 2003), and the number of new vessels produced in the ischemic penumbra is correlated to decreased morbidity and longer survival in stroke patients (Cramer et al., 1997;Krupinski et al., 1994;Weiller et al., 1993), suggesting that restoration of cerebral microvascular circulation is important for functional recovery after ischemic attack.
HOXD3 is a member of the highly conserved family of homeobox proteins, which regulate pattern formation and segmental identity along the anterior/posterior axis during development. HOX transcription factors regulate cell proliferation, differentiation, and migration (Gorski and Walsh, 2000;Prince, 2002). These transcription factors are involved in the final transcriptional control of the genes responsible for the vascular remodeling and angiogenesis (Gorski and Walsh, 2000;Prince, 2002). HOXD3 promotes angiogenesis by upregulating the expression of several proangiogenic molecules including integrin αVβ3 (Boudreau et al., 1997;Gorski and Walsh, 2000). αVβ3 is a heterodimeric receptor consisting of the 3 V and 33 subunits, which bind to a variety of extracellular matrix proteins including vitronectin, tenascin, and fibrinogen. This integrin promotes angiogenesis by regulating cell adhesion and migration (Carmeliet 2002;Travis et al., 2003). Function blocking antibodies against αVβ3 has been shown to block both tumor- or cytokine-mediated neovascularization (Brooks et al., 1994b, 1995;Eliceiri et al., 1998). Whereas the α V subunit is constitutively expressed in many endothelial cells, expression of the β 3 subunit is the rate-limiting step in synthesis of the functional surface receptor (Carmeliet, 2002;Travis et al., 2003). Although αVβ3 expression is low in resting vessels, its expression is rapidly induced after exposure to angiogenic cytokines including bFGF and Del-1, both of which activate HOXD3 and transcription of the β3 subunit (Brooks et al., 1994a;Penta et al., 1999;Sepp et al., 1994). Studies in brain cerebral vessels also show that expression of integrin is rapidly induced in αVβ3 response to focal ischemia (del Zoppo and Mabuchi, 2003). Although, a direct role of αVβ3 in mediating angiogenesis in the brain has not been shown, our studies, which link HOXD3, β3 integrin expression, and increased cerebral vascular density, further support a role for this integrin in cerebral angiogenesis.
We observed very high levels of HOXD3 and αVβ3 integrin in association with the brain AVM tissues (Fig. 1). Also, our gene microarray studies showed that αVβ3 integrin was upregulated in brain AVM tissues (Hashimoto et al., 2004). Brain AVMs are complexes of tortuous, tangled vessels representing fistulous connections between arteries and veins and lacking a capillary bed. The underlying pathogenesis of the formation and progression of brain AVMs is unknown, but studies suggest that aberrant expression of angiogenic mediators, including VEGF and Ang-2, may contribute to the vascular morphology (Hashimoto et al., 2000, 2001). Unlike human brain AVMs, which usually display several tortuous, branching, high-flow arterial vessels of varying caliber and wall thickness supplying the central nidus, the vessels induced by HOXD3 gene transfer in our murine model are of normal morphology without vascular leakage. Furthermore, in contrast to the aberrant activities of various angiogenic factors expressed in brain AVMs, the regulated gene transfer of HOXD3 in our murine model may not lead to pathologic angiogenesis. It is worth noting that our previous studies in chick embryos showed that sustained high levels of HOXD3 expression led to the formation of cavernous vascular structures resembling hemangiomas (Boudreau et al., 1997). However, in subsequent work in mouse dermis (Hansen et al., 2003) and in the present study in adult mouse brain, no evidence of aberrant vascular morphology was observed after HOXD3 administration. One possible explanation for this finding may be that embryonic vessels are more susceptible to disruption and development of hemangioma than adult (Stoker et al., 1990). Alternatively, the limited degree of retroviral infection in the adult brain may not produce as sustained high level of HOXD3 as was achieved in embryonic tissue, which is undergoing extensive proliferation. Nonetheless, our current work shows that limited expression of HOXD3 may provide a means to induce functional, normal vessels in the adult brain after stroke.
Footnotes
Acknowledgments
The authors thank Carroll Schreibman and Broderick Belenson for editorial assistance and the collaborative support of the staff of the Center for Cerebrovascular Research.
