Abstract
Hyperthermia worsens outcome in clinical and experimental studies of ischemic stroke. Thus, we tested whether hyperthermia aggravates intracerebral hemorrhage (ICH) in rats. A striatal hemorrhage was produced via an infusion of bacterial collagenase. In a preliminary experiment, we compared brain and core temperatures (via telemetry) during heating (infrared lamp). The brain temperature rise exceeded that produced by enforced core hyperthermia, which was used subsequently. In these experiments up to three hyperthermia conditions (versus normothermia) were tested including: hyperthermia (>38.5°C) over the first (HYP-1) or second 24 h period (HYP-2) after ICH and 3 h of 40°C hyperthermia starting 12 h after ICH (HYP-3). The HYP-1, HYP-2, and HYP-3 treatments did not affect functional deficits (e.g., spontaneous forelimb use, skilled reaching) or the volume of injury at 30 days. Furthermore, the HYP-1 treatment did not aggravate injury or deficits at 7 days. Bleeding and inflammation, which contribute to pathology, were not significantly altered by HYP-1 and HYP-3 treatments. Bleeding was assessed at 1 day, and macrophages and neutrophils were counted at 2 and 4 days. Accordingly, hyperthermia, under the present conditions, did not worsen outcome after striatal ICH.
Introduction
Approximately 15% of strokes in Western populations are the result of an intracerebral hemorrhage (ICH); which is the most devastating and least treatable type of stroke (Broderick et al 1999). Current treatment of ICH is primarily supportive in nature (e.g., control of blood pressure) and outcome is generally poor. Despite the fact that increased body temperature frequently occurs in patients with an ICH (Schwarz et al, 2000), few studies have examined the prognostic significance of hyperthermia on ICH outcome. Several clinical studies of ICH patients with elevated body temperature found that hyperthermia does not independently predict mortality (Boysen and Christensen, 2001; Schwarz et al 2000; Szczudlik et al, 2002; Wang et al, 2000). The small number of investigations and conflicting results necessitate further investigation to determine the significance of hyperthermia after ICH.
Contrary to discordant findings in ICH, the relationship between fever and worsened outcome in ischemic stroke patients appears clear. Elevated body temperature on admission (Reith et al 1996; Wang et al, 2000) or in the days after ischemia (Azzimondi et al 1995; Castillo et al, 1998) increases morbidity and mortality. Given that hyperthermia of as little as 0.5°C significantly worsens outcome (Hindfelt, 1976; Reith et al, 1996), it is recommended that body temperature be maintained at normothermic levels after stroke (Ginsberg and Busto, 1998). However, it should be noted that there is no clinical evidence proving that antipyretic treatments improve outcome.
Likewise, small temperature elevations in rodent models of ischemic injury critically affect outcome (for a review see Ginsberg and Busto, 1998). Hyperthermia during or after global ischemia (Baena et al, 1997; Busto et al, 1987; Dietrich et al, 1990; Minamisawa et al, 1990) exacerbates neuronal injury. Similarly, spontaneous (Li et al, 1999; Reglodi et al, 2000; Zhao et al, 1994) and externally induced hyperthermia (Chen et al, 1991; Meden et al, 1994) increases focal ischemic injury, even when delayed for 24 h after ischemia (Kim et al, 1996). Hyperthermia is thought to aggravate ischemic injury through several mechanisms including: increased blood-brain barrier disruption (Dietrich et al, 1990, Dietrich et al, 1991), enhanced release of excitotoxic neurotransmitters such as glutamate (Takagi et al, 1994), increased production of free-radicals (Globus et al, 1995), increased spreading depression (Back et al, 1996; Chen et al, 1993), greater cytoskeletal degradation (Eguchi et al, 1997), and impaired metabolism (Back et al, 1995).
In experimental ICH, the majority of damage is rapid and is caused by compression and displacement of brain tissue after the sudden development of an intracerebral mass. However, several components of hemorrhage resemble ischemic injury. For instance, hemorrhage triggers secondary events such as disruption of the blood-brain barrier (Xi et al, 2001), edema formation (Xi et al, 1998), excessive production of free radicals (Peeling et al, 1998), and pronounced inflammation (Del Bigio et al, 1996; Rosenberg et al, 1990). Accordingly, we examined whether mild to moderate hyperthermia affects factors known to influence outcome after ICH, such as inflammation (Peeling et al, 2001) and bleeding (Auriat et al, 2005; MacLellan et al, 2004). We assessed outcome at multiple time points using histopathology (e.g., lesion volume) and a battery of behavioral tests sensitive to hemorrhagic injury (Auriat et al, 2005; DeBow et al, 2003; MacLellan et al, 2004; Peeling et al, 2001). We hypothesized that hyperthermia would worsen functional and histologic outcome in part by increasing inflammation.
Methods
Animals
All procedures followed the Canadian Council on Animal Care guidelines and were approved by a local animal care committee. A total of 144 male Sprague-Dawley rats were obtained locally and weighed between 350 and 400 g (~16 weeks old) at the time of ICH. Rats were randomly assigned to treatment groups.
There were four separate experiments in this study (see Table 1, detailed methods follow) that each tested up to four groups: normothermia (NORMO; temperature kept >36.5°C via 175 W infrared lamp), mild hyperthermia (38.5°C) for the first 24 h (HYP-1) or second 24 h period (HYP-2) after ICH, and moderate brief hyperthermia (40°C for 3 h) starting 12 h post-ICH (HYP-3). Rats were otherwise maintained at normothermia for up to 48 h after ICH (e.g., for the first 24 h period in the HYP-2 treatment condition). In a preliminary study (N = 3 total), we examined the relationship between brain and body temperature under post-ICH hyperthermia conditions. Experiment 1 (N = 62) examined whether the three hyperthermia treatments worsen long-term (i.e., 30 days) histologic and functional outcome after ICH (n = 15 to 16 each). We assessed skilled reaching, walking, swimming, limb use asymmetry, as well as the volume of tissue lost. To determine if there were transient effects of hyperthermia that could not be detected at later times, we assessed functional (walking and limb use asymmetry) and histologic (lesion volume) outcome 7 days after ICH in NORMO (n = 8) and HYP-1 (n = 8) treatment conditions (Experiment 2). In Experiment 3, we measured neurologic deficits 1 to 4 days after ICH and assessed parenchymal infiltration of macrophages and neutrophils at 2 and 4 days in NORMO (n = 12), HYP-1 (n = 12), and HYP-3 (n = 13) rats. In Experiment 4 (NORMO, n = 8; HYP-1, n = 9; HYP-3, n = 9), we used a spectrophotometric assay of hemoglobin (MacLellan et al, 2004) to determine whether hyperthermia affects the extravasation of blood into the brain after ICH.
Summary of treatment conditions and endpoints in each experiment
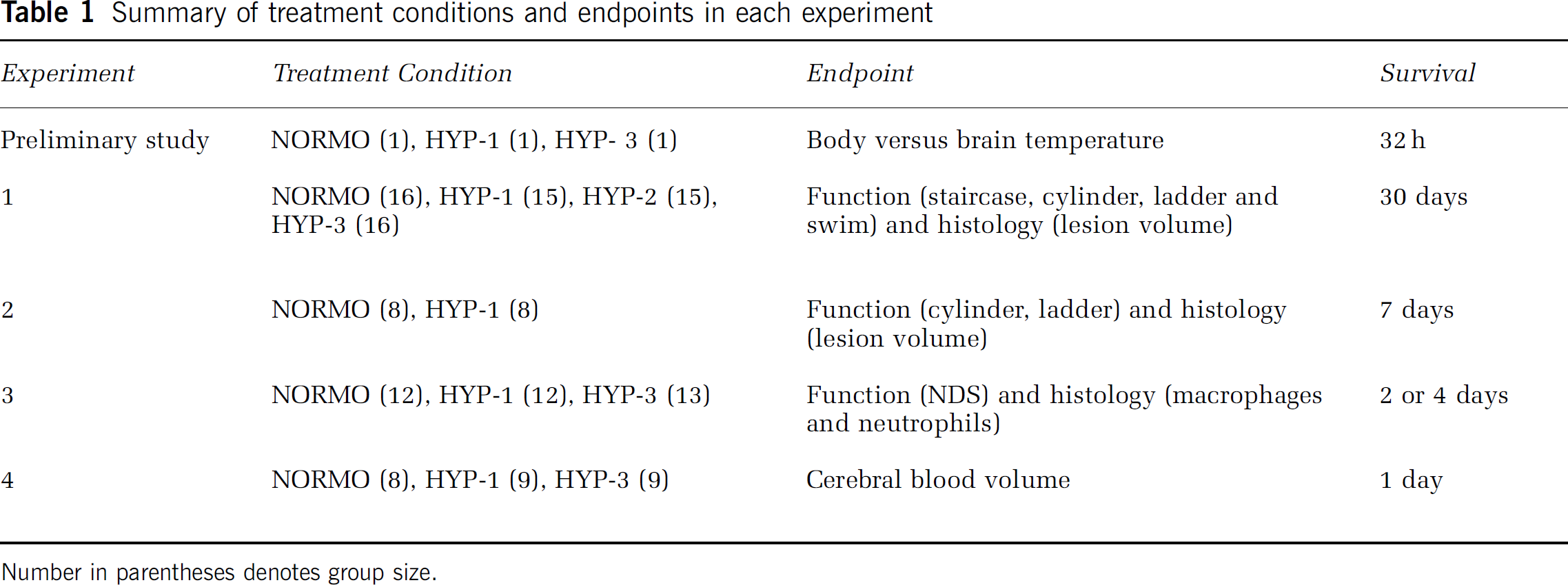
Number in parentheses denotes group size.
Surgical Procedures
All surgical procedures were performed under isoflurane anesthesia (4% induction; 2% maintenance in 70% N2O and 30% O2) using aseptic technique.
Telemetry temperature probe implantation: All animals had core temperature telemetry probes (model TA10TA-F40; Transoma Medical, St Paul, MN, USA) implanted into the peritoneal cavity 3 days before ICH surgery as described previously (MacLellan et al, 2004). After surgery, rats were individually housed on receivers interfaced to a computer running A.R.T. software (Transoma Medical) that sampled temperature every 30 secs. Baseline core temperature was recorded for a complete 24 h period before ICH. After ICH, temperature was monitored for up to 7 days (Experiments 1 and 2) or until euthanasia in experiments with shorter survival times. In Experiment 1, core temperature telemetry probes were removed under brief anesthesia 7 days after ICH.
In a pilot study, several rats were implanted with a core temperature telemetry probe as well as a telemetry brain probe (model XM-FH; Mini Mitter Co., Inc., Bend, OR, USA) immediately after ICH so that the temperature of the contralateral dorsal striatum was also measured in NORMO, HYP-1, and HYP-3 rats (n = 1 each). Continuous and simultaneous sampling of brain and core temperature is not possible owing to signal interference. Instead, core temperature was sampled every 30 secs followed by intermittent (every 4 h) brain temperature recordings (DeBow and Colbourne, 2003). We found a strong relationship between brain and core temperature measured near-simultaneously after ICH (NORMO; r = 0.892), although brain temperature was ~0.9°C lower than core temperature (mean of 36.5°C versus 37.4°C, respectively). However, as core temperature was elevated in the HYP-1 and HYP-3 rats, brain temperature also increased and eventually exceeded core temperature. Owing to technical difficulty in securing a probe on the head while allowing access to the striatum for collagenase infusion, we elected to regulate core temperature in other experiments.
Intracerebral hemorrhage: All rats were anesthetized and placed in a stereotaxic frame. A hole was drilled 3 mm lateral (right) to bregma and a 26-gauge needle (Hamilton syringe, Hamilton, Reno, NV, USA) was lowered 5.5 mm below the surface of the skull. To create an ICH, 0.8 μL of sterile saline containing 0.16 U bacterial collagenase (Type IV-S, Sigma, Oakville, ONT, Canada) was infused into the striatum over 5 mins. After infusion, the needle remained in place for an additional 5 mins, then a metal screw (model MX-080-2, Small Parts, Miami Lakes, FL, USA) was used to seal the hole. The scalp wound was treated with Marcaine (Sanofi, Markham, ONT, Canada) and closed with staples. Rats were not intubated during surgery to minimize invasiveness and duration of anesthetic.
Behavioral testing
Staircase task (Experiment 1): Three weeks before ICH surgery (Experiment 1), rats were food deprived to 90% of free-feeding weight and trained in the staircase test, to measure independent forelimb reaching ability. Briefly, rats received two 15-min trials per day separated by 4 to 5 h, 5 days per week (DeBow et al, 2003; MacLellan et al, 2004). Ten rats failed to reach the criterion of 9 pellets (45 mg each; Bio-Serv, Frenchtown, NJ, USA) per side out of a possible 21 by the last 3 consecutive days of training, and were thus excluded from just this test.
Skilled reaching deficits were assessed 24 to 28 days post-ICH. Testing consisted of 10 trials (two 15-min trials per day, separated by 4 to 5 h) during which rats were maintained at 90% of their free-feeding weight. We recorded the total number of pellets retrieved on each side for each trial, and expressed performance as a percent of baseline (average of the last 10 training trials).
Swimming task (Experiment 1): Rats in Experiment 1 were trained to swim to a visible platform located at one end of a rectangular aquarium (length: 123, width: 46, height: 57 cm) for 3 days before core probe implantation. Each rat was trained (~10 consecutive trials/day for 2 days) to swim from one end of the tank directly to the platform without touching the walls (Gonzalez and Kolb, 2003). Three baseline trials were videotaped for each animal on the third day. Normally, the hind limbs are used to propel the rat through the water while the forelimbs are held under the jaw. Damage to the motor cortex, often accompanied by injury to the dorsolateral striatum, causes asymmetry in forelimb inhibition and use of the impaired limb during swimming (Gonzalez and Kolb, 2003). Thirty days after ICH, three trials were videotaped for each animal as they swam directly to the platform. We recorded the number of strokes made with each forelimb and calculated an asymmetry score for forelimb inhibition:
Number of contralateral forelimb strokes
– number of ipsilateral forelimb strokes
Forelimb use asymmetry test (Experiments 1 and 2): At 7 (Experiment 2) or 30 days (Experiment 1) after ICH, rats individually explored a transparent cylinder (45 cm in height and 20 cm in diameter) for 10 mins. A video camera set up below the cylinder recorded each animal's movements, which were analyzed according to our previous work (MacLellan et al, 2004). Briefly, independent forelimb use for weight support during exploration of the walls was expressed as:
(Number of contacts with contralateral limb/ipsilateral + contralateral limb use) × 100
Horizontal ladder walking test (Experiments 1 and 2): At 7 (Experiment 2) or 30 days (Experiment 1) after ICH, rats were videotaped walking across a 1 m horizontal ladder with variably spaced bars ranging from 3 to 5 cm apart. For each forelimb, the percentage of footfalls (limb slips completely through the bars) made over four trials was determined (DeBow et al, 2003; MacLellan et al, 2004).
Neurologic deficits score (Experiment 3): Neurologic deficits were measured 1 to 4 days after ICH (Del Bigio et al, 1996; Peeling et al, 2001) in Experiment 3. Tests included (1) spontaneous ipsilateral circling, graded from 0 for no circling to 3 for continuous circling; (2) hind limb retraction, graded from 0 for immediate replacement to 3 for no retraction after the hind limb was moved laterally 2 to 3 cm; (3) bilateral forepaw grasp, graded from 0 for normal grasping behavior to 3 for a rat unable to grasp with the forelimbs; (4) contralateral forelimb flexion, graded from 0 for uniform extension of forelimbs to 2 for full wrist flexion and shoulder adduction when the rat was gently lifted by the base of the tail; and (5) ability to walk a 70 cm long × 2.4 cm wide beam; graded from 0 for a rat that readily traversed the beam to 3 for a rat unable to stay on the beam for >10 secs. Scores for each component were added for a maximum of 14 (greatest impairment).
Histopathology
Volume of brain tissue destruction (Experiments 1 and 2): Rats were euthanized 7 (Experiment 2) or 30 days (Experiment 1) after ICH with sodium pentobarbital (80 mg/kg) and were transcardially perfused with saline and then 10% neutral buffered formalin. Forty micron coronal sections were taken with a cryostat every 600 μm and stained with cresyl violet. Using Scion Image J 4.0 (Scion Corporation, Frederick, MD, USA) the volume of lesion plus atrophy (e.g., ventricular enlargement) was quantified and expressed as follows (MacLellan et al, 2004):
Volume of tissue lost =remaining volume of normal
hemisphere-remaining volume
of lesioned hemisphere:
Volume of a hemisphere =verage(area of the complete
coronal section of the hemisphere
area of ventricle-area of damage)
×interval between sections
number of sections analyzed
Inflammatory cell infiltration (Experiment 3): Rats in Experiment 3 were euthanized as described above 2 or 4 days after ICH. A 4-mm-thick section of tissue encompassing the lesion was taken from each brain and embedded in paraffin. Ten micron sections were taken with a microtome and stained with Perl's Prussian blue for ferric iron and Leder's stain (Leder, 1964) for chloracetate esterase activity (Kit 91-C, Sigma, Oakville, ONT, Canada). These particular stains have been used to quantify components of inflammation after ICH in several other studies (Del Bigio et al, 1996; Peeling et al, 2001). In consecutive sections, we counted the total number of activated macrophages (stained blue) and viable neutrophils (stained red) in the damaged hemisphere at the level of maximum hematoma diameter.
Spectrophotometric assay of hemoglobin (Experiment 4): Rats in Experiment 4 were subjected to an ICH and then 24 h later were overdosed with sodium pentobarbital and transcardially perfused with 100 mL of 0.9% saline. Brains were extracted, dissected free of olfactory bulbs and cerebellum, and homogenized (Bio-Spec, Racine, WI, USA) for 60 secs in a test tube containing distilled water (total volume of 3 mL). After centrifugation (15,800g for 30 mins; model CR20B2, Hitachi, Japan), Drabkin's reagent (400 μL; Sigma) was added to 100 μL aliquots of the supernatant (at least four samples per brain) and allowed to react for 15 mins. The absorbance of this solution was read using a spectrophotometer (540 nm; model DU-8; Beckman Coulter Ltd, London, UK) and the amount of blood in each brain was calculated using a curve generated previously using known blood volumes (MacLellan et al, 2004).
Statistical Analysis
Data are expressed as the mean±s.d. Data were analyzed using a one-way ANOVA or with t-tests (two-tailed) that did not assume equality of variance in the case of a significant Levene's test (SPSS, v. 11.5). The Kruskal-Wallis test was used for heterogeneous data (e.g., ipsilateral limb error rate in the ladder task) and nonparametric data (e.g., NDS).
RESULTS
Experiment 1
Baseline core temperature recorded before surgery was similar among groups (overall mean = 37.0°C±0.3°C; data not shown), and was regulated as desired after ICH (Figure 1A). On the first day after ICH, the NORMO group was slightly (0.4°C) but significantly warmer than baseline temperature (P<0.001; Figure 1B). Temperature was forcibly elevated (versus baseline) in the HYP-1, HYP-2, and HYP-3 groups by 1.4°C, 1.4°C, and 2.9°C, respectively (average of hyperthermia period). One HYP-3 rat died one day after ICH of unknown cause.
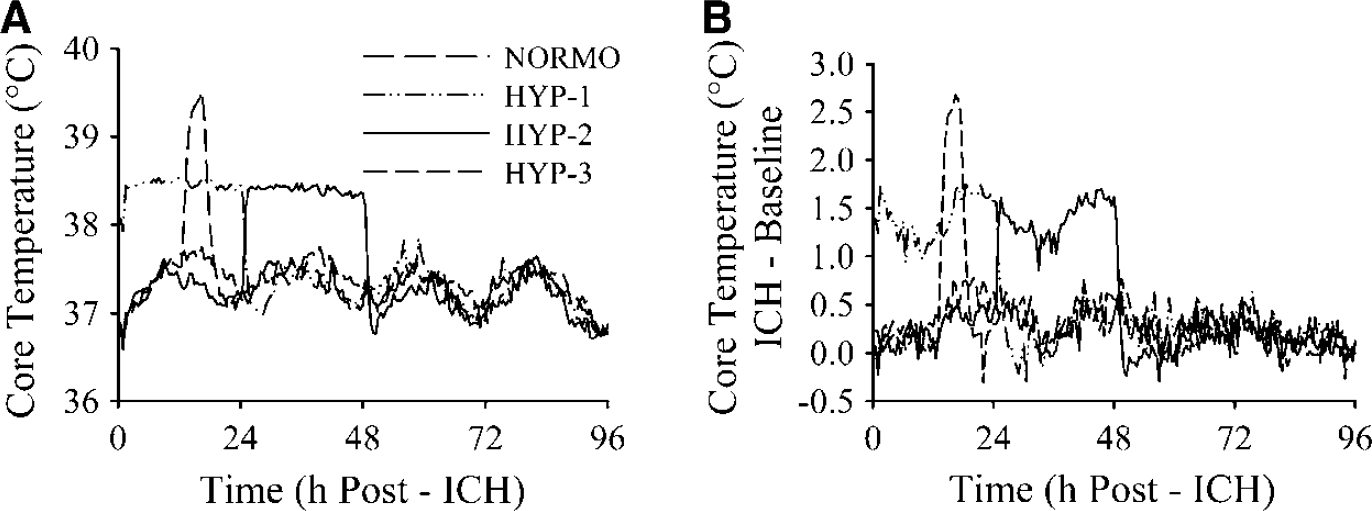
Core temperature (°C) averaged every 30 mins (sampled every 30 secs) for 4 days after ICH (
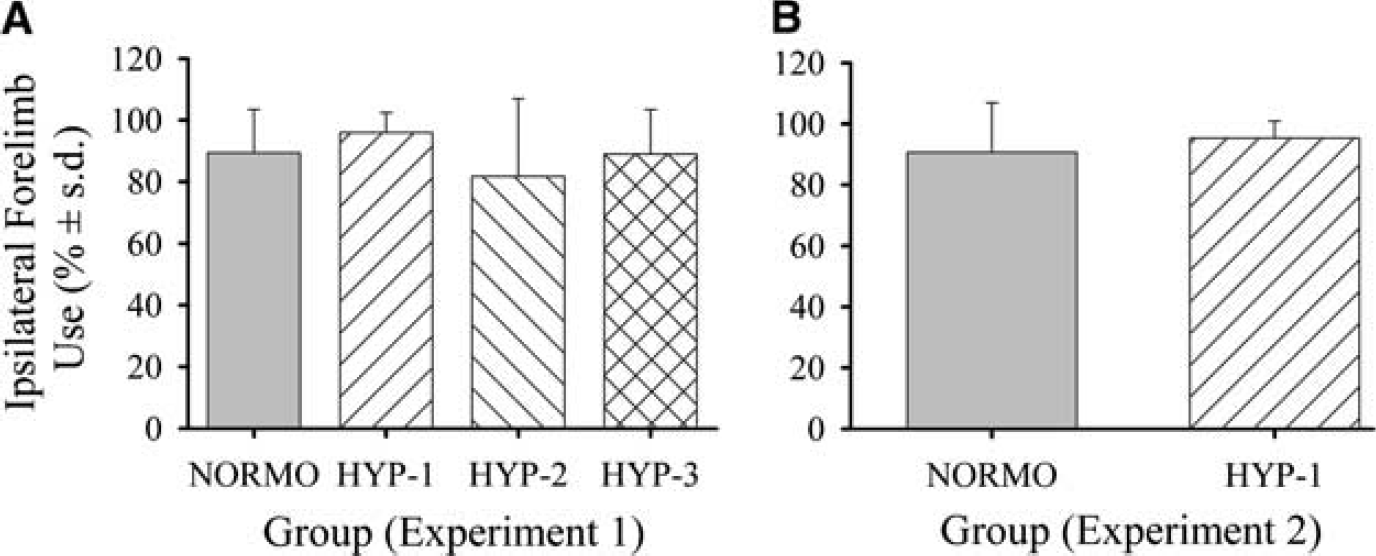
Rats performed similarly during staircase training (data not shown). Intracerebral hemorrhage (NORMO) caused significant reaching impairments with the contralateral (31.8% of baseline; P<0.001 versus baseline) and ipsilateral (78.5% of baseline; P = 0.019) forelimbs during testing (days 24 to 28 post-ICH). The hyperthermia treatments did not affect reaching success with either forelimb (P≥0.151; Figure 2). Untreated ICH caused significant impairments with the contralateral limbs (P<0.001 versus ipsilateral limb) in the ladder-walking test. NORMO rats had a mean slip rate of 19.6%±10.3% with the contralateral forelimb (Figure 3A). Performance was similar (mean of 18.6%±11.7%) with the contralateral hind limb. Error rates were small with the ipsilateral forelimb (overall mean = 0.8%±0.6%) and hind limb (overall mean = 1.7%±0.9%). The hyperthermia treatments did not significantly affect error rate of any limb (P≥0.156). Similarly, there was no main effect of group in the limb-use asymmetry task (P = 0.114; Figure 4A). After ICH, rats displayed a substantial asymmetry favouring use of the ipsilateral limb (overall mean = 89.5%±14.1%). Finally, all groups exhibited an asymmetry in forepaw inhibition in the swim test after ICH (overall mean = 4.8±5.1 strokes; P≤0.018 versus baseline performance), however, there was no difference among groups (P = 0.293).
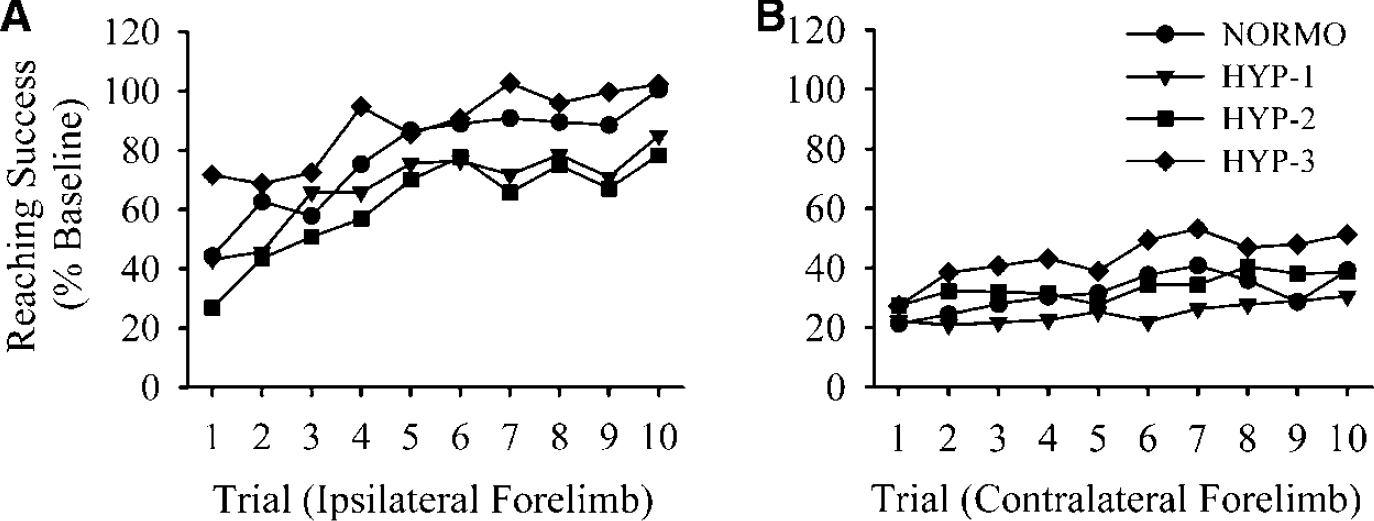
Reaching success (% baseline) with the ipsilateral (
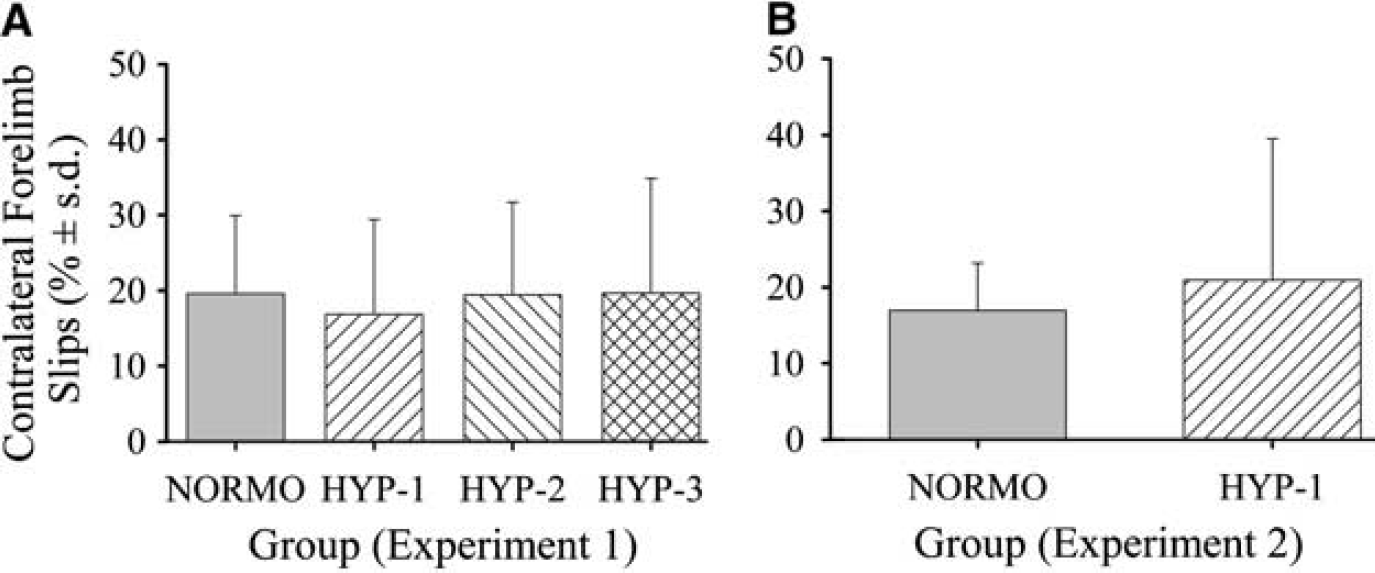
Error rate (% falls through bars±s.d.) made with the contralateral forelimb on the horizontal ladder 30 days (
Spontaneous ipsilateral forelimb use (% independent limb use±s.d.) in the cylinder test 30 days (
Volume (mm3±s.d.) of tissue lost 30 days (
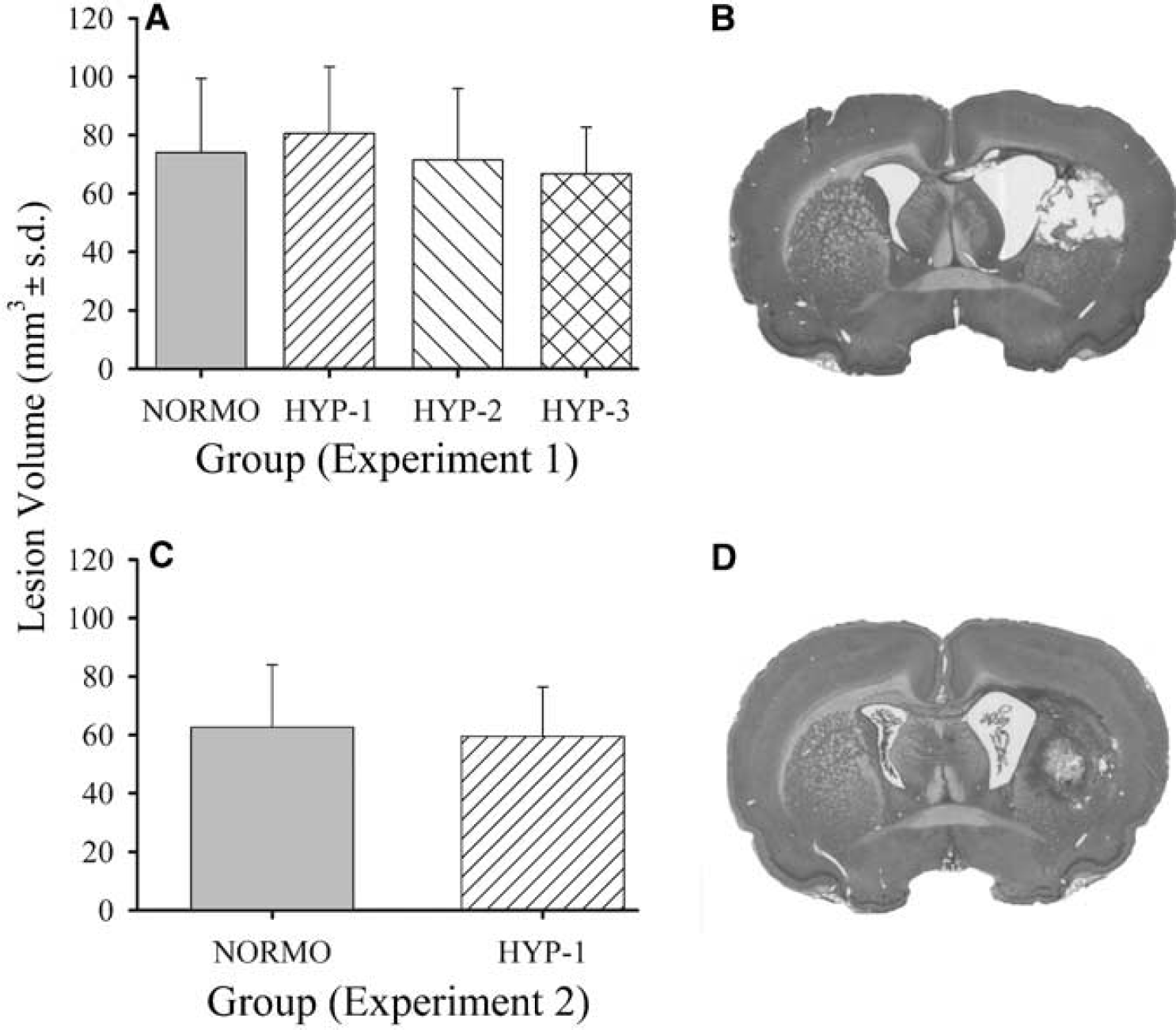
Intracerebral hemorrhage resulted in damage primarily to the striatum and also to the internal capsule, corpus callosum, globus pallidus, and thalamus. Tissue lost 30 days after ICH was not significantly different among groups (overall mean = 73.3±22.4 mm3; P = 0.356; Figures 5A and 5B).
Experiment 2
Temperature of NORMO and HYP-1 groups was regulated according to Experiment 1 (data not shown). Seven days after ICH, NORMO rats had a contralateral forelimb error rate of 17.0%±6.2% in the ladder test (Figure 3B). Performance was similar (20.9%±18.6%) with the contralateral forelimb in the HYP-1 group. Error rates of each limb were similar between groups (P≥0.285). One HYP-1 rat was excluded from this task for failure to cross the ladder. Both the NORMO (90.7%±16.2%) and HYP-1 (95.3%±5.6%) groups displayed increased usage of their ipsilateral forelimb in the limb-use asymmetry task, but were not significantly different from each other (P = 0.493; Figure 4B). One HYP-1 and two NORMO rats did not explore the cylinder wall and were thus excluded from analysis. The NORMO group sustained a 62.6±21.3 mm3 lesion by 7 days post-ICH (Figures 5C and 5D). The HYP-1 treatment did not increase the volume of tissue lost (59.5±17.0 mm3; P = 0.752).
Experiment 3
Temperature was regulated as described above (data not shown). Rats exhibited significant neurologic deficits 1 to 4 days after ICH (P<0.001 versus baseline; data not shown). The HYP-1 and HYP-3 conditions did not affect neurological deficit scores on any day (P≥0.253; Figure 6). The number of macrophages observed in the injured hemisphere was not significantly different among groups at either 2 (P = 0.798) or 4 days (P = 0.489) after ICH (Figure 7). Similarly, hyperthermia did not significantly affect the number of neutrophils present at 2 (overall mean 951.00±552.45 neutrophils; P = 0.845) or 4 days (overall mean 1095.60±1052.20; P = 0.573). Four brains could not be evaluated because of poor tissue quality.
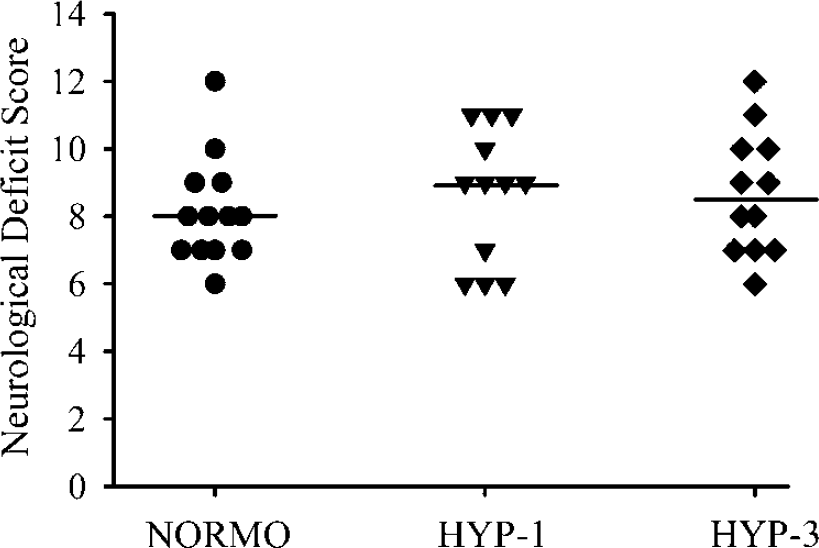
Neurologic deficit scores (bar denotes group median) at 2 days after ICH (Experiment 3). Data were similar on other days (data not shown). Hyperthermia conditions did not affect neurologic deficits at any time.
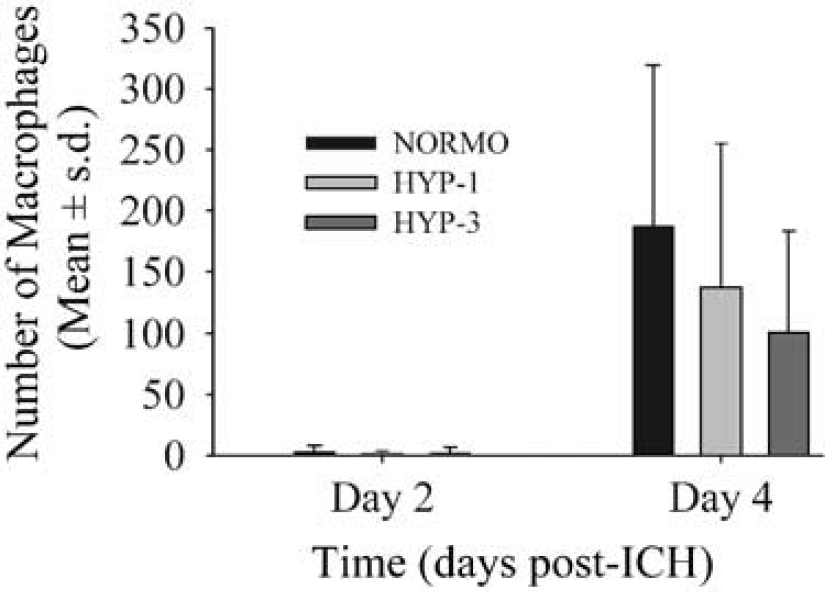
Number of activated macrophages (mean±s.d.; Experiment 3) in the lesioned hemisphere 2 and 4 days after ICH. There were no group differences at either time.
Experiment 4
Post-ICH temperature was regulated as in Experiment 1 (data not shown). The NORMO group had 56.5±34.1 μL of blood in the brain 24 h after ICH. This was not significantly different (P = 0.587) in the HYP-1 (39.2±18.6 μL) or HYP-3 groups (42.9±47.8 μL). One HYP-3 rat died in this experiment and was not included in the analysis.
DISCUSSION
This is the first study to assess the effects of mild to moderate hyperthermia on outcome after experimental ICH. Contrary to our expectations, hyperthermia did not aggravate bleeding, macrophage or neutrophil infiltration, and had no discernable effect on long-term histologic or functional outcome. These findings contrast with the overwhelming evidence that even mild elevations in temperature markedly worsen outcome after cerebral ischemia in humans and rodents. However, our findings are consistent with several clinical studies of ICH in which hyperthermia did not independently predict mortality (Boysen and Christensen, 2001; Szczudlik et al, 2002; Wang et al, 2000).
The differing effects of hyperthermia after ischemic and hemorrhagic stroke are likely due to fundamental differences in the underlying pathophysiology. For instance, it is the penumbra (mildly ischemic region) that is recruited by hyperthermia after focal ischemia (Reglodi et al, 2000). Notably, experimental (Patel et al, 1999) and clinical ICH studies (Qureshi et al, 1999) fail to find a significant penumbral region surrounding the hematoma. As well, the majority of hemorrhagic injury occurs rapidly and is caused by direct tissue destruction and space-occupying effects of the hematoma. Thus, mechanisms thought to aggravate ischemic injury (e.g., increased inflammation) may not substantially worsen outcome after ICH. Indeed, many antiinflammatory agents fail to lesson lesion size after ICH (Mayne et al, 2001; Peeling et al, 2001). Additionally, hyperthermia aggravates ischemic injury through increased excitotoxicity (Ginsberg and Busto, 1998); yet the role of the less dramatic increases in excitatory amino acids found soon after ICH has yet to be defined (Qureshi et al, 2003). Accordingly, we expect that mild and moderate hyperthermia, under the conditions tested, do not substantially and directly aggravate key mechanisms contributing to injury after ICH (e.g., bleeding, mechanical destruction). Furthermore, as the insult was not worsened (i.e., volume of injury did not change), one would not expect a concomitant increase in inflammation. Perhaps mild hyperthermia does not directly aggravate inflammation after focal cerebral ischemia. Instead, an enlarged lesion (e.g., because of enhanced excitotoxicity) may result in greater inflammation, which may then act to further worsen outcome.
Hyperthermia may aggravate mild hemorrhagic insults, whereas more severe insults may not be noticeably altered. Although the insult produced in this study is large, we have previously created larger lesions (e.g., greater rostral—caudal extension of damage) with more severe impairments (e.g., in skilled reaching) and greater mortality (unpublished data). Thus, injury and mortality could have been increased in this study. Hyperthermia may have also affected other mechanisms that we did not assess, or perhaps there was an effect that would have been detected with other functional tests, histologic measures, or additional survival times. We evaluated functional outcome using five tests sensitive to hemorrhagic injury (DeBow et al, 2003; MacLellan et al, 2004; Peeling et al, 2001) and measured the volume of tissue lost at a short-term (e.g., 7 day) and long-term (e.g., 30 day) survival. Furthermore, we assessed the effects of hyperthermia on mechanisms such as bleeding and inflammation, which contribute to hemorrhagic injury. The clear lack of effect suggests that mild to moderate hyperthermia does not worsen histologic or functional outcome, and other mechanisms or undetected effects likely do not contribute significantly to injury.
Near-simultaneous measurement of core and brain temperature after ICH in a pilot study showed that brain temperature was slightly cooler (by <1°C) than body temperature, as previously shown using this method in normal rats (DeBow and Colbourne, 2003). In accordance with previous work from this (Plahta et al, 2004) and other labs (Kim et al, 1996), we found that increasing body temperature disrupts the normally good relationship between body and brain temperature. Indeed, brain temperature was relatively overheated (versus core temperature) when body temperature was elevated via infrared lamps. Further study is needed to determine the relationship between body and brain temperature when other methods of temperature measurement (e.g., rectal or temporalis muscle) and regulation (e.g., using a heating pad) are used. For example, perhaps an even greater overheating of the brain occurred in ischemic studies that used rectal temperature measurements or alternative heating methods.
Our previous work showed that delayed hypothermia reduces the volume of tissue lost and improves functional outcome in this model of ICH (MacLellan et al, 2004). However, hypothermia induced soon after ICH increases the volume of extravasated blood because of adverse physiologic effects of the treatment (e.g., elevated blood pressure). Additionally, we recently showed that 17-β estradiol attenuates bleeding in this model and thereby lessens injury (Auriat et al, 2005). Accordingly, because injury and bleeding can be improved or worsened, it is reasonable to expect that hyperthermia could have affected outcome in this model of ICH. Differences in experimental design may explain why hypothermia affected outcome whereas hyperthermia did not. Notably, the magnitude of the enforced temperature change was not equivalent in these studies. Temperature was reduced by up to 4°C (to 33°C) during prolonged hypothermia (MacLellan et al, 2004) whereas temperature was persistently elevated by 1.5°C in the HYP-1 and HYP-2 conditions in the present study. Second, the duration of temperature regulation was not the same in these studies. Thus, greater effects may have been detected had we kept rats at greater hyperthermic levels or for more protracted periods. It should be noted, however, that infarct volume and neurologic deficits are increased when brain temperature is elevated to 40°C for 3 h starting 24 h after focal ischemia in rats (Kim et al, 1996). Our similar HYP-3 treatment was induced sooner after ICH, yet had no discernable effect on outcome. Finally, in our opinion, prolonged mild hypothermia provides substantially less benefit after ICH (MacLellan et al, 2004) than after global (e.g., Colbourne et al, 1999) and focal (e.g., Colbourne et al, 2000) cerebral ischemia. Hemorrhagic injury simply appears to be less sensitive to temperature manipulations, which is in agreement with the majority of clinical studies on hyperthermia after ICH.
Future studies should evaluate alternate causes (e.g., infection) and complications (e.g., cardiovascular instability) of hyperthermia as well as its effects on other hemorrhagic events such as hemorrhagic transformation after ischemic stroke and traumatic brain injury (e.g., see Kinoshita et al, 2002). Furthermore, hyperthermia may differentially affect male and female rodents as gender (Nakamura et al, 2004) and estrogen (Auriat et al, 2005) affects ICH insults. Such experimental studies are needed in conjunction with clinical findings to clarify the independent significance of hyperthermia on mortality and functional outcome after ICH.
In summary, mild to moderate increases in body temperature did not worsen brain injury, bleeding, inflammation, or functional deficits after ICH in rats. These results are consistent with clinical findings that hyperthermia is not an independent prognostic factor in ICH patients. The failure of hyperthermia to critically affect outcome in this study suggests that treating mild to moderate hyperthermia may provide no cytoprotective benefit. However, treating the causes of fever (e.g., infection) or life-threatening fever is recommended.
Footnotes
Acknowledgements
The authors thank D. Bruins, L. Davies, J. Feniak, H. Huynh, S. Kirkland, B. Rollins, R. Yan, and T. Yokota for technical assistance, and Drs. C. Sturdy and D. Wong-Wylie for use of equipment.
