Abstract
Few effective treatment options exist for stroke beyond the hyperacute period. Radical generation and myeloperoxidase (MPO) have been implicated in stroke. We investigated whether pharmacologic reduction or gene deletion of this highly oxidative enzyme reduces infarct propagation and improves outcome in the transient middle cerebral artery occlusion mouse model (MCAO). Mice were treated with 4-aminobenzoic acid hydrazide (ABAH), a specific irreversible MPO inhibitor. Three treatment regimens were used: (1) daily throughout the 21-day observational period, (2) during the acute stage (first 24 hours), or (3) during the subacute stage (daily starting on day 2). We found elevated MPO activity in ipsilateral brain 3 to 21 days after ischemia. 4-Aminobenzoic acid hydrazide reduced enzyme activity by 30% to 40% and final lesion volume by 60% (P<0.01). The MPO-knockout (KO) mice subjected to MCAO also showed a similar reduction in the final lesion volume (P<0.01). The ABAH treatment or MPO-KO mice also improved neurobehavioral outcome (P<0.001) and survival (P=0.01), but ABAH had no additional beneficial effects in MPO-KO mice, confirming specificity of ABAH. Interestingly, inhibiting MPO activity during the subacute stage recapitulated most of the therapeutic benefit of continuous MPO inhibition, suggesting that MPO-targeted therapies could be useful when given after 24 hours of stroke onset.
Introduction
Stroke is the fourth most common cause of mortality and a leading cause of long-term disability in the United States. 1 Inflammation has been shown to correlate with adverse stroke outcomes in animal models and human studies,2, 3 and has been implicated both in propagation of tissue damage and in postischemic tissue repair. 4
Myeloperoxidase (MPO) is highly expressed and secreted by neutrophils, monocytes, activated macrophages, and brain microglia in response to injury and inflammation. Elevated MPO activity concomitant with leukocyte influx into the ischemic brain has been found in animal models of stroke.5, 6 As such, MPO has been used as an inflammatory marker for stroke.7, 8 Interestingly, leukocyte depletion in stroke markedly decreased MPO activity and resulted in smaller infarct size, 5 suggesting that MPO may not just be a bystander. Myeloperoxidase interacts with hydrogen peroxide to generate highly reactive species including hypochlorite and radicalized oxygen species, and its activity can promote apoptosis and nitrotyrosination of proteins.9, 10 Thus, increased MPO activity may contribute to infarct progression through protein alterations and oxygen radical generation that leads to cell death. In humans, serum levels of MPO are elevated after stroke 11 and these levels reportedly correlate with infarct volume 8 , particularly for certain MPO genotypes. 12 Additionally, MPO has also been implicated in other cardiovascular diseases in humans, including myocardial infarction, 13 congestive heart failure, 14 and peripheral vascular disease. 15
Despite evidence supporting a deleterious effect of inflammation and associated elevation of MPO levels in stroke,2, 7, 8, 12, 16, 17 it is not yet clear whether MPO participates in infarct propagation or serves only as a marker for inflammation. Indeed, one investigation reported that infarct volume is actually larger in MPO-knockout (KO) mice after transient cerebral ischemia based on the area of infarction and edema within 24 hours after infarct induction. 18 Because MPO activity peaks later, around 3 to 7 days after stroke, 19 we tested whether reduced MPO activity corresponds to a reduction in infarct size when examined over longer time periods, and whether MPO inhibition or deficiency would be beneficial for ischemic stroke, particularly beyond the acute stage after 24 hours when MPO and inflammation peak. 19
Materials and methods
Animal Model of Stroke
The protocol for the animal experiments was approved by the Massachusetts General Hospital Subcommittee on Research Animal Care and conducted according to National Institute of Health guidelines. In all, 8- to 11-week-old male animals were purchased from the Jackson Laboratory (Bar Harbor, ME, USA) and housed in a facility approved by the Association for Assessment and Accreditation of Laboratory Animal Care International. Experiments were performed in a total of 103 male mice (Figure 1). Among these, right-sided cerebral ischemia was induced in 72 C57B/L6 wild-type mice and 17 MPO-KO mice (13th generation backcross on the C57B/L6 background, The Jackson Laboratory) by occluding the right middle cerebral artery temporarily for 30 minutes using a thread occlusion model, followed by reperfusion, as previously described. 19 An additional 14 C57B/L6 wild-type mice underwent the surgical procedure without actual occlusion of the middle cerebral artery (sham mice). None of the mice developed gross intracranial hemorrhage either on imaging or at killing. Please refer to Figure 1 for distribution and numbers of animal in different treatment and control groups.
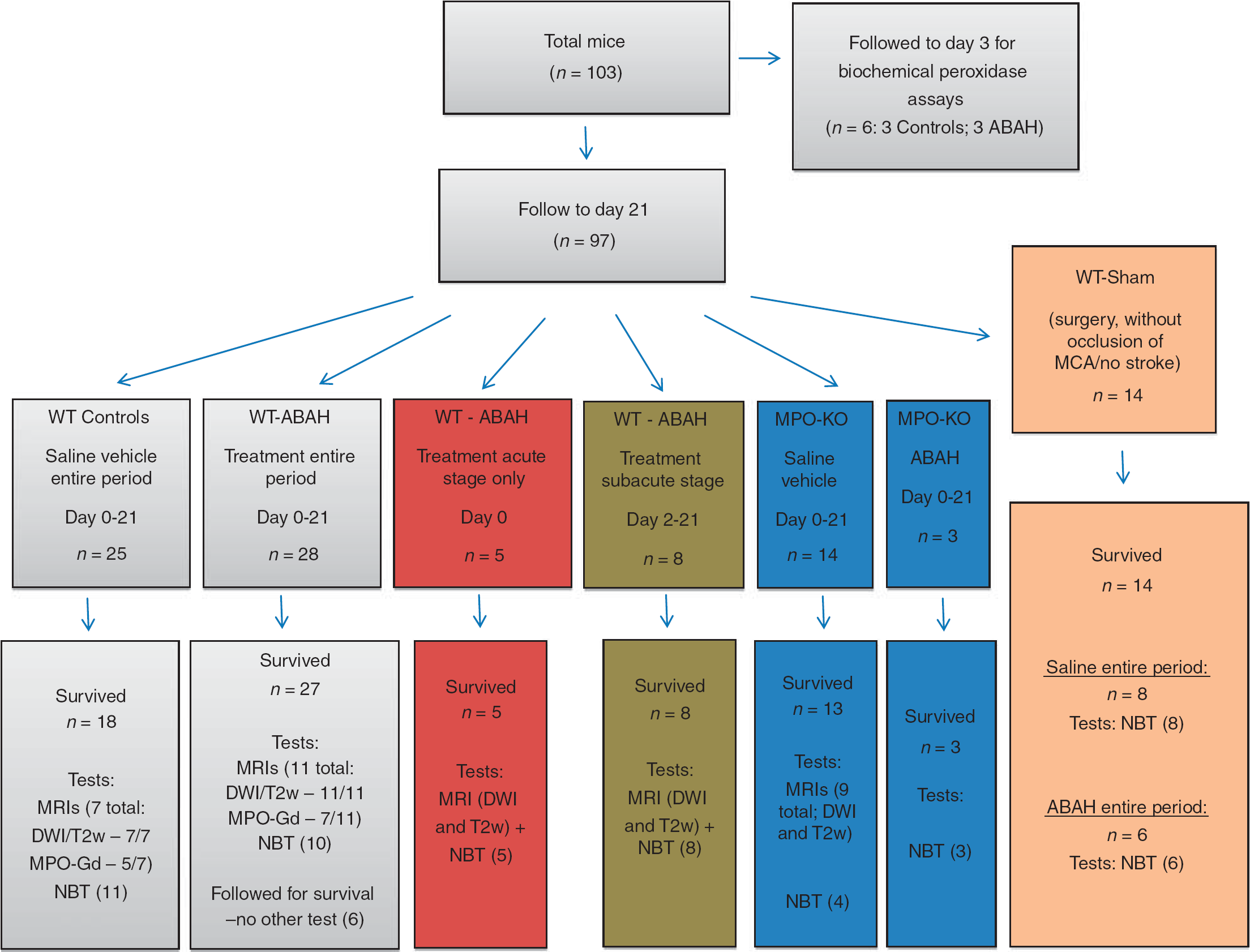
Figure summarizing the number of animals in different treatment groups and experiments performed. A subset of animals underwent specific tests such as MRI, NBT, or both. The number of animals in each group that underwent tests is provided in the bottom row, in brackets, next to the test(s) performed. ABAH, 4-aminobenzoic acid hydrazide; DWI, diffusion-weighted images (MRI); MPO, myeloperoxidase; MPO-Gd, activatable MPO specific MRI contrast agent; MPO-KO, MPO-knockout mice; MRI, magnetic resonance imaging; NBT, neurobehavior testing; T2w, T2-weighted images (MRI).
Treatment Protocol and Myeloperoxidase Inhibition
Wild-type C57B/L6 mice were randomly selected for treatment with either 400 μL of saline vehicle (controls) or 400 μL of the specific irreversible MPO inhibitor 4-aminobenzoic acid hydrazide (ABAH) 20 at a dose of 40 mg/kg, twice daily, injected intraperitoneally. Three MPO-KO mice were also treated with ABAH as an additional control (Figure 1). This dose of ABAH has been shown to result in substantial inhibition of MPO activity in vivo in a murine model of demyelinating disease. 21 4-Aminobenzoic acid hydrazide appears to be able to cross the intact blood—brain barrier (BBB) (Supplementary Figure S1), and passage into the central nervous system would be expected to be further facilitated during the subacute stage of infarction at which time there can be significant BBB breakdown. Three treatment regimens were used: (1) daily injections throughout the entire observational period (21 days) beginning within half an hour after completion of stroke induction, (2) two injections during the first day (day 0), administered 30 minutes after stroke induction followed by another injection 8 hours later (acute stage), and (3) daily injections starting on day 2 and continuing to day 21 after infarct induction (subacute stage). To ensure that the injection procedure and schedule is identical and controlled for in all groups, in groups 2 and 3, saline vehicle was injected during days when ABAH was not administered. In addition to these groups, in a fourth group, ABAH was administered to mice from day 0 to day 3 after infarct induction at which time they were killed and the brain used for biochemical peroxidase assays. Administration of ABAH does not significantly alter physiologic parameters (temperature, heart rate, and respiratory rate) of the mouse (Supplementary Figure S2). The mice were assessed daily and tolerated the injections well. In neither group was there any evidence of associated inflammation or infection at the injection sites. To control for any potential unexpected effects of the injection procedure, all animals received a similar injection regimen, either ABAH or the saline vehicle as a control, including sham animals.
Conventional Brain Magnetic Resonance Imagings and Determination of Lesion Volumes
A subset of animals underwent magnetic resonance (MR) imaging (MRI) of the brain (Figure 1). Magnetic resonance imaging was performed up to 21 days after stroke induction, including days 1, 3, 7, 14, and 21, on a 4.7-T animal MRI scanner (Bruker, Billerica, MA, USA) with a dedicated mouse head coil. Coronal diffusion-weighted images (DWI) were obtained on days 1 and 3 and coronal T1w and T2w images were obtained at all time points. The scans in a subset of animals also included MPO-Gd molecular imaging as described in the next section. Mice were anesthesized using isoflurane gas (0.5% to 2.5%) which was adjusted throughout the imaging process to keep a consistent respiratory rate while being monitored by the Model 1025 MRI compatible small animal monitoring and gating system (SA Instruments, Stony Brook, NY, USA). Rectal temperature was also monitored and maintained at 37°C by adjusting an MR-compatible small animal heating system (SA Instruments) throughout the imaging procedure. After scanning, the mice were placed under oxygen and heated using the Air-Therm ATX (World Precision Instruments, Sarasota, FL, USA) until they were conscious. Image analysis and segmentation was performed using the OsiriX DICOM viewer (http://www.osirix-viewer.com/). Total lesion volume was calculated by multiplying the sum total cross-sectional area of the lesion by slice thickness over the entire brain.
The evolution of cerebral infarction was assessed first on DWI/apparent diffusion coefficient (ADC) images on day 122, 23. Diffusion-weighted image is highly accurate in identifying the area of irreversible ischemia, shows good correlation with damage on histopathologic specimens, and is widely used in clinical practice to determine infarcted tissue area.24, 25 To determine the day 1 infarct volume, the area of restricted diffusion represented by the hypointense area on the ADC map on day 1 after stroke induction was segmented using the OsiriX DICOM viewer and used to calculate the volume, as described above. Thereafter, at all other time points starting on day 3 after stroke induction, the infarcted area was determined based on abnormally hyperintense areas on T2w images representing infarcted tissue and vasogenic edema. Final lesion volume was determined by segmentation of the residual abnormal hyperintense tissue on T2w images on day 21, after resolution of edema, representing gliosis and infarcted tissue. To correct for potential variations in the initial ictus, the normalized final volume was also calculated by dividing the day 21 volume in each mouse by the volume on the ADC map on day 1 in that mouse, expressed as a percentage. The images were evaluated by three authors independently and blinded to the groups.
Molecular Magnetic Resonance Imaging of Neuroinflammation with MPO-Gd
This is described in greater detail in Supplementary methods and in published studies.19, 21, 26, 27 Briefly, in a subset of animals imaged (five saline-treated control mice and seven ABAH-treated mice), MR molecular imaging was performed using the activatable MR contrast agent bis-5HT-DTPA-Gd (MPO-Gd) up to day 21, a sensitive and specific contrast agent for evaluation of MPO activity in vivo. After the intravenous administration of 0.3 mmol/kg of MPO-Gd, coronal T1w images were obtained at 6, 15, 30, 45, 60, and 75 minutes sequentially. Because early enhancement (6 minutes after contrast agent injection) represents mostly leakage through BBB breakdown, whereas delayed enhancement is derived mostly from agent retention caused by MPO activation,19, 21, 27in vivo MPO activity was determined semiquantitatively by calculating the ‘activation ratio’, 19 by dividing the contrast-to-noise ratios (CNRs) of lesions on delayed (60 minutes) images by the early (6 minutes) postcontrast images. The CNR at each time point was calculated using the formula SI(lesion)−SI(normal brain)/stdev noise, where SI(lesion) is the signal intensity of the lesion, SI(normal brain) is the signal intensity with the region of interest placed in the contralateral normal appearing brain parenchyma, and stdev noise is the standard deviation of noise obtained by placing the region of interest outside the mouse cranium in an empty area within the field of view.
Peroxidase Activity Assays
Peroxidase activity assays were performed on the extracellular and intracellular brain fractions prepared from fresh perfused brain samples as described in published studies21, 28 and as described in greater detail in Supplementary methods. A total of six mice (three controls and three ABAH) were evaluated on day 3 after stroke induction. The activity for each sample reaction was calculated and normalized by dividing by the day 1 infarct volume based on the areas of restricted diffusion on MRIs performed for each mouse on day 1 after stroke.
Neurobehavioral Testing
Neurobehavioral tests were performed by an investigator who was blinded to the experimental groups on three cohorts of wild-type mice, consisting of sham (n=8; receiving saline vehicle), saline vehicle-treated stroke (n=11), and ABAH-treated stroke (n=10) mice on days 3, 7, and 21 after infarction (Figure 1), as described in published studies. 29 Similar testing was also performed in six additional sham mice treated with ABAH as an additional control. Furthermore, testing was performed in four MPO-KO mice (receiving saline vehicle) and three MPO-KO mice receiving ABAH.
Neurobehavioral tests were also performed and compared in mice treated only during the acute stage (n=5) or subacute stage (n=8). The sham mice underwent the surgical procedure without actual occlusion of the middle cerebral artery. Neurologic severity scores were determined by a number of tests to assess posture, position, and movement. Animals were suspended vertically by the tail and flexion of forelimb, head movement >10° to vertical axis, and circling toward paralytic side were assessed. Three tests were performed in an open field to assess abnormal gait, circling toward the paralytic side, and frequency of falling over; pinna reflex (a head shake upon touching the auditory meatus) and visual placement test (stretching of forelimbs to meet an approaching object) were also evaluated. Each test was scored as 1 for normal and 0 for abnormal, yielding a summed injury score from 0 to 8 with a maximal deficit score of 0. 29 Mice were pretrained 1 day before surgery and mice with seizures were excluded from the analysis.
Statistical Analysis
Results were reported as mean±standard error of measurement. For groups with a Gaussian distribution (n≥6), means were compared using the t-test (one-tailed for comparison of MPO activity after addition of the inhibitor ABAH; two-tailed for all other outcome measures, including final infarct volume). For groups with non-Gaussian distributions (n<6), comparison was also made with the nonparametric Mann—Whitney test. For simultaneous comparison of more than one group, a one-way analysis of variance (ANOVA) test was used with post hoc analysis for pairwise comparison of the subgroups. Two-way ANOVA test with multiple comparisons was performed for Figure 6A. For survival analysis, data were pooled from all mice assigned to the saline vehicle group (controls; n=25), mice assigned to the ABAH treatment group (n=28), and MPO-deficient mice (n=10). The analysis of the survival distribution was performed in each group using the log-rank (Mantel-Cox) test. A P value of ≤ 0.05 was considered to be statistically significant. When not provided in the manuscript text, the t-statistic, degrees of freedom, and F-statistic where applicable are provided in Supplementary materials. The number of mice used for each group was calculated to achieve a power of 90% (30% difference in mean and standard deviation of 15%, resulting in n=3 per group). We used Graphpad Prism version 6.00 for statistical analysis (GraphPad Software, La Jolla, CA, USA, www.graphpad.com).
Results
Stroke Induction and Temporal Evolution of Infarct and Associated Neuroinflammation
A subset of mice were serially imaged on days 1, 3, 7, 14, and 21 after ischemia-reperfusion to evaluate infarct and the evolution of MPO activity (Figures 2 and 3). Consistent with a previous published study, 19 MPO activity and neuroinflammation within the infarct bed peaked between 3 and 7 days after middle cerebral artery occlusion (MCAO) (Figure 3). The decline in MPO activity was much more gradual compared with the change in infarct/edema volume with diminished but persistent residual inflammation on day 21 (Figure 3A).
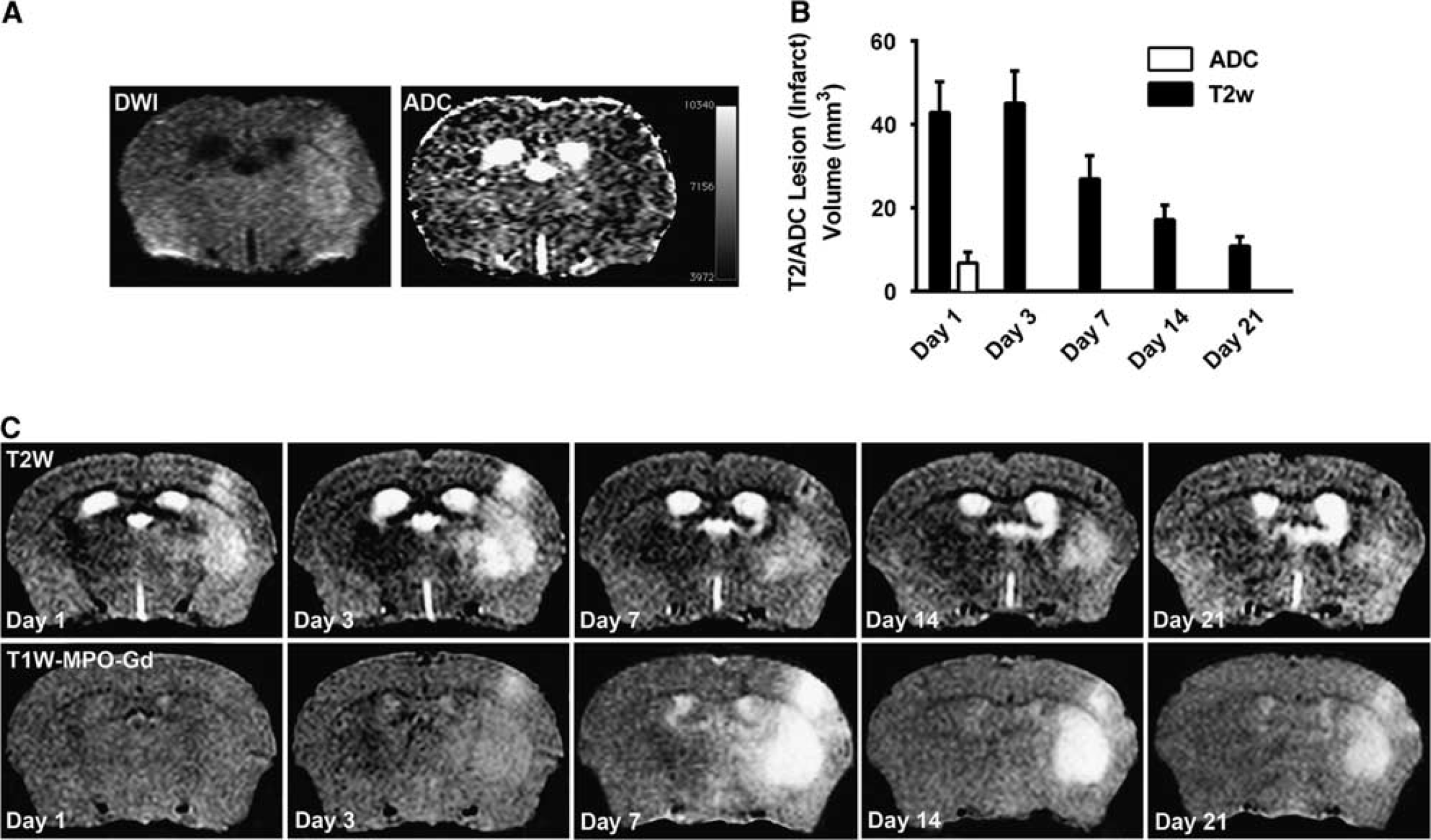
Temporal evolution of infarction and associated neuroinflammation using MPO-Gd molecular imaging. (
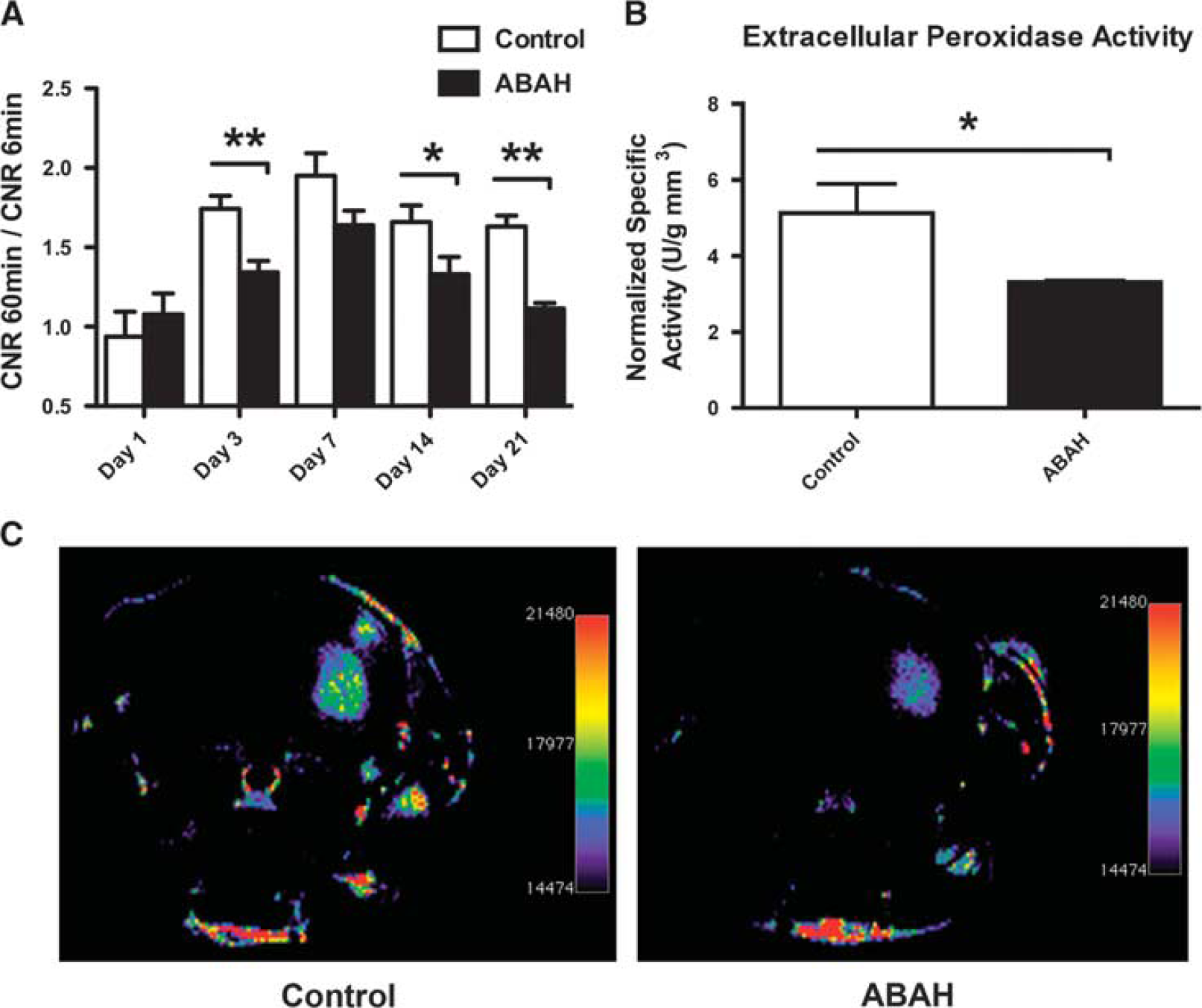
Partial inhibition of myeloperoxidase (MPO) activity after treatment of stroke mice with 4-aminobenzoic acid hydrazide (ABAH) beginning within half an hour after completion of stroke induction. (
4-Aminobenzoic Acid Hydrazide Results in Inhibition of Myeloperoxidase Activity In Vivo after Stroke Induction
4-Aminobenzoic acid hydrazide is a specific and irreversible inhibitor of MPO activity. 20 On day 1 after stroke induction, there was no significant MPO-specific enhancement and no significant difference between control and ABAH-treated mice (Figure 3). Similarly, no significant difference in infarct size was found at 1 day after stroke induction between the groups (Supplementary Figure S2). However, on days 3, 7, 14, and 21, there was reduction of MPO activity in the ABAH-treated mice based on the semiquantitative CNR analyses of approximately 30% (day 3, P=0.003, Mann—Whitney test), 19% (day 7, P=0.07), 25% (day 14, P=0.05), and 46% (day 21, P<0.001), (Figure 3).
Consistent with the imaging findings and our previous experiments demonstrating partial MPO inhibition with ABAH in vivo, 21 there was a significant decrease in peroxidase activity in the ABAH group (P=0.05; Mann—Whitney test) with approximately 35% decline in peroxidase activity, similar to the 30% inhibition found from MPO-Gd MRI.
Reduction of Myeloperoxidase Activity Results in a Decrease in Infarct Volume
We next evaluated whether MPO inhibition affects infarct volume. On days 3 to 7, during peak MPO activity, we found MPO inhibition with ABAH reduced T2 hyperintense lesions. For day 3, the reduction was 32% (45.1±7.7 mm3 in the saline control group compared with 30.5±9.9 mm3 in the ABAH treatment group, P=0.31; Figure 4A). On day 7, the reduction was similarly at 33% (27.0±5.5 mm3 in the control group compared with 18.0±7.1 mm3 in the ABAH-treated group, P=0.39; Figure 4B). Interestingly, at these early time points, there was a much larger reduction in the MPO-KO mice. On day 3, there was a reduction of 76% (10.6±3.2 mm3, P=0.0005; Figure 4A). On day 7, the reduction was 79% (5.6±1.8 mm3, P=0.0012; Figure 4B). On day 21, we found ABAH significantly decreased the final lesion volume (4.4±0.7 mm3 in the ABAH-treated group compared with 10.9±2.3 mm3 in controls, P=0.0051; Figure 5A). Similarly, there was also a significant decrease in final lesion volume in MPO-KO mice compared with controls (3.96±0.96 mm3, P=0.0085; Figure 5A). One-way ANOVA of all three groups with Tukey's post hoc analysis confirmed the statistical significance of these observations (P=0.0020, F (2,24)=8.2).
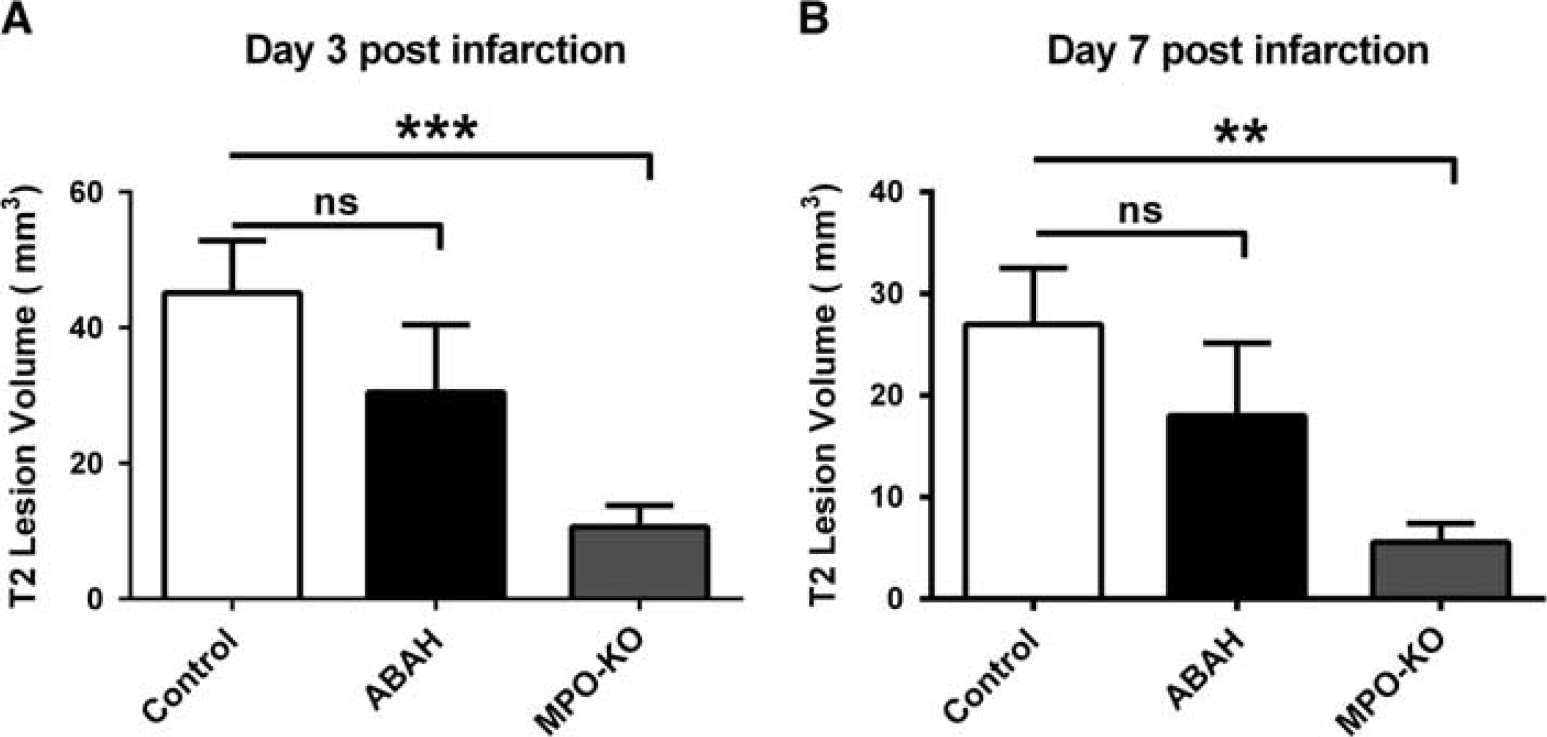
T2 lesion volumes on days 3 and 7 measured on T2w images. (
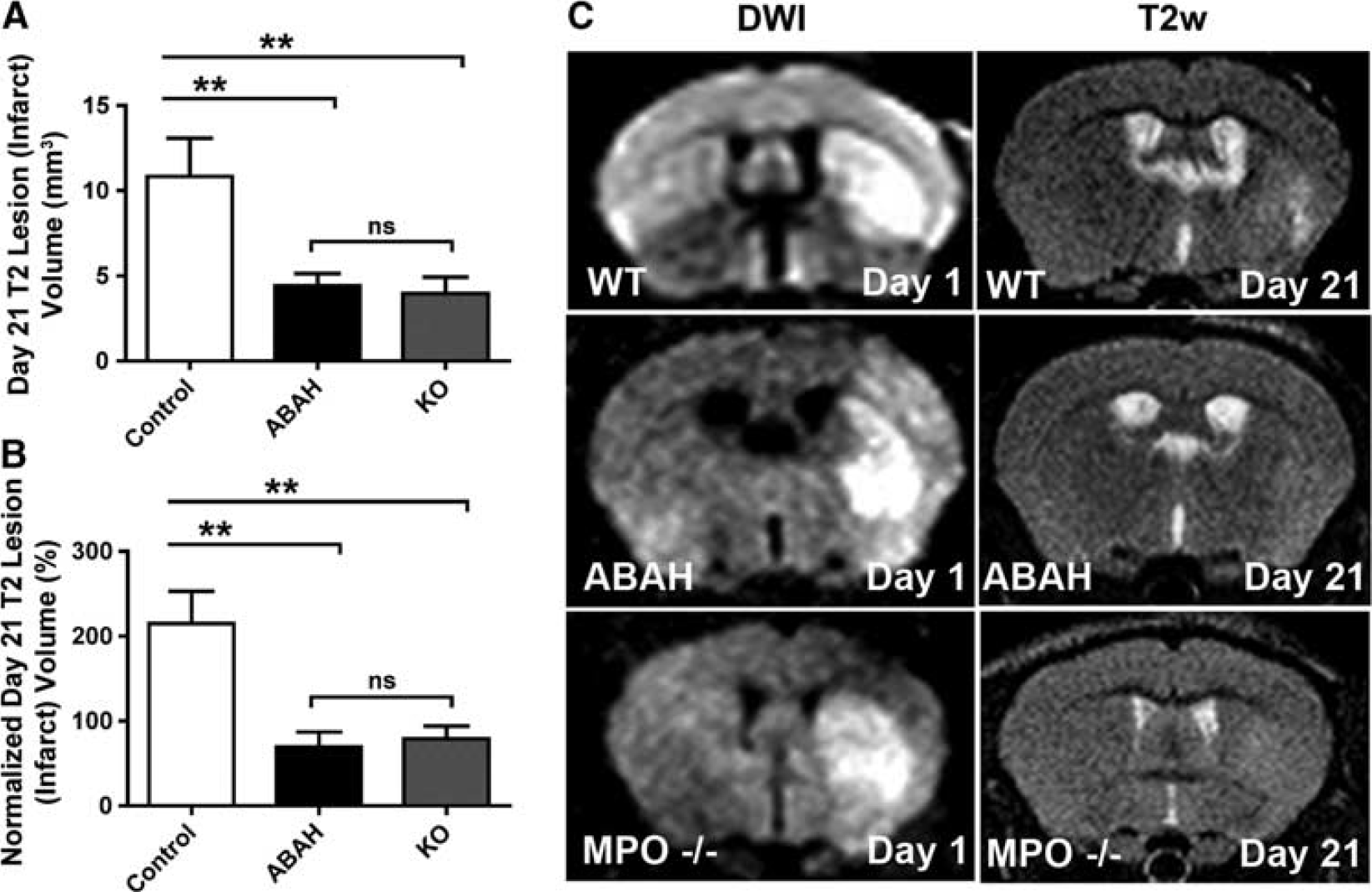
T2 lesion volume on day 21 is significantly reduced after inhibition of myeloperoxidase (MPO) activity for the entire 21-day period after infarction and in MPO-knockout (MPO-KO) mice. Comparison of both (
To ascertain that the observed outcomes were not secondary to differences in experimental variations in the infarct induction between groups, we calculated the difference in mean flow reduction during experimental stroke induction. We found no significant difference between the groups (Supplementary Figure S3a). The day 1 infarct volume was likewise not significantly different between the three groups (Supplementary Figure S3b). Finally, we also calculated the normalized day 21 lesion volume as a percentage of the day 1 infarct volume on the ADC map to account for any variations in the infarct induction. Consistent with the above observations, there was a significant decrease in the normalized day 21 lesion volume in the ABAH group compared with controls (P=0.0013; Figure 5B). There was likewise a significant decrease in normalized day 21 lesion volume in MPO-KO mice compared with controls (P=0.0027). Once again, ANOVA with Tukey's post hoc analysis confirmed these observations (P=0.0004, F (2,24)=11.13).
Inhibition of Myeloperoxidase Activity Results in Improvement in Clinical Outcome and Survival after Stroke
We next assessed the effects of reducing MPO activity on behavioral outcome in three cohorts of mice, consisting of sham, saline vehicle treated, and ABAH-treated mice. As expected, in stroke mice treated with vehicle, there was significant impairment (P<0.0001; Figure 6A). However, there was significant improvement in behavior outcome in the ABAH-treated mice, which had nearly normal functional outcome on day 21 with only a small residual deficit (P<0.0001; two-way ANOVA with multiple comparisons; Figure 6A). Similar improvement was seen in saline vehicle (P<0.0001) or ABAH-treated (P<0.0001) MPO-KO mice compared with wild-type, saline vehicle-treated controls (Figure 6A). Evaluation of interaction followed by multiple comparisons showed a strong statistically significant effect of ABAH in wild-type stroke mice but no significant effect on the MPO-KO background, further supporting the specificity of the intervention to MPO blockage.
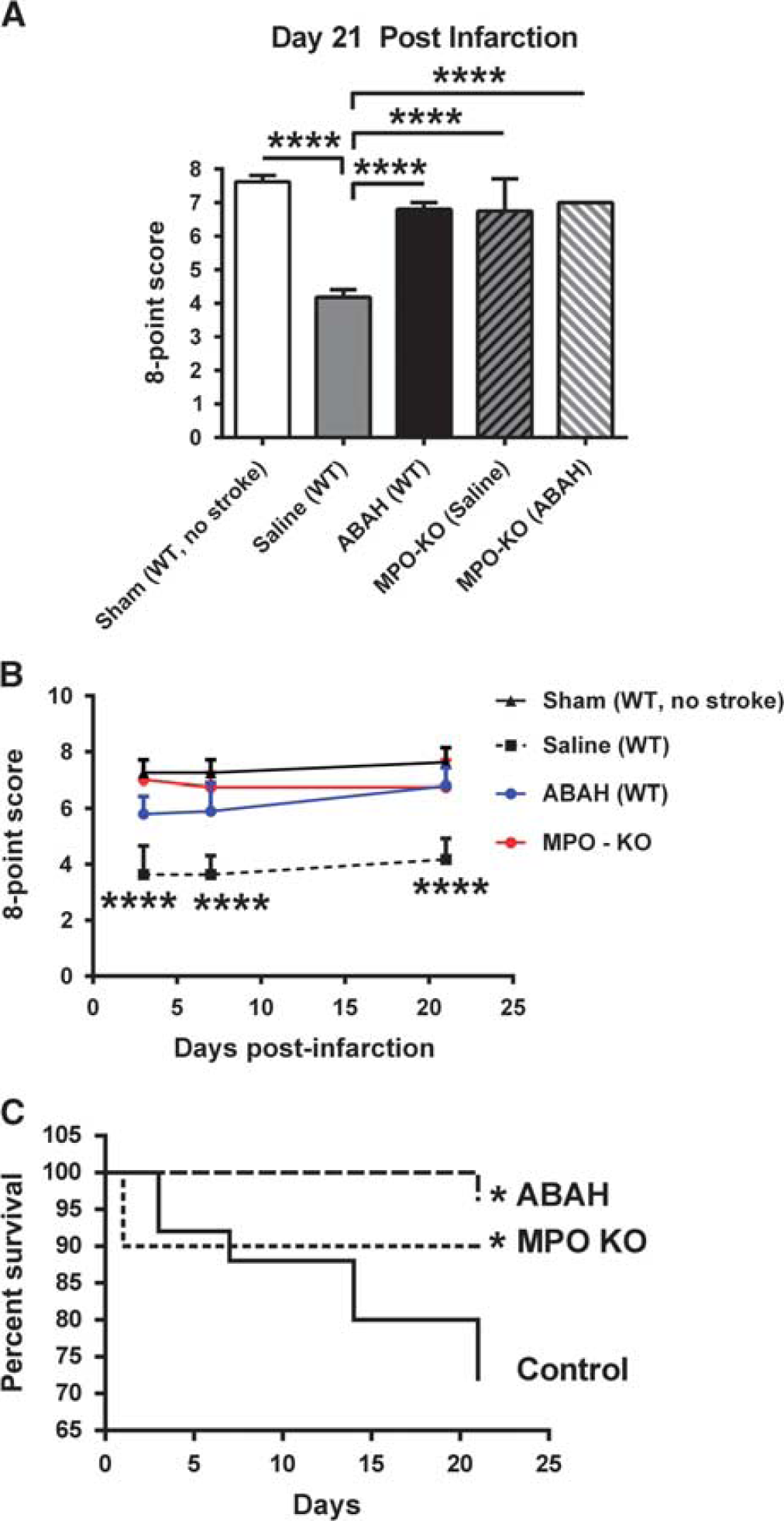
Neurobehavioral evaluation and survival analysis show beneficial effect of reduced myeloperoxidase (MPO) activity on stroke outcome. (
Furthermore, neurobehavioral improvement was evident at different time points in ABAH-treated group and MPO-KO mice compared with saline vehicle wild-type stroke mice, including days 3, 7, and 21 after infarction (P<0.0001; one-way ANOVA; Figure 6B). In addition to improved outcome on neurobehavioral testing, there was also an overall significant improvement in survival in the ABAH group compared with controls (P=0.01; Figure 6C). Of a total of 25 saline (control)-treated mice, only 18 survived, compared with 27 out of 28 ABAH-treated mice and 9 out of 10 MPO-KO mice.
Myeloperoxidase Inhibition During the Subacute Stage of Stroke
To assess whether the impact of MPO inhibition on infarct volume is greatest when the inhibitor is given during the acute or subacute stages, we administered ABAH either immediately after MCAO on day 0 (acute stage), or starting on day 2 after stroke induction (subacute stage). Subacute treatment resulted in significantly smaller day 21 lesion volume (P=0.0088; Figure 7A) compared with acute treatment. Both acute (P<0.01) and subacute (P<0.0001) treatments improved the neurobehavioral outcome on day 21 compared with saline-treated mice (Figure 7B), but subacute treatment resulted in almost similar improvement as that of treatment throughout the entire period (compare Figure 6A with Figure 7B), and was significantly better than treatment in the acute period alone (P<0.01; Figure 7B). Analysis of variance with Tukey's post hoc analysis confirmed these observations (P<0.0001, F (3,28)=0.318).
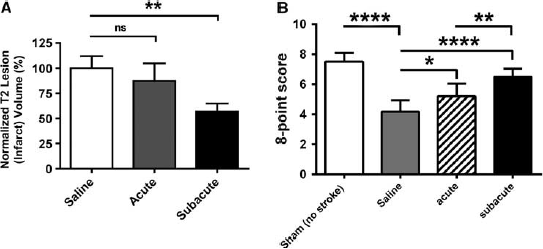
Myeloperoxidase (MPO) inhibition has the greatest impact when treatment was begun during the subacute stage of stroke (2 days) and continued for 21 days. (
Discussion
In this study, we found that progression of the ischemic lesion beyond the initial 24-hour period is accompanied by persistently elevated MPO activity in the untreated animals. Treatment with the irreversible MPO inhibitor ABAH significantly reduced the infarct volume and improved neurobehavioral outcome, suggesting that MPO has an important role in infarct propagation and associated tissue damage. Importantly, similar observations were made in MPO-KO mice, and there was no additional beneficial effect of ABAH in MPO-KO mice, confirming the specificity of the finding and directly implicating MPO and its reaction products in the underlying pathophysiology rather than as a bystander molecule or inflammatory marker without therapeutic significance.
Myeloperoxidase inhibition as a treatment strategy is unique in that it appears to be most effective during the subacute stage of stroke evolution. We found that ABAH can cross intact BBB and its action is also likely enhanced by BBB breakdown in stroke. Interestingly, neurobehavioral improvement is evident even during the subacute stages of infarction on days 3 and 7 in ABAH-treated or MPO-KO mice. Taking into account this observation and the fact there is significant MPO activity on days 3 and 7, this again implicates MPO activity as having a deleterious role in infarct propagation, and suggests that treatment against MPO would be beneficial until MPO activity has subsided, at or beyond the 21-day postinfarct period in our model.
In the absence of treatment, the final infarcted area usually includes the area of restricted diffusion and often the area surrounding the restricted diffusion on DWI imaging.22, 23, 30 This trend was reflected in the control saline-treated mice. However, the average final lesion was smaller in both ABAH-treated mice and MPO-KO mice compared with the infarct volume on day 1 DWI imaging, suggesting that MPO inhibition or the absence not only helps to prevent infarct propagation (secondary injury) but may actually result in some reversibility of the initially injured area.
Myeloperoxidase promotes injury because of its pleiotropic role in oxygen radical generation, inflammation, and vasomotor function. It is a multifunctional enzyme that promotes oxidative stress via its enzymatic products such as hypochlorous acid, hypothiocyanous acid, and chlorotyrosine. These reactive species can also modify lipids, 31 activate metalloproteinases, 32 and induce BBB dysfunction. 33 In addition to its direct enzymatic functions of generating radicals and oxidizing agents, inhibiting its enzymatic activity has been shown to decrease leukocyte recruitment and ameliorate demyelination, 21 indicating a role of MPO activity as a positive feedback for promoting inflammation and damage. The MPO molecule also has been shown to have a cytokine-like role in signaling to attract neutrophils 34 and delay neutrophil apoptosis 35 to prolong inflammation. Myeloperoxidase-deficient humans exhibit preserved vasomotor function, 36 further suggesting a role for MPO in the integrity of vascular responsiveness.
Myeloperoxidase enzymatic activity depends on the presence of hydrogen peroxide, likely generated from NADPH oxidase activity. Therefore, MPO acts downstream of NADPH oxidase, a major pro-oxidant enzyme in brain. Unlike NADPH oxidase, which is membrane bound, MPO is secreted into the extracellular environment and can exert its effects at a distance. 37 While both NADPH oxidase and MPO may be therapeutic targets in stroke, NADPH oxidase deficiency results in chronic granulomatous disease that often causes death at a young age. However, MPO deficiency in humans does not significantly impact life expectancy, 38 making MPO potentially a better target for translation. We also found that treatment with an MPO inhibitor during the subacute stage conferred significant beneficial effects, demonstrating that MPO-mediated inflammation is most damaging during the subacute stage of infarct evolution. Our conclusions differ from those of Takizawa et al 18 but are concordant with the results from Matsuo et al. 5 Our study provides longitudinal in vivo data in living animals on imaging, neurobehavior, survival, and different treatment regimens experiments that extend the previous studies beyond day 1 after infarction. In our model, MPO activity remains elevated on day 21, and both short-term and long-term data are consistent with MPO inhibition/absence being beneficial for stroke.
Interestingly, we found a much larger reduction in infarct volume on days 3 and 7 in the MPO-KO mice compared with ABAH-treated mice. This may reflect less than ideal onset of pharmacological inhibition. Moreover, the MPO inhibitor we used for this study only suppressed MPO activity by 30%. This suggests the need for developing more potent and faster acting MPO inhibitors for future study. In this regard, the absence of a more powerful long-term effect on injury volume in the KO mice could be due to upregulation of other compensatory molecules in mice that are congenitally devoid of MPO. For example, it has been found that during sepsis there is increased inducible nitric oxide synthase expression and nitric oxide production, 39 as well as increased T cell-mediated inflammation in MPO-KO mice. 40 There may also be a limit to the extent of improvement conferred by MPO inhibition as other molecules and mechanisms have been implicated in promoting additional damage 41 including other inflammatory pathways mediated by NADPH oxidase. 42 Most likely, our observations reflect a combination of these mechanisms.
In conclusion, our results show that MPO, a downstream effector of the inflammatory cascade, is a key mediator of tissue damage and secondary infarct propagation and consolidation in stroke. Myeloperoxidase is therefore an attractive target mitigating tissue damage, in particular during the subacute stage of stroke evolution, during which few effective therapies currently exist.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank Dr Lorette Noiret for assistance with statistical analysis and Ms. Veronika Glyudza for preparation of the figures.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
