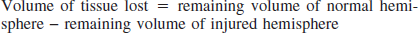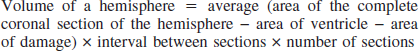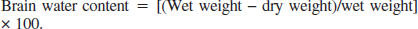Abstract
Prolonged hypothermia reduces ischemic brain injury, but its efficacy after intracerebral hemorrhagic (ICH) stroke is unresolved. Rats were implanted with core temperature telemetry probes and subsequently subjected to an ICH, which was produced by infusing bacterial collagenase into the striatum. Animals were kept normothermic (NORMO), or were made mildly hypothermic (33–35°C) for over 2 days starting 1 hour (HYP-1), 6 hours (HYP-6), or 12 hours (HYP-12) after collagenase infusion. Others were cooled for 7 hours beginning 1 hour after infusion (BRIEF). Skilled reaching, walking, and spontaneous forelimb use were assessed. Normothermic ICH rats sustained, on average, a 36.9-mm3 loss of tissue at 1 month. Only the HYP-12 group had a significantly smaller lesion (25.5 mm3). Some functional improvements were found with this and other hypothermia treatments. Cerebral edema was observed in NORMO rats, and was not lessened significantly by hypothermia (HYP-12). Blood pressure measurements, as determined by telemetry, in BRIEF rats showed that hypothermia increased blood pressure. This BRIEF treatment also resulted in significantly more bleeding at 12 hours after ICH (79.2 μL) versus NORMO-treated rats (58.4 μL) as determined by a spectrophotometric hemoglobin assay. Accordingly, these findings suggest that early hypothermia may fail to lessen lesion size owing to complications, such as elevated blood pressure, whereas much-delayed hypothermia is beneficial after ICH. Future experiments should assess whether counteracting the side effects of early hypothermia enhances protection.
Intracerebral hemorrhage (ICH) occurs when a blood vessel ruptures, releasing blood into surrounding brain tissue. ICH commonly occurs in the striatum, pons, thalamus, and cerebellum, and accounts for approximately 15% of strokes in Western cultures (Mayo et al., 1996). Mortality is approximately 50% at 30 days and only 10% of survivors live completely independently (Broderick, 1994). Neurologic impairments result from direct tissue destruction, surrounding tissue compression, cerebral edema, and reduction of cerebral blood flow (Diringer, 1993). Unfortunately, therapies aimed at treating these perturbations have not substantially improved outcome in patients. Several putative cytoprotectants have been tested in rodents, including free-radical scavengers (Peeling et al., 1998, 2001a), anti-inflammatory agents (Del Bigio et al., 1999;Mayne et al., 2001;Peeling et al., 2001b;Power et al., 2003), thrombin inhibitors (Kitaoka et al., 2002), anti-apoptotic agents (Rodrigues et al., 2003), aspiration of the hematoma (Altumbabic et al., 1998), and hypothermia (Kawai et al., 2001;MacLellan et al., 2002). Although several of these therapies persistently reduce gross neurologic impairments (Mayne et al., 2001;Peeling et al., 1998, 2001a, Peeling et al., b), such as spontaneous rotation, none have improved performance in more demanding functional tests (e.g., skilled reaching) or provided substantial long-term histologic protection. Long-term assessment is necessary because treatments may simply delay and not stop injury (Dietrich et al., 1993;Valtysson et al., 1994).
Prolonged postischemic hypothermia improves functional recovery and reduces injury after global (Colbourne and Corbett 1995;Colbourne et al., 1999;Hickey et al., 2000) and focal ischemia (Colbourne et al., 2000;Corbett et al., 2000;Maier et al., 1998) in rodents to an extent that is arguably unsurpassed by pharmacologic treatments. Hypothermia is a safe and feasible treatment for stroke (Georgiadis et al., 2001;Schwab et al., 1998, 2001) and global ischemia (The Hypothermia After Cardiac Arrest Study Group, 2002) in humans. Notably, recent clinical trials report improved outcome when prolonged hypothermia was induced after cardiac arrest (Bernard et al., 2002;The Hypothermia After Cardiac Arrest Study Group, 2002). Because prolonged hypothermia is remarkably efficacious after global and focal ischemia, it might provide benefit after hemorrhagic stroke. Hypothermia may reduce secondary consequences of ICH such as ischemia in the perihematoma region, inflammation, oxidative damage, edema, blood—brain barrier disruption, and increased intracranial pressure. For example, hypothermia reduces edema and blood—brain barrier permeability after focal cerebral ischemia (Karibe et al., 1994). Furthermore, prolonged hypothermia (24 hours) reduced vasogenic edema formation after intrastriatal thrombin injection in rats (Kawai et al., 2001, 2002). Unexpectedly, however, when induced 1 hour after infusion of bacterial collagenase into rat striatum, prolonged hypothermia (2 days) failed to reduce volume of tissue lost or behavioral deficits (MacLellan et al., 2002).
We hypothesized that although hypothermia effectively treats components of hemorrhagic injury, our failure to find protection in a previous study (MacLellan et al., 2002) might have been due to deleterious physiologic consequences of the early phase of the prolonged hypothermia treatment. For example, cooling conscious rats with fans and water misters may cause acute increases in systemic blood pressure that promote further bleeding after cerebral hemorrhage in rats (Benveniste et al., 2000). Furthermore, hypothermia is well known to inhibit enzymatic reactions of the coagulation cascade, causing abnormalities that exacerbate bleeding (Kirkpatrick et al., 1999;Schubert 1995). Such adverse effects of hypothermia likely contribute to increased insult severity and may counteract the beneficial effects of prolonged cooling before the ICH has clotted.
Infusion of bacterial collagenase into the striatum disrupts vasculature and causes bleeding in surrounding brain tissue (Rosenberg et al., 1990). The progression of injury (Del Bigio et al., 1996) and concomitant behavioral deficits (DeBow et al., 2003b;Peeling et al., 2001a) are well characterized in this model of ICH. Whereas some behavioral effects (e.g., spontaneous rotation) largely recover over weeks, other functional deficits are long-lasting, such as contralateral-forelimb stepping errors while walking across a beam or horizontal ladder, deficits in skilled reaching, and diminished spontaneous use of the contralateral forelimb during exploration. Therefore, we used this model and an appropriate array of functional tests to assess the efficacy of various mild hypothermia treatments induced 1 to 12 hours after ICH. We hypothesized that cooling rats soon after ICH, during active bleeding, would exacerbate injury, whereas cooling many hours after injury would improve outcome. Thus, prolonged hypothermia delayed for 12 hours after ICH was expected to be most beneficial, and early, long-lasting hypothermia was expected to provide no benefit. Finally, we hypothesized that early and brief hypothermia would be deleterious because it might aggravate bleeding or increase lesion size but not last long enough to protect against the delayed consequences of the ICH.
MATERIALS AND METHODS
Animals
In total, 214 male Sprague-Dawley rats (Ellerslie, Edmonton, Alberta, Canada) were used in this study. Rats weighed between 375 and 450 g and were approximately 16 weeks old on the day of ICH. All procedures used were in accordance with the Canadian Council on Animal Care guidelines, and were approved by the Biological Sciences Animal Policy and Welfare Committee at the University of Alberta.
Montoya staircase training
Rats were food deprived to 90% of their free-feeding weight over 3 days and weighed daily to ensure stability at 90%. They were trained in the staircase test, which is a measure of independent forelimb reaching ability (Montoya et al., 1991), over 15 days for two 15-minute trials per day separated by 4 or 5 hours, 5 days per week (Colbourne et al., 2000). Fifteen rats were excluded because they failed to reach the criterion of 9 pellets (45 mg each; Bio-Serv, Frenchtown, NJ, U.S.A.) per side out of a possible 21 by the last 3 consecutive days during training.
Core temperature telemetry probe implantation
Seven days after the last staircase training session, rats were briefly (10 minutes) anesthetized with isoflurane (4% induction; 1.5–2% maintenance) in 70% N2O, 30% O2. Core telemetry temperature probes (model TA10TA-F40; Transoma Medical, St. Paul, MN, U.S.A.) were implanted into the peritoneal cavity (DeBow and Colbourne, 2003). The wound was infiltrated with a local anesthetic (Marcaine; Sanofi Canada, Markham, Ontario, Canada) and sutured closed. Rats were then housed individually on receivers (RPC-1; Transoma Medical) interfaced to a computer running telemetry software (A.R.T. 2.2; Transoma Medical) that sampled temperature every 30 seconds. Data from the complete day before ICH surgery served as a baseline.
Intracerebral hemorrhage
Rats were anesthetized with isoflurane (1.5–2% maintenance) and placed in a stereotaxic frame. Throughout surgery (30 minutes) core temperature was maintained near normothermia (37°C) with a rectal temperature probe and heating blanket. Under aseptic conditions a midline scalp incision was made and a hole was drilled in the skull 3 mm lateral (right) and 0.2 mm anterior to bregma. A 26-gauge needle (Hamilton syringe; Hamilton, Reno, NV, U.S.A.) was lowered 5.5 mm below the surface of the skull and 1.0 μL sterile saline containing 0.2 U bacterial collagenase (Type IV-S; Sigma, Oakville, Ontario, Canada) was infused into the striatum over 5 minutes to create an ICH (DeBow et al., 2003b;MacLellan et al., 2002). The needle remained in place for an additional 5 minutes after infusion. A metal screw (model MX-080–2; Small Parts, Miami Lakes, FL, U.S.A.) was inserted into the thickness of the skull bone and the scalp wound was treated with Marcaine and closed with staples. To minimize invasiveness and duration of anesthetic, rats were not intubated during surgery. Rats were randomly assigned to treatment condition and the abdomen and back were shaved to facilitate subsequent hypothermia treatment or to prevent knowledge of group identity in all rats.
Temperature control after surgery
All animals were maintained near normothermia for 1 hour after infusion of bacterial collagenase. Some rats were kept above 37°C for an additional 11 hours and were then allowed to regulate their own temperature (NORMO, n = 17). Other rats were slowly cooled beginning 1 hour after infusion, by a rate of 1°C every 30 minutes to a core temperature of 33°C and maintained at this level for 24 hours. These rats were then slowly warmed (1°C/30 min) to 35°C and kept at that temperature for an additional 24 hours before slow warming (HYP-1, n = 18). In some rats, the hypothermia treatment (as described above) was initiated 6 hours (HYP-6, n = 15) or 12 hours (HYP-12, n = 17) after ICH induction. Other rats were cooled to 33°C starting 1 hour after ICH, but maintained at this level for only 7 hours before being heated to normothermia (BRIEF, n = 17). Temperature was precisely (± 0.3°C) regulated by a servo-controlled system that used fans and fine water misters for cooling and infrared lamps for warming (DeBow and Colbourne, 2003). Temperature was monitored in all animals for 7 days after surgery before telemetry probes were quickly removed under isoflurane anesthesia. One additional rat was excluded because of technical problems during surgery. An additional nine rats were excluded because of aberrant lesion placement. There was no mortality due to the ICH or treatment procedures in this part of the study.
Behavioral testing
On days 7, 14, 21, and 28, the forelimb use asymmetry test (cylinder test) (DeBow et al., 2003b;Tillerson et al., 2001) and the horizontal-ladder walking test (DeBow et al., 2003b;Metz and Whishaw 2002) were used to assess functional outcome. Rats were also tested in the staircase test on days 21 through 25.
Horizontal-ladder walking test
Each testing session was videotaped and consisted of three consecutive trials of rats walking across a horizontal ladder with variably spaced bars ranging from 1 to 3 cm. For each forelimb, the error rate (limb slips completely through the bars) made while traversing a 0.5-m segment of the ladder over three trials was determined.
Forelimb use asymmetry test
Rats were placed in a transparent cylinder (height, 45 cm; diameter, 33 cm) for 5 minutes and allowed to spontaneously explore the cylinder. A video camera set up below the cylinder recorded all forelimb placements on the wall, which were subsequently analyzed according to our previous work (DeBow et al., 2003b). Briefly, we counted independent and initial fore-limb use (exploration of wall after rearing) for contacting the cylinder wall during each test session. The percent of contralateral and ipsilateral forelimb use when exploring the cylinder walls was analyzed as [(number of contacts with contralateral limb/ipsilateral + contralateral limb use) × 100].
Staircase test
Rats were food-deprived to 90% of their free-feeding weight 5 days before staircase testing. This consisted of 10 trials (two 15-minute trials per day, separated by 4 or 5 hours) on days 21 through 25 after ICH. The total number of pellets retrieved on each side was recorded for each trial, and performance was expressed as a percent of baseline (average of the last 10 training trials when performance reaches asymptotic level).
Histopathology
Thirty days after ICH, rats were euthanized with an overdose of sodium pentobarbital (Somnotol; MTC Pharmaceuticals, Cambridge, ON, Canada; 80 mg/kg, intraperitoneal) and were transcardially perfused with 0.9% saline and then 10% neutral buffered formalin. Forty-micrometer coronal sections were taken with a cryostat every 400 μm starting at +1.7 mm to bregma and extending back to −4.8 mm to bregma. Sections were stained with hematoxylin and eosin and by using Scion Image J 4.0 (Scion Corporation, Frederick, MD, U.S.A.), the volume of lesion plus atrophy (e.g. ventricular enlargement) was quantified and expressed as follows (DeBow et al., 2003b;MacLellan et al., 2002):
The brains of four additional rats, which were only subjected to an ICH, were grossly dissected and visually examined 6 hours after collagenase injection to determine whether the ICH had fully clotted at that time.
Brain water content
Brain water content was determined in nine unoperated rats, which served as a control group. An additional 44 rats were used to assess the effects of an ICH and hypothermia treatment on edema formation. The 44 rats were implanted with core temperature telemetry probes and subjected to an ICH 3 days later as described previously. All rats were kept at normothermia for 12 hours after surgery. Half of these then regulated their own temperature whereas the others received hypothermia treatment as for the HYP-12 group. Fresh brain tissue was removed at either 2 or 3 days after ICH. Using a brain matrix (Ted Pella Inc., Redding, CA, U.S.A.) a 4-mm section (2 mm anterior and 2 mm posterior to the needle injection site) was obtained and divided into each hemisphere. The weight of each fresh block was determined (i.e., wet weight). Brain blocks were placed in an oven (100°C for 24 hours) and then reweighed (i.e., dry weight). The water content of each block was determined as follows (Xi et al., 1998):
Blood pressure
To characterize acute changes in blood pressure after ICH, eight rats were implanted with a telemetry probe that measures temperature and blood pressure simultaneously (model TML2-C50-PXT; Transoma Medical). Three days later, rats were subjected to ICH and were maintained at normothermia for 12 hours (NORMO; n = 4) or they received a brief hypothermia treatment (BRIEF; 33°C for 7 hours, induced 1 hour after ICH; n = 4). Temperature and blood pressure were sampled every 30 seconds and the complete day before surgery served as a baseline. These rats were not part of the behavioral or histologic analysis.
Intracerebral blood volume
A modified spectrophotometric assay (Choudhri et al., 1997) was used to determine the volume of blood (hemoglobin) in the brain after ICH. Briefly, rats were overdosed with Somnotol (80 mg/kg, intraperitoneal) and transcardially perfused with 100 mL of 0.9% saline to remove blood from the vasculature. The brain was then extracted and distilled water was added to it to reach a total volume of 3 mL. This solution was homogenized for 30 seconds (Brinkman Instruments, Ontario, Canada) and then centrifuged (13,000 g for 30 min; model CR20B2, Hitachi, Japan). Several (minimum of three) samples of the supernatant (100 μL) were collected and reacted with Drabkins reagent (400 μL; Sigma) at room temperature for 15 minutes. Finally, absorbance readings at 540 nm (corrected for the background reading) were taken in a spectrophotometer (model DU-8; Beckman Coulter Ltd., London, U.K.) from several samples per rat and averaged. First, we generated a curve using known quantities of blood (0, 2, 4, 8, 16, 32, 64, 128 μL), which were obtained from a cardiac puncture, added to saline perfused normal rat brains (n = 11). Using the equation of the best-fitting linear regression line from this data, we determined the volume of blood remaining in other rats based on absorbance readings. Thus, additional rats, which survived for 12 hours after ICH, were subjected to an ICH with (n = 13) or without (n = 15) the BRIEF hypothermia treatment. One additional rat died in the BRIEF group and was not included in the analysis. These rats were previously implanted with core temperature probes as described earlier. They were not part of the behavioral or histologic volumetric analysis.
Data analysis
All data are expressed as a mean ± SD and were analyzed with an analysis of variance and by planned comparisons (SPSS 11.0; SPSS Inc., Chicago, IL, U.S.A.).
RESULTS
Temperature
Baseline core temperature collected on the day before ICH surgery was similar among NORMO, BRIEF, HYP-1, HYP-6 and HYP-12 groups (overall mean, 37.14° ± 0.14°C; data not shown). Similarly, temperature during the 1 hour after collagenase injection was similar among groups (overall mean, 37.12° ± 0.1°C;Fig. 1). Temperature was regulated as desired after the surgical procedure (see Materials and Methods). However, for 12 hours after surgery, the HYP-12 group was slightly (37.46° vs. 37.10°C), and significantly (P < 0.001) warmer than the NORMO group.
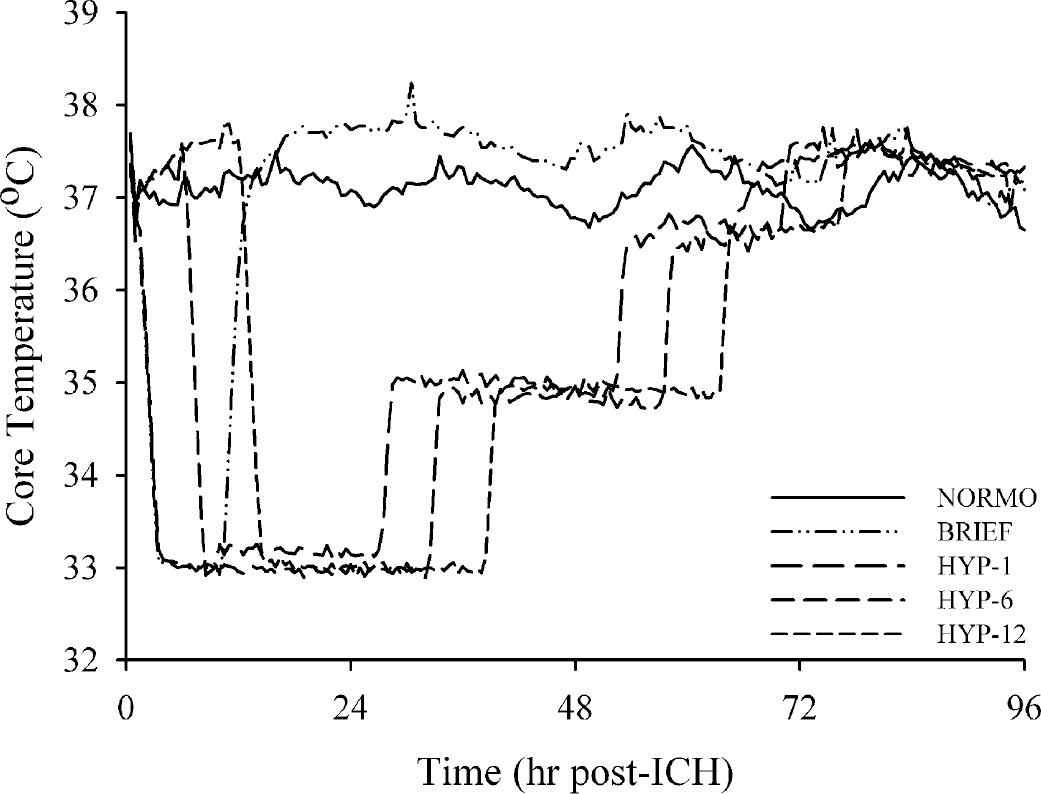
Core temperature (°C) for 4 days after ICH. Temperature from 5 to 7 days was similar (data not shown). See Materials and Methods for temperature manipulations. The temperature profiles (data not shown) of the rats used for collecting blood pressure readings, edema measurements, or estimating blood volume with spectrophotometry were similar to those of their respective groups shown here.
Body weight
Body weight averaged 90.3% of baseline (± 0.83%) during staircase training. Body weight was similar among groups (P = 0.324) on the day of ICH (overall average = 418 ± 18.54 g). Postoperative weight (Fig. 2) quickly returned to normal in the NORMO group and more gradually returned to normal in the hypothermia-treated groups. All groups were above surgery day weight by day 14.
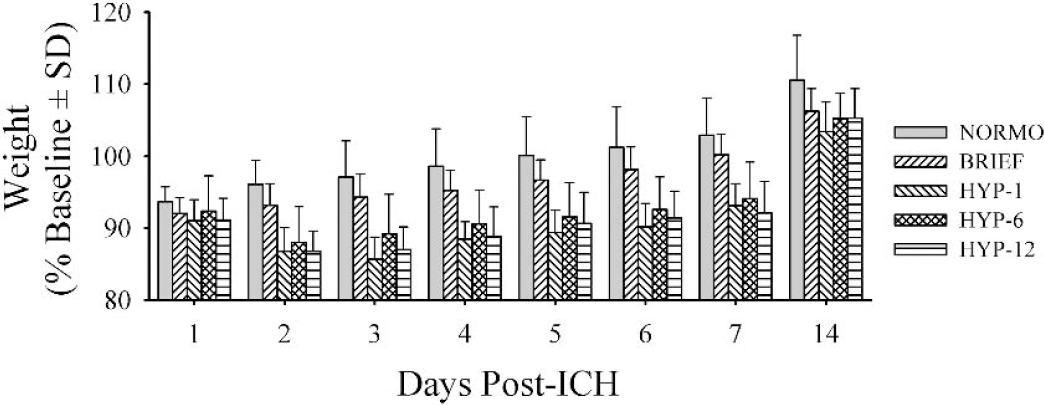
Postoperative weight (g) on post-ICH days 1 through 7 and 14. See Results for a description of statistical findings.
Staircase testing
All groups performed similarly during training (data not shown). The test data were analyzed as a percentage of this baseline performance (last 10 trials). The ICH procedure (NORMO) resulted in a substantial reaching impairment with the contralateral limb (63.16% of baseline; P = 0.001; see Fig. 3). The ipsilateral forelimb success was not significantly lessened in the NORMO group (85.48%; P = 0.118). Only the HYP-12 group appeared to be improved by hypothermia treatment. However, planned comparisons (HYP-12 vs. NORMO) showed no significant effect with either the contralateral (81.27% of baseline; P = 0.123) or ipsilateral forelimb (101.47% of baseline; P = 0.060).
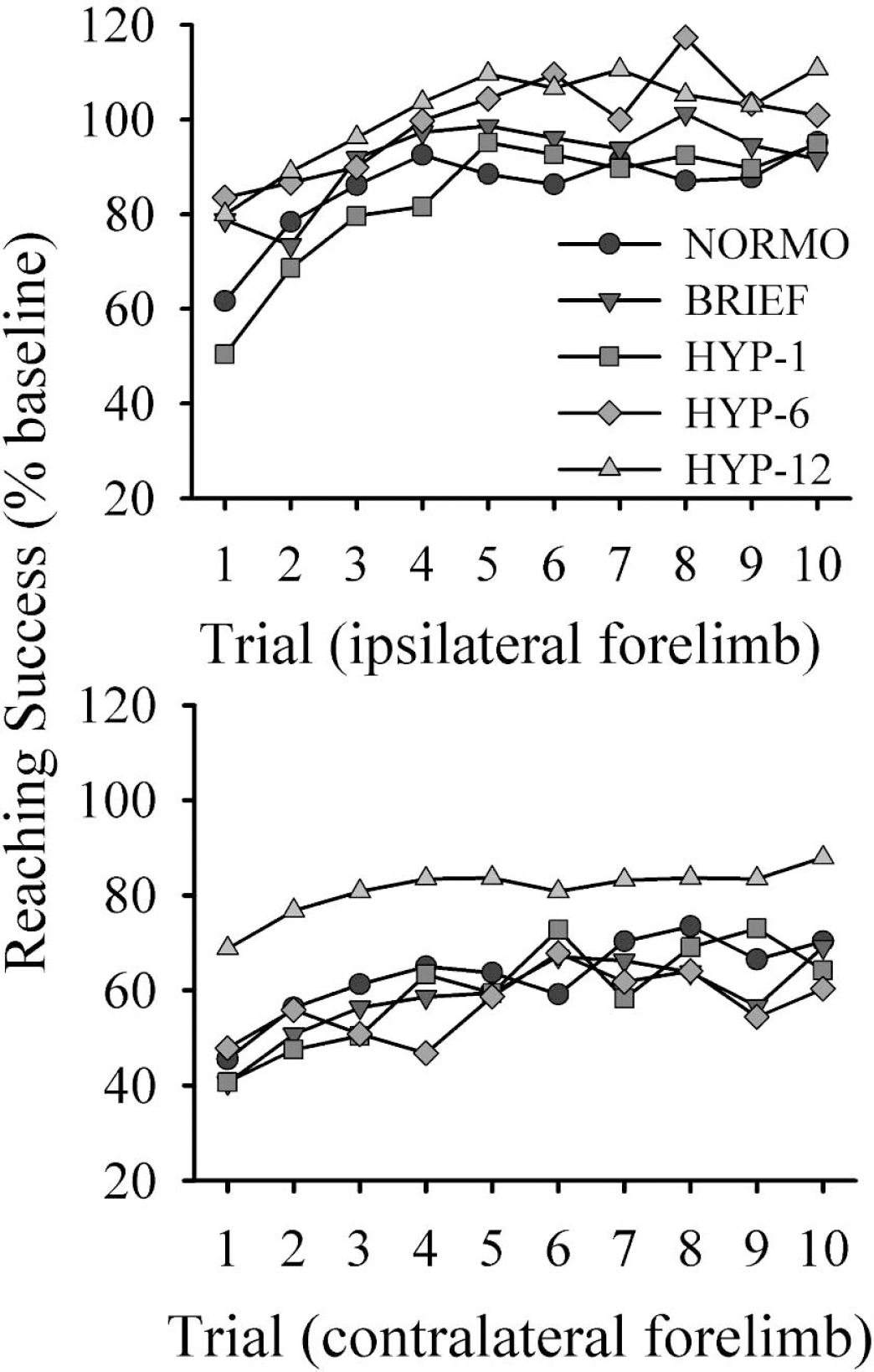
Success (% baseline) with the ipsilateral (top) and contralateral (bottom) forelimbs in the Montoya staircase apparatus over 10 test trials. See Results for a description of statistical findings.
Horizontal-ladder walking test
The NORMO group had a mean error rate of 9.76% with the contralateral forelimb (Fig. 4). The HYP-1 and HYP-12 groups had significantly lower error rates at, on average, 5.39% (P = 0.012 vs. NORMO) and 6.14%, respectively (P = 0.039). The ipsilateral forelimb error rate did not differ significantly among groups (data not shown; P = 0.172).
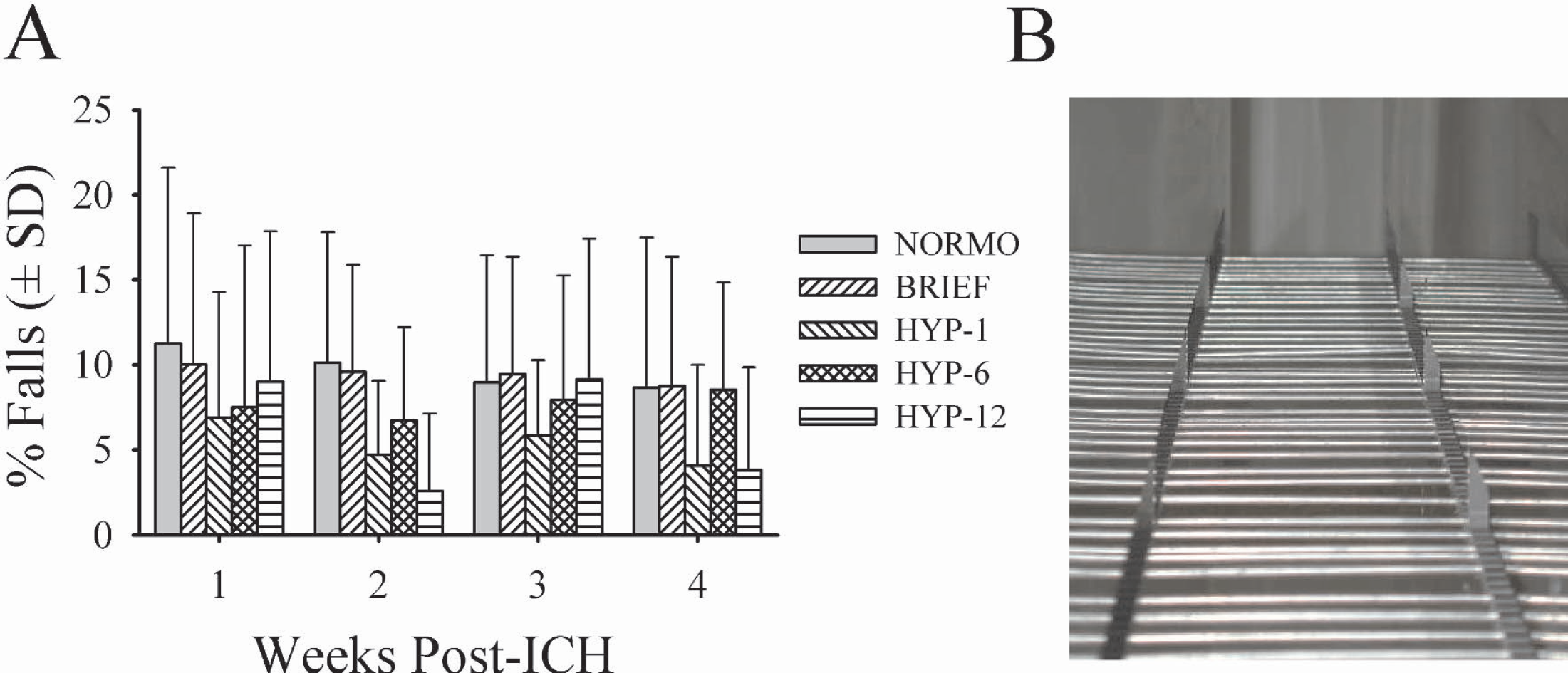
Error rate (% falls through bars;
Limb-use asymmetry test
There was a significant group main effect for independent use of the contralateral limb (P = 0.005;Fig. 5). The BRIEF (P = 0.021), HYP-1 (P = 0.001), and HYP-12 (P = 0.003) groups were significantly improved overall (i.e., they used their contralateral forelimb more frequently than NORMO rats).
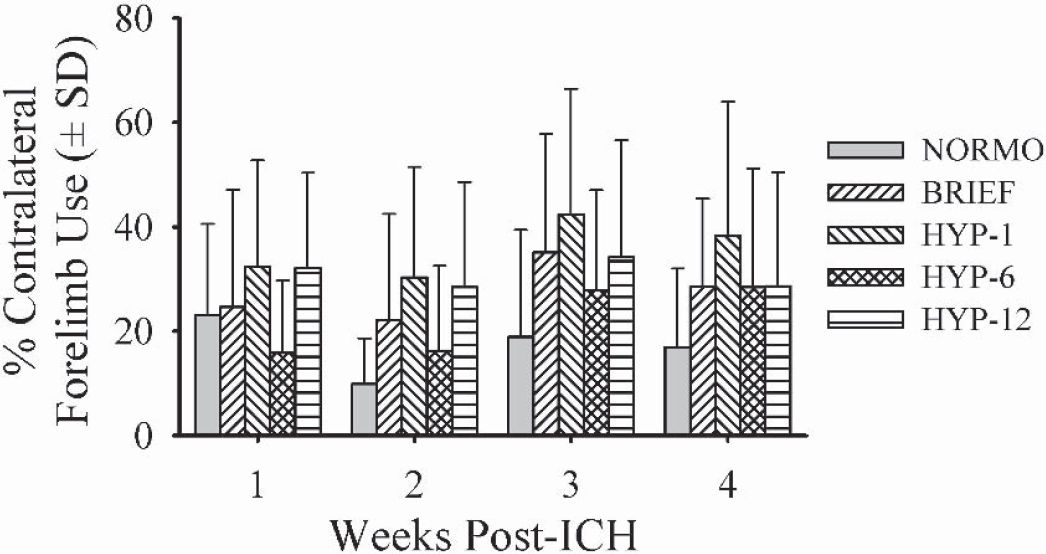
Spontaneous contralateral forelimb use [(number of contacts with contralateral limb /ipsilateral + contralateral limb use) × 100] in the cylinder test on post-ICH days 7, 14, 21, and 28.
Composite behavioral score
All rats that had undergone behavioral testing (n = 85) were ranked from best to worst on their average performance (across trials) for each of the three behavioral tests. These three ranks were then averaged and then analyzed by analysis of variance. The purpose of this procedure was to assess overall performance, and with this analysis only the HYP-1 (mean rank, 36.71 ± 16.87; P = 0.040) and HYP-12 (34.71 ± 15.62; P = 0.020) groups were significantly better (i.e., performed better) than the NORMO group (50.17 ± 18.68).
Histopathology
The lesion volumes at 30 days after ICH are given in Fig. 6A. Untreated NORMO rats sustained damage primarily to the striatum, but also to the internal capsule, globus pallidus, thalamus, and corpus callosum (Fig. 6B). Ventricular dilation was common ipsilateral to the lesion, but frequently occurred in the contralateral hemisphere as well. Prolonged hypothermia delayed 12 hours after ICH reduced the volume of injury by 30.88% (P = 0.047 vs. NORMO). The HYP-1 (P = 0.345), HYP-6 (P = 0.810), and BRIEF groups were not significantly different than the NORMO group, though there was a trend (P = 0.063) toward a larger lesion (by 28.46%) in the BRIEF group.
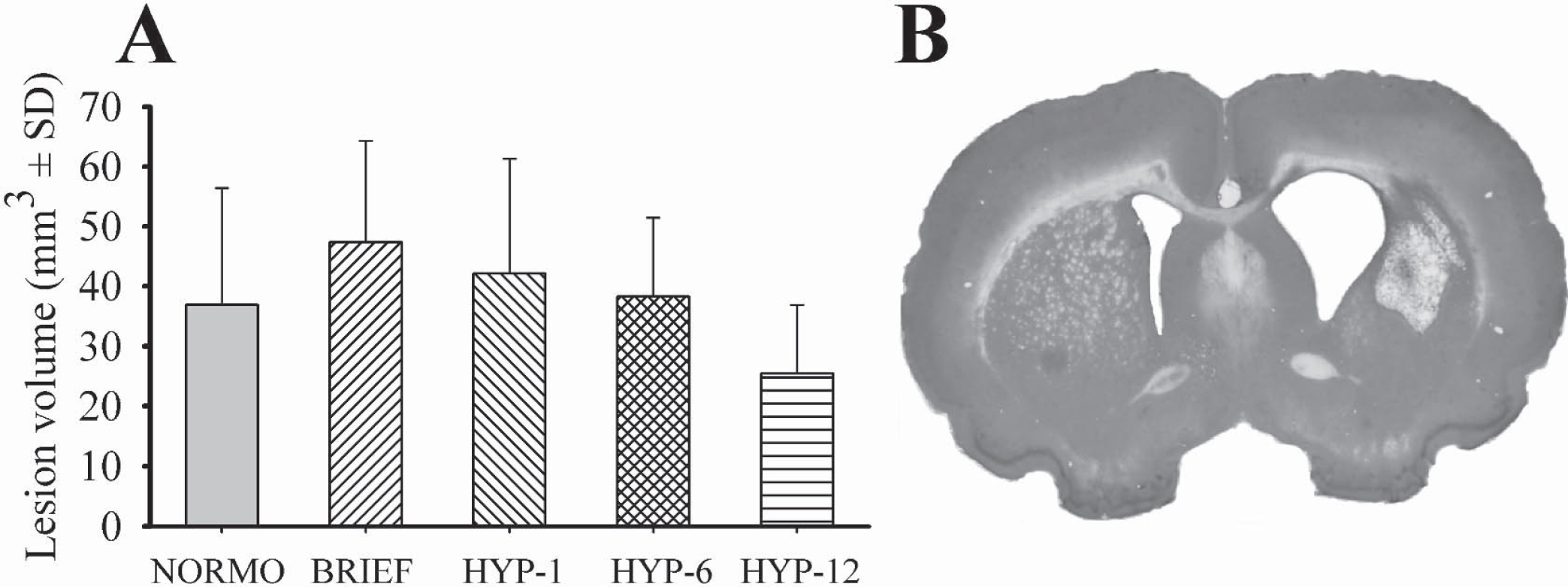
Lesion volume (mm3;
A visual inspection of several brains at 6 hours after ICH indicated that the blood released into the brain had yet to be fully clotted, but was nearly so. The area of blood released into the brain also appeared to exceed that which was eventually destroyed (Fig. 6B).
All three behavioral tests significantly, though modestly, correlated with the volume of tissue lost when all groups were considered in the analysis. The Pearson r values were 0.26 (P = 0.008), −0.244 (P = 0.012), and −0.466 (P < 0.001) in the ladder, cylinder, and staircase tests, respectively. Thus, there were greater functional deficits in rats with larger lesions.
Brain water content
The water content (one hemisphere block) in unoperated controls was 60.57% ± 4.89%. Water content significantly increased to 65.50% ± 6.52% (P = 0.050 vs. control) and 66.09% ± 5.66% (P = 0.026) at 2 and 3 days, respectively, after untreated ICH (lesioned hemisphere). Brain water content was not significantly reduced by the HYP-12 treatment on either day 2 (66.25% ± 4.28%; P = 0.754 vs. NORMO) or day 3 (62.97% ± 4.89%, P = 0.189). There were no significant differences among groups in the contralateral hemisphere (data not shown).
Blood pressure
Baseline blood pressure, collected the complete day before surgery, was similar among groups (108.78 ± 9.32 mm Hg; P = 0.367). To determine if hypothermia significantly increased blood pressure, we averaged data collected over the first 6 hours after the induction of hypothermia. This period encompassed the maximal elevation in pressure apparent approximately 1 to 1.5 hours after the induction of hypothermia (Fig. 7). Post-ICH pressure (107.51 ± 6.82 mm Hg) in normothermic ICH rats did not significantly differ (P = 0.528) from baseline pressure. Brief hypothermia induced 1 hour after ICH caused a significant and prolonged (at least 6 hours) blood pressure hypertension relative to untreated rats (118.76 ± 4.83 mm Hg; P = 0.036).
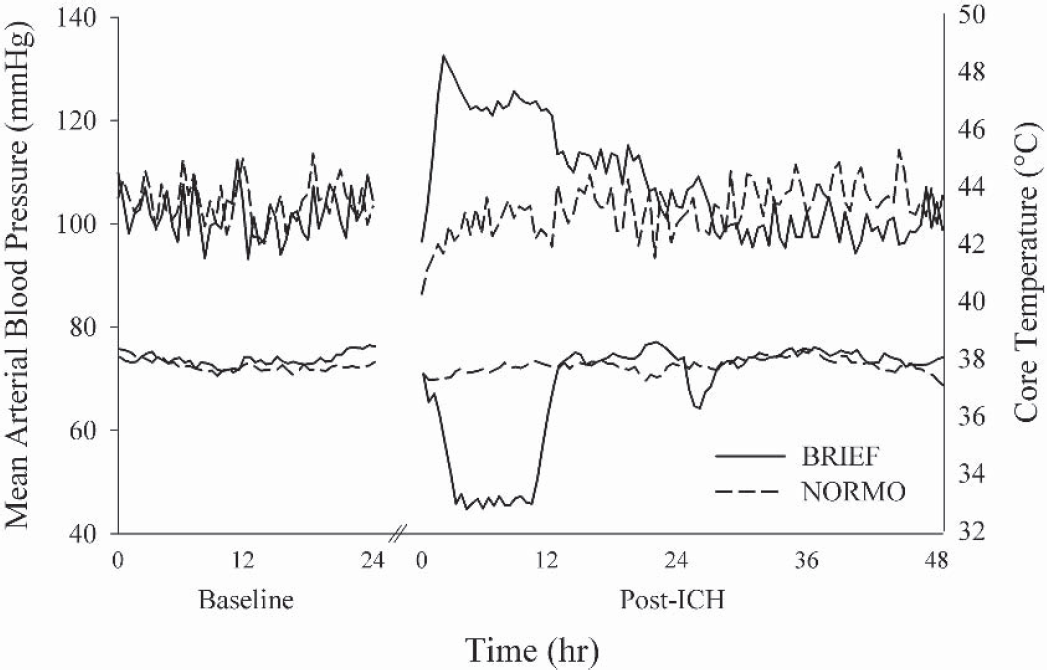
Mean arterial blood pressure (mm Hg; left y-axis) and core temperature (°C; right y-axis) in two representative ICH rats (one NORMO and one BRIEF) during baseline and after ICH. Hypothermia caused a significant elevation in blood pressure. See Results for a description of statistical findings.
Intracerebral blood volume
Rats subjected to the BRIEF cooling treatment after ICH had an estimated blood volume of 79.2 ± 25.2 μL whereas normothermic ICH rats has a volume of 58.4 ± 20.5 μL (P = 0.023). Thus, the BRIEF treatment significantly increased bleeding by 35.6% at 12 hours after ICH.
DISCUSSION
Prolonged and delayed (12 hours) mild hypothermia reduced the ICH lesion volume and concomitant behavioral deficits after a moderate-sized ICH in rats. To date, this is the only cytoprotective therapy that persistently (i.e., 30-day survival time) reduces the volume of injury after ICH. Remarkably, the greatest histologic protection occurred with a treatment starting 12 hours after the ICH, whereas earlier treatments failed to lessen the volume of tissue lost. This contrasts with findings after both global and focal cerebral ischemia, where the greatest protection occurs when hypothermia begins as early as possible (Colbourne et al., 1997, 1999;Dietrich et al., 1996;Maier et al., 1998). Thus, the ideal starting time of hypothermia therapy in the clinic may critically depend on the type of insult.
We suspected that side effects in the early phase of hypothermia treatment would negate the beneficial effects of the therapy after ICH resulting in no net protection. Indeed, we previously observed (MacLellan et al., 2002) no histologic protection with a 1-hour delayed treatment, and this was again found in the HYP-1 group. Furthermore, neither the BRIEF group nor the HYP-6 group had histologic protection. One possible side effect is coagulopathy (slowed clotting time), which is well documented during hypothermia (Kirkpatrick et al., 1999;Schubert 1995). Our method of cooling using fans and water misters (DeBow and Colbourne, 2003) is also stressful to conscious rats temporarily increasing blood pressure in cooled ICH rats (present study) as well as in normal rats (MacLellan et al., 2003). Notably, acutely high blood pressure increases hemorrhage volume after needle biopsy in rats (Benveniste et al., 2000). Therefore, both coagulopathy and hypertension likely exacerbated bleeding in our study with the BRIEF treatment and contributed to increased insult severity in treated ICH rats that were still actively bleeding (i.e., BRIEF, HYP-1, and HYP-6 groups). Finally, visual observations made 6 hours after ICH indicated that the hematoma was not fully formed at that time in this model, which may account for the lack of structural protection in the BRIEF, HYP-1, and HYP-6 groups. Further experiments designed to determine whether these and other side effects of hypothermia exacerbate hemorrhagic injury were beyond the scope of this efficacy study but are planned (e.g., regulating blood pressure).
We suspect that hypothermia (even when delayed for 12 hours) counteracts multiple mechanisms of hemorrhagic tissue damage (e.g., inflammation, oxidative stress, ischemia). Perhaps it is because hypothermia counters many injurious cascades that long-term histologic and functional protection occurs, whereas most pharmacologic agents that generally target one component of injury, such as oxidative damage (Peeling et al., 2001a), fail to provide histologic benefit against hemorrhagic stroke. One prominent contributor to a poor outcome is edema, which hypothermia is well known to reduce (Colbourne et al., 1997;Dietrich et al., 1996). For instance, hypothermia reduces swelling after experimental focal ischemia (Inamasu et al., 2000;Karibe et al., 1994;Kollmar et al., 2002) and intrastriatal thrombin injections (Kawai et al., 2001, 2002). Thus, it was surprising that the reduction in edema (3% at 3 days after ICH) by hypothermia was not statistically significant in this study. Perhaps more animals were needed in each group or the sampling times (2 and 3 days after ICH) were not ideal. However, pilot work in our laboratory indicates that edema is minimal at 1 and 4 days after ICH. Finally, it is possible that hypothermia reduces edema, but that it rebounds during the rewarming phase, which is when we sampled.
One limitation of this study is that we did not determine the extent by which 12-hour-delayed hypothermia reduces striatal cell death. There was a significant reduction in the volume of tissue lost; however, it is also possible that hypothermia reduced injury outside of the primary lesion site (e.g., reduced dendritic atrophy of surrounding neurons and less axonal degeneration). Indeed, the volume of the tissue lost increases over days to weeks in this ICH model (Del Bigio et al., 1996;Felberg et al., 2002), and this is unlikely to be due solely to continuing cell death. Thus, the long-term protection observed in the HYP-12 group could result from reduced cell death and limiting delayed degenerative events. These possibilities are not easily dissociated. Regardless, the primary purpose of this study was to determine whether hypothermia could persistently improve outcome, which it did. Future studies should determine whether protection is permanent (e.g., 6-month survival time). Cell counts should be used in those studies to determine the locus of protection.
There are several limitations with the collagenase ICH model to consider. Most importantly, the model we used, which resulted in greater injury than many studies, produced variably sized lesions that did not consistently affect one structure (e.g., lateral striatum). This variability is not surprising given the nature and severity of the insult. Such inherent variability causes more variable behavioral deficits (e.g., cylinder data) and contributes to the imperfect correlations between behavioral scores and lesion volume in this study, a finding that is common in the literature (Corbett and Nurse, 1998). For instance, functional deficits on simple behavioral measures (e.g. neurologic deficit score) often spontaneously recover despite substantial brain injury (Cregan et al., 1997). Several studies of hemorrhagic stroke have found significant reductions in neurologic deficits without histologic protection (Peeling et al., 2001a, 2001b;Mayne et al., 2001). In addition to variability in the size of a lesion, one must consider that there are individual differences in brain organization, motivation, recovery mechanisms, and the presence of undetected damage that contribute to discordant histologic and functional outcomes. Therefore, it is not surprising that we detected behavioral, but not histologic, improvement with the HYP-1 treatment, whereas both were found in the HYP-12 group. We attempted to overcome these problems by using relatively large group sizes, multiple testing sessions, and three sensorimotor tests. We also used a composite behavioral score. The use of both histologic and multiple functional assessments are required to assess the effectiveness of any therapy (Corbett and Nurse, 1998;DeBow et al., 2003a).
An additional problem with this model was that the lesion sometimes affected the contralateral hemisphere (i.e., apparent bilateral atrophy). This effect appeared to be least common in the HYP-12 group. Accordingly, we may have underestimated the volume of lesion more so in the NORMO group and thereby underestimated the protective effect of the HYP-12 therapy. Unfortunately, there is no simple way to overcome this problem. An analysis of only the lesioned side does not correct for individual differences in brain size (i.e., compared with the contralateral hemisphere) or differences in coronal section selection.
A final consideration is whether prolonged hypothermia would similarly affect other models of hemorrhagic stroke (e.g., subarachnoid hemorrhage) or even alternative ways of modeling a striatal ICH (e.g., blood-injection method). Unfortunately, neither the collagenase nor the blood injection model perfectly reflects the clinical situation. We predict that early hypothermia may provide greater benefit after direct intrastriatal autologous blood injection than after collagenase injection, because in the latter there is more active bleeding that would presumably be increased by hypothermia. This hypothesis is currently being investigated, and these issues must be resolved before hypothermia is applied to patients with ICH. Finally, other insults that cause intracranial bleeding, notably traumatic brain injury, may not be similarly affected by hypothermia because hypothermia reduced, not increased, extravasated hemoglobin levels in a rat model of traumatic brain injury (Kinoshita et al., 2002). This difference may relate to the time course, quantity or location of bleeding in traumatic brain injury versus ICH as well as to method of cooling. Thus, the latter parameter will be investigated in ICH in our laboratory.
In summary, when delayed 12 hours, prolonged mild hypothermia provides persistent histologic and functional protection after striatal ICH in rats. Hypothermia is thus the only treatment, at present, that persistently reduces injury and improves functional outcome after experimental ICH. Although these findings suggest that clinical investigation is warranted, there are a number of issues that must be resolved first. Notably, in addition to identifying the optimal hypothermia profile (delay, depth, and duration) and the mechanisms of action, investigators should study pharmacologic means of cooling or alternatively to counteract known side effects (e.g., coagulopathy) of whole-body hypothermia during the treatment. A mechanistic understanding of hypothermia's beneficial and deleterious effects could lead the way in finding alternative, less risky therapies. We also suggest that hypothermia be tested in larger gyrencephalic animal models of hemorrhagic stroke with more attention to clinically relevant issues (e.g., sex, age, co-morbidities such as hypertension) (DeBow et al., 2003a;Stroke Therapy Academic Industry Roundtable, 1999).
Footnotes
Acknowledgments
The authors thank D. Clark, K. Cuthbert, S. Haji, I. Harding, M. Iseke, S. McGie, and K. Wiltshire for technical assistance, Dr. P. Hurd for statistical advice, and Dr. W. Gallin for the use of his spectrophotometry equipment.