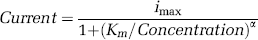Abstract
The pathophysiology of peri-lesion boundary zones in acute brain injury is highly dynamic, and it is now clear that spreading-depression-like events occur frequently in areas of cerebral cortex adjacent to contusions in the injured human brain. An automated method to assay microdialysate from peri-lesion cerebral cortex in 11 patients with intracranial haematomas requiring surgery was used. Perfusate (2 μL/min) flowed directly into a flow-injection system for assay of glucose and lactate at intervals typically of 30 secs each. Four channels of electrocorticogram (ECoG) were recorded from a subdural strip adjacent to the catheter. Several patterns of change in metabolites were identified in different time domains. Overall, the number of transient lactate events was significantly correlated with the number of glucose events (r2=0.48, P=0.027, n=10). Progressive reduction in dialysate glucose was very closely correlated with the aggregate number of ECoG events (r2=0.76, P=0.0004, n=11). It is proposed that the recently documented adverse impact of low dialysate glucose on clinical outcome may be because of recurrent, spontaneous spreading-depression-like events in the perilesion cortex.
Keywords
Introduction
Despite improvements in mortality and morbidity from serious head injury, and the availability of increasingly sophisticated acute care for patients with head injury and stroke, secondary deterioration in the days after onset of both these conditions remains a serious and sometimes fatal complication, with pathophysiology that is not fully understood. In consequence, treatment of ischemic and traumatic ‘brain swelling’ is still to a large extent empirical, and of limited benefit; a need for translational research, linking experimental models and clinical studies, has been identified (MRC(UK), 1998).
In the work reported here, a collaboration between groups in London, Denmark, and Switzerland, we addressed this challenge by seeking to use information and technical experience gained from an experimental stroke study to implement a novel application of microdialysis for clinical research in acute brain injury. An automated flow-injection assay of microdialysate from the cerebral cortex for glucose and lactate, each typically at 30 secs intervals (‘rapid-sampling microdialysis’, rsMD), was applied first in the ischemic penumbra after middle cerebral artery occlusion (MCAO) in cats (Hopwood et al, in press). Subsequently, we applied the method in perilesion cerebral cortex in patients requiring emergency craniotomy for traumatic or spontaneous intracerebral haematoma. Specifically, we reasoned that since peri-infarct depolarisations (PIDs) are a prominent feature of the penumbra in experimental MCAO in non-human primates (Branston et al, 1977; Strong et al, 2000), in the cat (Saito et al, 1995; Strong et al, 1983) and in the rat (Gill et al, 1992; Iijima et al, 1992; Nedergaard and Astrup, 1986) and contribute to its gradual recruitment into the core territory (Back et al, 1996; Busch et al, 1996; Takano et al, 1996), we should first apply rsMD experimentally to characterise the time course of dialysate glucose and lactate in the penumbra, with special reference to PIDs. In this report, we describe our subsequent experience with the application of the same method in 11 patients, with the aim of rapidly and automatically acquiring glucose and lactate time series data for two purposes. These were, first, to examine at high temporal resolution the dynamic changes in both metabolites during the evolution of a clinically significant brain injury in order to improve understanding of the pathophysiology, and second, to seek evidence for occurrence of the experimentally acquired glucose/lactate ‘signature’ of a depolarisation event in the data.
We have recently reported (Strong et al, 2002) detection of transient suppressions of amplitude of the electrocorticogram (ECoG) with the essential features of cortical spreading depression (CSD) of Leão (1944); seven of the present ECoG data sets were included in the earlier report. Thus, it was possible to attempt comparisons of ECoG and metabolite data acquired simultaneously, and at closely adjacent locations in the periphery of a focal contusion, or of a site of surgical access to a deeper (spontaneous) haematoma. A prominent feature of the results is the demonstration of a relationship between the aggregate number of depolarisation-like events and progressive depletion of brain glucose as measured in the dialysate.
Clinical procedures and methods
Patient Selection and Consent
All research procedures were approved by the local Research Ethics Committee of King's College Hospital. Criteria for admission to the study were (1) clinical indications for emergency craniotomy for traumatic or spontaneous intracerebral haematoma, (2) postoperative ventilation, (3) Glasgow Coma Score not less than 4, and (4) age range 16 to 70 years. An opinion from a relative or close friend that the patient, were they competent and aware of the purposes and risks of the study, would have agreed, was signed and documented as (surrogate) consent.
Procedures for Postoperative Care in the Intensive Care Unit
All patients were treated with endotracheal intubation, sedation, and intermittent positive pressure ventilation throughout the period of monitoring. Sedation was generally with fentanyl and midazolam, at dosages determined by patterns of response to suction and similar care routines. Propofol was substituted for these agents before withdrawal of sedation. Use of anticonvulsants was at the discretion of the attending surgeon.
Microdialysis Probe Location
Towards the completion of surgery, a sterile, flexible CMA 70 microdialysis probe (CMA Microdialysis, Stockholm, Sweden) with a 10 mm membrane and a 60 mm probe shaft length was inserted (under direct vision) obliquely to full membrane depth through a minimal pial incision, so as to lie largely within the cerebral cortex. The catheter was sited so as to be capable of withdrawal by gentle external traction, and was exteriorised through a stab incision peripheral to the craniotomy incision. The aim was to site the probe within approximately 1 cm of nonviable tissue, thus locating it in any ‘traumatic penumbra’.
On-Line Sampling
The probe was perfused with sterile artificial cerebral spinal fluid (aCSF) (CMA Microdialysis, Sweden) at 2 μL/min using a CMA 107 mini pump (CMA Microdialysis). The outlet tubing of the probe, which normally has a sample vial holder, was adapted to connect to a dual on-line assay system. The vial holder was cut from the tubing with a sharp scalpel and the remaining tubing was then attached to a length of microdialysis tubing using an adaptor built in-house. The electrical impedance and mains leakage of 0.5 m of the microdialysis tubing from the probe outlet to the assay inlet was thoroughly tested by a duly authorised third party and found sufficient to isolate the patient from the assay equipment. As a consequence, this was the shortest length that could be used clinically. Typically, a 1 m length was used to facilitate patient movement, and this resulted in a constant, net delay of 9 mins between sample dialysis and readout.
Dual On-Line Microdialysis Assay
For this work, we developed a novel dual on-line assay system, shown schematically in Figure 1A. Full details are given elsewhere (Jones et al, 2002) but briefly, the assay comprised a custom built valve (Valco Inc., Switzerland) that allowed rapid flow injection of small volumes of dialysate into two separate enzyme bed systems. The system injected glucose and lactate samples in alternation, each at 30 secs intervals. A buffer containing a ferrocene compound was pumped at high flow rates through the enzyme bed system using an HPLC pump (Flux, Switzerland). At each injection, the buffer flow carried the dialysate sample rapidly into the enzyme bed, mediated the enzyme turnover within the bed, and then carried the resulting ferrocinium species to a downstream radial flow electrode (BAS, USA) giving a current peak related to concentration in the dialysate. Data collection was with a Powerlab A/D converter (16 S/P, ADInstruments, NSW, Australia), controlled by a portable computer (Apple G4 Powerbook) running Chart 4.0 software (ADInstruments).
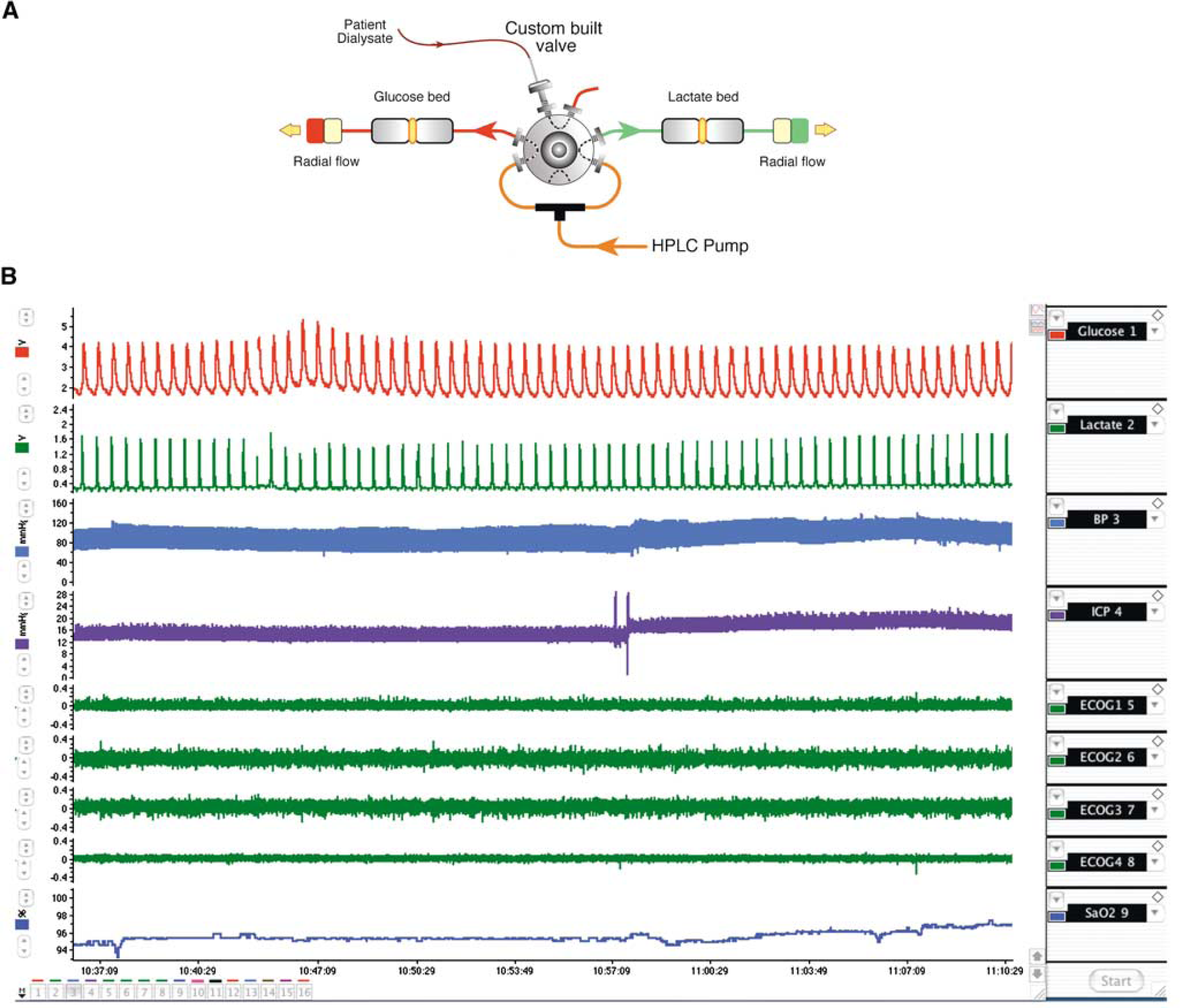
(
The assay was calibrated at least once daily with standards in an appropriate range (typically 50 × 10−6 to 2 × 10−3 mol/dm3). A nonlinear regression of the resulting peak current concentration data was then obtained using the Hill equation
The derived parameters (imax, K m , α) were then used to convert patient current peaks to concentration (Georganopoulou et al, 2000).
Electrocorticography
A linear strip of six stainless-steel electrodes (AdTech, Racine, USA) was placed on the cortex at the conclusion of surgery, as described previously (Strong et al, 2002), and connected to two ADInstruments Dual Channel bioamplifiers in bipolar montage. Importantly, every effort was made to locate the ECoG strip and microdialysis catheter closely adjacent; it was usually possible to site them on the same gyrus. This yielded four data channels from consecutive sites along the strip. Data were logged continuously into the same files as the microdialysis and systemic data. The data were analysed in Copenhagen (MF, ML) independently of the biochemistry, and ECoG events (rapidly developing transient reductions in amplitude of 50% or more, followed by gradual recovery) were classified as nonspreading (N: event occurs at two or more electrodes, with no clear evidence of propagation between them), possibly spreading (P: sequential onset at two adjacent electrodes), or clearly spreading with the characteristics of CSD (C: sequential onset at three or four adjacent electrodes) (Strong et al, 2002).
Duration of Monitoring
The duration of rsMD monitoring is stated for each patient in Table 1.
Patients studied
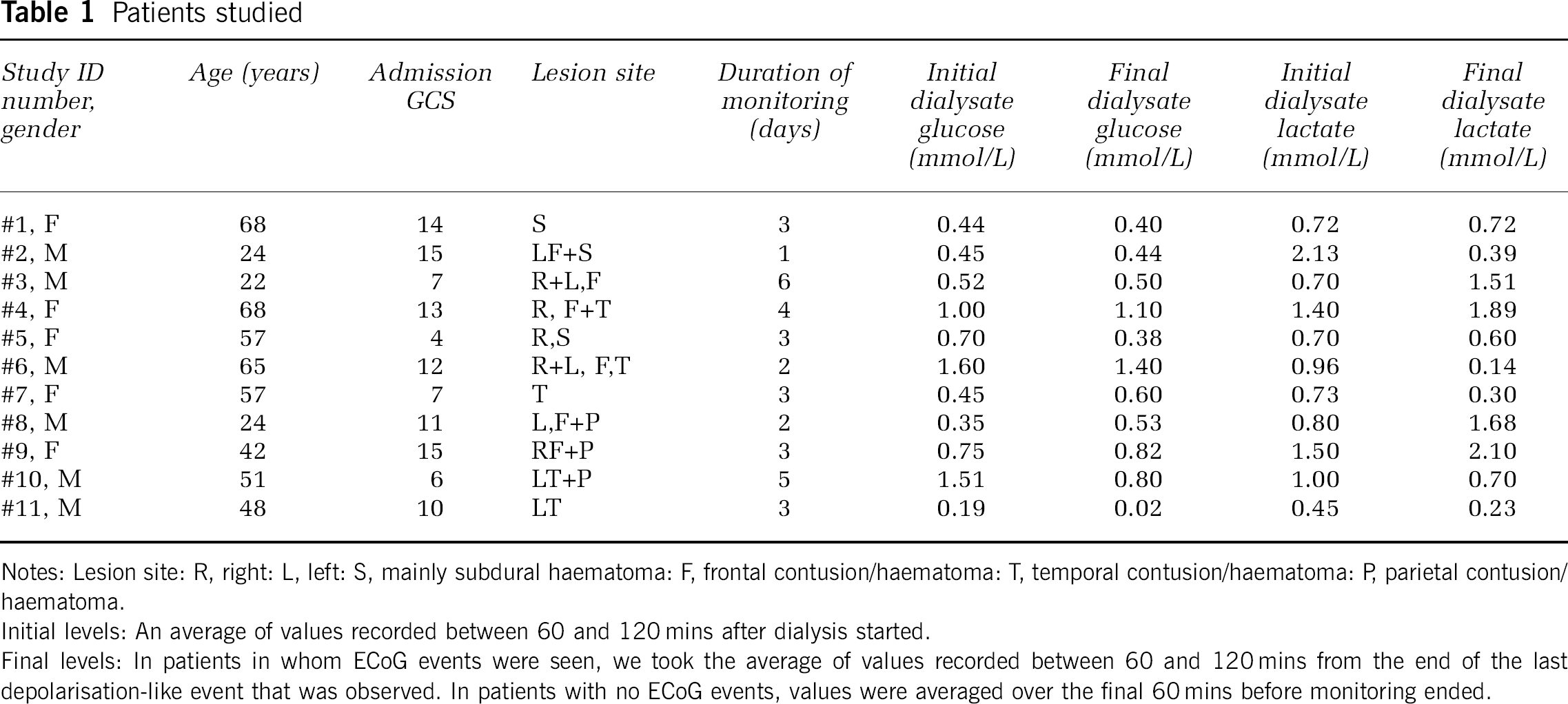
Notes: Lesion site: R, right: L, left: S, mainly subdural haematoma: F, frontal contusion/haematoma: T, temporal contusion/haematoma: P, parietal contusion/haematoma.
Initial levels: An average of values recorded between 60 and 120 mins after dialysis started.
Final levels: In patients in whom ECoG events were seen, we took the average of values recorded between 60 and 120 mins from the end of the last depolarisation-like event that was observed. In patients with no ECoG events, values were averaged over the final 60 mins before monitoring ended.
Assessment of Clinical Outcome
For future use, when a larger data set becomes available, clinical outcome was assessed at 6 to 9 months by telephone interview with the patient and also, on most occasions, with a relative, close acquaintance or care taker, using the extended Glasgow Outcome Scale (eGOS) (Teasdale et al, 1998; Wilson et al, 1998).
Statistical Analysis
The relationships between numbers of metabolite transient events and of transient suppressions of ECoG were examined by linear correlation analysis using Statview (SAS, USA). Values of P less than 0.05 were taken to indicate significance. All values are quoted as mean±s.d.
Results
Eleven patients were monitored for periods of not less than 24 h, and for up to 7 days. Clinical details and global microdialysis variables are summarised in Table 1.
Dialysate Glucose and Lactate
Dialysate concentrations are related to extracellular concentrations by the extraction fraction or in vivo recovery of the particular probe/flow-rate combination. With the exception of nonstandard circumstances, the in vivo recovery cannot be approximated by a measured in vitro recovery. Consequently, all values of glucose and lactate concentrations are reported here in terms of the dialysate concentration obtained by perfusion of aCSF at 2 μL/min. A typical 24 h glucose and lactate trace is shown in Figure 2.
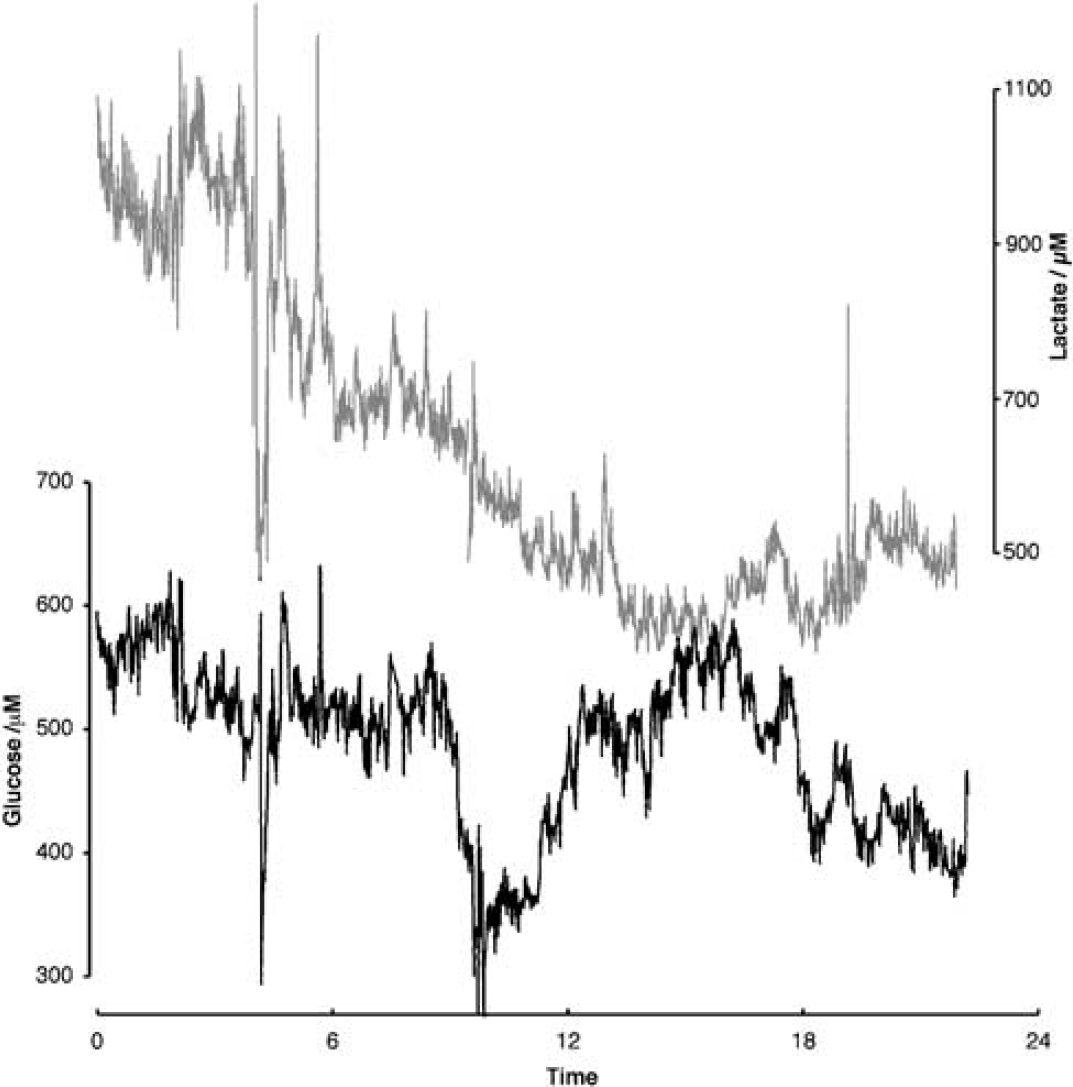
Patient dialysate glucose (black) and lactate (grey) trace recorded over a 24-h period as a continuous session. Both slow and rapid changes in levels are visible, and their significance is considered in the Discussion.
In patients with a minimum of 24 h of dialysis, initial glucose levels (defined in the legend to Table 1) lay between 0.19 mmol/L and 1.6 mmol/L. The final glucose levels ranged from 0.02 to 1.4 mmol/L. We saw similar concentration ranges for the initial dialysate lactate levels, from 0.45 to 2.13 mmol/L. Final lactate levels ranged from 0.14 to 2.1 mmol/L. In a number of patients, lactate levels increased quite considerably during monitoring.
Spontaneous Dynamic Changes in Dialysate Glucose and Lactate
The main focus of this study was the examination of rapid, spontaneously occurring, and dynamic changes in glucose and lactate levels in dialysate. The rapid sampling nature of our assay system allowed us to record a large number of these events in our patient population. We categorised these events as transient increases, transient decreases, combinations, or oscillations (defined, respectively, as ‘Types A…D’). Figure 3 shows typical examples of the four event types we considered. The event types are defined, and the occurrence within our patient groups given, in Table 2. Metabolite transients that had the same time course, percentage amplitude, and polarity were excluded from analysis on the basis that they might reflect fluctuation in recovery resulting from variation in dialysate perfusion rate (Parkin et al, 2003). Overall, the number of glucose events was significantly correlated with the number of lactate events (r2=0.48, P=0.027, n=10).
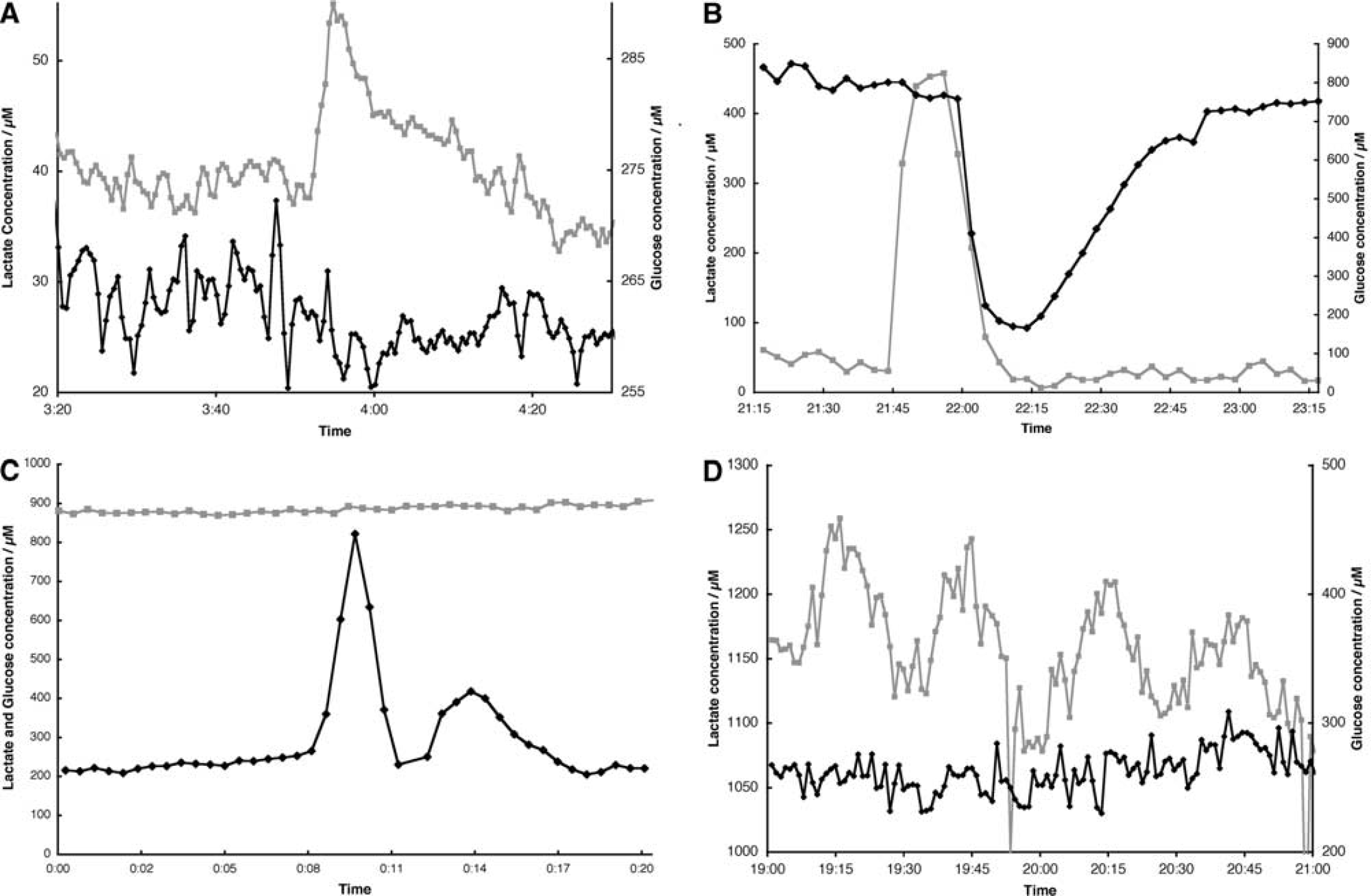
Examples of spontaneous dynamic events seen in dialysate glucose (black, diamonds) and lactate levels (grey, squares). The classification of these events is stated in Table 2. (
Dynamic events
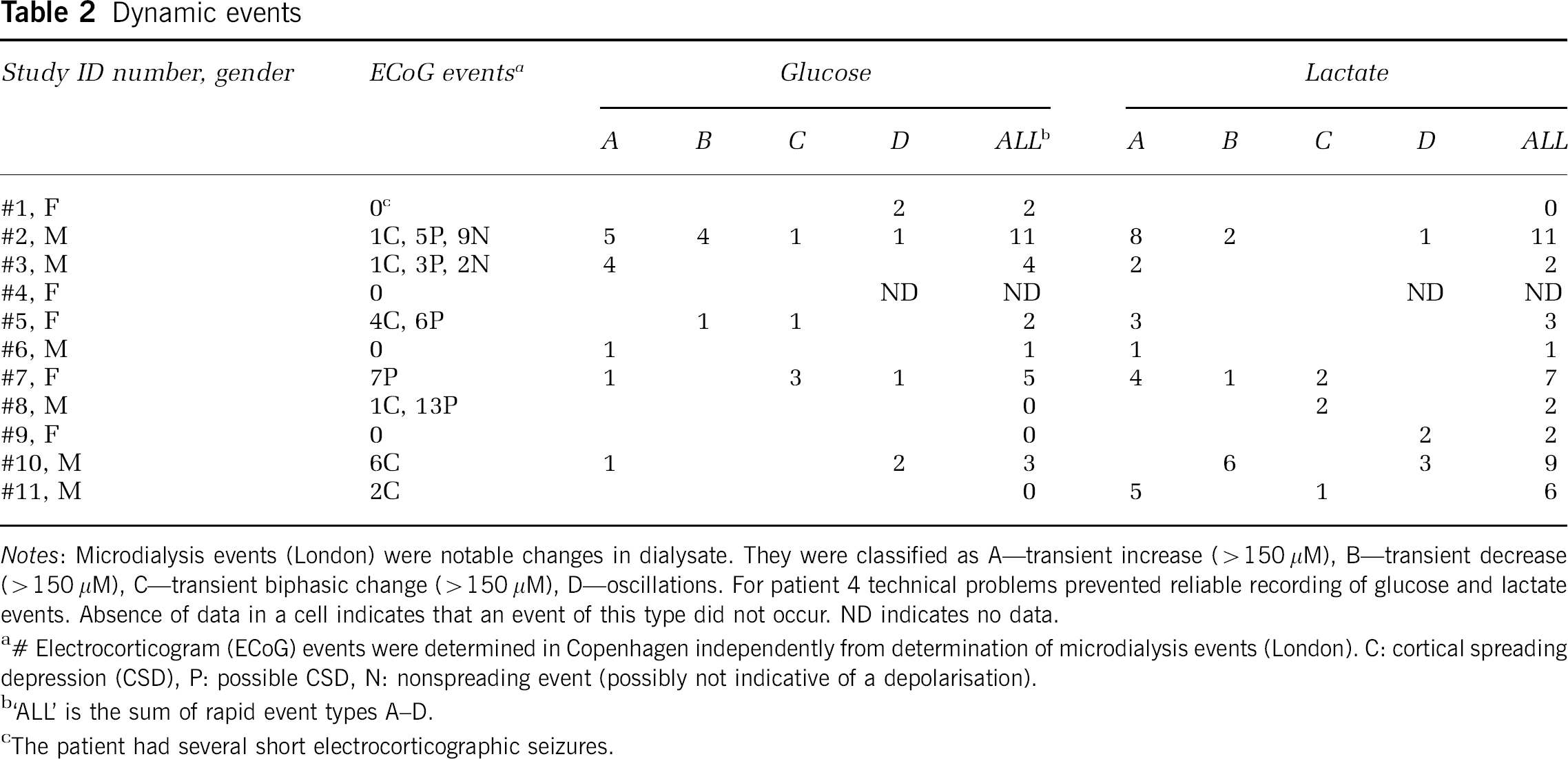
Notes: Microdialysis events (London) were notable changes in dialysate. They were classified as A—transient increase (>150 μM), B—transient decrease (>150 μM), C—transient biphasic change (>150 μM), D—oscillations. For patient 4 technical problems prevented reliable recording of glucose and lactate events. Absence of data in a cell indicates that an event of this type did not occur. ND indicates no data.
# Electrocorticogram (ECoG) events were determined in Copenhagen independently from determination of microdialysis events (London). C: cortical spreading depression (CSD), P: possible CSD, N: nonspreading event (possibly not indicative of a depolarisation).
‘ALL’ is the sum of rapid event types A–D.
The patient had several short electrocorticographic seizures.
In two patients (#2, #11), ECoG suppression events were clearly associated with transient increases in glucose and lactate. One such event is shown in Figure 4 (patient 2). Here, the ECoG suppression (Figure 4A) is associated with dialysate changes in which a glucose increase briefly precedes a lactate increase (Figure 4B). Arterial pressure and intracranial pressure show transient falls at this point. A second event in this patient with similar ECoG features was associated with a glucose transient resembling that illustrated, but without any simultaneous lactate transient.
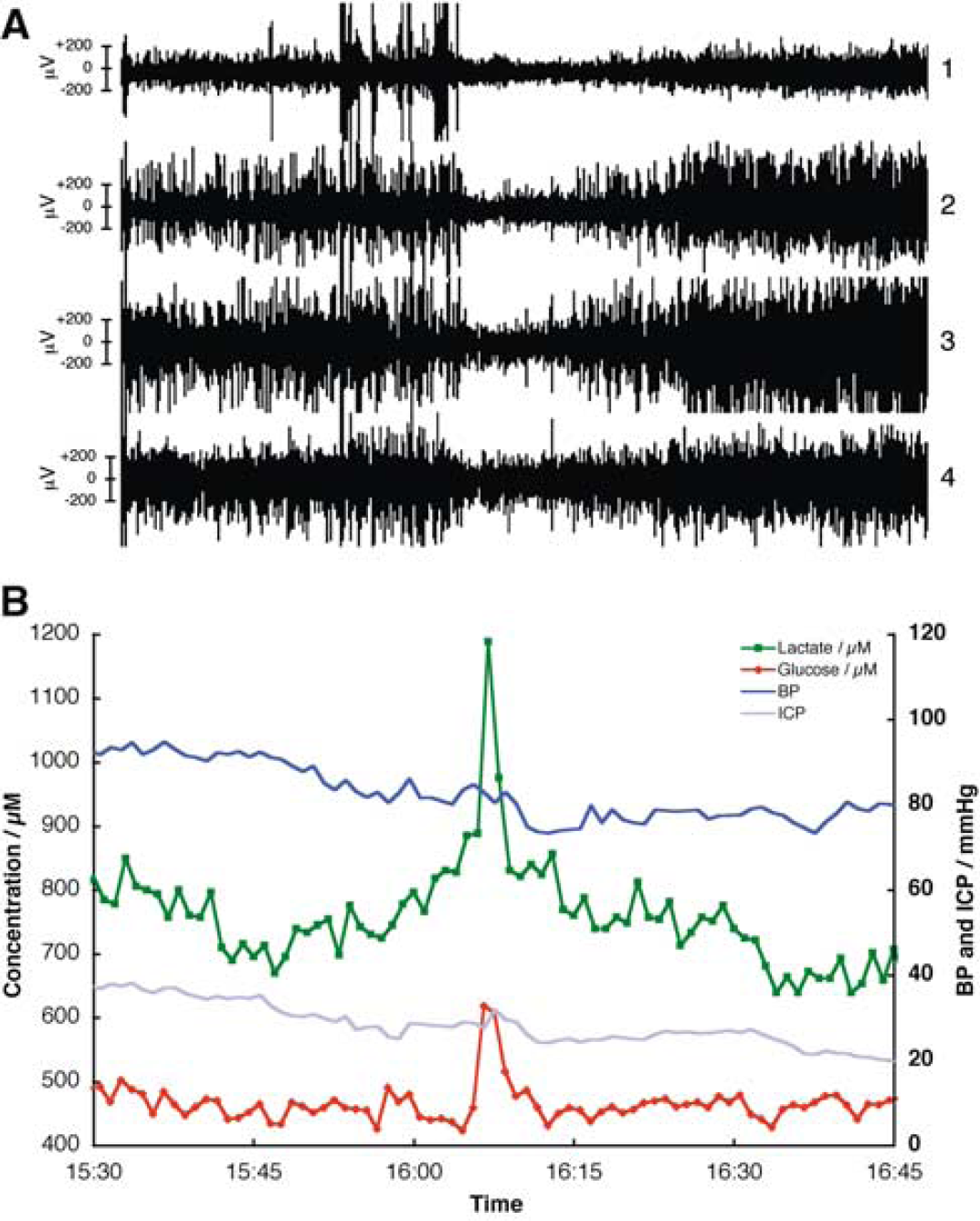
Recording from patient 2, 91 h after trauma and 27 h after surgery for left frontal subdural and intracerebral haematomas. (
Relationship of Changes in Dialysate Content with Transient Cortical Events Detected by Electrocorticography
We performed a specific analysis of the relationship of change in dialysate glucose with a number of depolarisation-like ECoG events. The procedure we followed for this was based on the observation in some patients, late in the monitoring period, of a marked rise in dialysate glucose and partial decrease in lactate, suggesting tissue ‘homogenisation’/infarction, and loss of blood–brain barrier function, leading to partial equilibration of the brain extracellular space with plasma. This finding was sometimes coupled with CT scan observation of delayed, haemorrhagic conversion localised in the perilesion cortex in which the microdialysis catheter had been placed. Therefore, since recurrent depolarisation activity must necessarily cease at some time before full ‘equilibration’, the use of such a ‘final’ value to indicate a possible depletion in cortical extracellular glucose content related to the occurrence of depolarisations is potentially misleading. Instead, in patients in whom spreading depolarisation events were seen, we took as the late dialysate glucose value the average of values recorded between 60 and 120 mins from the end of the last depolarisation-like event that was observed. In patients with no ECoG events, values were averaged over the final 60 mins before monitoring ended. We carried out a linear correlation analysis between microdialysis measurables (levels, and numbers of events determined in London) and the number of ECoG events (determined independently in Denmark). The change in glucose levels as calculated above was very highly significantly correlated with the number of ECoG events that could be confirmed as spreading (type C), with more ECoG events being associated with a greater decrease in dialysate glucose levels (r2=0.76, P=0.0004, n=11). This is shown in Figure 5 and suggests that each spreading event leads on average to a 0.11 mmol/L decrease in glucose. Changes in lactate were not correlated with ECoG events. However, in this restricted data set the number of energy metabolite events (glucose or lactate) were at the borders of significance when compared with all ECoG events and not correlated in the smaller number of cases with confirmed spreading events.
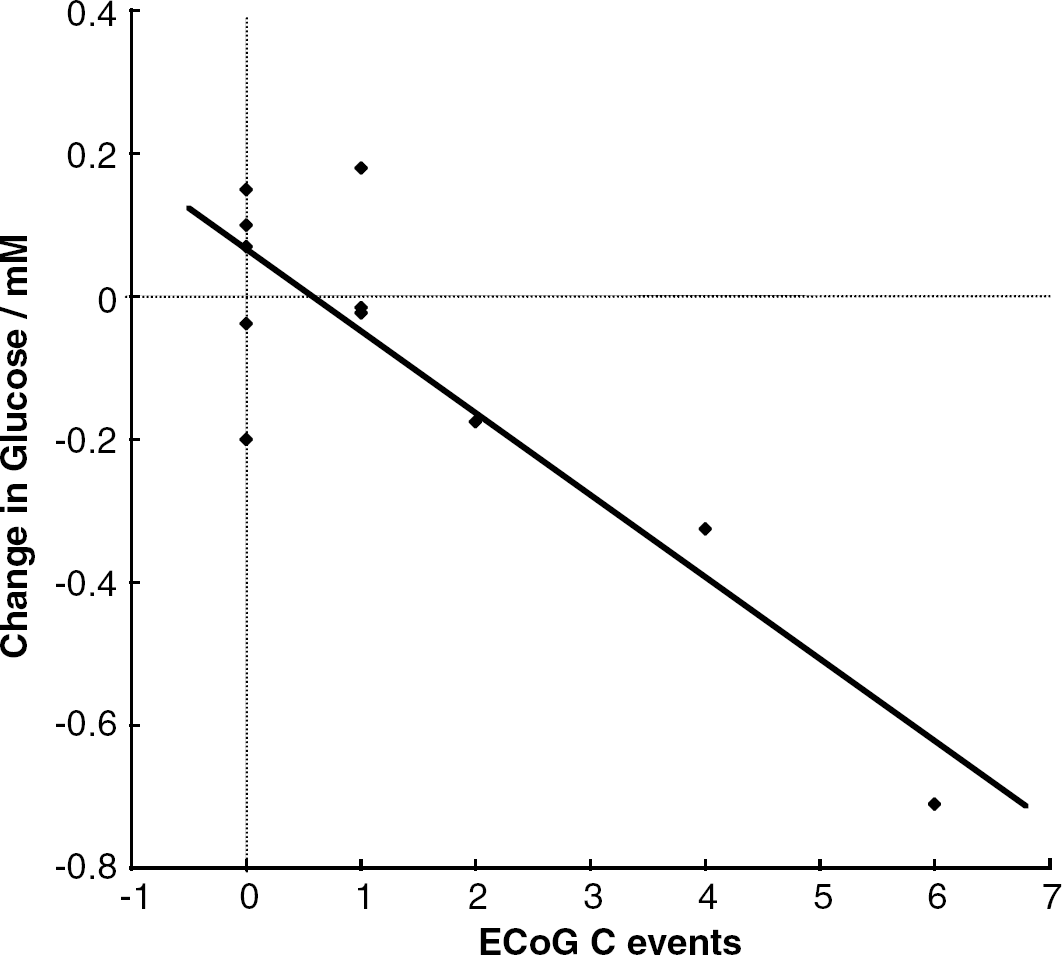
Regression plot comparing the change in dialysate glucose and the number of electrocorticogram (ECoG) events. The value for change in dialysate glucose is the mean value calculated over the second hour of monitoring, less the mean value recorded between 60 and 120 mins after the last depolarisation-like ECoG event, or at the conclusion of monitoring if no events occurred (please also see text). Line represents the equation of linear regression (r2=0.76, P=0.0004, n=11).
Discussion
The introduction of a method for prompt and very frequent sampling of dialysate from the injured brain in patients receiving intensive care offers potential dividends in two related domains—understanding of pathophysiology, and delivery of care. The second of these will require consideration in a separate report. The potential impact of this and similar work on our understanding of the pathophysiology of the injured human cerebral cortex will be discussed, after the technical aspects of the method described here have first been addressed.
Methods
Application of rapid-sampling microdialysis methodology in a neurosciences intensive or high-dependency care unit:
For research purposes, a general picture of current and historic glucose and lactate trends, and of the occurrence of dynamic events was available in real time on the ward. In subsequent processing, peak heights were converted to dialysate concentrations using the calibration information, and peak times were corrected for the 9-min time lag because of the 1 m outlet tubing.
Recovery of extracellular metabolites in the dialysate:
Values are reported here as microdialysate concentration rather than as an extracellular concentration derived using a measure of probe efficiency. The latter depends on (a) dialysis variables such as membrane length and probe length and flow rate (Benveniste, 1989); (b) normal tissue variables such as tortuosity, excluded volume, and local uptake rates (Boutelle and Fillenz, 1996; Bungay et al, 1990; Yang et al, 1998); and (c) damaged tissue variables such as thickness of damage layer near probe and extent of tissue oedema (Bungay, 2001; Khan and Michael, 2003). Thus, it is never appropriate to use an in vitro measurement of probe recovery to determine in vivo extracellular concentrations.
Rapid sampling of microdialysate from peri-lesion cerebral cortex:
Since its first introduction by Ungerstedt and colleagues (Ungerstedt and Pycock, 1974; Tossman et al, 1983), the technique of cerebral microdialysis has yielded important information in a remarkable variety of fields within basic and clinical neuroscience (Hillered et al, 1990; Reinstrup et al, 2000). The many published reports of assays of brain microdialysate from patients with head injury have until now relied on data from catheters located in cerebral white matter, with dialysate aliquots typically integrated over periods of 60 mins but occasionally over 20 mins (Hutchinson et al, 2002), and assayed most commonly for glucose, lactate, pyruvate, glycerol, and glutamate. Results from brain considered normal provide evidence for local production and consumption of lactate (Abi-Saab et al, 2002) and vary, for example, in their support for a relationship between dialysate lactate (considered in isolation) and outcome in human TBI (Goodman et al, 1999; Vespa et al, 2003).
Knowledge of the dynamic nature of perfusion and metabolism in the cerebral cortex in experimental models of traumatic and ischemic brain injury (Katayama et al, 1990; Strong et al, 1996) led us to develop a variant of our existing method for frequent automated assay of glucose and lactate in a continuous dialysate stream, so as to make it suitable for clinical use. We have used this new system to analyse a continuous clinical dialysate stream drawn from a catheter with 10 mm membrane length, inserted obliquely under direct vision to some 7 to 8 mm depth, and hence located so far as possible within (1) the cerebral cortex and also (2) still viable but ‘at-risk’ tissue in the neighbourhood of a focal contusion. This is in contrast to most early clinical microdialysis studies where probes were placed in the ‘standard right frontal burrhole site’ regardless of the location of the traumatic lesion. Recent findings from comparison of the effects of ischaemia on superficial brain and underlying white matter suggest that a wider dynamic range of normal and abnormal values becomes available from the cortex, with faster responses (Dohmen et al, 2001). As a component of a clinical research strategy, we intend that the data emerging from metabolic monitoring should increasingly be set beside simultaneously acquired data on the electrophysiological state of the cortex, important clinical variables such as cerebral perfusion pressure and arterial oxygen saturation, and clinical outcome data.
Transient changes in dialysate glucose and lactate
An examination of the time series data in Figure 2 indicates changes in dialysate levels occurring in three time domains:
There are slow global changes over periods of several hours—these are the changes reported by classical microdialysis.
There are transient increases, decreases and oscillations with time scale of 1 min to approximately an hour. The largest of these changes are of the same magnitude as the slow global changes.
There is variation between consecutive measurement points. This appears as ‘grass’ in Figure 2, and might be thought of as noise. Further study of the nature of this category of variation is required.
These three classes of information present a problem of data analysis. Simple averaging over time, either by explicit combination of our data points or by pooling dialysate into longer sampling periods, effectively filters out all but slow global changes (class 1). In this paper, we seek to describe for the first time, in the human brain, class 2 changes, and report the existence of class 3 changes.
We have counted class 2 events according to type (A–D in Table 2). Without other data (such as the fluorescence transients in the accompanying paper) to align events we feel that it is not appropriate to average these events together as they are of different magnitudes and durations. It is our hope that in future studies ECoG will fulfil this alignment role.
It is clear from the data in Table 2 and the illustrations in Figure 3 that several patterns of transient change in glucose and lactate occur. Although we are at present unable to offer satisfactory proposals for the mechanisms of the various events we report and classify as ‘A–D’ here, the detection of oscillations in both variables suggests the presence—perhaps partially impaired—of homeostatic systems or unstable equilibria. The fact that oscillations occurred only in one channel at a given time (either in glucose or lactate but never simultaneously) supports an interpretation as true findings rather than as artefact relating to fluctuations in microdialysis perfusion rate (Parkin et al, 2003). It also suggests that the concentrations of glucose and lactate may be independently regulated—at least during periods when oscillations are observed.
Pathophysiology and Evolution of Acute Brain Injury
The system that we have applied for experimental and clinical use has been evolved in order to progress our understanding of the nature of depolarisation phenomena in the normal and injured brain. Although the occurrence and propagation of spontaneous depolarisation-like events in the injured human brain has been clearly documented (Mayevsky et al, 1996; Strong et al, 2002), the pattern of metabolic disturbance and hence the capacity of any given event to increase pathology in a patient with acute brain injury is not known. In short, is a given ECoG event likely to increase damage and prejudice outcome? Our goal has therefore been to explore the use of rsMD as a means of examining this issue in both experimental and clinical settings.
Understanding of the potential for additional damage from spontaneous, transient depolarisation events in the injured brain derives first from the original demonstration by Leão, in rabbits, of a wave of depolarisation of the cortex spreading at a rate of some 3 mm/min from a selected site of electrical stimulation of the cortex, manifest in his work as a period of depression of amplitude of ECoG activity—Leão's ‘spreading depression’ (Leão, 1944). Second, studies of the effects of MCAO in several animal models of stroke have repeatedly yielded evidence of spontaneous events with electrophysiological features similar to those of CSD, but, probably unlike CSD, with proven capacity to augment ischemic damage—PIDs (Back et al, 1996; Busch et al, 1996; Takano et al, 1996). The potential contribution of depolarisation events spreading in the injured cerebral cortex to adverse outcome from acute brain injury in humans is discussed in a review (Strong and Dardis, 2005).
The proposed mechanism of damage in focal cerebral cortical ischaemia—amounting to maturation or extension of infarction into the penumbra—has been reviewed by Hossmann (1996). Nedergaard and Astrup (1986) emphasised the likelihood of tissue glycopenia in association with PIDs, and application of our method to monitor glucose and lactate concentrations in dialysate from peri-infarct cortex after permanent (experimental) MCAO has demonstrated transient, coupled reductions in glucose and increases in lactate linked to PIDs, and occurring on a background of progressive depletion of the extracellular glucose pool (Hopwood et al, in press). Dependence of the ischemic brain on anaerobic glycolysis, and the much less efficient utilisation of glucose which is inherent in this, renders such areas vulnerable to reduced glucose availability. Furthermore, the frequency of PIDs increases with relatively mild reductions in plasma glucose (Hopwood et al, in press; Strong et al, 2000), exacerbating the challenge to vulnerable brain. In the case of spreading depression occurring in the normally perfused brain, no damage occurs (Nedergaard and Hansen, 1988)—unlike the situation in the ischemic brain.
The present project was undertaken in parallel with another, related clinical study designed to detect evidence for the occurrence of Leão's CSD or related phenomena in the injured human brain, using subdural ECoG strips. The results of that study (Strong et al, 2002) confirm unequivocally that depolarisation events do indeed occur in the injured human brain. Although the nature of the pathogenic effect of these events on the evolution of brain injury remains to be confirmed, the issue clearly requires study, and it follows that, where feasible, physiological and chemical methods to monitor the injured brain should now possess sufficient sensitivity and temporal resolution to allow analysis of transient changes in the injured brain—of whatever origin.
How far does the introduction of rsMD take us towards this goal? The glucose and lactate transients linked to PIDs in our experimental study had a 5 mins time-to-maximum, with the lactate increases being some two times the magnitude of the glucose decreases. A probe with a shorter membrane (2 mm) placed normal to the cortical surface was used in the experimental studies, in contrast to the 10 mm length of the probe used clinically and placed obliquely into the cortex (and most probably subcortical white matter). We now suspect that this has compromised our ability to detect with rsMD all transient events propagating through the cortex in patients, as a typical CSD would take up to 4 mins to pass along the membrane length, if propagation were along the membrane axis. The long membrane thus mixes contributions from cortex (a) ahead of the wave, (b) at the height of depolarisation, and (c) in the hyperpolarisation phase in the same dialysis sample, and probably from subcortical white matter, all of these factors reducing the magnitude of the measured neurochemical change. This would affect the smaller glucose response to a greater extent than lactate, pushing the changes below our limit of detection. This suggestion is supported by the finding that in this clinical study glucose and lactate events were usually not simultaneous, whereas they were in our experimental study.
For reasons given above, we believe that rsMD, as currently implemented clinically, fails to detect many ECoG-defined depolarisation events. However, it seems likely that rsMD detected depolarisation events in two patients. As illustration, marked, time-coupled increases in glucose and lactate occurred in Patient 2 (Figure 4), and coincided with an episode of ECoG amplitude suppression (Figure 4A). The metabolite transients were well above the class 3 background variation (Figure 4B). A second instance of ECoG suppression in this patient (not shown) was accompanied by transient increases in ICP and in glucose, but with little change in lactate. The events in this patient are compatible with increased delivery of glucose and metabolism to lactate, associated with ‘adequate’ or normal recruitment of perfusion and glucose availability in response to the transient but intense activation stimulus of CSD; the patterns are not those of a PID, in which dialysate glucose decreases (Hopwood et al, in press), although later deterioration may result in appearance of ‘PID-like’ patterns in perfusion and NADH fluorescence (Mayevsky et al, 1996).
Significance of Changes in Dialysate Glucose
The principal finding to emerge from this work is the very striking relationship of progressive glucose depletion with the number of confirmed CSD-like ECoG events (Figure 5). That this relationship does not extend to all ECoG events requires explanation. Our current operational definition of a confirmed ECoG event (C) specifies sequential detection at three adjacent electrode pairs. There is thus a higher probability that such a CSD wave will affect the rsMD probe than for a wave seen at fewer electrode sites (‘N’ and ‘P’ events). Moreover, since perilesion tissue is highly heterogeneous (Tomlinson et al, 1993), it is indeed possible that some events are highly localized, and may not propagate between the dialysis catheter and all—or even any—of the electrodes.
A decrease in dialysate glucose has been associated (Langemann et al, 1995) and definitively correlated with poor clinical outcome from severe head injury (Vespa et al, 2003), but in most of their patients, the latter group was unable to identify causes for the glucose reductions, since their values for lactate:pyruvate ratio did not suggest ischaemia as a cause of increased (anaerobic) glucose utilisation. Our recent demonstration of ECoG suppressions suggesting CSD or PID events in patients with head injury (Strong et al, 2002), the association of transient reductions in dialysate glucose with PIDs in our MCAO experiments, and the associated progressive decline in ‘inter-transient’ dialysate glucose (Hopwood et al, in press) suggest to us that recurrent depolarisation events in their patients could account for the findings of Vespa and his colleagues. The finding in this present study of a very robust relationship between decline in dialysate glucose and a number of confirmed, spreading depolarization-like ECoG events strongly supports such a hypothesis. The same reasoning also points by inference to the possibility that recurrent transient ECoG suppressions prejudice outcome, as has been discussed above in relation to PIDs in experimental stroke. We did not find a relationship between ECoG event burden and any overall increase in lactate; this would be in keeping with the absence of evidence of ‘ischaemia’ in Vespa and colleagues' data.
The same group describes hyperglycolysis in association with experimental (Yoshino et al, 1991) and clinical (Bergsneider et al, 1997) brain injury. Our present demonstration of confirmed ECoG depolarisation events and glucose depletion, together with the association of increased glucose utilisation with PIDs reported earlier by Nedergaard and Astrup (1986), suggests that recurrent depolarisation activity may be one factor accounting for hyperglycolysis in acute brain injury.
The outcome data collected in this limited series (but not described in detail here, and intended for future use) are at present insufficient to support or exclude an association of adverse outcome with a number of ECoG events (but do confirm, as is very well known, a strong association of increasing age with adverse outcome).
Could possible changes in glucose transport capacity at the blood–brain barrier influence these considerations? Existing data on the relationship of transport capacity for glucose with cerebral perfusion suggest the likelihood of depletion of brain glucose—in whatever cortical compartment—under ischemic conditions, and particularly during any recovery from a depolarisation event; such conditions now seem very likely to exist in the peri-contusion cortex. Thus Betz et al (1973) showed a linear relationship of unidirectional glucose transport into the brain (dogs) with perfusion, such that its rate would decrease to some 60 to 80 μmol/100 g min, or lower, at the levels of ischaemia likely to prevail in the present conditions (20 to 30 ml/100 g min). Nedergaard et al (1986) recorded values for local cerebral glucose utilisation (LCGU) of some 200 μmol/100 g min in peri-infarct zones in rats subjected to MCAO; timing of LCGU measurements did not necessarily coincide with periods of recovery from depolarisation events, implying the likelihood of even greater depletion of cortical glucose by depolarisation events. In view of the dependence of inward glucose transport on perfusion, it is unlikely that any upregulation of glucose transport capacity, were it to occur, would significantly influence this concept. Furthermore, on the timescale of the transient depolarisation events (mins) it is not likely that transporter capacity would change. On the longer timescale (3 h) of, for example, Figure 6 in Hopwood et al (in press), any change in transporter capacity would be likely to be an increase. This would in turn increase glucose delivery to the brain and hence attenuate the possible decrease in brain glucose to that seen in that figure.
In the light of these findings and discussion, we conclude, first, that the introduction of rapid-sampling microdialysis for monitoring glucose and lactate in patients with severe head injury represents an important new clinical research tool. Second, these data, together with the accompanying experimental report (Hopwood et al, in press) and existing published reports, provide support for the suggestion that where progressive decline of brain extracellular glucose concentration occurs in marginally injured brain, this may be explained—and also accelerated—by the occurrence of spontaneous depolarisations. There is now a clear need for a detailed examination of the characteristics of spontaneous depolarisation events in the injured brain and of their relationship with outcome, in a substantial number of patients. The Co-operative Studying Brain Injury Depolarisations (‘COSBID’, www.cosbid.org) seeks to address this need.
Footnotes
Acknowledgements
The authors thank the Wellcome Trust (SEH) and EPSRC (MCP) for their support, together with the Golden (now Rosetrees) Trust, Roche Research Trust, Glaxo SmithKline, and King's College London. The authors are especially grateful to the nursing and medical staff of the Intensive Care Unit and neurosurgical team, and to the Medical Equipment and Management Service at King's College Hospital. The authors thank ADInstruments (Australia) for construction of interface hardware.