Abstract
Brain levels of glucose and lactate in the extracellular fluid (ECF), which reflects the environment to which neurons are exposed, have never been studied in humans under conditions of varying glycemia. The authors used intracerebral microdialysis in conscious human subjects undergoing electro-physiologic evaluation for medically intractable epilepsy and measured ECF levels of glucose and lactate under basal conditions and during a hyperglycemia–hypoglycemia clamp study. Only measurements from nonepileptogenic areas were included. Under basal conditions, the authors found the metabolic milieu in the brain to be strikingly different from that in the circulation. In contrast to plasma, lactate levels in brain ECF were threefold higher than glucose. Results from complementary studies in rats were consistent with the human data. During the hyperglycemia–hypoglycemia clamp study the relationship between plasma and brain ECF levels of glucose remained similar, but changes in brain ECF glucose lagged approximately 30 minutes behind changes in plasma. The data demonstrate that the brain is exposed to substantially lower levels of glucose and higher levels of lactate than those in plasma; moreover, the brain appears to be a site of significant anaerobic glycolysis, raising the possibility that glucose-derived lactate is an important fuel for the brain.
The central nervous system is an obligate glucose consumer that depends almost entirely on the supply of glucose from the systemic circulation to maintain normal function (Edvinsson et al., 1993). However, relatively little is known about the metabolic fate of glucose in the brain once it has passed from the systemic circulation, across the blood–brain barrier into the central nervous system. It has been suggested that astrocytes are anatomically positioned to sense neuronal activity and uptake glucose because the endfeet processes of astrocytes surround both capillaries and neurons in the brain (Golgi, 1886; Sala, 1891). The hypothesis advanced by Pellerin and Magistretti (Pellerin and Magistretti, 1994) posits that during brain activation, neurons release glutamate, which is removed from the synaptic cleft by astrocytes. The energy needed for this process is derived from glycolysis—i.e., increased glucose use and lactate production—in astrocytes, which in turn, release lactate in the extracellular fluid (ECF) to be used by neurons (Tsacopoulos and Magistretti, 1996). Thus, lactate, produced and secreted by glia, may provide an important source of fuel for neuronal energy metabolism. However, much of the data implicating metabolic coupling between astrocytes and neurons originate from in vitro studies highlighting the need for investigating this issue in vivo in both animals and humans.
Several in vivo methods have been used to measure cerebral glucose metabolism in humans. These methods include the Fick principle, which measures tissue blood flow and arteriovenous differences across the brain, to calculate total brain glucose metabolism. More recently, brain imaging techniques such as positron emission tomography, functional magnetic resonance imaging, and magnetic resonance spectroscopy (MRS) have played a major role in advancing our understanding of human brain metabolism in vivo. Microdialysis is a powerful complementary in vivo technique that enables more direct sampling of the brain microenvironment, beyond the constraints imposed by the blood–brain barrier. Although microdialysis cannot specifically monitor the neurochemical changes within the synapse, it provides unique information about the local microenvironment surrounding these synapses. Hence, microdialysis has become the preferred in vivo method to study chemical changes in brain ECF in animals, and the focus of this work has been predominantly on neurotransmission. In human brain, microdialysis has been used infrequently and those studies have been conducted in extreme conditions of head injury or during intraoperative neurosurgical procedures (During, 1991; Landolt and Langemann, 1996; Hamani et al., 1997; Hillered and Persson, 1999a; Hillered and Persson, 1999b). Although several studies measured dialysate levels of glucose and lactate in humans (During et al., 1994; Langemann et al., 1995; Goodman et al., 1996, 1999; Enblad et al., 1996; Zauner et al., 1997; Mendelowitsch et al., 1998; Sarrafzadeh et al., 1998; Valadka et al., 1998; Berger et al., 1999; Hutchinson et al., 1999; Menzel et al., 1999), there has been no study to date that used established microdialysis techniques that allow determination of true basal ECF levels of metabolites [e.g., the zero-flow (Jacobson et al., 1985) or the no-net-flux (Lonnroth et al., 1987) methods] in awake humans. In addition, there have been no studies in humans that examined the effects of varying glycemia on ECF concentrations of glucose and lactate.
We have adapted a microdialysis system for the in vivo study of brain in conscious human subjects (During, 1991, 1992; During et al., 1994; During and Spencer, 1993). Studies were performed in patients undergoing neurosurgical assessment for medically intractable complex partial seizure disorder. For this purpose, depth electrodes were stereotactically positioned intracerebrally; microdialysis catheters were coupled to these electrodes. During the postoperative period, we carried out studies with the specific goal of directly measuring levels of glucose and lactate in brain ECF of conscious human subjects at baseline and under conditions of varying glycemia. We focused on data generated from brain regions considered to be free of disease. As a means of validation, we complemented our findings in patients with similarly devised studies in conscious rats.
MATERIALS AND METHODS
Human studies
Zero-flow studies. Six patients were studied, with one patient having two microdialysis probes in nonepileptic areas. Three probes were located in the hippocampus, two in Heschel's gyrus, one in the thalamus, and one in the parietal cortex. To estimate basal levels of glucose and lactate, we used the zero-flow technique (Jacobson et al., 1985). Sterile artificial ECF was infused at a rate of 2.5 μL/min for 2 hours to achieve steady state. Fifteen microliters of dialysate were collected and then the flow was decreased to 1.5 μL/min, 0.75 μL/min and then 0.25 μL/min, each time allowing a 1-hour period for equilibration between changes in flow rates. Dialysates were stored over dry ice, then transported to a −70°C freezer where they were kept until they were analyzed. ECF levels of glucose and lactate at each flow level were measured as described in the next section. Basal levels were determined using regression analysis to a flow of zero (steady-state) as described by Jacobson and colleagues (Jacobson et al., 1985).
Hyperglycemic–hypoglycemic clamp studies. These studies were conducted in six patients who had hippocampal microdialysis probes. An intravenous line was inserted into a dorsal hand vein and the hand was put inside a heated box that was maintained at approximately 60°C to 65 °C to ensure arterialization of venous blood (McGuire et al., 1976). The catheter was kept patent with a small volume of infused saline. A second intravenous line was inserted in the antecubital space for infusion of glucose and insulin. After an initial baseline period (60 minutes), the procedure involved a hyperglycemic phase (90 minutes) followed by glucose descent to a hypoglycemic phase (60 minutes). The hyperglycemic phase of the protocol was achieved by infusing 20% glucose to raise plasma glucose from baseline euglycemic values to approximately 200 mg/dL (approximately 11 mmol/L). At the end of this phase, a primed continuous infusion of insulin (human regular) was then commenced at a rate of 3 mU/kg min−1 and plasma glucose was lowered during a period of 60 minutes to a value of 50 mg/dL (approximately 2.8 mmol/L). Plasma glucose was maintained at this hypoglycemic plateau, after which the insulin infusion ceased and euglycemia was restored. Brain dialysate was collected every 10 minutes for measurement of glucose and lactate throughout the baseline, hyperglycemic, and hypoglycemic phases of study, taking into account the 10-minute delay corresponding to the 25-μL dead space of the microdialysis tubing.
Animal studies
Microdialysis materials
Microdialysis catheters used in humans were attached to depth electrodes. In brief, the catheters comprised vitreous silica inlet and outlet tubings inserted into a hollow fiber of dialysis membrane (300 μm o.d., 5 KD, Cuprophan, Enka Glanstoff, Germany), the distal end of which was plugged with ultraviolet adhesive (Locktite, Stonington, CT, U.S.A.). The proximal end of the dialysis fiber was sealed with cyanoacrylate (Locktite Medical Adhesive). The dialysis membrane length in this system was 10 mm for the zero-flow studies and 30 mm for the clamp studies. The dialysis catheter was attached to the depth electrode (Spencer probe; Adtech Medical Instrument Corp., Racine, WI, U.S.A.) using adhesive. In rats, we used the commercially available microdialysis catheters (CMA 12, Carnegie, MA, U.S.A.). These probes have an active dialysis membrane length of 4 mm. In rat and human studies, dialysis catheters were perfused with precision pumps using a sterilized and pyrogen-free artificial ECF (147 mmol/L NaCl, 3 mmol/L KCl, 1.0 mmol/L MgCl2, 1.2 mmol/L CaCl2, 200 μmol/L ascorbate and a sodium phosphate buffer to pH 7.4).
Determinations and analyses
Plasma glucose and lactate were measured using the Beckman Glucose II Analyser (Beckman, Fullerton, CA, U.S.A.), and a microfluorometric enzyme linked assay, respectively (Maggs et al., 1995). Dialysate glucose and lactate were measured by HPLC in the initial four human studies but in subsequent studies and for animal studies were measured microfluorometrically (Maggs et al., 1995). The three methods used to measure glucose (glucose oxidase, hexokinase, and HPLC) correlate extremely well (r > 0.9). Plasma insulin, growth hormone, glucagon, and cortisol were measured by established radioimmunoassay. Plasma catecholamines were measured by a radioenzymatic assay (Amersham, Arlington Heights, IL, U.S.A.).
RESULTS
In human subjects, under basal fasting conditions before the sequential hyperglycemic–hypoglycemic clamp study (n = 6), ambient plasma levels of glucose and lactate were 5.64 ± 0.39 mmol/L and 0.96 ± 0.20 mmol/L, respectively (mean ± SD). In brain dialysate, glucose and lactate levels were 0.82 ± 0.27 mmol/L and 1.38 ± 1.2 mmol/L, respectively, at a perfusate flow rate of 2.5 μL/min. Therefore, the ratios of lactate to glucose were 0.17 ± 0.03 and 1.68 ± 0.5 in plasma and dialysate, respectively (Fig. 1). Taking into consideration the recovery rate of our microdialysis probes, which is estimated to be 25% to 40%, it becomes apparent that brain ECF glucose levels are substantially lower than ambient plasma levels, whereas ECF lactate levels are markedly higher. To further evaluate this issue in six additional subjects, we used the zero flow technique (Jacobson et al., 1985), which estimates basal ECF levels of these substrates. Glucose levels were calculated to be 1.57 ± 0.76 mmol/L, whereas lactate levels were 5.10 ± 1.37 mmol/L (Fig. 2) with a recovery rate of 27% and 18%, respectively, at a flow of 2.5 μL/min (two-tailed t test, P = 0.34). Similarly, in complementary microdialysis studies conducted in awake rats, estimated ECF levels of glucose were significantly lower than plasma levels (1.66 ± 0.32 mmol/L versus 7.84 ± 0.37 mmol/L), whereas the reverse was true for lactate (2.70 ± 0.86 mmol/L versus 1.05 ± 0.24 mmol/L), and the molar content for lactate in the ECF was more than 50% higher than that of glucose (P < 0.05) (Fig. 3). This disparity, which was seen in the hippocampus and the frontal cortex, suggests that lactate is locally produced from glucose in brain ECF and, hence, might play a role as an important fuel source in the brain.
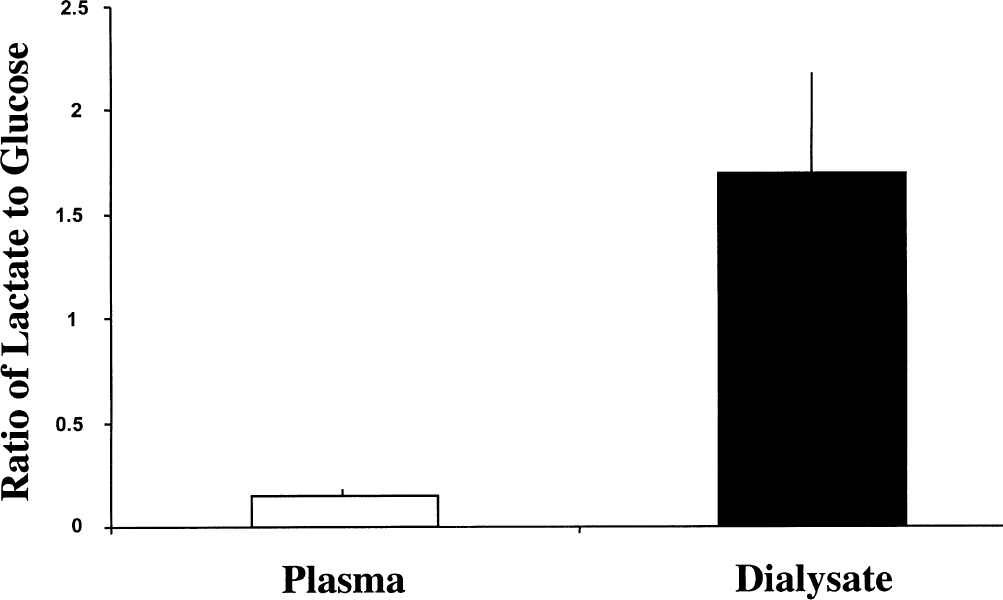
Marked difference in the ratio of lactate to glucose in plasma and dialysate from hippocampal microdialysis probes in six fasting human subjects with complex partial seizure disorder. Dialysate values were not corrected for recovery. Values are represented as mean ± SD.
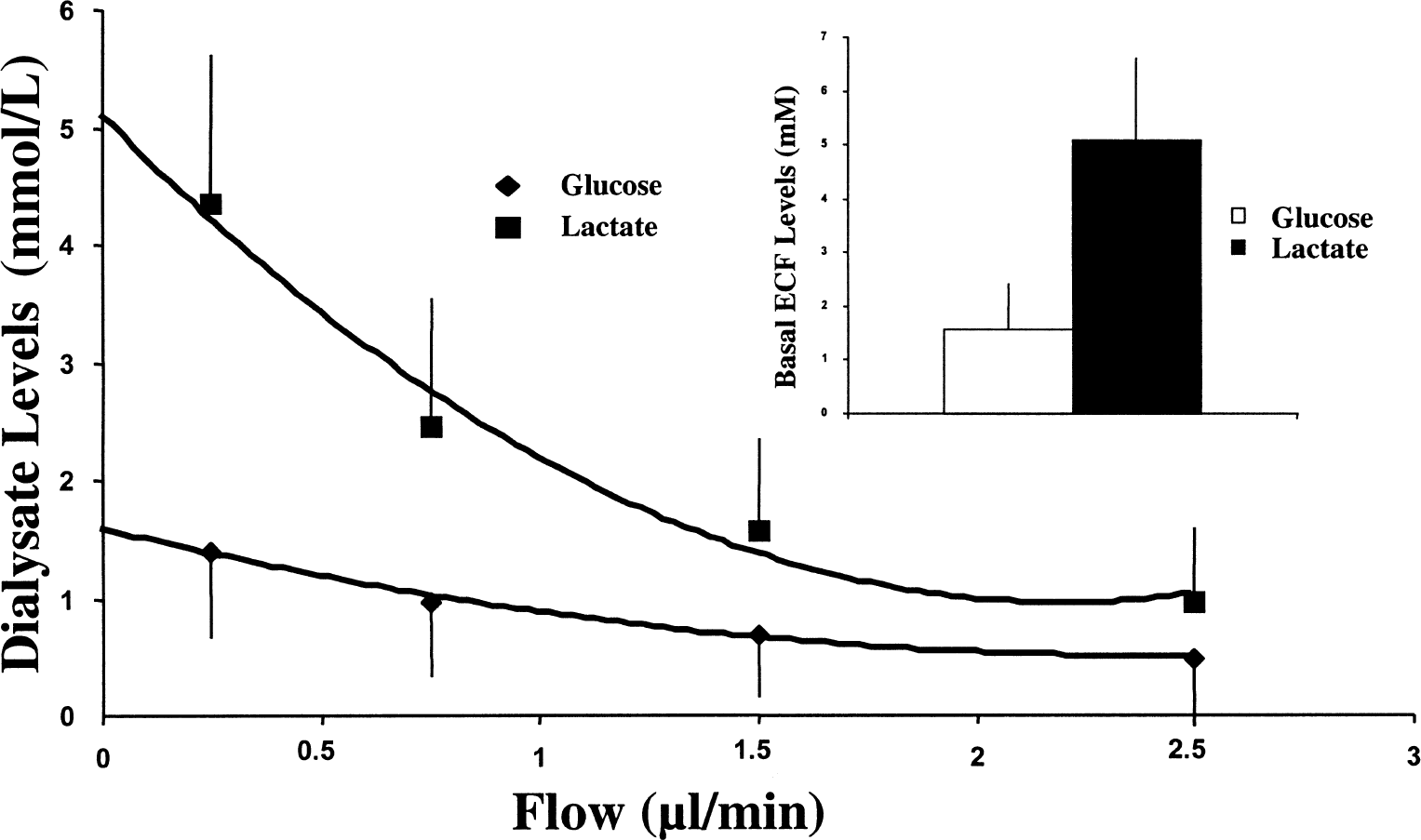
Dialysate levels of glucose and lactate measured using the zero-flow technique in six conscious human subjects with complex partial seizure disorder. Basal levels of each substrate at steady state (zero flow) were calculated using a second-degree polynomial curve fit and determining the Y axis intercept. The calculated basal extracellular fluid (ECF) levels are plotted separately (inset). Values are represented as mean ± SD.
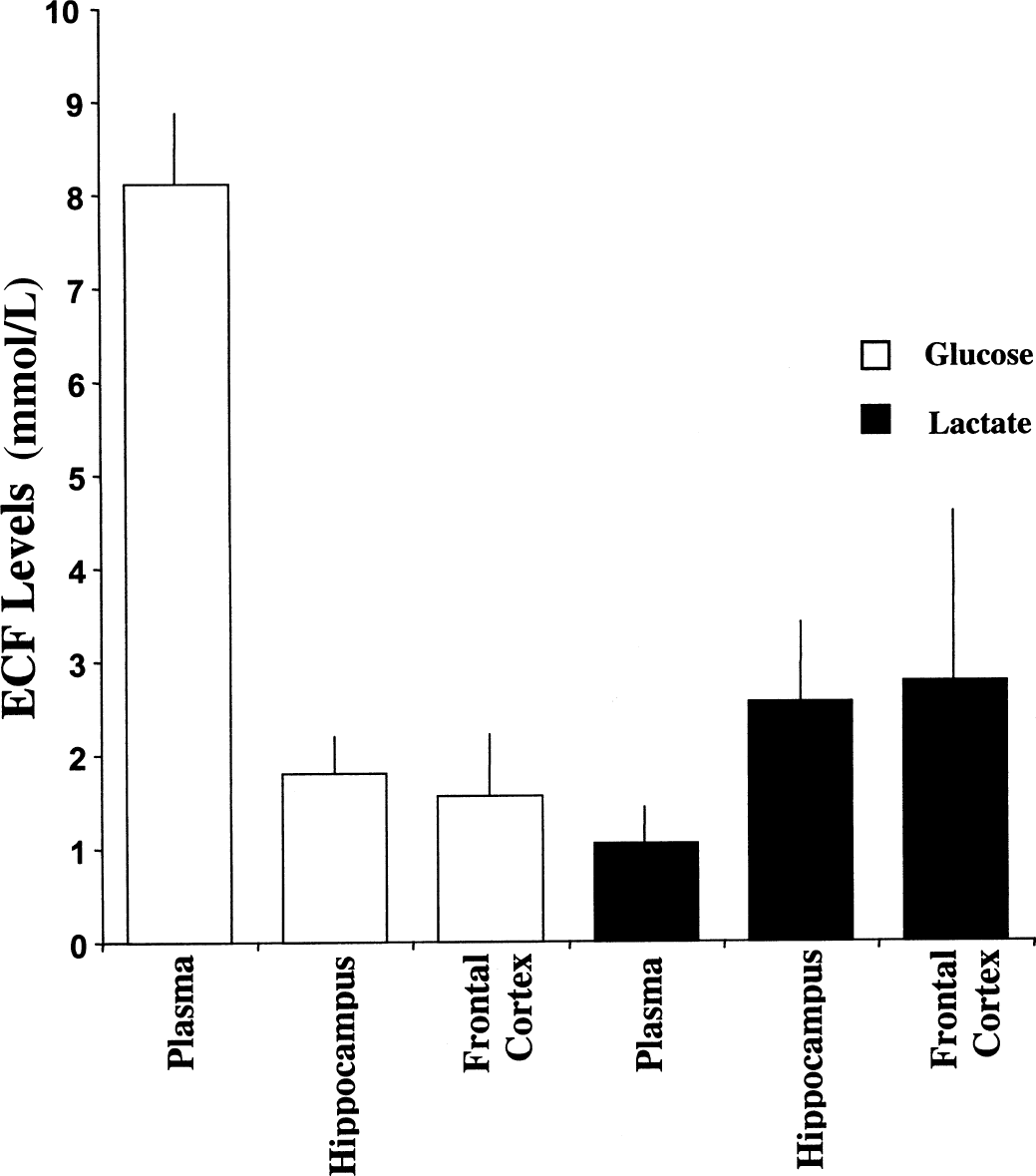
Estimated basal extracellular fluid (ECF) (from in vitro recovery) and plasma levels of glucose and lactate from the hippocampus (n = 7) and the frontal cortex (n = 12) of awake, freely moving rats. Values are represented as mean ± SD.
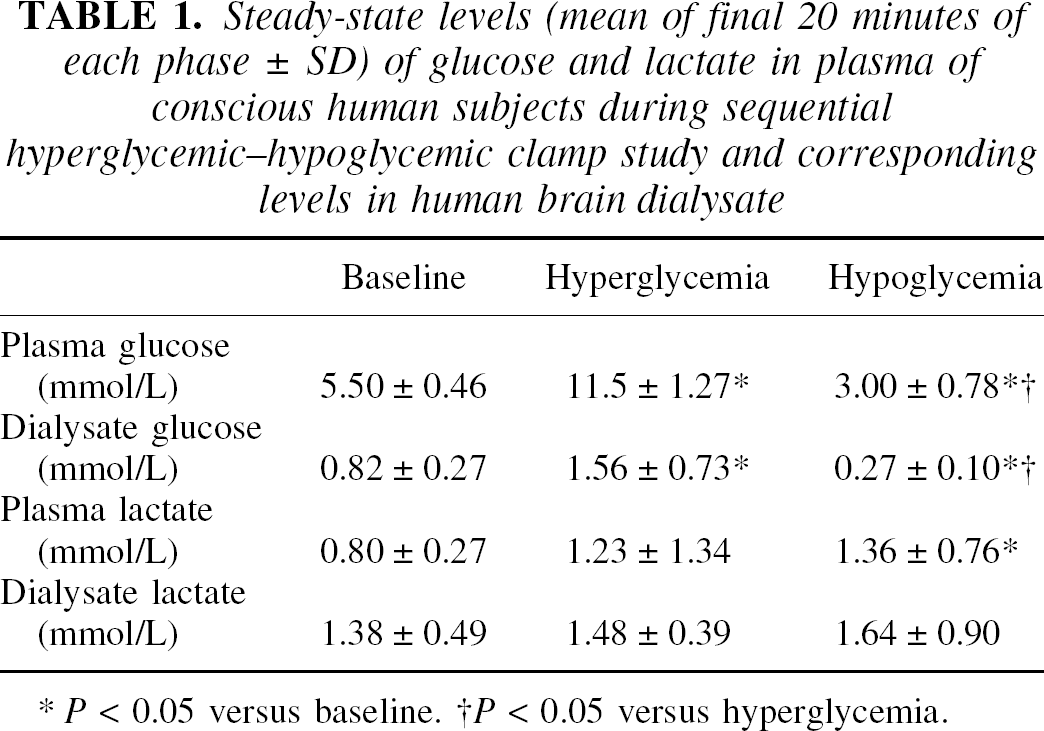
During the sequential hyperglycemic–hypoglycemic clamp study there were proportional changes in plasma and dialysate glucose. However, glucose levels in dialysate required more time to reach a steady state (Fig. 4). When exogenous glucose was infused, plasma glucose rapidly rose to 11.5 ± 1.22 mmol/L or 232 ± 7% above baseline values. In contrast, the rise in brain dialysate glucose was delayed. After 25 minutes, while plasma glucose was 233 ± 39% of baseline, ECF glucose had risen to only 164 ± 49% of baseline values. Only after 50 to 60 minutes did plasma and brain dialysate glucose change proportionately during the hyperglycemic phase of the study. As shown in Figure 4, when the glucose infusion was discontinued and exogenous insulin was given, plasma glucose rapidly declined. Plasma glucose decreased to baseline values in approximately 40 minutes and reached a stable hypoglycemia level of approximately 3 mmol/L approximately 30 minutes later. During the glucose descent in plasma there was also a delay in the descent in glucose in brain dialysate. The concentration of glucose in the ECF reached baseline approximately 30 min later than did plasma. Moreover, once a hypoglycemic steady state was reached, there was a 20-to 30-minute lag before the levels of glucose in the brain ECF had stabilized. Plasma lactate significantly increased from baseline during the hypoglycemic phase of the study, whereas dialysate lactate did not significantly change from baseline values during the hyperglycemic and hypoglycemic phases of study (Table 1). Expected changes in insulin and counterregulatory hormones during the hyperglycemia–hypoglycemia clamp study were seen, as illustrated in Table 2.
Steady-state levels (mean of final 20 minutes of each phase ± SD) of glucose and lactate in plasma of conscious human subjects during sequential hyperglycemic–hypoglycemic clamp study and corresponding levels in human brain dialysate
P < 0.05 versus baseline.
P < 0.05 versus hyperglycemia.
Levels of insulin and counterregulatory hormones (mean ± SD) at baseline and during sequential hyperglycemic–hypoglycemic clamp study
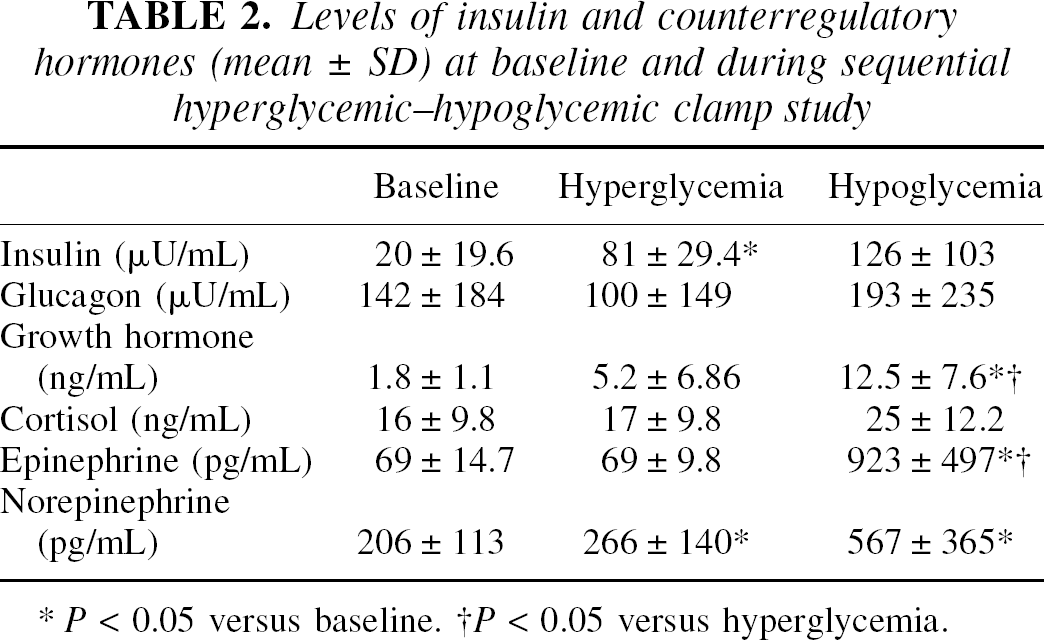
P < 0.05 versus baseline.
P < 0.05 versus hyperglycemia.
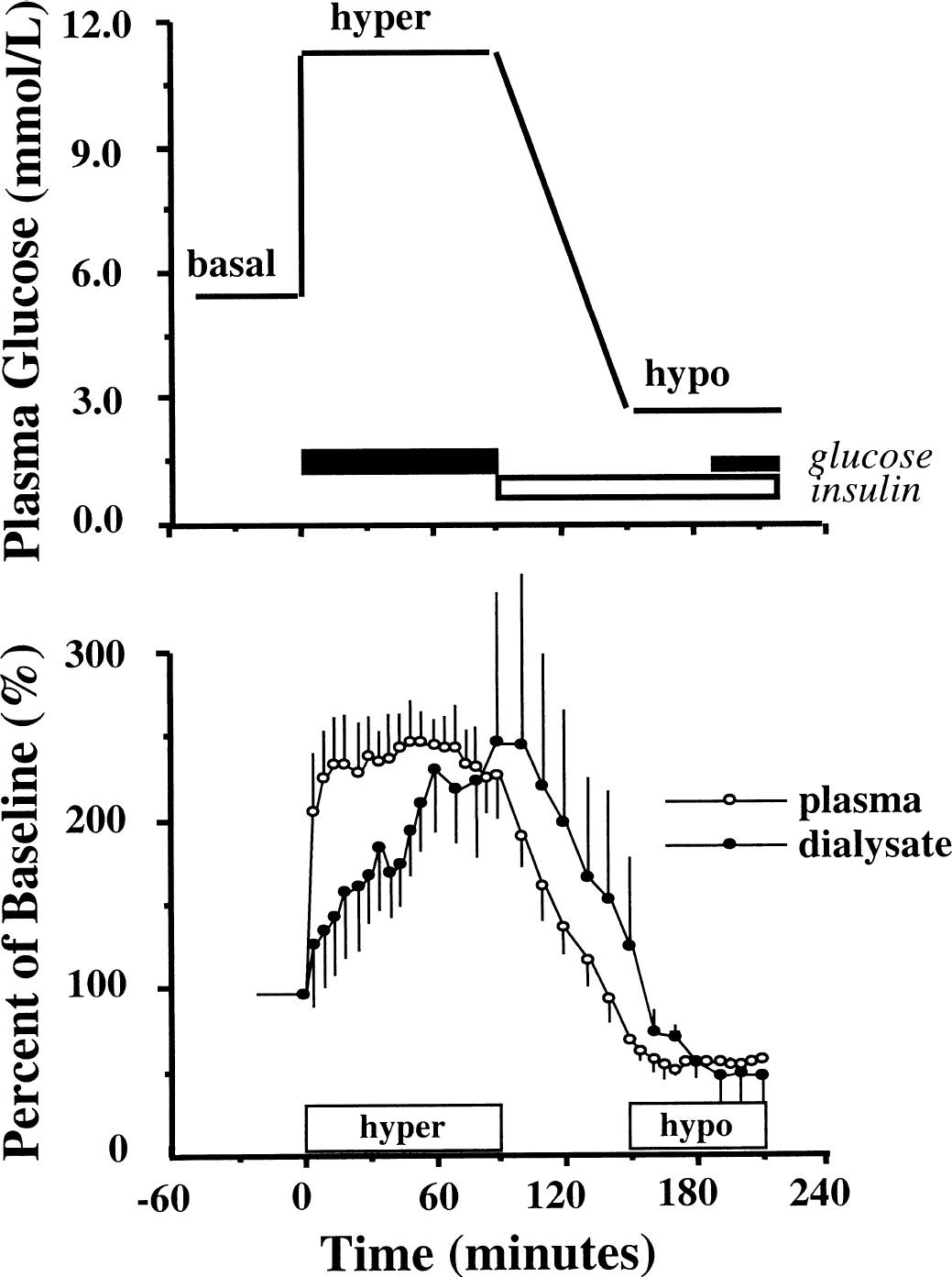
Representation of sequential hyperglycemic–hypoglycemic clamp study protocol (top panel) with actual levels of glucose in plasma and brain extracellular fluid of fasting human subjects with complex partial seizure disorder (n = 6) expressed as percent of baseline values (bottom panel). After baseline, hyperglycemia was induced (0–90 minutes) by intravenous infusion of glucose (20%). At 90 minutes, glucose was allowed to fall (90–150 minutes) by stopping glucose infusion and initiating intravenous insulin (Human Regular 3 mU/kg min−1). At the hypoglycemic plateau (150–210 minutes), glucose (20%) was restarted at a variable infusion rate to maintain plasma glucose. Plasma glucose was restored to normal at 210 minutes. Dialysate values are adjusted for a dead-space volume of 25 μL corresponding to 10-minute delay. Values are represented as mean ± SD.
DISCUSSION
In this study, we used the zero-flow microdialysis technique (Jacobson et al., 1985) in the central nervous system of conscious human subjects to estimate the basal concentrations of glucose and lactate in brain ECF. Our findings demonstrate a lower glucose concentration in brain ECF as compared to ambient plasma levels and a reversal in the concentration gradient for lactate, with the molar content of lactate in brain ECF in excess of that of glucose under basal conditions. These observations are consistent with prior studies in humans using plain microdialysis techniques showing higher levels of lactate than glucose in brain dialysate fluid (Goodman et al., 1999; Alessandri et al., 1999; Reinstrup et al., 2000; Langemann et al., 2001). The current data extend those findings by defining the absolute levels of metabolic fuels in brain ECF of conscious human subjects. These data suggest that the brain ECF milieu is quite different from that in the circulation and highlights the intriguing possibility that lactate may be locally produced and serve as an important source of fuel in the human brain. Our findings in humans are supported by similar findings in conscious rats using a variation of the same microdialysis technique.
Although previous in vitro and animal studies have suggested that lactate is an important fuel for the central nervous system (Schurr et al., 1997), the current data provide more direct support for this concept in humans. This hypothesis posits that a metabolic coupling exists between astrocytes and neurons, whereby glucose is used and lactate is released into the ECF by the astrocyte. Lactate is then taken up by neurons, where it serves as an important source of fuel (Tsacopoulos and Magistretti, 1996). It has long been postulated that astrocytes are likely a primary uptake site for metabolites from blood into the brain because their endfeet surround the intraparenchymal capillaries (Andriezen, 1893). Therefore, astrocytes appear to form the first cellular barrier that glucose encounters when entering the brain. As such, they are ideally positioned to provide coupling between neuronal activity and glucose uptake. Ample evidence from in vitro studies support this view (Tsacopoulos et al., 1988; Veuthey et al., 1994; Lopes-Cardozo et al., 1986; Pellerin and Magistretti, 1996). There is also good evidence that the primary 3-carbon metabolite produced by astrocytes is lactate (Walz and Mukerji, 1988; Dringen et al., 1993), produced at 10-fold higher rates than other 3-carbon compounds (Shank and Campbell, 1984; Shank et al., 1993; Sonnewald et al., 1991). In vitro work also demonstrates that flux of lactate occurs from astrocytes to neurons even in the presence of glucose (Larrabee, 1983, 1995), and that isolated photoreceptor neurons preferentially use lactate over glucose (Poitry-Yamate et al., 1995). Although our findings are far removed from in vitro work highlighting the apparent metabolic coupling of astrocytes and neurons, the surprisingly high levels of lactate in human (and rat) brain ECF are consistent with the potential importance of lactate as a source of fuel in the human brain. This view is also in keeping with studies demonstrating that lactate is an effective alternate fuel for the central nervous system in human subjects under experimental hypoglycemic conditions, where exogenous lactate infusion has been shown to offset the effects of hypoglycemic neuroglycopenia (Maran et al., 1994; Veneman et al., 1994; King et al., 1997).
Our ECF lactate levels are markedly higher than total brain tissue lactate in humans, estimated to be only approximately 0.5 to 1.0 mmol/L using MRS (Hanstock et al., 1988). This significant difference is consistent with the view that lactate is actively transported (from astrocytes) into the extracellular space against a gradient. Alternatively, it is possible that our findings of elevated ECF lactate could be because of the methods we used or the population we studied. Recently, several studies have characterized the family of monocarboxylic acid transporters through which lactate transport occurs (Broer et al., 1997; Gerhart et al., 1997; Gerhart et al., 1998; Koehler-Stec et al., 1998; Pellerin et al., 1998; Bergersen et al., 1999; Broer et al., 1999; Halestrap and Price, 1999; Leino et al., 1999; Hanu et al., 2000). However, the distribution of the different subtypes of these transporters in the human brain is still unclear because of between species and discrepant results between in vitro and in vivo studies. Pellerin and colleagues have shown that in the adult rats, both astrocytes and neurons continue to express monocarboxylic acid transporters at a time when lactate can no longer cross easily from the circulation, suggesting that lactate is formed within the brain parenchyma and is actively transported into the ECF where it is readily available in relatively high concentrations for use by neurons when needed (Pellerin et al., 1998).
It is possible that our findings of high lactate levels in brain ECF may be in part secondary to the technique we used. For example, microdialysis might cause significant tissue disruption leading to a leak of lactate from cells and artificially increasing lactate levels in the ECF. However, this explanation seems unlikely. In other studies using the same technique, we estimate basal glutamate levels to be approximately 5 μmol/L (unpublished observations), values several orders of magnitude lower than measurements of total brain tissue glutamate by MRS, which are approximately 9 mmol/L (Mason et al., 2000). Furthermore, studies in animals suggest that the effects of trauma on the brain are negligible 2 hours after implantation of the microdialysis probes (Benveniste, 1989). Finally, lactate levels in cerebrospinal fluid (reflecting contribution from brain ECF and the systemic circulation) in healthy human subjects are approximately 1.6 mmol/L to 1.7 mmol/L (Calabrese et al., 1991; Stover et al., 1997; Livingston et al., 1989), values higher than those normally present in plasma and those in total brain tissue as measured by MRS.
The subjects included in this study had complex partial seizures. Despite the fact that we only included data collected from microdialysis probes located in what was believed to be nonepileptic regions, the underlying seizure disorder may have influenced our results. In this regard, two studies are worth discussing. First, Reinstrup and colleagues used a low flow dialysis system at 0.3 μL/min with a 10-mm dialysis probe and calculated “basal” ECF lactate in normal frontal cortex of nine awake subjects to be 2.9 ± 0.9 mmol/L (Reinstrup et al., 2000). Using a 10-mm probe in humans, one would expect a recovery rate 70% (Hutchinson et al., 2000). Therefore, the corrected “true” lactate values estimated from that study would be approximately 4.1 mmol/L, which are slightly lower (although unlikely to be significantly so) than ours. Langemann and colleagues used the no net flux (six patients) and low flow (two patients) methods in anesthetized patients (Langemann et al., 2001). They estimated basal glucose and lactate levels to be approximately 1.7 mmol/L and 2 mmol/L, respectively. However, these measurements were obtained in acute surgical settings under general anesthesia, thereby, limiting their applicability to conscious humans. Thus, we believe that the contribution of the underlying seizure disorder to elevations in ECF lactate levels in our patients is most likely small. Regardless, this issue would not affect our main finding of significantly higher molar content of lactate in the ECF as compared with total brain lactate and to plasma levels.
To validate our findings in humans, we designed similar studies in normal rats and measured basal ECF levels of glucose and lactate from the hippocampus and the frontal cortex. Data from these complementary studies further support our main findings of reversal of molar content of glucose and lactate in ECF as compared with plasma, and of higher levels of lactate than glucose in the ECF (Fig. 3). It is noteworthy, however, that although brain ECF glucose levels were very similar in the human and rat studies, brain ECF lactate levels observed in the rat were lower than those in humans. Although these results might be explained by differences between the techniques used or difference between the two species, they raise the possibility that lactate levels in our subjects might be further elevated by their underlying seizure disorder. Nonetheless, it is important to highlight differences in the application of the microdialysis technique in the two experimental settings, which might have contributed to these results. One major difference is that in humans, the microdialysis catheter was implanted days before the actual metabolic study whereas, in rats, the catheter guide probe was implanted days before study but the actual microdialysis catheter was inserted acutely just before the metabolic study. In addition, the calibration methods applied to estimate recovery and in turn calculate levels of glucose and lactate in brain ECF in the human and rat studies also differed. In the rats, we used measurement of in vitro recovery immediately after in vivo study as an estimate of in vivo recovery at the same low perfusate flow rate (0.3 μL/min), whereas in humans we used the zero-flow technique (Jacobson et al., 1985). Regardless of the explanation for the differences in the estimation of brain ECF levels in humans and in rats, the main finding of reversal of molar content of glucose and lactate between plasma and ECF remains unaffected.
The current ECF glucose data obtained under basal conditions and during the hyperglycemic–hypoglycemic clamp study indicate that ECF levels are only approximately 30% those in plasma. The gradient between plasma and brain ECF was substantially greater than that previously reported by our group between plasma and muscle adipose ECF (Maggs et al., 1997), presumably reflecting the effects of the blood brain barrier and the high rate of brain glucose metabolism. These data are in close agreement with MRS measurements of total brain tissue glucose obtained both at baseline and during glucose infusion (Gruetter et al., 1993). The marked plasma–brain ECF glucose gradient has implications with regard to the importance of glucose transport for brain glucose metabolism during hypoglycemia. It suggests that brain glucose transport may become rate limiting at higher plasma levels of glucose than previously thought. The close agreement between our data and MRS measurements also support the hypothesis that after glucose crosses the blood–brain barrier, it freely diffuses into brain cells and extracellular space (Lund-Andersen and Kjeldsen, 1976).
The significant lag time between acute changes in glucose levels in plasma and those in brain ECF has significant implications for the interpretation of data from studies that examine brain function during hypoglycemia. It is possible, for example, to underestimate the glycemic level below which brain function becomes impaired if assessment of brain function took place immediately after the desired hypoglycemic level has been reached (i.e., before ECF glucose levels have reached steady state). Our data suggest that an equilibration period of at least 20 to 30 minutes should be allowed after the desired hypoglycemic levels are acutely achieved before studying brain function. In addition, the lag time between acute changes in glycemia and changes in ECF glucose levels have implications for interpreting data collected using subcutaneous glucose sensors that have recently been developed to monitor glycemia in patients with diabetes. These sensors, which measure glucose levels in the interstitial fluid of subcutaneous tissues, appear to do so with a lag time of up to 20 minutes with respect to plasma levels in humans (reviewed in Rebrin et al., 1999). Therefore, based on our data, it appears that acute changes in brain ECF glucose levels are more closely mirrored by subcutaneous sensors than by plasma glucose.
In summary, these data provide unique information about the metabolic milieu in the conscious human brain at baseline and in response to varying glycemia. The data demonstrate that metabolic fuels within the brain microenvironment do not mirror those in the systemic circulation and suggest that this disparity may reflect the brain's unique need for glucose to drive the glutamate–glutamine cycle as well as to maintain neuronal function. To our knowledge, this is the first in vivo study in humans to support the hypothesis that lactate is locally generated in the brain, where it provides an important source of fuel. Furthermore, this study indicates that a lag time exists between changes in glucose levels in the plasma and those in the brain ECF. Such a lag time as well as the local availability of lactate might play a protective role to limit brain injury during severe hypoglycemia.
Footnotes
Acknowledgments:
The authors thank Drs. Robert Shulman and Ian Simpson for their review and helpful comments on earlier versions of this article; Dr. Elizabeth Wallace for assistance in conducting this study and data collection; Xiaoning Fan for the preparation of the microdialysis probes; and Yan-ping Pu, Michael Cassaday, and David Ansonoff for technical support in collecting and analyzing the microdialysis samples.
