Abstract
Astrocytes play many roles essential for normal brain activity. The ability of these cells to recover after temporary focal cerebral ischemia is likely to be one important determinant of the extent of brain dysfunction and tissue damage. We have assessed astrocytic function based on the incorporation of radiolabel from 1-14C-acetate into glutamine at 1 hour of recirculation after middle cerebral artery occlusion for 2 or 3 hours in rats. There were marked differences in the response between subregions within the tissue subjected to ischemia, but the overall pattern of changes was similar after each ischemic period. The striatum, which forms part of the severely ischemic focal tissue during arterial occlusion, showed a large (44% to 68%) decrease in glutamine labeling compared with equivalent tissue from the contralateral hemisphere. In contrast, 14C-glutamine content was not significantly altered in perifocal tissue in the cerebral cortex, which was subjected to more moderate ischemia. Cortical focal tissue also was not significantly affected, but the response was much more variable between rats. In these brain subregions, the extent of recovery of the 14C-acetate metabolism after ischemia was not a good predictor of the likelihood of subsequent infarct development. Interestingly, a similar pattern of responses persisted when recirculation was extended to 4 hours. These results indicate that many astrocytes, particularly in the cortex, remain viable and capable of at least some complex oxidative metabolism during the first few hours of recirculation.
Introduction
Ischemic stroke usually produces a nonselective cell loss, resulting in a region of tissue infarction surrounding the occluded artery. The size and location of the infarcted tissue are major determinants of the long-term consequences of this disorder. Significant advances have been made in understanding the molecular events contributing to cell death (Leker and Shohami, 2002; Lipton, 1999), but the picture remains far from complete. Reperfusion during the first few hours of focal ischemia can reduce infarct volume (Anderson and Sims, 1999; Memezawa et al, 1992) and early thrombolytic intervention has been found to improve the outcome in stroke in humans (Lees, 2000). Furthermore, at least in animal models of stroke, some treatments initiated during reperfusion after ischemic periods up to 3 hours can produce dramatic reductions in the subsequent tissue damage (Markgraf et al, 1998; Yoshimoto and Siesjo, 1999). Thus, many of the key events leading to cell death after temporary ischemia apparently develop during the reperfusion period.
Although infarct formation involves the loss of astrocytes and other glia as well as neurons, the timing of the death of these cells and their contributions to the generation of ischemic damage have undergone only limited investigation. Astrocytes have long been recognized as playing important roles in normal neurotransmission through functions including the uptake of neurotransmitters such as glutamate and GABA, the provision to neurons of glutamine and other essential metabolic precursors and the buffering of extracellular potassium (Fields and Stevens-Graham, 2002; Sykova, 2001). Furthermore, recent studies have revealed additional complexities in the interchange of many metabolites with neurons and the release of possible neuromodulators (Fields and Stevens-Graham, 2002; Muyderman et al, 2001). Thus, under the abnormal conditions during ischemia and reperfusion, astrocytes have the potential to ameliorate or exacerbate damage to neurons and other cells, depending on the extent to which key functions are preserved.
Astrocytes in culture show a greater resistance than neurons to some ischemic-like insults (Goldberg and Choi, 1993; Xu et al, 2001). Evidence to suggest that this might also be the case in the intact mature brain is provided by the better preservation in animal models of stroke of markers for astrocytes detected immunohistochemically compared with those for neurons (Chen et al, 1993; Lee et al, 2003; Li et al, 1995). However, these findings offer little insight into the extent of functional preservation of the cells. Furthermore, evidence for a greater susceptibility of at least some astrocyte subpopulations to ischemia in vivo has also been obtained (Liu et al, 1999; Lukaszevicz et al, 2002) and the vulnerability of these cells in culture can be markedly increased by acidosis (Giffard et al, 1990; Swanson et al, 1997). In the present study, we have directly evaluated astrocytic functional recovery after temporary focal cerebral ischemia in vivo, based on the oxidative metabolism of intravenously injected 1-14C-acetate and the incorporation of radiolabel into glutamine. This technique allows important aspects of astrocytic metabolism to be simultaneously evaluated in multiple brain regions.
Radiolabeled acetate in astrocytes is converted to acetyl CoA and further metabolized via the tricarboxylic cycle. Glutamine is rapidly labeled in these cells by the conversion of α-ketoglutarate to glutamate and the subsequent action of the synthetic enzyme, glutamine synthetase. The selectivity of this approach is based on the ability of glia, but not neurons, to take up acetate (Dienel and Hertz, 2001; Fonnum et al, 1997; Waniewski and Martin, 1998), and on the preferential localization of glutamine synthetase within astrocytes (Norenberg and Martinez-Hernandez, 1979; Petito et al, 1992). A similar approach has been used in some previous investigations of cerebral ischemia in which the products of acetate metabolism were analyzed using NMR (Haberg et al, 1998; Pascual et al, 1998). Only one of these studies (Pascual et al, 1998) has examined a response to reperfusion and this was an investigation of the whole cerebral hemisphere under conditions in which ischemia was only partially reversed.
For our studies, focal ischemia was induced by a modification of the thread occlusion method of Zea Longa et al (1989), which allows a complete reversal of the ischemia. Tissue for analysis was obtained from regions of the striatum and the cerebral cortex that formed part of the severely ischemic core or focus during occlusion of the middle cerebral artery (MCA). Adjacent cortical ‘perifocal’ tissue, which receives a more moderate reduction in flow during MCA occlusion, was also studied. Two ischemic periods were investigated because these differ in the size of the infarcts that subsequently develop. Thus, these measurements allow us to evaluate the effects of the duration and severity of ischemia on astrocytic oxidative metabolism and to examine the relationship of changes to the distribution of subsequent tissue infarction.
Materials and methods
Surgical Procedure
All experimental procedures were approved by the Animal Welfare Committee of Flinders University and are consistent with the Code of Practice of the National Health and Medical Research Council (Australia). Male Sprague–Dawley rats (265 to 295 g) were supplied by the Animal Resource Centre (Gilles Plains, South Australia) or bred in-house (School of Medicine, Flinders University) from the same stock. Rats were fasted overnight before surgery. In preparation for surgery, they were intubated and ventilated with a mixture of 23% oxygen/77% nitrous oxide (vol./vol.) containing 1% to 1.5% halothane. Body temperature was maintained throughout surgery using a heating lamp connected to a rectal temperature probe. A polyethylene catheter was placed in the right femoral artery for physiological monitoring of blood gases, blood pressure and blood glucose. A second catheter was placed in the femoral vein, externalized to the tail and taped in place for later administration of 1-14C-acetate.
Reversible focal cerebral ischemia was achieved by occluding the origin of the MCA using the intraluminal filament technique of Zea Longa et al (1989), with minor modifications (Anderson and Sims, 1999). A monofilament nylon thread (Dynek sutures, Adelaide, Australia) coated with poly-L-lysine (Belayev et al, 1996) was introduced into the right external carotid artery and advanced through the internal carotid artery to occlude the origin of the right MCA. The wounds were closed, infused locally with 0.5% bupivicaine and the volatile anesthetic discontinued. Animals were tested at 2 hours for anticlockwise circling as an indicator of successful MCA occlusion. Rats not meeting this criterion were excluded from the study. Body temperature was monitored postoperatively for 6 hours or until euthanasia. Most rats developed hyperthermia as reported previously (Anderson and Sims, 1999). When the temperature exceeded 37.8°C, rats were placed in an insulated cooling box at a temperature of 7°C to 12°C to limit the magnitude of this response.
For reversal of ischemia, rats were briefly reanesthetized with 2% to 2.5% halothane in 33% oxygen/67% nitrous oxide administered via a face mask and the intraluminal thread withdrawn. Three sham-treated rats underwent the same surgical procedure, except that the thread was inserted only a short distance and therefore did not occlude the MCA. Reperfusion was mimicked by withdrawing the thread 2 hours later. The rats were injected with radiolabeled acetate and killed for analysis of metabolic products after a further hour.
Assessment of Tissue Damage
For assessment of tissue damage, rats were decapitated 48 hours after occlusion, the brains removed from the skulls, placed in a perspex brain-cutting template and the forebrain cut into 1.5-mm-thick coronal sections using a razor blade. The slices were incubated in 3% 2,3,5-triphenyl 2-H-tetrazolium chloride (TTC; in 0.9% NaCl and 20 mmol/L Tris–HCl, pH 7.4) in darkness for 20 mins. The slices were then placed in fixative (0.5% glutaraldehyde, 4% formaldehyde in 0.1 mol/L phosphate buffer) and were subsequently digitally scanned. The area of infarction was calculated using Adobe Photoshop software.
Preparation of Tissue Extracts from Brain Subregions
After the required period of recirculation, the rats were injected intravenously with 400 μL of 60 μCi 1-14C-acetate (45 to 60 mCi/mmol, NEN, Boston, USA) dissolved in 0.3 mmol/L sodium acetate and 0.9% NaCl. Rats were decapitated 5 mins after the injection. The brains were rapidly removed within 90 secs, transferred into a perspex brain mold and frozen in liquid nitrogen. A 3-mm coronal section extending caudally from the rostral limit of the striatum was dissected from the frozen brain. Tissue regions were defined based on a previous study (Anderson and Sims, 1999) and were dissected from the frozen section in a cold box (−10°C). These regions are illustrated in Figure 1. In the hemisphere subjected to MCA occlusion, samples were obtained from the striatum and from part of the cerebral cortex (‘cortical focal tissue’) that fell within the severely affected core region during ischemia. Adjacent perifocal tissue, from an area of the cortex subjected to more moderate ischemia, was also sampled. Corresponding tissue regions were also obtained from the contralateral hemisphere to provide comparisons of the acetate metabolism in tissue that had not been ischemic.
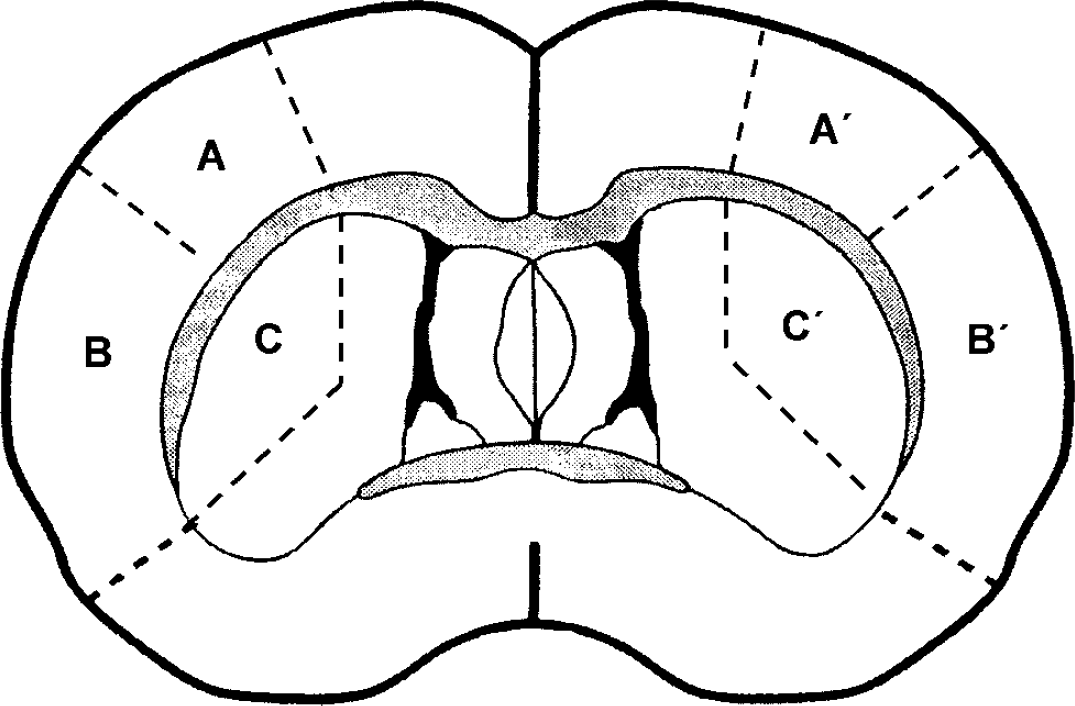
The diagram represents a coronal section of rat brain showing the tissue subregions analyzed in studies of 1-14C-acetate metabolism. Samples were removed from a single 3-mm-thick section in which the posterior face corresponded to the section shown in this figure. The region marked A′ is cortical perifocal tissue in the hemisphere subjected to MCA occlusion. Region B′ is cortical focal tissue and region C′ is striatal tissue. The areas marked A, B and C represent the corresponding regions sampled from the contralateral hemisphere.
Each tissue sample was added to 200 μL of ice-cold 0.05 mol/L HClO4 and sonicated for 20 secs. Homoserine was added (20 μL, 5 mmol/L) as an internal standard. A 20 μL aliquot was removed to determine total tissue radioactivity. The sample was centrifuged (15,000g for 5 mins at 4°C) and the supernatant placed on ice. The pellet was reextracted using 100 μL 0.05 mol/L HClO4 and recentrifuged. The supernatants from the two centrifugations were pooled and freeze dried. The samples were stored at −80°C and analyzed within two weeks. The pellets from the extraction were solubilized using 1 mL 2 mol/L NaOH and protein determined by the method of Lowry et al (1951).
Measurement of Radiolabel Incorporation into Glutamine and Glutamate
Amino acids were derivatized with o-phthaldialdehyde (OPA) before separation by HPLC and electrochemical detection using a modification of the method of Donzanti and Yamamoto (1988). In preliminary studies, this procedure was adapted to provide reproducible derivatization and suitable separation of the substantial amounts of amino acids in the freeze-dried tissue extracts. Under the final conditions that were developed (see below), measurements using radiolabeled glutamine demonstrated that OPA derivatization and HPLC separation resulted in 82% recovery (range: 80% to 84%) of the radiolabel in a single peak that corresponded to the position of derivatized glutamine standards detected electrochemically and was well separated from the solvent front. The remainder of the radiolabeled glutamine eluted with the front. The recovery in the main glutamine peak was the same for tissue samples and for standard mixtures of amino acids. The final results have been corrected for this incomplete recovery of the amino acid.
Before derivitization, the freeze-dried tissue extracts were dissolved in 70 μL phosphate buffer (0.5 mol/L, pH 7.4) and filtered using 0.45 μm hydrophilic polyproprylene filters (GHP Nanosep MF, Pall Life Sciences, USA). The derivatization reagent was prepared by dissolving 5.4 mg OPA in 200 μL methanol followed by the addition of 2 μL 2-mercaptoethanol and 1.8 mL 0.1 mol/L sodium tetraborate. This reagent was prepared daily and kept sealed in darkness. For determination of radiolabel incorporation, 50 μL of the sample was mixed with 75 μL of OPA reagent and injected after approximately 75 secs via a Waters 712 Wispy autosampler (Waters, Milford, MA, USA).
Separation was achieved using a 100 × 4.6 mm2 C-18 reversed phase column (3 μm Exsil, SGE Australia) with a guard column (3 μm, SGE Australia). The mobile phase for isocratic elution consisted of 0.1 mol/L Na2HPO4, 0.13 mmol/L Na2EDTA in 28% methanol (pH 6.4) delivered at 0.8 mL/minute using an LC1100 HPLC pump (ICI Instruments, Melbourne, Australia). Derivatized amino acids were detected electrochemically (BAS LC-4B amperometric detector, BAS, West Lafayette, IN, USA) at an applied potential of 600 mV. Samples of eluate were collected at 1-min intervals for 15 mins. Those fractions corresponding to the glutamine and glutamate peaks were added to 9 mL Readysafe scintillation fluid (Beckman Coulter, Fullerton, CA, USA) and 1 mL water. The incorporated radioactivity was measured using a Beckman LS3801 scintillation counter.
When this procedure was applied to tissue extracts, 14C-glutamine and 14C-glutamate were completely separated from each other and were also separated from other radiolabeled metabolites at baseline or near-baseline level. The peak electrochemical response of the amino acids in these extracts was too large to allow accurate assessment of the amino-acid concentrations. Thus, a second 1 to 2 μL aliquot of the extract was sampled and diluted with phosphate buffer (0.5 mol/L, pH 7.4) to a final volume of 30 μL. This diluted extract was derivatized (45 μL OPA reagent) and subjected to HPLC separation to allow tissue contents of the amino acids to be calculated. Standard mixtures of amino acids were prepared daily from 1-mmol/L stock solutions and treated for analysis as for the tissue extracts. The content of amino acids in the effluent from the HPLC was calculated from comparisons with standards that were analyzed on the same day. The tissue content of amino acids and the incorporation of radioactivity into these amino acids were corrected for recovery of the internal standard, homoserine.
Statistical Analysis
Results are expressed as mean±s.d. Paired t-test was used in comparing results from equivalent regions in the occluded and nonoccluded hemispheres.
Results
Tissue infarction produced by 2 or 3 hours of ischemia was assessed at intervals during these studies based on the staining of brain sections with TTC at 48 hours after MCA occlusion. Some representative sections showing TTC staining at 48 hours after either 2- or 3-hour MCA occlusion are presented in Figure 2. In agreement with previous findings from our group (Anderson and Sims, 1999), 3 hours of focal ischemia produced widespread tissue infarction in all rats. This completely encompassed all of the MCA perfusion territory in most animals (seven out of nine). Such damage fully incorporated the cortical perifocal tissue as well as the regions of the striatum and cortex that were sampled as focal tissue for the assessment of acetate metabolism. The shorter 2-hour ischemic period produced more variable outcomes. In most rats, there was substantial involvement of the focal tissue in the striatum and cortex, but in the perifocal tissue there was either no infarction (two rats) or partial involvement (four rats). These observations were reflected in the measures of infarct volume (expressed as a percentage of the total volume of the tissue region). The infarct volume in the striatum was similar at 48 hours after either 2 hours (61%±39%, n=6) or 3 hours (78%±16%, n=9) of ischemia, but in the cortex was substantially less after the shorter ischemic period (2-hour ischemia: 30%±25%; 3-hour ischemia: 55%±20%).
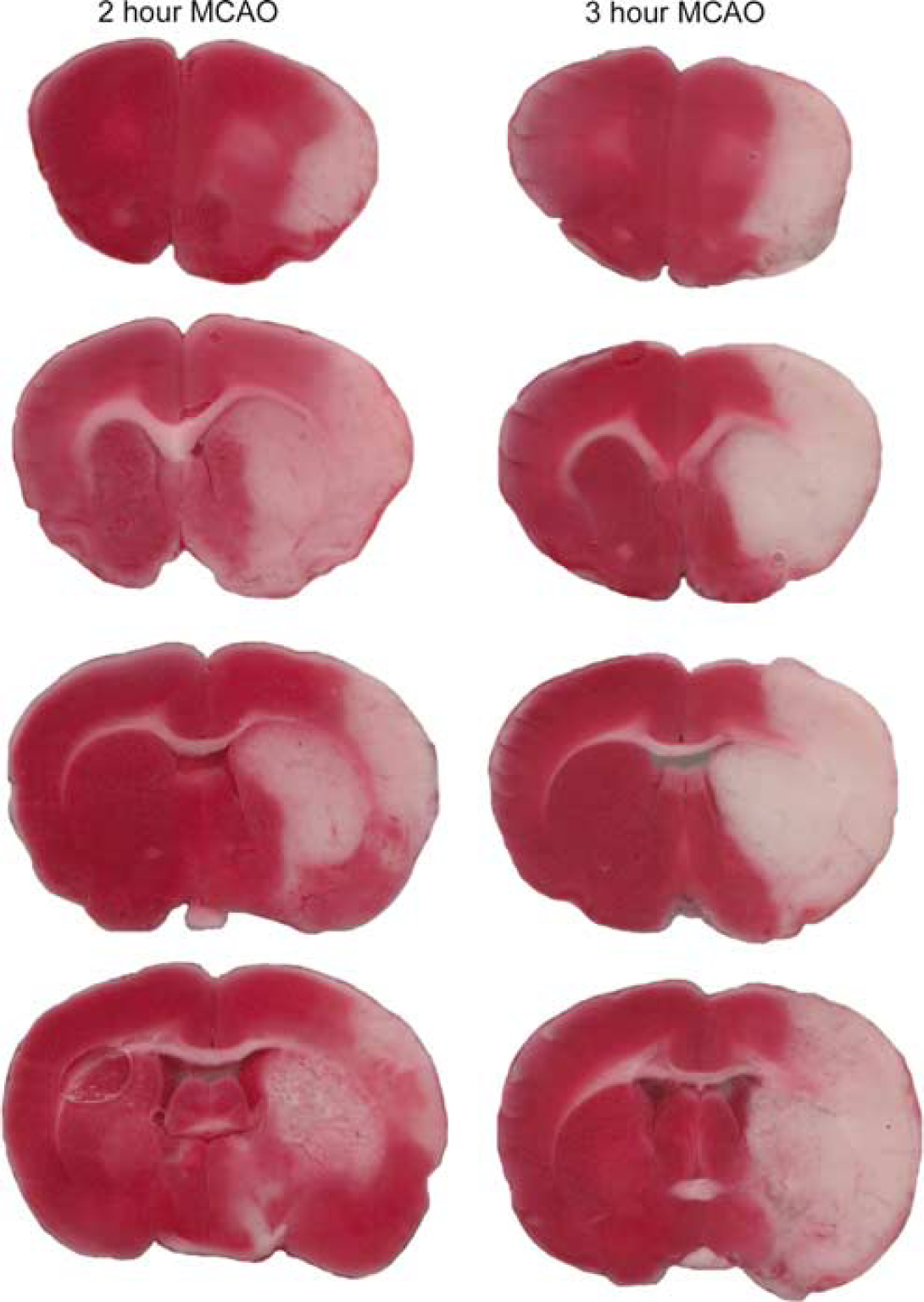
Coronal sections of rat brains stained with TTC for the assessment of infarct size at 48 hours after right MCA occlusion. The red areas represent nonischemic regions and white areas (absence of staining) identify infarcted tissue. The images presented are from two representative experiments for rats subjected to either 2 or 3 hours of MCA occlusion. The coronal sections were cut at 1.5-mm intervals.
The physiological parameters for the groups of rats studied are presented in Table 1. Blood pressure, blood gases, pH and glucose are similar for all of the groups investigated. Body temperature increased during ischemia, consistent with previous investigations using the thread occlusion model (Anderson and Sims, 1999; Oliff et al, 1996; Zhao et al, 1994).
Physiological parameters for rats used in the studies of [1-14C]acetate metabolism following MCA occlusion
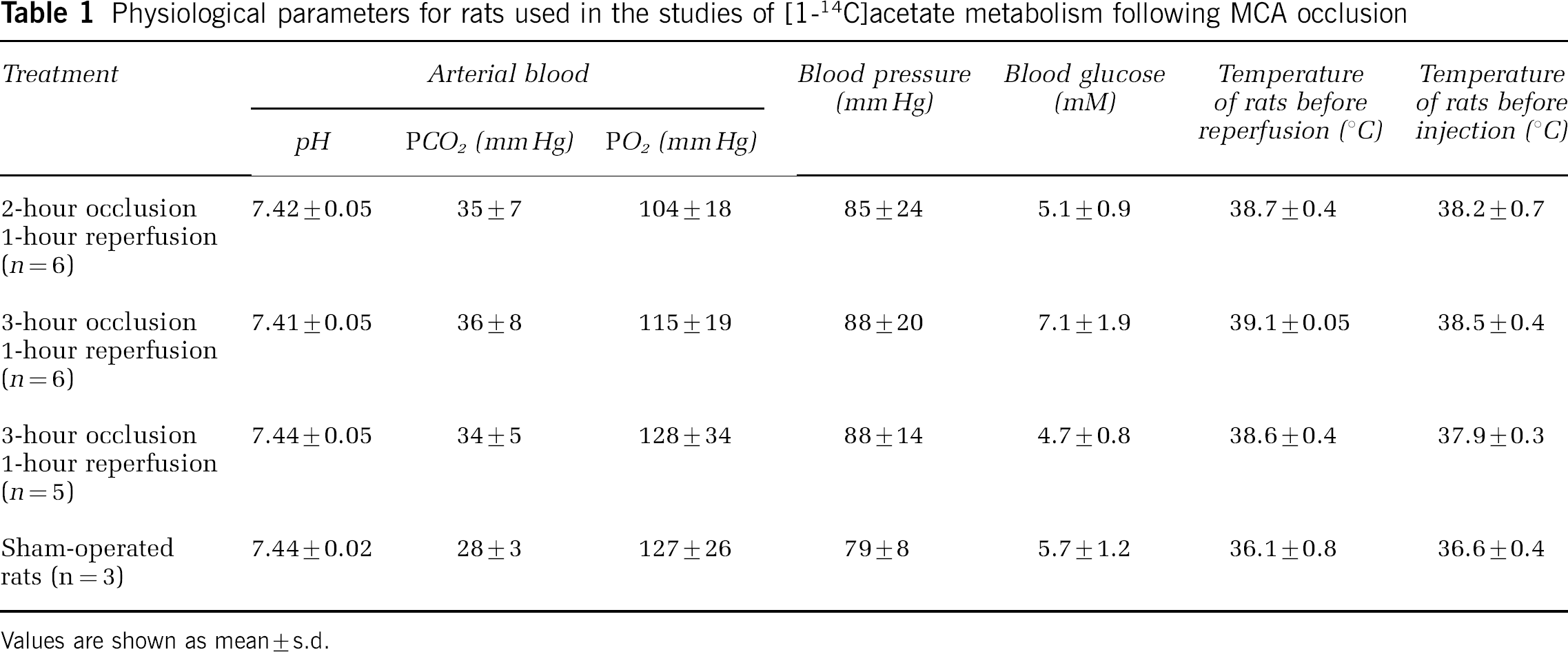
Values are shown as mean±s.d.
In earlier studies, the maximum specific activity of glutamine was seen at 5 mins after intravenous injection of 1-14C-acetate and then declined slowly with a half-life of more than an hour (Berl and Frigyesi, 1969). Approximately 80% of label remaining in the tissue at 5 mins was contained in the amino acids glutamate and glutamine. In preliminary studies, we confirmed that the maximum specific labeling of glutamine was produced at approximately 5 mins after injection, with similar values seen at 10 mins. Thus, rats were routinely killed for tissue extraction at 5 mins after injection of radiolabel in our subsequent investigations.
Not surprisingly, given the complexity of the metabolic processes being investigated, there was considerable variability (relative s.d.=15% to 37%) between rats in the measured 14C-glutamine content even in the hemisphere that was not subjected to MCA occlusion. Nonetheless, comparisons between hemispheres in individual rats revealed consistent effects of MCA occlusion and reperfusion on 14C-acetate metabolism in two of the three brain subregions (the striatum and perifocal cortical tissue) under each of the conditions investigated.
Effects of Ischemia and 1-hour Recirculation on Astrocytic Oxidative Metabolism
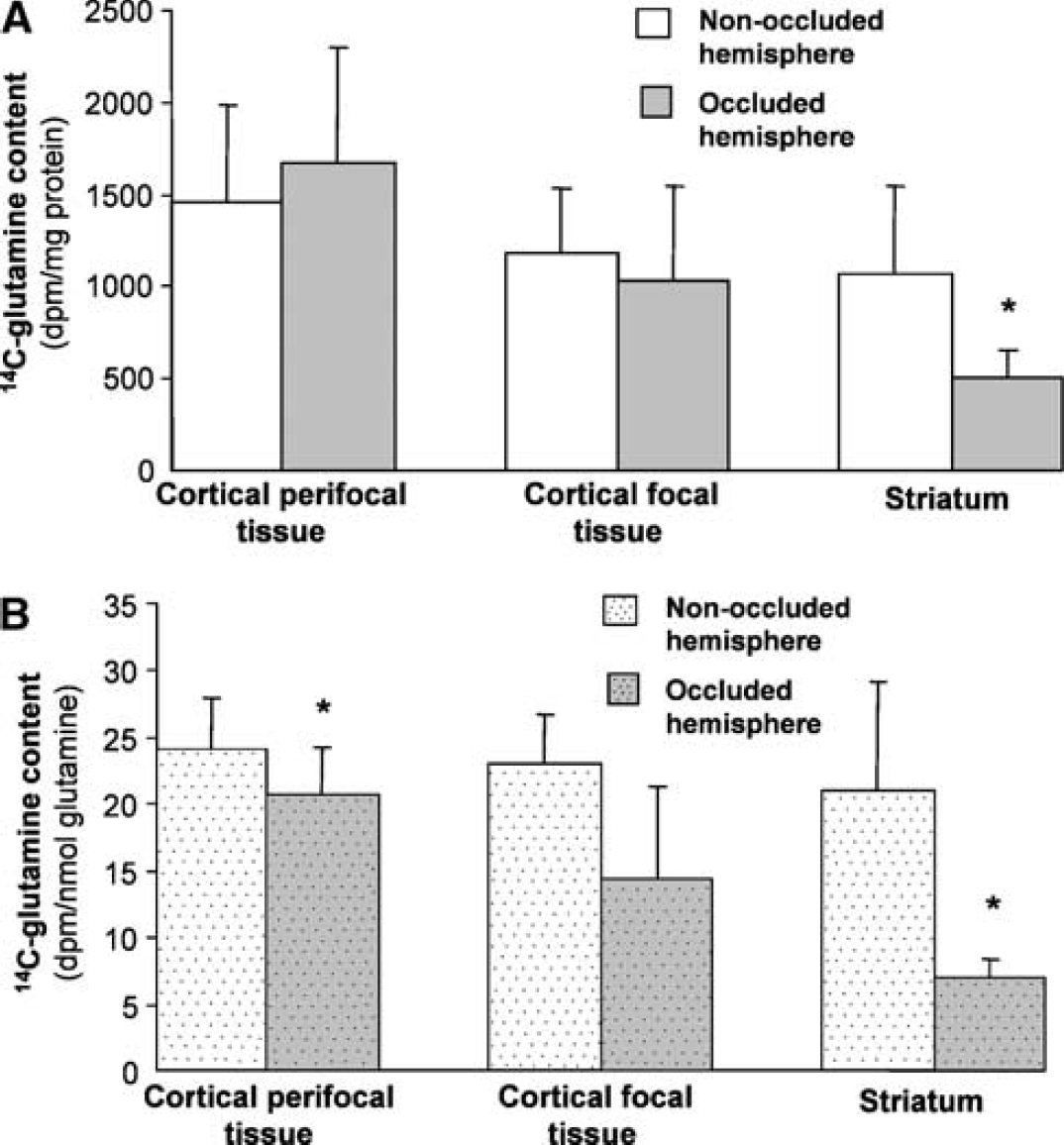
The 14C-glutamine specific activity in the tissue subregions from the two hemispheres after 2- and 3-hour MCA occlusion is shown in Figure 3. The metabolic response to ischemia and reperfusion differed markedly between the three tissue subregions but the overall pattern of responses across the affected brain tissue was very similar after either 2 or 3 hours of focal ischemia.
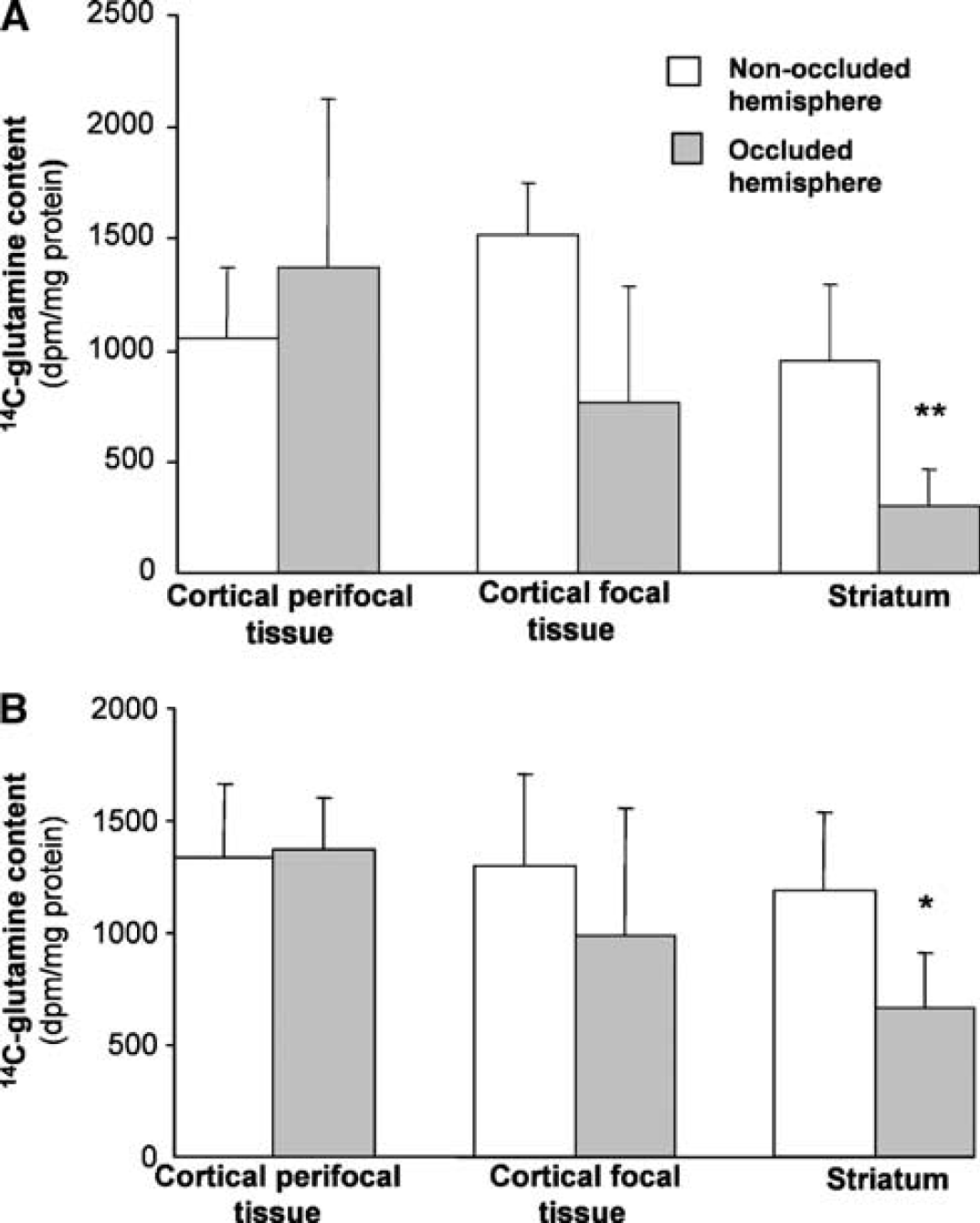
Incorporation of radiolabel from 1-14C-acetate into glutamine at 1 hour of reperfusion after either (
The largest effect was seen in the striatum (Figure 3). For all of the rats studied after both of the ischemic insults, 14C-glutamine specific activity in the striatum subjected to occlusion was less than in the corresponding tissue from the contralateral hemisphere. The pooled results revealed an overall decrease of 68% during early recirculation after 2 hours of ischemia and 44% after 3 hours of ischemia. In contrast, the radioactivity in glutamine in the cortical perifocal tissue was similar in the two hemispheres. There was also no significant difference between hemispheres in radiolabel incorporation into glutamine in the focal tissue from the cortex. However, the response was much more variable between animals in this region. In four of the rats studied during early recirculation after 3 hours of ischemia and two rats after 2 hours of ischemia, 14C-glutamine in cortical focal tissue from the treated hemisphere was either increased relative to the contralateral hemisphere or showed small (<10%) decreases. However, in the remaining animals, large reductions were seen (30 to 90% decreases) in the tissue from the hemisphere subjected to MCA occlusion.
The incorporation of radiolabel into glutamine in rats that underwent sham occlusion of the MCA was similar in the two hemispheres (Figure 4). Thus, the differences seen in the MCA occluded rats were not because of the experimental procedure alone.
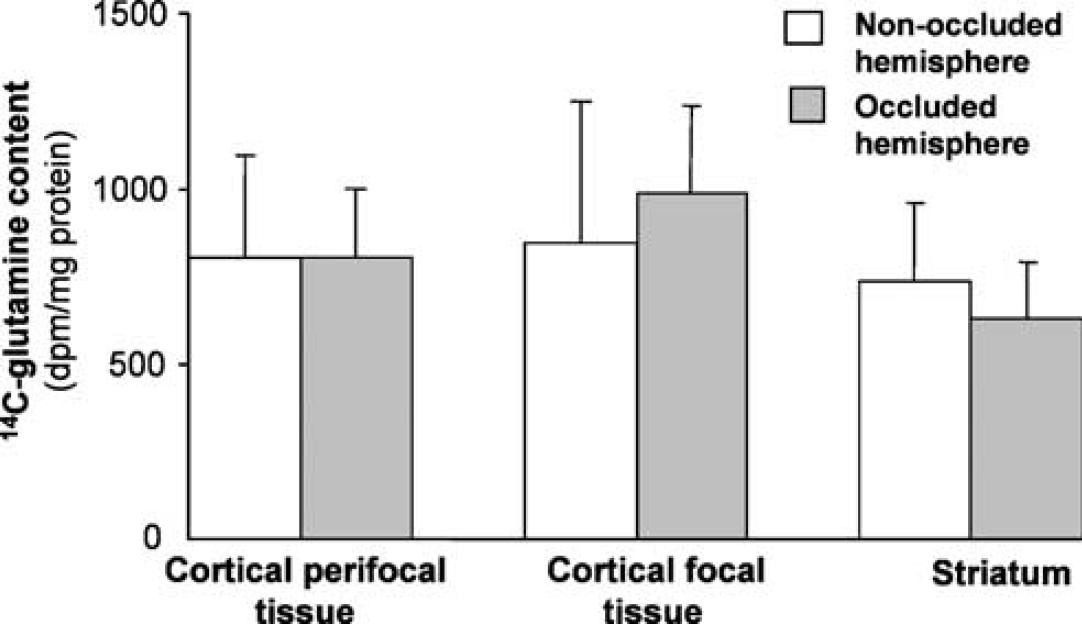
Incorporation of radiolabel from 1-14C-acetate into glutamine in sham-operated rats. The content of 14C-glutamine in three subregions from the hemisphere subjected to sham MCA occlusion is compared with 14C-glutamine in the equivalent subregions from the contralateral hemisphere. Values are shown as mean+s.d. (n=3).
The total radioactivity detected in samples of unfractionated tissue homogenates before extraction of the amino acids was generally similar in the equivalent samples from the two hemispheres, although in a few regions there was a small but statistically significant increase in the samples from the hemisphere subjected to MCA occlusion (Figure 5). These findings suggest that changes observed in radiolabel incorporation into glutamine were not likely to have resulted from altered availability of 1-14C-acetate in the hemisphere previously subjected to MCA occlusion.
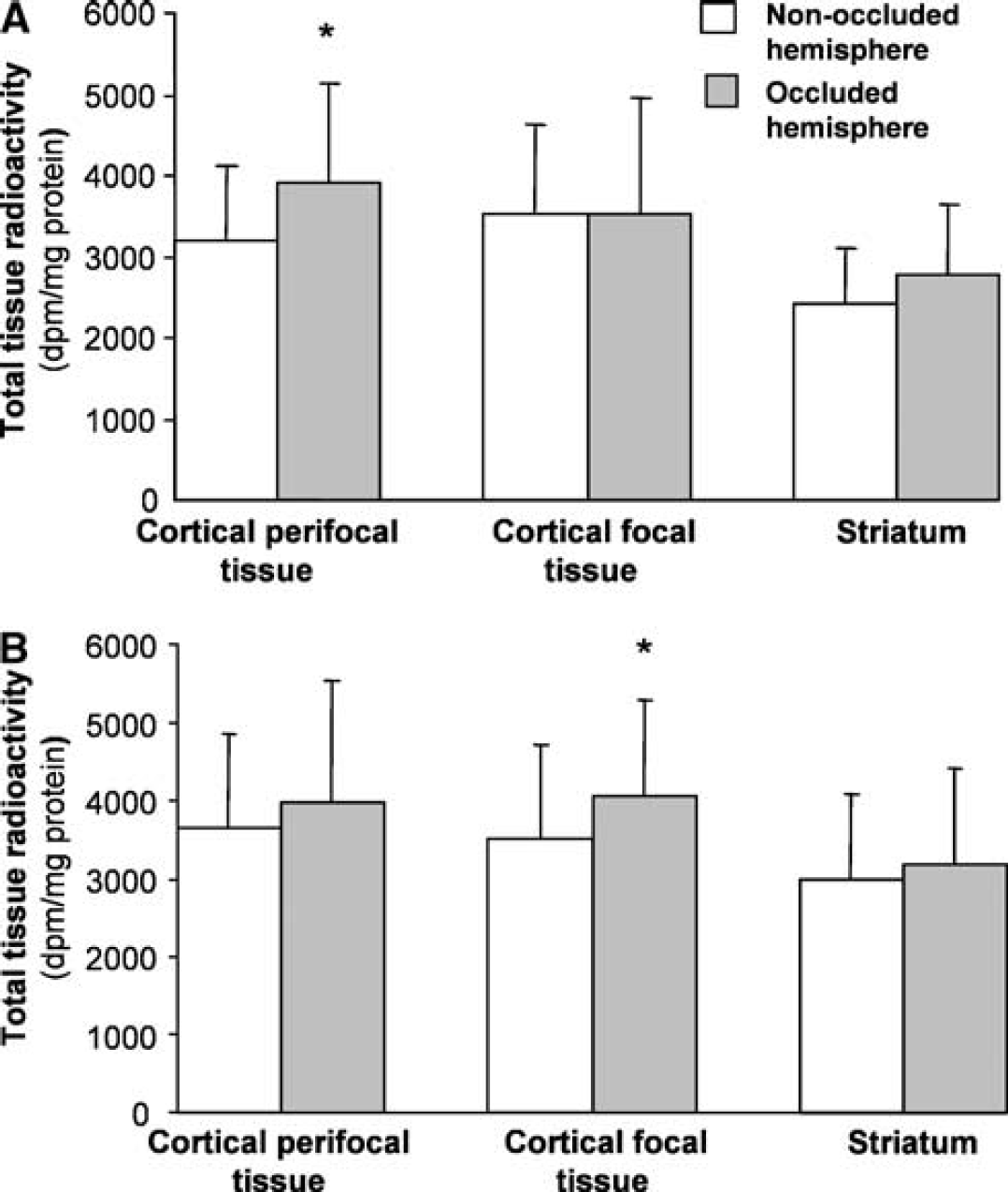
Total radioactivity in tissue subregions at 1 hour of reperfusion after either (
The magnitude of the changes in 14C-glutamine content in the tissue subjected to ischemia could possibly be moderated by diffusion of labeled glutamine directly from adjacent brain tissue or via the circulation from other tissues. To test this possibility, 14C-acetate metabolism was assessed in two rats at 48 hours after 3-hour MCA occlusion, a time at which extensive astrocytic loss in the infarcted tissue was expected. Most of the glutamine labeling was lost in the three regions exposed to ischemia in these two rats with an average decrease of 84%±11% compared with the corresponding contralateral regions. (Values were expressed relative to tissue wet weight to avoid the effects of protein loss at this late post-ischemic time.) The effects were particularly pronounced in the cortical focal tissue, which showed an average decrease for the two rats of 92%. Marked changes were also seen in the striatum and perifocal cortical tissue, with both regions showing average decreases of 79% compared with corresponding contralateral samples. Despite the damage produced at this late reperfusion time, total tissue radioactivity in the affected regions showed only moderate reductions (averaging 23%±18%, compared with values in the equivalent contralateral regions in the two rats). These findings indicate that almost all of the 14C-glutamine production detected in the studies was the result of local astrocytic metabolism. The minor residual activity seen at 48 hours after MCA occlusion might have been because of small numbers of preserved astrocytes in the tissue that was sampled or the diffusion of 14C-glutamine from other sites.
The total tissue contents of glutamine and glutamate were measured in the samples obtained at 1 hour of recirculation to assess possible changes in these metabolic pools that may influence the incorporation of 1-14C-acetate into glutamine. The results are presented in Table 2. Small but significant reductions in glutamate content were seen in the striatum after both ischemic periods with cortical perifocal tissue also showing a significant decrease after 3 hours of ischemia. Glutamine content was very similar in comparable regions from the two hemispheres after both 2 and 3 hours of ischemia. Thus, the pattern of 14C-glutamine specific labeling expressed relative to glutamine content (results not shown) was not substantially different from that expressed per mg tissue protein (Figures 3 and 4).
Effects of 2 or 3 hours of MCA occlusion and 1 hour of reperfusion on total glutamine and glutamate content (mmol/mg protein) in brain tissue subregions
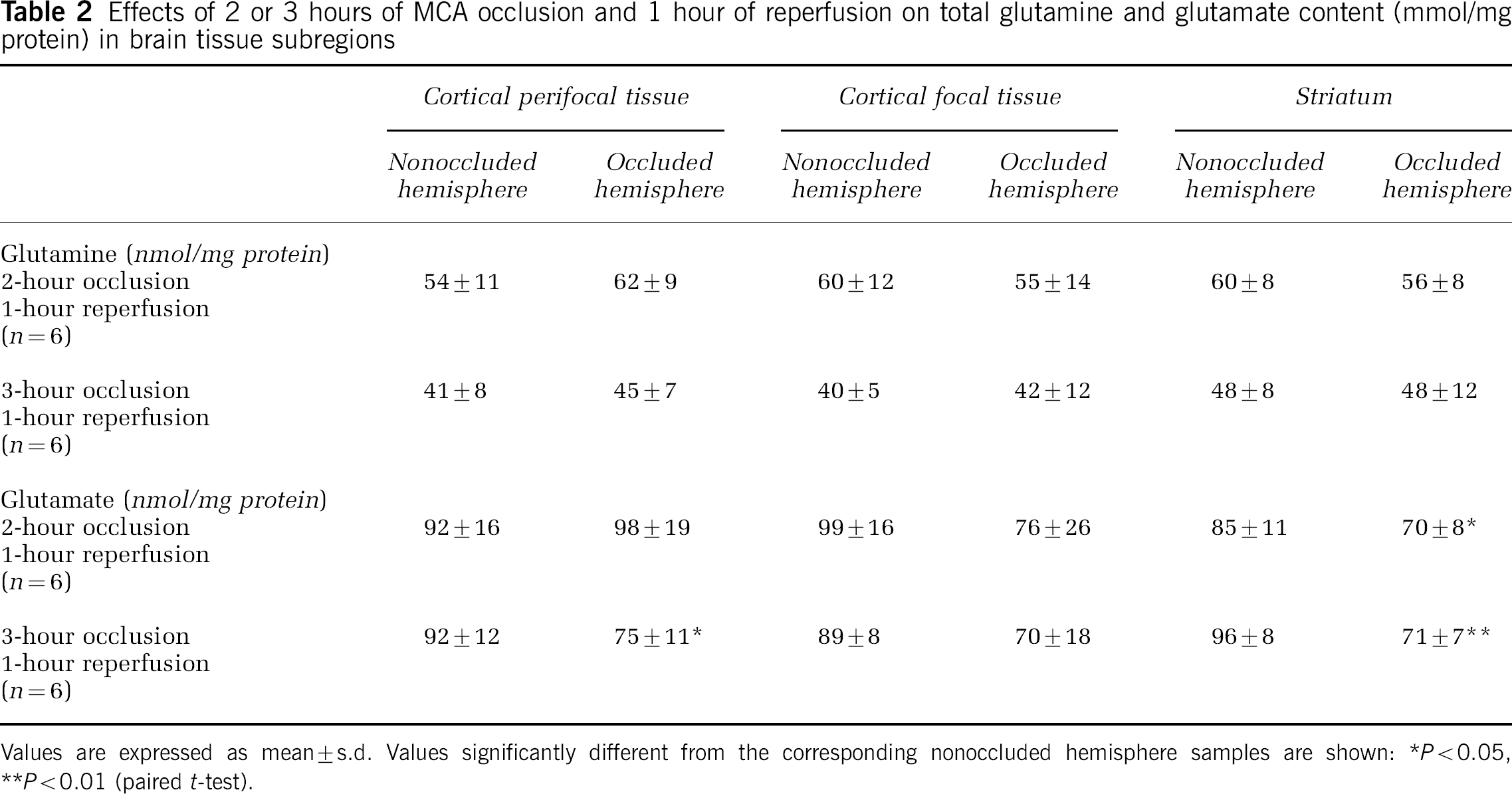
Values are expressed as mean±s.d. Values significantly different from the corresponding nonoccluded hemisphere samples are shown:
P<0.05,
P<0.01 (paired t-test).
As expected from previous studies of the metabolism of 1-14C-acetate (Berl and Frigyesi, 1969; Haberg et al, 2001; Hassel et al, 1995), 14C-radiolabel was also incorporated into glutamate, but at a lower specific activity than glutamine. The initial production of 14C-glutamate from 14C-acetate is also predominantly astrocytic based on the substantial impairment produced by the selective glial aconitase inhibitor, fluorocitrate (Fonnum et al, 1997). Consistent with the findings with glutamine, incorporation of radiolabel into glutamate in the present study was substantially decreased in the striatum and was essentially preserved in the cortical perifocal tissue (Figure 6). However, a significant decrease was seen in radiolabel in glutamate in the cortical focal tissue after both ischemic periods when expressed relative to total protein (Figure 6). This difference was also significant (P<0.05) after 2-hour ischemia but not 3-hour ischemia when expressed based on the glutamate content of the tissue (results not shown).
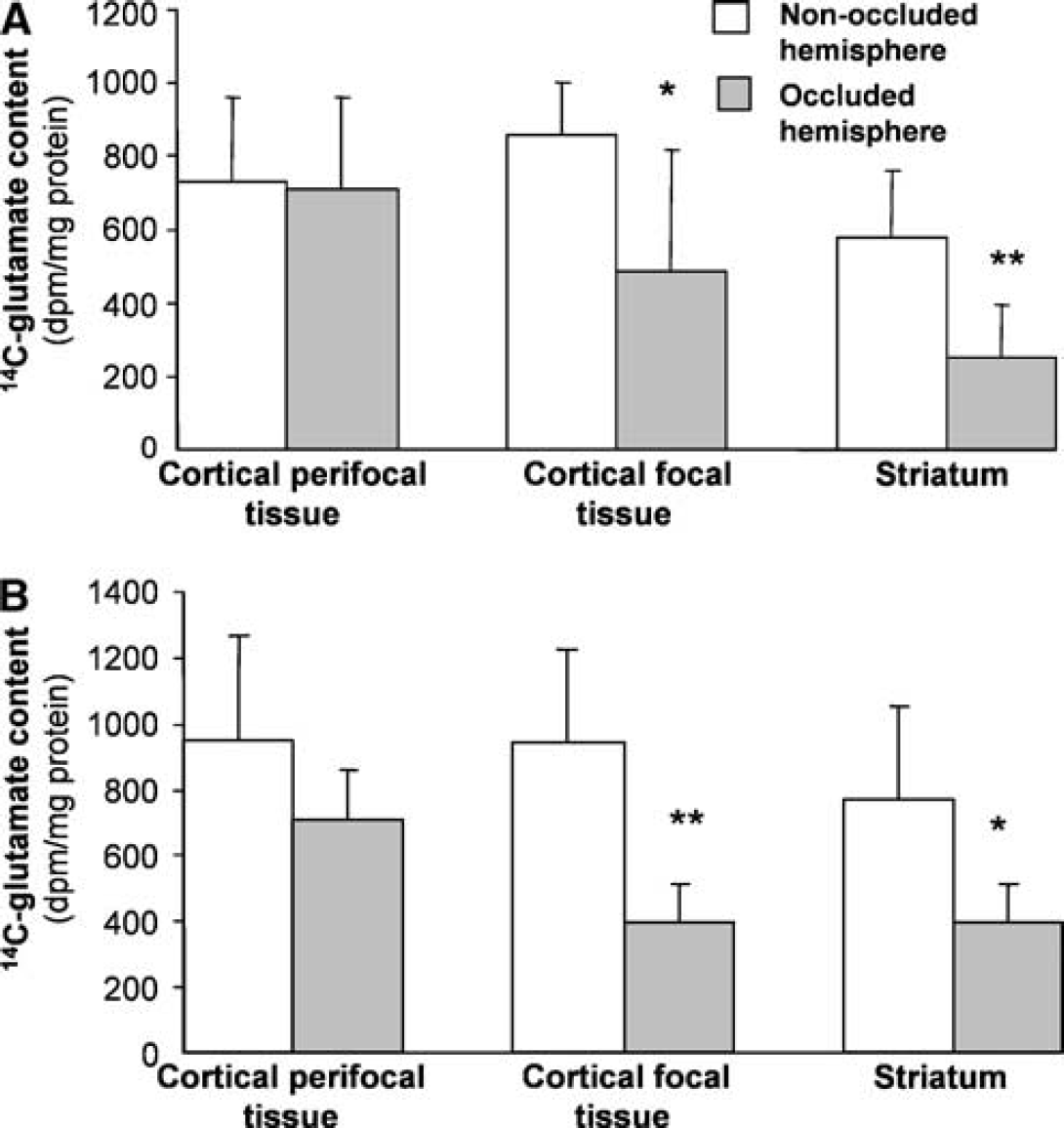
Incorporation of radiolabel from 1-14C-acetate into glutamate at 1 hour of reperfusion after either (
Effects of Ischemia with Extended Recirculation on Astrocytic Oxidative Metabolism
The metabolism of 1-14C-acetate was subsequently investigated at 4 hours of reperfusion after 3 hours of ischemia to test for possible deterioration of astrocytic function during development of the infarct. Interestingly, the relative metabolic activity in the treated and contralateral hemisphere was very similar to that seen at 1 hour of reperfusion (Figure 7). In the striatum, there was again a significant decrease in incorporation of radiolabel, with all rats showing lower values for the hemisphere subjected to ischemia compared with tissue from the contralateral hemisphere. There was no significant difference in either of the cortical regions investigated. As at the earlier recirculation time, the relative response of the two hemispheres was more variable in the focal tissue, with two of the five rats exhibiting large reductions (47% and 69% decreases) in the hemisphere subjected to MCA occlusion.
Incorporation of radiolabel from 1-14C-acetate into glutamine at 4 hours of reperfusion after 3 hours of MCA occlusion. The content of 14C-glutamine in three subregions from the hemisphere subjected to MCA occlusion is compared with 14C-glutamine in the equivalent subregions from the contralateral hemisphere. Values have been expressed relative to (
Glutamate content was not significantly different between the two hemispheres for any of the regions studied at 4 hours of recirculation. However, there was a tendency for glutamine content to be higher in each of the regions and this difference was statistically significant for the cortical focal tissue (occluded hemisphere: 71±5 nmol/mg protein; contralateral hemisphere: 53±17 nmol/mg protein; P<0.05). When 14C-glutamine labeling was expressed relative to the glutamine content, there was a large decrease in the striatum in the occluded hemisphere (Figure 7B). This reduction resulted primarily from the marked decrease in 14C-glutamine production (as seen in Figure 7A) with a superimposed decrease because of subtle increases in glutamine content. A small but significant decrease in the cortical perifocal tissue was also seen when 14C-glutamine was expressed relative to total glutamine content (Figure 7B).
Discussion
This study indicates that astrocytic function, as assessed from 14C-acetate metabolism, is substantially different between brain subregions during the first few hours after focal cerebral ischemia. In the striatum, this metabolic response was consistently impaired under each of the conditions investigated. In contrast, 14C-acetate metabolism was generally preserved in the cortical perifocal tissue that was subjected to more moderate ischemia. The focal tissue within the cortex showed more variability in response between animals but metabolic activity was also largely preserved in more than half of the rats investigated. Thus, in these animals, most astrocytes in the cortex apparently remained viable after the extended periods of ischemia and retained the capacity to oxidatively metabolize acetate via the tricarboxylic acid cycle. In each of the brain subregions investigated, 14C-acetate metabolism was similarly affected at 1 and 4 hours of reperfusion after 3 hours of cerebral ischemia. Thus, for at least several hours of recirculation, this metabolic property showed little deterioration beyond the changes that had already been induced by ischemia or initial reperfusion.
The greater susceptibility of the astrocytic response in the striatum is consistent with other evidence for more rapid progression toward cell dysfunction and death in this region after temporary cerebral ischemia. Thus, other metabolic properties, including mitochondrial respiratory capacity (Anderson and Sims, 1999) and mitochondrial glutathione content (Anderson and Sims, 2002), have been found to be affected to a greater degree in the striatum than in the cortex during early recirculation. We have not directly measured cerebral blood flow during MCA occlusion in the present study. Nonetheless, the differential sensitivity of cells in this region compared with the cortical perifocal tissue seems likely to relate at least partially to differences in the severity of the ischemic insult in the two regions. Whether such differences in ischemia also contribute to the lesser effects in focal cortical tissue compared with striatum is not resolved by the present study. Previous investigations indicate that there are at most only subtle differences in reductions in flow between the focal tissue in the cerebral cortex compared with those in the striatum during MCA occlusion (Belayev et al, 1997; Bolander et al, 1989; Nagasawa and Kogure, 1989). Thus, differences in the cellular composition and properties between these regions may also contribute to the more consistent and larger changes in the striatum. Regional heterogeneity in astrocytic properties has been reported (Porter and McCarthy, 1997; Wilkin et al, 1990; Xu et al, 2001) and could contribute to the differential responses.
We have also recently identified more pronounced changes in some cell-specific proteins in the striatum compared with the cerebral cortex during early recirculation after 3 hours of MCA occlusion (Lee et al, 2003). However, the astrocytic marker, glial fibrillary acidic protein, was not altered and glutamine synthetase was increased in this brain subregion. Thus, the metabolic changes we have observed in the present study are probably not because of an early loss of astrocytes, and provide an indicator of functional deterioration in these cells.
There was little difference in the pattern of metabolic responses seen after either 2 or 3 hours of ischemia and no obvious relationship of these changes to the pattern of tissue infarction that subsequently developed. This is most obvious for the cortical perifocal tissue. Tissue infarction, which involves the death of all cells including astrocytes, usually develops throughout this region after 3-hour ischemia but is absent or incomplete after 2 hours of ischemia. However, the astrocytic response was similarly preserved after both ischemic insults. Thus, the deterioration and death of these cells is clearly markedly delayed in this region. Both insults typically produce infarcts encompassing focal tissue within the striatum and the cortex. The difference in response between these two subregions further suggests that early changes in this aspect of astrocytic oxidative metabolism are not essential for subsequent progression to tissue infarction.
While the findings of the present study suggest good preservation of the activity of parts of the tricarboxylic acid cycle in some affected brain regions, this does not necessarily indicate that the metabolism of the glucose will be similarly preserved in these cells. Temporary MCA occlusion results in marked decreases in the utilization of deoxyglucose (Belayev et al, 1997) and increases in lactate content (Folbergrova et al, 1992; Franke et al, 2000; Selman et al, 1990). Both of these findings suggest substantial alterations to glucose metabolism, although neither provides information on possible differential responses of cell subpopulations. In a recently completed study (in preparation), we have found marked decreases in the incorporation of radiolabel from glucose into glutamine after reversal of MCA occlusion even in tissue regions in which acetate metabolism was intact. This finding indicates that there are probably additional alterations affecting astrocytic metabolism of glucose at or before the generation of acetyl CoA.
The metabolism of 14C-acetate and incorporation of radiolabel into glutamine was chosen to provide a highly selective measure of astrocytic function that allowed simultaneous assessment across multiple brain regions in vivo. Comparisons of astrocytic function would have been improved by determination of actual rates of 14C-glutamine production but this would require measurements at multiple time points. The choice of a 5-min period between injection of 14C-acetate and the determination of labeled glutamine content provided a useful estimate of cell function that encompassed the period of maximum production of glutamine from the labeled acetate and ensured adequate radioactivity in glutamine for reliable detection. Because of the nonlinearity of 14C-glutamine production during the 5-min period (Berl and Frigyesi, 1969), the magnitude of observed changes is not necessarily directly proportional to the extent of functional impairment. Nonetheless, our findings of the consequences of advanced infarction on 14C-glutamine generation provide one indicator of the usefulness of this approach in detecting severe loss of astrocytic function. Furthermore, similar approaches examining 14C-glutamine content at short periods (5 to 10 mins) after injection of radiolabeled precursors have successfully detected more subtle functional impairment because of treatment with the aconitase inhibitor, fluorocitrate (Fonnum et al, 1997; Willoughby et al, 2003), further validating the usefulness of this method.
A possible complication with the approach measuring radiolabel incorporation arises if the treatment under investigation substantially alters the pool size of metabolites between acetate and glutamine. This would lead to differential dilution of the radiolabel during metabolism in treated and control tissue. Several observations strongly suggest that such changes are not a significant issue in the present study. The glutamine and glutamate content was not greatly altered under any of the conditions investigated, suggesting that gross disruption of relevant metabolite pools did not occur. Small but statistically significant decreases in glutamate, which is the major intermediate pool in this series of reactions, were seen in some subregions at 1 hour but not 4 hours after ischemia. However, these changes showed no obvious association with the pattern of responses in specific labeling of 14C-glutamine. Such decreases, if also present in the smaller pool of glutamate contained in the astrocytes, would be expected to increase the rate of incorporation of radioactivity into glutamine. Therefore, this could not account for the significant reductions that were consistently detected in the striatum. Finally, the pattern of specific labeling of glutamate relative to samples from the contralateral hemisphere was generally similar to that seen for glutamine, further indicating that the size of the glutamate pool did not substantially influence the glutamine-specific activity.
To our knowledge, the present study provides the first detailed investigation of the effect of focal ischemia and reperfusion on an important aspect of astrocytic cell function in affected brain subregions. The findings provide evidence that astrocytic oxidative metabolism is significantly impaired in parts of the postischemic brain, particularly in the striatum. The effects on this aspect of astrocyte function in the three subregions investigated were not obviously influenced by extending the ischemic period from 2 to 3 hours and were not good predictors of the extent of tissue damage that would subsequently develop. Many astrocytes, especially in the cerebral cortex, apparently maintained this activity for many hours after prolonged temporary ischemia. These astrocytes would seem to provide a target for interventions to limit cell death with the potential to support recovery of any neurons that also remain viable during early reperfusion or to promote longer term plastic responses in the damaged tissue.
