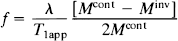Abstract
We developed a fibrin-rich thrombotic focal cerebral ischemic model with reproducible and predictable infarct volume in rats. In male Wistar rats (n = 77), a thrombus was induced at the origin of the middle cerebral artery (MCA) by injection of thrombin via an intraluminal catheter placed in the intracranial segment of the internal carotid artery (ICA). Thrombus induction and consequent ischemic cell damage were examined by histopathological analysis and neurological deficit scoring, and by measuring changes in cerebral blood flow (CBF) using laser–Doppler flowmetery (LDF), perfusion-weighted imaging (PWI), and by diffusion weighted imaging (DWI). Histopathology revealed that a fibrin-rich thrombus localized to the origin of the right MCA. Regional cerebral blood flow (rCBF) in the right parietal cortex was reduced by 34–58% of preinjection levels after injection of thrombin in rats administered 30 U of thrombin (n = 10). Magnetic resonance imaging (MRI) showed a reduction in CBF and a hyperintensity DWI encompassing the territory supplied by the right MCA. The infarct volume in rats administered 80 U of thrombin was 31.29 ± 12.9% of the contralateral hemisphere at 24 h (n = 13), and 34.7 ± 16.4% of the contralateral hemisphere at 168 h (n = 6). Rats administered 30 U of thrombin exhibited a hemispheric infarct volume of 34.0 ± 14.5% (n = 9) at 24 h and 29.7 ± 13.9% (n = 8) at 168 h. In addition, thrombotic rats (n = 3) treated with recombinant tissue plasminogen activator (rt-PA) (10 mg/kg) 2 h after thrombosis showed that CBF rapidly returned towards preischemic values as measured by PWI. This model of thrombotic ischemia is relevant to thromboembolic stroke in humans and may be useful in documenting the safety and efficacy of thrombolytic intervention as well as for investigating therapies complementary to antithrombotic therapy.
Up to 80% of human strokes are caused by thrombosis or embolism (Mohr et al., 1978; Sloan, 1987). Administration of recombinant tissue plasminogen activator (rt-PA) within three hours after onset of ischemic stroke improves neurological outcome in patients (NINDS, 1995). With the expected use of antithrombotic agents for treatment of stroke, animal models of thrombotic cerebral ischemia will be required to assess the safety and efficacy of antithrombotic therapies and to evaluate combination therapies. The beneficial effects of thrombolytic agents on increasing cerebral blood flow (CBF), reducing infarct volume, and improving neurological outcome have been demonstrated using cerebrovascular thromboembolic models induced by autologous or heterologous blood clot embolization in rabbits and rats (Zivin et al., 1985; Papadopoulos et al., 1987; Overgaard et al., 1993; Hamilton et al., 1994). However, a limitation of these models is the lack of reproducibility and uniformity in the size and location of the infarcts, since placement and ultimate lodgement of the multiple emboli are not controlled (Hill et al., 1955; Penar and Greer, 1987; Phillips et al., 1988; Overgaard et al., 1992). Photochemically-induced emboli are platelet-rich and lack fibrin (Watson et al., 1985) and, therefore, are not accessible to thrombolysis with rt-PA. Commonly-used models generate multiple and scattered emboli, and there is a need for a model to mimic the large cerebral artery thrombosis of human stroke. We report the development of a rat model of thrombotic focal cerebral ischemia, in which the middle cerebral artery (MCA) is selectively occluded by thrombosis. The formation of a thrombus at the origin of the MCA and the reproducibility of the resulting volume of infarction were confirmed by histopathology. Thrombolysis of the occluded MCA in this model was also demonstrated by means of magnetic resonance imaging (MRI) after intravenous administration of rt-PA.
MATERIAL AND METHODS
Animal model
Male Wistar rats, weighing 300–450 g, (n = 77) were used in the experiments. Animals were anesthetized with 3.5% halothane, and anesthesia was maintained with 1.0% halothane in 70% N2O and 30% O2 (vol/vol) using a face mask. Rectal temperature was maintained at 37 ± 0.5°C throughout the surgical procedure using a feedback-regulated water heating system. The right femoral artery and vein were cannulated with a PE-50 catheter for continuous monitoring of blood pressure and measurement of blood gases (pH, P
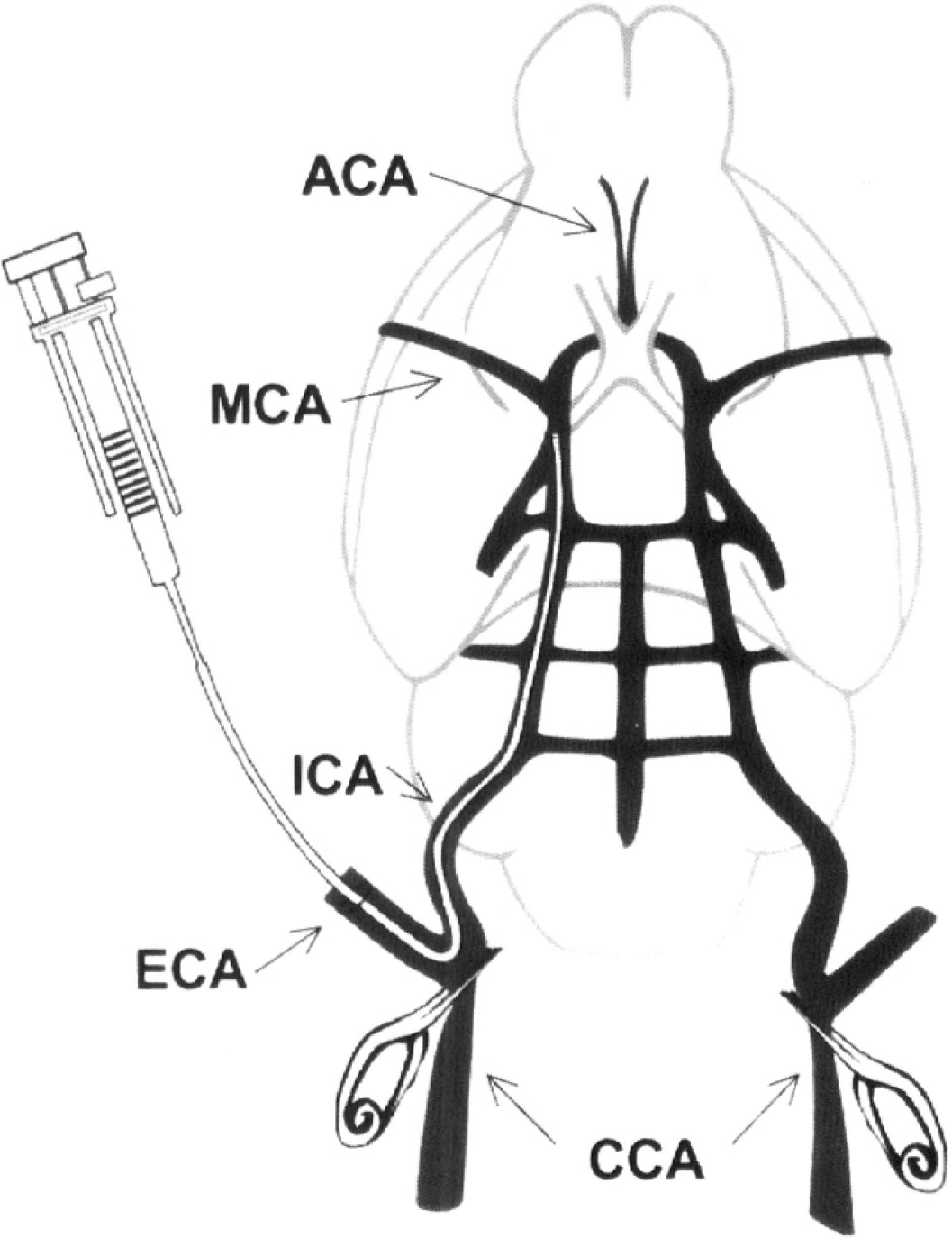
Schematic drawing of the catheter placed into the external carotid artery (ECA) and the internal carotid artery (ICA) of the rat, with its tip at 2 mm proximal to the origin of the middle cerebral artery (MCA). Thrombin is injected into the catheter. The right and left common carotid arteries (CCAs) are temporarily clipped during injection of thrombin.
Neurologic deficits
Neurologic examinations were performed at 2, 24, and 168 h after injection of thrombin. The neurologic findings were scored on a five-point scale: no neurologic deficit, 0; right Horner's syndrome, 1; failure to extend left forepaw fully, 2; turning to left, 3; and circling to left, 4.
Blood analysis
Arterial blood gas (pH, P
In order to assess the effect of thrombin on the blood clotting system, plasma levels of fibrinogen and crosslinked D-dimer were measured in rats without injection of thrombin (n = 5), with injection of 80 U of thrombin (n = 4), and with injection of 30 U of thrombin (n = 6). Blood from the femoral vein (2.7 ml) was collected in a tube containing 3.2% sodium citrate anticoagulant at 3 h after injection of thrombin. Blood samples were cetrifuged at 1,000 g at 10°C for 15 min. The supernatant fractions were stored at −70°C. The plasma level of fibrinogen was measured on a fibrometer using a fibrinogen assay kit (Pacific Hemostasis, Hunterville, NC, U.S.A.) (Clauss, 1957) and the plasma level of D-dimer was measured using a D-dimer assay kit (Pacific Hemostasis).
Monitoring of regional CBF (rCBF) by laser–Doppler flowmetery (LDF)
Relative rCBF, f, was measured using LDF. LDF was performed with a PeriFlux PF4 flowmeter (Perimed AB, Jrflla, Sweden) with relative flow values expressed as perfusion units. The rat was mounted on the stereotactic frame. A 1.5-mm diameter burr hole was placed 1.0 mm posterior and 5.5 mm lateral to the bregma in each hemisphere (Paxinos and Watson, 1986). The dura was left intact and mineral oil was placed onto the burr hole. Using a micromanipulator, two probes were positioned 0.5 mm above the dural surface, with care taken to avoid any large vessels. rCBF of two hemispheres was simultaneously measured prior to injection of thrombin (to calculate baseline flow) and 30 min to 2 h after injection of thrombin. LDF probes were repositioned as accurately as possible so that after injection of thrombin, relative flow could be measured. We found <10% changes in rCBF (p > 0.05) when probes were repositioned several times on rats without thrombus induction. rCBF was recorded every 5 min and was expressed as a percentage of preischemic baseline values.
MRI measurements
Diffusion-weighted imaging (DWI) and perfusion-weighted imaging (PWI) were performed with or without rt-PA treatment. MRI measurements were performed using a 7 T, 20-cm bore superconducting magnet (Magnex Scientific, Abingdon, U.K.) interfaced to a console (SMIS, Surrey, England). A 12-cm bore gradient coil set, capable of producing magnetic field gradients up to 20 gauss/cm and a birdcage coil with 5 cm interdiameter were used. Images were produced using a 3.2-cm field of view (FOV), 2-mm slice thickness and 128 × 64 image matrix. Stereotaxic ear bars were used to minimize movement during the imaging procedure. While in the magnet, animals were anesthetized using a face mask with a 70% of N2O and 30% of O2 (vol/vol) gas mixture, and halothane (0.75–1%). Rectal temperature was maintained at 37°C using a feedback-controlled water bath. A modified FLASH (Frahm et al., 1986) imaging sequence was employed for reproducible positioning of the animal in the magnet at each DWI session.
DWI was performed prior to injection of thrombin, continuously for 5 h, and at 24, 48, and 168 h after administration of thrombin, and with or without rt-PA treatment.
Perfusion measurements were performed using a technique described by Williams et al. (1992). This method for measuring relative CBF is based on the selective inversion of blood water protons at the level of the carotid arteries prior to [1H] MRI measurement in the brain. Perfusion images were obtained prior to injection of thrombin and at various times between 1–5 h and at 1, 2, and 7 days after administration of thrombin (and rt-PA). Perfusion measurement MRI parameters were: repetition time (TR) = 2 s, echo time (TE) = 30 ms, 64 × 64 image matrix, 3-mm slice thickness, and a 3.2-cm field of view. The duration of the inversion pulse was 2 s at a power level of 0.5 W. Relative CBF, f, was calculated using the equation (Williams et al., 1992)
where Mcont and Minv are the control and inversion image intensities, respectively; λ is the blood–brain partition coefficient; and Tlapp is the apparent T1 relaxation time constant of brain. Values of 0.9 g ml–1 for A and 1.7 s for Tlapp (Williams et al., 1992) were used for CBF calculation. After the final MRI measurement, the brain was removed, and 6-μ thick coronal sections were obtained and stained with hematoxylin and eosin (H&E) for evaluation of ischemic cell damage.
Histopathology
Light microscopy.
Animals were anesthetized with an intramuscular injection of ketamine (44 mg/kg/body wt/body wt) and xylazine (13 mg/kg body weight). Rats were transcardially perfused with heparinized saline and 10% buffered formalin, and brains were removed. Using a rat brain matrix, each brain was cut into 2-mm thick coronal blocks, for a total of seven blocks per animal. The brain tissue was processed, embedded, and 6-μm thick paraffin coronal sections from each block were cut and stained with H&E for histopathological evaluation.
Gross hemorrhage, defined as blood evident to the unaided eye on two faces of each block and on the H&E stained sections, was evaluated in each animal.
Formation of thrombi in the origin of the MCA was examined on the H&E-stained coronal sections. Phosphotungstic acid hematoxylin (PTAH) staining was performed on 6-μm thick coronal cerebral sections to examine fibrin deposition (Futrell et al., 1989; Heye et al., 1993). Other organs (heart, lung, liver, spleen, and kidney) were also evaluated for fibrin deposition.
Electron microscopy.
Rats were perfused with saline and 3% glutaraldehyde in cacodylate buffer. A 5-mm segment of the right intracranial ICA, including the origin of the MCA, was removed and fixed in 3% glutaraldehyde in cacodylate buffer for 24 h.
For scanning electron microscopy, the segment was longitudinally exposed. The tissue was then postfixed in 1% buffered osmium tetroxide for 2 h, dehydrated through progressive concentrations of ethanol for 5 min each, immersed in hexamethyldisilizane (HMDS) (Polysci, Warrington, PA, U.S.A) for another 5 min, and dried in air at room temperature. The vessel was sputter-coated with gold and examined under a JSM-840 scanning electron microscope (JEOL, Peabody, MA, U.S.A).
For transmission electron microscopy, blocks were postfixed in 1 % buffered osmium tetroxide for 2 h, dehydrated in a graded series of ethanol, and embedded in araldite. Ultrathin sections, counterstained with uranyl acetate and lead citrate, were examined using a Philips EM 201C transmission electron microscope (Philips Electronic Instruments, Mahwah, NJ, U.S.A.).
Confocal microscopy.
Evans blue dye (1 mg/kg body wt) was injected intravenously 10 min prior to injecting thrombin (30 U). Then, 2 h after injection of thrombin, animals were perfused with buffer containing 4% paraformaldehyde. This procedure removes Evans blue dye from patent vessels, with focal retention of the dye, indicating occluded cerebral vessels and ischemic tissue not accessible to reperfusion. The brain was removed and fixed in the same buffer for 3 days. Vibratome coronal sections (100 μm) were analyzed with a Bio-Rad MRC 1,024 laser scanning confocal microscope mounted onto a Zeiss microscope (Bio-Rad, Cambridge, MA, U.S.A.). The Evans blue dye produced a bright red color during excitation with the following second filter combinations: a 545-nm primary filter, a 580-nm dichroic interference mirror, and a 590-nm secondary filter. A 10 × objective was used for data acquisition. Areas of interest were scanned in 512 × 512 pixel format in the x/y direction using a 4× frame scan average. Twenty thin optical sections were scanned through a specimen along the z-axis with a 5-μm step size.
Measurement of infarct volume
Volume of cerebral tissue infarction was measured using a Global Lab Image analysis program (Data Translation, Marlboro, MA, U.S.A.). Each H&E-stained coronal section was evaluated at 2.5 × magnification. The area of infarction and the area of both hemispheres (mm2) were calculated on H&E-stained sections by tracing these areas on the computer screen; volumes (mm3) were determined by integrating the appropriate area with the section interval thickness. To reduce errors associated with processing of tissue for histological analysis, the infarct volume was corrected using the method of Swanson et al. (1990). The area of infarction in each section was expressed as the percentage of the infarct to the area of the contralateral hemisphere, and the infarct volume was also expressed as the percentage of infarct to the contralateral hemisphere (Jiang N et al., 1994).
Triphenyltetrazolium chloride (TTC) staining was also performed to demonstrate infarction 24 h after thrombin injection. Brain sections from selected animals were incubated in isotonic phosphate-buffered saline (pH 7.4) containing 2% TTC (Sigma) at room temperature for 30 min and then stored in 10% neutral buffered formalin. Brain sections were photographed 3–5 h after fixation.
rt-PA treatment
At 2 h after successful induction of thrombosis documented by PWI and DWI measurements, rt-PA (Genentech, Inc., South San Francisco, CA, U.S.A.) was administered at a dose of 10 mg/kg body wt as a 10% i.v. bolus and the remainder continuously infused (i.v) over a 30-min interval using a Harvard pump (Harvard Apparatus, South Natick, MA, U.S.A.).
Experimental protocols
To evaluate thrombosis formation and the subsequent induction of a focal ischemic lesion, the following experiments were performed:
High-dose of α-thrombin (80 U). Animals were killed 24 h (n = 18) and 168 h (n = 8) after injection of thrombin and the volume of cerebral infarction was measured.
Low dose α-thrombin (30 U). Animals were killed 24 h (n = 12) after injection of thrombin and the volume of cerebral infarction was measured.
CBF measurement to correlate relative rCBF reduction measured by LDF with cerebral infarct volume. Animals (n = 10) were killed 168 h after injection of 30 U thrombin and infarct volume was measured.
Confocal microscopy to examine the retention of Evans blue dye in the occluded MCA territory. Animals (n = 2) were killed 2 h after injection of 30 U of thrombin.
Electron microscopy to examine the composition of the thrombus. Animals (n = 3) were killed 2 h after injection of 30 U of thrombin.
Thrombolysis to evaluate, by means of MRI, the effect of administration of t-PA on thrombolysis (n = 3).
Control experiments. To (a) evaluate the effect of α-thrombin (80 U) on ischemic cell damage when α-thrombin was injected through an extra cranial segment of the ICA (n = 4) and (b) determine the effect of the transient reduction of CBF on ischemic cell damage when the right and left CCA were temporarily clamped without injection of thrombin (n = 4).
Statistics
Data were analyzed using one-way analysis of variance (ANOVA) followed by a Bonferroni t-test correction, paired t-test, and linear regression analysis. Values are presented as means ± SD, except for the neurological deficit scores, which are reported as medians and interquartile ranges, the 25–75th percentiles. Statistical significance was set at p < 0.05.
RESULTS
Physiological parameters
Arterial blood gas values were within normal physiological ranges (Table 1). The mean arterial blood pressure (MABP) was within a normal physiological range prior to injection of thrombin (102 ± 6.9 mm Hg) and 2 h after injection of 30 U of thrombin (103 ± 6.0 mm Hg).
Physiological parameters
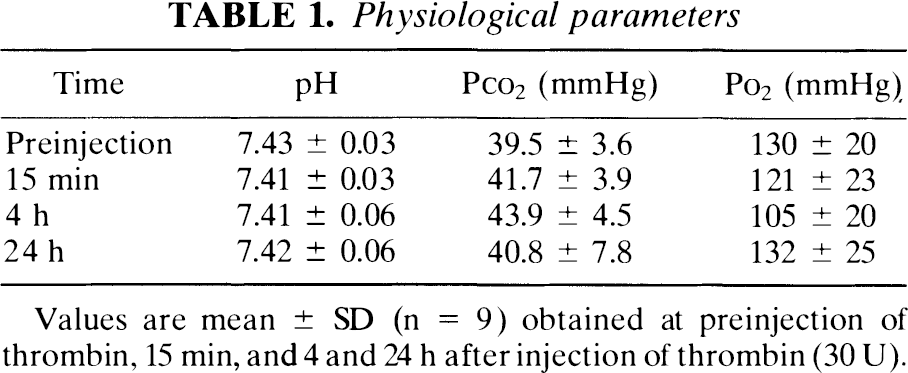
Values are mean ± SD (n = 9) obtained at preinjection of thrombin, 15 min, and 4 and 24 h after injection of thrombin (30 U).
Plasma levels of fibrinogen and D-dimer
The plasma level of fibrinogen was 222 ± 11.7 mg/dl in the control group (animals were not subjected to surgical procedures) (n = 5), 68 ± 20.2 mg/dl in the 80 U thrombin group (n = 4) and 188 ± 15.1 mg/dl in the 30 U thrombin group (n = 6), respectively. There was a significant (p < 0.05) difference in plasma fibrinogen levels between the control and the thrombin groups. The plasma level of D-dimer was <500 ng/ml in all groups.
Neurological deficits
Animals (n = 15) killed at 24 h in the group administered 80 U of thrombin exhibited a median neurological deficit score of 3 with an interquartile range of (2,4) at 2 h and 2 (2,2) at 24 h; animals in 30 U group (n = 10) exhibited scores 3 (2,3) at 2 h and 2 (1,2) at 24 h; rats killed at 168 h in 80 U group (n = 7) had scores of 3 (2,4) at 2 h and 2 (1,2) at 168 h.
Histopathology
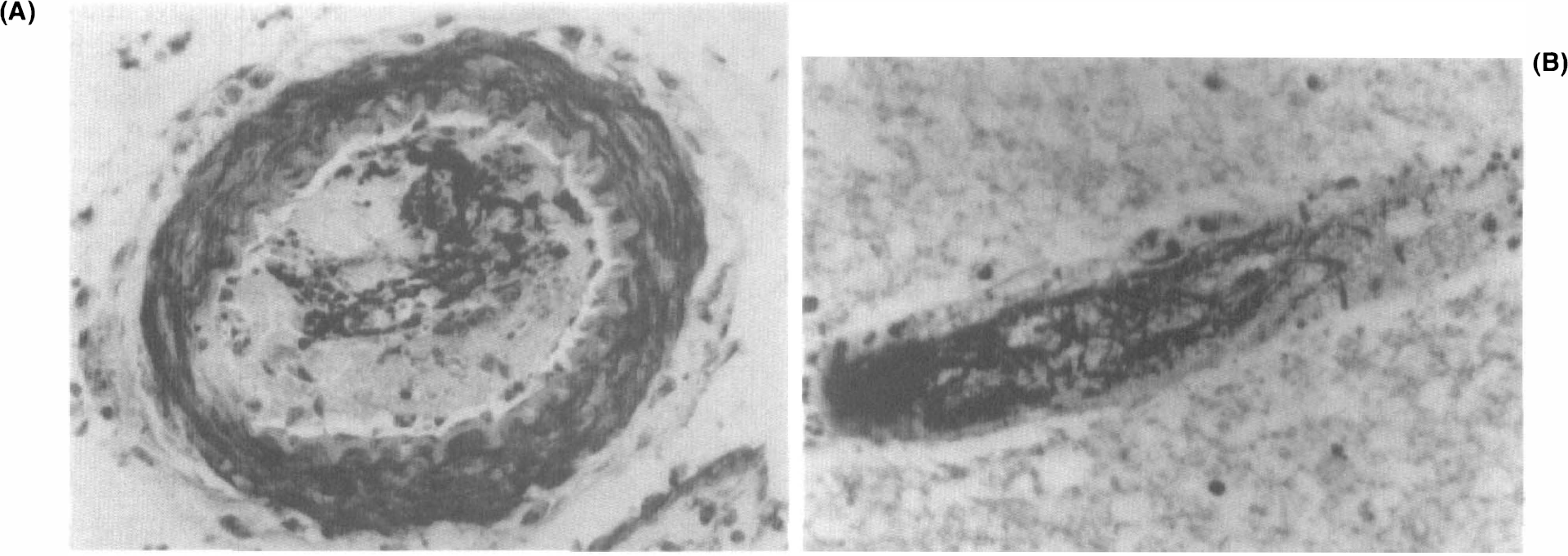
Administration of thrombin resulted in the formation of a thrombus at the origin of the MCA. Scanning and transmission electron microscopy of the thrombosed intracranial ICA segment revealed the thrombus to be composed of fibrin, platelets, leukocytes, and erythrocytes (Fig. 2A, B ). Fibrin deposition in the thrombus was confirmed in the ipsilateral hemisphere by PTAH staining (Fig. 3). Fibrin deposition evaluated by PTAH staining was not detected in the contralateral hemisphere or in the heart, lung, kidney, spleen, and liver of rats receiving 30 U of thrombin. However, fibrin deposition was found in the lung and kidney, but not in the contralateral hemisphere in rats given 80 U of thrombin.

Scanning electron micrograph of the right intracranial segment of the ICA at the origin of MCA 2 h after injection of thrombin (30 U).
Phosphotungstic acid hematoxylin (PTAH) staining of a coronal section of a rat 24 h after injection of thrombin (30 U).
Confocal microscopy revealed that fluorescent materials (Evans blue stain and erythrocytes) were present in the lumen of the ipsilateral cerebral microvasculature in the MCA territory (Fig. 4), but not in the contralateral hemisphere. Brightly diffuse fluorescence appeared and was primarily localized to the preoptic area and striatum.
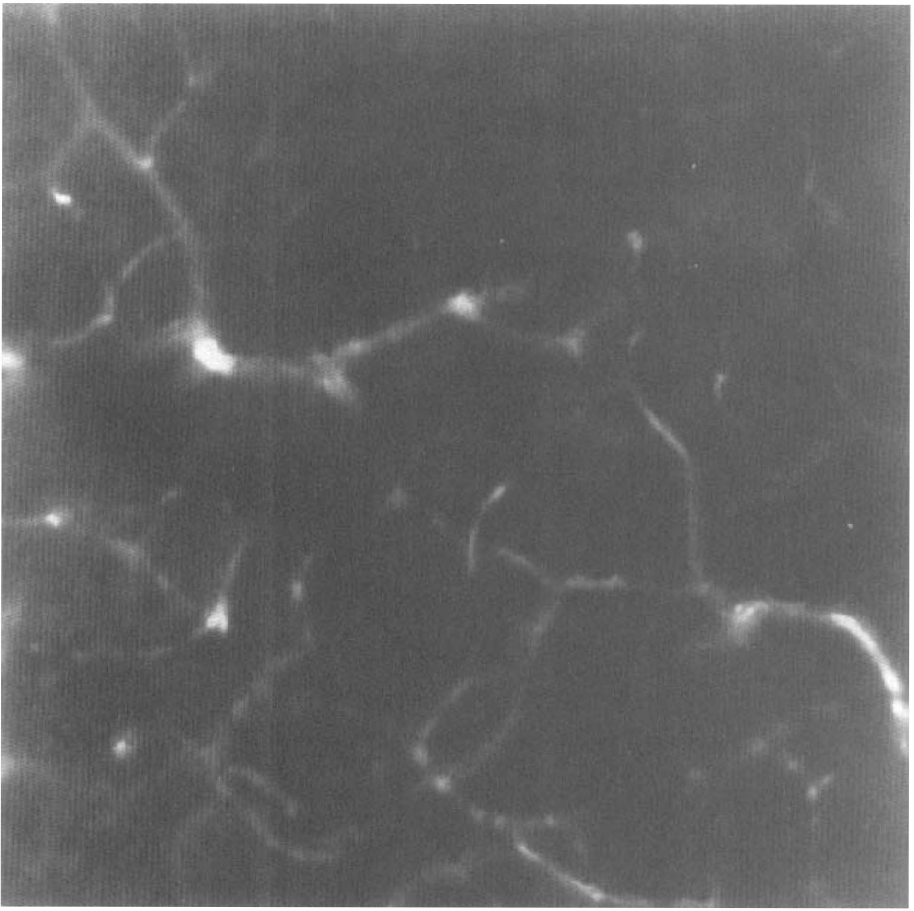
Confocal microscopy of a vibratome coronal section (100 μm) of a rat 2 h of after injection of thrombin (30 U). Fluorescent material filled the lumena of the microvasculature in the ipsilateral stratum. Diffuse fluorescence can be seen on the left side in the picture.
A thrombus was found in four of four (100%), five of six (83%), six of nine (67%), and 0 of 18 (0%) rats in the origin of the right MCA at 2, 4, 24, and 168 h after injection of thrombin, respectively. Gross hemorrhage in the ipsilateral lesion was detected in five of 12 (41%) and two of six 33% rats receiving 80 U of thrombin at 24 and 168 h, respectively, and two of nine (22%) and one of eight (13%) rats receiving 30 U of thrombin at 24 and 168 h, respectively.
rCBF and infarct volume
As measured by LDF, injection of 30 U thrombin resulted in a significant reduction in rCBF in the right parietal cortex by 58, 44, and 34% of preinjection levels at 60, 90, and 120 min after injection of thrombin, respectively; reduction in rCBF in the contralateral parietal cortex was 2, 0, and 6% of preinjection levels at 60, 90, and 120 min, respectively (Fig. 5). Linear regression analysis revealed a statistically significant (p < 0.05) correlation (r = 0.76) between rCBF reduction 75 min after administration of thrombin and cerebral infarct volume.
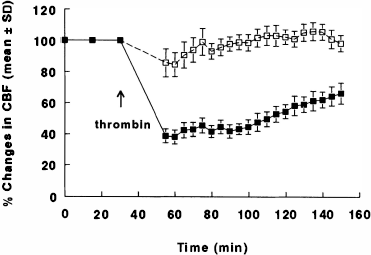
rCBF measured using LDF in the parietal cortex 30 min prior to and 2 h after injection of thrombin (30 U). n = 10; ▪, ipsilateral; and □, contralateral.
Cerebral infarction induced by thrombin administration was demonstrated by TTC (Fig. 6). Table 2 summarizes the absolute values of the contralateral and the ipsilateral hemispheric volumes, indirect infarct volumes, and percent infarct volume to the contralateral hemisphere as measured on H&E-stained coronal brain sections. In contrast, ischemic cell damage was not detected in animals infused with 80 U of thrombin through the extracranial segment of the ICA or in rats with the right and left CCAs clamped for 5 min. Three rats administered 80 U of thrombin and three rats administered 30 U of thrombin did not exhibit infarction. Rats without infarction exhibited a neurological deficit score of 0–1 at 2 h and 0 at 24 h after injection of thrombin. All other rats with infarction exhibited higher (2–4) neurological deficit scores at 2 h. Thus, a 100% success rate of infarction is achieved when neurological deficit scoring is used as an adjunctive measure. Two rats that were administered 80 U of thrombin exhibited infarction in the area supplied by the right anterior cerebral artery (ACA). These rats were excluded from the infarct volume calculation of the 24-h group. Two rats administered 80 U of thrombin and one rat administered 30 U of thrombin died after 24 h of injection of thrombin; autopsy revealed subarachnoid hemorrhage in one and massive ipsilateral hemispheric edema in two.
Absolute hemispheric volumes, indirect infarct volumes, and percent infarct volume of the contralateral hemisphere

Values are mean ± SD. 30 U and 80 U stand for rats administered 30 U of thrombin and 80 U of thrombin, respectively.
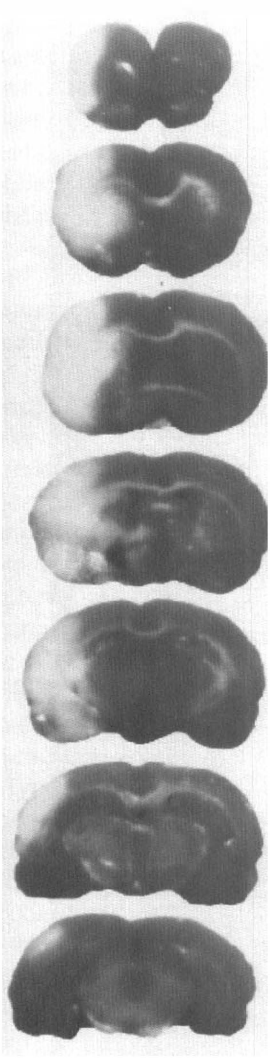
Triphenyltetrazolium chloride (TTC) staining of seven 2 mm coronal sections from a rat 24 h after injection of thrombin (30 U). Infarction was located in the territory of the right MCA.
MRI
Serial MRI perfusion measurements revealed a reduction in CBF encompassing the territory supplied by the right MCA (Fig. 7A). CBF values in the ipsilateral hemisphere after injection of thrombin declined from preinjection CBF values and remained low 3 h after injection. However, CBF in the right cortex increased towards preinjection values at 24, 48, and 168 h after injection of thrombin. CBF values in the subcortex remained low 168 h after injection of thrombin. The contralateral hemisphere exhibited a small reduction in CBF after injection of thrombin.
Thrombolysis resulting from administration of rt-PA was evident by measuring CBF with perfusion MRI (Fig. 7B). During infusion of rt-PA, CBF returned rapidly towards preischemic values. CBF values approached preischemic values in the cortex 3 h after infusion of rt-PA. Hyperemia was detected at 48 h after infusion of rt-PA. CBF was reduced in the ipsilateral hemisphere at 166 h after administration of rt-PA when compared with the preinjection CBF value.
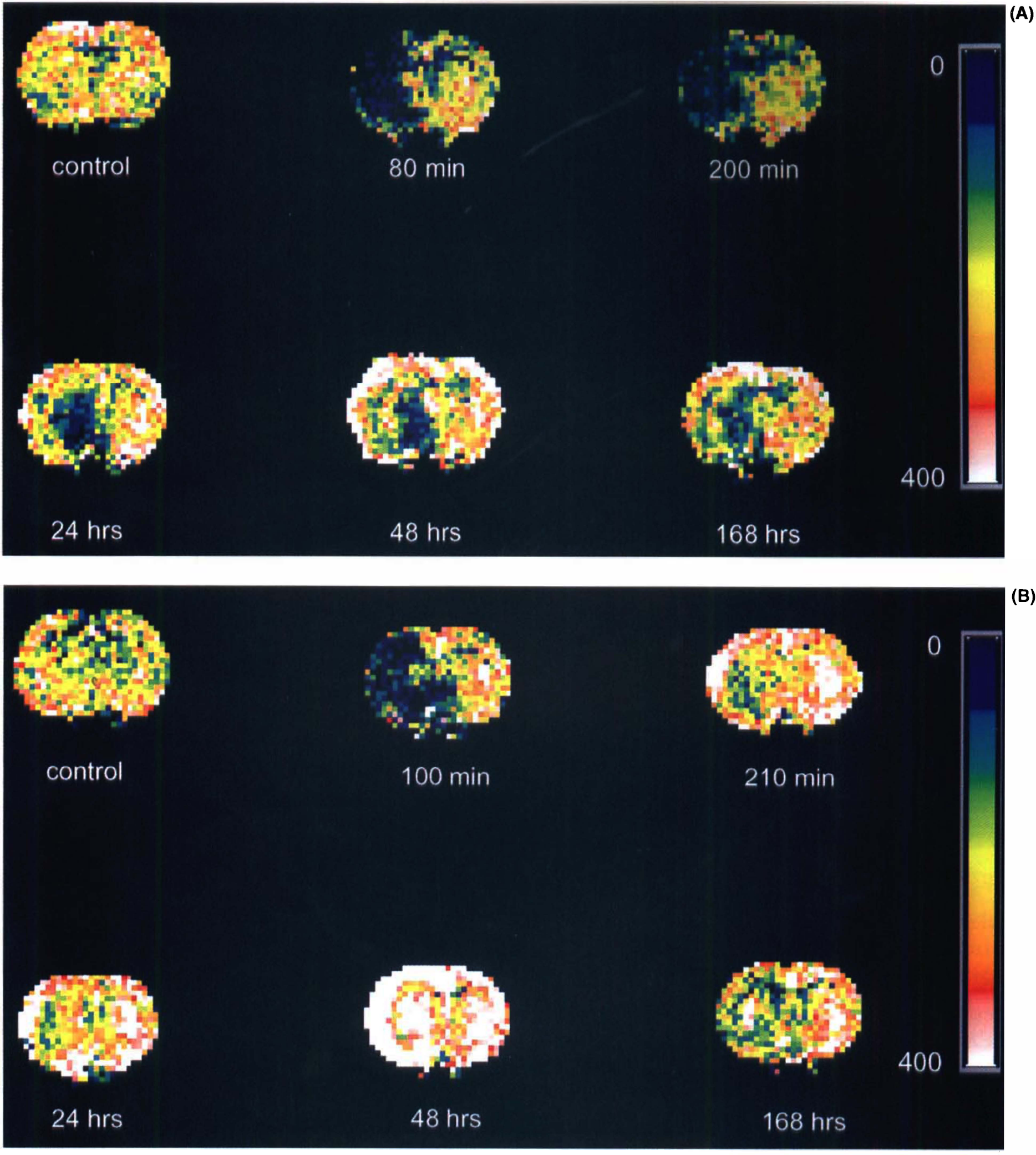
Perfusion-weighted images (PWIs) of coronal sections from a control rat brain (
Figure 8 presents DWI from rats that showed a reduction of CBF (Fig. 7A) and thrombolysis ensuing from administration of rt-PA (Fig. 7B) obtained at 80 min, and 2, 3, 24, 48, and 168 h after initiation of thrombin injection. In the non-rt-PA-treated rat, hyperintensity was apparent by DWI in the ipsilateral MCA region at 80 min after injection of thrombin; image contrast incorporating T2-weighting tended to increase as time progressed and persisted up to 168 h after injection of thrombin. In the rt-PA-treated rat, hyperintensity in the ipsilateral MCA territory was apparent within 2 h after injection of thrombin. Image contrast increased 1 h after administration of rt-PA. However, the area of hyperintensity appeared smaller at 48 h after administration of thrombin than at 100 min after administration of rt-PA. Figure 8C and D show the corresponding histological sections from the DWI and CBF images as noted in Figs. 7 and 8A,B.
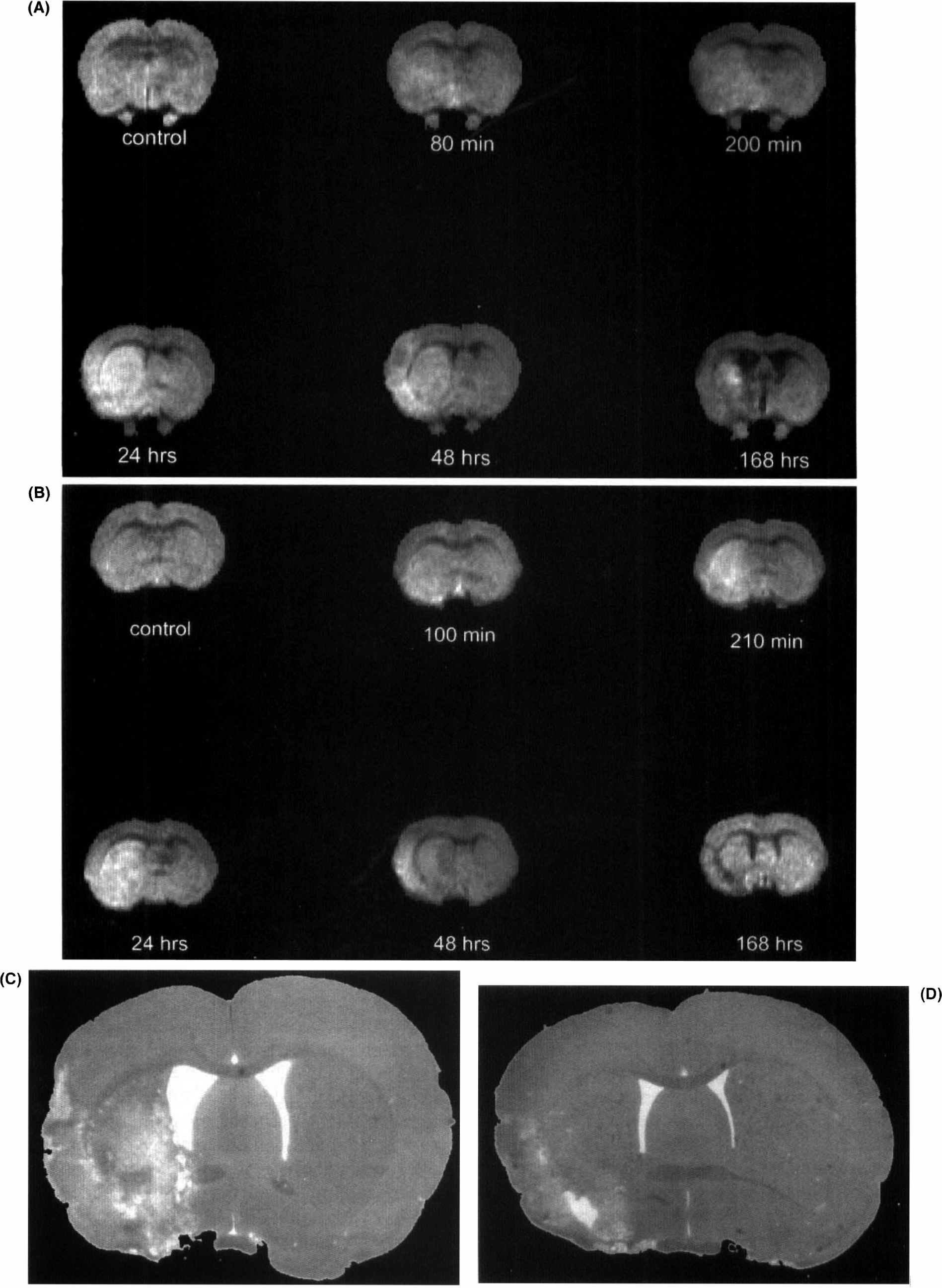
DWIs of coronal sections from the animal brains shown in Fig. 7 with (
DISCUSSION
This study presents a new model of thrombotic focal cerebral ischemia in the rat, in which a thrombus is induced at the origin of the MCA by intraluminal injection of thrombin. Thrombus induction and consequent ischemic cell damage were confirmed by histopathological analysis, neurological examination, and measuring changes in CBF (LDF, MRI, and DWI). Thrombin-injected animals in this model treated with rt-PA exhibited a rapid improvement of CBF. Therefore, this model enables quantitative study of the safety and efficacy of antithrombotic therapies in thrombotic focal cerebral ischemia.
This rat model of thrombotic focal cerebral ischemia has advantages over other thrombotic models: (a) a large vessel thrombosis similar to thromboembolic stroke in humans is produced, and the thrombus contains fibrin and is amenable to thrombolysis with rt-PA; (b) the model provides a reproducible and predictable infarct volume within the territory supplied by the MCA; and (c) the development of thrombosis and thrombolysis can be assessed noninvasively by measuring changes in DWI and PWI.
Light and electron microscopic examination confirmed that the thrombus is composed of fibrin, platelets, leukocytes, and erythrocytes. PTAH staining demonstrated a high fibrin content of the thrombus. Specimens of carotid artery plaques obtained from humans reveal a similar composition to the rat thrombin-induced thrombus (Fisher et al., 1993). Retention of Evans blue dye in the microvasculature and parenchyma in the ipsilateral preoptic area, striatum, and cortex 2 h after injection of thrombin is consistent with impairment of the blood–brain barrier (BBB) found in other models of MCA occlusion (Del Zoppo et al., 1986). Ischemic cell damage was not detected by light microscopy for animals subjected to 5-min bilateral ligation of the carotid arteries; this is consistent with the finding that 5-h bilateral carotid artery ligation does not result in cerebral ischemic lesions (Ogata et al., 1976). Taken together, these data demonstrate that a fibrin-rich thrombotic focal cerebral ischemic model is produced by precisely delivering a small volume (3–8 μl) of thrombin into the segment of the intracranial ICA 2–3 mm proximal to the origin of the MCA.
A significant reduction of CBF in the ipsilateral MCA territory was confirmed by both LDF and MRI immediately after injection of thrombin. A significant reduction of CBF persisted at least 2 h after injection of thrombin. There was a consistent correlation between the reduction of rCBF and infarct volume, with a correlation coefficient of 0.76. Reduction in rCBF after induction of a thrombus is less than that reported in the suture model of MCA occlusion (Yang and Betz, 1994). Although the LDF probe was placed 5.5 mm from the midline of the ipsilateral hemisphere, this region may not be within the ischemic core area. Consequently, the percentage reduction in rCBF may represent the perifocal area. CBF was slightly reduced in the contralateral hemisphere in the present model. Light microscopy did not reveal ischemic changes in the contralateral hemisphere. Hypoperfusion in the contralateral hemisphere may possibly be attributed to diffusion of thrombin. However, CBF reduction in the contralateral MCA territory was also observed in the suture model of MCA occlusion (Nagasawa and Kogure, 1989; Laing et al., 1993). Liang et al. (1993) reported that CBF in the contralateral MCA territory declined by 12% (at 1 h) and 11% (at 2 h) from preocclusion values measured by the hydrogen clearance method (Laing et al., 1993). Therefore, hypoperfusion in the contralateral hemisphere may represent interhemispheric diaschisis during ischemia (Slater et al., 1977).
Infarct volume in the present model is consistent with that obtained in the suture model of MCA occlusion in rats (Chopp et al., 1994; Zhang et al., 1995). Since a thrombus blocks an intracranial segment of ICA at the origin of the MCA, a relatively uniform predictable infarct volume can be produced. This is an important endpoint if therapeutic interventions, such as t-PA administration, are to be investigated. Infarct volumes are variable in other thromboembolic models, in which introduction of emboli via the extracranial segment of the ICA results in multifocal cerebral ischemia, with no control over the ultimate location of the emboli (Penar and Greer, 1987; Phillips et al., 1988; Overgaard et al., 1992). In addition, emboli induced by activation of a photosensitive dye do not contain fibrin and, thus, are not amenable to thrombolysis by administration of t-PA (Watson et al., 1985).
Evolution of the thrombotic lesion and thrombolysis in this model can be monitored by measuring DW- and PW-MRI. The hyperintensity observed on DWI after thrombin injection is consistent with data obtained from the suture model of MCA occlusion (Jiang Q et al., 1994). Treatment with rt-PA at 2 h after thrombus formation caused an increase in parenchymal perfusion, as measured by MRI.
Previous studies of rt-PA treatment of thromboembolic ischemia in the rat have shown effective thrombolysis of a heterologous blood clot, but not of an autologous blood clot (Papadopoulous et al., 1987; Penar and Greer, 1987). Using a human blood clot, Papadopoulos et al. (1987) showed that regional CBF increased significantly when rt-PA (1.5 mg/kg) was infused at 2 h after a thromboembolic occlusion of the rat MCA (Papadopoulos et al., 1987). However, Penar et al. failed to find an effect on patency of the occluded vessels when rt-PA (10 μg/kg/min for 30 min) was administered immediately following injection of autologous blood clots in a rat cerebral thromboembolic model (Penar and Greer, 1987). The fibrinolytic effect of rt-PA is species-dependent. In vitro data indicate that the specific fibrinolytic effect of human rt-PA is at least ten times less in the rat than in the human (Korninger and Collen, 1981). The therapeutic concentration of rt-PA for effective thrombolysis is 1–2mg/kg in humans (NINDS, 1995). We, therefore, employed a dose of 10 mg/kg of rt-PA. The affinity of t-PA for circulating plasminogen is low relative to the affinity of t-PA for fibrin-bound plasminogen (Sloan, 1987). In our model, the thrombotic material is fibrin-rich and is, therefore, suitable for investigating the effects of fibrinolytic agents, e.g., rt-PA, on thrombolysis. Overgaard et al. (1993) demonstrated a significant reduction of infarct volume when rt-PA (20 mg/kg) was infused 2 h after fibrin-rich clots were injected via the extracranial ICA (Overgaard et al., 1993). Since t-PA is a serine protease, it plays a role not only in fibrinolysis but also in cell migration, epithelial differentiation, tumor invasion, and a variety of other physiological processes. Therefore, a high dose of rt-PA may affect tissue in ways other than thrombolysis. However, we have not observed any effect on infarct volume or physiological response after administration of same dose of rt-PA to rats subjected to transient MCA occlusion by an intraluminal suture (n = 6) (unpublished observation). The major complication in the use of rt-PA in thrombotic stroke is intracerebral hemorrhage (Slivka and Pulsinelli, 1987; Zivin et al., 1988). With the small number (n = 3) of animals treated with rt-PA in this study, our purpose in the present study was not to evaluate the effect of t-PA either on hemorrhagic transformation of ischemic cerebral infarct or on ischemic cell damage, but, rather, to demonstrate the utility of this model for investigating thrombolytic therapy.
Gross hemorrhagic infarct was observed in 13–41% of animals. This is in agreement with data obtained from patients with ischemic stroke. Autopsy studies have revealed that 18–42% of infarcts are grossly hemorrhagic in patients with ischemic stroke (Lodder et al., 1986; Lyden et al., 1987). Gross hemorrhage occurs most often in embolic stroke in humans. Fisher and Adams (1987) reported that in two-thirds of 57 embolic stroke cases, the infarct was partly hemorrhagic (Fisher and Adams, 1987). The incidence of gross hemorrhage increases in large cerebral infarct, since large infarcts place more cerebral vessels at risk than do smaller ones (Lodder, 1984; Hornig et al., 1986). Secondary hemorrhage worsens the clinical symptoms of patients with hemorrhagic infarction through formation of hematomas (Hornig et al., 1986). However, the pathogenesis of hemorrhagic infarction is poorly defined. In the present study, we did not quantify the area of hemorrhage or correlate hemorrhage with infarct volume. Nonetheless, there were no grossly apparent differences in infarct size between hemorrhagic and nonhemorrhagic infarctions.
In our study, evaluation of neurological deficit scores revealed values similar to those reported by Zea Longa et al. (1989) in the suture model of MCA occlusion (Zea Longa et al., 1989). They observed that neurological deficit mean scores were 1.6 (circling to the left) at 2 h and decreased to 0.7 (failure to extend left forepaw fully) at 72 h after the onset permanent MCA occlusion. In comparison, we found neurological deficit median scores of 3 (circling to the left) at 2 h and 2 (failure to extend left forepaw fully) at 24 and 168 h after the onset of thrombus, respectively.
Thrombin cleaves specific arginine-glycine bonds in fibrinogen, releasing fibrinopeptides A and B (FPA and FPB from the N-terminals of the α and β chains and yielding fibrin monomer (Nossel, 1981). Thrombin has been used to induce experimental intravascular thrombosis (Collen et al., 1983; Golzarian et al., 1994). Since thrombin is injected into an end of a ligated vessel, it only reacts with the blood in the ligated vessel to form a thrombus. A potential drawback of the present method of inducing thrombosis by injecting thrombin is the difficulty in localizing thrombin, since the distal end of the ICA (intracranial segment of the ICA) is open. We overcame this difficulty by withdrawing a small volume blood (10 μl) to form a clot in the catheter and injecting this clot into the intracranial segment of the ICA to serve as a plug. Thrombin is injected along with the clot to form a local thrombus. Thrombin has a half-life of seconds when infused in vivo, due to rapid reversible binding to the vascular endothelium (Krishnamurti et al., 1993). Our technique of initial intraluminal clot formation minimizes the effects of thrombin on the circulating blood.
The plasma level of fibrinogen was within a normal range in the control animals (Baker et al., 1979). Rats administered 80 U of thrombin demonstrate a decrease in plasma level of fibrinogen associated with renal and lung fibrin deposition. These data indicate that 80 U of thrombin not only induces a thrombus in brain but also produces thrombi in other organs. However, rats administered 30 U of thrombin exhibit a mild decrease in circulating fibrinogen, without evidence of fibrin deposition in other organs except the ipsilateral hemisphere. The lack of fibrin deposition in other organs suggests that 30 U of thrombin induces a thrombus without producing systemic thrombi. In the absence of other coagulation defects, when the fibrinogen level is >100 mg/dl, the blood coagulation system is not altered (Sloan, 1987). Thus injection of 30 U of thrombin in our model produced a thrombus localized to the MCA without emboli distributed to the contralateral hemisphere or other organs. In addition, the 30 U dose of thrombin should not affect systemic blood coagulation.
D-dimer is a byproduct of the digestion of crosslinked fibrin by plasmin (Whitaker et al., 1984; Conard and Samama, 1987). It is thought to be a marker of fibrinolysis as reflected by plasmin activity on fibrin (Ono et al., 1991). The absence of change in plasma D-dimer levels found in our study may be explained by the possibility that the antibody against the human D-dimer used in our study may not cross react with rat D-dimer; alternatively, endogenous fibrinolysis may not be activated after 3 h of administration of thrombin.
Studies in stroke patients suggest that infarcted brain might activate endogenous thrombin activity. A significant increase in plasma thrombin activity has been demonstrated during the acute phase of ischemic stroke by measuring plasma levels of FPA, a marker reflecting thrombin activity (Lane et al., 1983; Feinberg et al., 1989; Tohgi et al., 1990). However, thrombin is a multifunctional protease (Coughlin, 1994) that mediates not only hemostasis and thrombosis but also inflammatory and proliferative responses (Cohen et al., 1991). One of the actions of thrombin, among the many proinflammatory actions, is to rapidly upregulate P-selectin (Sugama et al., 1992), a granular component of the Weibel-Palade body in endothelial cells and azurophilic granules of platelets (Bonfanti et al., 1989), which promotes leukocyte adhesion to endothelial cells. Leukocytes may contribute to ischemic cell damage. Therefore, one of the potential issues to be addressed is whether thrombin exerts stimulatory actions in the upregulation of P-selectin and, consequently, causes more leukocyte accumulation in this model than in other models of focal cerebral ischemia induced by non-thrombin techniques. We are currently engaged in studies that address this question.
The rCBF was higher at 2 than at 1 h after thrombosis. This suggests initiation of spontaneous thrombolysis. The frequency of the thrombus in the origin of the MCA is 83% at 4 h after injection of thrombin. Autolysis may also explain why some animals did not exhibit infarction. Spontaneous recanalization has also been observed after injection of autologous blood clots in rats (Overgaard et al., 1994). Emboli mainly blocked the ICA immediately after injection of clots. However, emboli only blocked MCA branches 3 h after injection of clots, as measured by an angiography (Overgaard et al., 1994).
In summary, we have demonstrated that a rat model of thrombotic focal cerebral ischemia induced by thrombin results in a reproducible and predictable infarct volume and permits continuous observation of the process of thrombolysis after administration of rt-PA. This model of thrombotic ischemia is relevant to thromboembolic stroke in humans and may be useful in documenting the safety and efficacy of thrombolytic intervention as well as investigating therapies complementary to antithrombotic therapy.
Footnotes
Acknowledgment:
This work was supported by National Institute of Neurological Disorders and Stroke (NINDS) grants PO1 NS23393, RO1 NS33627, and RO1 NS34184. The authors wish to thank Cecylia Powers for technical assistance, Drs. Shi-Ting Qiun and Khang-Loon Ho for electron microscopy assistance, Genentech Inc. for providing rt-PA, and Denice Janus for manuscript preparation.