Abstract
Osmotherapy is the cornerstone of medical management for cerebral edema associated with large ischemic strokes. We determined the effect of duration of graded increases in serum osmolality with mannitol and hypertonic saline (HS) on blood-brain barrier (BBB) disruption and regional cerebral edema in a well-characterized rat model of large ischemic stroke. Halothane-anesthetized adult male Wistar rats were subjected to transient (2-h) middle cerebral artery occlusion (MCAO) by the intraluminal occlusion technique. Beginning at 6 h after MCAO, rats were treated with either no intravenous fluids or a continuous intravenous infusion (0.3 mL/h) of 0.9% saline, 20% mannitol, 3% HS, or 7.5% HS for 24, 48, 72, and 96 h. In the first series of experiments, BBB permeability was quantified by the Evans blue (EB) extravasation method. In the second series of experiments, water content was assessed by comparing wet-to-dry weight ratios in six predetermined brain regions. Blood-brain barrier disruption was maximal in rats treated with 0.9% saline for 48 h, but did not correlate with increases in serum osmolality or treatment duration with osmotic agents. Treatment with 7.5% HS attenuated water content in the periinfarct regions and all subregions of the contralateral nonischemic hemisphere to a greater extent than mannitol did with no adverse effect on survival rates. These data show that (1) BBB integrity is not affected by the duration and degree of serum osmolality with osmotic agents, and (2) attenuation of increases in brain water content with HS to target levels > 350 mOsm/L may have therapeutic implications in the treatment of cerebral edema associated with ischemic stroke.
Introduction
Cerebral edema is a major cause of morbidity and mortality in patients with large hemispheric ischemic infarctions from elevated intracranial pressure or lethal compartmental shifts (herniation syndromes), resulting in compromise of vital brainstem function (Hacke et al, 1996). Osmotherapy is the cornerstone of medical therapy for cerebral edema (Bhardwaj and Ulatowski, 2004; Harukuni et al, 2002; Qureshi and Suarez, 2000; Paczynski, 1997; Schell et al, 1996). Acute administration of osmotic agents produces a potent antiedema action, primarily on undamaged brain regions with an intact blood-brain barrier (BBB), theoretically causing egress of water from the interstitial and extracellular space into the intravascular compartment, thereby improving intracranial elastance (Bhardwaj and Ulatowski, 2004; Harukuni et al, 2002; Qureshi and Suarez, 2000; Paczynski, 1997; Schell et al, 1996). In addition to causing ‘dehydration’ of the brain, osmotic agents exert beneficial nonosmotic cerebral effects, such as augmentation of cerebral blood flow (CBF), resulting in enhanced oxygen delivery, free radical scavenging, and diminished formation, and enhanced reabsorption of cerebrospinal fluid (Schmoker et al, 1991; Bhardwaj and Ulatowski, 2004; Harukuni et al, 2002; Qureshi and Suarez, 2000; Paczynski, 1997; Schell et al, 1996; Prough et al, 1991; Toung et al, 2002).
We have previously shown that maintenance of a hyperosmolar state with continuous mannitol and hypertonic saline (HS) infusion ameliorates cerebral edema associated with experimental ischemic stroke (Toung et al, 2002, 2005). Historically, it has been recommended that serum osmolality should not exceed 320 mOsm/L (Ropper et al, 2003; The Brain Trauma Foundation, 2000). However, we recently showed improved mortality and potent antiedema action with HS treatment to target levels of > 350 mOsm/L (Toung et al, 2005).
Accordingly, the present study was designed to investigate whether duration of treatment and graded increases in serum osmolality with osmotic agents alter BBB integrity after large experimental ischemic stroke. Furthermore, we studied the effects of the institution and maintenance of a hyperosmolar state with mannitol and HS therapy on regional cerebral edema in our well-characterized model of large hemispheric ischemic stroke.
Materials and methods
General Preparation and Animal Surgery
The experimental protocol was approved by the Institutional Animal Care and Use Committee, and conformed to the National Institutes of Health guidelines for the care and use of animals in research. All techniques were conducted as described previously (Toung et al, 2002, 2005). In brief, adult male Wistar rats (280 to 320 g) were anesthetized with halothane (1.0% to 2.0%) in oxygenenriched air and allowed to ventilate spontaneously. Using an aseptic surgical technique, the right femoral artery and femoral vein were cannulated. The femoral artery was ligated and the catheter was removed before emergence from anesthesia. The femoral venous catheter was exteriorized in the posterior mid-thorax and suturefixed onto a swivel at the midscapular region, which allowed the animal to move freely. Rectal temperatures (37.0°C to 38.0°C) were maintained with a heating lamp throughout the surgical procedures, and animals were housed in separate cages at ambient room temperature after emergence from anesthesia.
Focal Ischemia
Adequacy of middle cerebral artery occlusion (MCAO) was determined by laser-Doppler flowmetry (LDF) over the ipsilateral parietal cortex (Toung et al, 2002, 2005). Transient focal ischemia (2 h) was produced by MCAO using an intraluminal suture technique (Longa et al, 1989) with modifications (Toung et al, 2002, 2005; Alkayed et al, 1998). After confirmation of reperfusion for 30 mins, rats were allowed to emerge from anesthesia in separate cages and given free access to food. Rats that did not show a significant reduction in LDF signal (< 40% of baseline) during MCAO were not included in the final analysis. This degree of reduction in LDF signal over the ipsilateral cortex is necessary to ensure a consistent infarct volume in our model of MCAO (Alkayed et al, 1998).
Assessment of Blood-Brain Barrier Integrity
Blood-brain barrier permeability was assessed by the Evans blue (EB) extravasation method (Uyama et al, 1988). Briefly, EB (2% in saline; 3 mL/kg) was administered intravenously 3 h before killing. Via a thoracotomy under halothane anesthesia, intracardiac perfusion was then performed through the left ventricle with saline to remove intravascular EB dye, and continued until the fluid from the right atrium became colorless. Rats were then decapitated, the brains were quickly removed, and dissected into right (ischemic) and left (nonischemic) hemispheres. Each hemisphere was weighed and homogenized in 4 mL of 50% trichloroacetic acid solution. After centrifugation at 10,000g for 30 min, the supernatants were diluted with ethanol (1:3), and EB concentration was determined with a spectrophotometer at 620 nm for absorbance against a standard curve. Evans blue extravasation was expressed as the ratio of absorbance intensity in the ischemic hemisphere to that in the nonischemic hemisphere (EB extravasation index).
Assessment of Regional Brain Edema
After treatment with osmotic agents for desired durations, rats were killed by decapitation under deep halothane (5%) anesthesia. The brain was quickly removed and dissected into six regions, which represented the ‘ischemic core’ and the ‘periinfarct zones', from the ischemic hemisphere and from the corresponding regions of the nonischemic hemisphere (Figure 1). Brain edema was assessed by comparing wet-to-dry weight ratios (Lin et al, 1993; Toung et al, 2002, 2005). Tissues were weighed with a scale to within 0.1 mg. The dry weight of the brain regions was determined after heating the tissue for 3 days at 100°C in a drying oven. Tissue water content was then calculated as % H2O = (1-dry wt/wet wt) × 100% (Toung et al, 2002, 2005; Lin et al, 1993).
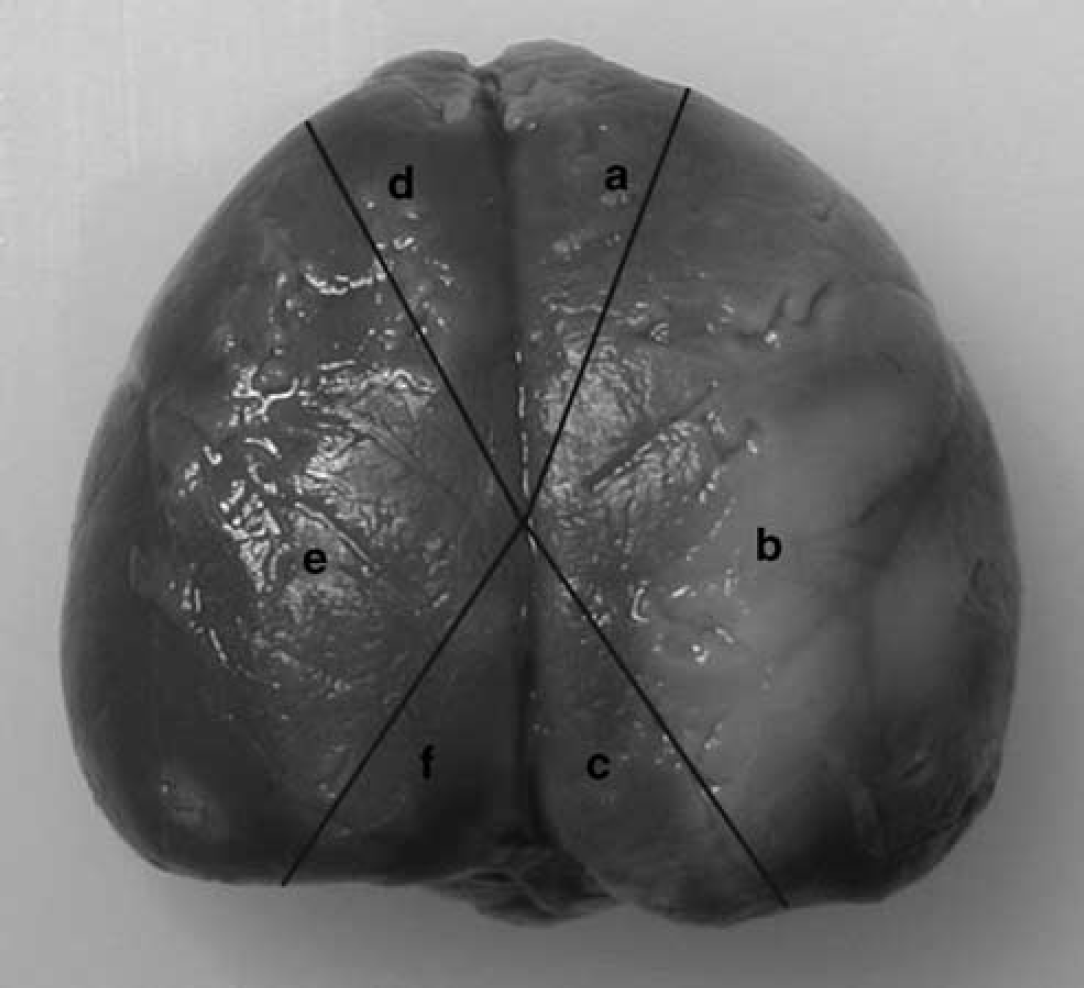
Illustrative photomicrograph of triphenyltetrazolium chloride-stained rat brain, delineating six subregions for assessment of brain edema. Region (b) represents the ‘ischemic core', while regions (a) and (c) represent ‘borderline zones' or ‘periinfarct’ regions; (d) to (f) represent corresponding regions from the contralateral nonischemic hemisphere.
Assessment of Plasma Osmolality
At the end of the experiment, a sample of blood was drawn by cardiac aspiration to determine serum osmolality (mOsm/L) with an automated freezing point depression micro-osmometer (Advanced Instruments, Inc., Norwood, MA, USA) (Toung et al, 2002, 2005).
Experimental Groups
In the first series of experiments (n = 115), all rats subjected to MCAO were randomized to receive either no intravenous fluids or continuous intravenous infusion of 0.3 mL/h (≈1.0 mL/kgh) of 0.9% saline (NS; 308 mOsm/L), 20% mannitol (1098 mOsm/L), 3% HS (1023 mOsm/L), or 7.5% HS (2310 mOsm/L). Rats that did not receive intravenous fluids were given free access to drinking water ad libitum. Hypertonic saline was instituted as a mixture of acetate:chloride (50:50; pH = 6.5 to 7.0) to prevent hyperchloremic acidosis. Treatments were started at 6 h after MCAO and continued for 24, 48, 72, and 96 h (n = 5 with each of the above treatments and at each time point). Blood-brain barrier integrity was assessed by the EB method at desired end points. Sham-operated rats (n = 5) that had all surgical procedures except for MCAO served as controls. In the second series of experiments (n = 135), rats were subjected to 2 h MCAO and treated with a continuous infusion of NS, 20% mannitol, 3% HS, and 7.5% HS. Treatments were started at 6 h, and regional brain water was assessed by wet-to-dry ratios in the six predetermined brain regions (Figure 1). Sham-operated rats served as controls (n = 10).
Statistical Analysis
All values are expressed as mean ± s.d. Physiologic parameters, plasma osmolality, and mean LDF measurements among groups were subjected to repeated-measures analysis of variance (ANOVA). Differences in the regional brain water content and EB extravasation among treatment groups were determined by two-way ANOVA with the post hoc Newman-Keuls test. The Pearson correlation coefficients between % brain water content and serum osmolality were determined. The criterion for statistical significance was P < 0.05.
Results
In the first series of experiments, physiologic parameters, including the mean arterial blood pressure (MABP), arterial carbon dioxide (PaCO2) and oxygen (PaO2), pH, rectal temperature, and hemoglobin, were within normal ranges in all experimental groups treated for 24, 48, 72 (data not shown), and 96 h (Table 1). The mortality rates before the completion of the experiment were as follows: 72 h: 3% HS—1/5; 96 h: mannitol—1/5, 7.5% HS—1/5. Laser-Doppler flowmetry signal was not attenuated to < 40% of baseline in 10 rats; therefore, they were not included in the final analysis. Serum osmolality was significantly elevated in rats treated with 7.5% HS as compared with other treatments (Table 2). The Eb extravasation index was 0.98 ± 0.2 in surgical sham controls. Blood-brain barrier disruption was maximal in rats treated with NS and mannitol for 48 h as compared with other treatment groups (Figure 2). Blood-brain barrier disruption did not correlate with increases in serum osmolality.
Summary of physiologic variables (means ± s.d.) at baseline, during ischemia, and at immediate reperfusion in rats subjected to 2-h MCAO and treated for 96 h
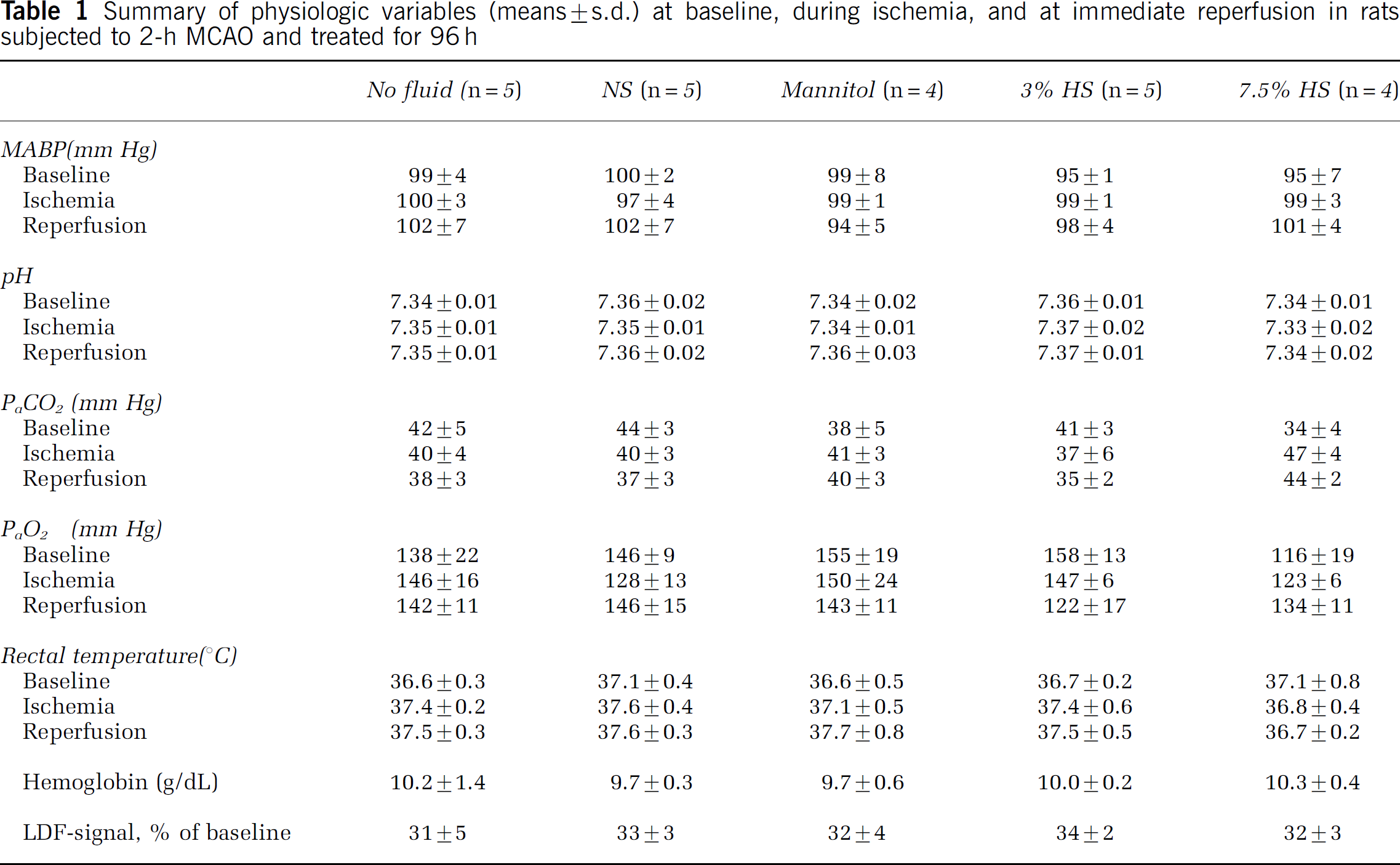
Serum osmolalities (mOsm/L; means ± s.d.) in experiments on BBB disruption
*P < 0.05, **P < 0.01, ***P < 0.001 versus 7.5% HS.
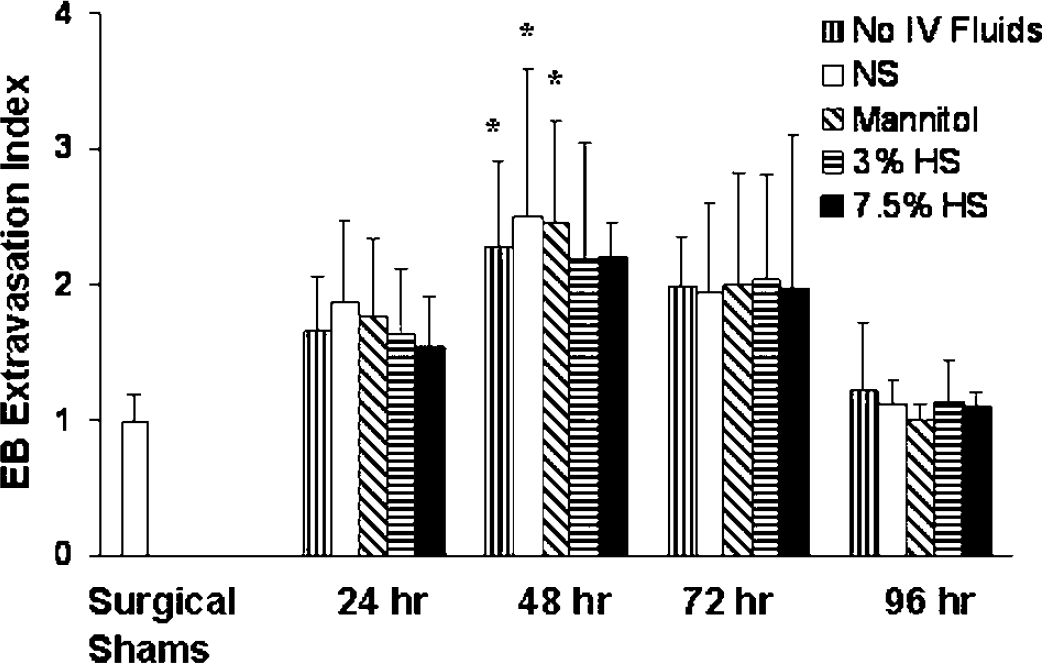
Blood-brain barrier disruption as estimated by EB extravasation, expressed as the ratio of absorbance intensity in the right (ischemic) hemisphere to that in the left (nonischemic) hemisphere (EB extravasation index). *P < 0.05 versus surgical shams (n = 5) and corresponding treatments at 96 h.
In the second series of experiments, MABP, PaCO2, PaO2, rectal temperature, and hemoglobin were within normal ranges in all treatment groups (data not shown). The LDF signal was not attenuated to < 40% of baseline in eight rats; therefore, they were not included in the final analysis. The mortality rates before the completion of the experiment were as follows: 24 h: NS—2/8, mannitol—1/7, 3% HS—1/7, 7.5% HS—2/8; 48 h: NS—2/8, mannitol—2/8, 3% HS—1/7, 7.5% HS—1/7; 72 h: NS—2/8, mannitol—2/8, 3% HS—1/7, 7.5% HS—0/6; and 96 h: NS-1/7, mannitol—2/8, 3% HS—0/6, 7.5% HS—1/7. Sham-operated rats had a serum osmolality of 302 ± 1 mOsm/L. Regional brain water content (Figure 1) was accentuated in all brain regions in rats treated with NS and was maximal at 24 h after MCAO. Treatment with 7.5% HS attenuated water content in the periinfarct regions (regions a, c) and all subregions of the contralateral nonischemic hemisphere (regions d to f) to a larger extent than mannitol and 3% HS treatment (Figure 3). There was a significant correlation between serum osmolality and brain water content for subregions c to f (Table 3; Figure 4).
Serum osmolalities (mOsm/L; means ± s.d.) in experiments on regional cerebral edema (n = 6 per treatment at each time point)
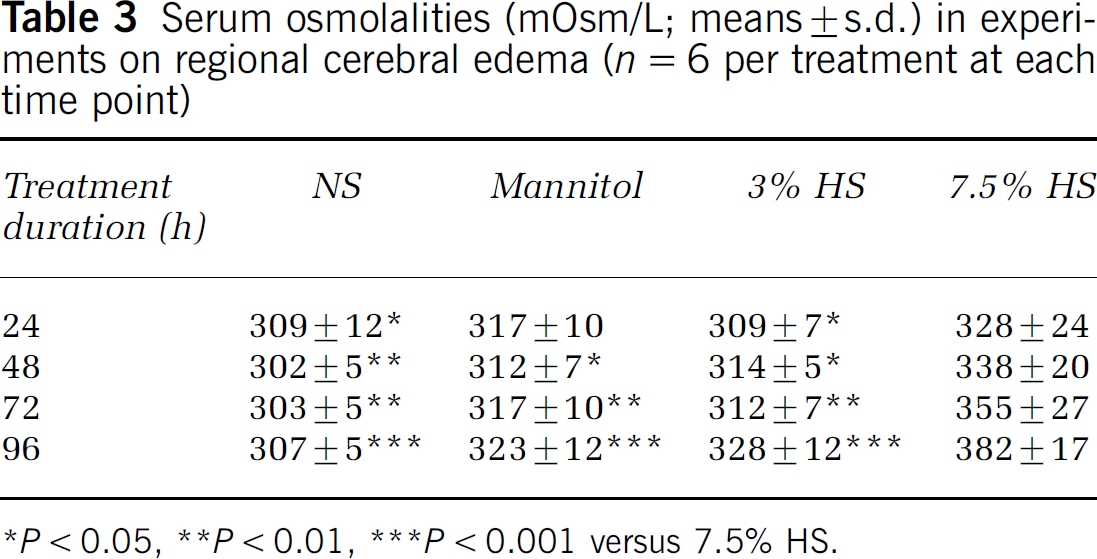
*P < 0.05, **P < 0.01, ***P < 0.001 versus 7.5% HS.
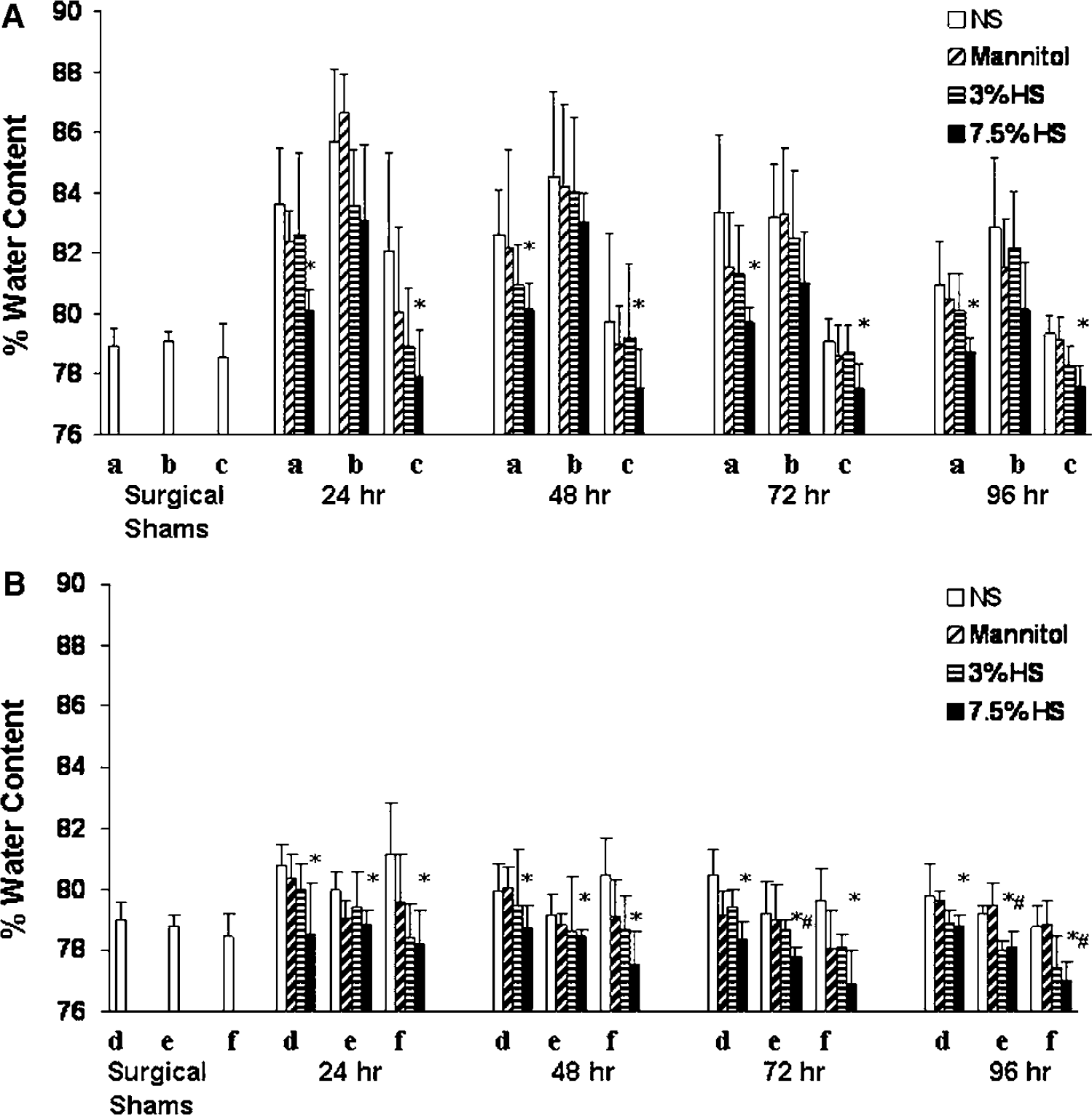
Water content in the 6 predetermined brain subregions from rats subjected to 2-h MCAO and treated with NS, 20% mannitol, 3% HS, and 7.5% HS. (
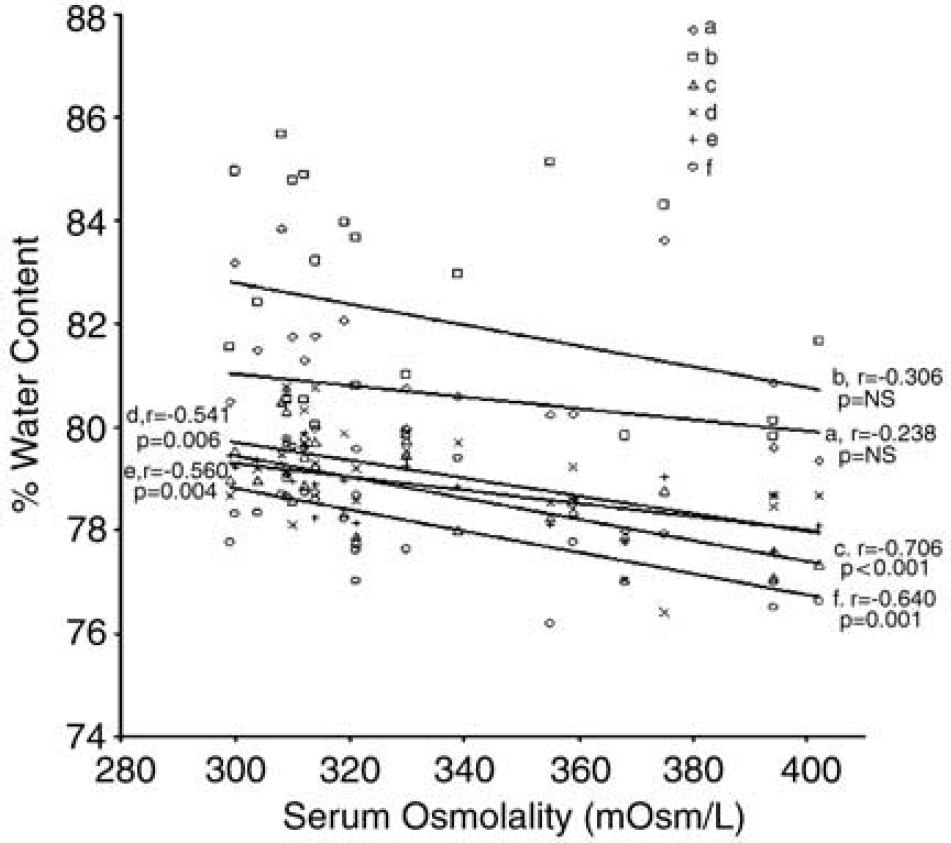
Scatter plot comparing osmolality with regional brain water content in the 4 treatment groups at 96 h. The Pearson correlation coefficient (r) is displayed on the plot and is statistically significant in regions (c) to (f).
Discussion
This study shows two novel and important findings. First, BBB integrity is not affected by graded increases in serum osmolality with osmotherapy after large experimental stroke. Second, institution and maintenance of a hyperosmolar state with HS to levels well beyond those suggested and reported in the literature attenuates the increases in regional brain water associated with ischemic stroke. These data may have important implications for the treatment of cerebral edema associated with large ischemic stroke.
Blood-Brain Barrier Integrity During Osmotherapy
Blood-brain barrier disruption after hypoxia-ischemia involves a cascade of events in which cytokines, vascular endothelial growth factor (VEGF), and nitric oxide are important mediators (Ballabh et al, 2004). Disruption of BBB and consequent vasogenic edema are known to intensify 3 to 72 h after ischemic stroke (Preston and Webster, 2004; Hofmeijer et al, 2004). Therapeutic maneuvers proposed for BBB stabilization and modulation include hypothermia (Preston and Webster, 2004) and corticosteroids (Qizilbash et al, 2003), although these strategies are not established in the ischemic stroke paradigm. Conversely, osmotherapy has been used for BBB opening and modification for more effective chemotherapy in brain tumors (Rapoport, 2001). In our model of large ischemic stroke, we observed maximal BBB disruption at 48 h after MCAO in all treatment groups. We did not observe differences in BBB permeability between different experimental groups with graded increases in serum osmolality. However, maximal BBB disruption at 48 h was observed in rats treated with no intravenous fluids, NS, and mannitol, but not with 3% or 7.5% HS (Figure 2, Table 1).
Cerebral Edema After Ischemic Stroke
In keeping with our previous studies (Toung et al, 2002, 2005), we observed increases in brain edema in the ipsilateral (ischemic) and contralateral (nonischemic) hemispheres after large experimental ischemic stroke, which were responsive to osmotherapy. The mechanisms of cerebral edema in the nonischemic hemisphere after focal ischemia are complex and not completely elucidated, but possibly include impedance of cerebral venous return from cerebral swelling, intrahemispheric diaschisis (Abe et al, 2000), increased BBB permeability by inflammatory mediators (Abbott, 2000), neurohumoral responses (Bemana and Nagao, 1999), induction of proteins, such as VEGF (van Bruggen et al, 1999), and upregulation of aquaporin-4 (Taniguchi et al, 2000).
Hypertonic saline solutions have received renewed attention as hyperosmolar agents and are being increasingly used clinically as a therapeutic modality for cerebral edema in a variety of brain injury paradigms (Bhardwaj and Ulatowski, 2004; Harukuni et al, 2002; Qureshi and Suarez, 2000; Paczynski, 1997; Schell et al, 1996; Prough et al, 1991). As sodium chloride (reflection coefficient =1.0) (Bhardwaj and Ulatowski, 2004; Harukuni et al, 2002; Qureshi and Suarez, 2000; Paczynski, 1997; Schell et al, 1996; Prough et al, 1991; Zornow, 1996) is completely excluded from brain regions with an intact BBB, it has been proposed that HS might be a more favorable osmotic agent compared with the conventional osmotic agent mannitol (reflection coefficient = 0.9). Nonosmotic properties of HS may also have a beneficial effect in brain injury, similar to those shown with mannitol (Bhardwaj and Ulatowski, 2004; Harukuni et al, 2002; Qureshi and Suarez, 2000; Paczynski, 1997). For example, HS solutions have been shown to have antiinflammatory responses, which, in turn, modulate BBB function and its permeability after brain injury (Arbabi et al, 1999). Furthermore, HS might be a more desirable agent for maintaining a ‘euvolemic hyperosmolar’ state in a variety of brain injury paradigms (Bhardwaj and Ulatowski, 2004; Harukuni et al, 2002; Qureshi and Suarez, 2000). Accordingly, in the present study, we examined the effect of mannitol and HS therapy on regional cerebral edema (injured and uninjured brain). In our previous study, the temporal profile of ischemiaevoked increases in brain water content (Toung et al, 2002) suggested a major vasogenic etiology, rather than cytotoxic edema secondary to postischemic energy failure. We have previously shown that institution of a hyperosmolar state with continuous infusion of HS when started at the onset of reperfusion in our rat model of transient focal ischemia worsened infarct volume (Bhardwaj et al, 2000). The explanation for the observed worsening of infarct volume is not readily evident at present, but the mechanism of this detrimental effect was not due to maldistribution of regional CBF It should be noted that HS was instituted as an intravenous bolus in addition to a continuous infusion at the onset of reperfusion in our previous study. Furthermore, little is known about the differential response of neurons and glia to HS solutions during an ‘evolving’ cerebral infarction. For example, in vitro studies have shown that hypertonic-hyperoncotic saline differentially affects healthy and glutamate-injured hippocampal neurons and astrocytes (Himmelseher et al, 2001). There might be competing effects of HS solutions in ischemic stroke, and the beneficial osmotic effects on stroke-associated cerebral edema might be dependent on the timing of the onset and the duration of therapy in relation to ‘maturation’ of the lesion after ischemic stroke. Based on our postulate from previous studies, we initiated osmotherapy to target vasogenic edema at 6 h after MCAO and provided effective osmotherapy for up to 96 h after the insult. However, data from the present study indicated that cerebral edema was maximal at 24 h and disruption of BBB maximal at 48 h after MCAO, suggesting that cytotoxic component plays a dominant role in stroke-associated cerebral edema in our animal model. Therapy with high-dose mannitol and 3% and 7.5% HS was used to maintain a constant osmotic gradient to cause egress of water from the brain. While a hypoosmolar state clearly amplifies brain injury (Ropper et al, 2003), a hyperosmolar state also adversely affects outcome (Bhalla et al, 2000). Although some literature suggests that a serum osmolality of 300 to 320 mOsm/L is optimal in patients with brain injury (Ropper et al, 2003; The Brain Trauma Foundation, 2000), this recommendation is not supported by systematic laboratory-based or clinical studies. Much of the evidence for this goal is indirectly supported by studies that used mannitol where its beneficial effects were offset by significant systemic side effects (Paczynski, 1997), when serum osmolality exceeded 320 mOsm/L. However, these observations have not been substantiated (Diringer and Zazulia, 2004).
We observed graded attenuation of brain water content to a hyperosmolar state with HS therapy. Wet-to-dry weight comparisons were used as a simple and reproducible assessment of brain water in brain subregions from both hemispheres. Therapy with 7.5% HS had a more robust effect on water content in the subregions of the contralateral hemisphere than in the ‘ischemic core’ and ‘periinfarct’ regions, presumably reflecting regions with intact BBB. As in our previous study (Toung et al, 2005), a serum osmolality of > 350 mOsm/L was well tolerated, with no adverse effect on survival rates in animals treated with HS. In this study, brain water content was attenuated by 1% to 2% in rats treated with mannitol and HS. As brain volume is a geometric function of water content, even minute changes in brain water content translate into relatively large alterations in brain volume. Our observations are in keeping with previous studies that have shown that a reduction of 1.6% in brain water content can translate into a 90-mL reduction in brain volume (Paczynski, 1997). The translational significance of these findings is immense, in that it can be life-saving for patients with poor intracranial elastance, resulting from large hemispheric strokes.
Our study has limitations. We did not measure serum sodium because mannitol causes a ‘pseudohyponatremia’ and hypernatremia with its use indicates volume depletion due to systemic dehydration (Paczynski, 1997). Although we instituted intravenous fluids to ensure euvolemia in our animal model, our protocols included limited measures of hydration status. Our study cannot address whether institution of osmotic agents as continuous intravenous infusion is more efficacious than bolus administration. While some investigators have shown efficacy with bolus administration, we have shown that maintenance of a constant osmotic gradient with continuous infusion of HS is beneficial in a variety of brain injury paradigms (Bhardwaj and Ulatowski, 2004; Harukuni et al, 2002; Qureshi and Suarez, 2000). We cannot comment on the diuretic effects of HS because we did not monitor urinary output in our animal model. We cannot address the potential and occurrence of ‘rebound’ cerebral edema with abrupt cessation of osmotherapy in our experimental paradigm. Although never characterized with HS therapy, a theoretical potential exists for ‘rebound’ brain uptake of water by the entry and accumulation of a nonmetabolized osmotic agent in damaged tissue through the disrupted BBB and/or secondary accumulation of idiogenic tissue osmoles (Paczynski, 1997; Schell et al, 1996). Further studies are being planned to determine the long-term outcomes after treatment in this injury paradigm.
In conclusion, our data show that maintenance of serum osmolality > 350 mOsm/L with continuous intravenous infusion of HS is efficacious in ameliorating stroke-associated cerebral edema, without causing further disruption of the BBB. These findings may have important significance in the treatment of patients with large ischemic stroke.
Footnotes
Acknowledgements
The authors thank Tzipora Sofare, MA, for her editorial assistance in preparing this manuscript and Ellen Gordes for her technical help.
