Abstract
Osteopontin (OPN) is a secreted extracellular phosphoprotein involved in diverse biologic functions, including inflammation, cell migration, and antiapoptotic processes. Here we investigate the neuroprotective potential of OPN to reduce cell death using both
Introduction
Osteopontin (OPN) is a multifunctional cytokine and adhesion protein that has been linked to a variety of physiological and pathological processes (Lee et al, 2003; Mazzali et al, 2002; O'Regan and Berman, 2000). It is a secreted glycosylated phosphoprotein found in all body fluids and exists as an immobilized extracellular matrix protein in mineralized tissues (Mazzali et al, 2002). Stimuli known to regulate OPN expression include pro-inflammatory mediators, hormones, growth factors and mechanical stressors (reviewed in Mazzali et al, 2002). Osteopontin acts on a number of cell types (e.g. endothelial cells, fibroblasts, smooth muscle cells, macrophages) via an RGD (arginine–glycine–aspartic acid) motif that binds several distinct integrins
Several observations suggest that OPN might be involved in the cellular response to ischemic injury (Ellison et al, 1999; Noiri et al, 1999; Persy et al, 1999; Wang et al, 1998). The net effect of OPN in conditions of ischemia has been difficult to ascertain because, in addition to effects that appear to promote cell survival, OPN has actions that are both proinflammatory and ant-iinflammatory. In renal ischemic injury, a protective role for OPN is supported by the finding that increased OPN expression occurs in the distal tubules, which contain cells that are resistant to ischemic injury (Padanilam et al, 1996; Persy et al, 1999). In contrast, proximal tubules, which are less tolerant to ischemic injury, show very little OPN expression after ischemia. Furthermore, a renoprotective effect of OPN is supported by the finding that OPN knockout mice display greater renal damage and dysfunction after acute renal ischemia (Persy et al, 1999).
The response to ischemic brain injury is also associated with increased OPN expression (Lee et al, 1999; Wang et al, 1998), which is observed primarily in microglia and macrophages found in the infarct and periinfarct regions. Osteopontin expression parallels the time course of macrophage infiltration into the infarct and expression of CD44 (an OPN receptor) (Wang et al, 1998). In addition, astrocyte expression patterns of another receptor for OPN, integrin
Osteopontin and integrin receptors have both also been ascribed functions in cell survival. Such functions might be attributed to effects that oppose apoptotic pathways (Liaw et al, 1995; Ophascharoensuk et al, 1999, 1998; Persy et al, 2003; Scatena et al, 1998) or enhance cell growth. Osteopontin acts through multiple intracellular pathways and downstream effectors, leading to diverse outcomes that could confer protection against ischemic injury. Examples of such pathways are: the reduction of nitric oxide synthase expression, activation of the phosphatidylinositol 3-kinase (PI3K) and NF-
We postulated that OPN may promote neuronal survival in the setting of injury because OPN is increased in the brain after ischemia and can act through
Materials and methods
Neuronal Cell Culture
Cortical neuronal cultures were prepared from 1- to 3-day-old Sprague–Dawley rat pups using the method of Goslin (1998), as described previously (Stenzel-Poore et al, 2003). Briefly, cortices were dissected from 10 to 12 rat pups, dissociated with papain (Worthington Biochemicals, Lakewood, NJ, USA) and grown in Neurobasal-A/B27 media (Invitrogen, Carlsbad, CA, USA) for 7 to 10 days, when cultures consist of 80% to 90% neurons as assessed by NeuN versus GFAP staining (not shown). Cells were plated out at a density of 400,000 cells per coverslip for cell viability assays, or 5,000,000 cells per 10-cm culture dish (Primara; Becton Dickinson, San Jose, CA, USA) for Western blotting.
Oxygen–Glucose Deprivation
Oxygen–glucose deprivation (OGD) was performed by washing the cells with phosphate-buffered saline (PBS) (0.5 mmol/L CaCl2, 1.0 mmol/L MgCl2; pH 7.4) and placing culture dishes in an anaerobic chamber for 120 minutes (Forma Scientific, Marjetta, OH, USA) (85% N2, 5% H2, 10% CO2; 35°C). Anaerobic conditions in the chamber were monitored using Gaspack anaerobic indicator strips (Becton Dickinson, San Jose, CA, USA). Oxygen–glucose deprivation was terminated by removing cells from the chamber, replenishing with Neurobasal A media without B27 and placing them back in the normoxic incubator. Mouse recombinant OPN (Calbiochem, San Diego, CA, USA) (0.01 to 1.0
Cell Viability Assay
For cell viability assays, coverslips containing cortical cells were incubated with propidium iodide (1.5
Western Blotting
Western blotting was performed as described previously (Meller et al, 2002). Tissue samples were lysed in a nondenaturing buffer containing the protease inhibitors phenylmethylsulfonylfluride (100
Mice
C57Bl/6 mice (male, 8 to 10 weeks) were obtained from the National Cancer Institute for experiments with OPN administration. Osteopontin-deficient mice (OPN−/−) were obtained from Dr Lucy Liaw, Maine Medical Center Research Institute, Scarborough, Maine (Liaw et al, 1998). OPN−/− mice were backcrossed five generations onto C57Bl/6. Littermates were used as controls. All procedures met NIH guidelines with the approval of the Oregon Health & Science University Institutional Animal Care and Use Committee.
Surgery
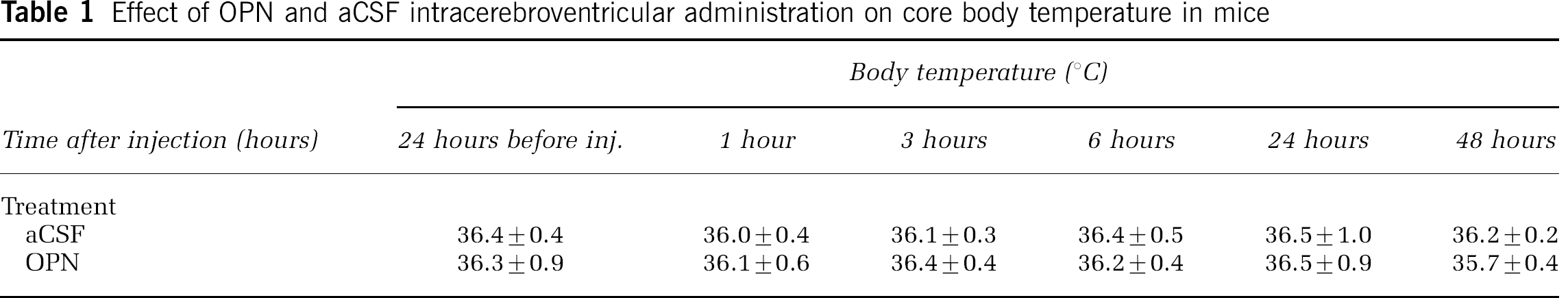
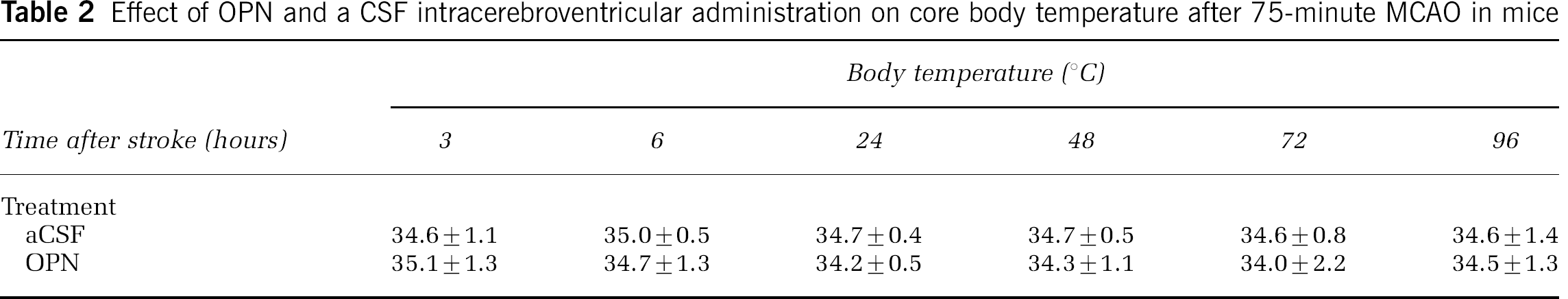
Cerebral focal ischemia was induced by middle cerebral artery occlusion (MCAO) as published previously (Clark et al, 1997). Briefly, mice were anesthetized by halothane inhalation (4%/litre O2) and maintained with 1.5%/litre O2. The middle cerebral artery (MCA) was blocked by threading silicone-coated 7-0 monofilament nylon surgical suture through the external carotid to the internal carotid, and finally blocking its bifurcation into the MCA and anterior cerebral artery. The filament was maintained intraluminally for either 60 minutes (OPN-deficient mice) or 75 minutes, while the mice were maintained under anesthesia. The filament was then removed, thereby restoring blood flow. Cerebral blood flow (CBF) was monitored throughout the surgery by laser Doppler flowmetry (Periflow 5000; Perimed, Sweden). Band body temperature was monitored throughout surgery using a rectal thermometer (Hi-Lo Temp 8200 Patient Temperature Monitor, Sensortek Inc, Lake forest, CA, USA). After surgery, mice were kept on a thermal barrier pad with free access to soft food and were killed after either 24 or 96 hours. Body temperature of mice was measured at 3, 6, 24, 48, 72, and 96 hours after MCAO surgery. In a second group of animals, body temperature was measured at 1, 3, 6, 24, and 48 hours after administration of either artificial cerebrospinal fluid (aCSF) or OPN, but not subjected to MCAO. Blood glucose was measured (One Touch SureStep, Lifescan, Milpitas, CA, USA) 1 hour and 24 hours after i.c.v. administration of OPN or aCSF.
Intracerebral Ventricular Injections
Mouse recombinant OPN (Calbiochem, San Diego, CA, USA), human albumin (ZLB Bioplasma AG, Berne, Switzerland), or vehicle (aCSF) was injected into the left lateral ventricle (coordinates: 0 mm bregma, 1 mm lateral, and 2.5 mm ventral) using a 27-gauge needle. Injections (0.5
Infarct Measurement
Mice were deeply anesthetized with isoflurane, then perfused via the ascending aorta with saline containing 2 U/mL heparin at a flow rate of 9 mL/minute. Brains were rapidly removed, placed on a tissue slicer (Stoelting, Wood Dale, IL, USA) and covered with soft agarose. The olfactory bulb and cerebellum were removed and discarded. The remaining brain was sectioned into 1-mm slices beginning from the rostral end. To visualize the region of infarction, sections were placed in 1.5% 2,3,5-triphenyltetrazolium chloride (TTC) in 0.9% phosphate-buffered saline and stained for 15 minutes at 37°C (Bederson et al, 1986). After staining, the sections were transferred to 10% paraformaldehyde. Images of the sections were scanned, and the areas of the infarct and the ipsilateral hemisphere were measured using NIH image 1.62. The measurements were multiplied by the section thickness (1 mm) and then summed over the entire brain to yield volume measurements. The percent infarct was calculated as: (Infarct Volume)/(Ipsilateral Hemisphere Volume) × 100.
Statistical Analysis
Data are shown as means±s.d. of
Results
Osteopontin is Protective in an In Vitro Model of Ischemia
We investigated whether OPN protects neurons from injury in an
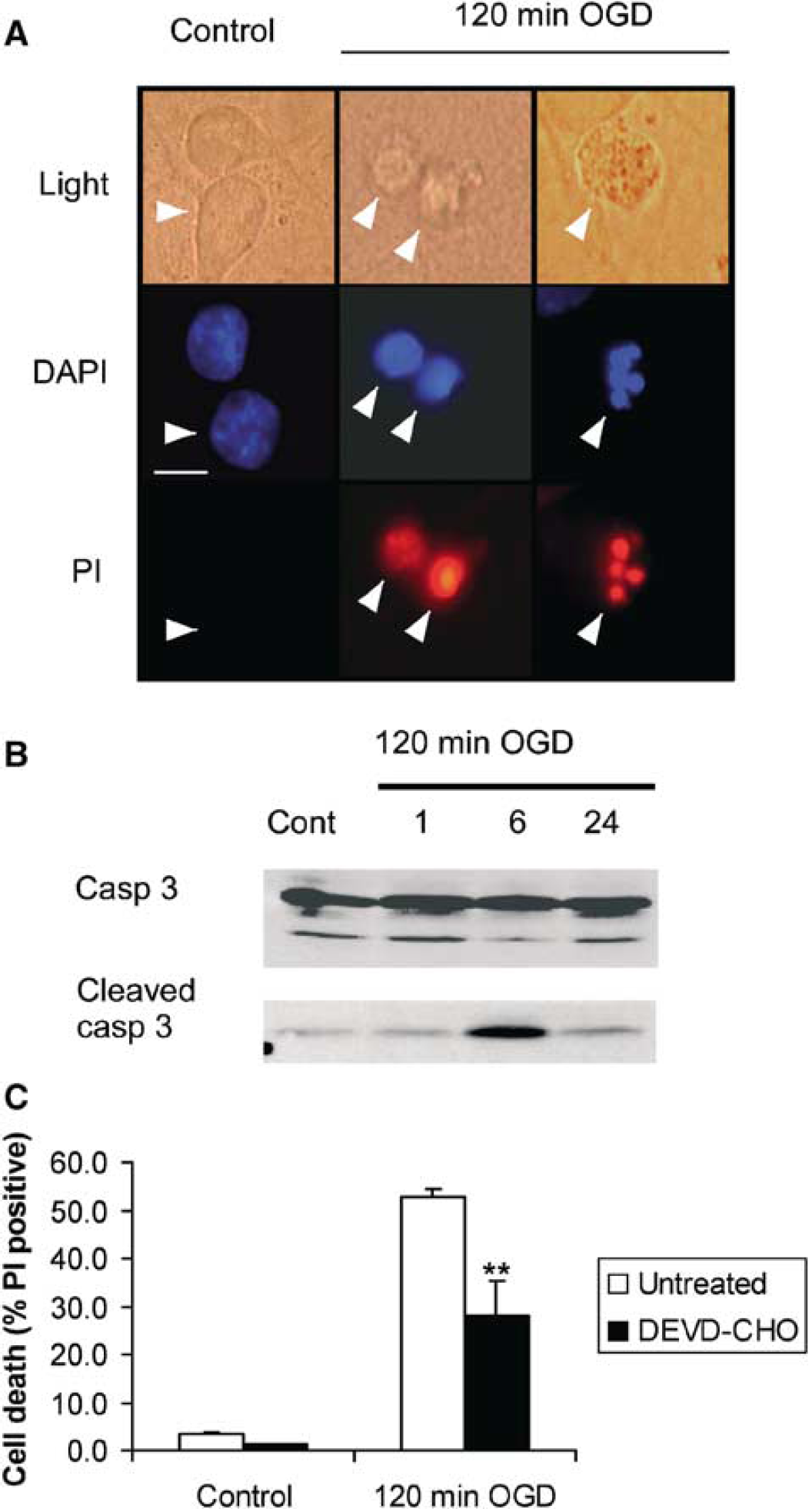
Effect of 120-minute OGD on neuronal cultures. (
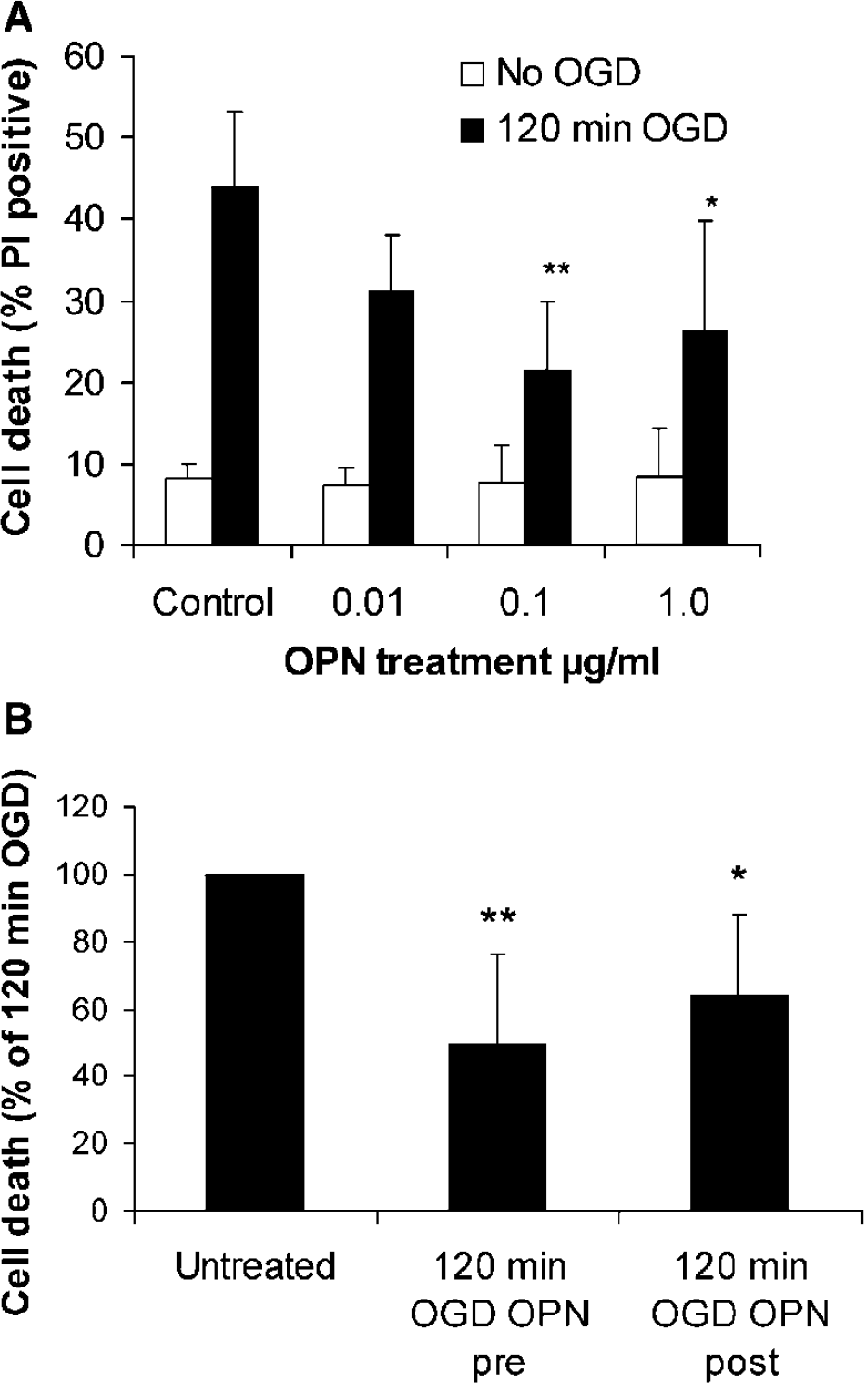
Pretreatment of cortical cultures with OPN (0.01 to 1.0
(
Osteopontin is Neuroprotective Administered After Ischemic Challenge In Vitro
Our data suggest that
The Neuroprotective Effect of Osteopontin is Blocked by an RGD-containing Peptide
To determine whether the neuroprotective effect of OPN
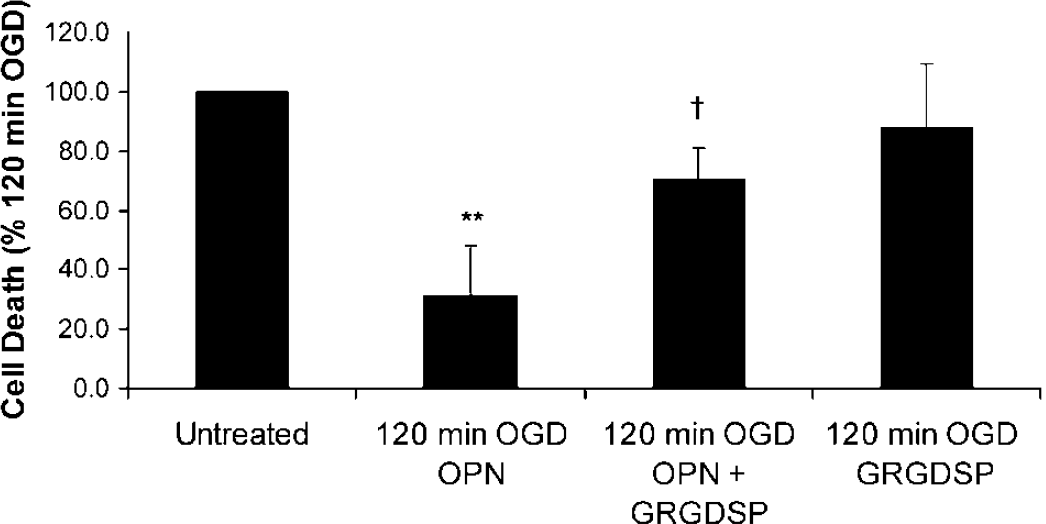
The neuroprotective effect of OPN is blocked by the RGD containing hexapeptide GRGDSP. Cells were treated with GRGDSP (10 nmol/L) for 10 minutes before addition of OPN (0.1
Activation of Akt and p42/p44 MAPK in Osteopontin-induced Neuroprotection
We tested whether the neuroprotective actions of OPN occur through PI3K, a known downstream mediator of OPN's effects in certain cell types, which, in turn, activates Akt (protein kinase B) (Das et al, 2003; Lin and Yang-Yen 2001; Zheng et al, 2000). Osteopontin treatment of cortical cells failed to protect against OGD-induced cell death in the presence of LY294002 (10
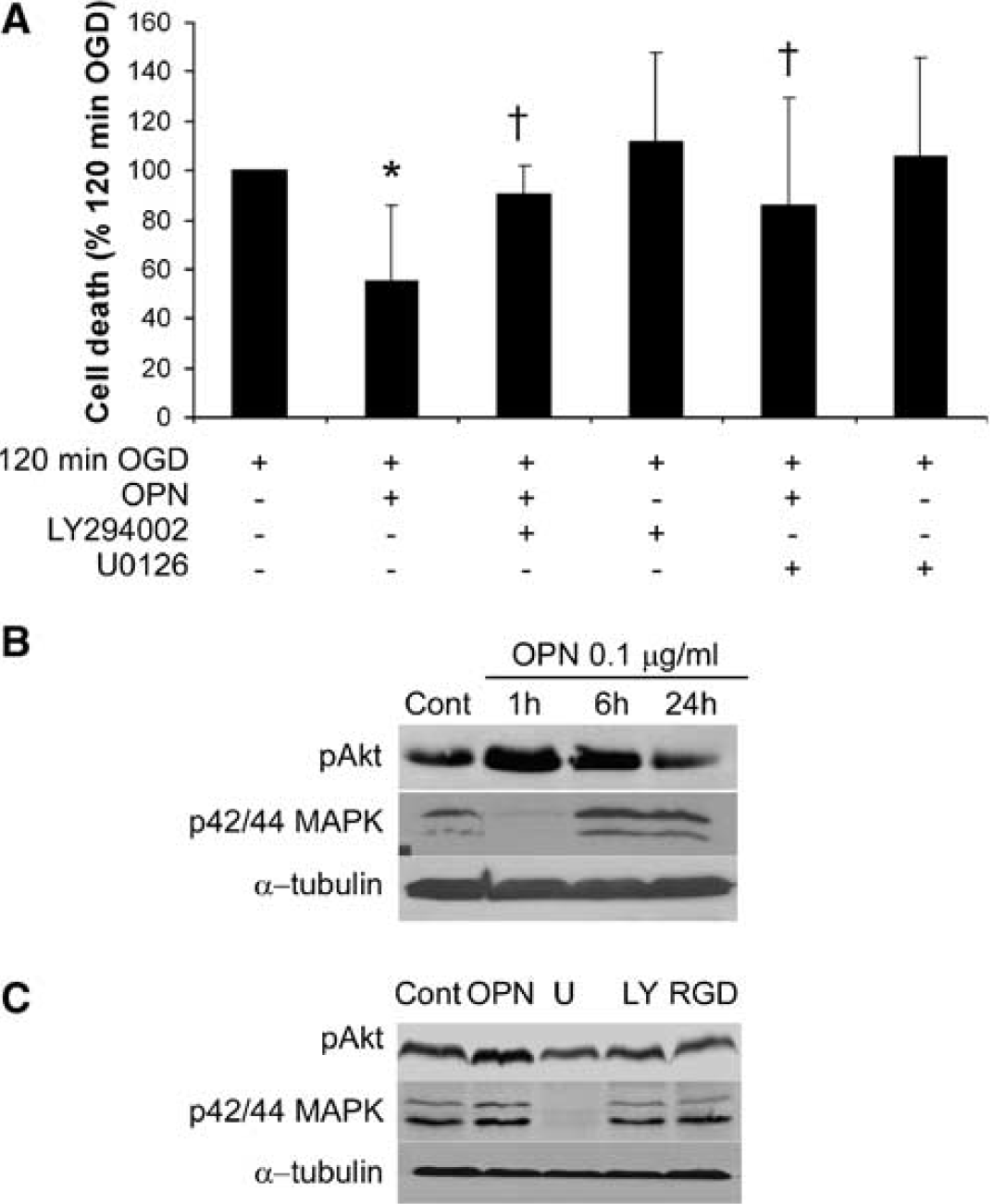
The neuroprotective effect of OPN is blocked by the PI3K inhibitor LY294002 and the Mek inhibitor U0126. (
To further test whether treatment of cells with OPN leads to activation of Akt or p42/p44 MAPK, we probed Western blots with antibodies specific for phosphorylated Akt (Ser 473) and phosphorylated p42/p44 MAPK. Cortical cultures treated with OPN display increased Akt phosphorylation (Figure 4B). Phosphorylation of p42/p44 MAPK by OPN was biphasic, with an initial reduction, followed by an increased phosphorylation (Figure 4B). Further, the effect of OPN on p42/44 MAPK phosphorylation was reduced by LY294002 and GRGDSP peptide and abolished by U0126, whereas the effect of OPN on Akt phosphorylation was reduced by LY294002 and GRGDSP peptide (Figure 4).
Finally, the neuroprotective effect of OPN against OGD-induced cell death was blocked when cortical cultures were treated with OPN in the presence of the protein synthesis inhibitor cycloheximide (data not shown). These data suggest that the neuroprotective effects of OPN require the activation of the Akt and p42/p44 MAPK cascades, as well as new protein synthesis.
Exogenously Administered Osteopontin Protects Against Focal Ischemic Injury
Given the neuroprotective effects of OPN that we observed
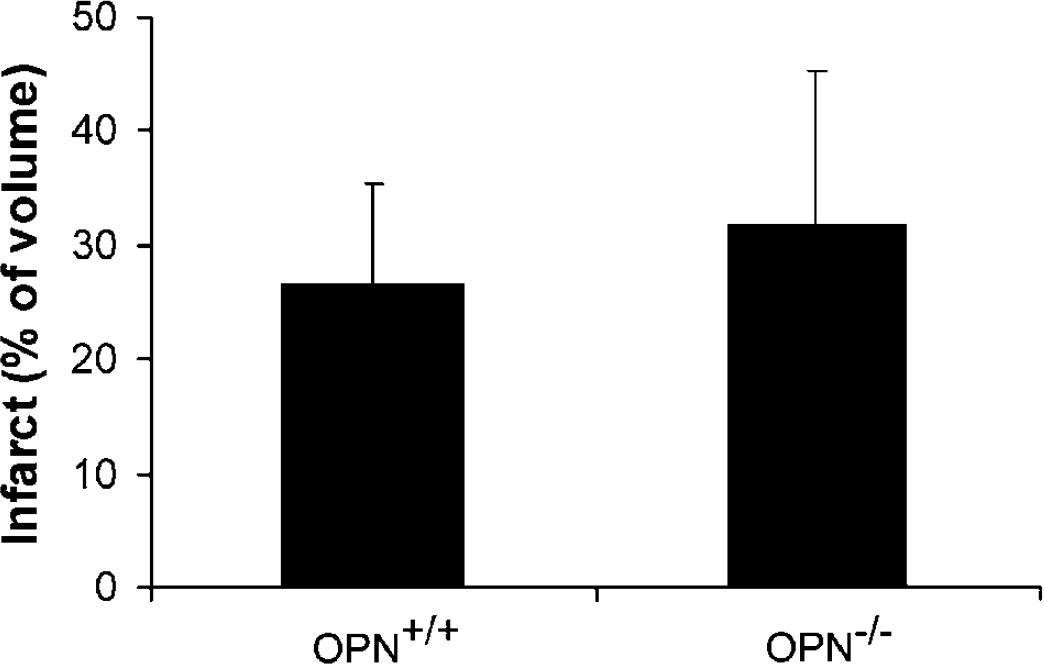
Osteopontin-deficient mice show similar infarct size to wild-type littermate controls. OPN−/− (
To determine if administration of exogenous OPN conferred protection against ischemic injury, we injected OPN (2 × 50 ng) or aCSF into the lateral ventricle just before and immediately after MCAO (75 minutes). Mice treated with OPN had significantly smaller infarcts (∼2-fold) compared with mice treated with aCSF at 24 and 96 hours after MCAO (Figure 6). As a control, we injected albumin, a protein with an equivalent weight to OPN, (2 × 50 ng) into the lateral ventricle before and after MCAO (75 minutes). Infarct size in mice injected with albumin did not differ from aCSF-treated animals (data not shown).
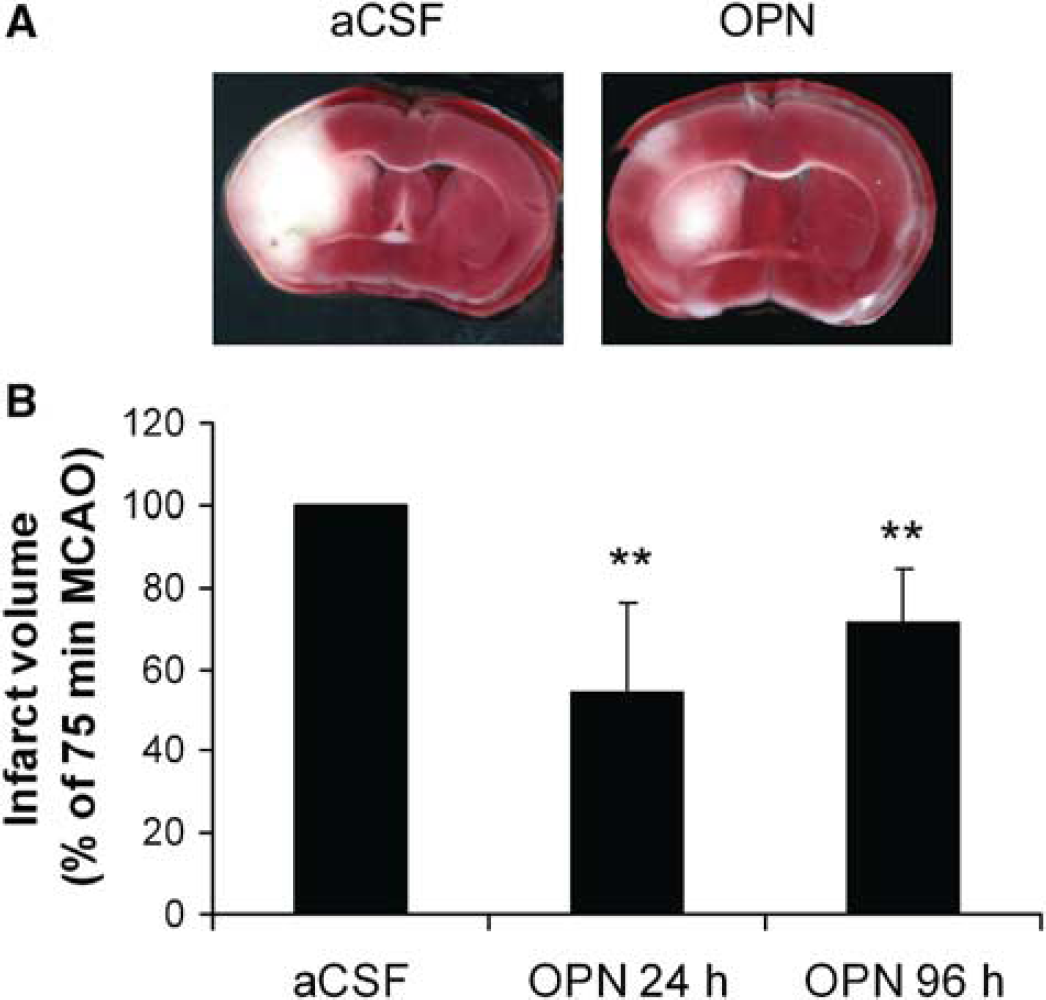
Osteopontin reduces infarct volume after stroke. Mice were administered OPN (2 × 50 ng) or CSF (
The effect of OPN and aCSF on body temperature was measured in the presence or absence of focal ischemia and found not to differ (Tables 1 and 2). Furthermore, no differences in blood glucose levels were observed after OPN administration (data not shown); hence, the protective effect of OPN is unlikely to be due to changes in body temperature and blood glucose availability. Thus, OPN administered at the time of stroke acts as a potent neuroprotectant in focal ischemia.
Effect of OPN and aCSF intracerebroventricular administration on core body temperature in mice
Effect of OPN and a CSF intracerebroventricular administration on core body temperature after 75-minute MCAO in mice
Discussion
There is a dearth of effective neuroprotective therapies for the treatment of stroke. Here, we have investigated the potential of OPN as a neuroprotective therapy for acute brain ischemia. Osteopontin reduced ischemic cell death significantly in
We investigated the neuroprotective potential of OPN on the basis of its increased expression in the brain after ischemia (Stenzel-Poore et al, 2003; Wang et al, 1998). We hypothesized that increased expression of OPN after ischemia may affect neuronal survival in brain ischemia. Given that OPN contains an integrin receptor-binding motif (Denhardt et al, 2001; Hwang et al, 1994; Malyankar et al, 2000), it was conceivable that OPN acts on neurons via interactions with integrin receptors. It has been shown previously that activation of integrin receptors on neurons renders them tolerant to glutamate toxicity (Gary and Mattson, 2001). Thus, OPN may induce a neuroprotective effect via integrin receptor activation. In our studies, OPN was neuroprotective after injurious 120-minute OGD in cultured rat cortical cells. Osteopontin also reduced infarct volume after MCAO-induced ischemia in mice. Taken together, these data suggest OPN has therapeutic potential as a stroke therapy. A protective role of OPN in ischemia has been suggested in the kidney (Kleinman et al, 1995; Persy et al, 1999) because of its increased presence after ischemia and preferential localization with cells destined to survive (Padanilam et al, 1996). Further, the absence of endogenous OPN appears to increase sensitivity to ischemia, as OPN-knockout animals have greater kidney damage after renal ischemia (Noiri et al, 1999). Interestingly, in our studies reported here, the absence of OPN does not appear to predispose mice to greater injury in the CNS, as we found similar infarcts in wild-type and OPN-knockout mice after MCAO (Figure 5).
Previous reports suggest that OPN may activate multiple prosurvival mechanisms, including decreased induction of nitric oxide synthase (Denhardt et al, 1995), increased NF-
Exogenous administration of OPN directly into the brain has a marked neuroprotective effect in ischemia. These results offer promise in the identification and development of neuroprotectants in stroke. Therapeutic use can be limited for proteins that must be delivered directly into the brain. Efficient delivery to the brain in animal models and humans has been achieved with a number of proteins using intranasal administration (Liu et al, 2001). This approach has already been used successfully with a limited number of proteins and peptides in the treatment of CNS diseases or injury, including stroke (Liu et al, 2001), and may allow the rapid administration of OPN into the brain.
Further drug development is needed in the case of OPN. Pursuit of an OPN-mimetic with the similar biological properties of OPN, but improved potency and pharmacokinetics, would be a viable therapeutic agent for further development. Interestingly, the biological effects of OPN have been mimicked by a 20-amino-acid peptide containing the RGD integrin receptor-interacting motif of OPN (Hwang et al, 1994). Hence, OPN or short OPN analogues may lead the way to the design of a drug as an effective therapy for use in stroke. As such, further investigation of the therapeutic potential of OPN or an OPN mimetic is warranted.
Footnotes
Acknowledgements
This work was supported by the Medical Research Foundation of Oregon (RM) and by NIH grants NS24728 (RPS), NS35965 (RPS, MS-P) and NS046827 (MS-P). The work reported in this manuscript was also supported in part by Virogenomics, Inc., a company that may have a commercial interest in the results of this research. OHSU and investigators, MS-P and RPS, have a financial interest in Virogenomics. This potential conflict of interest has been reviewed and managed by the OHSU Conflict of Interest in Research Committee.
