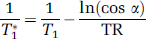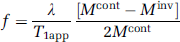Abstract
Suppression of platelet activation improves the efficacy of thrombolytic therapy for stroke. Thus, combination treatment with recombinant tissue plasminogen activator (r-tPA) and 7E3 F(ab′)2, a GPIIb/IIIa inhibitor that binds the platelet to fibrin, may improve the efficacy of thrombolytic therapy in embolic stroke. Magnetic resonance imaging (MRI) was used to monitor treatment response in rats subjected to embolic middle cerebral artery (MCA) occlusion (MCAo). Animals were randomized into treated (n = 12) and control (n = 10) groups and received intravenous combination therapy or saline, respectively, 4 hours after MCAo. Magnetic resonance imaging (MRI) measurements performed 1 hour after MCAo showed no difference between groups. However, an increased incidence (50%) of MCA recanalization was found in the treated group at 24 hours compared with 20% in the control group. The area of low cerebral blood flow at 24 and 48 hours was significantly smaller in the combination treatment group, and the lesion size, as indicated from the T2 and T1 maps, differed significantly between groups. Fluorescence microscopy measurements of cerebral microvessels perfused with fluorescein isothiocyanate-dextran and measurements of infarct volume revealed that the combination treatment significantly increased microvascular patency and reduced infarct volume, respectively, compared with the control rats. The efficacy of combination treatment 4 hours after ischemia is reflected by MRI indices of tissue perfusion, MCA recanalization, and reduction of lesion volume. The treatment also reduced secondary microvascular perfusion deficits.
Keywords
Introduction
Thrombolytic therapy using recombinant tissue plasminogen activator (r-tPA) is efficacious when administered within 3 hours of stroke onset (NINDS, 1995). Experiments in rats subjected to embolic cerebral middle artery (MCA) occlusion (MCAo), however, have demonstrated that treatment at 4 hours after stroke onset fails to reduce the volume of cerebral infarction or produce therapeutic benefit (Jiang et al, 2000; Zhang L et al, 2001). Furthermore, r-tPA treatment may increase hemorrhagic transformation (HT), the most common adverse effect of thrombolytic therapy (NINDS, 1997; Jiang et al, 2002). Therefore, it would be desirable to improve the efficacy and safety, as well as to increase the therapeutic window for thrombolytic therapy of stroke.
Studies on experimental and human stroke suggest that platelets play a role in the development and progression of cell damage after focal cerebral ischemia (del Zoppo, 1998). Platelets aggregate in regions of low blood flow during the postischemic period in animal models of focal cerebral ischemia (Dietrich et al, 1993; Jafar et al, 1989; Kochanek et al, 1988; Obrenovitch and Hallenbeck, 1985), and acute stroke patients show evidence of platelet activation (Dougherty et al, 1977; Fisher et al, 1982). Platelets aggregate dynamically at the site of an embolus occluding the MCA, and may contribute to resistance to thrombolysis with duration of ischemia, since platelets retract clots and release plasminogen activator inhibitor 1 (PAI-1), α2-antiplasmin, and factor XIII, all of which reduce thrombolysis (Zhang ZG et al, 2001). In addition, the platelet and fibrin thrombosis in cerebral microvessels downstream of the occluded MCA increase time-dependently after ischemia and promote progressive impairment cerebral microcirculation (Zhang ZG et al, 2001). Thus, suppression of platelet deposition and activation at the initial site of obstruction and in the downstream microvessels may improve the efficacy of thrombolytic therapy (Coller, 1995; AISI, 2000; Choudhri et al, 1998; Zhang L et al, 2003).
Hemorrhagic cerebrovascular sequelae of thrombolytic therapy is a major obstacle for extending the time from the onset of symptoms to the initiation of therapy. Upregulation of PAI-1 acutely after onset of focal embolic ischemia contributes to intravascular fibrin deposition, which directly obstructs cerebral microcirculation (Zhang ZG et al, 1999). Activated platelets not only secrete PAI-1 but also release matrix metalloproteinase 2 (MMP2) and can induce upregulation of MMP9 (Sawicki et al, 1997). Increases in MMP2 and MMP9 activity after focal cerebral ischemia contribute to disruption of the blood-brain barrier (BBB) (Lampugnani et al, 1991). Therefore, in addition to its role of thrombosis, platelet aggregation in the ischemic vessels promotes disruption of the BBB.
The platelet glycoprotein (GP) IIb/IIIa receptor, which is the most abundant platelet receptor, acts as the final common pathway for platelet aggregation (Coller, 1995). Treatment of embolic stroke in the rat with a GPIIb/IIIa inhibitor or r-tPA alone fails to provide any therapeutic benefit when the treatment is initiated at 4 hours after stroke onset, that is, beyond the therapeutic window for thrombolysis with r-tPA. Combination therapy with r-tPA and a GPIIb/IIIa inhibitor extends the tPA therapeutic window in experimental stroke (Zhang L et al, 2003). These data indicate that therapeutic benefit of the combination treatment derives from a super-additive interaction between the r-tPA and the GPIIb/IIIa antagonist. What, however, has not been investigated is the relationship between MCA patency after embolic stroke and microvascular occlusion. We propose that the downstream microvasculature is hypoperfused even when the MCA is patent after embolic stroke, and that a contributing factor to this hypoperfusion is the platelet GPIIb/IIIa receptor binding of fibrin. Thus, we hypothesize that a combination of r-tPA and anti-GPIIb/IIIa yields increased cerebral tissue perfusion, even with nontreated embolic stroke.
We have previously demonstrated using MRI and non-MRI approaches that treatment of embolic stroke with r-tPA alone at 4 hours after stroke onset fails to improve CBF and reduce infarct volume compared with saline-treated animals (Jiang et al, 2000, 2002; Ding et al, 2004; Zhang ZG et al, 2002; Zhang L et al, 2003). In the present study, using magnetic resonance imaging (MRI), we investigated the effects of combination treatment of 7E3 F(ab′)2 and r-tPA by means of MRI including the measurements of the apparent diffusion coefficient of water (ADCw) by diffusion-weighted imaging (DWI), cerebral blood flow (CBF) by perfusion weighted imaging (PWI), magnetic resonance angiography (MRA), relaxation time constants of T1 by T1-weighted imaging (T1WI) and T2 by T2-weighted imaging (T2WI), as well as fluorescence microscopy and histology.
MATERIALS AND METHODS
All studies were performed in accordance with institutional guidelines for animal research under a protocol approved by the Institutional Animal Care and Use Committee (IACUC) of Henry Ford Hospital.
Animal Model and Experimental Groups
Male Wistar rats (N = 22, 300 to 350g) were subjected to embolic stroke and investigated using a 7-T MRI system. Rats were randomized into one of two groups: group 1 (n = 10) was comprised of animals that received embolization but used saline instead of r-tPA treatment; group 2 (n = 12) was comprised of animals that received embolization, followed by r-tPA and GPIIb/IIIa inhibitor treatment. The F(ab′)2 fragment of the murine monoclonal antibody 7E3, which reacts with GPIIb/IIIa, was administered intravenously at a bolus dose of 6.0 mg/kg 4 hours after embolic MCAo and was followed by a second intraperitoneal dose 6.0 mg/kg at 12 hours after the first dose. Recombinant human tPA (tPA, Genentech, San Francisco, CA, USA) was infused intravenously at a total dose of 10 mg/kg (10% bolus 4 hours after ischemia, and the remainder at a continuous infusion over a 30-min interval using a syringe infusion pump; Harvard Apparatus, South Natick, MA, USA). All animals were killed at 48 hours after MCAo. The dosing protocol for the 7E3 F(ab′)2 used in this study was selected based on the literature (Yang et al, 2001) and our previous protocol (Zhang L et al, 2003). We have previously demonstrated that this dosing protocol (Zhang L et al, 2003) is effective in extending the therapeutic window for r-tPA and enhances microvascular integrity and cerebral perfusion.
The method of embolic stroke induction has been previously described (Jiang et al, 1998). Briefly, an aged white clot, which was prepared 24 hours before ischemia, was slowly injected into the internal carotid artery to block the MCA. This model of embolic stroke provides a relatively reproducible infarct volume localized to the territory supplied by the MCA (Zhang RL et al, 1997).
Magnetic Resonance Imaging on a 7T system
Magnetic resonance imaging measurements were performed using a 7-T, 20-cm bore superconducting magnet (Magnex Scientific, Abingdon, UK) interfaced to a Bruker console (Bruker Company, Boston, USA), with a 12-cm bore actively shielded gradient coil set capable of producing magnetic field gradient up to 200 mT/m. A birdcage radiofrequency (RF) coil was used as the transmitter and a surface coil as the receiver. Stereotaxic ear bars were used to minimize movement during the imaging procedure. During MRI measurements, the anesthesia was maintained using a gas mixture of nitrous oxide (69%), oxygen (30%), and halothane (0.75% to 1.00%). Rectal temperature was kept at 37°C ± 1.0°C using a feedback controlled water bath. The right femoral artery and vein were cannulated with a PE-50 catheter for monitoring of blood pressure and gases (pH, partial pressure of oxygen (PO2), carbon dioxide (PCO2)), and drug administration, respectively.
A tri-pilot scan of imaging sequence was used for reproducible positioning of the animal in the magnet at each MRI session. A complete set of MR images were performed before ischemia, repeatedly for 1 hour after the onset of embolization, and at 24 hours, as well as 48 hours after embolization for all animals. A complete set of all sequences, including DWI, T1WI, Look-Locker (L-L) T1 measurements, multiecho T2 measurement, PWI, MRA, and magnetization transfer (MT) MRI, required approximately 2 hours of scan time. Contrast-enhanced MRI was performed acutely, and 24 and 48 hours after ischemia, respectively.
Magnetic Resonance Imaging Techniques
Measurement of MRA employed a three-dimensional (3D) gradient echo imaging sequence with calculated gradient trims. This sequence comprises the first-order flow compensation achieved via gradient-moment nulling in the frequency-encoding, phase-encoding and slice directions (Haase et al, 1986). Thus, the effect of the flow compensation is maximized whenever imaging materials possess linear flow characteristics. The acquisition matrix was set as 256 × 192 × 64 for fitting the field of view (FOV) 32 × 32 × 16 mm3 in coronal-head-foot orientation. Repetition time (TR) and echo time (TE) were 50 and 4.5 ms, respectively. The 500 ms Gaussian RF pulse generated a flip angle of approximately 40°.
The arterial spin tagging (AST) technique (Williams et al, 1992) was used for quantifying blood flow in cerebral tissue. The adiabatic inversion of arterial water protons (Dixon et al, 1986) was accomplished via an axial gradient of ±0.3 kHz/mm, and a continuous wave (CW) RF power of approximately 0.3 kHz at a frequency offset of ±6 kHz. A hyperbolic secant (sech) shaped pulse of 1-second duration was employed as an adiabatic fast passage RF pulse, followed by a spin echo imaging sequence with TR/TE = 1000/20 ms. The labeled slice was 2 cm distal from the imaging slice. To minimize the effects of MT, the CBF of the central slice (0 offset, 1 mm slice thickness) was measured. To remove the asymmetry of gradient in the axial direction, an image average was applied by switching around the gradient polarities. The FOV is 32 mm × 32 mm, matrix 64 × 64.
Quantitative estimates of tissue T1 values were generated using an imaging variant of the Look-Locker technique (Look and Locker, 1970). This method provides an efficient and unbiased pixel-by-pixel estimate of T1 (Crawley and Henkelman, 1988). The sequence begins with a RF inversion pulse followed by a series of low-angle excitation RF pulses. With this sequence, a set of five slices of T1 maps (32-mm FOV, 128 × 64 matrix, 2-mm slice thickness) was obtained in 11 mins. Interleaved slices were acquired. At least five dummy cycles were applied before the start of data acquisition. Inversion of the longitudinal magnetization was accomplished using a nonselective hyperbolic secant adiabatic pulse of 8 ms duration. One phase-encode line of 32 small-tip-angle gradient-echo images (TE of 2.2 ms) was acquired at 80-ms intervals after each such adiabatic inversion. The total recovery time is 11 secs.
The trace ADCw was measured using the method described by LeBihan et al (1986) with diffusion gradient in each x, y, z direction. A two-dimensional Fourier transform (2DFT) multislice spin-echo sequence (13 slices, 32-mm FOV, 128 × 64 matrix, 1.5 secs repetition time, 40 ms echo time) was modified to include two 10-ms diffusion-weighted gradient pulses, one on either side of the refocusing 180° RF pulse. The diffusion-weighted gradients were increased in a nonlinear manner from 0 to approximately 100mT/m to obtain a series of three images with gradient b-values of 0, 600, and 1,200 s/mm2. Each image required a scan time of 5 mins for completion, and the total time needed for the entire three directions trace map sequence was approximately 15 mins.
T2 was measured using a standard 2DFT multislice (13 slices) multiecho (6 echoes) MRI. The echo times were 20, 40, 60, 80, 100, and 120 ms and the repetition time was 0.7 secs. Images were produced using a 32-mm FOV, 1-mm slice thickness, and a 128 × 64 image matrix. The total time for the entire sequence was approximately 9 mins.
Measurement of Microvascular Patency
To examine patency of cerebral microvessels, fluorescein isothiocyanate (FITC)-dextran (2 × 106 molecular weight, Sigma, St Louis, MO, USA; 1 mL of 50 mg/mL) was administered intravenously to the rats 5 min before being killed. Three coronal sections (100 μm) at the bregma –0.2, –0.8, and –2.8 mm from each rat that encompass main territory supplied by MCA were analyzed with a MicroComputer Imaging Device (MCID) system (Imaging Research Inc., St Catherines, ON, Canada). Briefly, fluorescence was digitized using a × 5 objective (Zeiss Plan Neoflura) attached to a fluorescent microscope (Zeiss, Axioplan 2) via a digital CCD camera (Hammatsu, Sunayama, Japan) connected to the MCID system. Ten fields of view (4.4 mm2) from each coronal section were acquired in the territory supplied by the right MCA. A threshold was applied to each digitized image to ensure that the numbers of FITC pixels reflected the original FITC-dextran perfused patterns. Data are presented as the numbers of FITC pixels divided by the total numbers of pixels within the FOV, expressed as a percentage. Two control rats (one patent MCA at 24 hours, one not) and four rats from the combination treatment group (one patent MCA with reduced CBF at 24 hours, one occluded MCA, and two patent MCA and recovered CBF at 24 hours) were chosen for measurement of microvascular patency.
Histological Measurement of Lesion Size
After the final MRI measurement, animals, except those used for the tissue perfusion study of microvascular patency, were anesthetized with ketamine (44mg/kg intraperitoneal) and xylazine (13mg/kg intraperitoneal), and were transcardially perfused with heparinized saline followed by 10% neutral buffered formalin. The head was immersed in formalin solution for 1 hour, after which a total of seven 2-mm-thick blocks of brain tissue were cut, processed, and embedded in paraffin. Coronal sections (6 mm thick) were cut from each block and stained with hematoxylin and eosin (H&E) for the evaluation of ischemic cell damage and hemorrhage. The lesion volumes were measured using MCID by a person who was masked on the study groups. Each H&E-stained section was evaluated at × 2.5 magnification. The area of infarction and the area of the ipsilateral hemisphere (mm2) were measured by tracing the areas for each section on the computer monitor screen (Zhang RL et al, 1994).
Data Analysis
All MR images were reconstructed using a 128 × 128 matrix. The ADCw and T2 maps were produced on a pixel-by-pixel basis using a linear least-squares fit to the plot of the natural logarithm of normalized image intensity versus gradient b-value (ADCw) or TE (T2) values, respectively. Look-Locker procedures result in a set of images that recover with a time constant T*1 toward a steady-state magnetization. T*1 and the steady-state magnetization both have a dependency on the tip angle of the readout as well as on T1 and the equilibrium magnetization. T*1 is related to T1 as follows:
where α is the tip angle and TR is repetition time. A nonlinear optimization procedure was used to estimate the parameters of interest for each slice and a ‘nuisance’ variable, the tip angle. For all such procedures, home-written software employing a Simplex curve-fitting algorithm was used, as explained in a previous publication (Gelman et al, 2000), but with a nonnegative constraint added.
The CBF map (ƒ-value) was calculated according to
where δ is the blood/brain partition coefficient for water and T1app is the apparent T1 relaxation time of brain water in the absence of flow or exchange between blood and brain. Mcont and Minv are the control and inversion image intensities, respectively. Values of 0.9 g/mL for δ and 1.7 secs for T1app were used for CBF calculation (Williams et al, 1992).
The sizes of abnormal parenchyma on MRI parametric ADCw, T1, and T2 maps were calculated based on the mean value and standard deviation (s.d.) of the contralateral parenchyma. Normal pixels were defined within the mean value plus 2.0 s.d. of the contralateral parenchyma for T1, T2 maps and mean value minus 2.0 s.d. for ADCw maps. For CBF, abnormal parenchyma was identified if its CBF value is lower than 50 mL/100 g min. The threshold of 50 mL/100 gmin is chosen based on the threshold of CBF, which induces an abnormal physiological state. Although the threshold of CBF to induce ischemic damage is below 20 mL/100g min (Branston et al, 1974; Sundt et al, 1974), CBF below 50mL/100gmin will cause ‘misery perfusion syndrome', a syndrome of impending or manifest failure of adequate oxygen supply (Baron et al, 1983). Therefore, we choose a CBF threshold of 50 mL/100 gmin.
The MRA image is presented by means of maximum intensity projection (MIP) reconstruction of the 3D imaging data. The situation was also confirmed by viewing the 3D images slice by slice.
Statistical Analysis
Observations are summarized as mean ± s.d. The statistical comparisons were performed using the Student's t-test for the effects of combined treatment with r-tPA and 7E3 F(ab′)2 versus treatment with saline. The significance level was set at 0.05.
RESULTS
Our MRI experiments showed that r-tPA and 7E3 F(ab′)2 work together to increase the efficiency of dissolving the embolus, as well as to increase CBF. Among the 12 rats from the combination treatment treated group, the MCA of 6 rats (50% of the group) recanalized by 24 hours after the onset of ischemia, as indicated by the MRA images. For the control group, only 2 of 10 rats (20% of the group) showed MCA recanalization. Figures 1D-1F and 1J-1L present typical MRA images obtained at 1, 24 and 48 hours after embolization. The MRA images acquired at 1 hour after embolic stroke onset showed that the right branches of the MCA (straight arrow in the figures) for both treated rat (Figure 1D) and control rat (Figure 1J) were occluded. However, by 24 hours after embolization, half the animals (50%) from the treated group and two animals (20%) from the control group had patent right branches of the MCA (Figure 1E). The remaining animals (50% of treated and 80% of control group) had occluded right MCA branches (Figure 1K). No significant change was found in the MRA images from 24 to 48 hours for each group (e.g., Figure 1F for patent right MCA and Figure 1L for occluded right MCA at 48 hours.
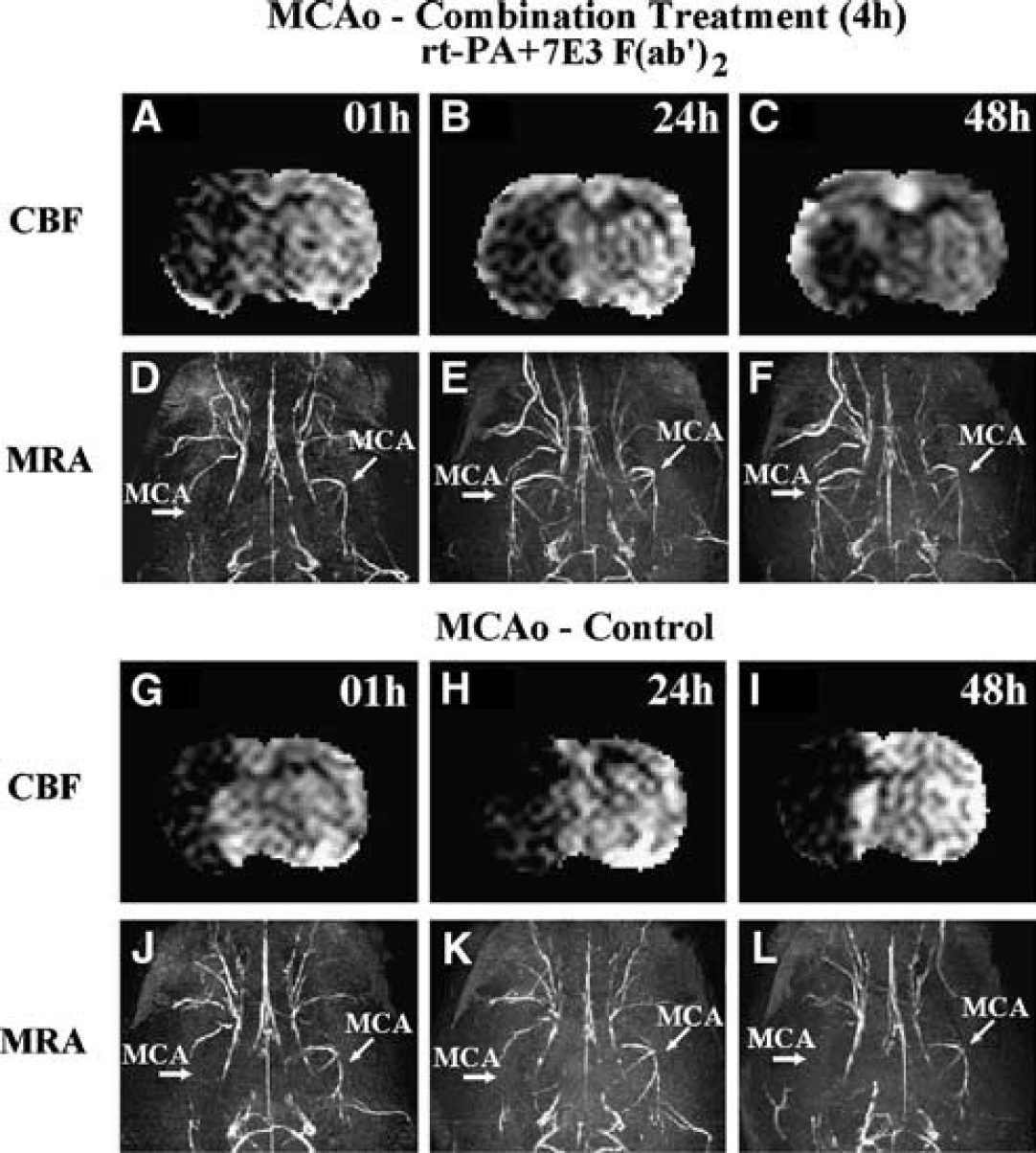
Cerebral blood flow (CBF) maps acquired by magnetic resonance imaging (MRI) at 1 hour (
Furthermore, in the treated group, 4 of 6 (67%) rats, whose right branches of MCA were patent as indicated using MRA, showed significant recovery of ipsilateral CBF at 24 hours after the onset of stroke (4 rats with 193.8% ± 30.0% versus 8 rats with 110.6% ± 10.7% normalized to 1 hour CBF values, P<0.005). Figures 1A-1C present CBF obtained at 1, 24, and 48 hours after embolization from a rat subjected to a combination treatment of r-tPA and 7E3 F(ab′)2. In Figure 1A, the CBF in the right (ipsilateral) brain hemisphere was lower than in the left (contralateral) brain hemisphere. The corresponding MRA image acquired following CBF at 1 hour after MCAo (Figure 1D) confirmed that the right branch of MCA had been occluded, while the contralateral MCA was patent (tilted arrow in figures). Treatment of embolic stroke with r-tPA and 7E3 F(ab′)2 increases the local CBF value in the ipsilateral hemisphere (Figures 1B and 1C). Recanalization of the MCA (Figures 1E and 1F) and increased CBF (Figures 1B and 1C) in the cortex of the ipsilateral hemisphere were evident at 24 and 48 hours, respectively, after embolization. For the treated group, the average ipsilateral CBF values are 140.3%±19.7% at 24 hours and 188.5%±21.7% at 48 hours compared with the acute values (i.e., acquired at 1 hour after MCAo).
In contrast, most rats (80%) in the control group had occluded right branches of the MCA at 24 (Figure 1K) and 48 hours (Figure 1L) after embolization (Figure 1J for 1-hour MRA). The ipsilateral hemisphere remained hypoperfused and CBF (1 hour Figures 1G, 24 hours Figures 1H and 48 hours Figure 1I) did not recover by 48 hours after the stroke onset. For the 2 rats (20%) of the control group with saline treatment, whose MRA images indicated that the ipsilateral MCA was patent at 24 hours, none of them showed recovery of CBF at 24 hours (2 rats with 91.1%±14.5% versus 8 rats with 100.2%±23.3% normalized to 1 hour CBF values, P≥0.42). For a control rat, the 1-hour CBF map (Figure 2A) shows low CBF values in the ipsilateral hemisphere, and the 1-hour MRA image (Figure 2D) confirms blockage of the right MCA. The MRA images at 24 hours (Figure 2E) and 48 hours (Figure 2F) of this rat show a patent MCA. However, the CBF maps at 24 hours (Figure 2B) and 48 hours (Figure 2C) reveal that a very low CBF persists throughout the MCA territory. The average ipsilateral CBF values for the control group are 98.4%±18.5% at 24 hours and 118.9%±17.7% at 48 hours compared with the 1 hour CBF.
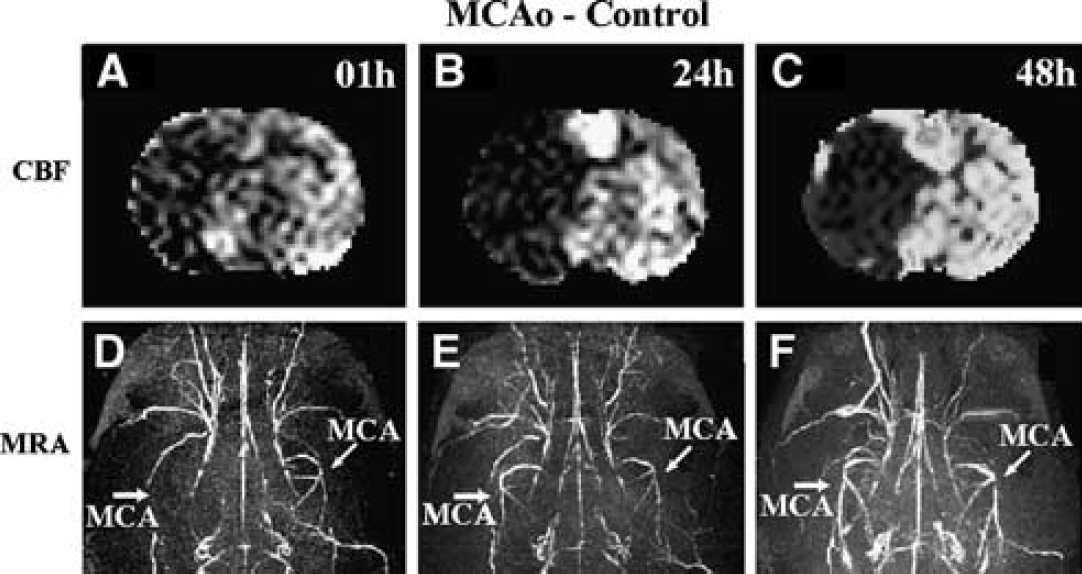
Images of cerebral blood flow (CBF) maps at 1 hour (
Measurement of microvascular patency indicates that combined r-tPA and 7E3 F(ab′)2 therapy improved microvascular patency of rat brain (Figure 3). Figure 3 shows CBF data from two rats subjected to embolic MCA occlusion that received either r-tPA and 7E3 F(ab′)2 (Figure 3A) or saline (Figure 3C). Both rats show low CBF in the ipsilateral hemispheres at 1 hour after embolic stroke onset. However, the CBF maps acquired at 48 hours after embolization differ between the treated rat (Figure 3B) and control rat (Figure 3D) in the ipsilateral hemispheres. For rats treated with r-tPA and 7E3 F(ab′)2, the 48-hours CBF shows an elevated cerebral perfusion of the ipsilateral cortex (Figure 3B), and the microcirculation of ipsilateral cortex recovered at 48 hours (Figure 3E) after MCA occlusion compared with the contralateral cortex, as illustrated in Figure 3F. The cortical tissue of both hemispheres exhibits bright microvessels because of fluorescence in the microvessels. The ipsilateral striatal tissue is darkened without any bright microvessels (in Figure 3E) because of low blood perfusion, that is, low fluorescence. For the control rat, the CBF map acquired at 48 hours after stroke onset (Figure 3D) shows no improvement of cerebral microcirculation perfusion in the ipsilateral hemisphere at 48 hours after embolization in both cortex and subcortex of the ipsilateral hemisphere. The damaged tissue does not reveal bright microvessels (Figure 3G) compared with the bright microvessels in the image of normal tissue in the contralateral hemisphere (Figure 3H). Cerebral blood flow maps measured by spin tagging MRI (Figures 3B and 3D) matched the fluorescent immunohistochemical perfusion images (Figures 3E-3H) in both control and combination treated rats at 48 hours after stroke. The fluorescence microscopy measurements of cerebral microvessels perfused with FITC-dextran revealed that the combination treatment significantly increased cortical microvascular patency (19.5±4.6 area percentage to FOV) compared with microvascular patency (12.4±3.5) in the control rats (P<0.05).
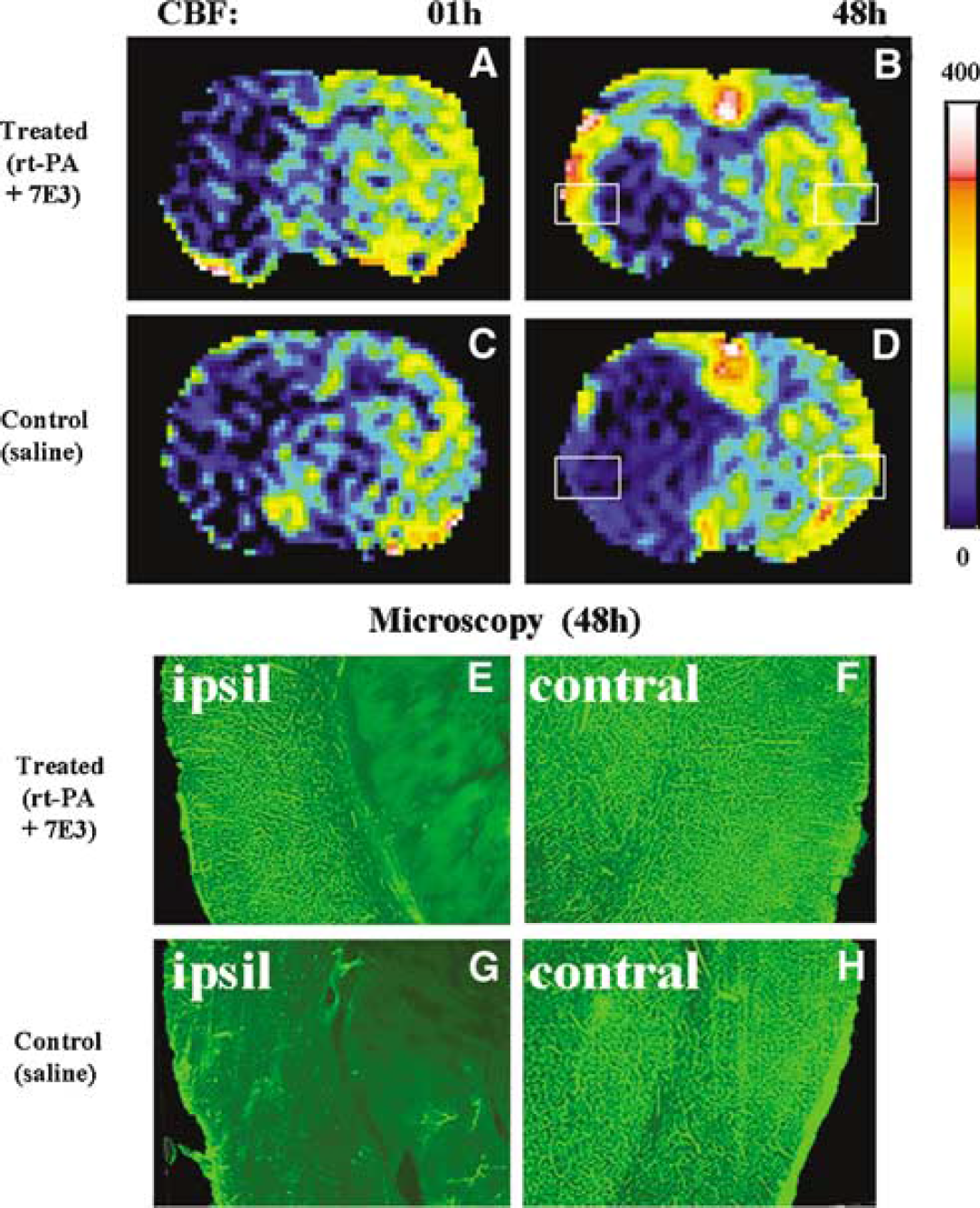
Cerebral blood flow (CBF) maps match to microscopy pictures of microvascular patency for both treated and control rats. For a rat treated with r-tPA and 7E3 F(ab′)2, the typical CBF maps at 1 hour (
Treatment of embolic stroke in rat at 4 hours after MCA occlusion with r-tPA and 7E3 F(ab′)2 significantly reduced ischemic lesion volumes compared with saline-treated rats. Figure 4 presents the relative evolution of four common MRI parameters. Each time point gives the group averages for the r-tPA and 7E3 F(ab′)2-treated group and the saline-treated control group, respectively. The average values of CBF and ADCw were normalized to the acute values. Our data reveal a significant decrease in the average relative area with low CBF (Figure 4A) at 24 hours (P<0.01) and 48 hours (P<0.02) in the treated group (0.64±0.12 at 24 hours, 0.48±0.11 at 48 hours) compared with the control group (1.07±0.09 at 24 hours, 0.88±0.11 at 48 hours). At the acute period of stroke, the T1 and T2 maps of both groups showed negligible changes between the ipsilateral and contralateral hemisphere of rat brain. The decrease of ischemic lesion size indicated from the T2 map (Figure 4B) for the treated group (0.46±0.07 at 24 hours and 0.48±0.07 at 48 hours, ratio to ipsilateral hemisphere area) was significantly lower than in the control group (0.66±0.04 at 24 hours and 0.72±0.02 at 48 hours) at 24 hours (P<0.03) and 48 hours (P<0.01). The T1 map showed similar results (Figure 4C), P<0.03 at 24 hours (0.45±0.07 for the treated group, 0.65±0.04 for the control group) and P<0.05 at 48 hours (0.47±0.07 for the treated group, 0.63±0.12 for control group). The data from the ADCw map (Figure 4D) did not show any significant difference between the treated group and the control group after the acute period of embolic stroke (24 hours P>0.3 and 48 hours P>0.4). The infarction volumes measured from the H&E histologic coronal sections using the MCID system are 19.9%±12.3% for the treated group and 38.8%±13.3% for the control group (P<0.002).
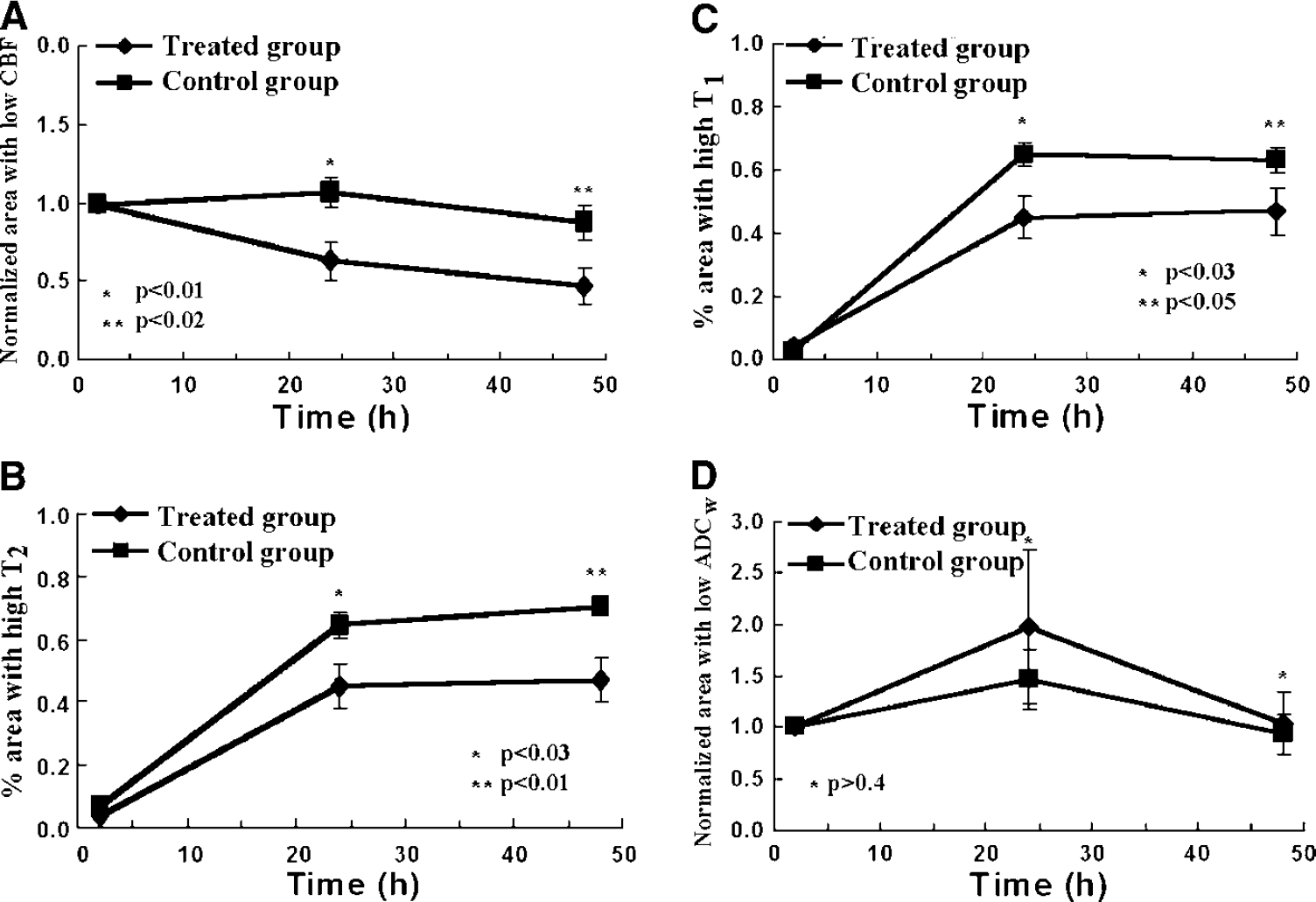
Lesion area measured from magnetic resonance imaging (MRI) parameter maps including Images of cerebral blood flow (CBF) (
Using H&E staining, we found that 4 control rats (40% of control group) and 3 treated rats (25% of treated group) showed gross hemorrhage (macroscopic), defined as blood evident to the unaided eye on the H&E-stained coronal sections and confirmed by microscopy. Our Gd-DTPA enhanced T1-weighted imaging also showed contrast agent leakage, which identifies the BBB disrupted region.
DISCUSSION
A major finding of the present study is that persistent hypoperfusion in the microvasculature induced after embolic occlusion of the MCA probably is attributed to platelet aggregation and secondary thrombosis in the downstream microvasculature. Even if the MCA is patent at 24 or 48 hours after stroke, hypoperfusion persists within the territory of the MCA. In sharp contrast, treatment with a combination of r-tPA and the GPIIb/IIIa receptor antagonist, 7E3 F(ab′)2, reduces the perfusion deficits within the cortical region of the MCA territory. An open MCA after treatment with the 7E3 F(ab′)2 antibody and r-tPA yields significantly enhanced cortical perfusion compared with control animals (Figures 1C and 2C). To our knowledge, this is the first demonstration, in vivo, of the therapeutic benefit of combination treatment targeting the cerebral microvasculature. Hypoperfusion after stroke is a well-documented event, with a long history (Ames et al, 1968). However, the coupling of embolic occlusion of the MCA with the downstream microvascular perfusion deficits, and the therapeutic effects of blocking the platelet-fibrin receptor (i.e., GPIIb/IIIa) on microvascular perfusion, even with an open MCA, has until now not been demonstrated.
Our previous study has shown by means of immunohistochemistry that platelet aggregation and fibrin deposit are key events leading to microvascular thrombosis and progressive impairment of downstream microvascular perfusion after stroke (Zhang ZG et al, 1999, 2001; Zhang L et al, 2003). Those data indicate that individual treatment of r-tPA alone or 7E3 F(ab′)2 alone when instituted 4 hours after stroke provide no therapeutic benefit, and do not reduce the ischemic lesion volume compared with saline-treated animals (Zhang L et al, 2003). Treatment with r-tPA alone outside the therapeutic window can also evoke adverse effects, that is, increase the incidence of hemorrhagic transformation and increase ischemic lesion size (Jiang et al, 2002). Our previous data have shown a significant therapeutic benefit of combination r-tPA and 7E3 F(ab′)2 treatments; however, MRI, MRA, and tissue perfusion measurements were not performed. In this MRI study including MRA and CBF, we have investigated the effects of r-tPA and 7E3 F(ab′)2 on the treatment of embolic stroke in vivo, and the evolution of the effect of a combination therapy on brain perfusion and to relate the cerebral tissue perfusion to MCA patency.
Our results indicate that the treatment with r-tPA and 7E3 F(ab′)2 of rats subjected to embolic stroke at 4 hours after MCA occlusion increases the rate of upstream thrombolysis of the MCA, improves ipsilateral CBF, and reduces ischemic lesion. Fifty percent of combination-treated rats versus twenty percent of control rats with the MCA recanalized by 24 hours after the stroke, shown from MRA, indicate that 7E3 F(ab′)2, a potent GPIIb/IIIa antagonist, increases the efficacy of r-tPA thrombolysis of the embolus. This is consistent with observations that platelets deposit within the embolus 1 hour after MCA occlusion and persist in the region for at least 24 hours (Zhang ZG et al, 2001). Under combination treatment, 7E3 F(ab′)2 suppresses the aggregation of platelets activated by fibrinolysis with r-tPA. Activated platelets retract clots and release PAI-1, α2-antiplasmin, and factor XIII, all of which are known to contribute to the resistance of thrombolysis (Zhang ZG et al, 2001). Our MRA images show increased canalization in the combination-treated group. Thus, MRA results confirm that inhibition of platelet aggregation by 7E3 F(ab′)2 increases the efficiency of r-tPA thrombolysis, which may increase the probability of earlier recanalization after embolic stroke.
Suppression of platelet aggregation may also inhibit downstream microvessels from undergoing rethrombosis after fibrinolytic therapy. Our CBF and immunohistochemistry data show that combination treatment with 7E3 F(ab′)2 and r-tPA significantly increases downstream CBF and microvascular patency, indicating that the combination therapy reduces platelet-mediated thrombosis and improves cerebral microvascular patency. Secondary thrombosis is an evolving process. Platelet aggregation downstream in microvessels increases significantly from 1 to 4 hours after MCAo (Zhang ZG et al, 2001). The earlier MCA recanalization and suppression of platelet aggregation in microvessels may promote CBF recovery and reduction of edema and infarction volume. Our results show that combination treatment with 7E3 F(ab′)2 and r-tPA significantly increased downstream CBF and microvascular patency, indicating that the combination therapy reduces platelet-mediated thrombosis and improves cerebral microvascular patency. Among the six rats with the MCA recanalized by combination treatment, four animals (67%) showed significant recovery of ipsilateral CBF at 24 hours after onset of stroke. However, since there is no 7E3 F(ab′)2 to suppress the platelet aggregation in downstream microvessels for control animals, two rats in the control group whose MCAwere patent indicated by 24 hours MRA images did not show CBF recovery at 24 hours after MCA occlusion. This failure to reperfuse may be caused by secondary thrombosis due to platelet aggregation before recanalization. Thus, our results demonstrate that combination treatment with 7E3 F(ab′)2 and r-tPA at 4 hours after ischemia significantly enhances CBF recovery.
Dividing each animal group into two subgroups, that is, MCA opened or occluded as indicated by MRA at 24 hours, we found no statistically significant differences in the ipsilateral hemisphere CBF values among subgroups, such as combination-treated without recanalization, saline-treated with or without recanalization. The combination-treated with recanalization subgroup shows increased ipsilateral CBF compared with all other animals at 24 hours after MCAo (193.8%±30.0% versus 103.8%±11.1% normalized to 1 hour CBF, P<0.005). This result indicates that recanalization alone or too late, or prevention platelet aggregation alone is not sufficient to achieve effective reperfusion. Thus, both early MCA recanalization and suppression of platelet aggregation downstream are important for thrombotic therapy. In this study, we show, both by MRI as well as FITC fluorescence perfusion data, that even if the MCA is patent, microvascular hypoperfusion is present. A factor mediating this hypoperfusion is the activation and aggregation of the platelet. Using detailed histologic analyses, we have previously shown that the platelet provokes secondary thromboses (Zhang ZG et al, 1999, Zhang ZG et al, 2001; Zhang L et al, 2003) We now show, using the intervention of an agent that blocks the final pathway to platelet aggregation (i.e., a GPIIb/IIIa receptor antagonist), that thrombolysis with rtPA, even at a time beyond the therapeutic window (i.e., 4 hours), increases the likelihood of recanalization of the MCA. Importantly, compared with control animals, microvascular cerebral perfusion is significantly increased whether the MCA is patent or not. Treatment of embolic stroke in the rat with rtPA at 4 hours after stroke onset fails to improve and likely worsens histologic and neurologic outcome (Zhang RL et al, 1999; Jiang et al, 2000; Zhang ZG et al, 2002; Zhang L et al, 2003) and yields a persistent and severe hypoperfusion (Jiang et al, 2000). Our data presented in this study explicitly show by means of MRI and fluorescence perfusion measurements, for the first time, that at 4 hours after embolic stroke we can significantly reduce microvascular perfusion deficits mediated by the GPIIb/IIIa receptor.
The ability of T2-weighted MRI to identify infarcted tissue is primarily based on vasogenic edema (Hoehn-Berlage et al, 1995). Vasogenic edema develops when the BBB is disrupted. Based on the T2 measurement in the present study, the decrease in ischemic lesion size for the treated group was significantly lower than in the control group at 24 hours (P<0.03) and 48 hours (P<0.01). This suggests that the combination treatment inhibits the developing vasogenic edema and prevents progressive disruption of the BBB. Thus, 7E3 F(ab′)2 preserves the BBB by reducing platelet activation.
The reduction of platelet deposition and activation in microvessels diminishes MMP9 activity, because platelets are a cellular source of MMP9 (Sawicki et al, 1997; Fernandez-Patron et al, 1999). Reduction of MMP9 decreases degradation of collagen type IV since MMP9 cleaves collagen type IV. Collagen type IV is one of the major basal lamina components of cerebral microvessels (Foellmer et al, 1983). Degradation of collagen type IV increases disruption of the BBB and causes hemorrhagic transformation after stroke (Zhang ZG et al, 2000, 2001). The increased risk of hemorrhagic transformation limits the clinical use of tPA (NINDS, 1995). Previous studies by our group (Zhang L et al, 2003) and others (Sawicki et al, 1997; Fernandez-Patron et al, 1999) indicate that the combination treatment significantly reduced degradation of collagen type IV and the number of MMP9 immunoreactive vessels.
Compared with our previous gross hemorrhage data of 40% for control group rats and 62% for treated group rats with r-tPA alone at 4 hours after MCA occlusion (Jiang et al, 2002), the gross hemorrhage data in the present study are 40% for control group rats and 25% for combination treatment rats. This suggests that the combination therapy does not increase and may reduce the incidence of hemorrhagic transformation. Furthermore, since 7E3 F(ab′)2 could amplify the efficacy of fibrinolytic therapy with r-tPA, the dose of r-tPA may be reduced, thereby further reducing the incidence of hemorrhagic transformation (Levy et al, 1994).
A method to assess the risk of HT in ischemic cerebral tissue after stroke would improve the safety of thrombolytic therapy. Increased acceptance of thrombolytic therapy will depend in large part on the ability of clinicians to identify patients at risk of developing hemorrhagic complications and the development of techniques to decrease such risks. Magnetic resonance imaging data show that large increases in kinv, the inverse of the apparent forward transfer rate for magnetization transfer, was detected at 3 hours after embolism and the signal intensity in the contrast-enhanced MRI was detected at 4 hours after embolism in the region where gross hemorrhage was confirmed histologically in the rat model of embolic stroke (Jiang et al, 2002). However, accurate detection and early prediction of BBB disruption and HT using MRI requires further investigation.
The therapeutic efficacy of combined r-tPA and 7E3 F(ab′)2 treatment of embolic stroke can be monitored and detected by MRI. Based on our previous MRI studies, the administration of r-tPA alone at 4 hours after onset of embolic ischemia via intravenous infusion did not reduce the ischemic infarction volume (Jiang et al, 2000, 2002) of rats. However, the combination treatment protocol of the present study administering r-tPA and 7E3 F(ab′)2 at 4 hours after embolic stroke onset significantly reduced the lesion size. These results indicate that the combination treatment may extend the therapeutic window for thrombolysis therapy to embolic stroke. Animal care concerns precluded additional MRI measurements to be performed prior to 24 hours after stroke, for example, at 12 hours.
Using the MRA and CBF images, our data show that the status of MCAo and microcirculation thrombosis can be monitored in the focal embolic stroke model in rat. The CBF and ADCw maps identify the difference of lesion area between control and treated group rats within the first few hours after MCAo. However, 24 hours after stroke onset, the difference in ischemic lesion between treatment and control groups could not be distinguished by ADCw maps. This lack of discrimination may be caused by the pseudonormalization of ADCw in damaged tissue after 24 hours ischemia. T1 and T2 maps are insensitive to lesioned tissue at an early time after stroke onset. Although the T1 maps cannot identify the ischemic region at 1 hour after MCAo, the T1 map seems more sensitive to embolic stroke than the T2 map, based on our MRI images. After 24 hours of stroke onset, both T1 and T2 maps clearly distinguish the differences in lesion size between control and treated rats.
CONCLUSIONS
Magnetic resonance imaging, fluorescence microscopy, and histology data indicate that the combined r-tPA and 7E3 F(ab′)2 treatment at 4 hours after embolic MCAo significantly reduced microvascular perfusion deficits compared with the control animals. This may be attributed to suppression of the aggregation of platelets, which increases the efficacy of fibrinolytic therapy with r-tPA and protects microvessels downstream of the occluded MCA from secondary occlusion. The MRI CBF, and T1, T2, and ADCw maps can clearly identify reduction of ischemic lesion and enhanced tissue perfusion after the combined treatment for treated animals from the control ones. Our data also show for the first time that combination therapy reduces secondary microvascular thrombosis.