Abstract
A novel photothrombotic ring stroke model was characterized by multiparametric magnetic resonance imaging, imaging of cerebral blood flow (CBF), adenosine triphosphate (ATP), pH, and histology. Ischemia was initiated by transosseous irradiation of a predefined brain area intravenously perfused by the photosensitive dye erythrosin B in male Wistar rats. In the region of the primary ring-lesion, the phototoxic reaction caused necrosis reflected by low relative ATP levels (28 ± 15%), alkalosis (pH: 7.35 ± 0.50), and histologic evidence at 14 days after lesion induction. In the ring-encircled interior region (region-at-risk), spontaneous tissue reperfusion (relative CBF: 93 ± 3%) enabled partial tissue preservation. This was demonstrated by a less impaired energy metabolism (ATP: 65 ± 23%), normal pH (7.01 ± 0.50), and still normal cellular structures shown by histologic staining. Analysis of the temporal characteristics within the region-at-risk revealed a slow continuous increase of the apparent diffusion coefficient of water (ADC) to 144 ± 16% of control (14d) and an early vasogenic edema, reflected by an increase of the T2 relaxation time to 143 ± 17% of control (2d). Both final ADC and T2 correlated well with the tissue pH within the region-at-risk, thus emphasizing the usefulness of this multiparametric noninvasive imaging approach.
In various experimental stroke models it has been demonstrated that a persisting perfusion deficit leads to irreversible cell damage from necrosis, apoptosis, or a combination of both. Thereby, cell damage depends on the severity and duration of ischemia as well as on the cell type (Hossmann, 1994). After experimentally induced thromboembolic stroke, the tissue within the lesion core is mostly irreversibly damaged even shortly after ischemic onset, despite treatment attempts (Niessen et al., 2002; Pillekamp et al., 2001). However, tissue that is in the transition between reversible and irreversible damage is believed to be potentially salvageable and may, therefore, benefit from appropriate therapeutic effort. This region-at-risk, i.e., the ischemic penumbra, has been defined as a region, in which spontaneous or evoked electric and synaptic transmissions disappear because of decreased blood flow, while energy metabolism is maintained at a degree that suffices to maintain the structural cell survival (Astrup et al., 1981).
Unfortunately, the extent and position of both the ischemic core and penumbra is neither predictable in the particular subject nor reproducible between different experiments. Since 2 decades, therefore, photochemical reactions have been used to achieve reproducible thrombosis for the investigation of stroke mechanisms (Watson et al., 1985). The pathophysiology of these stroke models has been characterized extensively without treatment (Dietrich et al., 1986, 1987a,b) but also in combination with various therapeutic interventions (Belayev et al., 1995; Dietrich et al., 1989; Hurwitz et al., 1991). Although these approaches are well suited to study the processes in the infarct core, they do not enable a thorough analysis of the ischemic penumbra. Therefore, Wester et al. established a ring stroke model in the adult rat (Wester et al., 1995). The advantage of this photothrombotic approach is its production of an anatomically predefined region-at-risk, encircled by a primary lesioned ring (ring-lesion). The pathomechanisms involved in the evolution of the ischemic damage are considered to be relevant to the evolution of clinical thromboembolic stroke with penumbra. This novel photothrombotic stroke model has been modified in order to study the consequences of instantly occurring sustained blood flow reduction, followed by late spontaneous reperfusion in the region-at-risk, i.e., from 48 to 72 hours after ischemic onset and thereafter (Gu et al., 1999a,1999b,1999c; Hu et al., 1999, 2002).
In order to characterize the temporal evolution of brain damage and potential restoration in the region-at-risk and the ring-lesion after photothrombotic ring stroke, we used noninvasive multiparametric magnetic resonance imaging (MRI). We measured tissue perfusion, water diffusion, and the T2 relaxation time, all of which have been previously used for the characterization of neuro-pathophysiologic situations (Hoehn et al., 2001). In combination with biochemical imaging and histology, we were able to provide a comprehensive description of this new stroke model.
METHODS
Animal model
All experiments were performed in accordance with the NIH animal protection guidelines and were approved by the local governmental authorities.
Surgical procedures have been described in detail elsewhere (Gu et al., 1999a,1999b; Hu et al., 2001; Wester et al., 1995). Briefly, male Wistar rats (n = 14, 280–340 g) were anesthetized initially with 4% halothane in a mixture of 65% nitrous oxide and 31% oxygen, subsequently reducing the halothane concentration to maintenance levels of 1%. Body temperature was kept at 37°C using a rectal temperature probe regulated by a thermostatically controlled heating pad. A femoral arterial catheter was inserted into the vessel, tunneled under the skin of the back, and exited through a small incision at the nape of the neck. For repeated measurements, the arterial catheter was instilled with a highly viscous fluid (Polyvinylpyrrolidone, Sigma-Aldrich Chemicals, Deisenhofen, Germany) and sealed, preventing blood from retrogradely clogging the catheter. The viscous fluid was changed every other day. In the same surgical procedure a venous line was placed in the femoral vessel for drug administration. The intravenous line was removed at the end of the first postirradiation measurement. Arterial blood pressure and blood gases were monitored acutely during the surgical procedure, and during the subsequent measurements in the magnet (see below). Blood gases were recorded every 30 minutes to ensure physiological equilibrium (pH, pao2, paCO2, glucose; 288 Blood Gas System, Ciba Corning, Fernwald, Germany). Before mounting in the stereotaxic frame, the anesthetized animal was intubated with a 14-gauge polyethylene tubing (B. Braun Melsungen, Melsungen, Germany), and mechanically ventilated (Inspira asv, Harvard Apparatus, Hollistan, MA, U.S.A.) under immobilization with pancuronium bromide (0.6 mg/kg initial bolus; maintenance 0.3 mg kg−1 h−1). After median incision of the scalp, the skull was exposed and thoroughly abraded to minimize dispersion artifacts caused by remaining blood and debris.
For the induction of the photothrombotic ring lesion, animals (n = 12) were infused with the photosensitizing dye erythrosin B (ErB, 17 mg/ml/kg b.w.) for 30 seconds, and, simultaneously irradiated for 2 minutes by a circular laser beam (outside diameter 5 mm, ring thickness 0.35 mm) at Λ = 514 nm with a ring intensity of 0.90 W/cm2 (Melles Griot, 43 Series Ion Laser, Carlsbad, CA, U.S.A.). The induced type-II photochemical reaction resulted in occlusion of the microvessels by platelet-rich thrombi (Gu et al., 1999a; Hu et al., 1999; Wester et al., 1995). The ring beam was angulated perpendicularly to the skull bone marked off by the bregma and the lambda suture, as well as by the sagittal suture and the parietal bone crest, the ring enclosing a small osseous torus. Care was taken to angulate the beam exactly, to ensure a continuous ring lesion on the cortex of the brain. Figure 1 shows the irradiation of the exposed skull of a Wistar rat. After the irradiation and the closure of the scalp wound, the animal was placed in a nonmagnetic stereotaxic holder to minimize head movement within the magnet during the MRI measurements. The venous line was removed after the acute (5-hour) MRI measurements, and, as soon as the muscle relaxant had worn off, the rat was extubated and reawakened. Sham-operated animals were only infused with erythrosin B (n = 1) or submitted to irradiation without erythrosin B infusion (n = 1).
In the 14-day observation period after lesion induction (MR measurements at 2 days, 3 days, 7 days, and 14 days after laser irradiation) the animals were reanesthetized with halothane, but instead of intubating and immobilizing the animal, they were placed in the magnet spontaneously breathing, while monitoring body temperature and arterial blood pressure, as long as the arterial line was patent.
Magnetic Resonance Imaging
All MRI measurements were performed in a 4.7-T magnet (BioSpec 47/30, Bruker Medical, Ettlingen, Germany), equipped with actively shielded gradient coils (100 mT/m; rise time <250 μs). RF transmission was achieved with a Helmholtz coil (diameter: 12 cm), and the signal was detected with a 22-mm-diameter surface receiver coil, positioned above the skull of the animal. The RF coils were decoupled from each other: the Helmholtz coil actively, and the receiver coil passively. Gradient echo fast imaging pilot scans were used for accurate positioning of the animal's head in the magnet.
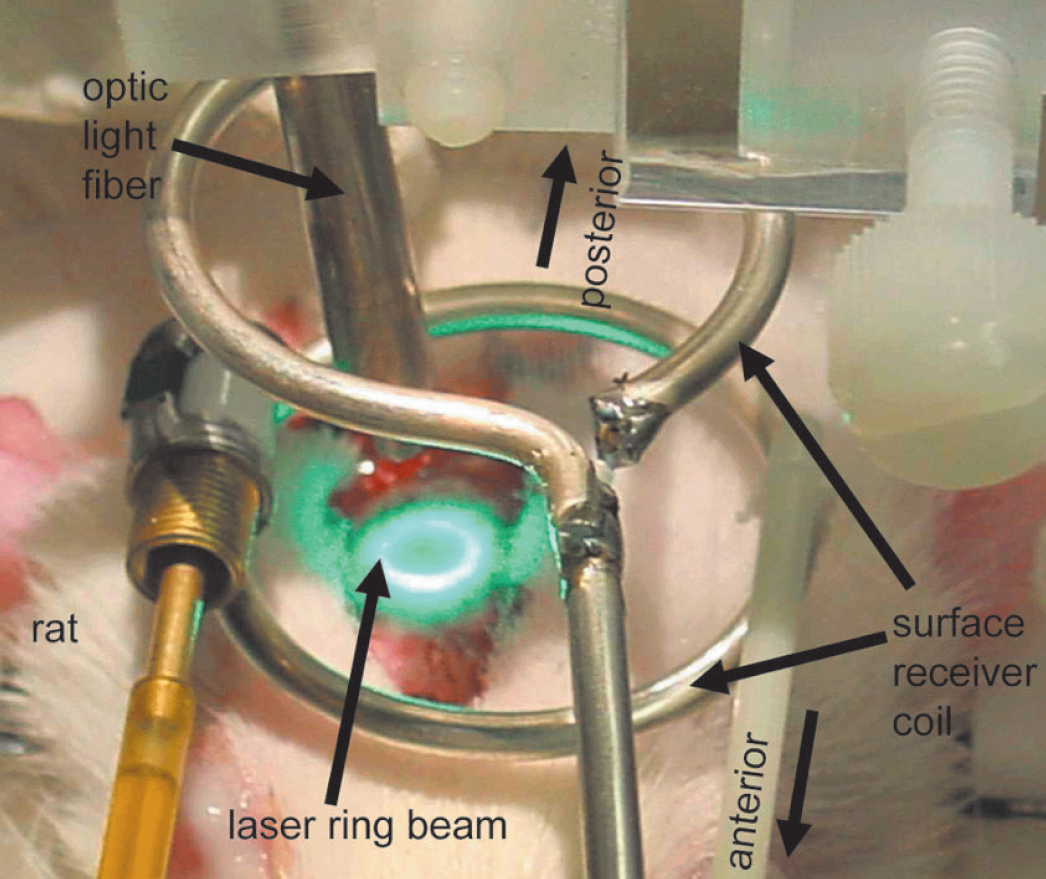
Experimental setup of a photothrombotically induced ring stroke. Optic light fiber was carefully positioned to ensure irradiation perpendicular to the skull surface. Furthermore, the position of the magnetic resonance surface receiver coil is shown.
Perfusion-weighted imaging (PWI) was performed with arterial spin labeling (labeling time: 3 seconds). During the first acquisition, arterial spins flowing through the neck were inverted adiabatically (tagging) and the inflow of labeled spins was detected with a snapshot fast low-angle shot imaging sequence (Kerskens et al., 1996): repetition time = 8.9 milliseconds, echo time = 5.3 milliseconds, field of view = 4 × 4 cm2, slice thickness: 2 mm, matrix = 128 × 64. The second acquisition left the inflowing spins undisturbed by changing the sign of the frequency offset (untagged image). Eight tagged and untagged images each were averaged to improve the signal-to-noise ratio. PWI was recorded in four different coronal slices covering the region of the ring-lesion and the surrounding area.
Diffusion-weighted images were recorded with a multislice Stejskal-Tanner spinecho sequence: repetition time = 2325 milliseconds, echo time = 35.2 milliseconds, field of view = 4 × 4 cm2, slice thickness: 1.21 mm, interslice distance = 1.21 mm, matrix: 128 × 128. Eight coronal and eight horizontal slices were acquired covering a volume extending 10 mm in the rostrocaudal (dorsoventral, respectively) direction. Coronal slices were centered around the ring-lesion, whereas horizontal slices were aligned with the skullcap. Two sets of images were acquired with two different diffusion-encoding gradient strengths (b = 30, 1,500 s/mm2) in the dorsoventral (rostrocaudal, respectively) direction, resulting in 10 minutes experimental time for one complete data set.
T2-weighted images were acquired with a multislice multiecho CPMG sequence: repetition time = 3,000 milliseconds, echo time = 12.5 milliseconds, 16 echoes, field of view = 4 × 4 cm2, slice thickness: 1.21 mm, interslice distance = 1.21 mm, matrix: 128 × 128. Data were recorded in the same coronal and horizontal slices as chosen for diffusion-weighted images.
Measurement protocol
Multiparametric MRI data sets consisting of PWI, diffusion-weighted images and T2-weighted images were acquired at 5 hours, and 2, 3, 7, and 14 days after initiation of photothrombotic ring stroke.
Cerebral blood flow-autoradiography
Before the final scanning protocol at 14 days, an additional arterial and two venous lines were placed to measure regional cerebral blood flow by quantitative autoradiography (Sakurada et al., 1978). During intravenous ramp infusion of [14]C-iodoantipyrine (50 μCi/kg b.w. in 1 mL saline), arterial blood samples were collected on preweighed filter paper for 60 seconds. At the end of tracer infusion, rats were frozen in liquid nitrogen and stored at -80°C for further processing.
Biochemical imaging
Brains were removed from the skull in a cold box at -;20°C and sliced into 20-μm thin coronal sections using a cryostat microtome. These slices corresponded to the coronal apparent diffusion coefficient (ADC) and T2 images obtained during the final MRI measurement. The regional distribution of ATP was evaluated by inducing substrate-specific bioluminescence (Kogure and Alonso, 1978). Changes of tissue pH were measured using the umbelliferone fluorescence technique by Csiba et al. (Csiba et al., 1983). Tissue alkalosis was then defined as a pH greater than 7.2.
Histology
Brain material frozen at -80°C was used for the histologic evaluation of morphologic changes induced by the photothrombotic reaction. Care was taken to stain the brain slices corresponding exactly to the acquired MR images. Twenty-micron thin coronal brain sections were cut at 1.21-mm intervals according to the slice thickness of the MR images and stained with hematoxylin and eosin, and with silver staining techniques.
Image analysis
Perfusion-weighted images were calculated by subtracting the averaged tagged image from the averaged untagged image using homemade software written in IDL (Research Systems Inc., Boulder, CO, U.S.A.). For compensation of signal loss caused by the use of the surface receiver coil, this difference image was then divided by the untagged image. Quantitative maps of the ADC were calculated pixelwise based on the monoexponential intravoxel incoherent motion model (Le Bihan et al., 1988), and T2 maps were calculated from the 16 acquired echoes, both according to described algorithms (Eis and Hoehn-Berlage, 1995).
Region-of-interest (ROI) analysis of the various MRI parameters and the biochemical images was carried out in affected and unaffected tissue regions using the image analysis software Scion Image for Windows (Version 4.0.2, Scion Corporation, MD, U.S.A.). Thereby, the exact knowledge about the real lesion position (referring to the bregma, lambda, and midline sutures) was used to transfer ROIs of identical shape in the analyzed brain slices. In biochemical images, ROI analysis was performed in coronal slices, comprising a small area within the primary ring-lesion region (ring-lesion), a region within the geometrical center of the ring-encircled interior region (region-at-risk), and a homologous area in the contralateral cortex, which was not affected by the ipsilateral ring stroke (control). The positions of the ROIs are outlined in Fig. 2A (upper right panel). All values were expressed in percent of the contralateral control region measurement. To include a maximum number of voxels, ROI analysis of MR images was done in horizontal slices (only region-at-risk) for ADC and T2. For PWI, only coronal images were recorded and thus used for ROI analysis. By this ROI definition (Fig. 2A, upper left panel) it was possible to enclose the entire region-at-risk. The temporal MR characteristics of primarily damaged tissue (equivalent to the ring-lesion) have been described earlier (Schroeter et al., 2000).
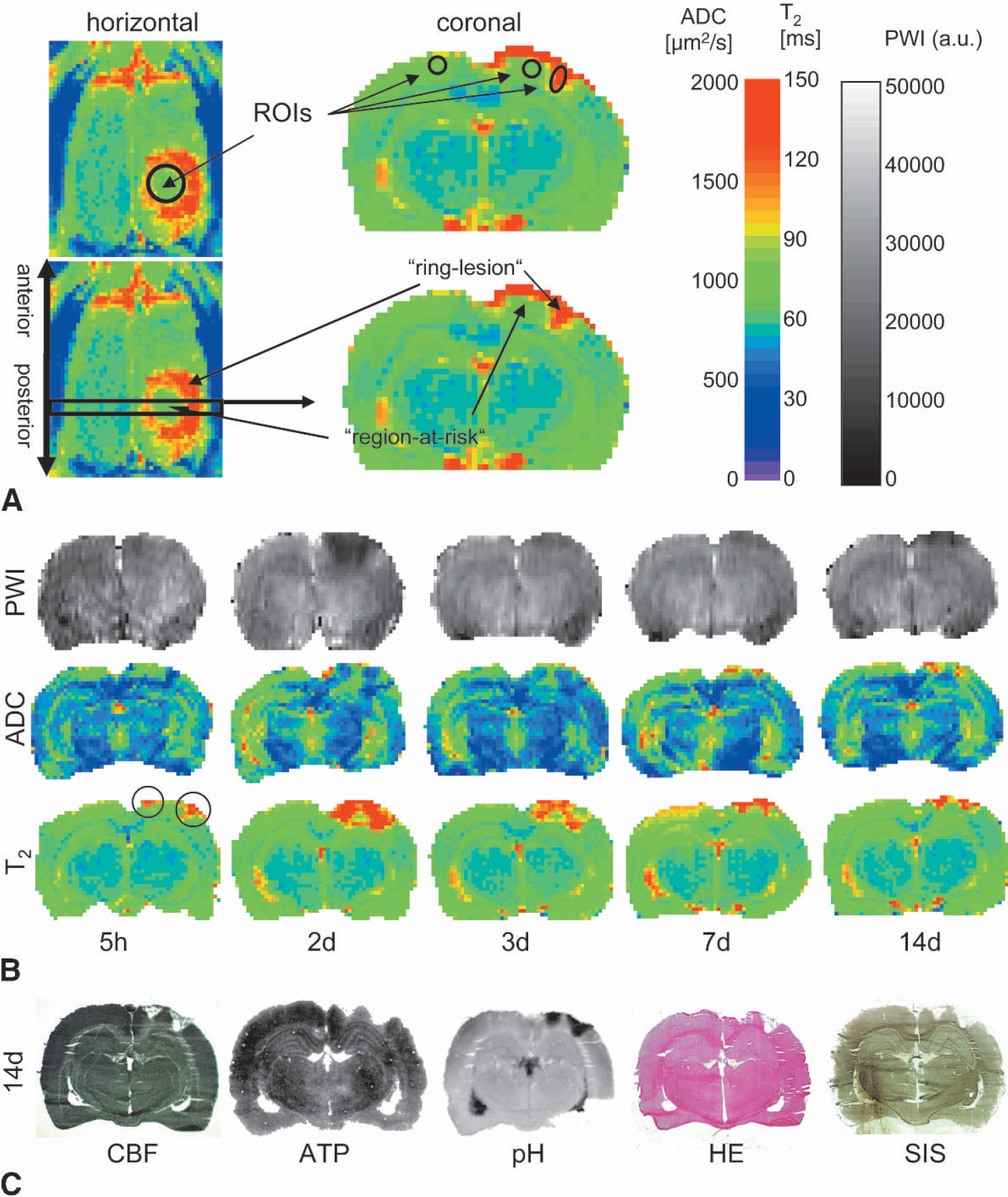
(A) The primary ring-lesion region coincides with the laser ring beam, as shown by an increase in T2 relaxation time (T2) in a cortical horizontal slice (left column). In the corresponding coronal slices, the primary ring-lesion region appears as small wedges surrounding the region-at-risk (right column). Areas for region of interest analysis are plotted in the coronal/horizontal slice (upper panel).
The results of the ROI analysis have been tested for statistical significance using a nonparametric Kruskal-Wallis test (StatView, SAS Institute Inc., Cary, NC, U.S.A.).
RESULTS
Physiologic recordings and survival times
Physiologic parameters recorded before the irradiation procedure were within normal range (n = 14): mean arterial blood pressure = 102 ± 14 mm Hg, HR = 367 ± 31 bpm, body temperature = 36.9 ± 0.3°C, paO2 = 129 ± 14 mm Hg, paCO2 = 38.2 ± 3.1 mm Hg, pH = 7.40 ± 0.04, base excess = 1.5 ± 0.8 mmol/L, glucose = 98.4 ± 11.6 mg/dL. Animals placed within the magnet, spontaneously breathing under halothane anesthesia, showed paCO2 values of ~46 mm Hg, i.e., in the higher normal range. Functional impairment of the animals upon reawakening was not observed. From a total of 14 studied animals, two rats that had been submitted to photothrombotic ring stroke died before the final MRI at 14 days, from respiratory failure. Therefore, no biochemical data was collected from these animals.
Multiparametric MRI
In all animals, photothrombotic ring stroke caused pronounced changes in MR parameters in an annular region coinciding with the laser ring beam (ring-lesion). This ring can be visualized by MRI of horizontal slices as shown in Fig. 2A (left column). The T2 relaxation time was intensely increased at 5 hours after lesion induction in this animal. The inner, only indirectly affected area (region-at-risk) showed no major changes at this early time point. Correspondingly, changes in coronal slices appear as small wedges (Fig. 2A right column, see arrows).
In Fig. 2B, multiparametric MR data of one representative animal are shown. Five hours after induction of ring stroke, the PWI signal showed only small changes. ADC decrease and T2 increase were restricted to the primarily damaged region (ring-lesion, see black circles). At 2 days after lesion induction, PWI showed a pronounced decrease in both the ring-lesion and the region-at-risk. Following the enlarged area of PWI decline, the area of decreased ADC had spread inwards, thereby sparing the center of the region-at-risk. Simultaneously, a massive T2 increase covered nearly the entire dorsal cortex except a small triangle of normal T2. The intense recovery of the perfusion signal at 3 days was accompanied by an attenuation of the T2 increase, whereas the area of decreased ADC remained unchanged. At 7 days after stroke onset, perfusion remained unchanged; ADC hypernormalized in the ring-lesion, and because of a broadening of the hyperintense wedges, parts of the region-at-risk showed also elevated ADC and T2 values. Interestingly, the perfusion signal deteriorated again 14 days after photothrombotic lesioning in the ring-lesion, whereas both ADC- and T2-maps did not show any further substantial changes. Therefore, the findings at the end of the observation period were as follows: severe hypoperfusion, pronounced elevation of ADC, and T2 in the ring-lesion, but only discretely reduced perfusion, and slightly elevated ADC and T2 in the region-at-risk.
The biochemical data at 14 days after ischemic onset (Fig. 2C) corresponded well with the MR changes: total cerebral blood flow loss, ATP depletion and alkalosis (pH > 7.3) within the ring-lesion, but complete cerebral blood flow preservation, partially maintained ATP level, and normal pH in the region-at-risk. This was also confirmed by histologic evidence, which showed severe cell changes caused by ischemia and partial cyst formation in the ring-lesion, whereas cells in the midmost part of the region-at-risk appeared to be salvaged (Fig. 2C, HE and SIS).
Biochemical and histologic characterization at 14 days after irradiation
Quantitative ATP measurements within the control region showed normal values (2.8 ± 0.3 μmol/g). In the following, ATP levels are therefore expressed as percent of control. In the ring-lesion, breakdown of brain metabolism was reflected by low ATP levels (28 ± 15% of control), while the disturbance of the acid—base equilibrium in this region was indicated by an alkalotic pH (7.35 ± 0.55) (Fig, 3, black bars). In consideration of the histologic findings, the severely impaired energy metabolism in the primarily affected ring-lesion obviously results in irreversible tissue damage.
The heterogeneous histologic findings within the region-at-risk were also reflected by the biochemical data. Although cerebral blood flow was almost completely restored in this region (relative cerebral blood flow = 93 ± 3%), we found a wide range of decreased ATP content (65 ± 23% of control). The pH was normal (pH = 7.01 ± 0.50) and the reduced but not depleted ATP levels suggest that at least a fraction of cells has been salvaged within the region-at-risk (Fig. 3, gray bars).
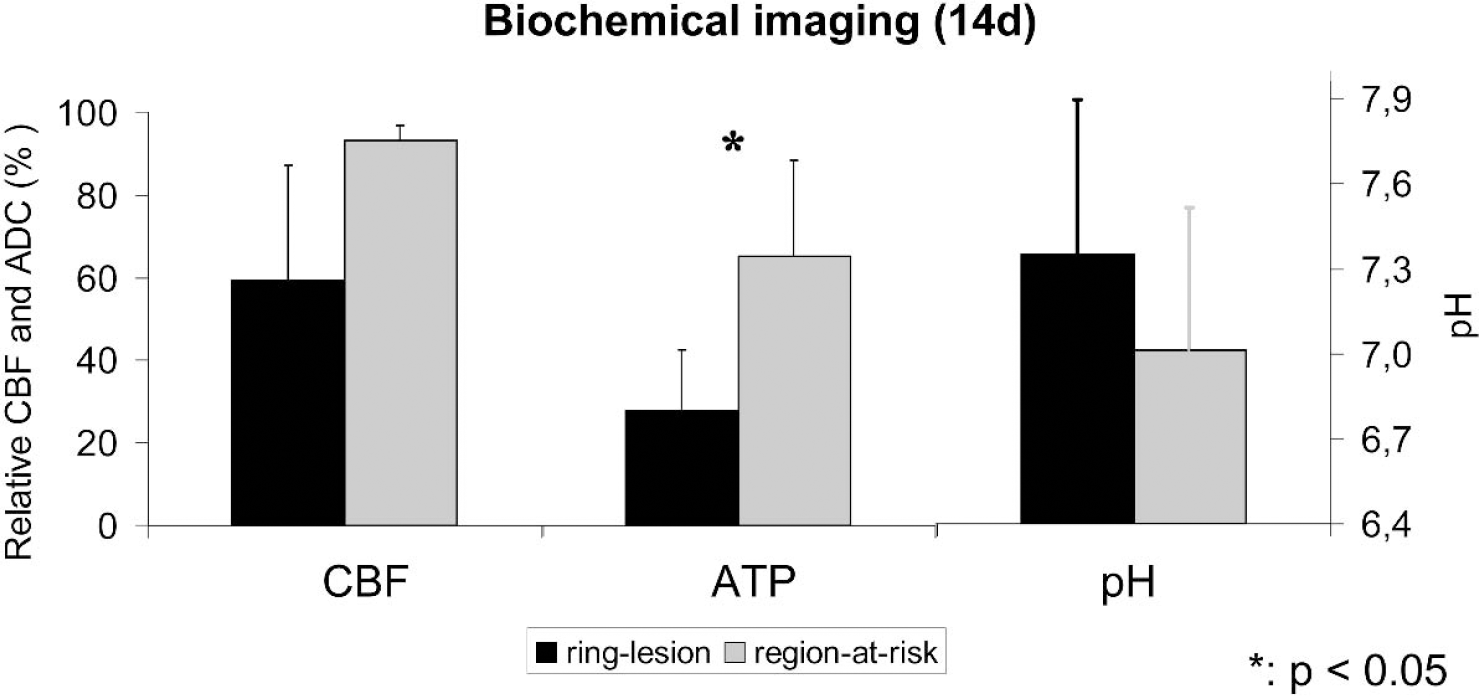
Averaged data of biochemical imaging at 14 days after photothrombotic ring stroke: cerebral blood flow (CBF) and adenosine triphosphate (ATP) are decreased in the ring-lesion, while pH is increased (black bars). In the region-at-risk, close to normal CBF, significantly higher ATP levels, and normal pH were found (gray bars).
The differences between the ring-lesion and the region-at-risk were statistically significant at P < 0.05 only for the relative ATP content.
Time course of MR changes and correlation to biochemical data
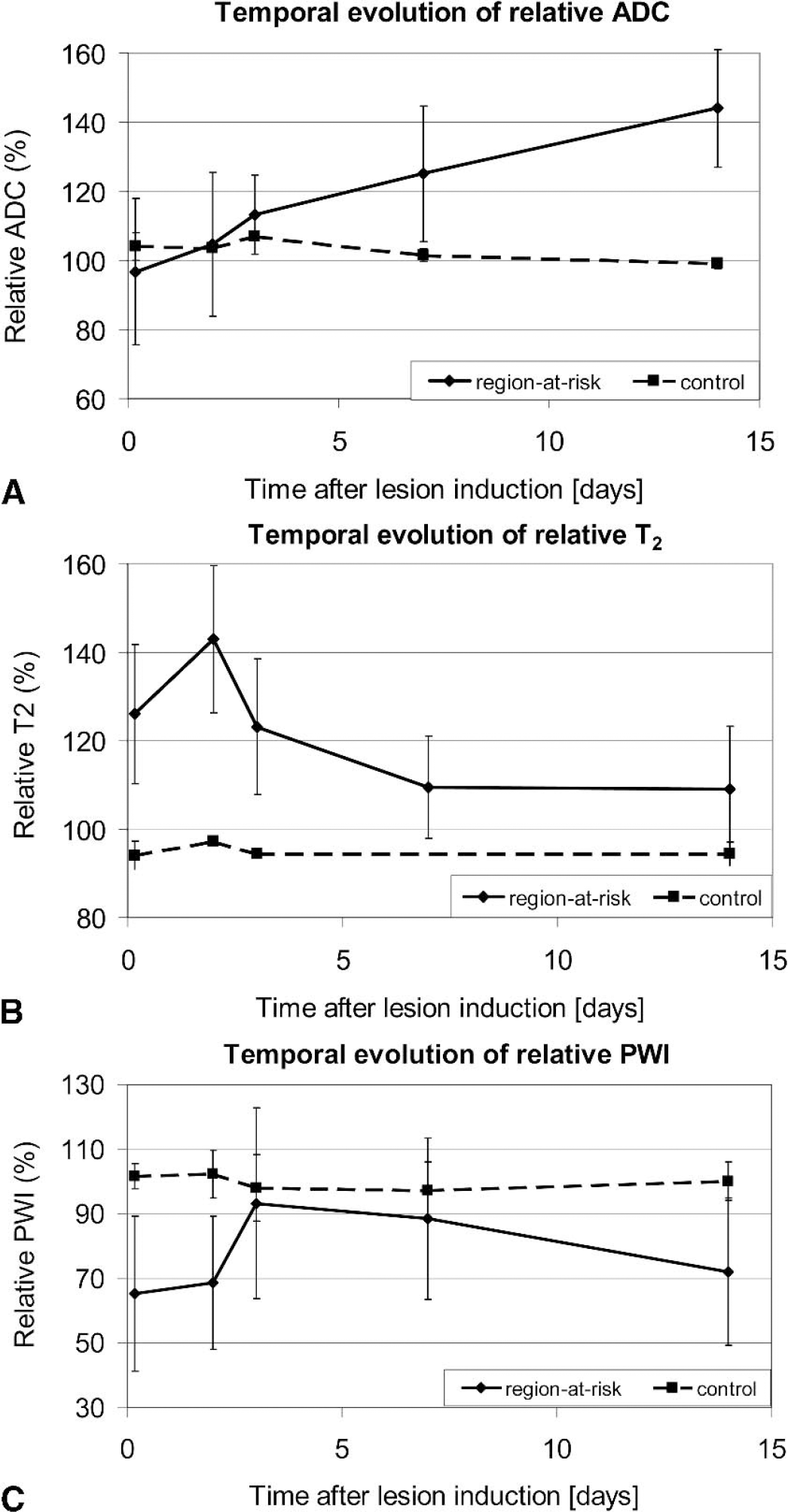
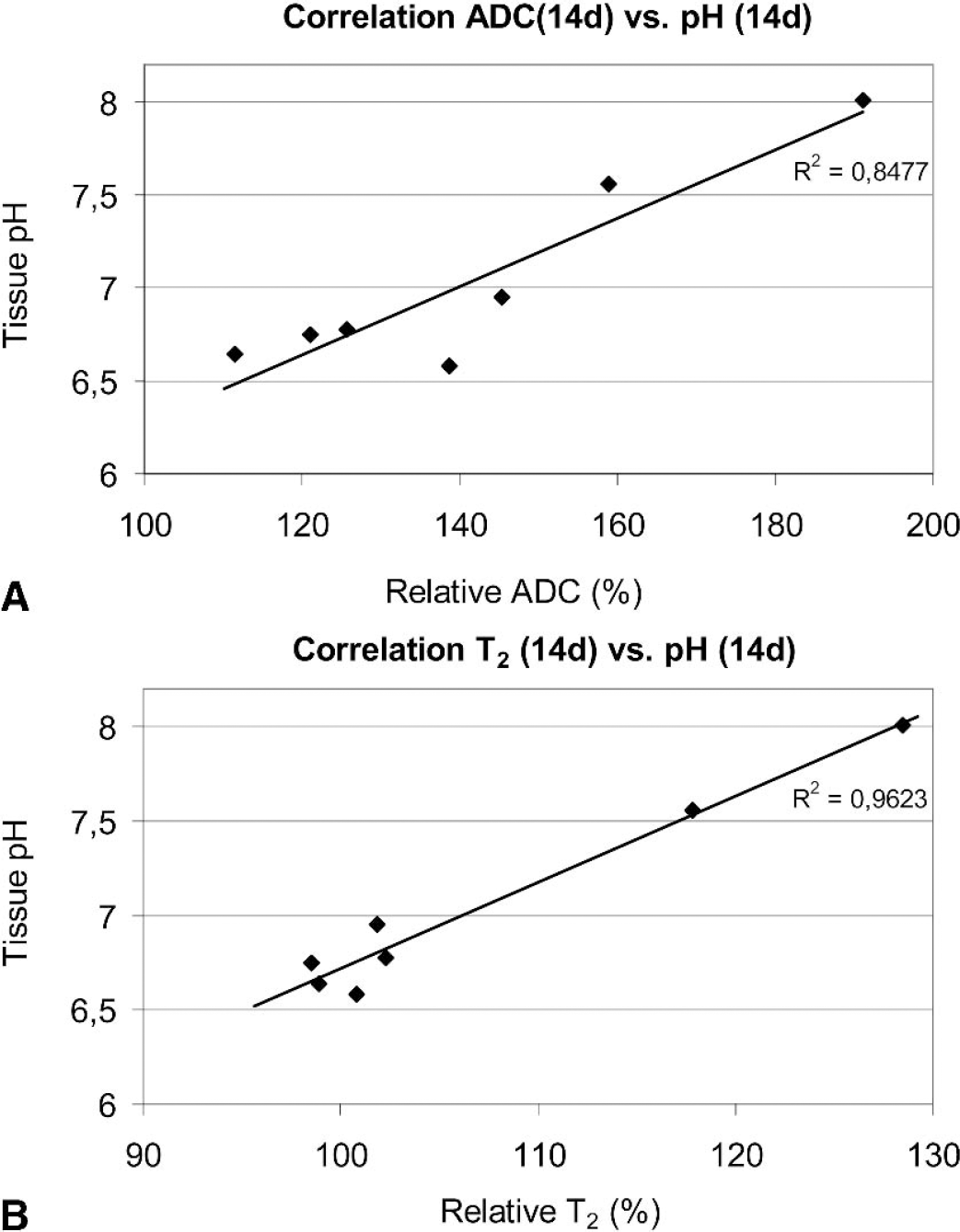
Correlation of the final MR results (14d) with the biochemical findings revealed that both relative ADC and T2 showed only a weak inverse linear correlation (r2 ≈ 0.5) to the ATP content. This can be explained by the sigmoidal shape of the relation between these parameters, indicating a sudden decline of the ATP content because of its depletion rather than a linear relation between ADC and ATP. In contrast, both MR parameters (14 days) correlated well to the tissue pH measured at 14 days after photothrombotic ring stroke (ADC vs. pH: r2 = 0.85; T2 versus pH = r2 = 0.96; cf. Fig. 5A and Fig. 5B). Both biochemical and MR (ADC, T2) parameters therefore reflect the pathophysiologic situation at this time point.
DISCUSSION AND CONCLUSIONS
In the present study we have characterized a novel photothrombotic ring stroke model, which permits the study of a reproducible region at risk, by means of multiparametric MR imaging, biochemical imaging, and histology. Biochemical and histologic analysis revealed significant differences between the ring-lesion and the region-at-risk, regarding the ATP levels. The final MR data at 14 days after ischemic onset correlated well with the histologic and biochemical outcome. This first noninvasive study of the ring stroke model showed that MRI is an appropriate tool for studying its temporal characteristics of pathophysiologic alterations.
Methodological aspects
The exact positioning of the laser ring beam on the animal's skull was crucial to achieve a reproducible lesion. Interindividual variation in the curvature of the skull bone may account for the different degrees of photothrombotic injury observed in this study. Although the laser ring beam intensity always appeared homogeneous along the annulus, slight deviations from the exact orthogonal incidence of the laser light on the skull bone might have occurred, and may have caused an asymmetric lesion resulting in an imperfectly closed ring of the photothrombotically induced microvessel occlusions. An additional contributing factor to the variation of photothrombotic injury between different individuals is the nonlinear dose-effect response of the photochemical type II reaction, i.e., between the laser light excitation and the photosensitizing dye on the one hand, and the platelet aggregation and thrombus formation on the other (Watson, 1998).
Another critical point was the repetitive exact repositioning of the animals in the magnet. With the rhinal fissure of the rats serving as reference point, we achieved a high reliability of reproducing the position in the anteroposterior direction (accuracy of 1 pixel ≈ 300 μm). Additionally, correction within the coronal planes was done by in-plane translation or rotation of imaging frames.
Histological and biochemical characterization
Correlation between MR changes and histologic/biochemical data
The partial maintenance of brain metabolism can be explained by viable cells having survived the initial photothrombotic ring stroke, whereby the proportion of these viable cells may be reflected indirectly by the relative ADC. However, there is also strong evidence for morphologic tissue recovery in the region-at-risk. The initially modest lesion severity may induce factors that are responsible for an increased ischemic tolerance (Johansson et al., 2000). Additionally, DNA repair may rescue nerve cells in this region (Chopp et al., 1996) or even induce cortical neurogenesis in the region-at-risk, as demonstrated for the first time in the photothrombotic ring stroke model (Gu et al., 2000) and also after transient middle cerebral artery monofilament occlusion (Jiang et al., 2001).
In conclusion, the photothrombotic ring stroke led to tissue necrosis in the primary ring-lesion of almost all animals. In the region-at-risk, energy metabolism was impaired, but did not break down, indicating partial cell survival, which was confirmed by histologic evidence. The pathophysiologic processes were further characterized by the noninvasive ADC mapping, thereby suggesting that the number of viable cells is inversely related to the ADC increase. Moreover, MRI of the T2 relaxation time revealed an early vasogenic edema in the ring-lesion, which spreads out into the region-at-risk. Although the existence of viable cells in the region-at-risk has been demonstrated by this approach, it remains uncertain, whether—or to what degree—these cells survived the initial photothrombotic ring stroke or are newly formed neurons. Further studies should address this question and, in addition, evaluate the influence of therapeutic interventions within this tissue region.
Footnotes
Acknowledgments
We thank Anke Stollenwerk for biochemical and histologic work, Ulla Uhlenküken for supporting the noninvasive MR-measurements, Wei Jiang and XiaoLei Hu for technical support when establishing the ring stroke model in Cologne, Bernd Radermacher for solving mechanical problems, and Dr. WeiGang Gu for fruitful discussion.
