Abstract
The regulatory interplay between laminar shear stress and proinflammatory cytokines during homeostatic maintenance of the brain microvascular endothelium is largely undefined. We hypothesized that laminar shear could counteract the injurious actions of proinflammatory cytokines on human brain microvascular endothelial cell (HBMvEC) barrier properties, in-part through suppression of cellular redox signaling. For these investigations, HBMvECs were exposed to either shear stress (8 dynes/cm2, 24 hours) or cytokines (tumor necrosis factor-α (TNF-α) or interleukin-6 (IL-6), 0 to 100 ng/mL, 6 or 18 hours). Human brain microvascular endothelial cell ‘preshearing’ ± cytokine exposure was also performed. Either cytokine dose–dependently decreased expression and increased phosphorylation (pTyr/pThr) of interendothelial occludin, claudin-5, and vascular endothelial-cadherin; observations directly correlating to endothelial barrier reduction, and in precise contrast to effects seen with shear. We further observed that, relative to unsheared cells, HBMvECs presheared for 24 hours exhibited significantly reduced reactive oxygen species production and barrier permeabilization in response to either TNF-α or IL-6 treatment. Shear also downregulated NADPH oxidase (nicotinamide adenine dinucleotide phosphate-oxidase) activation in HBMvECs, as manifested in the reduced expression and coassociation of gp91phox and p47phox. These findings lead us to conclude that physiologic shear can protect the brain microvascular endothelium from injurious cytokine effects on interendothelial junctions and barrier function by regulating the cellular redox state in-part through NADPH oxidase inhibition.
INTRODUCTION
The integrity of the cerebrovascular blood–brain barrier (BBB) is dynamically responsive to a broad range of circulatory parameters (e.g., shear, humoral factors, and infectious agents). Laminar shear stress, for example, has previously been shown to enhance brain microvascular endothelial barrier function, 1 while proinflammatory cytokines have been shown to cause endothelial barrier injury. 2 Notwithstanding these reports, extensive observational and experimental diversity is evident across BBB models contributing to limited conclusions. In this respect, a single comprehensive study model profiling the contrasting effects of laminar shear stress and proinflammatory cytokines at the molecular level of the human brain microvascular endothelium is arguably lacking within the scientific literature, as is a coherent understanding of the precise regulatory ‘interplay’ between these potent stimuli during homeostatic maintenance of the BBB.
With regard to the latter interplay concept, previous studies indicate that shear stress may help to protect the endothelium from cytokine injury via suppression of signaling pathways. Steady laminar shear has been shown to inhibit cytokine-mediated activation of MAP kinases (and specifically, JNK) in both human umbilical vein endothelial cells (HUVECs) 3 and rabbit aorta. 4 Shear-dependent upregulation of pregnane X receptor, Krüppel-like factor-2, and Akt,5–7 as well as downregulation of sphingosine kinase-1, thioredoxin-interacting protein, and adhesion molecules8–10 have also been linked to suppression of cytokine signaling in endothelial cells. Consistent with these observations, cessation of shear has been associated with cytokine-induced imbalance in MMP/TIMP (matrix metalloproteinase/tissue-inhibitor of MMP) levels leading to BBB disruption. 11 With the exception of this latter study, these previous investigations have all focused primarily on macrovascular endothelial cells and ex vivo arterial models, which are phenotypically distinct from microvascular endothelial cells of the BBB. As such, a closer investigation into the homeostatic relationship between shear stress and proinflammatory cytokines within the human brain microvascular endothelium is warranted, both to confirm the phenomenon and to identify the putative signaling mechanisms involved. Recent work from our group has shown that treatment of human brain microvascular endothelial cells (HBMvECs) with either tumor necrosis factor-α (TNF-α) or interleukin-6 (IL-6) could dose-dependently activate NADPH oxidase (nicotinamide adenine dinucleotide phosphate-oxidase) to induce reactive oxygen species (ROS) production, with direct injurious consequences for interendothelial junctional protein expression and paracellular permeability. 12 By contrast, other researchers have shown that exposure of HUVECs and bovine aortic endothelial cells to steady laminar shear can significantly decrease expression of NADPH oxidase subunits, gp91phox and p47phox, in parallel with reduced superoxide generation.13,14 In view of these observations, we therefore hypothesized that laminar shear stress could protect the brain microvascular endothelium against cytokine injury in-part via suppression of NADPH oxidase–mediated ROS production.
To address these specific issues, the following paper undertakes a series of interlinked objectives using primary-derived HBMvECs: (1) we comprehensively profile HBMvECs about the contrasting influences of shear stress and proinflammatory cytokines on the expression, localization, and phosphorylation of interendothelial junction proteins—the latter posttranslational modification in particular, is known to be robustly linked to the proper assembly and function of paracellular adherens and tight junction complexes; (2) we show the ability of laminar shear to suppress both cytokine-induced ROS production and cytokine-induced monolayer permeabilization; and (3) we show the shear-dependent attenuation of NADPH oxidase activation.
MATERIALS AND METHODS
Chemicals
Unless otherwise stated, all reagents were purchased from Sigma-Aldrich (Dublin, Ireland). Cytokines (TNF-α and IL-6) were purchased from Millipore (Cork, Ireland). Primary antisera were purchased from the following sources: antioccludin IgG, anticlaudin-5 IgG, antiphosphotyrosine IgG, and antiphosphothreonine IgG (Life Technologies, Paisley, UK); anti-VE-cadherin IgG (Abcam, Cambridge, UK) and anti-GAPDH IgG (Santa Cruz Biotechnology, CA, USA). HRP-conjugated secondary antisera were purchased from Cell Signaling Technology (Danvers, MA, USA). Alexa Fluor 488/546–conjugated secondary antisera for immunocytochemistry purposes were purchased from Life Technologies.
Cell Culture
Primary-derived HBMvECs were cultured as previously described. 12 Cells were obtained from Cell Systems Corporation (WA, USA, cat no. ACBRI 376) and routinely grown in EndoGRO MV basal medium (Millipore, SCME004) supplemented with 5% fetal bovine serum, antibiotics, and supplements. All cells (passages 5 to 12) were grown on attachment factor–coated tissue culture grade plasticware and maintained in a humidified atmosphere of 5% CO2/95% air at 37°C. For experimental purposes, HBMvECs were routinely treated with either proinflammatory cytokines (either TNF-α or IL-6, 0 to 100 ng/mL, 6 or 18 hours) or steady laminar shear stress (8 dynes/cm 2 , 24 hours). Moreover, an investigation of how preshearing of cells (8 dynes/cm 2 , 24 hours) may effect cytokine-induced changes in ROS production and barrier function was also undertaken. Application of shear stress by orbital rotation has been described previously.15,16 After experiments, cells were routinely harvested for analysis of both mRNA and protein.
Quantitative Real–Time Polymerase Chain Reaction
Extraction of total RNA, preparation of cDNA, and amplification of target cDNA sequences using gene-specific primers by RT-qPCR has been described previously. 17 Polymerase chain reaction mixtures (25 μL) were as follows: 12.5 μL of FastStart Universal SYBR Green/Rox Mastermix (Roche Diagnostics, West Sussex, UK), 8.5 μL of RNase-free water, 2 μL of cDNA, and 1 μL each of 10 μmol/L forward and reverse primers. Polymerase chain reaction conditions were as follows: denaturation at 95°C for 10 minutes followed by 40 cycles of (1) denaturation at 95°C for 15 seconds, (2) annealing at 59°C for 60 seconds, and (3) elongation at 72°C for 15 seconds. Each cDNA sample was assayed in triplicate and results were analyzed by the comparative CT method. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was routinely used for normalization purposes. All primer pairs were screened for correct product size (1% agarose gel electrophoresis) and underwent melt-curve analysis for primer–dimers; GAPDH (238 bp): forward 5′-gagtcaacggatttggtcgt-3′, reverse 5′-gagtcaacg gatttggtcgt-3′; occludin (224 bp): forward 5′-ccttcacccccatctgacta-3′, reverse 5′-gcaggtgctctttttgaagg-3′, claudin-5 (240 bp): forward 5′-gaggcgt gctctacctgttt-3′, reverse 5′-agtacttcacggggaagctg-3′; and VE-cadherin (185 bp): forward 5′-cagcccaaagtgtgtgagaa-3′, reverse 5′-cggtcaaactgcc caactt-3′.
Immunocytochemistry
Human brain microvascular endothelial cells were seeded onto attachment factor–coated glass coverslips and grown to confluency. After treatment (100 ng/mL TNF-α or IL-6 for 0, 6, or 18 hours; 0 or 8 dynes/cm 2 of steady laminar shear stress for 24 hours), coverslips were slide mounted and cells were washed with prewarmed phosphate buffered saline (PBS) before in situ fixation with ice-cold 3.7% v/v paraformaldehyde for 10 minutes. Human brain microvascular endothelial cells were then washed twice with PBS before 50 mmol/L ammonium chloride was added for 10 minutes. After two further PBS washes, the fixed cells were treated for 30 minutes with a permeabilizing/blocking solution (0.1% v/v saponin, 0.25% v/v fish gelatin, and 0.02% w/v sodium azide in PBS) before incubation overnight at 4°C with primary antisera diluted 1:50 in permablock solution (mouse anti-VE-cadherin IgG and mouse anticlaudin-5 IgG). The next day, fixed HBMvECs were washed twice with permablock solution before incubation for 1 hour in the dark with secondary antisera diluted 1:500 in permablock solution. Cells were also routinely counterstained for the actin cytoskeleton and nuclei using 1:50 Alexa Fluor 488/546 phalloidin (Life Technologies) and 1:2,000 DAPI, respectively, before a final wash with permablock and addition of DAKO fluorescent mounting media (DAKO, Carpinteria, USA). All slides were stored at 4°C until visualized by confocal microscopy (Zeiss 710 confocal, Carl Zeiss, Cambridge, UK).
Western Immunoblotting
After cytokine treatment, endothelial cell lysates were harvested and analyzed by Western immunoblotting as previously described. 12 Primary antisera were prepared in TBST (Tris-buffered saline+0.1% Tween 20, Sigma-Aldrich, Dublin, Ireland) containing 1% BSA: 0.5 μg/mL antioccludin mouse monoclonal IgG, 0.5 μg/mL anticlaudin-5 mouse monoclonal IgG, 0.2 μg/mL anti-VE-cadherin mouse monoclonal IgG, 1 μg/mL antiphosphotyrosine mouse IgG, 1 μg/mL antiphosphothreonine mouse IgG, and 0.2 μg/mL anti-GAPDH rabbit monoclonal IgG. Secondary antisera were prepared in TBST (+1% BSA): 1:2,000 HRP-conjugated goat antimouse IgG (occludin, claudin-5, and VE-cadherin), 1:1,000 HRP-conjugated goat antimouse IgG (pTyr and pThr), and 1:3,000 HRP-conjugated goat antirabbit IgG (GAPDH). Membranes were developed using a Luminata Western HRP kit (Millipore) followed by chemiluminescent imaging using a G:Box gel documentation system (Syngene, Cambridge, UK). Scanning densitometry of Western blots was routinely performed using NIH ImageJ software, with GAPDH routinely used as a loading control to facilitate densitometric normalization of bands.
Immunoprecipitation
After HBMvEC cytokine treatments, column immunoprecipitation (IP) was used in conjunction with Western immunoblotting to monitor changes in pTyr and pThr levels for occludin, claudin-5, and VE-cadherin. All IPs were performed using a Co-IP kit (Pierce, Cheshire, UK) and all relevant beaded agarose columns were prepared in accordance with manufacturer's instructions. Briefly, after treatment HBMvEC protein lysates were routinely volume adjusted to give a total protein concentration of 200 μg/300 μL using IP lysis/wash buffer. Lysates were then transferred to individual preequilibrated IP columns (containing either antioccludin, anticlaudin-5, or anti-VE-cadherin ‘pull-down’ antisera derivatized to agarose beads), which were subsequently sealed and rotated for 4 hours at 4°C. After incubation, the columns were placed in fresh collection tubes and centrifuged at 1,000 g for 1 minute. Columns were then washed thrice with 200 μL of IP lysis/wash buffer with each wash collected by centrifugation (1,000 g for 1 minute). Columns were then transferred to fresh collection tubes and 60 μL of elution buffer was added for 5 minutes and centrifuged accordingly. The collected eluent was then stored at −80°C for subsequent analysis by Western immunoblotting. For monitoring pTyr and pThr levels in IP eluants, Western immunoblotting using anti-pTyr and anti-pThr antisera was used as described above. Membranes were then stripped using Restore Western blot stripping buffer (Thermo Scientific, Loughborough, UK) and reprobed with antioccludin, anticlaudin-5, or anti-VE-cadherin antisera as described above. Finally, according to a previously described method, 12 column IP was also used in conjunction with Western immunoblotting to monitor changes in the coassociation of NADPH oxidase subunits (gp91phox and p47phox) in response to shear treatment of HBMvECs.
Transendothelial Permeability Assay
Analysis of HBMvEC permeability after cytokine and shear treatments was based on the Transwell method of Walsh et al, 1 with minor modifications described by Rochfort et al. 12 By using this method, the impact of cytokines on HBMvEC transendothelial exchange of fluorescein isothiocyanate-dextran 40 kDa (% transendothelial exchange of FITC-dextran 40 kDa) in the absence and presence of HBMvEC shear preconditioning was monitored.
Flow Cytometry
For analysis of ROS generation after either cytokine or shearing treatments, flow cytometry was used in conjunction with 5 μmol/L 2′,7′-dichlorofluorescein diacetate for ROS detection, as described previously. 12 For 2′,7′-dichlorofluorescein diacetate, excitation and emission wavelengths were 492 and 517 nm, respectively (CFDA) (i.e., FITC spectral range). All flow cytometry data analysis used FlowJo LLC software (Ashland, OR, USA).
Statistical Analysis
Each investigation entailed three independent experiments (n = 3). Moreover, within any individual experiment, assays were normally conducted in triplicate. Results are expressed as mean ± s.d. Statistical comparisons between control and experimental groups were by analysis of variance in conjunction with a Dunnett's post hoc test for multiple comparisons. Student's t-test was also routinely performed for pairwise comparisons. A value of P ≤ 0.05 was considered significant.
RESULTS
Tumor Necrosis Factor-α and Interleukin-6 Reduce Expression and Increase Phosphorylation of Occludin, Claudin-5, and VE-Cadherin in a Dose-Dependent Manner in Human Brain Microvascular Endothelial Cells
The effect of proinflammatory cytokines on mRNA expression was initially monitored. Treatment of confluent HBMvECs with either TNF-α or IL-6 (0 to 100 ng/mL, 18 hours) showed a dose-dependent reduction in mRNA levels for all three target proteins (occludin, claudin-5, and VE-cadherin) as monitored by RT-qPCR. At the upper treatment conditions (100 ng/mL, 18 hours), reductions of 40% to 50% and 35% to 45% were noted for TNF-α and IL-6 treatment, respectively (Figures 1A and 1B). Effects on target protein phosphorylation after similar cytokine treatment regimens were also considered. Treatment with either TNF-α or IL-6 significantly increased pTyr and pThr levels for all three target proteins in a dose-dependent manner as monitored by IP and Western blotting. At the upper treatment concentration for TNF-α (100 ng/mL, 18 hours), significant increases of up to 2.9 and 3.3 fold were noted for pTyr and pThr levels, respectively (Figure 2B, right). Likewise for IL-6, significant increases of up to 2.9 and 3.1 fold were noted for pTyr and pThr levels, respectively (Figure 2B, left). About cytokine effects on both mRNA (Supplementary Figure S1) and phosphorylation (Supplementary Figure S2) levels, similar trends were also noted for shorter cytokine treatments (6 hours).
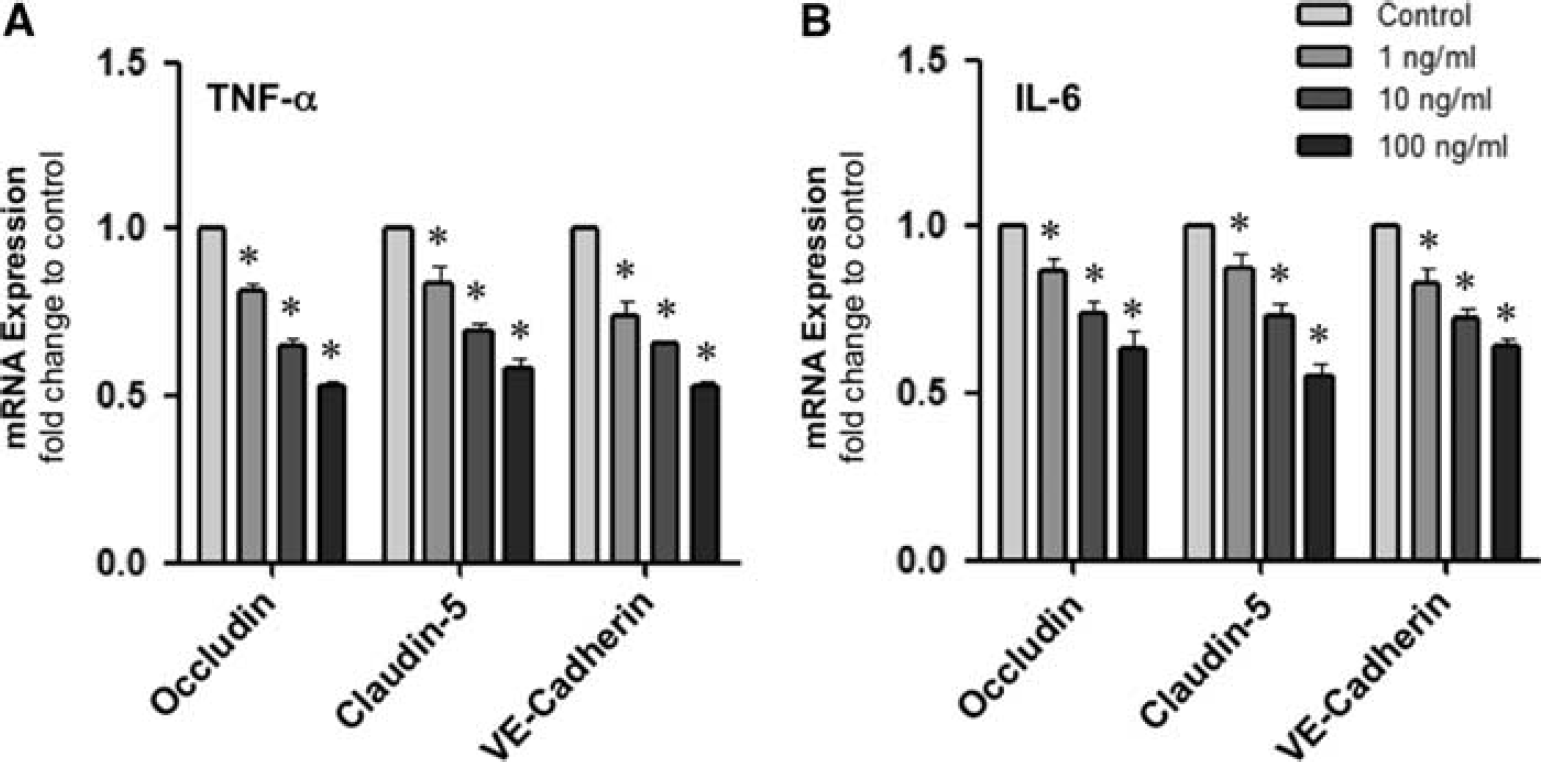
Effect of cytokines on the expression of interendothelial junction proteins in human brain microvascular endothelial cells (HBMvECs). Confluent cells were treated with (
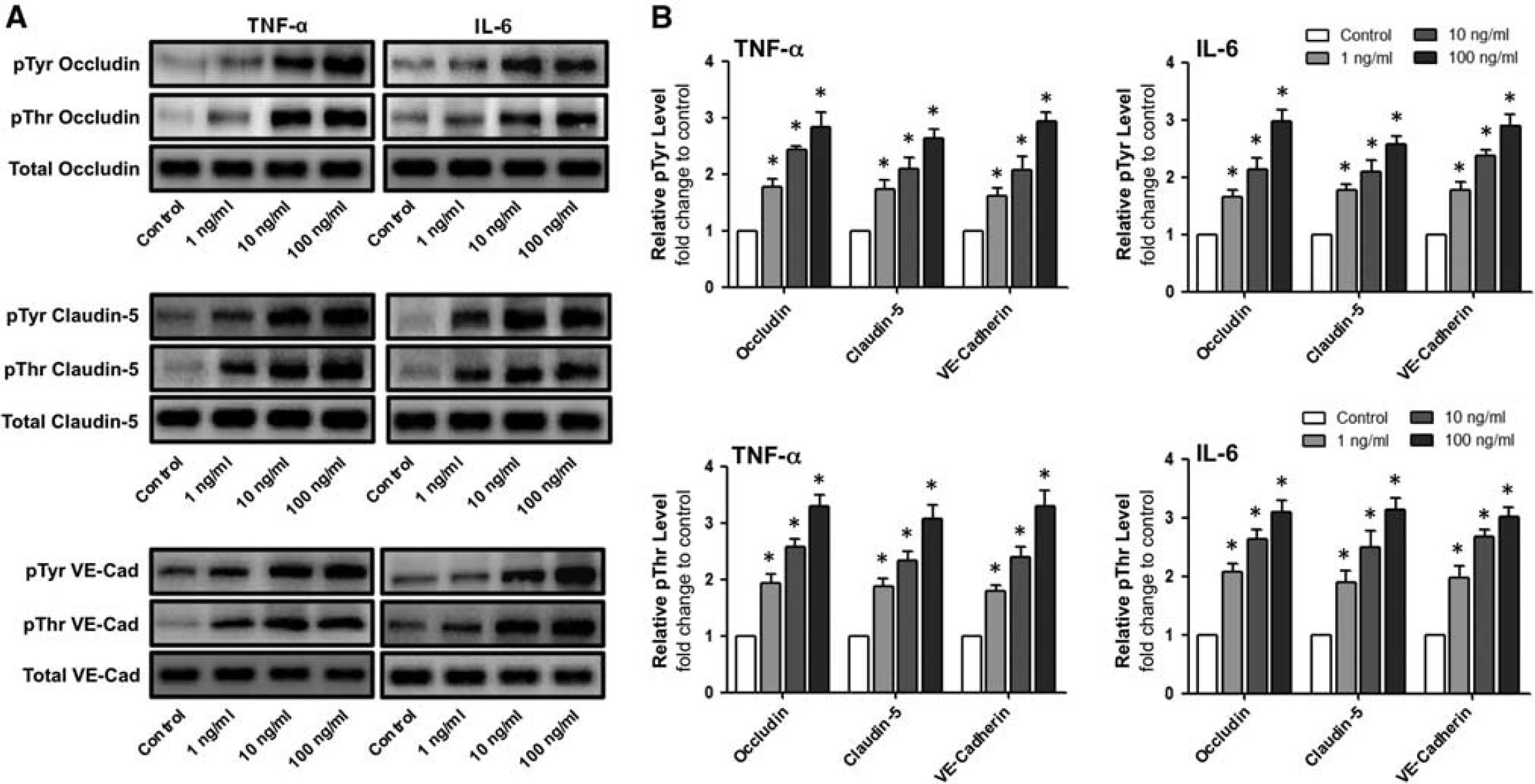
Effect of cytokines on the posttranslational modification of interendothelial junction proteins in human brain microvascular endothelial cells (HBMvECs). (
Localization of Claudin-5 and VE-Cadherin to the Human Brain Microvascular Endothelial Cell–Cell Border is Strongly Depleted by Either Tumor Necrosis Factor-α or Interleukin-6 and Enhanced by Shear Stress
In addition to transcriptional and posttranslational effects, the impact of proinflammatory cytokines on the localization of interendothelial protein targets to the cell–cell border was also monitored by confocal microscopy. We noted an almost complete diminution of both claudin-5 and VE-cadherin immunoreactivity at the HBMvEC membrane after 18-hour treatment with 100 ng/mL of either cytokine (Figure 3A). By contrast, exposure of cells to steady laminar shear stress at 8 dynes/cm 2 for 24 hours significantly enhanced localization of either protein to the cell–cell border (Figure 3B).
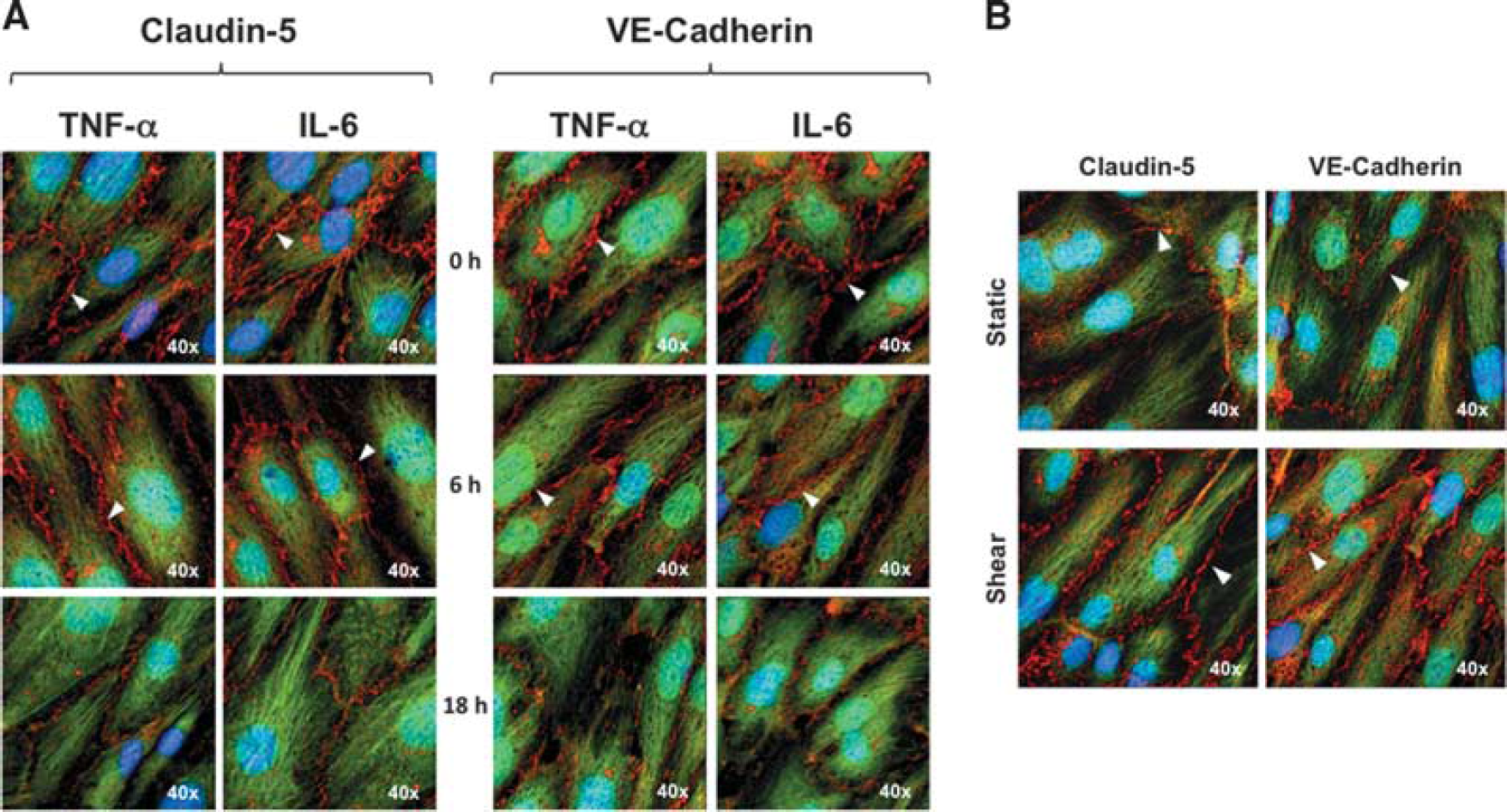
Effect of cytokines and shear on the localization of claudin-5 and VE-cadherin in human brain microvascular endothelial cells (HBMvECs). Confluent cells were treated with (
Shear Stress Increases Expression and Reduces Phosphorylation of Occludin, Claudin-5, and VE-Cadherin in Human Brain Microvascular Endothelial Cells
The effect of shear stress (8 dynes/cm 2 , 24 hours) on the expression of occludin, claudin-5, and VE-cadherin was next monitored. By using RT-qPCR, mRNA levels for all three proteins were increased under shear conditions (Figure 4A), with fold changes ranging from 1.15 (occludin), 1.45 (claudin-5), and 1.90 (VE-cadherin). Likewise, Western blotting showed elevated protein levels in shear (Figure 4B), with fold changes ranging 1.65 (occludin), 1.80 (claudin-5), and 1.75 (VE-cadherin). In contrast to the elevated expression levels, shear stress (8 dynes/cm 2 , 24 hours) was found to strongly reduce the pTyr and pThr phosphorylation level of all the three interendothelial proteins by 60% to 70% and 50% to 60%, respectively (Figures 4C and 4D).
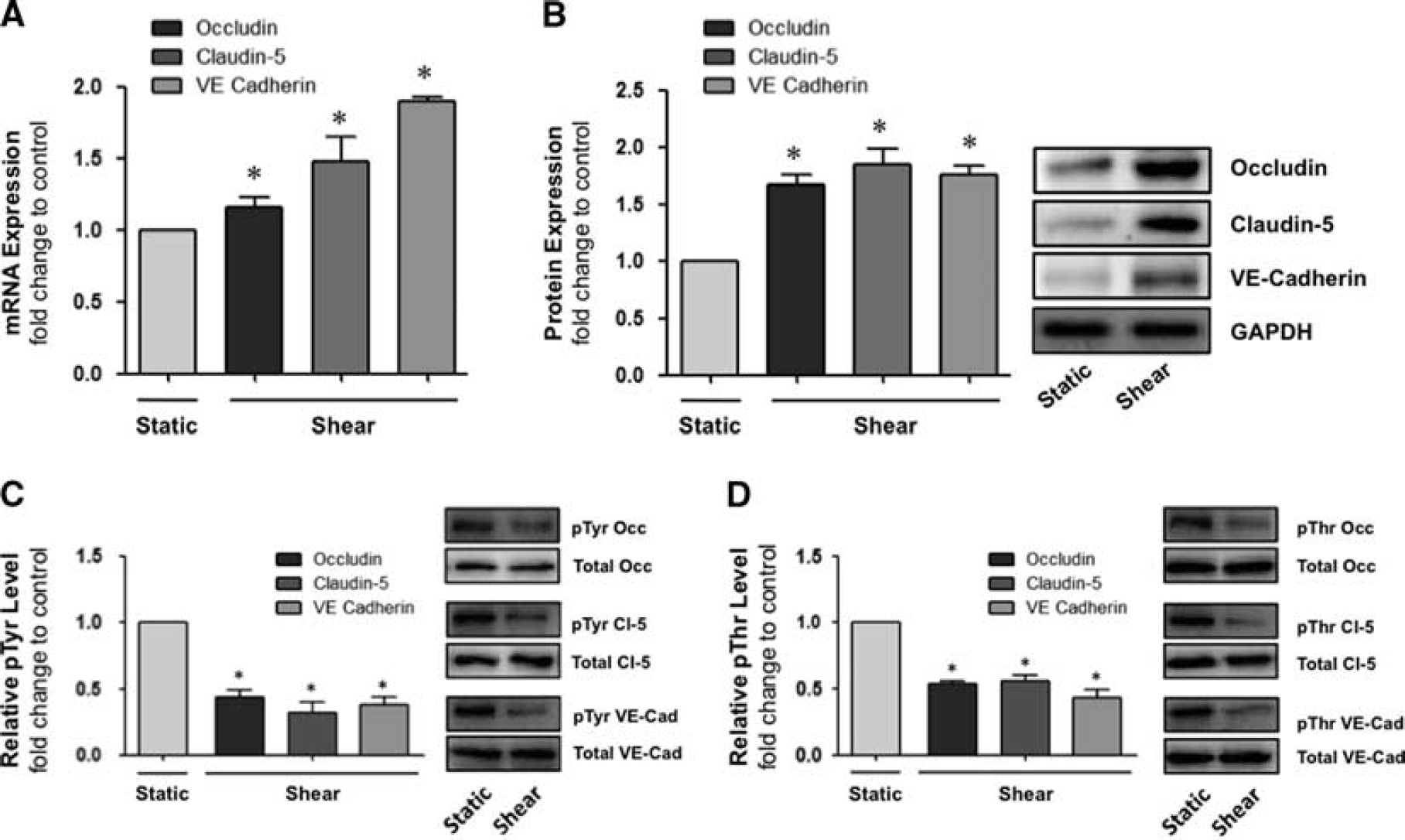
Effect of shear on the expression and posttranslational modification of interendothelial junction proteins in human brain microvascular endothelial cells (HBMvECs). Confluent cells were exposed to steady laminar shear stress (8 dynes/cm
2
, 24 hours). After treatment, total cell protein and mRNA was harvested for immunoprecipitation (IP)/Western blotting and RT-qPCR analysis, respectively. Upper histograms represent (
Shear Stress Attenuates Cytokine-Induced Permeabilization in Human Brain Microvascular Endothelial Cells
The influence of shear stress on cytokine-induced permeabilization of HBMvECs was investigated. Confluent cells were preconditioned for 24 hours under either static or shear (8 dynes/cm 2 ) conditions before being treated for 18 hours with either TNF-α or IL-6 (0 to 100 ng/mL), with static and shear conditions being maintained for the duration of cytokine treatments (Figure 5A). After treatment, cells were trypsinized and replated at high density into transwell filters Millipore (Danvers, MA, USA) for assessment of transendothelial permeability to FITC-dextran 40 kDa as previously described. 1 Preshearing of HBMvECs was observed to attenuate cytokine-induced permeabilization by up to 60% at 1 ng/mL for either TNF-α or IL-6. At higher cytokine concentrations (10 or 100 ng/mL), the shear-dependent attenuation of cytokine-induced permeabilization was slightly lower at 43% to 47% (Figure 5B).
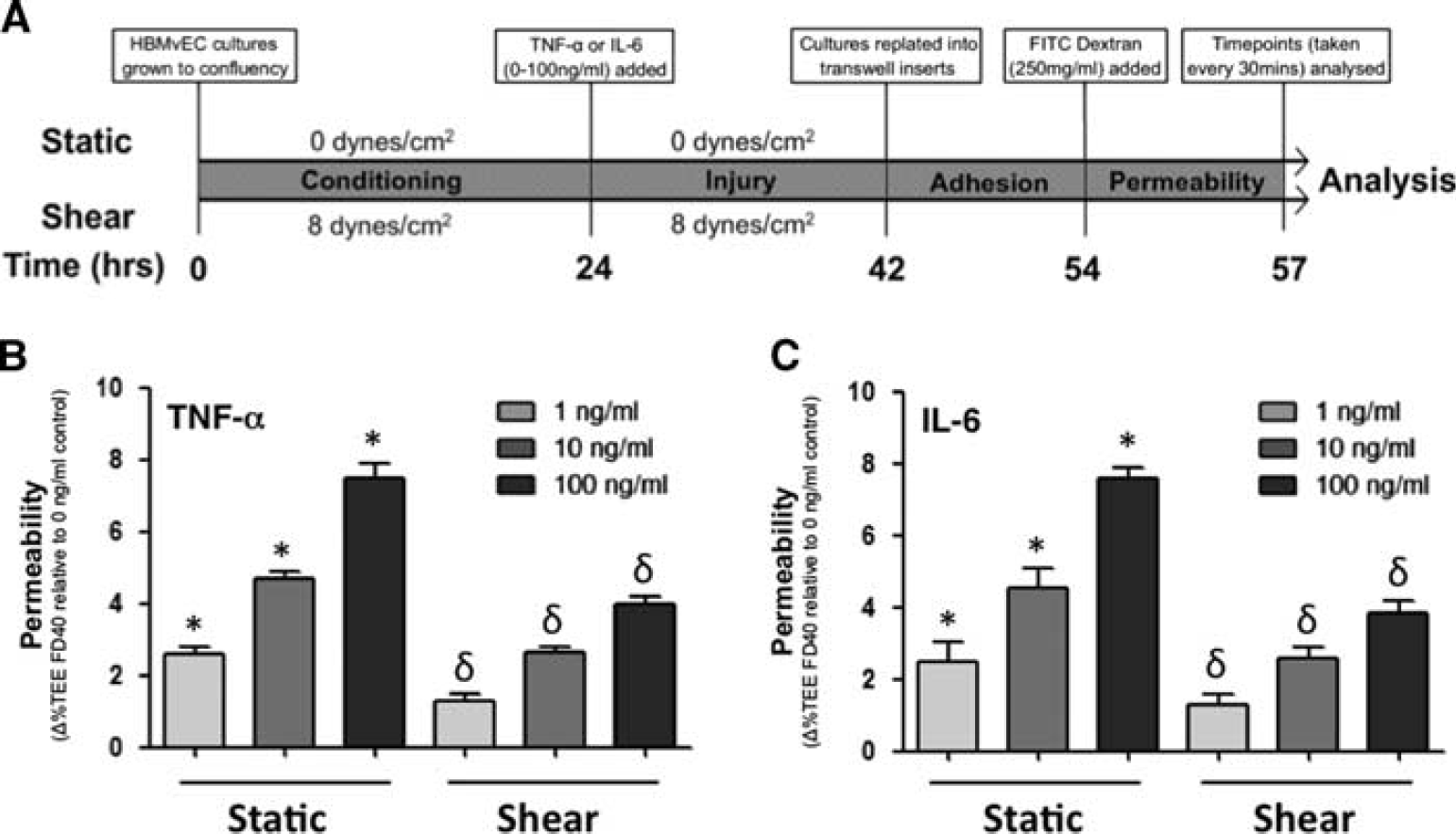
Effect of shear preconditioning on cytokine-induced human brain microvascular endothelial cell (HBMvEC) permeabilization. (
Shear Stress Attenuates Cytokine-Induced ROS Generation and NADPH Oxidase Activation in Human Brain Microvascular Endothelial Cells
By using flow cytometry in conjunction with CFDA to monitor ROS generation in HBMvECs, preshearing of cells (8 dynes/cm 2 , 24 hours) was shown to significantly reduce ROS generation induced by 18-hour treament with either TNF-α or IL-6 by 57% or 54%, respectively (Figures 6A and 6B). The effect of shear stress on the expression and coassociation of gp91phox and p47phox in HBMvECs was also investigated. These subunits constitute membrane-bound and cytosolic components of the NADPH oxidase complex, respectively, and as such, constitute a useful index of NADPH oxidase activation. 18 After treatment of confluent HBMvECs with shear (8 dynes/cm 2 , 24 hours), cells were harvested for protein expression analysis by Western blotting. Shear stress decreased protein expression of gp91phox and p47phox by 35% and 25%, respectively (Figure 7A). Shear also decreased gp91phox/p47phox subunit coassociation, as monitored by co-IP, by ∼30% to 35%, irrespective of which subunit was used as the ‘pull-down’ target and which as the Western blot target (Figure 7B).
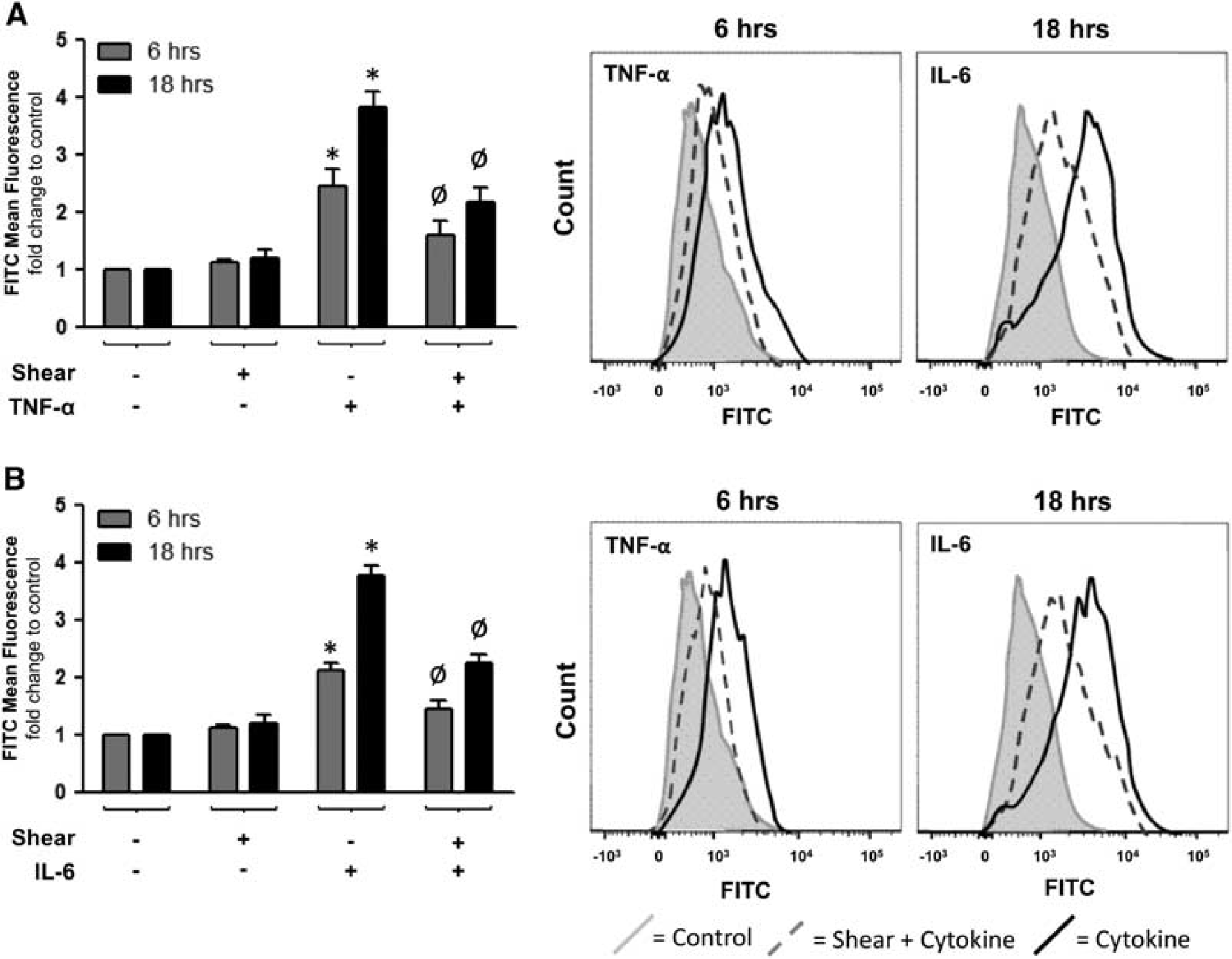
Effect of shear preconditioning on cytokine-induced ROS generation in human brain microvascular endothelial cells (HBMvECs). Confluent cells were preconditioned for 24 hours under static or shear conditions (0 or 8 dynes/cm
2
, 24 hours) before treatment of cells with tumor necrosis factor-α (TNF-α,
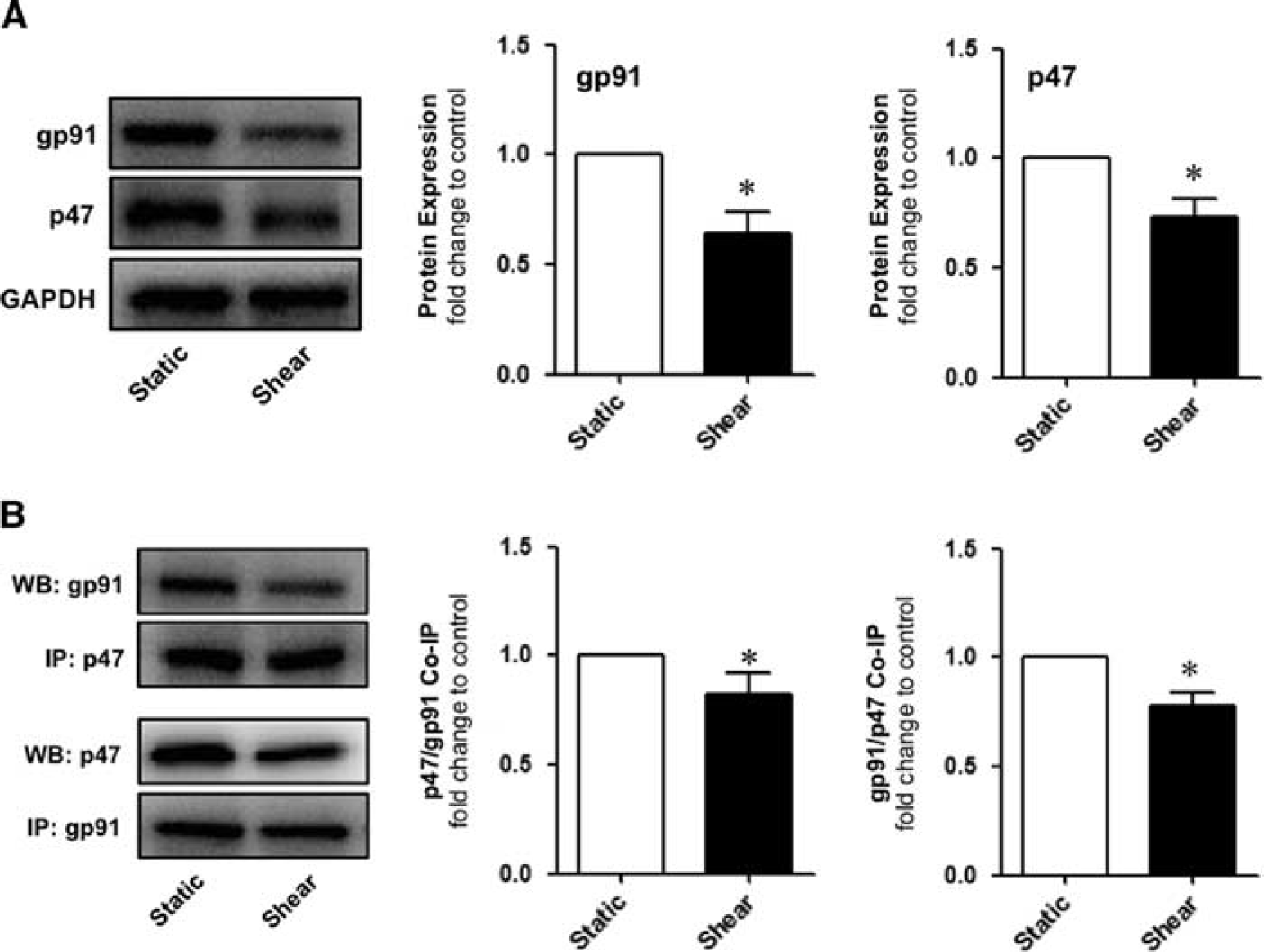
Effect of shear on NADPH oxidase activation in human brain microvascular endothelial cells (HBMvECs). Confluent HBMvECs were exposed to steady laminar shear stress (8 dynes/cm
2
, 24 hours). After treatment, cells were harvested for analysis of NADPH oxidase subunit expression and coassociation, as monitored by Western blotting and co-IP, respectively. (
DISCUSSION
Blood flow–associated shear stress is a potent differentiative stimulus capable of modulating endothelial phenotype and responsiveness to circulatory conditions. Indeed, various studies present evidence that shear plays a beneficial role in the homeostatic regulation of the cerebral microvasculature, contributing to its pro-barrier, antiinflammatory, and antithrombotic properties.1,11,16 It is noteworthy that the precise regulatory interplay between laminar shear stress and proinflammatory cytokines at the BBB interface remains largely undefined. In this respect, a number of recent studies indicate that shear may potentially shield the vascular endothelium against injurious cytokine signaling,3–10 although these papers do not focus on endothelial barrier integrity, and primarily use macrovascular endothelial cell models, which are phenotypically quite distinct from microvascular endothelial cells of the BBB. A closer mechanistic examination of how shear stress may protect the human brain microvascular endothelium against cytokine challenge is therefore warranted. Such an experimental model is currently absent from the scientific literature and would help to elucidate the mechanisms underlying cerebrovascular homeostasis and disease. Recent work from our laboratory has confirmed that proinflammatory cytokines can dose-dependently induce NADPH oxidase activation and ROS production in HBMvECs leading to barrier disruption. 12 In the current study, we therefore hypothesized that physiologic shear could counteract the injurious permeabilizing actions of proinflammatory cytokines on the brain microvascular endothelium in-part through suppression of NADPH oxidase–mediated ROS production.
We began our investigations by comprehensively profiling the contrasting effects of laminar shear stress versus proinflammatory cytokines at the molecular level of the human brain microvascular endothelium, an analysis currently absent from the scientific literature. Our initial experiments clearly showed cytokine-dependent decrease in the mRNA expression of interendothelial junction proteins (occludin, claudin-5, and VE-cadherin), in parallel with a significant increase in their pTyr and pThr phosphorylation levels. Similar observations were made for both TNF-α and IL-6 treatments, and all observations were dose dependent up to 100 ng/mL of cytokine. Either cytokine also caused almost complete loss of claudin-5 and VE-cadherin immunostaining at the cell–cell border where adherens and tight junctions assemble. These results are consistent with the increased microvascular endothelial permeability previously reported by our laboratory under similar cytokine treatment conditions. 12 In stark contrast, laminar shear stress had strongly opposing effects to those seen with cytokine treatments, significantly increasing the expression (mRNA and protein) of occludin, claudin-5, and VE-cadherin, while simultaneously decreasing their pTyr and pThr phosphorylation levels. Moreover, exposure to shear upregulated localization of claudin-5 and VE-cadherin immunostaining at the HBMvEC cell–cell border. These results are consistent with the decreased microvascular endothelial permeability previously reported by our laboratory under similar shearing conditions. 1 Our observations for shear stress are also consistent with earlier studies showing shear-dependent upregulation of occludin and VE-cadherin expression in bovine brain microvascular endothelial cells 16 and HUVECs, 19 respectively, while shear-dependent upregulation of VE-cadherin expression at interendothelial junctions in vivo has been reported in atheroprotected aortic regions. 20
The starkly opposing effects of either stimulus on the phosphorylation state of junctional proteins in HBMvECs warrants further discussion. Phosphorylation changes have been directly linked to the dynamic plasticity of intercellular junctions and endothelial permeability. Elevated pTyr levels of junctional proteins for example have been widely reported to correlate with BBB failure in response to a variety of permeabilizing stimuli including hypoxia, 21 TGF-β1, 22 alcohol, 23 and cerebral ischemia. 24 Comparable observations have also been made in various non-BBB endothelial models.25,26 Elevated pThr levels of zonula occludens-1 (ZO-1), occludin, and claudin-5 have also been linked to barrier injury in various BBB and non-BBB endothelial models.27–29 Unlike tyrosine phosphorylation, however, disparate observations on the impact of threonine phosphorylation on junctional assembly have been reported,30,31 highlighting stimulus-specific influences on the phosphorylation dynamics of endothelial junctions. Suprisingly, there are no existing studies that comprehensively address the direct influence of proinflammatory cytokines on junctional phosphorylation events within the human brain microvascular endothelium. To our knowledge, the current paper is the first to do so. Moreover, our findings with TNF-α and IL-6 are consistent with studies by Nwariaku et al 32 and Lo et al, 33 who respectively show in HUVECs, the ability of TNF-α and IL-6 to induce elevated pTyr phosphorylation of VE-cadherin in parallel with elevated permeability. Our observation of significantly reduced pTyr/pThr levels for occludin, claudin-5, and VE-cadherin in response to shear stress is also consistent with recent studies using bovine brain microvascular endothelial cells 1 and in vivo mouse carotid arteries, 34 and has clear parallels with in vivo models of flow-mediated regulation of endothelial BBB pathophysiology. 35 Finally, it can be noted that the functional relevance of the observed phosphorylation changes was underscored by our ability to consistently block (≥ 40%) the permeabilizing action of either cytokine using genistein (a receptor tyrosine kinase inhibitor), as well as the barrier-enhancing action of shear stress using dephostatin (a tyrosine phosphatase inhibitor; data not shown). In conclusion, we believe these results highlight how phosphorylation status may potentially serve as a flexible cellular mechanism mediating between barrier-stabilizing and barrier-disruptive forces within the human brain microvascular endothelium.
By using our HBMvEC model, we next addressed the hypothesis that shear can protect the BBB against cytokine injury through modulation of cellular redox state. As previously indicated, recent work from our laboratory has meticulously shown that either TNF-α or IL-6 can dose dependently induce NADPH oxidase activation, ROS production, and barrier permeabilization in cultured HBMvECs, events that were linked to observed reductions in protein expression of occludin, claudin-5, and VE-cadherin. 12 Moreover, all of the aforementioned cytokine effects in that study were found to be comparably attenuated using either pharmacological antioxidants or siRNA-targeted blockade of NADPH oxidase, 12 clearly linking the ROS-induction events to junctional protein downregulation and elevated permeability. In view of these observations, we therefore decided to investigate if shear stress exhibited protective antioxidant effects in HBMvECs that could potentially ameliorate cytokine-induced oxidant injury. In this respect, we observed that, relative to unsheared cells, HBMvECs preconditioned for 24 hours with shear stress (and continually exposed to shear for the 18-hour duration of cytokine treatment) exhibited significantly lower levels of ROS production and barrier permeabilization in response to treatment with either cytokine. Shear was also found to downregulate NADPH oxidase activation in HBMvECs, as reflected in the reduced expression and coassociation of gp91phox and p47phox subunits. This latter finding is consistent with a study by Duerrschmidt et al, 14 who report reduced gp91phox and p47phox subunit expression, in parallel with reduced superoxide generation, after exposure of HUVECs to laminar shear for 24 hours. A related paper by Hwang et al 13 has also shown reduced expression of gp91phox and Nox4, with accompanying reduction of superoxide generation, after exposure of bovine aortic endothelial cells to just 4 hours of laminar shear stress. Not surprisingly, several papers within the literature also report upregulation of endothelial NADPH oxidase activation under conditions of ‘disturbed’ shear, ranging from shear cessation (simulated ischemia) to oscillatory shear.36,37 In relation to these studies, it is noteworthy that previous investigations concerning the interplay between shear stress and NADPH oxidase appear to focus primarily on arterial models, with no apparent studies on the brain microvascular endothelium. Again, to our knowledge, the current paper is the first to do so. Furthermore, the relevance of our in vitro findings to the in vivo situtation is underscored by the antioxidant and antiinflammatory effects of physical exercise–induced shearing within the cerebrovasculature. 38 In conclusion, our collective findings lead us to propose that laminar shear can protect the brain microvascular endothelium from injurious cytokine effects on interendothelial junctions and barrier integrity by regulating the cellular redox state, in-part through NADPH oxidase inhibition.
In addition to the NADPH oxidase downregulation highlighted in this paper, the net antioxidant effect of shearing almost certainly draws on additional cellular mechanisms. These may include shear-dependent modulation of intracellular bioenergetic pathways to yield reductant equivalents capable of neutralizing harmful ROS, 39 blockade of thioredoxin-interacting protein expression, 9 and induction of antioxidant response element-mediated genes (e.g., heme oxygenase-1 and glutathione-S-transferase) with antiinflammatory potential. 40 Thus, we propose that during homeostatic regulation of the BBB, steady laminar shear may generate a compensatory antioxidant defense within the brain microvascular endothelium, which may in turn suppress both upstream and downstream proinflammatory cytokine signaling pathways that would otherwise lead to reduced expression and disassembly of interendothelial junction proteins that govern paracellular permeability. A future in vivo study using a relevant animal model of BBB pathophysiology will enable a fuller validation of this hypothesis.
In summary, this research paper presents novel findings pertaining to the contrasting effects of biomechanical and humoral circulatory factors on HBMvEC barrier integrity at the paracellular adherens and tight junction protein level. Moreover, it provides findings in support of the hypothesis that the injurious effects of functionally distinct proinflammatory cytokines (TNF-α and IL-6) on HBMvEC barrier integrity can be attenuated in-part through shear-mediated suppression of cellular redox signaling. These findings offer an important insight into the largely undefined interplay between shear and cytokines during homeostatic regulation at the BBB interface.
Footnotes
KDR and PMC conceived and designed the experiments, and prepared the manuscript. KDR, LEC, and AM performed the experiments. KDR, LEC, and PMC performed data analysis. PMC contributed to reagents/materials/analysis tools.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
