Abstract
The ABC-transporter, p-glycoprotein-1 (pgp-1), is expressed on brain endothelium and is reported to be induced by several cytotoxic drugs, which are themselves substrates of pgp-1. Pgp-1 was increased on a human brain endothelial cell line (hCMEC/D3) after treatment with puromycin or verapamil. However, flow cytometry showed that the apparent upregulation caused by puromycin was not because of a global increase in expression levels, but selective cell death of a subpopulation of endothelium expressing the lowest levels of pgp-1. If a cytotoxic substrate of pgp-1 increases pgp-1 expression
Introduction
The ABC-transporter, p-glycoprotein-1 (pgp-1), is an important element of the blood–brain barrier. It is expressed primarily on the lumenal surface of brain endothelium and it actively prevents many toxic molecules and potentially useful drugs from entering the CNS (Löscher and Potschka, 2005). Expression is constitutive on brain endothelium but is generally absent from vascular endothelium that lacks barrier functions and continuous tight junctions (Holloway et al, 2007). Pgp-1 is also reported to be transcriptionally induced by a wide variety of toxic molecules, as well as cytokines, steroids, cellular stressors, heat-shock, and ultraviolet irradiation (Sarkadi et al, 2006; Uchiumi et al, 1993). However, most of these studies were carried out using tumor cells with induced pgp-1, or other cell types that constitutively express it, such as gut epithelium. It is sometimes assumed that transcriptional activators that act on one cell type will act in a similar way on other cell types, but this is often not true.
Many of the inducers of pgp-1 are also substrates for pgp-1, for example, puromycin and verapamil. If such drugs increase pgp-1 on brain endothelium, it potentially creates problems in achieving sustained delivery of the drugs to the CNS, because a drug used for treatment induces the transporter that prevents it from entering the CNS. Moreover, because the substrate specificity of pgp-1 is low, induction of the pgp-1 gene (MDR1) by one drug could affect permeability of other drugs (Begley, 2004).
While studying transcriptional controls of the brain-endothelium phenotype, we aimed to identify tissue-specific elements in the pgp-1 promoter and distinguish them from elements that control induced expression. The study used the human brain endothelial cell line hCMEC/D3 (Weksler et al, 2005) and puromycin, a drug that is both a proposed inducer and substrate of pgp-1, and which has been extensively used to select and purify brain endothelium (Perriere et al, 2005). We compared pgp-1 induction by puromycin with the results produced by verapamil a calcium channel blocker, which is a substrate and functional activator of pgp-1.
Materials and methods
The human brain endothelial cell line, hCMEC/D3, was used at passages 23 to 33 (Weksler et al, 2005). Cells were plated onto collagen-coated 96-well plates for cell surface ELISA or 6-well plates for fluorescence activated cell sorting (FACS) and grown to confluence in EBM2 medium (Cambrex, Wokingham, UK), containing half of the standard growth factors. At this stage the cells were switched into EBM2 without growth factors or antibiotics, but retaining hydrocortisone and sodium ascorbate, for the remainder of the experiment. Cells were rested for 48 h before treatment with puromycin, verapamil, or UV light. Puromycin or verapamil were either present throughout the experiment, with medium changes every third day, or the concentration was increased at each medium change for ramped treatments.
Cell surface ELISA was performed as described earlier (Male et al, 1987), using 70
FACS analysis was performed by collecting cells with trypsin/EDTA, washing twice in PBS and fixing in 2% paraformaldehyde for 30 mins. Washed cells (2 × 105 in 100
Results
To assess the induction of pgp-1, hCMEC/D3 were initially cultured for 3 days in 0 to 1,000 ng/mL puromycin, 0 to 200
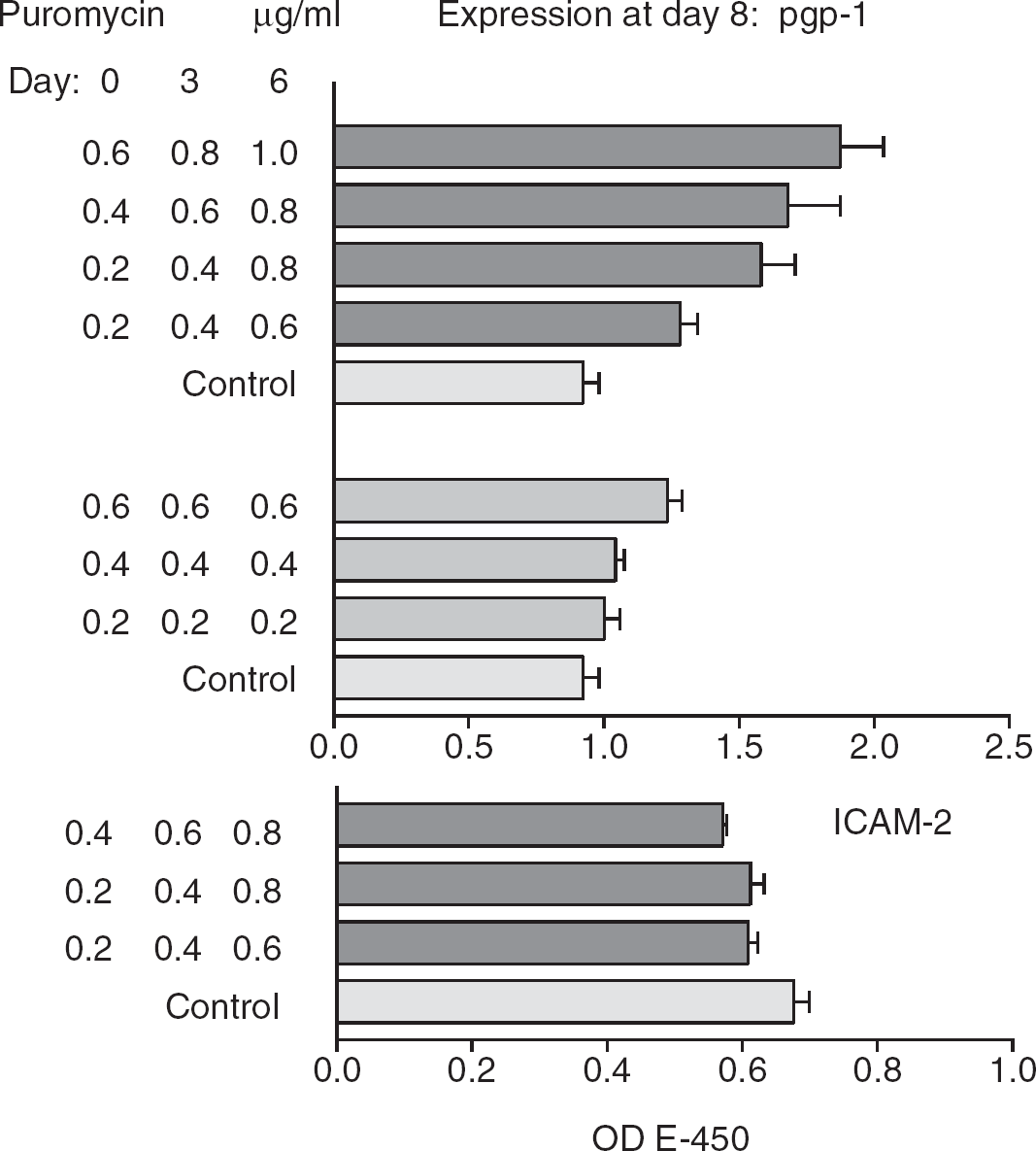
ELISA of pgp-1 and ICAM-2 expression at day 8, after treatment of hCMEC/D3 endothelial cells with the indicated doses of puromycin. Results show mean and s.e.m. (controls
At first sight, the increased expression of pgp-1 suggested an increase in transcription or translation, but the induction was surprisingly slow—8 days. Consequently, pgp-1 expression was examined in more detail by FACS. Figure 2A shows that the range of pgp-1 expression on resting cells was very variable. Moreover, a ramped puromycin treatment over 8 days (0.4 to 1.0
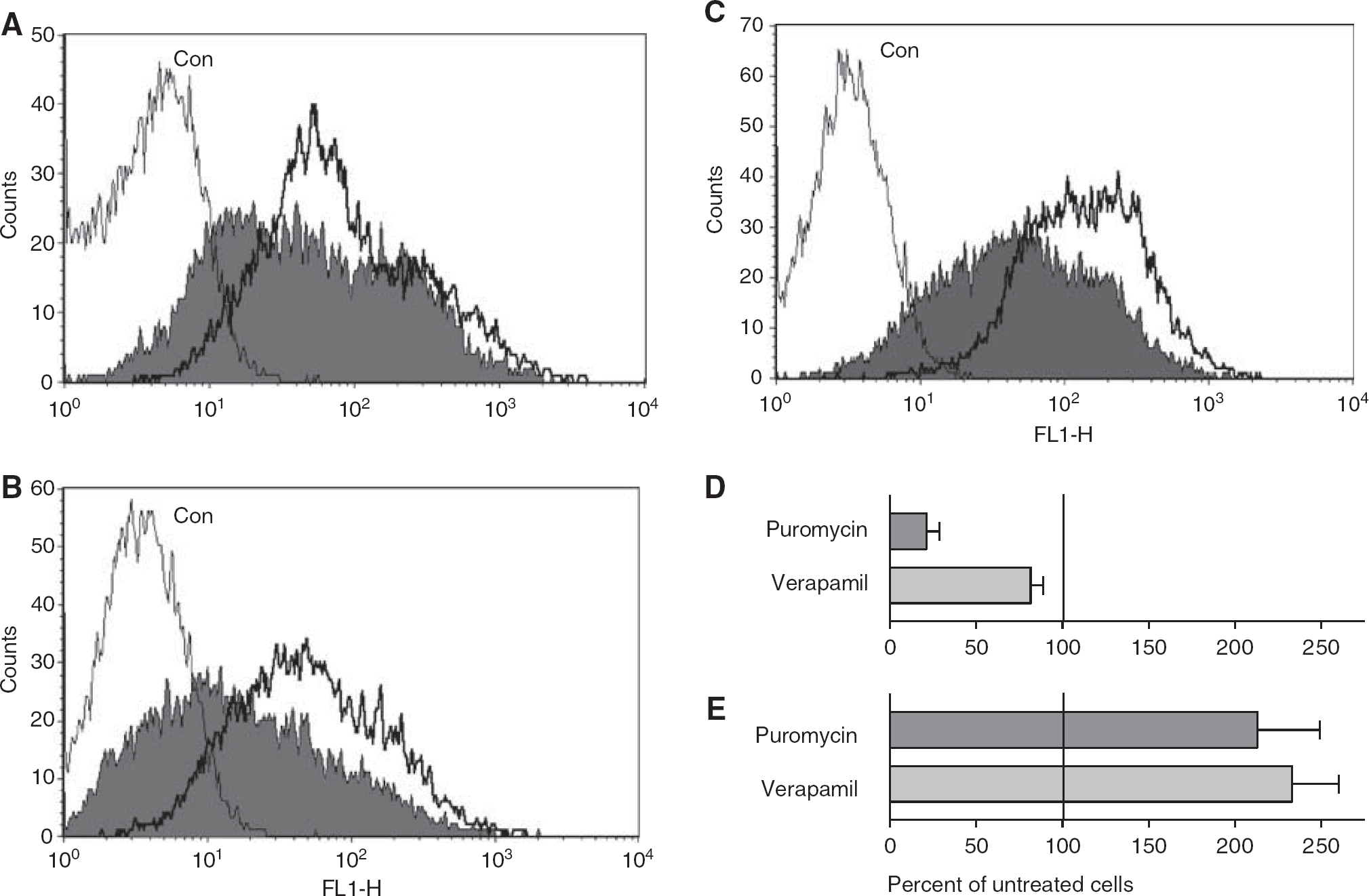
FACS analysis of pgp-1 expression on hCMEC/D3 cells. Untreated cells (filled histograms) were compared with puromycin- or verapamil-treated cells (unfilled histograms). Cells stained with isotype-control antibody (Con) are indicated. (
Further investigation of the cells at day 8 showed that the number of cells recovered from puromycin-treated wells was only 14 to 28% (range) of that on matched, untreated wells, even though the monolayers remained confluent. The puromycin-treated cells were significantly larger when analyzed by flow cytometry (forward scatter 654±23, compared with 472±33 in untreated cells—mean and coefficient of variance,
To determine whether the higher expression of pgp-1 after puromycin treatment was a stable characteristic, puromycin-treated cells were regrown in medium without puromycin and pgp-1 expression was reanalyzed by FACS. Cells treated with ramped puromycin (0.4 to 1.0
Treatment of the cells with 100 to 200
Discussion
Many pgp-1 substrates are thought to induce pgp-1 expression, based on results seen in tumor cells (Scotto, 2003). However, transcriptional controls in cells that constitutively express a protein are generally different from controls on induced expression. This study suggests that brain endothelial cells, which constitutively express high levels of pgp-1, are not further induced by puromycin. Rather, the apparent slow induction of pgp-1
Verapamil was originally developed as a calcium channel blocker to treat cardiac conditions, but was subsequently found to act as a substrate for pgp-1, and has been used as a competitive inhibitor of pgp-1 activity
The transcriptional control of pgp-1 is complex. The MDR1 promoter seems to require activation through transcription factors interacting with a CCAAT-box and a GC-rich box (Sundseth et al, 1997), which are modulated by several other elements as part of the MDR1 enhanceosome (Scotto, 2003). On rodent brain endothelium, pgp-1 can be further induced through the pregnane-X receptor (Bauer et al, 2004) — cell surface expression was induced over 3 to 6 h by dexamethasone or pregnenolone-16
Drug resistance presents problems for drug delivery to the CNS, particularly for tumors and epilepsy (Löscher and Potschka, 2005), and it has been proposed that increased expression of multidrug transporters on the brain endothelium underlies resistance. In support of this view, an increase in pgp-1 expression has been noted on brain capillaries in epileptic subjects (Tishler et al, 1995), and it has been debated whether this is due to the condition itself, or induction by drugs. However, because the increase is local rather than global, it suggests that it is not because of the treatment (Sisodiya et al, 2002). The induction of pgp-1 on brain endothelium by its substrates
Footnotes
The author declares no conflict of interest.
