Abstract
The blood–brain barrier (BBB) has a critical role in central nervous system homeostasis. Intercellular tight junction (TJ) protein complexes of the brain microvasculature limit paracellular diffusion of substances from the blood into the brain. Hypoxia and reoxygenation (HR) is a central component to numerous disease states and pathologic conditions. We have previously shown that HR can influence the permeability of the BBB as well as the critical TJ protein occludin. During HR, free radicals are produced, which may lead to oxidative stress. Using the free radical scavenger tempol (200 mg/kg, intraperitoneal), we show that oxidative stress produced during HR (6% O2 for 1 h, followed by room air for 20 min) mediates an increase in BBB permeability
Introduction
The blood–brain barrier (BBB) is a physical and metabolic barrier comprised of brain microvascular endothelial cells that restrict the passage of substances from the blood to the brain and help maintain brain homeostasis. The BBB expresses a high number of ion channels and transporters, has a low rate of pinocytosis, and forms intercellular tight junction (TJ) protein complexes that limit paracellular permeability (Hawkins and Davis, 2005). It has previously been shown that disruption of the BBB is associated with hypoxia and reoxygenation (HR) (reviewed in Kaur and Ling, 2008). Hypoxia and reoxygenation leads to an increase in BBB permeability (Witt et al, 2003), which may result in neurotoxic substances leaking from the blood into the brain and contributing to vasogenic edema. Cerebral HR is a central component of many disorders or conditions affecting cognition. These include stroke (Kalaria and Ballard, 2001), cardiac arrest (Lim et al, 2004), postoperative cognitive dysfunction (Caza et al, 2008), acute respiratory distress syndrome (Hopkins et al, 2006), obstructive sleep apnea (El-Ad and Lavie, 2005), high-altitude cerebral edema, and acute mountain sickness (Hackett, 1999).
Much of the cellular damage caused by hypoxic insult is thought to occur during the subsequent reoxygenation phase. Hypoxia and reoxygenation is associated with an increased production of reactive oxygen species (ROS) (reviewed in Wong and Crack, 2008). These ROS contribute to brain injury by reacting with proteins, lipids, and nucleic acids, as well as by activating a number of redox-sensitive signaling pathways. A number of different laboratories have shown that production of ROS can affect BBB permeability by a variety of mechanisms, including modulation of TJ proteins (reviewed in Pun et al, 2009).
Previously, we have shown that HR causes an increase in BBB paracellular permeability
To examine the role that oxidative stress has in disrupting the functional integrity of the BBB, we administered the stable, membrane-permeable, water-soluble nitroxide, tempol to rats before the induction of HR. Tempol shows superoxide dismutase-like activity toward the superoxide anion (O2.−), as well as reactivity with the hydroxyl radical (OH.) (Saito et al, 2003), nitrogen dioxide (NO2.), and the carbonate radical (CO3.−) (Augusto et al, 2008). Tempol readily crosses the BBB (Zhelev et al, 2009), and has previously been shown to provide neuroprotection as a free radical scavenger in several models of brain injury and ischemia (Kwon et al, 2003; Deng-Bryant et al, 2008; Rak et al, 2000; Cuzzocrea et al, 2000).
In this study, we looked at the effects of ROS produced during HR in a model that has previously been shown to induce alterations in BBB permeability and the structural integrity of the TJ protein occludin. We use the
Materials and methods
Radioisotopes, Reagents, and Chemicals
The [14C]-sucrose was purchased from American Radio-labeled Chemicals (St Louis, MO, USA). Tempol was purchased from MP Biomedicals (Solon, OH, USA). Ts-2 tissue solubilizer was purchased from Research Products International (Mt. Prospect, IL, USA). Optiphase Supermix scintillation cocktail was purchased from Perkin Elmer (Shelton, CT, USA). Ethylenediaminetetraacetic acid (EDTA)-free complete protease inhibitors were purchased from Roche (Indianapolis, IN, USA). OptiPrep was purchased from Accurate Chemical (Westbury, NY, USA). The Coomassie Plus Better Bradford Assay Kit was purchased from Thermo Scientific (Rockford, IL, USA). Rabbit anti-occludin directed toward the C-terminus was purchased from Zymed (San Francisco, CA, USA) and mouse anti-platelet-endothelial cell adhesion molecule 1 was purchased from AbD Serotec (Raleigh, NC, USA). Mouse anti-heat shock protein 70 (anti-HSP-70) was purchased from R&D Systems (Minneapolis, MN, USA). Secondary antibodies for western blotting and immunofluorescence were purchased from Amersham (Pittsburgh, PA, USA) and Invitrogen (Carlsbad, CA, USA), respectively. Western Lightning enhanced chemiluminescence reagent was purchased from Perkin Elmer (Shelton, CT, USA). TO-PRO-3 and ProLong Gold were purchased from Invitrogen. The XT sample buffer, XT reducing agent and criterion gels were purchased from Bio-Rad (Hercules, CA, USA). All other antibodies or reagents, unless noted, were purchased from Sigma-Aldrich (St Louis, MO, USA).
Hypoxic Treatment
All experimental protocols were approved by the Institutional Animal Care and Use Committee at the University of Arizona. Adult female Sprague–Dawley rats (Harlan, Indianapolis, IN, USA) weighing 230 to 280 g were housed under standard 12:12 h light–dark conditions, and received food and water
In Situ Brain Perfusion
The
At the end of perfusion, the rat was killed and the brain was removed. The meninges and choroid plexuses were removed and the brain was divided and placed into preweighed vials. Tissue solubilizer (1 mL) was added to each vial and allowed to dissolve the brain sample for 48 h. After solubilization, 100 μL of 30% glacial acetic acid was added to each sample to quench chemiluminescence. Optiphase Supermix scintillation cocktail (2.5 mL) was added to each sample. Triplicate samples of 100 μL aliquots of the perfusion medium were treated in the same manner as the perfused brain samples. All samples were then measured for disintegrations per minute (dpm) (1450 LSC and Luminescence Counter; Perkin Elmer, Waltham, MA, USA). The ratio of the concentration of 14C-sucrose in tissue (Cbrain; in dpm/g) was compared with perfusate (Cperfusate; in dpm/mL) and expressed as a percent ratio Rbrain = (Cbrain/Cperfusate) × 100%. We have previously used capillary depletion analysis in this model to show that the increase in 14C-sucrose after HR was not because of increased trapping within the microvascular endothelial cells (Witt et al, 2003).
Microvessel Enrichment and Fractionation
After hypoxic or control treatment, rats were anesthetized with a 1.0 mL/kg i.p. injection of an anesthetic cocktail consisting of ketamine (78.3 mg/mL), xylazine (3.1 mg/mL), and acepromazine (0.6 mg/mL). They were then killed after 20 mins of reoxygenation and the brains were extracted. The cerebellum, meninges, and choroid plexuses were removed and the cerebral hemispheres were homogenized in 4mL of microvessel isolation buffer A (103 mmol/L NaCl, 4.7 mmol/L KCl, 2.5 mmol/L CaCl2, 1.2 mmol/L KH2PO4, 1.2 mmol/L MgSO4, 15 mmol/L 4-(2-hydroxyethyl) 1-piperazineethanesulfonic acid (HEPES), 2.5 mmol/L NaHCO3, 10 mmol/L D-glucose, and 1 mmol/L sodium pyruvate (pH 7.4)). Ice-cold buffer A (4 mL) with 26% dextran was added to the homogenates, which were vortexed and centrifuged at 5,800
Western Blot Analysis
Equal volume aliquots of each gradient fraction were mixed with equal volumes of 2 × perfluorooctanoic acid (PFO) extraction buffer (100 mmol/L Tris, 20% glycerol, and 4% PFO (pH 8.0)) and allowed to incubate at 25°C for 30 min. Samples were then centrifuged at 11,000
Immunofluorescence
All slides from control and treated rats were collected and processed in parallel. Primary antibody was omitted on some slides as a negative control. Fluorescent immunostaining for occludin was performed on microvessel preparations enriched as above. Briefly, microvessels were heat-fixed on glass slides at 95°C for 10 min, followed by fixation in ice-cold acetone for 10 mins. The slides were blocked in 2% goat serum and 1% bovine serum albumin before incubation in rabbit anti-occludin primary antibody (1:250) and the endothelial-specific marker mouse anti-platelet-endothelial cell adhesion molecule 1 (1:1,000) overnight in the cold; slides were incubated with the appropriate Alexafluor-conjugated secondary antibodies.
For HSP-70 immunostaining, microvessels were placed on a glass slide and allowed to air dry before being fixed in 3.7% formaldehyde for 10 min. The microvessels were permeabilized in 0.1% Triton X-100, blocked as above, and incubated overnight in the cold in mouse anti-HSP 70 (1:400) and in the endothelial-specific marker rabbit anti-von Willebrand factor (1:5,000). The slides were then incubated with the appropriate Alexafluor-conjugated secondary antibodies.
For hypoxia-inducible factor 1α (Hif-1α) immunostaining, microvessels were placed on a glass slide and allowed to air dry before being fixed in ice-cold acetone for 10 mins. The slides were blocked as above and incubated in mouse anti-Hif-1α (1:200) along with rabbit anti-von Willebrand factor (1:5,000) overnight in the cold. Slides were then incubated with the appropriate Alexafluor-conjugated secondary antibodies. The nucleus of the microvessels was then stained with the nuclear-specific dye TO-PRO-3 (10 μmol/L) for 10 min. All slides were mounted in ProLong Gold anti-fade reagent before applying the coverslip.
Confocal Microscopy
All slides were imaged on a Zeiss LSM 510 meta-NLO confocal microscope (Zeiss, Oberkochen, Germany), with filters appropriately set to avoid bleed-through. Only microvessels positive for the endothelial-specific markers platelet-endothelial cell adhesion molecule 1 or von Willebrand factor were used for analysis. Semiquantitative analysis of the mean fluorescent intensity of HSP-70 was performed according to previously published methods (Hawkins et al, 2004). Microvessels were randomly chosen from rats in each treatment group and the mean fluorescence intensity was analyzed using Zeiss LSM Image Browser software (Zeiss), with the data expressed as percentage of control. To investigate Hif-1α translocation to the nucleus, the Mander's colocalization coefficient for Hif-1α and TO-PRO-3 was determined using the Zeiss LSM Image Browser software in randomly chosen microvessels in each treatment group.
Statistical Analysis
All data analyses were performed using SigmaPlot software (Systat Software, Inc., San Jose, CA, USA). To determine statistical significance between treatment groups, data were analyzed using one-way analysis of variance (ANOVA), followed by Bonferroni's
Results
Hif-1α Expression and Translocation to the Nucleus
Hypoxia-inducible factor 1α is an oxygen-sensitive protein that is continuously synthesized and degraded under normoxic (Nx) conditions. Under hypoxic conditions, degradation of Hif-1α decreases and translocation to the nucleus occurs, wherein Hif-1α heterodimerizes with Hif-1β and binds to hypoxia-responsive elements of target genes. Our confocal imaging data (Figure 1) on our enriched microvessel preparations show that HR causes an increase in Hif-1α colocalization with the nuclear dye TO-PRO-3, suggesting that Hif-1α translocation to the nucleus is because of hypoxia. We measured the ratio of nuclear Hif-1α to total Hif-1α using the Mander's colocalization coefficient (Table 1). A value of 0 indicates no colocalization of Hif-1 with the nucleus, whereas a value of 1 indicates that all pixels colocalize. We obtained a Mander's colocalization coefficient of 0.142 ± 0.019 for saline-treated Nx (Nx + S) and 0.119 ± 0.015 for tempol-treated Nx (Nx + T) rats. Saline-treated HR (HR + S) rats showed a statistically significant increase in Hif-1α nuclear colocalization with a Mander's colocalization coefficient of 0.211 ± 0.22 than did Nx rats. Tempol-treated HR (HR + T) rats had a Mander's colocalization coefficient of 0.154 ± 0.012. Images are representative of six rats per treatment group (
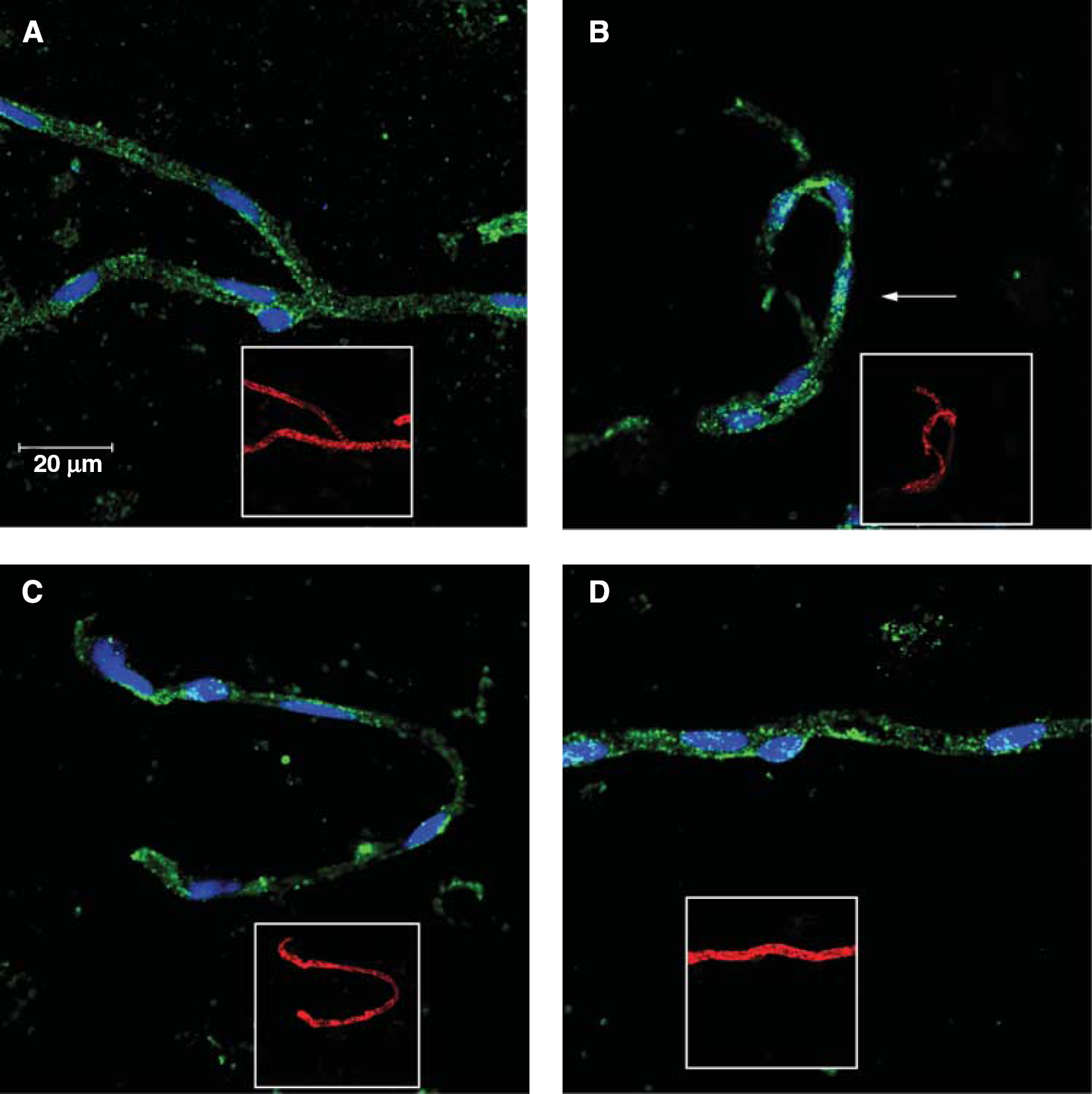
Hypoxia and reoxygenation (HR) induces hypoxia-inducible factor 1α (Hif-1α) translocation to the nucleus. Immunofluorescence of Hif-1α (green) and the nuclear dye TO-PRO-3 (blue) in brain microvessels of normoxic (Nx) plus saline (
Mander's colocalization coefficients for Hif-1α and TO-PRO-3
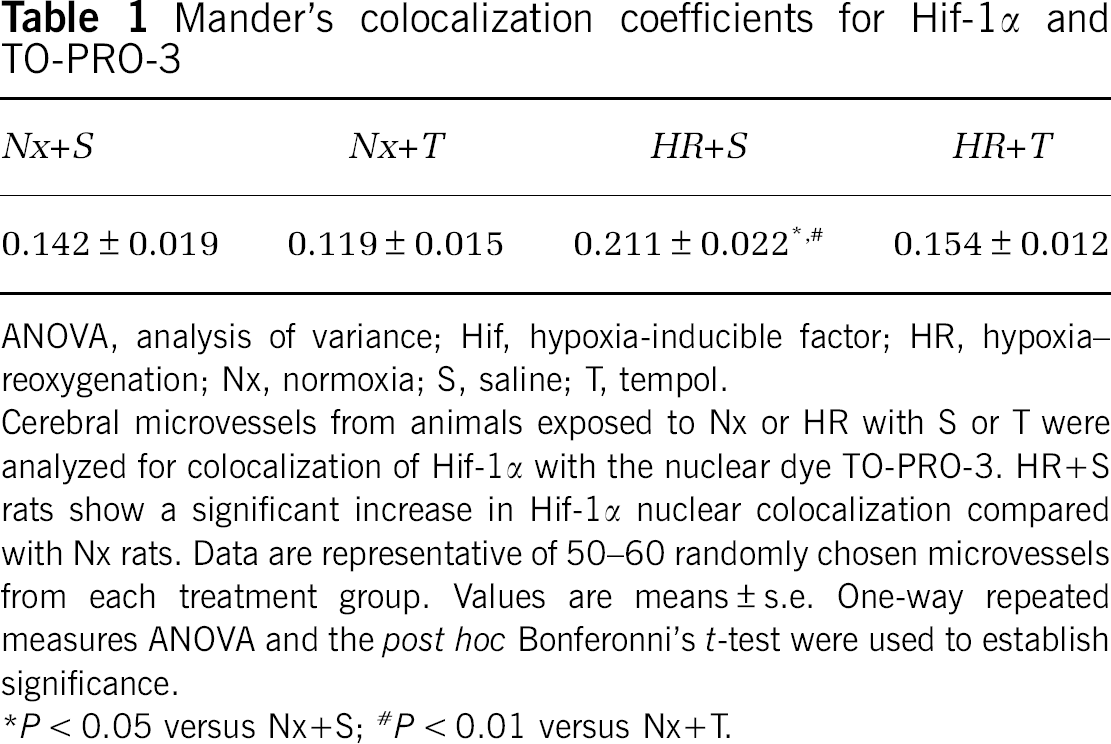
ANOVA, analysis of variance; Hif, hypoxia-inducible factor; HR, hypoxia–reoxygenation; Nx, normoxia; S, saline; T, tempol.
Cerebral microvessels from animals exposed to Nx or HR with S or T were analyzed for colocalization of Hif-1α with the nuclear dye TO-PRO-3. HR+S rats show a significant increase in Hif-1α nuclear colocalization compared with Nx rats. Data are representative of 50–60 randomly chosen microvessels from each treatment group. Values are means ± s.e. One-way repeated measures ANOVA and the
P < 0.01 versus Nx+T.
HSP-70 Expression
Upregulation of HSP-70 is associated with oxidative stress (Ronaldson and Bendayan, 2008; Amadio et al, 2008). We looked at levels of HSP-70 in the enriched microvessel preparations of all treatment groups using confocal microscopy (Figure 2A). Images are representative of six rats per treatment group (
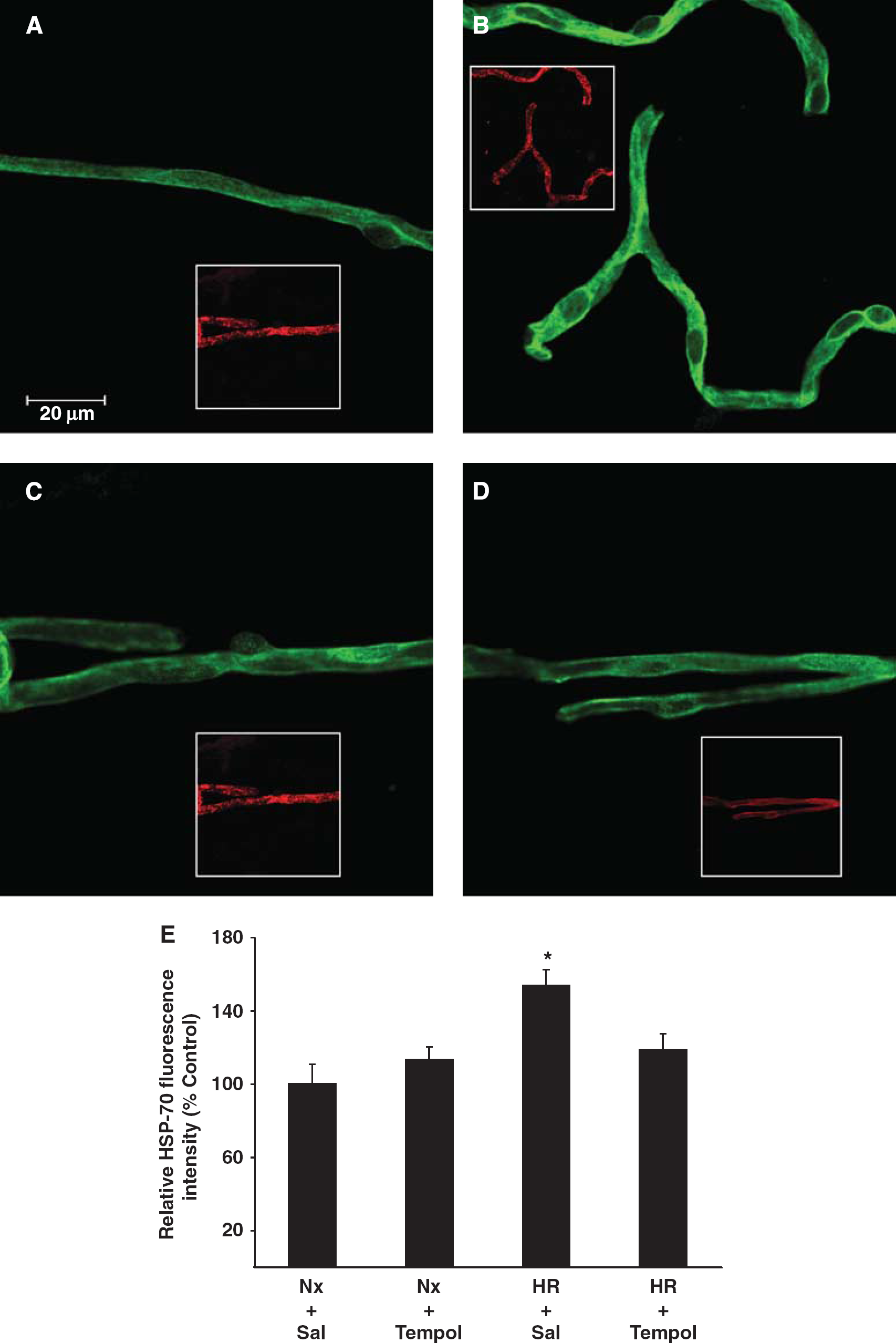
Oxidative stress induces expression of heat shock protein (HSP-70). Immunofluorescence of HSP-70 (green) in brain microvessels of normoxic (Nx) plus saline (Nx + S) (
In Situ Brain Perfusion
The role of oxidative stress in altering BBB permeability during HR was investigated using the
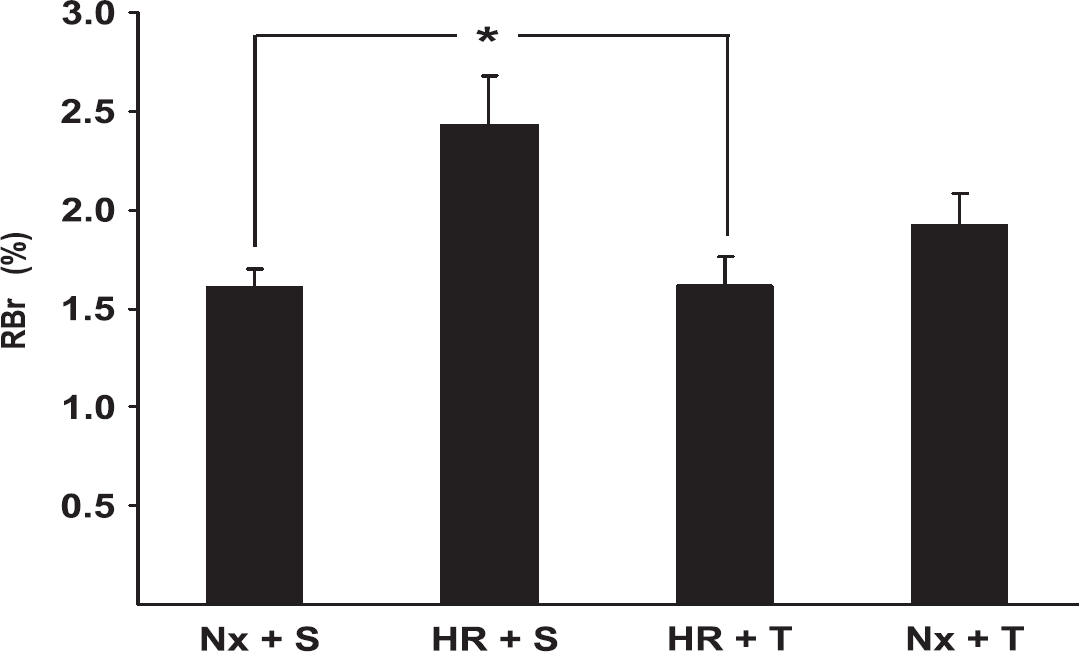
Effects of hypoxia and reoxygenation (HR) and tempol treatment on BBB permeability to 14C-sucrose. After treatment, normoxia (Nx) plus saline (S) (Nx + S), Nx plus tempol (T) (Nx + T), HR + S, and HR + T rats were anesthetized and subjected to
Occludin Microvascular Distribution
To examine the effects of HR and oxidative stress on the cellular localization of the TJ protein occludin, enriched microvessel preparations were placed on glass slides and stained for occludin. Slides were then examined under a laser scanning confocal microscope. Images are representative of five rats per treatment group (
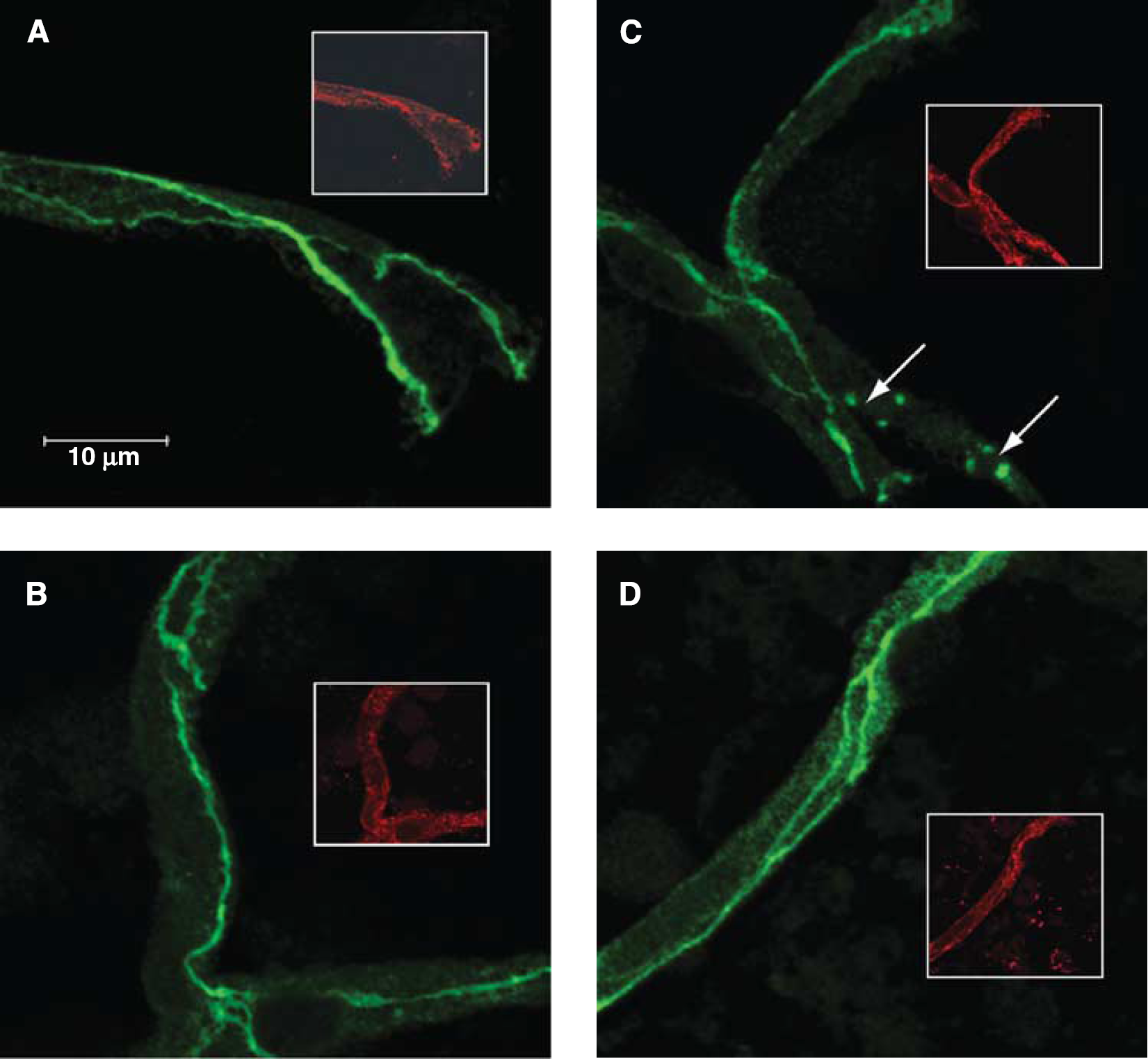
Oxidative stress induces changes in occludin localization. Immunofluorescence of occludin (green) and the endothelial-specific marker platelet-endothelial cell adhesion molecule 1 (PECAM-1) (red inset) in brain microvessels of normoxic (Nx) plus saline (
Percentage of cerebral microvessels exhibiting punctate staining of occludin

ANOVA, analysis of variance; HR, hypoxia–reoxygenation; Nx, normoxia; S, saline; T, tempol.
Microvessels were analyzed for punctate staining in Nx+S, Nx+T, HR+S, and HR+T. Values are means ± s.e.;
Density Gradient Fractionation
Density gradient centrifugation and fractionation of intact microvessels were performed to examine the distribution of occludin within plasma membrane microdomains. The protein concentration and refractive indices from intact microvessels subjected to density gradient fraction are presented as averages of three separate experiments, each containing three rats per treatment group (Figure 5A). Our data show that the protein concentration and refractive index from each fraction is similar for each treatment group. This shows that our analysis was performed on membrane microdomains that are similar across all treatment groups. We have previously shown that the TJs exist primarily in fractions 7 and 8 (McCaffrey et al, 2007). Using SDS-PAGE, we analyzed western blots of fractions 7 and 8 under reducing and nonreducing conditions to investigate the band patterns of PFO-soluble occludin oligomers (Figure 5B). Perfluorooctanoic acid has previously been shown to preserve the oligomeric structure of membrane proteins by stabilizing internal noncovalent interactions between component protein subunits (Ramjeesingh et al, 1999). During HR, a significant increase in the optical density of occludin oligomers under nonreducing conditions compared with Nx controls (4.9-fold in fraction 7 and 11.0-fold in fraction 8) is observed (Figure 5C). Occludin oligomers exhibit a broad, intense staining pattern resulting from HR. This suggests that extensive conformational changes in occludin occur during HR. Administration of tempol before HR served to significantly decrease the extent of oligomeric disassembly and to attenuate the increase in optical density of nonreduced occludin oligomeric isoforms compared with Nx + S rats (2.1-fold in fraction 7 and 4.1-fold in fraction 8). Comparison of occludin banding patterns on nonreducing with reducing blots reveals that the reducing agent TCEP increases the detection of occludin oligomeric isoforms in Nx rats, but not in HR rats. This suggests that HR causes conformational changes of occludin oligomeric assemblies, exposing more of the structure to the antibody.
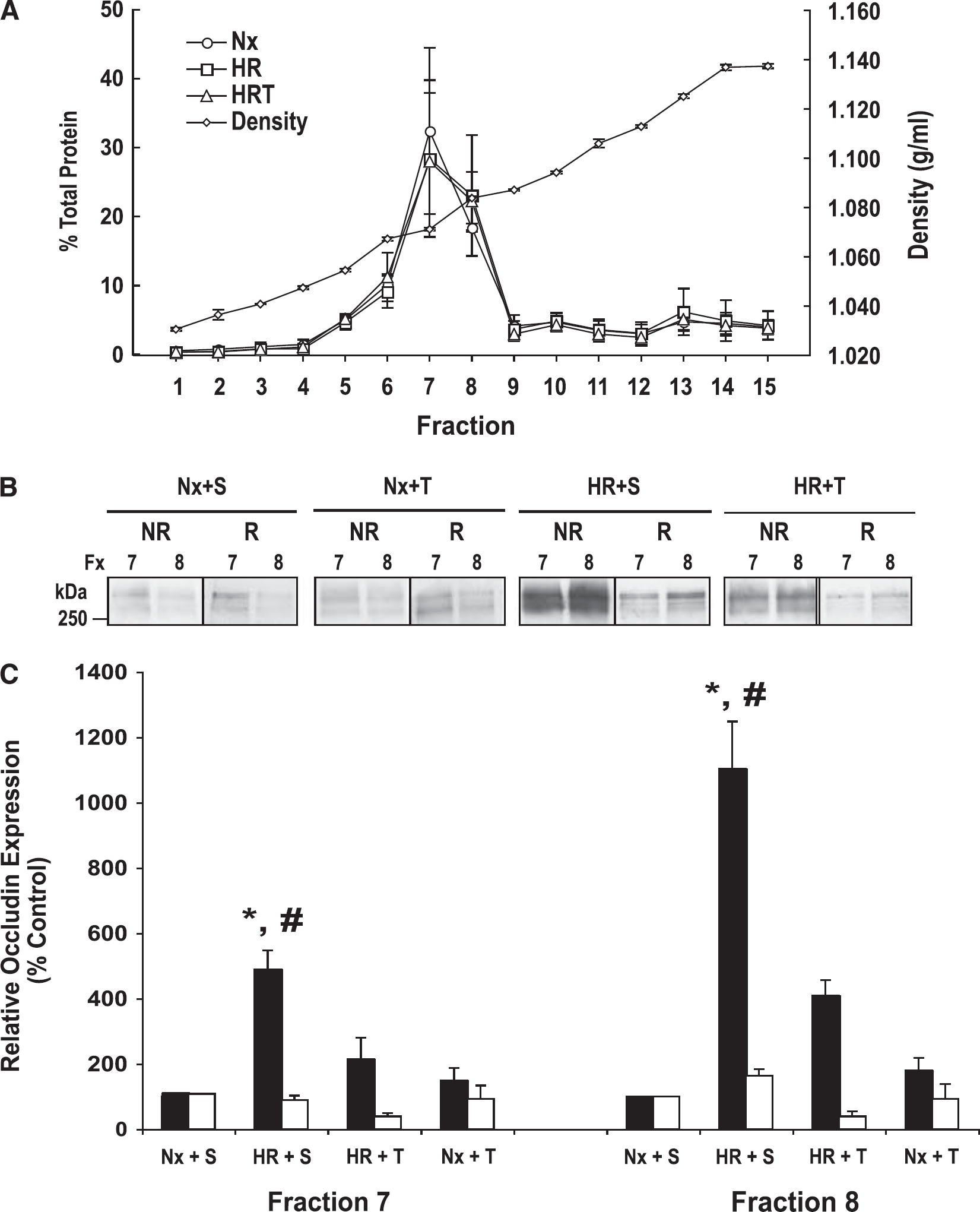
Density gradient fractionation of cerebral microvessels. (
Discussion
The BBB has a critical role in maintaining central nervous system (CNS) homeostasis. Inability of the BBB to perform its normal physiologic functions is thought to contribute to numerous disease states affecting the CNS, many of which have an HR component (Hawkins and Davis, 2005; Zlokovic, 2008). Cerebral HR occurs during stroke and microvascular infarcts, vascular dementia, cardiac arrest, postoperative cognitive dysfunction, acute respiratory distress syndrome, obstructive sleep apnea, and high-altitude cerebral edema.
We have previously shown that HR causes an increase in paracellular permeability at the BBB (Witt et al, 2003; Witt et al, 2008). We have also shown changes in occludin protein localization and oligomeric assemblies that are associated with HR (McCaffrey et al, 2009). Oligomers of occludin are key components of the TJ protein complexes that are critical for restricting parcellular diffusion of solutes from the blood into the brain. Little is known about the mechanisms that lead to changes in occludin oligomeric assembly at the BBB after HR. Hypoxia and reoxygenation is associated with the production of ROS, which can lead to oxidative stress and BBB disruption (Pun et al, 2009). Recent evidence indicates that the oligomerization of occludin is redox sensitive (Walter et al, 2009). These observations led us to investigate the influence of oxidative stress on BBB permeability and occludin structural integrity during HR. To examine this possibility, we used the free radical scavenger tempol to pharmacologically inhibit production of ROS.
To confirm that the brains of rats exposed to our HR treatment are indeed hypoxic, we stained isolated microvessels for Hif-1α. Hypoxia-inducible factor-1α is a transcription factor that is constitutively degraded under Nx conditions. Hypoxia causes Hif-1α to stabilize, which allows it to translocate to the nucleus, dimerize with Hif-1β, and regulate gene transcription. Our confocal microscopy data show increased colocalization of Hif-1α with the nuclear dye TO-PRO-3 in HR-treated rats, which indicates that Hif-1α is stabilized and does translocate to the nucleus in response to HR. Hypoxia and reoxygenation does not cause a significant increase in rats pretreated with tempol. This may be because of an increase in ROS production during HR. Reactive oxygen species have previously been shown to induce Hif-1α nuclear translocation independent of hypoxia (Haddad and Land, 2001). Because tempol acts as a ROS scavenger, our data suggest that the translocation of Hif-1α to the nucleus was attenuated because of a decrease in ROS production in HR + T rats.
As a marker of cellular stress, we also stained microvessels for HSP-70. Heat shock protein 70 has previously been shown to be upregulated because of stressors such as hypoxia and/or oxidative stress. Our data show prominent HSP-70 immunoreactivity in the brain microvasculature during HR; however, pretreatment with tempol attenuated the HR-associated increase in HSP-70 expression. This suggests a reduction in cellular stress in the brains of rats administered with tempol.
Using the
Paracellular permeability at the BBB is regulated, in part, by the TJs. Our data showing that tempol prevents the increase in BBB permeability to 14C-sucrose after HR treatment suggest that oxidative stress may also affect the molecular structure of the TJs. Previous work by our laboratory has indicated that the TJ protein occludin is affected by HR (McCaffrey et al, 2009), and its ability to oligomerize is redox sensitive (Walter et al, 2009). These observations led us to investigate the effects of oxidative stress on occludin at both the cellular and molecular level using confocal microscopy and density gradient subcellular fractionation followed by western blot analysis.
Confocal microscopy of isolated cerebral microvessels immunostained for occludin revealed continuous distribution of occludin along the margins of the TJ. Hypoxia and reoxygenation induced intracellular punctate staining of occludin in regions of the microvessel that were not associated with TJs. In contrast, microvessels from HR + T rats show staining of occludin at the TJ in a pattern similar to Nx + S rats, suggesting that tempol may prevent HR-associated changes in occludin localization. Taken together, our data imply that oxidative stress may induce changes in the cellular localization of occludin at the BBB.
We have previously used a detergent-free density gradient centrifugation method to examine the distribution of occludin in plasma membrane microdomains of cerebral microvessels. These studies have revealed valuable insight into the structure of occludin at the BBB. Density gradient fractionation of intact microvessels has shown that occludin exists in the TJ-associated lipid raft regions corresponding to fractions 7 and 8, in which it colocalizes with the lipid raft marker caveolin-1 and the TJ marker Rab-13 (McCaffrey et al, 2007). We have also used PFO (which stabilizes the quaternary structure of protein complexes) to solubilize occludin oligomeric assemblies (Ramjeesingh et al, 1999). Extracts of PFO-soluble occludin subjected to nonreducing PFO-PAGE have revealed that in TJ-associated fractions 7 and 8, occludin exists almost exclusively as high MW (> 250 kDa) oligomers (McCaffrey et al, 2009). It is only when occludin is solubilized in SDS and/or subjected to reducing conditions that lower MW isoforms are detected. These data strongly suggest that occludin exists as high MW oligomeric assemblies at the TJs of the BBB
Using a C-terminal antibody to occludin, we subjected equal amounts of protein in fractions 7 and 8 to SDS-PAGE under nonreducing and reducing conditions. Our data show very faint banding patterns of occludin oligomeric assemblies located primarily in fraction 7 of Nx rats. Under reducing conditions with the hydrophilic reducing agent TCEP, these bands become much more intense. This suggests that under nonreducing conditions, the antigen is less accessible to antibody, presumably because it is being masked. Reducing conditions cleave these disulfide bonds and allow the antibody greater access to the antigen. Remarkably, oligomeric assemblies of occludin can still be detected after occludin has been subjected to SDS-PAGE under hydrophilic reducing conditions. This suggests an inner core of hydrophobic disulfide bonds holding the oligomeric assemblies together. In contrast to what was observed in Nx rats, HR rats show a prominent band of occludin oligomers under nonreducing conditions. These data suggest that the antibody binds to nonreduced occludin more readily in rats subjected to HR. This indicates that HR may induce conformational changes in occludin oligomers, possibly through the cleavage of disulfide bonds. In addition, occludin redistributes from fraction 7 to the higher density fraction 8 in the HR rats. Taken together, these data suggest that HR affects both the localization and the structural integrity of occludin oligomeric assemblies. Tempol-treated HR rats show significantly less dense bands of occludin oligomers under nonreducing conditions compared with HR + S rats. This suggests that oxidative stress is involved in the conformational changes of occludin oligomeric assemblies, and free radical scavengers such as tempol can preserve the structural integrity of occludin at the BBB during an HR insult.
In summary, our data suggest that ROS produced during HR may cause disruption of the BBB, leading to an increase in permeability to 14C-sucrose. In addition, ROS may cause changes in localization and structure of the TJ protein occludin. Both the structure and the localization of occludin are critically important to its function in the maintenance of the BBB's ability to restrict permeability to solutes in the systemic circulation. Changes in occludin localization and structure during HR can be pharmacologically inhibited by pretreating rats with the stable, BBB-permeable molecule tempol. Hypoxia and reoxygenation and/or oxidative stress are important components of several disease states (i.e., cardiac arrest, stroke, high-altitude cerebral edema) that exhibit a breakdown in the BBB and subsequent neurotoxicity and cognitive impairment. It is possible that ROS produced during the HR phase of these pathologic conditions induces changes in occludin structure and localization, leading to a breakdown in the ability of the BBB to protect the CNS. Treatment of these conditions with antioxidant therapy may preserve the functional integrity of the BBB, which may ultimately decrease the amount of CNS damage associated with several pathologic conditions.
Footnotes
Acknowledgements
The authors thank Dr Patrick Ronaldson for his helpful advice on this manuscript.
The authors declare no conflict of interest.
