Abstract
Protein modifications cooperatively act to protect the proteome from cellular stress. Focal cerebral ischemia increases protein ubiquitination, resulting in formation of ubiquitin-rich aggregates. A concurrent elevation in small ubiquitin-related modifier (SUMO)-conjugated proteins has also been reported, but a potential connection to ubiquitin remains unexplored. Here we show that SUMO2/3 conjugates are present in postischemic ubiquitin-rich aggregates, physically associated with ubiquitin. The coaggregation of SUMO2/3 and ubiquitin is induced rapidly after ischemia, depends on reperfusion, and is also observed in the absence of ischemic damage. The association between SUMO and ubiquitin suggests overlapping functional roles after ischemia/reperfusion.
Introduction
Ischemic stroke caused by occlusion of major cerebral arteries produces rapid neuronal death in the center of the ischemic territory, while cell death is delayed in peripheral regions, the so called ischemic penumbra. 1 The slow evolution of the penumbra toward irreversible damage renders the tissue potentially salvageable, and elucidating the factors that influence the survival of penumbral tissue is of great importance for the development of new stroke therapies. One such factor might be the modification of proteins with ubiquitin and small ubiquitin-related modifier (SUMO), which is dramatically increased in the penumbral area after transient focal cerebral ischemia and not in the ischemic core.2, 3 Ubiquitin and SUMO attachment to proteins has the potential to influence protein stability, interaction and localization, and may therefore represent an effective way to rapidly adapt the cellular proteome to the changing, stressful environment after stroke. Indeed, elevated SUMO conjugation has been associated with neuroprotection,4, 5, 6 while the role of increased ubiquitin accumulation in stroke outcome has not been clarified.
Ubiquitin and SUMO were long thought to have separate targets and functions, but increasing evidence suggests that a crosstalk and interdependence exists between these two modifications.7, 8 Along those lines, the temporal profile and conjugation pattern of SUMO and ubiquitin after focal ischemia were found to be strikingly similar,2, 3 which warrants further examination of a potential connection between the two modifications in this setting. Ubiquitin is enriched in protein aggregates after focal ischemia,2, 9 however, it remains unclear if also SUMO is present within such inclusions. Therefore, the aim of the current study was to investigate whether SUMO is associated with postischemic ubiquitin aggregates in a mouse model of middle cerebral artery occlusion.
Materials and Methods
Animals
Experiments were performed in 6- to 8-week-old male C57Bl/6J mice (mean weight 22.8 g; Jackson Laboratory, Bar Harbor, ME, USA). All experimental procedures were approved by the IACUC (Institutional Animal Care and Use Committee) of Weill Cornell Medical College and were performed according to the IACUC, NIH, and ARRIVE guidelines (http://www.nc3rs.org/ARRIVE).
Middle Cerebral Artery Occlusion
Middle cerebral artery occlusion was induced by the intraluminal suture method as previously reported.
2
Briefly, mice were anesthetized with isoflurane (1.5% to 2%) and a 6-0 surgical nylon monofilament was introduced into the right internal carotid artery through the external carotid artery to occlude the origin of the middle cerebral artery. For transient ischemia, the filament was left in place for the indicated time points and then withdrawn. The filament was not retracted in mice subjected to permanent ischemia. Cerebral blood flow was measured in the ischemic hemisphere using transcranial laser-Doppler flowmetry (Periflux System 5010, Perimed, PA, USA). Only animals with 85% reduction in cerebral blood flow during middle cerebral artery occlusion and 80% cerebral blood flow recovery after 10 minutes of reperfusion were included in the study. In all mice, rectal temperature was kept at 37.0°C±0.5°C during surgery and in the recovery period until the animals regained consciousness. Out of a total of 76 animals used in this study 10 had to be excluded because they either did not meet cerebral blood flow criteria (
Isolation of Ubiquitin/SUMO-Containing Soluble and Aggregate Fractions and Detection of Proteins by Western Blotting
Soluble and aggregate fractions were obtained from the ipsilateral neocortex. The cortical sampling and isolation procedure is described in detail in Hochrainer
Equal amounts of proteins as determined by protein concentration measurement of the soluble fraction were loaded and separated by 4% to 20% (Life Technologies, Grand Island, NY, USA) or 7% Tris-glycine-sodium dodecyl sulfate (SDS) gel electrophoresis. After transfer of proteins to polyvinylidene difluoride membranes (EMD Millipore, Billerica, MA, USA), total ubiquitin, lysine 48-linked ubiquitin, lysine 63-linked ubiquitin, SUMO1, and SUMO2/3 were detected by western blotting with anti-ubiquitin (clone ubi-1, Life Technologies), lysine 48-specific anti-ubiquitin (clone Apu2; EMD Millipore), lysine 63-specific anti-ubiquitin (clone D7A11, Cell Signaling Technology, Danvers, MA, USA), anti-SUMO1, and anti-SUMO2/3 (both Cell Signaling Technology) antibodies, respectively. Protein bands from free ubiquitin/SUMO as well as protein band smear in the higher molecular weight range representing conjugated ubiquitin/SUMO (indicated by brackets in the figures) were quantified with Kodak 1D V3.6 image software (Kodak, Rochester, NY, USA). Densitometric data were obtained in the linear range of blot exposure. The background optical density of each blot was determined in an empty lane and the obtained value was subtracted from each specific signal. Ubiquitin and SUMO levels in experimental groups were expressed relative to levels in sham animals, which were set to 1. Free ubiquitin and SUMO were normalized to glyceraldehyde 3-phosphate dehydrogenase levels. Data are expressed as mean±s.d.
Coimmunoprecipitation of Ubiquitin and SUMO from Aggregate Fractions
Aggregate fractions were solubilized as specified previously in Shimada
Statistical Analysis
Sample size was determined based on previous published work on ubiquitination in ischemia by our laboratory.
2
Animals were randomly assigned to sham and stroke groups and analysis was performed in a blinded manner. Two group comparisons were statistically analyzed by the two-tailed Student's
Results
Transient Middle Cerebral Artery Occlusion Leads to Coaggregation of Ubiquitin and SUMO2/3 in the Ipsilateral Neocortex
To address whether SUMO conjugates found after ischemia are of soluble or, like ubiquitin conjugates, of insoluble nature we compared their levels in soluble cytosolic and Triton X-100 insoluble aggregate fractions from mouse neocortices after transient focal ischemia. SUMO2/3 conjugates were, like ubiquitin, significantly enriched in the aggregate fraction as opposed to its soluble counterpart (Figure 1A). At the same time, conjugated as well as free ubiquitin was depleted from the soluble fraction, while SUMO2/3 levels remained constant (Figure 1A). No significant changes were observed in either fraction for free and conjugated SUMO1 (Figure 1A). To evaluate the nature of ubiquitin conjugates after focal cerebral ischemia we probed blots with lysine 48- and lysine 63-specific antibodies. Interestingly, detection with both antibodies resulted in a strong signal (Figure 1B), indicating that aggregates contain ubiquitin chains linked via both lysine 48 and 63.
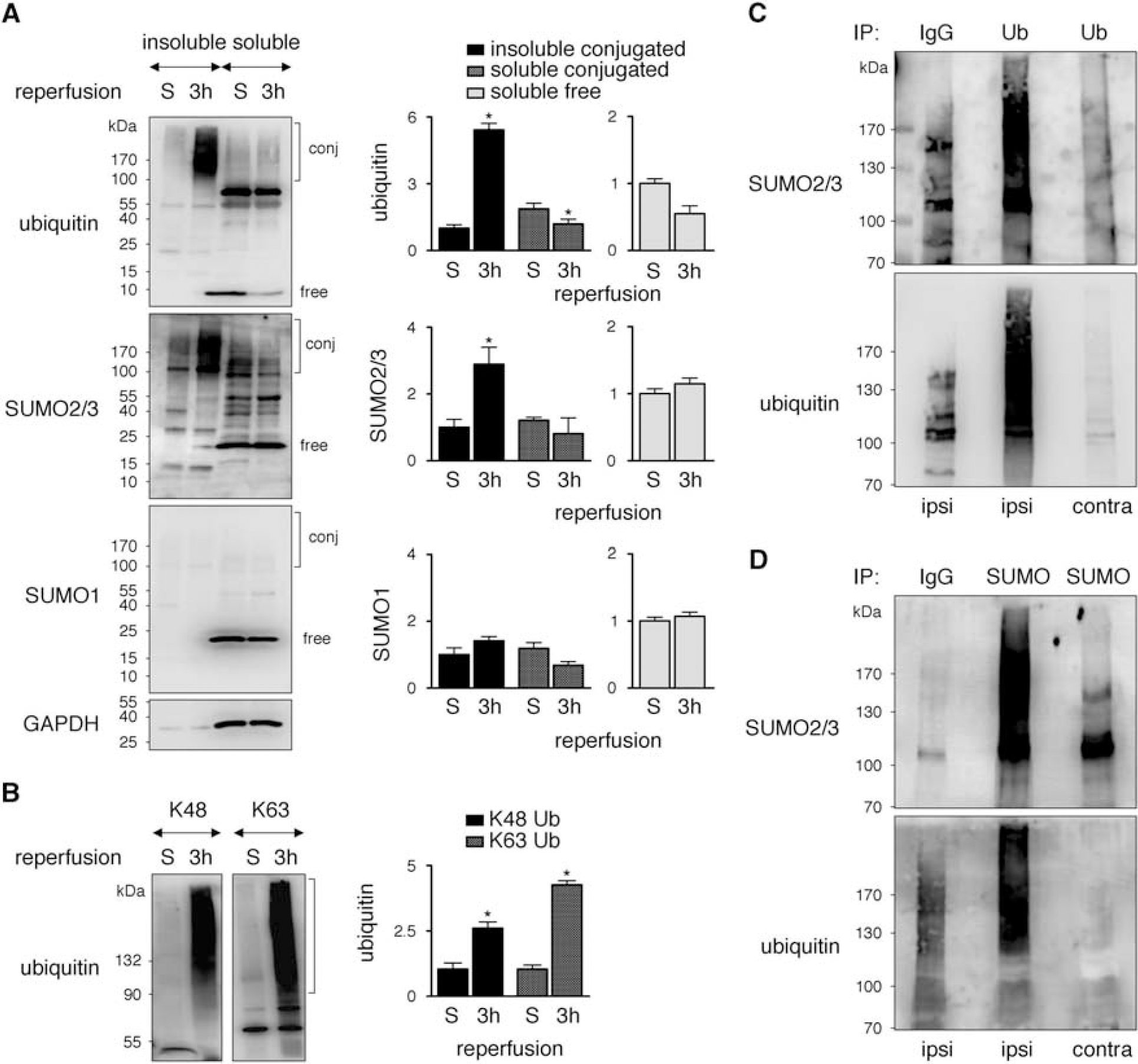
SUMO2/3, but not SUMO1, coaggregates and exhibits a direct interaction with ubiquitin in Triton X-100 insoluble aggregates in the ipsilateral neocortex after transient MCAO. (
SUMO2/3 and Ubiquitin Interact within Postischemic Aggregates
To examine the potential association between SUMOylation and ubiquitination after ischemia, we asked whether SUMO2/3 and ubiquitin could physically interact within postischemic aggregates. To this end, we solubilized aggregate fractions by SDS-mediated denaturation and ensured successful solubilization by exclusive detection of ubiquitin conjugates in the supernatant (data not shown). When ubiquitin was pulled down from SDS-solubilized contents we observed significant coprecipitation of SUMO2/3 (Figure 1C). Only minimal ubiquitin or SUMO was detected in the contralateral cortex or by pull down with an unrelated IgG control antibody from the ipsilateral cortex, attesting to the specificity of the interaction. To further confirm our data obtained with ubiquitin pull down we performed reciprocal immunoprecipitation with SUMO2/3 and detection of ubiquitin by western blotting. Analogous to the result with ubiquitin pull down, we found marked coprecipitation of ubiquitin with SUMO2/3 (Figure 1D). These results suggest a direct association between ubiquitin and SUMO2/3 within Triton X-100 insoluble protein aggregates after focal cerebral ischemia.
SUMO2/3 Accumulation within Ubiquitin-Rich Aggregates is Induced Rapidly with Reperfusion Independent of the Duration of Ischemia
Next, we investigated whether SUMO2/3 aggregates are formed under the same conditions established earlier for ubiquitin aggregation. 2 Since reperfusion was absolutely necessary for ubiquitin accumulation, 2 we first examined SUMO aggregation levels after transient ischemia with different periods of reperfusion or times of permanent ischemia. Reperfusion readily induced SUMO2/3 accumulation peaking after 1 and 3 hours, while permanent middle cerebral artery occlusion failed to increase SUMOylation levels at any time point (Figure 2A). SUMO2/3 as well as ubiquitin aggregation occurred rapidly and was observed as early as 5 minutes after reperfusion (Figure 2B). Finally we determined SUMO2/3 aggregation levels after ischemic episodes of different durations. As also observed for ubiquitin (Figure 2C and ref. 2), middle cerebral artery occlusion lasting 10 or 15 minutes, which produces either no or minimal tissue damage in our model, 2 was sufficient to trigger SUMO2/3 aggregation (Figure 2C).
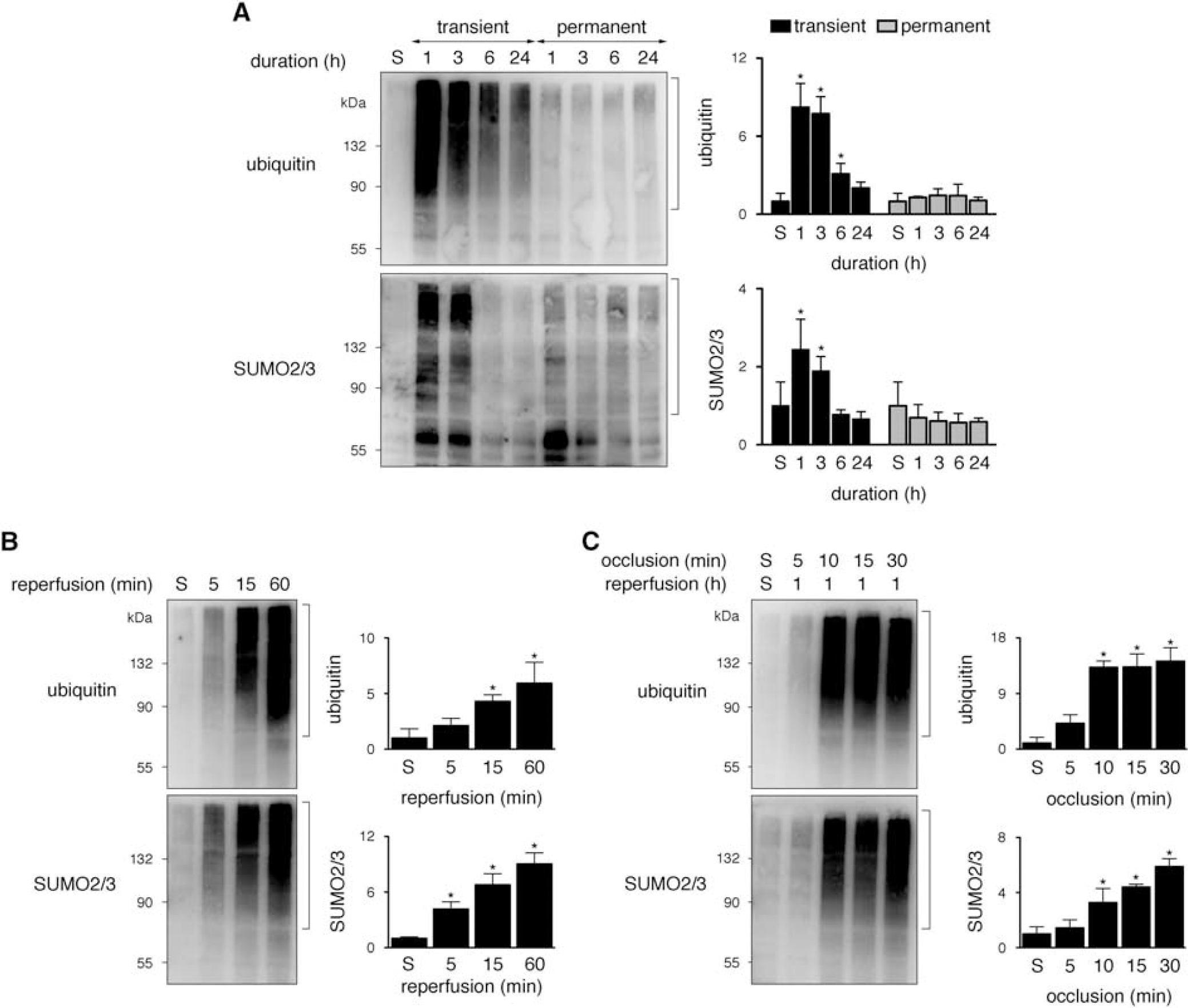
SUMO2/3 accumulation within ubiquitin-rich aggregates is rapidly induced dependent on reperfusion without a correlation to the duration and severity of ischemia. (
Discussion
Both SUMO and ubiquitin conjugates are dramatically increased after focal cerebral ischemia,2, 3, 6, 9 and here we provide evidence that these events might be linked. We find that SUMO conjugation is not only globally elevated after ischemia/reperfusion, but that it also, like ubiquitin, accumulates in insoluble protein aggregates. The accumulation is specific for SUMO2/3 since we could not identify any induction of SUMO1 conjugation. While SUMO2 and SUMO3 are identical except for three amino acids in the amino-terminus and are functionally indistinguishable, SUMO1 shares only 50% identity and has both distinct and overlapping targets. 11 Our finding of an induction of SUMO2/3, but not SUMO1 after ischemia is consistent with previous reports showing that SUMO2/3 constitutes a more abundant protein modification than SUMO1 in response to cellular stress.8, 12 Furthermore, our data in focal ischemia are in line with earlier observations in transient forebrain ischemia where no major changes in SUMO1 conjugation patterns were observed, while SUMO2/3 accumulation was significantly induced. 13
SUMO modification was first thought to be functionally distinct from the ubiquitin system. However, over the past decade it has become clear that in addition to their structural similarities, ubiquitin and SUMO have significant functional interactions.7, 14 These include (1) the competitive targeting of same attachment sites in common substrates, (2) the interdependent conjugation to distinct attachment sites in common targets, or (3) the sequential modification of different targets within the same signaling pathway, leading to antagonistic or synergistic effects on target protein properties. Our data of coprecipitation of SUMO2/3 and ubiquitin from SDS-denatured contents of the aggregate fraction strongly suggest a covalent, direct interaction between the two modifications. This could be in the form of SUMO2/3 and ubiquitin conjugated as monomers or mono-specific chains to different acceptor sites or as mixed chains to a common acceptor site within the same substrates. A potentially direct SUMO and ubiquitin interaction was suggested previously in a forebrain ischemia model where removal of SUMO conjugates led to a reduction in molecular weight of ubiquitinated proteins, indicating attachment to the same substrates. 15
An extensive crosstalk exists between SUMOylation and ubiquitination with potential implications for the functional consequences of the modification.7, 14 The significance of the accumulation of ubiquitin and SUMO2/3 conjugates for the outcome of cerebral ischemia, however, remains unknown. Of note, the induction of SUMO2/3 as well as ubiquitin conjugation is dependent on reperfusion, indicating that their presence might be associated with survival potential. Aggregation of SUMO and ubiquitin is rapid, transient, resolves before major tissue damage evolves and is not linked to cerebral infarction in our model, 2 suggesting that they are not causative for ischemic injury. In fact, elevated SUMO conjugation has been associated with neuroprotection, since its induction and suppression resulted in reduced and increased neuronal damage and infarction, respectively.4, 5, 6 The implications of ubiquitin aggregation for the outcome of focal cerebral ischemia are not clear. However, our previous data suggest that the presence of ubiquitin aggregates may in fact indicate a greater potential for tissue viability in this setting. 2 Considering the interaction of postischemic SUMO2/3 and ubiquitin modifications shown here, we speculate that SUMO2/3 and ubiquitin may work in concert to protect vital constituents of the cellular proteome from ischemic stress. Future studies must be directed at (1) understanding the contributing factors for the induction of ubiquitin/SUMO accumulation and (2) identifying modified substrates to evaluate the actual implications for brain survival after stroke.
Footnotes
The authors declare no conflict of interest.
