Abstract
Ubiquitylation is a posttranslational protein modification that modulates various cellular processes of key significance, including protein degradation and DNA damage repair. In animals subjected to transient cerebral ischemia, ubiquitin-conjugated proteins accumulate in Triton-insoluble aggregates. Although this process is widely considered to modulate the fate of postischemic neurons, few attempts have been made to characterize the ubiquitin-modified proteome in these aggregates. We performed proteomics analyses to identify ubiquitylated proteins in postischemic aggregates. Mice were subjected to 10 minutes of forebrain ischemia and 4 hours of reperfusion. The hippocampi were dissected, aggregates were isolated, and trypsin-digested after spiking with GG-BSA as internal standard. K-
INTRODUCTION
Transient cerebral ischemia activates various posttranslational protein modifications. These include ubiquitylation, small ubiquitin-like modifier conjugation, and ISGylation, all of which modify lysine residues in target proteins.1, 2, 3, 4 Transient cerebral ischemia triggers accumulation of ubiquitylated proteins that form Triton X-100-insoluble aggregates. 3 Aggregates of ubiquitylated proteins start to appear after ischemia during early reperfusion when cells are still morphologically intact. 5 Several groups have reported postischemic accumulation of ubiquitin-conjugated proteins in Triton X-100-insoluble aggregates,2, 3, 5, 6, 7, 8, 9, 10 but the significance of this process for the fate and functions of postischemic neurons has not yet been uncovered. No attempts have been made to perform proteomics analysis to characterize the ubiquitin-modified proteome in postischemic Triton X-100-insoluble aggregates.
Ubiquitin proteomic analysis is hampered by low levels of ubiquitylated proteins. Without efficient and specific enrichment before LC-MS/MS analysis, co-immunoprecipitation (IP) of proteins that are not ubiquitylated together with ubiquitylated proteins could be a major problem. To overcome problems associated with conventional ubiquitin proteomics analyses, a new strategy has recently been established that enables highly specific enrichment of ubiquitylated peptides derived from trypsin-digested ubiquitylated proteins.11, 12, 13 This approach takes advantage of antibodies that bind specifically to the di-glycine remnant (K-
MATERIALS AND METHODS
Animal Experiments
The following study was approved by the Duke University Animal Care and Use Committee (Durham, NC, USA) and complies with the Guide for the Care and Use of Animals published by the National Institutes of Health. Male C57Bl/6J mice (Jackson Laboratories, Bar Harbor, ME, USA) 8- to 10-weeks-old and weighing 20 to 25 g were fasted overnight with free access to water. 1 Mice were anesthetized with 5% isoflurane. The trachea was intubated with a 20-gauge intravenous catheter (Insyte-W, Becton-Dickenson, Sandy, UT, USA), and the isoflurane concentration was reduced to 1.8% of the inspired air. The rectal temperature was servocontrolled at 37°C by surface cooling or heating during ischemia and for 30 minutes after onset of reperfusion. The right internal jugular vein was cannulated to withdraw blood.
Forebrain ischemia was induced by bilateral common carotid artery occlusion and blood withdrawal to reduce mean arterial blood pressure to 30 mm Hg. After 10 minutes of ischemia, carotid arteries were deoccluded, withdrawn blood was reinfused, and catheters were removed. The wound was infiltrated with bupivacaine and closed. After 4 hours of reperfusion, mice were reanesthetized with 5% isoflurane and decapitated. The brains were quickly removed, and hippocampi were excised, immediately frozen, and stored at −80°C. Sham-operated control mice underwent the same procedures as experimental animals, except that the carotid arteries were not occluded and blood was not withdrawn. For the present study, animals were randomized and all mice exposed to sham or ischemia surgery for used for analysis. In earlier studies, using the same animal model, we found that ∼40% of CA1 hippocampal neurons were damaged.
Isolation of Triton X-100-Insoluble Protein Aggregates
Hippocampi from 6 to 8 animals were pooled, and Triton X-100-insoluble protein aggregates were isolated using three consecutive steps. Combined hippocampi were first weighed while still frozen and then homogenized with 800
Western Blot Analysis
For western blot analysis, homogenates and supernatant fractions were mixed 1:2 with 2 × Laemmli sample buffer and heated to 99°C for 10 minutes. Immunoblotting was performed using 4% to 15% SDS-PAGE gels (Bio-Rad, Hercules, CA, USA). After electrophoresis, proteins were transferred to polyvinylidene difluoride membranes (Bio-Rad). Membranes were blocked with 0.1% TBST and 5% skim milk powder, and then incubated with the primary antibody overnight at 4°C, followed by incubation with the secondary antibody for 1 hour at room temperature. Protein bands were visualized using the ECL western blotting detection reagents (GE Healthcare, Pittsburgh, PA, USA). The following primary and secondary antibodies were used: mouse anti-ubiquitin, rabbit anti-ubiquitin K48 linkage, rabbit anti-Nedd8, rabbit anti-ISG15, rabbit anti-CaMKII
Immunofluorescence Staining
Immunofluorescence staining was performed as described previously. 14 In short, after transcardial perfusion, brains were harvested and embedded in paraffin. After deparaffinization, sections were incubated with rabbit anti-hnRNP A/0 (1:400; Cell Signaling Technology), mouse anti-ubiquitin (1:500; Millipore, Billerica, MA, USA), or mouse anti-CaMKII (1:500; Abcam, Cambridge, MA, USA) at 4°C overnight. Sections were then incubated with Alexa Fluor 594-conjugated goat anti-rabbit IgG and Alexa Fluor 488-conjugated goat anti-mouse IgG (1:500; Invitrogen) at 20°C for 1 hour. Images were captured on a Leica SP5 confocal microscope (Leica Microsystems, Mannheim, Germany).
Proteomics Analysis
Generation of K-ε -GG-modified bovine serum albumin internal standard
Boc-Gly-Gly-OSu was prepared in the Duke Small Molecule Synthesis Facility by carbodiimide-mediated coupling of
Protein isolation and trypsin digestion
Ubiquitin proteomics analysis was performed on Triton X-100-insoluble protein aggregates isolated from hippocampi from sham and postischemic animals. Hippocampi from 6 to 8 animals were pooled and processed together to yield 1.1 mg of total protein in the insoluble aggregates/sample, which was considered one biologic replicate. To account for biologic variability in the response to ischemia three biologic replicates were processed per group. Triton X-100-insoluble pellets were resuspended in 1.2 mL urea (8 mol/L) supplemented with ammonium bicarbonate (50 mmol/L, AmBic). Samples were then diluted with 50 mmol/L AmBic to 1.8 mol/L urea. As an internal standard to control for quantitative recovery, 30 ng GG-BSA was added to each sample. Cysteine residues were reduced with 10 mmol/L dithiothreitol for 25 minutes at 32°C, and samples were alkylated at 20 mmol/L iodoacetamide for 20 minutes at room temperature in the dark. TPCK trypsin (USB Corporation, Santa Clara, CA, USA) was then added to each sample (25:1 substrate:enzyme ratio), and digestion was allowed to proceed at 32°C overnight. Samples were acidified to 0.5% v/v TFA, and desalted using 500 mg SEP-PAK C18 cartridges (Waters Corporation, Milford, MA, USA), per manufacturer's protocol. Peptide-containing eluates (2.25 mL, 50:49.9:0.1v/v/v MeCN/H2O/TFA) were diluted with 3 mL water, frozen at −80°C, and lyophilized overnight to yield peptide samples ready for IP.
K-ε -GG peptide immunoprecipitation
K-
Liquid chromatography tandem mass spectrometry analysis, database searching, and peptide quantitation
Label-free quantitative LC-MS/MS was performed in duplicate for each sample (5
The order of data collection was interwoven between conditions to minimize temporal bias, alternating sample analysis between biologic conditions with the technical replicates of each sample run back to back. After the 12 analyses, data were imported into Rosetta Elucidator v3.3 (Rosetta Biosoftware, Seattle, WA, USA), and all LC-MS/MS runs were aligned based on the accurate mass and retention time of detected ions using PeakTeller algorithm (Elucidator). The relative peptide abundance was calculated based on area-under-the-curve of aligned features across all runs. The overall data set had 50,569 quantified isotope (peptide) groups. In addition, 77,539 MS/MS spectra were acquired for peptide sequencing by database searching. These MS/MS data were searched against a custom NCBI RefSeq database with
Database searching was performed in an automated fashion from the Elucidator software package using Mascot search engine v2.2, assigning a precursor ion tolerance of 5 p.p.m. and product ion tolerance of 0.8 kDa. Searching allowed variable modification of N and Q (deamidation, +1 kDa), M (oxidation, +16 kDa), and K (diglycyl, +114 kDa). After aggregating all search results, assignment of peptide sequences to the quantitative MS signal was performed for peptides with an ion score greater than 24, to obtain a 1% false discovery rate at the peptide level according to the ratio of decoy database identifications to forward identifications. For quantification, the data were first curated to contain only K-
Bioinformatics analysis
Hierarchical clustering analysis was performed within Rosett Elucidator (Rosetta Biosoftware) using
RESULTS
Results from several experimental studies suggest that transient cerebral ischemia triggers accumulation of ubiquitylated proteins in Triton X-100-insoluble aggregates. This aggregation is believed to trap proteins required for physiologic cell functions. In the present study, we have used a model of transient global cerebral ischemia and focused ubiquitin proteomics analysis on the hippocampus, a brain region particularly affected in this model. We also used 4 hours of reperfusion to be able to compare our results with findings from an earlier study. 16
First, we verified that transient cerebral ischemia activated ubiquitin conjugation in our model. Hippocampi were dissected from sham and postischemic brains, and Triton-insoluble proteins were isolated as detailed in Materials and Methods. Western blots of whole-cell homogenates showed a marked postischemic activation of global ubiquitin conjugation (Supplementary Figure S1, ubiquitin). After differential centrifugation to isolate Triton X-100-insoluble protein aggregates, a significant fraction of ubiquitylated proteins was still present in postischemia samples but markedly less in sham samples (Supplementary Figure S1, ubiquitin T3). When we stripped membranes and re-probed with an antibody against polyubiquitin with K48 linkage, the pattern was quite similar to the ubiquitin pattern. We used eIF2
In the present study, we used a recently established strategy for specific enrichment of ubiquitylated peptides. This strategy takes advantage of an antibody that binds specifically to the di-glycine remnant on lysine residues, which is derived from trypsin digestion of ubiquitin-conjugated proteins. This di-glycine remnant is also generated by trypsin digestion of Nedd8- or ISG15-conjugated proteins. To determine whether Nedd8- or ISG15-conjugation could potentially contribute to K-
Next, we verified the extent of postischemic accumulation of ubiquitylated proteins in Triton X-100-insoluble protein aggregates by running western blots using all samples prepared from sham and postischemic hippocampi for proteomics analysis. Levels of ubiquitin-conjugated proteins and of K48 linkage polyubiquitin were dramatically increased in postischemic samples (Figure 1). Optical density measurements and statistical analysis revealed that transient ischemia caused a 11.0±0.4% increase in levels of ubiquitin-conjugated proteins in Triton X-100-insoluble aggregates (
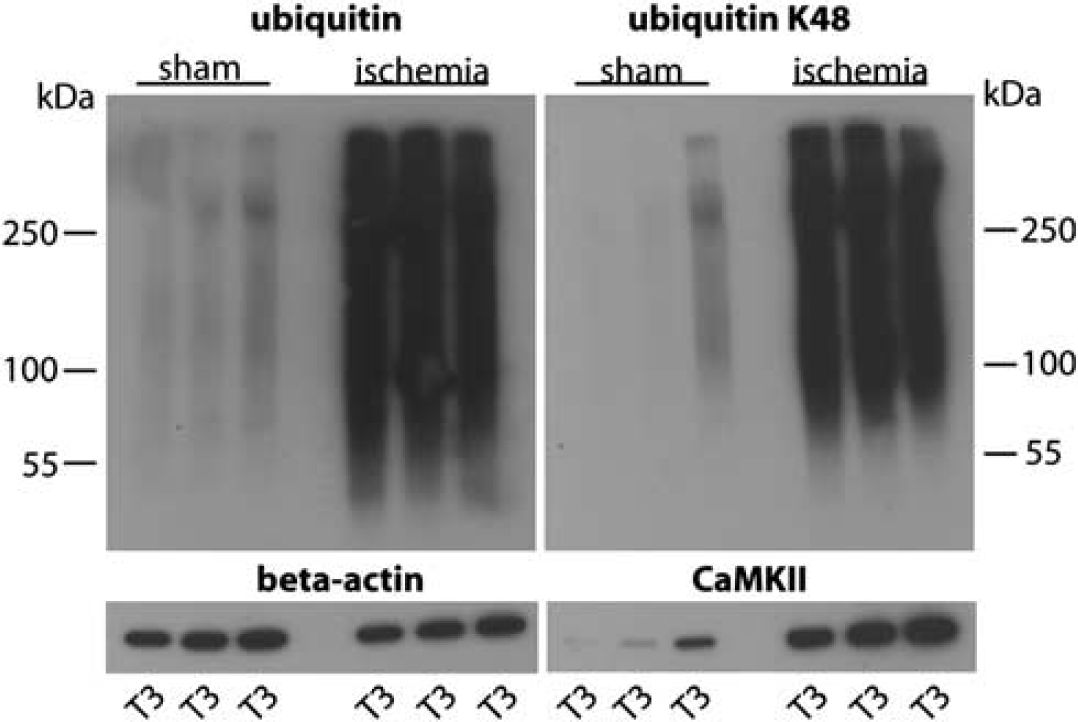
Transient cerebral ischemia results in accumulation of ubiquitin-conjugated proteins in Triton X-100-insoluble aggregates. Western blot analysis depicts the pattern of ubiquitin conjugation and K48 linkage polyubiquitylation in sham and postischemic T3 samples, representing Triton X-100-insoluble protein aggregates used for proteomics analysis. Note the dramatic increase in ubiquitin and K48 linkage ubiquitin conjugates in postischemic hippocampi. CaMKII was markedly enriched in postischemic T3 samples. Antibody against
Proteomics analysis identified a total of 5,483 peptides to 1,320 proteins, including 1,664 peptides to 520 proteins containing at least one K-
To assess the global pattern of ubiquitylated proteins in Triton X-100-insoluble aggregates and visually compare this pattern with that observed

Two-dimensional (2-D) hierarchical clustering across all K-
Next, we defined selection criteria of >fivefold increase and
Proteins with 10 or more K-

List of proteins enriched at least fivefold (
To visually illustrate the differential expression data obtained by LC-MS/MS analysis, Figure 3A shows an example of the raw chromatographic data for a double-ubiquitylated peptide to the ETS-related transcription factor Elf-1. The peptide K.GGAATILK∗PGNS[KAANPK]∗.D was virtually absent in the LC-MS/MS analyses of sham samples, and clearly abundant in the ischemia samples. In Figures 3B, C confocal microscopic images illustrate the distribution of ubiquitin and 2 of the ubiquitin-conjugated proteins identified in Triton X-100-insoluble aggregates—heterogeneous nuclear ribonucleoprotein A/0 (hnRNPA0; Figure 3B) and CaMKII, the protein that showed the most pronounced postischemic increase in ubiquitylation (Figure 3C). Ubiquitin immunoreactivity was markedly increased after ischemia (Figure 3B), as expected from ubiquitin western blots (Supplementary Figure S1, ubiquitin). No clear colocalization of ubiquitin and hnRNP A/0 was seen in the sham brain. After ischemia, however, many of the hnRNP A/0-positive nuclear dots were also positive for ubiquitin, as indicated by the yellow dots in the merged image (Figure 3B, arrowheads). The sham brain displayed strong CaMKII immunoreactivity; but after ischemia, CaMKII immunoreactivity disappeared almost completely (Figure 3C). This pattern is consistent with earlier observations that hypoxia-ischemia in neonatal rats triggered trapping of CaMKII in Triton X-100-insoluble aggregates and disappearance of CaMKII immunoreactivity. 19 This postischemic disappearance of CaMKII immunoreactivity may have been caused by inability of the antibody to bind to the protein because of the high ubiquitylation rate or the trapping in protein aggregates.
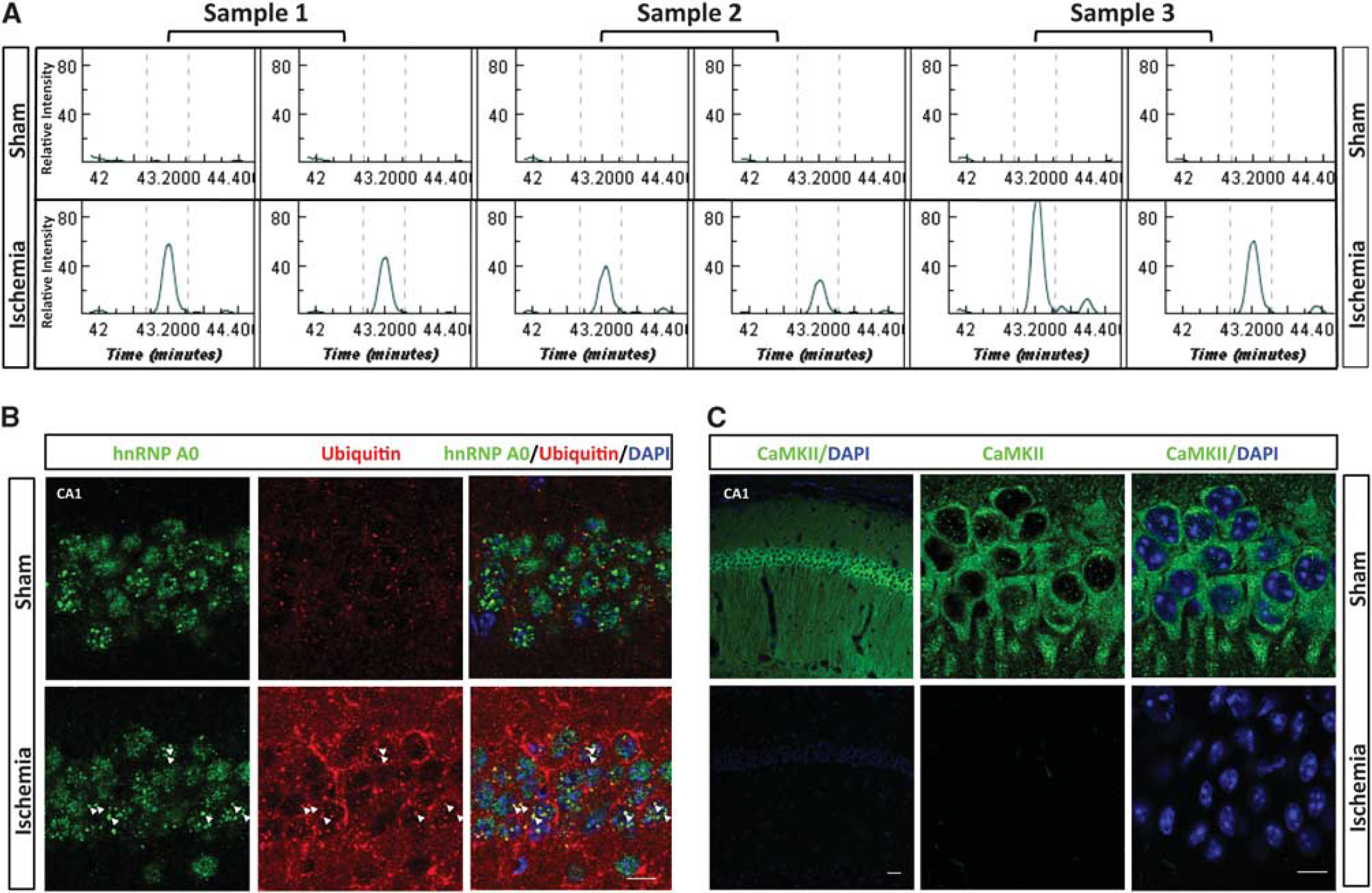
Visual illustration of ubiquitin proteomics results. (
As we were also interested to investigate whether any sequence patterns surrounding the ubiquitylation sites were preferentially targeted in postischemic aggregates, we performed a Motif-X analysis (http://www.motif-x.med.harvard.edu) using either the entire list of modified peptides identified (Supplementary Table S1) or the statistically significant data set (Supplementary Table S2).20, 21 Using these data sets as the input, there was a single AxK motif that was identified in the full data set (
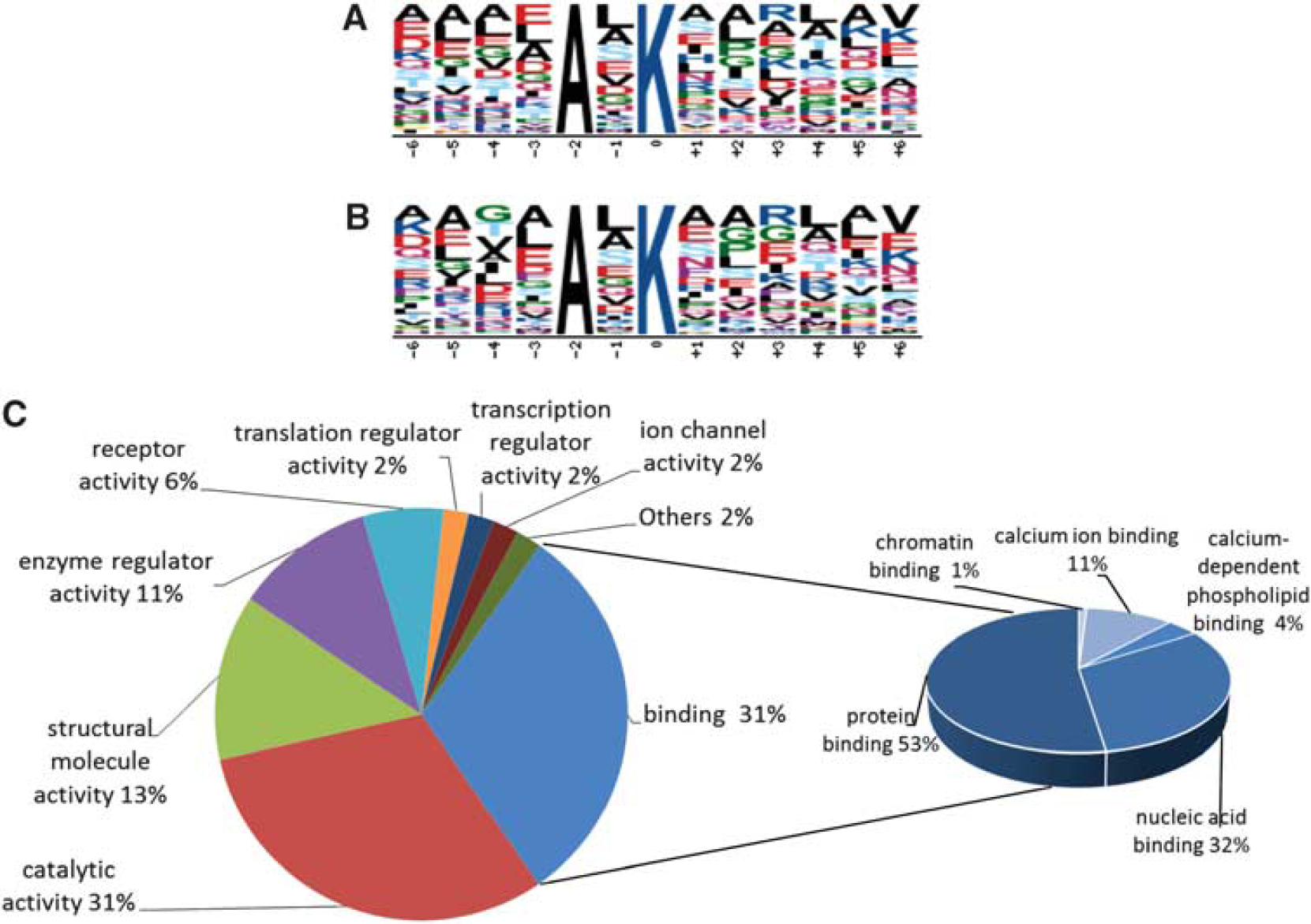
Bioinformatic analysis. Motif-X analysis of the entire ubiquitin-modified proteome of the Triton X-100-insoluble fraction (
Ingenuity Pathway Analysis was performed on the 272 proteins that fulfilled our strict selection criteria, to identify the neuronal functions and canonical pathways associated with the data set. Postischemic Triton X-100-insoluble protein aggregates were highly enriched with proteins associated with long-term potentiation (26 of the identified proteins;
DISCUSSION
We report here the first comprehensive analysis of the ubiquitin-modified proteome regulated by transient forebrain ischemia. Proteomics analyses were performed on Triton X-100-insoluble protein aggregates isolated from hippocampi, the brain region most vulnerable to even a short period of vascular occlusion.
23
We made slight modifications to a recently developed highly specific ubiquitin proteomics approach.
11
To account for inter-sample variability in the efficacy of di-glycine remnant motif IP and the consecutive proteomics analysis, we used a novel approach by adding a chemically distinct (exogenous) internal standard (GG-BSA) to all samples before IP. To account for biologic variability, we analyzed three independent samples/group, and defined stringent selection criteria for statistical significance in the ischemia-induced ubiquitin-modified proteome (fivefold increase in ischemia versus sham samples and
Lysine is the preferred amino acid modulated by various posttranslational protein modifications, including conjugation by acetate, ubiquitin, small ubiquitin-like modifier, ISG15, and Nedd8. Ubiquitin, ISG15, and Nedd8 all have the identical C-terminal sequence–RGG, and form K-
Earlier studies reported postischemic translocation of CaMKII and protein kinase C from the soluble to the particulate (insoluble) fraction.8, 10, 17, 18 We found these proteins to be highly ubiquitylated in postischemic Triton X-100-insoluble aggregates (Table 1). Furthermore, proteins of the translational machinery were reported to be highly ubiquitylated in postischemic aggregates, and it has been proposed that this trapping of components of the translational complex contributes to the irreversible suppression of proteins synthesis.16, 24, 25 In our list of 272 ubiquitylated proteins highly enriched in postischemic protein aggregates, we identified five chaperons (heat shock 70 kDa proteins 12A and 4L, and Dnaj homologs subfamily A member 1, and subfamily B members 2 and 6), three eukaryotic initiation factors (eIF4H, eIF4A-1, and eIF5A-1), and two elongation factors (eEFalpha 1 isoforms 3 and 9), thus supporting earlier observations.
The role of the postischemic accumulation of proteins in Triton X-100-insoluble aggregates for the fate and functions of postischemic neurons has not yet been uncovered. At least two different scenarios can be envisioned. Protein aggregates
The mechanisms underlying the postischemic accumulation of ubiquitylated proteins in Triton X-100-insoluble aggregates still need to be uncovered. Two different scenarios can be envisioned. The accumulation could be an active process or a non-specific sequestration owing to an overall postischemic activation of protein ubiquitylation. Indeed, we reported here that transient cerebral ischemia resulted in an increase in protein ubiquitylation, as indicated by the more pronounced smear of bands of ubiquitin-conjugated proteins on western blots of whole tissue homogenates from postischemic samples (Supplementary Figure S1, T1 ischemia versus T1 sham). This overall postischemic increase in levels of ubiquitylated proteins resulted from a specific and dramatic trapping of ubiquitin-conjugated proteins in Triton X-100-insoluble aggregates (Supplementary Figure S1, T3 ischemia versus T3 sham), while changes in the soluble fractions were minor (Supplementary Figure S1 ischemia versus S1 sham). This suggests the postischemic trapping of ubiquitylated proteins in insoluble aggregates to be an active process. Whether proteins were first sequestered in those aggregates and then ubiquitylated or vice versa needs to be elucidated in future studies. The observation that ubiquitylated CaMKII trapped in postischemic aggregates was not accessible any more for the CaMKII antibody (Figure 3C) supports a scenario whereby proteins were ubiquitylated before being sequestered in aggregates.
In the brain, ubiquitin conjugation is a physiologic process, and 1,786 K-ε-GG sites on 2,064 peptides to 921 proteins have recently been identified in normal rat brains, including CaMKII subunits alpha, delta, and gamma, and NMDA receptor subunits NR1 and NR2B. 22 CaMKII and the NMDA receptor that were found in postischemic insoluble aggregates (Table 1) form a complex that has a role in long-term potentiation. 26 Long-term potentiation is one of the major mechanisms underlying learning and memory. 27 Activity levels of neurons control the postsynaptic composition and signaling by modulating the composition of the postsynaptic density by reversible activation of ubiquitin conjugation/deconjugation. 28 Although the ubiquitin–proteasome system is an integral component of neuronal functioning, it is reasonable to predict that trapping ubiquitin-conjugated proteins in insoluble aggregates may impair neuronal function in the postischemic brain. Indeed, many of the ubiquitylated proteins that were trapped in postischemic Triton X-100-insoluble aggregates have pivotal roles in protein synthesis, and learning and memory processes, both of which are impaired after ischemia.29, 30, 31, 32
A hallmark of transient ischemia-induced neuronal cell death is an irreversible translational arrest.33, 34 This translational arrest is believed to be triggered by ischemia-induced impairment of endoplasmic reticulum functions that activates the unfolded protein response, resulting in shutdown of protein synthesis.35, 36, 37 The initiation factor eIF3 and the chaperons HSP40 and HSC70 were earlier reported to be trapped in postischemic protein aggregates. 16 We also found that many initiation and elongation factors and various chaperons were ubiquitylated after ischemia and trapped in Triton X-100-insoluble aggregates. Further, the calcium-binding protein hippocalcin was highly ubiquitylated after ischemia. Hippocalcin protects cells against endoplasmic reticulum stress-induced cell death and functions as a calcium sensor in synaptic plasticity.38, 39 Furthermore, 26 of the proteins that were highly ubiquitylated after ischemia and trapped in Triton X-100-insoluble aggregates have a role in long-term potentiation. It is therefore reasonable to conclude that the postischemic accumulation of ubiquitylated proteins in insoluble aggregates contributes to the pathologic process induced by transient cerebral ischemia and results in impaired endoplasmic reticulum function, suppression of protein synthesis, and impairment of neuronal functions associated with learning and memory.
Our ubiquitin proteomics analyses identified ubiquitin conjugated to ubiquitin at K6, K11, K48, and K63, indicating postischemic formation of polyubiquitin chains with different linkages. The K48 linkage, which targets proteins for degradation at the proteasome, was the most dysregulated with ischemia. Polyubiquitin chain formation with K6, K11, and K63 linkages are involved in various cellular processes, including DNA damage repair, endocytosis, inflammation, and kinase signaling. Of note, K63 linkage polyubiquitylation is involved in the formation of protein inclusions associated with neurodegenerative diseases. 40 Whether K6, K11, and K63 linkage polyubiquitylation and trapping of these modified proteins in postischemic aggregates indeed have a role in the fate and functions of postischemic neurons must be verified in future studies.
In conclusion, we presented here the first comprehensive proteomics analysis of the ubiquitin-modified proteome regulated by transient forebrain ischemia. We focused our analysis on the Triton X-100-insoluble fractions isolated from hippocampi of sham and postischemic brains, and we spiked samples, for the first time, with GG-BSA as internal standard before trypsin digestion—a step critical to successful label-free quantitative analysis. The 763 K-ε-GG-modified peptides to 272 proteins that fulfilled our selection criteria of fivefold increase and
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors thank Pei Miao for her excellent technical support and Kathy Gage for her excellent editorial contribution in the preparation of this manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
