Abstract
Geranylgeranylacetone (GGA) is an inducer of heat-shock protein 70 (HSP70) that has been used clinically for many years as an antiulcer treatment. It is centrally active after oral administration and is neuroprotective in experimental brain ischemia/stroke models. We examined the effects of single oral GGA before treatment (800 mg/kg, 48 hours before trauma) or after treatment (800 mg/kg, 3 hours after trauma) on long-term functional recovery and histologic outcomes after moderate-level controlled cortical impact, an experimental traumatic brain injury (TBI) model in mice. The GGA pretreatment increased the number of HSP70+ cells and attenuated posttraumatic α-fodrin cleavage, a marker of apoptotic cell death. It also improved sensorimotor performance on a beam walk task; enhanced recovery of cognitive/affective function in the Morris water maze, novel object recognition, and tail-suspension tests; and improved outcomes using a composite neuroscore. Furthermore, GGA pretreatment reduced the lesion size and neuronal loss in the hippocampus, cortex, and thalamus, and decreased microglial activation in the cortex when compared with vehicle-treated TBI controls. Notably, GGA was also effective in a posttreatment paradigm, showing significant improvements in sensorimotor function, and reducing cortical neuronal loss. Given these neuroprotective actions and considering its longstanding clinical use, GGA should be considered for the clinical treatment of TBI.
INTRODUCTION
Traumatic brain injury (TBI) is a major public health problem, with at least 1.7 million cases annually in the United States, contributing to almost a third of injury-related deaths, and resulting in enormous societal costs. 1 It causes cell death and neurologic dysfunction through direct, immediate physical damage as well as by delayed, potentially reversible molecular and cellular mechanisms that cause progressive tissue loss (secondary injury). 2 The mechanisms underlying secondary injury involve a complex cascade of biochemical, metabolic, and gene expression changes leading to not only initiation of intrinsic neuronal cell death, but also activation of both acute and chronic neuroinflammatory responses that contribute to further degeneration and neurologic dysfunction. 3
Heat-shock protein 70 (HSP70) is an abundant and highly conserved molecular chaperone involved in protein homeostasis that modulates the folding, function, and degradation of proteins. It is stress inducible and promotes cell survival under various pathologic conditions, such as tissue/organ trauma, ischemia–reperfusion injury, and during inflammation. It mediates its prosurvival effects by stabilizing proteins against denaturation and aggregation, and also by interacting with multiple components of caspase-dependent and -independent apoptotic pathways. 4 Exogenous HSP70 overexpression attenuated neuronal loss in models of cerebral ischemia or after kainic acid administration.5, 6 Conversely, HSP70−/− mice are associated with higher infarct volumes after cerebral ischemia. 7 Overexpression of HSP70 in cerebral ischemia models modulates several key neuronal cell death mechanisms; it inhibits the intrinsic caspase activation pathways by reducing release of cytochrome c8, 9 and inactivating Apaf-1 (apoptotic protease activating factor 1), 10 and attenuates caspase-independent pathways by sequestering apoptosis-inducing factor (AIF). 9 Studies suggest that HSP70-mediated neuroprotective effects that lead to improved neurologic function are primarily mediated via inhibition of apoptotic pathways (e.g., prevention of AIF translocation to the nucleus) rather than by its chaperone effects. 11 HSP70 also attenuates activation of microglia and astrocytes by decreasing activation/nuclear translocation of nuclear factor-κB, resulting in reduced proinflammatory signaling in these cells. 12
The preponderance of evidence suggests that HSP70 has significant neuroprotective effects across multiple models of brain ischemia/stroke by targeting many of the same secondary injury mechanisms that are present in TBI. Thus, HSP70-overexpression strategies hold significant promise as therapeutic interventions for TBI. Geranlygeranylacetone (GGA) is an acyclic isoprenoid compound originally developed in Japan as an antiulcer drug. 13 It penetrates the blood–brain barrier and is centrally active after oral administration, and induces HSP expression in many tissues, including the central nervous system (CNS).13, 14 It is well tolerated in humans and has no known side effects. 15 A single oral dose of GGA upregulates HSP70 expression in the CNS and promotes neuroprotective effects in response to various nontraumatic injuries. 16
In this study, we investigated the neuroprotective effects of GGA in a clinically relevant experimental model of TBI in mice. Single oral dose administration of GGA at 48 hours before TBI resulted in significantly reduced neuronal loss and microglial activation, and improved long-term functional recovery in injured mice. Notably, GGA posttreatment at 3 hours after injury also produced significant neuroprotective effects in various functional and histologic outcomes.
MATERIALS AND METHODS
Animals
The described experiments were performed in accordance with the Animal Welfare Act, Public Health Service (PHS) Policy on Humane Care and Use of Laboratory Animals, the Guide for the Care and Use of Laboratory Animals, and all other applicable regulations, policies, and procedures, and were approved by the University of Maryland School of Medicine Institutional Animal Care and Use Committee. Ten-week-old male C57BL/6 mice (20 to 25 g) were obtained from Taconic Farms (Derwood, MD, USA) and were handled as previously described. 17
Controlled Cortical Impact Injury
Controlled cortical impact (CCI) was performed as previously described. 17 The injury device consists of a microprocessor-controlled pneumatic impactor, driven by compressed air, with a 3.5 mm diameter tip. Moderate-level brain injury was induced by an impact velocity of 6 m/s and a deformation depth of 2 mm. Sham animals underwent the same procedure, including craniotomy, as injured mice except for the impact.
Drug Administration
In the pretreatment GGA study, mice received GGA (800 mg/kg in 5% gum arabic; n=10) or an equal volume of vehicle (n=9) orally 48 hours before injury. In the posttreatment GGA study, mice received same dose of GGA (n=6) or vehicle (n=7) orally 3 hours after injury. The dosing and administration of GGA was based on previous studies.13, 18 Both pre- and posttreatment GGA studies had six sham mice, respectively.
Motor Function Evaluation
Chronic motor function recovery was evaluated using a beam walking task, a method that is particularly good at discriminating fine motor coordination differences between injured and sham-injured animals. 17 The mouse was placed on the beam and the number of foot faults for the right hindlimb recorded over 50 steps. A basal level of performance was achieved after 3 days of training before surgery with an acceptance level of <10 foot faults per 50 steps. The test was performed at 0 (immediately before CCI), 1, 3, 7, 14, and 28 days after injury.
Morris Water Maze and Reversal Morris Water Maze Tests
Spatial learning and memory was assessed using the acquisition phase of the standard Morris water maze (sMWM) and reversal MWM (rMWM) tests as previously described. 17 The Morris water maze protocol included four phases: (1) standard hidden platform training (acquisition); (2) standard probe test; (3) reversal hidden platform training (reversal acquisition); and (4) reversal probe test. The swim path, latency to platform, time spent in each zone, and velocity were recorded by computer-based Any-Maze automated video tracking system (Stoelting, Wood Dale, IL, USA). Reference memory was assessed by a probe test on postinjury day (PID) 18. For the rMWM training, the hidden platform was moved from the northeast to southwest quadrant without changing any visual cues. A reversal probe test was performed on PID 23. Water maze search strategy analysis was performed as previously described. 17 The search strategies were analyzed on each of the 4 trials on day 4 of MWM and rMWM as previously described. 17
Novel Object Recognition Test
Novel object recognition (NOR) evaluated nonspatial hippocampal-mediated memory on PIDs 24 and 25, as previously described. 17
Tail-Suspension Test
The tail-suspension (TS) test assesses depression-like behavior in mice and is based on the observation that mice develop an immobile posture when placed in an inescapable hemodynamic stress of being hung by their tails. The TS was performed on PID 24 as described previously. 17
Lesion Volume Assessment
Mice were killed and transcardially perfused with saline and 10% buffered formalin phosphate solution (containing 4% paraformaldehyde; Fisher Scientific, Pittsburg, PA, USA) on PID 28. Lesion volume was determined based on the Cavalieri method as previously described. 19 The lesion area was outlined using the Stereologer 2000 program (Systems Planning and Analysis, Alexandria, VA, USA) to obtain the final volume measurements.
Assessment of Neuronal Cell Loss in the Hippocampal Subregions, Cortex, and Thalamus
Stereoinvestigator software (MBF Biosciences, Williston, VT, USA) was used to count the total number of surviving neurons in the cortex, thalamus, as well as Cornu Ammonis (CA) 1, CA2/3, and dentate gyrus (DG) subregions of the hippocampus using the optical fractionator method of unbiased stereology, as previously described. 19
Assessment of Microglial Morphology in the Cortex
Stereoinvestigator software (MBF Biosciences) was used to count the number of cortical microglia in each of the three microglial morphologic phenotypes (namely ramified, hypertrophic, and bushy) using the optical fractionator method of unbiased stereology as previously described. 20
Immunocytochemistry
Frozen coronal brain sections (20 μm) were stained with HSP70 antibody (1:100; Assay Design, Farmingdale, NY, USA) overnight and counterstained with 4,6-diamino-2-phenylindole (DAPI; 1:20,000, Sigma-Aldrich, St Louis, MO, USA). The slides were incubated with Alexa Fluor 546 Goat anti-mouse for 3 hours. Imaging was carried out using a Nikon Eclipse Ti and a Cool Snap EZ camera (Nikon, Melville, NY, USA). For quantitative image analysis, digital images were acquired at × 20 magnification (Nikon 20X Plan Apo objective) in multiple fields (n=5 per brain, a minimum of 1,474 cells per brain) around the cortical lesion regions (4 to 5 mice per group). The images were processed using the Nikon NIS Elements software V4.12.01 (Melville, NY, USA) to generate an unbiased quantification of the percentage of HSP70-positive cell in reference to the total DAPI-positive cells. The software automatically determined the threshold in each image and then counted the HSP70- and DAPI-positive cells. The HSP70-positive cells were identified by local threshold. Nuclei were identified using spot detection algorithm. The numbers obtained in each of the five fields were added to generate the total number of HSP70-positive cells for each animal. These numbers were then averaged to produce the values for each group (sham, Veh-TBI, pre-GGA-TBI, and post-GGA-TBI).
Western Blotting
At 6 hours after injury, sham+vehicle (n=3), sham+pre-GGA (n=3), CCI+vehicle (n=5), and CCI+pre-GGA (n=5) were killed and a 5-mm area surrounding the lesion epicenter on the ipsilateral cortex was rapidly dissected and immediately frozen on dry ice. Cortical tissue was homogenized in RIPA buffer and analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to nitrocellulose membrane. The immunoblots were probed with an anti-fodrin (1:3,000, Enzo Life Sciences International, Plymouth Meeting, PA, USA). β-Actin (1:20,000, Sigma-Aldrich) was used as an endogenous control. Chemiluminescence was captured on a Kodak Image station 4000R station (Carestream Health, Rochester, NY, USA), and protein bands were quantified by densitometric analysis using Carestream Molecular Imaging Software. The data presented reflect the intensity of the target protein normalized to protein levels of the endogenous control in each sample (expressed in arbitrary units).
Statistical Analysis
For the beam walk, acquisition, and reversal acquisition trials of the MWM test, repeated measures one-way analyses of variance (ANOVAs) were conducted, followed by Student–Newman–Keuls post hoc test to compare the differences between each group. One-way ANOVA analysis followed by Student–Newman–Keuls post hoc test was performed for the other behavioral tests and neuronal cell counts, microglia activation, and immunofluorescence quantification. One-tailed paired Student's t-test was used for the lesion volume. For the search strategy analysis, χ2 analysis was performed. For the composite score, nonparametric Kruskal–Wallis test was used. The Q-Q plots showed that the composite score data approached normality and showed equal variance; therefore, these data were subsequently analyzed using parametric statistics, and one-way ANOVA followed by Student–Newman–Keuls post hoc test was performed. Statistical analysis was performed using SigmaPlot Program, version 12 (Systat Software, San Jose, CA, USA) or GraphPad Prism software, version 4.00 for windows (GraphPad Software, San Diego, CA, USA). Data are expressed as mean±s.e.m., and significance was determined at P<0.05.
RESULTS
Geranylgeranylacetone Pretreatment Improved Spatial and Nonspatial Learning and Memory after Traumatic Brain Injury
To investigate the effect of GGA pretreatment on spatial learning and memory after moderate-level CCI, we performed the sMWM test from PID 14 to PID 18. The mean escape latency on the last day of training was 26.86±6.18 seconds for the sham-injured group, 66.38±6.96 for vehicle TBI group, and 46.78±8.93 for GGA pretreatment group (Figure 1A). Repeated measures one-way ANOVA showed a significant effect of treatment (F(2, 88)=12.84; P<0.001), day (F(2, 88)=12.40, P<0.0001), and the treatment–day interaction (F(6, 88)=2.75; P<0.05). Student–Newman–Keuls post hoc analysis revealed an improvement after GGA treatment with significant difference between the pre-GGA TBI and vehicle TBI groups on day 4 (P<0.05), and sham-injured and vehicle TBI groups on day 2 (P<0.05), day 3 (P<0.05), and day 4 (P<0.001) (Figure 1A). Probe tests were performed on PID 18 (sMWM) and reduced time spent in the target quadrant indicated impaired reference memory. The time spent in the target quadrant showed a significant effect of GGA treatment (Figure 1B; F(2, 24)=9.25, P<0.01), and Student–Newman–Keuls post hoc analysis revealed that the pre-GGA TBI group spent significantly more time in the target quadrant than the vehicle TBI group (P<0.05). Significant differences were also observed between sham-injured and vehicle TBI groups (P<0.01). We then performed search strategy analysis to evaluate the efficiency of locating the platform. Based on previously published criteria, 17 three search strategies were evaluated for each of the 4 trials on PID 17. Occasionally, mice changed search strategies during a trial. When this happened, the strategy that best described the major swimming path was assigned. Search strategy analysis showed statistically significant separation between the pre-GGA TBI and vehicle TBI groups (Figure 1C, χ2=265.3; P<0.001), with the vehicle TBI group exhibiting significantly higher reliance on looping search strategies than spatial and systematic search strategies when compared with the pre-GGA TBI group.
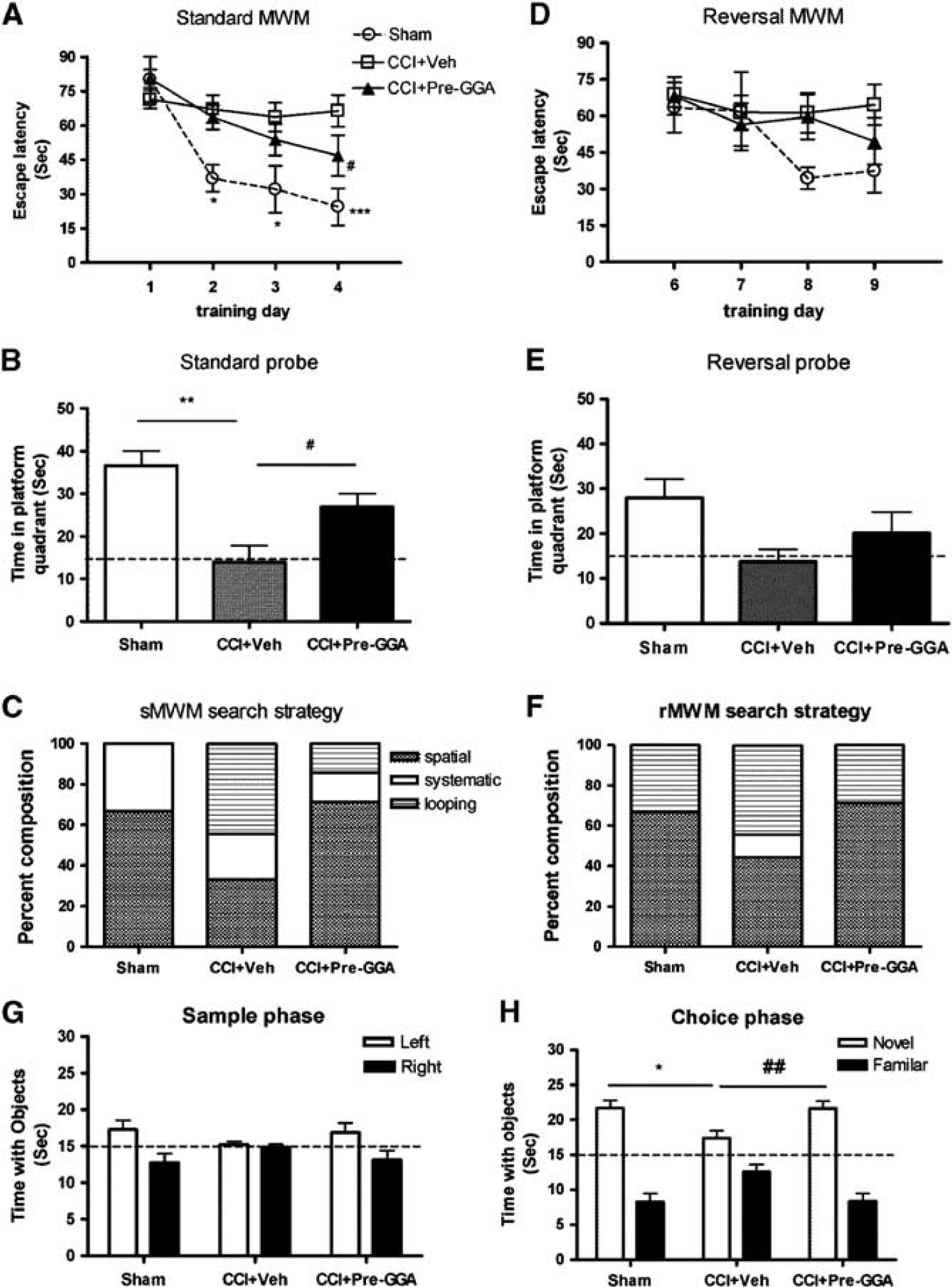
Geranylgeranylacetone (GGA) pretreatment improves cognitive function assessed by the standard Morris water maze (sMWM) and novel object recognition tests. (
The rMWM was performed from PID 19 to PID 23 to measure memory extinction and reacquisition in this study, and mean escape latency was recorded (Figure 1D). Repeated measures one-way ANOVA did not show a significant effect of treatment. A reversal probe test was performed on PID 23 (Figure 1E). The time spent in the target quadrant did not show a significant effect of treatment. There was an improvement after GGA treatment in the rMWM search strategy, and a significant difference between the pre-GGA TBI and vehicle TBI groups (Figure 1F, χ2=120.96; P<0.01). The vehicle TBI group had a higher reliance on looping search strategies than spatial and systematic search strategies than the pre-GGA TBI group. Finally, there were no significant differences in swim speeds across groups in the sMWM and rMWW trials (data not shown).
Hippocampal-mediated nonspatial learning and memory after TBI was assessed by the NOR test. Sham-injured, vehicle TBI, and pre-GGA TBI groups all spent similar times with two identical objects in the sample phase as shown in Figure 1G. The sham-injured group spent more time than chance (15 seconds) with the novel object 1 hour after training (choice phase), indicating intact memory (Figure 1H). The vehicle TBI group spent similar time with the novel and familiar objects, whereas the pre-GGA TBI group spent more time with the novel than familiar objects. Furthermore, one-way ANOVA analysis showed a significant effect of treatment on time with the novel object (F(2, 24)=5.16; P<0.05), and Student–Newman–Keuls post hoc analysis revealed that the pre-GGA TBI group spent significantly more time with the novel object when compared with the vehicle TBI group (P<0.01). Significant differences were also observed between sham-injured and vehicle TBI groups (P<0.05).
Geranylgeranylacetone Pretreatment Improved Motor Function, Attenuated Depressive-Like Behavior, and Improved Overall Performance in the Composite Behavioral Score after Traumatic Brain Injury
Mice were tested on the beam walk immediately before sham surgery or TBI and again on PIDs 1, 3, 7, 14, 21, and 28. Repeated measures one-way ANOVA showed a significant effect of treatment (Figure 2A; F(2, 154)=131.7; P<0.0001) and day (F(6, 154)=50.67, P<0.0001), and Student–Newman–Keuls post hoc analysis showed significant differences between the sham-injured and vehicle TBI groups on day 1 (P<0.001), day 3 (P<0.001), day 7 (P<0.001), day 14 (P<0.001), day 21 (P<0.01), and day 28 (P<0.01). Notably, GGA pretreatment improved motor function after TBI with a significant difference between the pre-GGA TBI and vehicle TBI groups on day 21 (P<0.05).
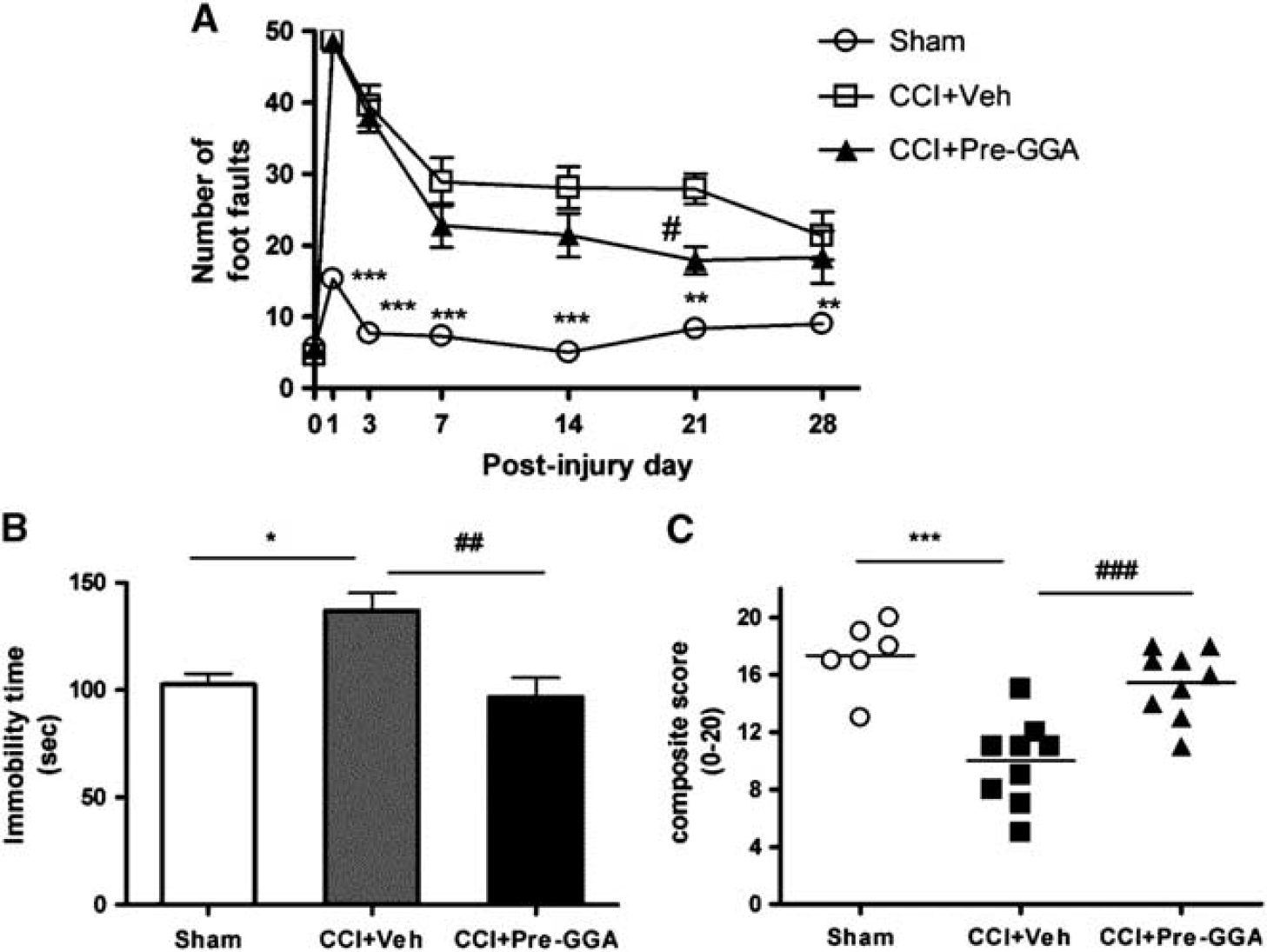
Geranylgeranylacetone (GGA) pretreatment improves motor function in beam walk test, reverses depressive-like behavior in the tail-suspension (TS) test, and improves overall behavioral outcomes in a composite score. (
To determine the long-term effects on depressive-like behavior after TBI, immobility time was tested in the TS test on PID 24. Increased immobility time in a 5-minute test is a measure of the degree of hopelessness and despair. One-way ANOVA analysis showed a significant effect of treatment in the TS test (Figure 2B; F(2, 24)=7.25; P<0.01), and Student–Newman–Keuls post hoc analysis showed significantly increased immobility times in the vehicle TBI group (P<0.05) when compared with the sham-injured group. We observed an improvement after GGA pretreatment, and there was a significant difference between the pre-GGA TBI and vehicle TBI groups (P<0.01).
A recently developed composite score (CS) 17 was used to comprehensively evaluate neurologic outcomes after TBI. We combined the motor (BW), cognitive (sMWM and rMWM), and affective (TS) test results into the CS. The score for each test was converted to an ordinal scale from 0 (most severe) to 5 for each behavioral test. The summation of these individual scores resulted in a CS ranging from 0 to 20. Sham-injured mice had a CS score of 18.17±0.48. As shown in Figure 2C, the CS showed significant separation between sham-injured, vehicle TBI, and pre-GGA TBI groups. Nonparametric Kruskal–Wallis analysis showed a significant difference among these groups (P<0.001). Q-Q plot analysis showed that the data displayed normality and equal variance; therefore, we analyzed these interval data using parametric statistics (one-way ANOVA; (F(2, 24)=24.87, P<0.0001). Student–Newman–Keuls post hoc analysis showed a significant difference between sham-injured and vehicle TBI groups (P<0.001). In addition, there was a significant difference between the pre-GGA TBI and vehicle TBI groups (P<0.001). Notably, the statistical differences in behavioral outcomes among sham-injured, vehicle TBI, and pre-GGA TBI groups were greater using the CS compared with any other behavioral task alone.
Geranylgeranylacetone Pretreatment Reduced Lesion Volume and Neuronal Loss in the Hippocampus, Cortex, and Thalamus after Traumatic Brain Injury
The TBI-induced lesion volume was quantified in cresyl violet–stained coronal brain sections from vehicle TBI and pre-GGA TBI groups at 28 days after injury by stereological methods. GGA pretreatment significantly reduced TBI-induced lesion volumes compared with the vehicle-treated group (Figure 3B; t(14)=1.77, P<0.05). Representative images from each group are shown in Figure 3A.
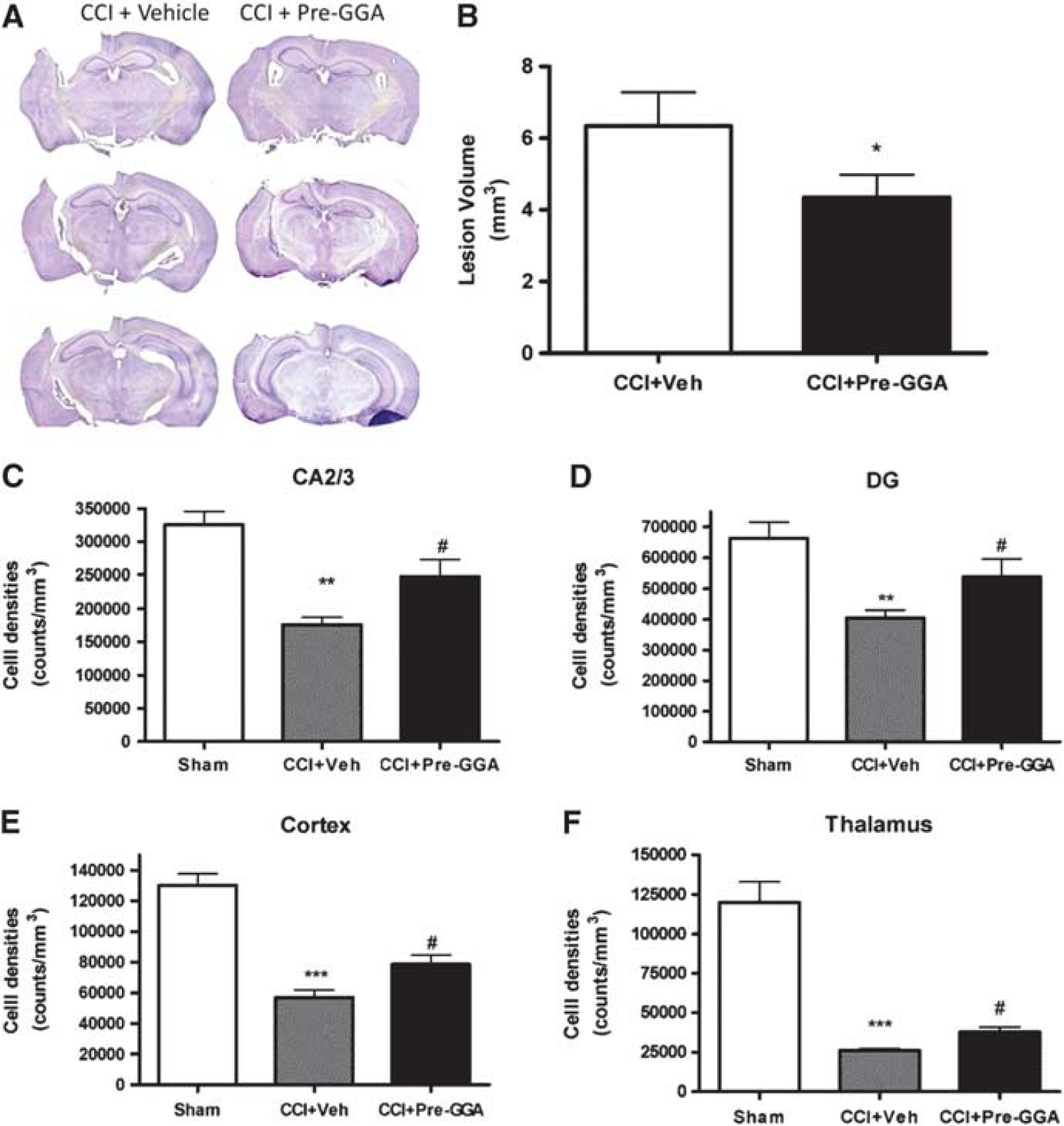
Geranylgeranylacetone (GGA) pretreatment reduces lesion size and attenuates neuronal cell loss in the Cornu Ammonis-2/3 (CA2/3) and dentate gyrus (DG) subregions of hippocampus, cortex, and thalamus after traumatic brain injury (TBI). Lesion volume was quantified using the Cavalieri method. Unbiased stereological assessment of lesion volume at 28 days after TBI was performed on cresyl violet–stained brain section. (
The TBI-induced neuronal loss in the CA1, CA2/3, and DG subregions of the hippocampus, cortex, and thalamus were quantified in cresyl violet–stained brain sections from sham-injured, vehicle TBI, and pre-GGA TBI groups 28 days after injury by stereological methods (Figures 3C–3F). One-way ANOVA analysis showed a significant effect of treatment in the CA2/3 (Figure 3C; F(2, 17)=14.46, P<0.001), DG (Figure 3C; F(2, 17)=7.49, P<0.01), cortex (Figure 3E; F(2, 17)=37.09, P<0.001), and thalamus (Figure 3F; F(2, 17)=44.52, P<0.001) neuronal densities, and Student–Newman–Keuls post hoc analysis showed a significant difference of neuronal densities between sham-injured and vehicle TBI groups in the CA2/3 (Figure 3C; P<0.01), DG (Figure 3D; P<0.01), cortex (Figure 3E; P<0.001), and thalamus (Figure 3F; P<0.001). Significant increased neuronal densities in the GGA pretreatment group were observed in the CA2/3 (P<0.05), DG (P<0.05), cortex (P<0.05), and thalamus (P<0.05) regions compared with vehicle TBI group in these regions. For the CA1 neuronal densities, although a significant effect of treatment was detected (Figure 3C; F(2, 17)=16.08, P<0.001), Student–Newman–Keuls post hoc test revealed a nonsignificant increase in neuronal density in the CA1 subregion in the pre-GGA TBI group (data not shown).
Geranylgeranylacetone Pretreatment Attenuated Microglial Activation after Traumatic Brain Injury
Traumatic brain injury results in microglial activation as shown by the transition of the microglial phenotypes from a ramified (resting form) cellular morphology predominant in noninjured brain to increasingly more activated phenotypes showing hypertrophic (moderate activation) or bushy (full activation) cellular morphologies. 20 Stereological assessment of microglial cell number and activation phenotype was performed in the ipsilateral cortex at 28 days after TBI. Traumatic brain injury in the vehicle group resulted in a significant decrease in ramified microglia (Figure 4A; F(2, 17)=6.08, P<0.01) and increase in hypertrophic (Figure 4B; F(2, 17)=14.45, P<0.001) and bushy microglia (Figure 4C; F(2, 17)=12.43, P<0.001) as compared with the sham-injured group. The GGA pretreatment significantly increased ramified (resting) microglia (Figure 4A; P<0.05) and reduced bushy (highly activated) microglia (Figure 4C; P<0.05) when compared with the vehicle TBI group. The GGA pretreatment did not significantly decrease the number of hypertrophic microglia (Figure 4B).
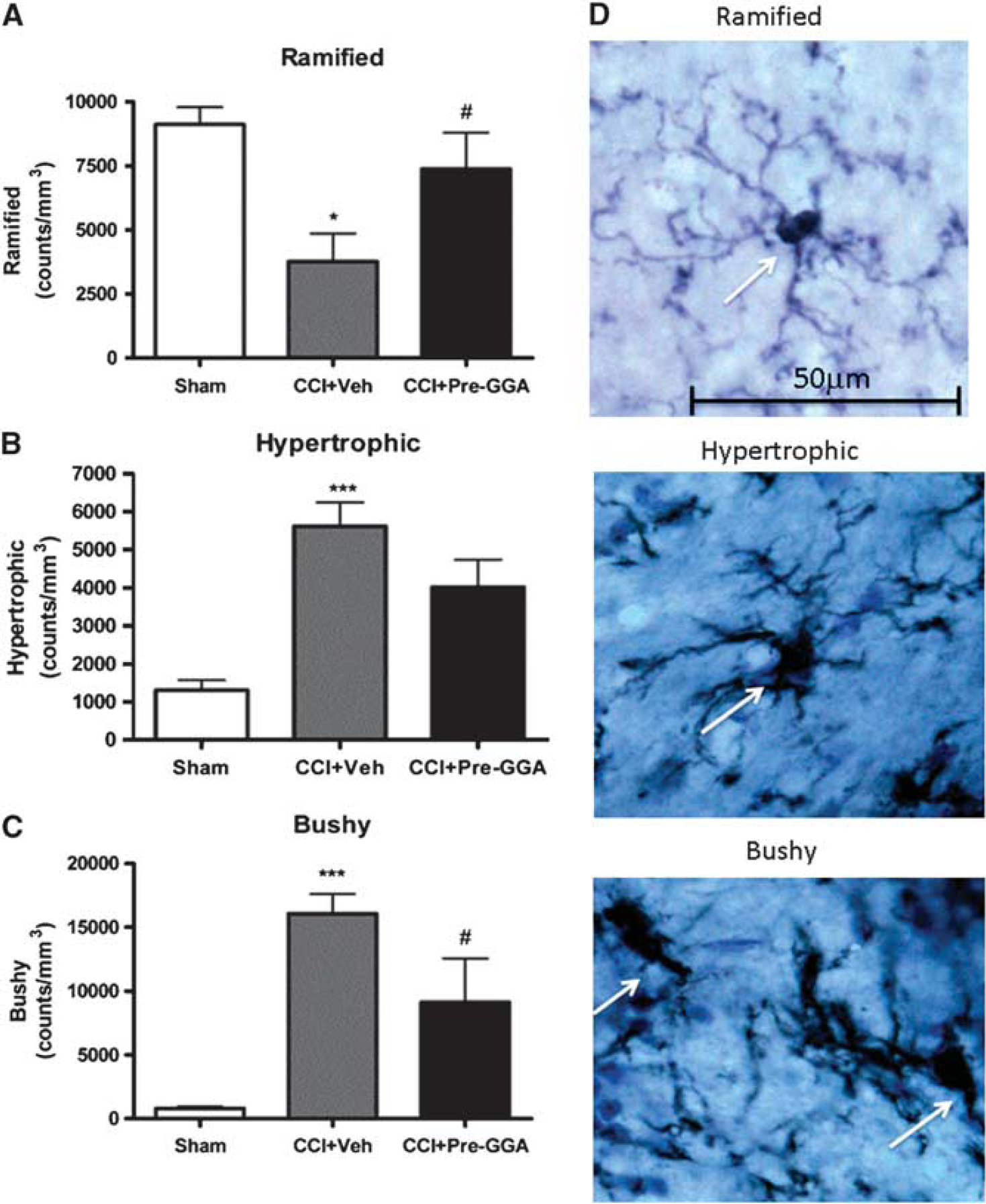
Geranylgeranylacetone (GGA) pretreatment attenuates microglia activation after traumatic brain injury (TBI). Unbiased stereological quantification of microglial cell number and activation status in the cortex at 28 days after TBI. Resting ramified (
Geranylgeranylacetone Pretreatment Increased HSP70 Expression and Attenuated α-Fodrin Cleavage after Traumatic Brain Injury
To determine whether GGA could increase the number of HSP70 cells in the ipsilateral cortex of TBI mice, HSP70 immunocytochemistry was performed on brain sections from animals killed at 6 hours after injury. The representative images showed that HSP70 expression was increased in vehicle TBI sections when compared with sham-injured sections and that HSP70 expression was further increased after GGA pretreatment (Figure 5A). One-way ANOVA analysis showed a significant effect of treatment in the percentage of HSP70+ cells analyzed by immunohistochemistry (Figure 5B; F(3, 13)=19.34, P<0.0001). Student–Newman–Keuls post hoc analysis showed a significant increase in the percentage of HSP70+ cells between sham-injured and vehicle TBI groups (P<0.01). Notably, GGA pretreatment significantly further increased the percentage of HSP70+ cells as compared with vehicle TBI group (P<0.05). No significant difference was found between GGA posttreatment and vehicle TBI groups. The effect of GGA pretreatment on TBI-induced caspase-dependent apoptosis was assessed by measuring fodrin cleavage products in the ipsilateral cortex from TBI mice at 6 hours after injury. Traumatic brain injury in the vehicle group significantly increased the 145/150 kDa fodrin cleavage product, a marker of calpain and caspase activity, 21 when compared with the sham-injured group (Figures 5C and 5D; F(3, 15)=18.69, P<0.001). The GGA pretreatment significantly reduced the level of 145/150 kDa fodrin cleavage products when compared with the vehicle TBI group (P<0.01).
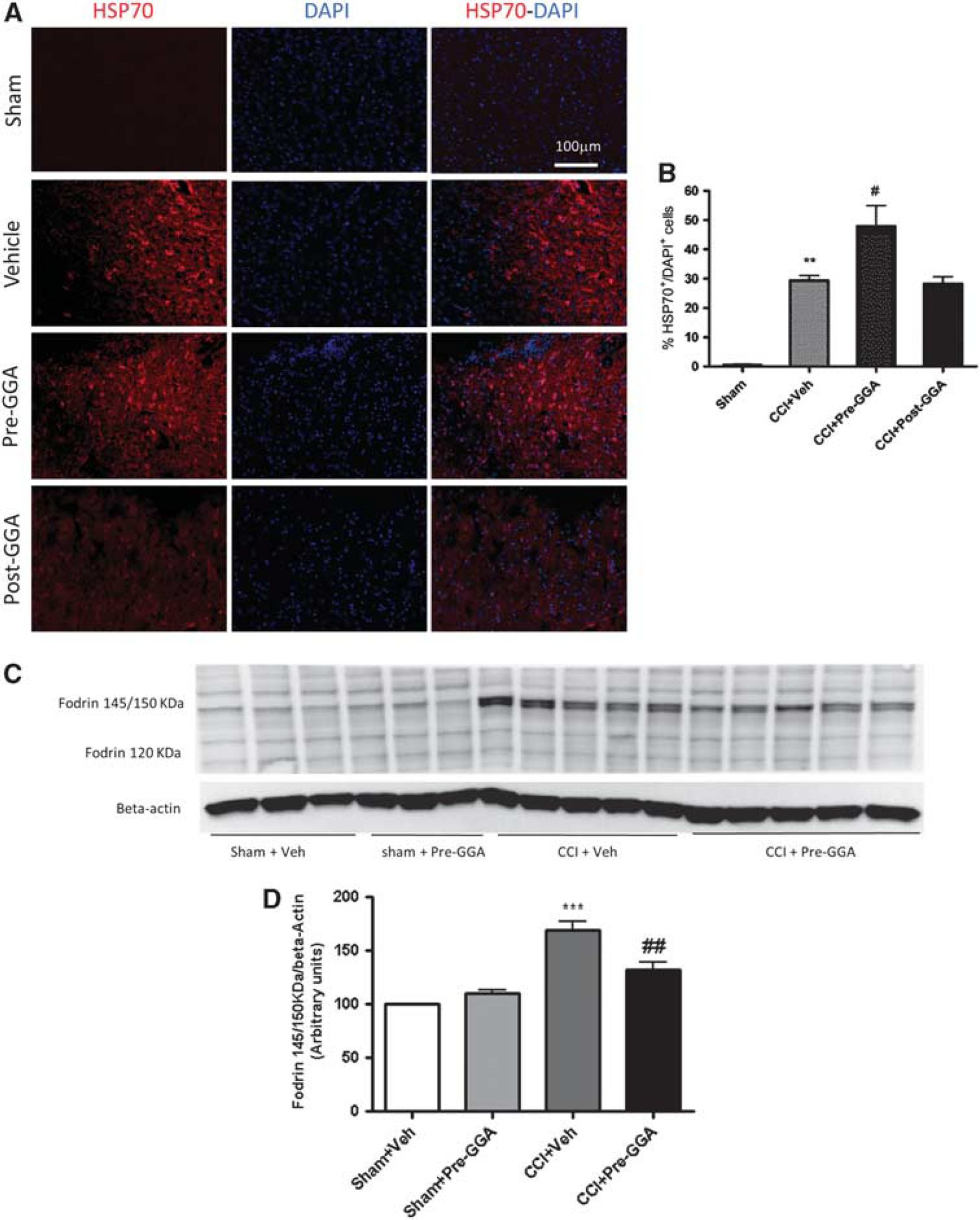
Geranylgeranylacetone (GGA) pretreatment upregulates heat-shock protein 70 (HSP70) expression and inhibits fodrin cleavage after traumatic brain injury (TBI). (
Geranylgeranylacetone Posttreatment Improved Motor Function Recovery after Traumatic Brain Injury
A separate group of animals was used to investigate the therapeutic potential of delayed administration of GGA after TBI. Geranylgeranylacetone (800 mg/kg) was delivered orally at 3 hours after injury, and the effect of GGA posttreatment on spatial learning and memory after TBI was examined using the sMWM test. In the escape latency, repeated measures one-way ANOVA showed a significant effect of treatment (Figure 6A; F(2, 60)=9.93; P<0.0001) and day (F(3, 60)=11.63, P<0.0001), but the treatment–day interaction failed to show significance. Student–Newman–Keuls post hoc analysis revealed a significant difference between the sham-injured and vehicle TBI groups on day 2 (P<0.01) and day 4 (P<0.01). No significant differences were observed between post-GGA TBI and vehicle TBI groups throughout. The sMWM probe trial was performed on PID 18. One-way ANOVA analysis failed to show a significant effect of treatment (Figure 6B; F(2, 17)=2.88; P>0.05). There were no significant differences in swim speeds across the groups (data not shown). In the search strategy analysis, GGA treatment improved the performance on this test and there was a significant difference between post-GGA TBI and vehicle TBI groups (Figure 6C, χ2=292.3; P<0.001). The GGA posttreatment failed to improve each of the outcomes in the rMWM when compared with the vehicle TBI group (Figures 6D–6F).
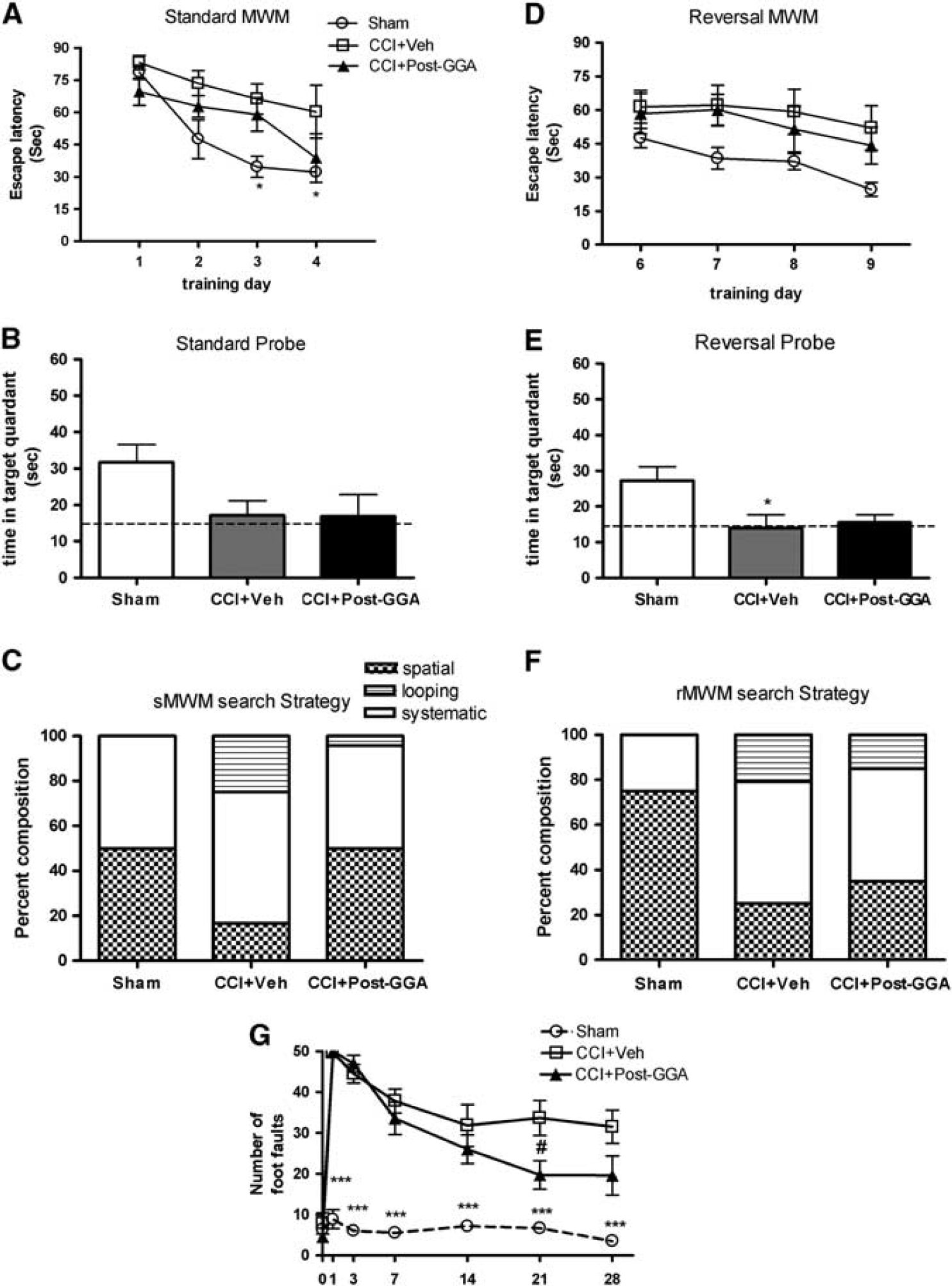
Geranylgeranylacetone (GGA) posttreatment improves motor function (beam walk test) after traumatic brain injury (TBI). (
The effect of GGA posttreatment on motor function recovery after TBI was assessed using the beam walk test, and repeated measures one-way ANOVA showed a significant effect of treatment (Figure 6D; F(2, 112)=177.00; P<0.0001) and day (F(6, 112)=35.83, P<0.0001). Student–Newman–Keuls post hoc analysis showed significant differences between the sham-injured and vehicle TBI groups on day 1 (P<0.001), day 3 (P<0.001), day 7 (P<0.001), day 14 (P<0.001), day 21 (P<0.001), and day 28 (P<0.001). Notably, GGA posttreatment improved motor function recovery after TBI, and there was a significant difference between post-GGA TBI and vehicle TBI groups on day 21 (P<0.05). The GGA posttreatment failed to improve TBI-induced nonspatial memory impairment as assessed by the NOR (data not shown). The GGA posttreatment also failed to reduce immobility times in the TS when compared with the vehicle TBI group (data not shown), indicating that GGA posttreatment did not have any beneficial effects on depressive-like behavior after TBI.
Geranylgeranylacetone Posttreatment Reduces Lesion Volume and Cortical Neuronal Loss after Traumatic Brain Injury
Traumatic brain injury–induced lesion volumes for each group were assessed at 28 days after injury using stereological methods. The GGA posttreatment resulted in a significant reduction in lesion size when compared with the vehicle TBI group (Figure 7B; t(11)=2.108, P<0.05). Representative images from each group are shown in Figure 7A. We also assessed TBI-induced neuronal loss in the CA1, CA2/3, and DG subregions of the hippocampus, cortex, and thalamus by stereological methods. One-way ANOVA analysis found that GGA posttreatment significantly attenuated neuronal densities in the cortex (Figure 7C; F(2, 17)=21.53, P<0.001). Student–Newman–Keuls post hoc analysis revealed a significant difference between the sham-injured and vehicle TBI groups (P<0.001), and between post-GGA TBI and vehicle TBI groups (P<0.05). No significant differences of neuronal densities were observed between vehicle TBI and GGA posttreatment group in CA1, CA2/3, DG subregions of the hippocampus, as well as thalamus (data not shown).
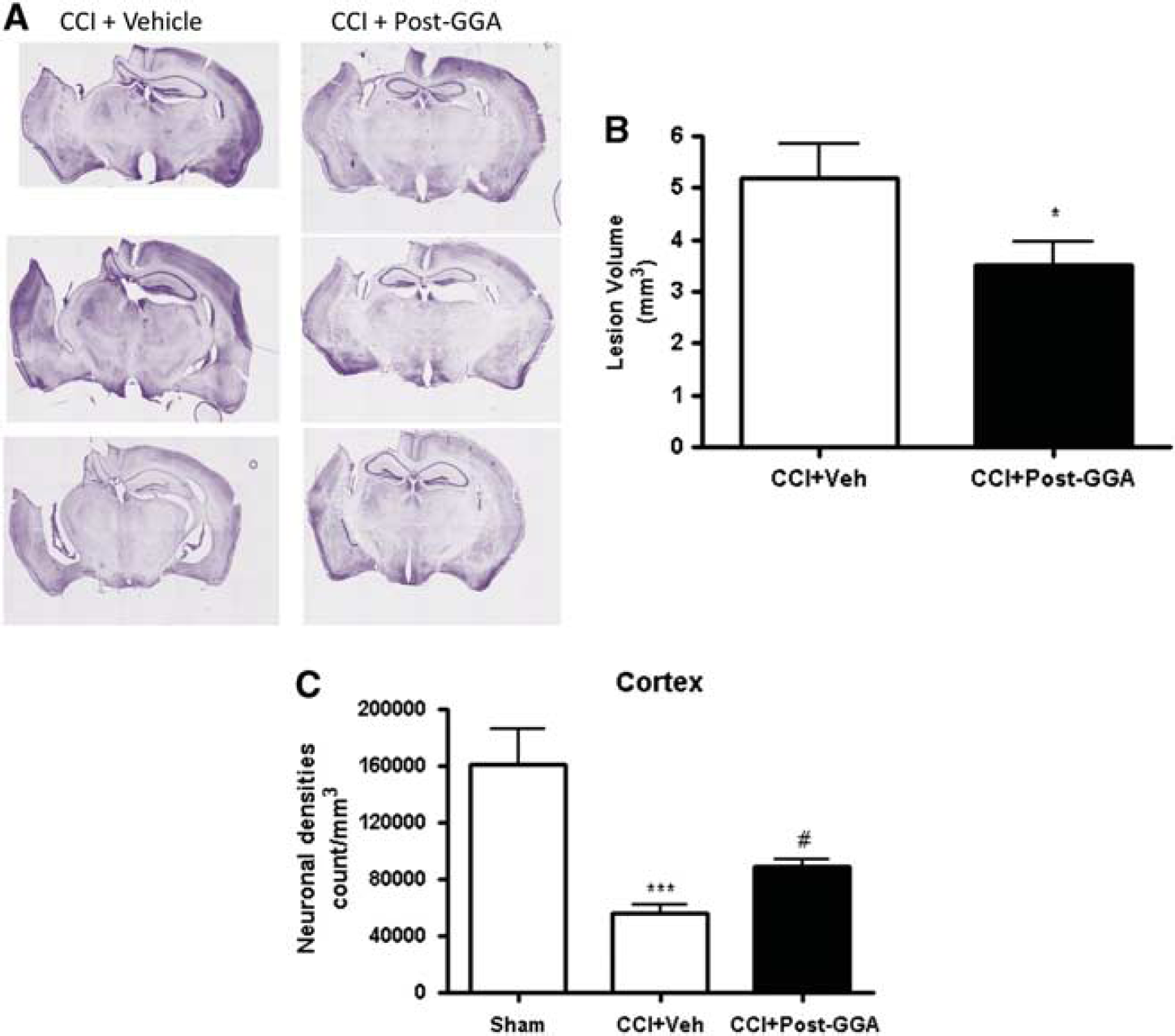
Geranylgeranylacetone (GGA) posttreatment reduces cortical lesion size and attenuates neuronal cell loss in the cortex after traumatic brain injury (TBI). Unbiased stereological assessment of lesion volume at 28 days after TBI was performed on cresyl violet–stained brain sections. (
DISCUSSION
Upregulation of HSP70 expression is a key stress response of the injured brain after both experimental and clinical TBI. 22 The evidence suggests that the temporal pattern of HSP70 expression is similar in human and rodent TBI, and these original studies hypothesized that HSP70 upregulation served as an important endogenous neuroprotective response after TBI. 22 Despite these key primary observations, few studies have focused on HSP70 overexpression as a therapeutic strategy for TBI. Our study is the first to explore the neuroprotective effects of a clinically tested pharmacological inducer of HSP70 expression (GGA) in a clinically relevant experimental TBI model in mice.
Geranylgeranylacetone is centrally acting after oral administration and has been used in the clinic as a treatment for peptic ulcer for many years without serious side effects. 15 This acyclic polyisoprenoid is a nontoxic HSP70 inducer that can augment the heat-shock response after stress, and because it is a lipid-soluble molecule it readily passes the blood–brain barrier to exert neuroprotective effects. 23 The ability of GGA to enhance the endogenous insult-induced HSP70 expression is faster and more robust than its intrinsic HSP70-inducing power in noninjured cells. 15 Pretreatment with the same GGA regimen used in this study showed significant neuroprotective activity after ischemic brain injury. For example, GGA pretreatment significantly attenuated neuronal death in the hippocampus after transient cerebral ischemia,18, 24 and reduced infract volumes and neurologic deficits after permanent middle cerebral artery occlusion.24, 25 To achieve optimal neuroprotective effects in transient cerebral ischemia, GGA should be administered 48 hours before the ischemic insult, and its effectiveness declines or disappears when delivered before or after that time point, respectively. 18 Other studies have suggested that the GGA treatment requirements are model dependent. The GGA pretreatment as little as 1 hour before injury was neuroprotective in permanent focal cerebral ischemia, increasing neuronal HSP70 expression and reducing infarct volumes; however, the neuroprotective effect was lost when GGA was administered at 1 hour after ischemia. 15 Furthermore, repeated doses of GGA (both before and after insult) are required to reduce brain edema, improve functional recovery, as well as reduce neuroinflammation and neuronal cell death in an experimental intracerebral hemorrhage (ICH) model. 13 Geranylgeranylacetone may also exert neuroprotective effects through upregulation/activation of other major HSPs, including HSP110, HSP90, and HSP40, as well as through modulation of HSP70-related neuroprotective pathways involving molecules such as thioredoxin-1, PKCs, Akt, and STAT3. 24
The main cellular target of GGA is HSP70 itself, and this interaction results in removal of HSP70 from the HSP70–HSF1 complex, freeing HSF1 to translocate to the nucleus to further enhance HSP70 transcription. 26 The key role of the HSP70 pathway as a mediator of GGA therapeutic effects was shown in models of transient renal ischemia (acute kidney injury) and experimental retinal detachment where GGA neuroprotective effects are attenuated by in HSP70−/− mice. 27
Early studies of GGA effects in the gastric mucosa suggested that cytoprotection is not only because of HSP70 overexpression, but may also involve additional mechanisms, including inhibition of neutrophil infiltration and attenuation of oxidative injury. 28 Similarly, GGA-dependent neuroprotection may also involve alternative mechanisms including HSF-1-mediated effects such as upregulation of other HSPs, as well as attenuation of tumor necrosis factor-α, interleukin (IL)-1β, and IL-6 inflammatory cytokine expression and secondary neurotoxicity via direct interaction with key transcription factors. 29 The GGA treatment before or after experimental ICH attenuated proinflammatory gene expression, including IL-6. 13
To examine the neuroprotective effects of GGA in experimental TBI, we used the well-established CCI model that produces a focal brain injury, resulting in significant neuronal cell loss and neuroinflammation that contributes to significant long-term neurologic dysfunction. 17 The CCI model has been extensively used to investigate the molecular and cellular mechanisms underlying secondary injury progression, and to develop novel therapeutic approaches for TBI. 30 Cognitive, affective, and motor functional deficits are common after clinical TBI. Here, we present a comprehensive assessment of the effect of a HSP70-inducing treatment on multiple neurologic outcomes using the MWM and NOR tests for cognition, the TS test for affective changes, and the beam walk test for motor function.
The sMWM, considered to be a test of anterograde spatial learning and memory, has been commonly used to examine learning and memory deficits in rodents after TBI. In this study, TBI mice showed significant increased latency to find the platform on day 4, and GGA pretreatment significantly reduced escape latency, suggesting that GGA pretreatment attenuated spatial memory deficit induced by TBI. In the probe test, GGA pretreatment showed improved memory retention. As the MWM results may not reflect pure cognitive deficits, but may be confounded by motor and affective impairment, we examined the search strategy used by TBI mice to locate the hidden platform. 17 The GGA-pretreated TBI mice used more spatial and less looping strategy than vehicle TBI mice in sMWM search strategy. These data showed that a pure cognitive deficit was observed after TBI and that GGA pretreatment alleviated this deficit. The NOR task evaluates nonspatial hippocampal memory, 17 and is therefore complementary to spatial memory assessment by the MWM test. This test is widely used to assess cognitive function in TBI animals. 31 In this study, GGA-pretreated TBI mice spent significantly more time with the novel than familiar object in the choice phase of the test, which was in stark contrast to the vehicle-treated TBI mice. These data showed that GGA pretreatment attenuated nonspatial memory deficits induced by TBI.
We also assessed long-term motor function recovery after TBI up to 28 days after injury using the beam walk test, and observed that TBI animals showed motor function deficits when compared with sham-injured controls. Importantly, GGA pretreatment significantly improved motor function recovery after injury when compared with the vehicle-treated TBI mice.
Depression is a common affective symptom after TBI. Even mild-to-moderate TBI can lead to development of major depression. 32 In this study, the TS test revealed that vehicle-treated TBI mice had depressive-like behaviors. Notably, GGA pretreatment significantly attenuated these changes in TBI mice.
Combinations of behavioral tests have been used in experimental brain and spinal cord injury models to increase sensitivity for discriminating between different injury mechanisms and to characterize the response to therapeutic interventions. 33 We have previously developed a behavioral composite score that combines multiple cognitive, affective, and sensorimotor functional outcomes. 17 This score provides an overall assessment of neurologic function and enhances the ability to observe significant difference between groups. In this study, this score showed a more robust separation between vehicle- and GGA-treated TBI groups compared with any individual behavioral test alone, underscoring the significance of the GGA-mediated neuroprotective effects.
This study used unbiased stereological techniques to generate a quantitative assessment of neuronal loss in several key brain regions at 28 days after injury. We have previously shown that neuronal cell loss in the cortex and DG was relatively modest in the first 24 hours and was followed by a significant and progressive neuronal cell death at later time points, therefore, highlighting the importance of examining late time points to obtain an accurate assessment of neuronal loss. 19 The GGA pretreatment significantly reduced delayed neuronal cell death in the CA2/3 and DG subregions of hippocampus, cortex, and thalamus. As hippocampus is one of the key centers responsible for the cognitive outcomes probed by the sMWM and NOR, 34 the GGA-dependent sparring of hippocampal neurons may explain the observed improvements in cognitive function in these tests. The cortex and thalamus are responsible for relaying sensory and motor signals between a variety of cerebral cortex and subcortical areas, 35 resulting in the coordination of sensorimotor function. The beam walk test used in this study is an excellent discriminator of sensorimotor function after TBI. In a rat TBI model, it was shown that damage in thalamus occurred after cortical injury. 36 Our observations are consistent with the hypothesis that GAA-mediated sparring of cortical and thalamic nuclei, as well as reduction of cortical lesion size, is responsible at least in part for attenuation of the sensorimotor deficits in GGA pretreatment TBI mice in the beam walk test. Because of the fact that multiple brain regions may influence functional outcomes, it is possible that the effects on other brain areas may in part contribute to the neuroprotective effects of GGA on the cognitive and sensorimotor tests.
The mechanisms of neuronal cell death after TBI may involve the activation of apoptotic pathways. Fodrin is a high-molecular-weight cytoskeletal protein that undergoes degradation catalyzed by activated caspases and other proteases during apoptosis, generating 145/150 kDa fragments. 33 In this study, we found that levels of 145/150 kDa fodrin cleavage products were increased 6 hours after TBI, and that GGA pretreatment significantly attenuated this increase. At the same time point, GGA pretreatment significantly increased the percentage of HSP70+ cells in the injured cortex, consistent with the hypothesis that GGA pretreatment reduced TBI-induced apoptotic pathways through HSP70 upregulation.
Chronic neuroinflammation and microglial activation after TBI is a key secondary injury mechanism that contributes to neuronal cell death and long-term neurologic dysfunction. 37 In a series of studies we characterized posttraumatic microglial activation based on cellular phenotypic changes that correspond to progressively increased microglial activation status: ramified, hypertrophic, and bushy.19, 20 Our quantitative analysis revealed that GGA pretreatment significantly reduced the numbers of bushy microglia cell morphologies as compared with vehicle-treated TBI mice. These data suggest that HSP70 induction via GGA pretreatment can efficiently attenuate TBI-induced chronic microglial activation, which may explain, in part, its neuroprotective actions.
Using a smaller group of animals, we also performed a proof-of-principle study to examine the effect of delayed oral administration of GGA after injury because it is a more clinically relevant treatment protocol. Even when GGA was administrated 3 hours after injury, it significantly improved performance on key motor and cognitive tests, and significantly attenuated TBI-induced lesion volumes and neuronal cell loss in the cortex. Overall, the neuroprotective effects of a single dose of GGA administered 3 hours after injury were more modest compared with GGA pretreatment, at least in part because of the delay in HSP70 upregulation. Further optimization of the dose and postinjury administration protocol (repeat dosing) may be important to improve outcomes further in the posttreatment protocol.
One of the limitations of this study is the large dose of GGA (800 mg/kg) used that is much higher than the clinical dose required to treat gastric ulcer (150 mg, t.i.d.). 38 However, it is not completely surprising that the dose required to achieve brain penetration is significantly higher than the dose that is effective locally on the gastric mucosa. Furthermore, the median lethal dose (LD50) of GGA is 15,000 mg/kg after oral administration in rodents. 39 The dose used in this study is ∼5% of the LD50 and no toxic and side effects were observed in our study. Another potential limitation is that our study cannot determine whether the observed reduction of neuroinflammation is because of direct effects on microglia versus indirect effects in response to attenuation in neuronal cell death.
Taken together, our results show that a single oral dose of GGA administrated 48 hours before TBI robustly attenuated cognitive, affective, and sensorimotor function deficits. Using quantitative stereological methods we showed that GGA pretreatment reduced cortical lesion volume, neuronal cell loss, and neurodegeneration in the CA2/3 and DG subregions of hippocampus, cortex, and thalamus, and attenuated microglial activation in the cortex. Notably, GGA also showed significant, although more modest, neuroprotective effects using a more clinically relevant postinjury administration protocol. Thus, HSP70 inducers, such as GGA, may be a promising pharmacotherapy for TBI.
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors thank Titilola Akintola, Stephanie Custer, and Shruti Kabadi for expert technical assistance.
