Abstract
Dysregulated protein synthesis is thought to be a core phenotype of fragile X syndrome (FXS). In a mouse model (
INTRODUCTION
Fragile X syndrome (FXS) is the most common inherited form of intellectual disability and a recognized monogenic cause of autism. The gene responsible,
Based on findings in the FXS mouse model, we hypothesized that rCPS are elevated in FXS subjects. We used the quantitative L-[1-
11
C]leucine positron emission tomography (PET) method12–14 to measure rCPS in FXS subjects. As most subjects with FXS could not remain motionless in the PET scanner for 2 hours while tracer was injected intravenously and arterial blood samples were drawn, PET studies of FXS subjects were conducted during deep sedation with propofol, a potent hypnotic that has no effect on rCPS in normal subjects.
15
Contrary to our hypothesis, we found as reported herein that rCPS were not increased in FXS subjects, but rather
MATERIALS AND METHODS
Human Studies
All procedures on human subjects were performed as described in a protocol approved by the National Institutes of Health Combined Neurosciences Institutional Review Board, the National Institutes of Health Radioactive Drug Research Committee, and the National Institutes of Health Radiation Safety Committee. All subjects or, in the case of subjects with FXS, their legal guardians gave written informed consent before study enrollment.
Positron emission tomography studies were performed on the ECAT High Resolution Research Tomograph (CPS Innovations, Knoxville, TN, USA). Data were acquired in list mode and reconstructed using the motion-compensated 3D ordinary Poisson ordered subset expectation maximum algorithm. 20 Images were reconstructed as 42 frames of data (16 × 15 seconds, 4 × 30 seconds, 4 × 60 seconds, 4 × 150 seconds, 14 × 300 seconds). All activities were decay corrected to the time of radiotracer injection. 3D frames of data were reconstructed to 207 slices 1.22 mm thick (no interleaved slices) with a pixel size of 1.21 × 1.21 mm. The full width at half maximum of the High Resolution Research Tomograph at the center of the field of view is ~2.5 mm, the transverse and axial dimensions of the field of view are 31.2 and 25.2 cm, respectively. 21
The kinetic model for the behavior of leucine in brain has been described in our previous work.12,23 The parameters of the model were estimated for each voxel in the whole brain volume by means of the Basis Function Method of Tomasi
Animal Studies
Male WT and
We used the autoradiographic L-[1-
14
C]leucine method for the
The experimental period was initiated by an intravenous pulse injection of 3.7 MBq/kg of L-[1- 14 C]leucine (specific activity, 2.22 GBq/mmol, Moravek Biochemicals and Radiochemicals, Brea, CA, USA). Timed arterial samples were collected during the following 60 minutes for determination of the time courses of plasma concentrations of leucine and [ 14 C]leucine. Labeled and unlabeled leucine concentrations in the acid-soluble fractions of arterial plasma were assayed by liquid scintillation counting and amino-acid analysis, respectively. At the end of the experimental interval, brains were removed and frozen, and serial sections, 20 μm in thickness, were prepared for quantitative autoradiography by means of a Leica cryostat (Leica Microsystems, Inc., Buffalo Grove, IL, USA). Sections were mounted on gelatin-coated slides, fixed and washed in 10% formalin, and exposed to Ektascan B/RA film (Eastman Kodak, Rochester, NY, USA) along with calibrated [ 14 C]methylmethacrylate standards as described previously. 10 Autoradiograms were digitized (MCID Analysis, Interfocus Imaging Ltd, Linton, Cambridge, UK), the concentration of 14 C in each ROI was determined, and rCPS were calculated by means of the operational equation of the method. 24 The value of lambda in the equation was 0.603 (ref. 10). Brain regions were identified by reference to a mouse brain atlas. 25
The rCPS data were analyzed by 3-way repeated-measures analysis of variance with genotype (WT, KO) and treatment (vehicle, propofol) as between-subjects factors and brain region as a within-subjects factor.
RESULTS
Human Studies
Control subjects were recruited from local universities and the NIH. Healthy volunteer and FXS subjects were well matched with respect to age and body weight (Table 1). Presence of a full mutation allele was confirmed in the FXS subjects by analysis of the CGG repeat length; all had greater than 200 CGG repeats. In control subjects repeat lengths ranged from 20 to 32 with a mean ± s.e.m. of 27 ± 1. Control subjects (
Subject population, psychological testing, and physiologic variables measured during PET studies of propofol-sedated subjects
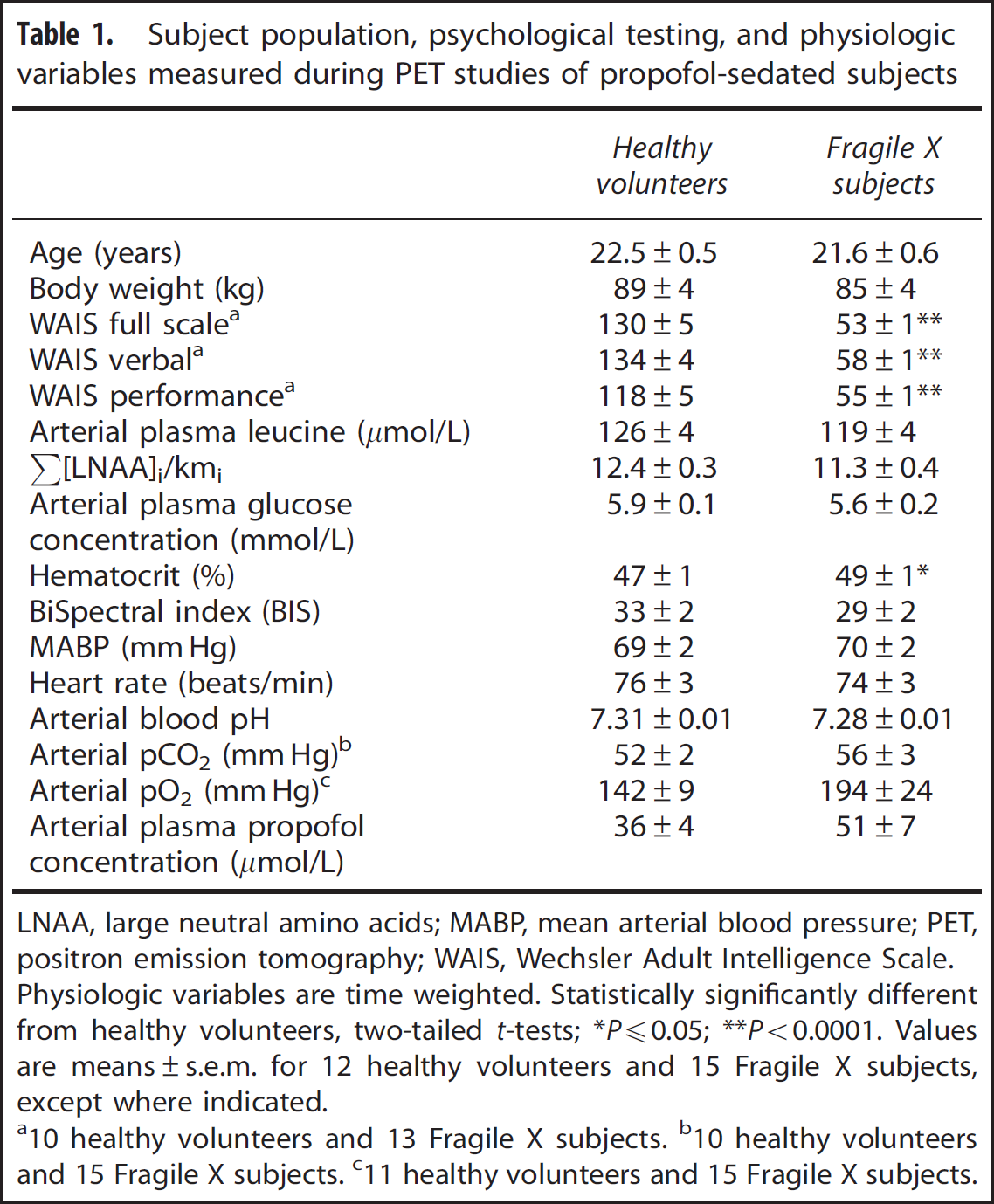
LNAA, large neutral amino acids; MABP, mean arterial blood pressure; PET, positron emission tomography; WAIS, Wechsler Adult Intelligence Scale.
Physiologic variables are time weighted.
Statistically significantly different from healthy volunteers, two-tailed
Values are means±s.e.m. for 12 healthy volunteers and 15 Fragile X subjects, except where indicated.
10 healthy volunteers and 13 Fragile X subjects.
10 healthy volunteers and 15 Fragile X subjects.
11 healthy volunteers and 15 Fragile X subjects.
Physiologic variables were averaged across the PET scan duration (Table 1). Values of the BIS indicate that, on average, subjects in both groups were deeply sedated and levels of sedation were similar in both groups. The mean time-weighted average arterial plasma concentration of propofol was 41% higher in the fragile X subjects, but the difference was not statistically significant (Table 1). Except for hematocrit which was slightly, but statistically significantly, higher in the fragile X subjects, values of other variables were similar between groups. Plasma leucine concentrations measured over the 90-minute PET scan remained fairly constant. Coefficient of variation for the 12 studies of healthy volunteers ranged from 2.0% to 6.9% (mean 4.6%) and for the 15 fragile X subjects ranged from 1.4% to 7.9% (mean 3.5%). Time-weighted average leucine concentrations were similar in both groups. The concentrations of large neutral amino acids that comprise the primary competitors for entry into brain via the L-amino acid transporter (methionine, valine, isoleucine, leucine, tyrosine, and phenylalanine) were also measured and the sums of the average concentration of each of the amino acids weighted by its
The following ROIs were drawn on MRIs from each subject: whole brain, cerebellum, frontal cortex, parietal cortex, thalamus, hippocampus, amygdala, hypothalamus, caudate nucleus, putamen, and corona radiata. The corona radiata was sampled, but other ROIs encompassed entire regions. We compared volumes of whole brain (not including the ventricles) and the 9 regions that were completely outlined in the 12 healthy volunteers and 15 FXS subjects (Table 2). Results show statistically significant differences in three regions. The volumes of putamen (–21.3%,
Region-of-interest volume and regional rates of cerebral protein synthesis
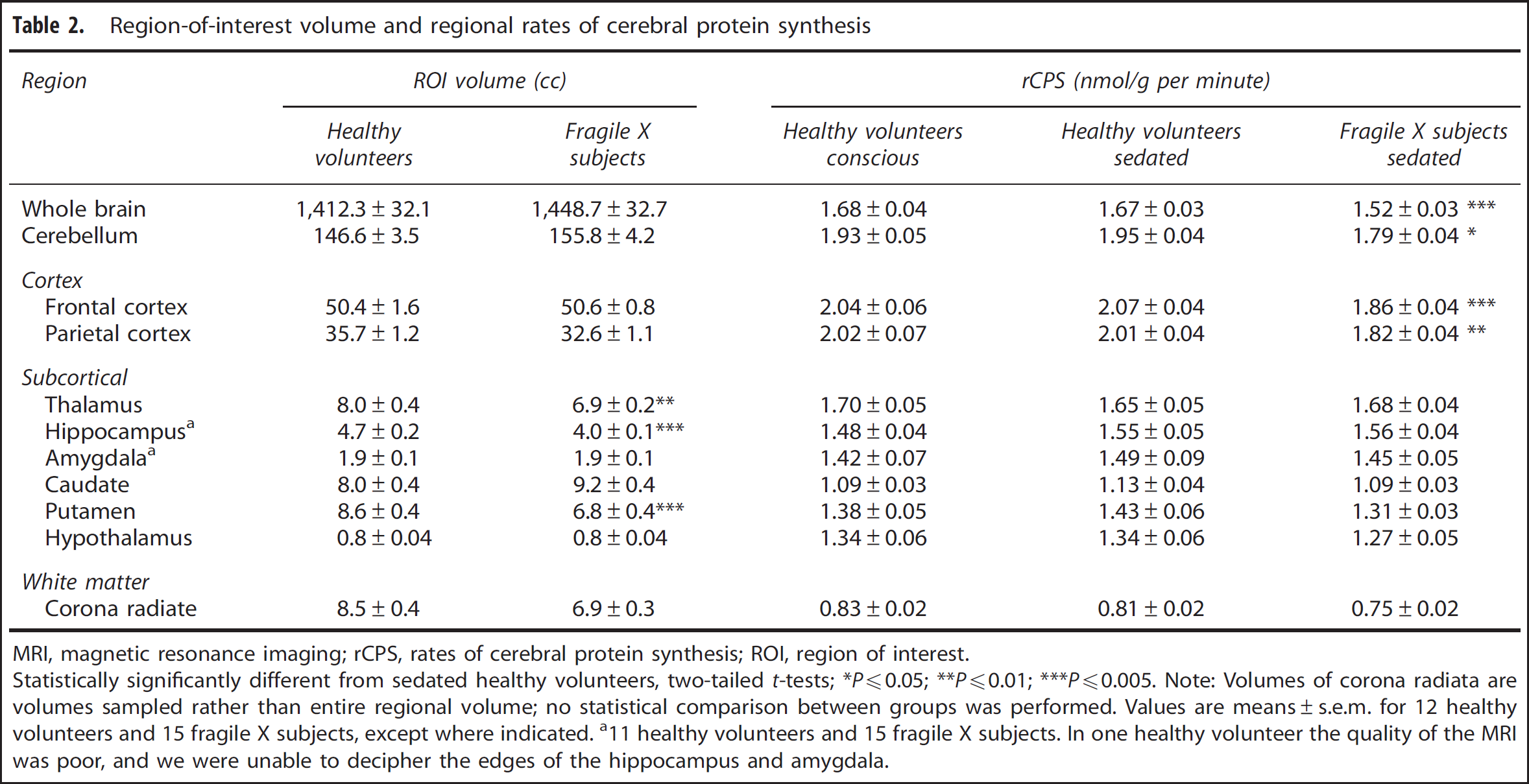
MRI, magnetic resonance imaging; rCPS, rates of cerebral protein synthesis; ROI, region of interest.
Statistically significantly different from sedated healthy volunteers, two-tailed
Note: Volumes of corona radiata are volumes sampled rather than entire regional volume; no statistical comparison between groups was performed.
Values are means±s.e.m. for 12 healthy volunteers and 15 fragile X subjects, except where indicated.
11 healthy volunteers and 15 fragile X subjects. In one healthy volunteer the quality of the MRI was poor, and we were unable to decipher the edges of the hippocampus and amygdala.
Rates of cerebral protein synthesis were measured with low intersubject variability: the coefficient of variation was 6% to 12% except in the very small (< 5 cc) subcortical regions where it was 9% to 20% (Table 2). Figure 1 illustrates the distribution of individual rCPS measurements in whole brain, cerebellum, frontal cortex, and thalamus; one notes the low variability in the measurements and the differences in mean rCPS between the sedated groups.
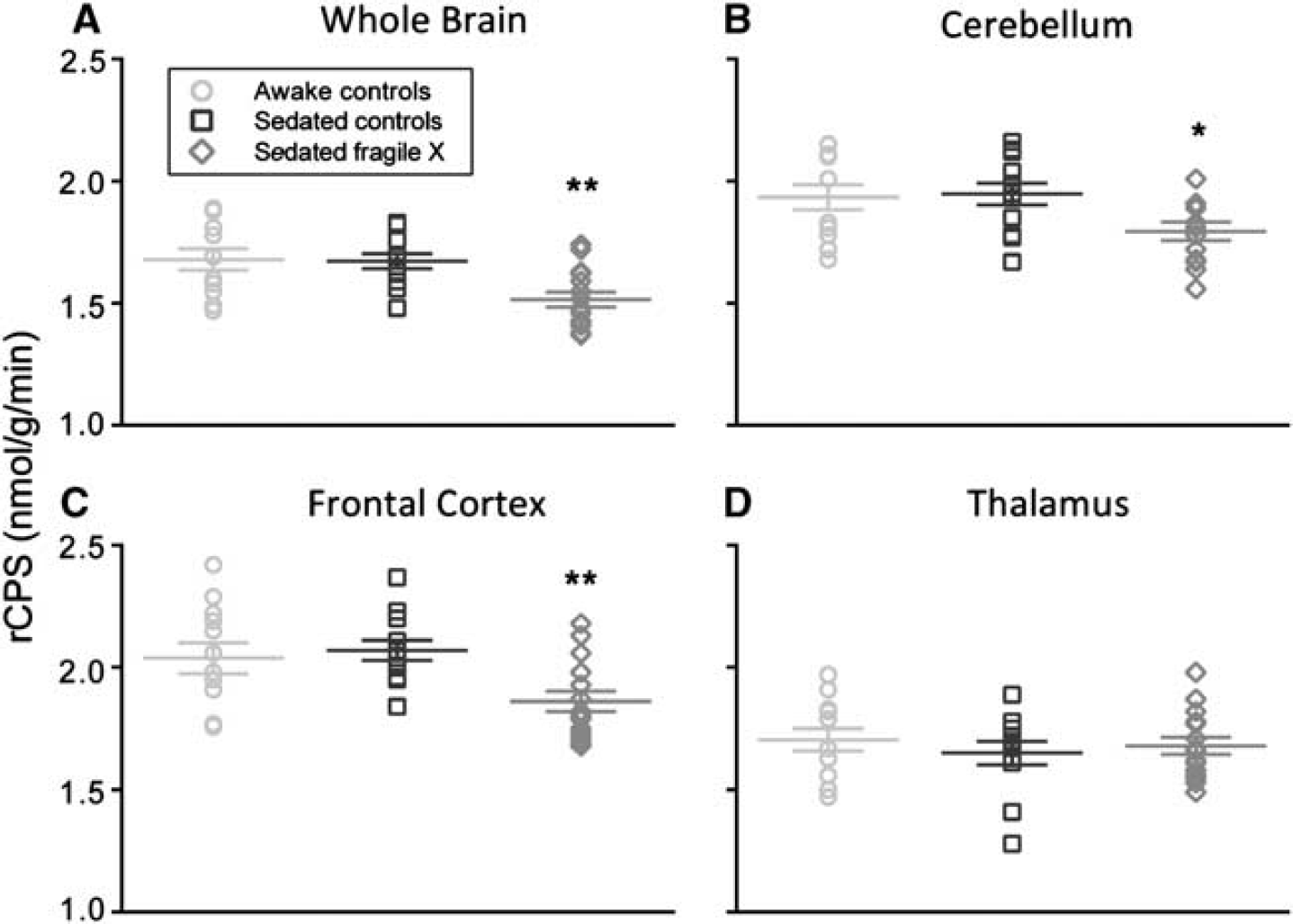
Effects of propofol sedation on rates of cerebral protein synthesis (rCPS) in (
Each healthy volunteer was studied twice with the [
11
C]leucine PET method, once awake and once under deep propofol sedation. We compared rCPS in the two states with paired
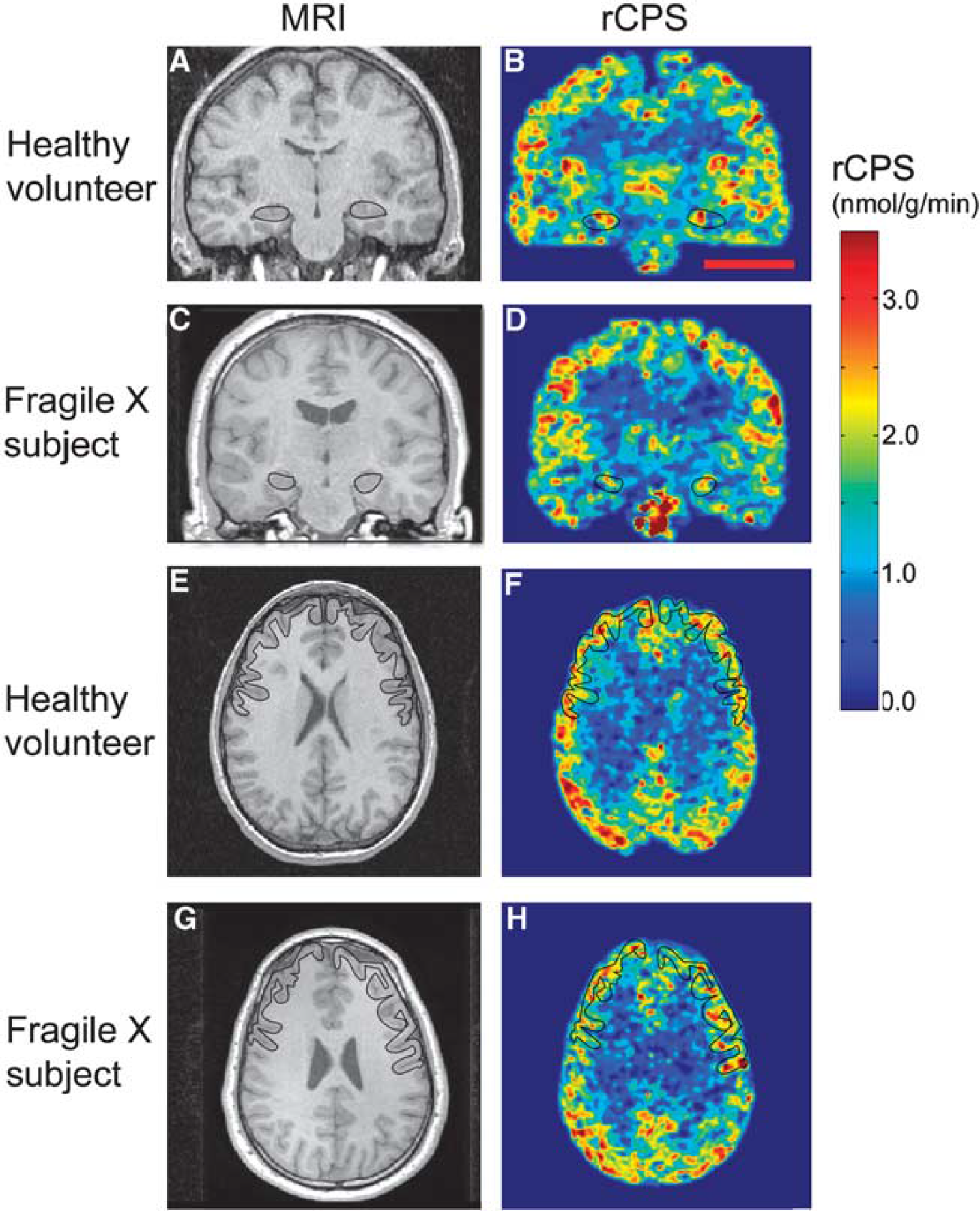
Parametric images of rates of cerebral protein synthesis (rCPS) in a typical sedated healthy volunteer and sedated fragile X subject. Magnetic resonance images (MRIs) on the left correspond to rCPS positron emission tomography (PET) images on the right. All PET images were resliced to match the subject's MRI, and kinetic model parameters were estimated voxelwise by the Basis Function Method (BFM). Resultant parametric images were smoothed with a 3D Gaussian filter (kernel 2 mm full width at half maximum (FWHM)). Parametric images are color coded for rCPS according to the colorbar on the right. Images at the level of the hippocampus (
Animal Studies
The findings in human subjects were contrary to our hypothesis that rCPS would be higher in FXS subjects compared with age-matched healthy volunteers. We considered the possibility that propofol could have a disparate effect on rCPS in FXS subjects thereby masking a baseline elevation in rCPS. We examined this possibility in the
Effects of propofol sedation on physiologic variables in
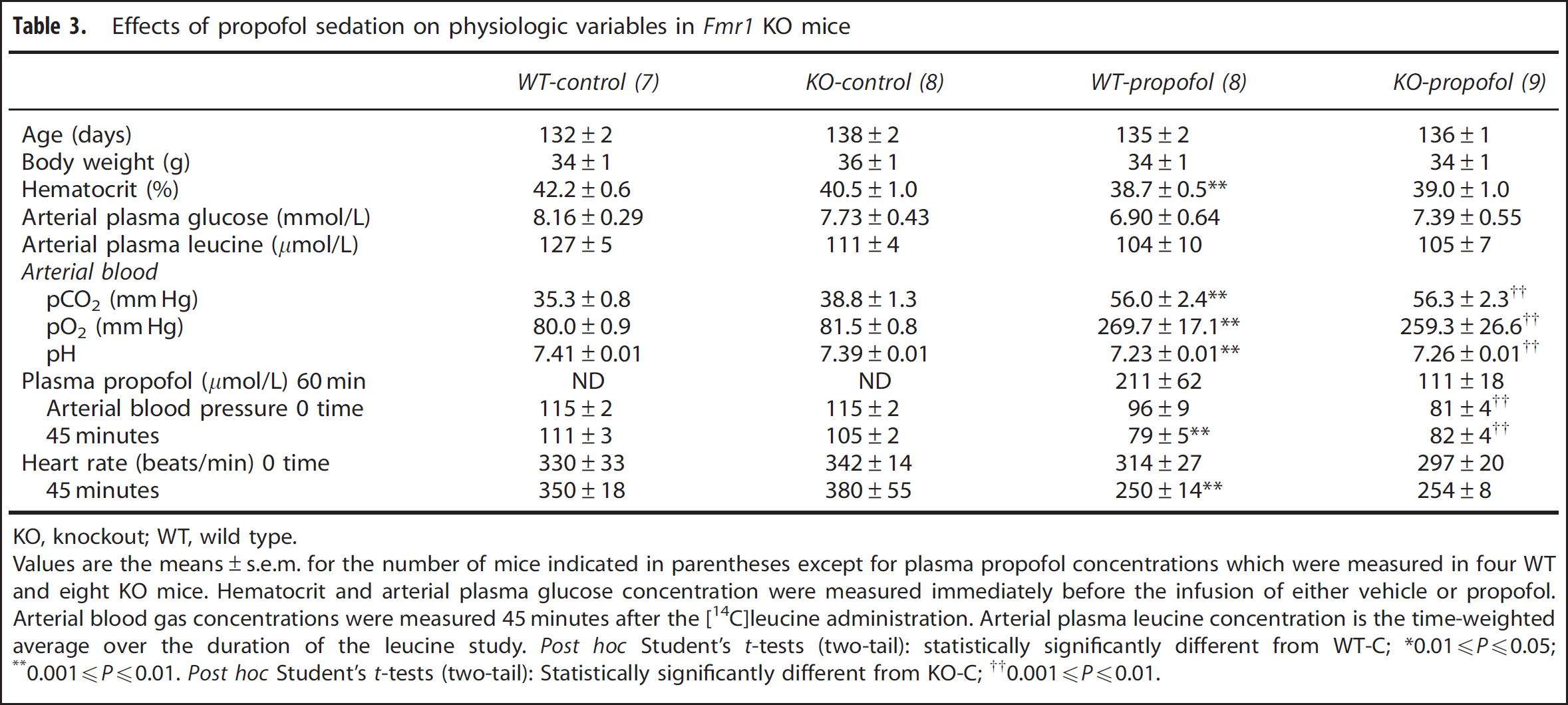
KO, knockout; WT, wild type.
Values are the means±s.e.m. for the number of mice indicated in parentheses except for plasma propofol concentrations which were measured in four WT and eight KO mice. Hematocrit and arterial plasma glucose concentration were measured immediately before the infusion of either vehicle or propofol. Arterial blood gas concentrations were measured 45 minutes after the [14C]leucine administration. Arterial plasma leucine concentration is the time-weighted average over the duration of the leucine study.
0.01
0.001
0.001
We confirm in the vehicle-treated mice our previous finding that rCPS were increased in the absence of FMRP in selective brain regions (Table 4). Statistically significantly (
Effects of propofol sedation on rCPS in
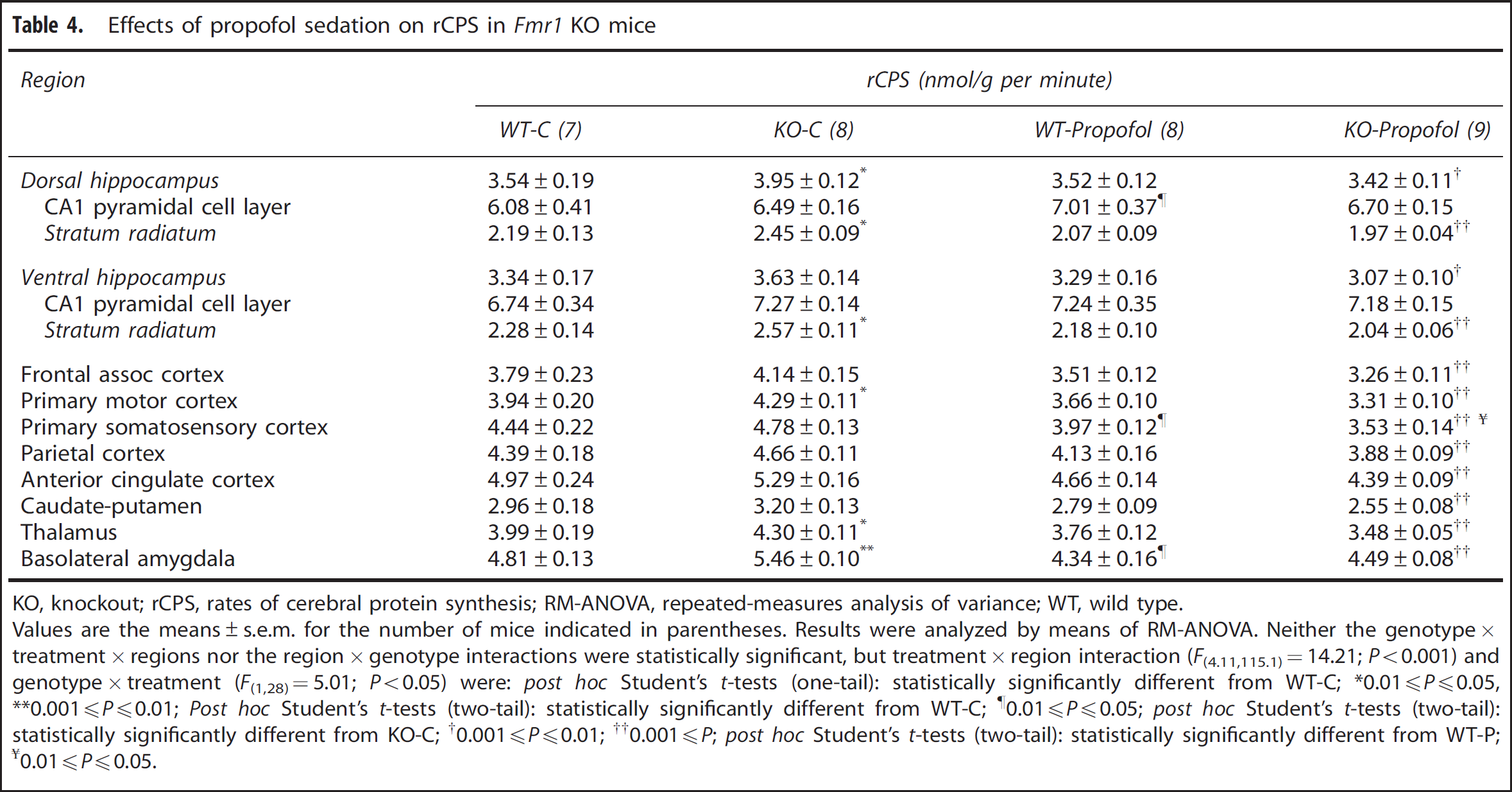
KO, knockout; rCPS, rates of cerebral protein synthesis; RM-ANOVA, repeated-measures analysis of variance; WT, wild type.
Values are the means±s.e.m. for the number of mice indicated in parentheses. Results were analyzed by means of RM-ANOVA. Neither the genotype × treatment × regions nor the region × genotype interactions were statistically significant, but treatment × region interaction (
0.01
0.001
0.01
0.001
0.001
0.01
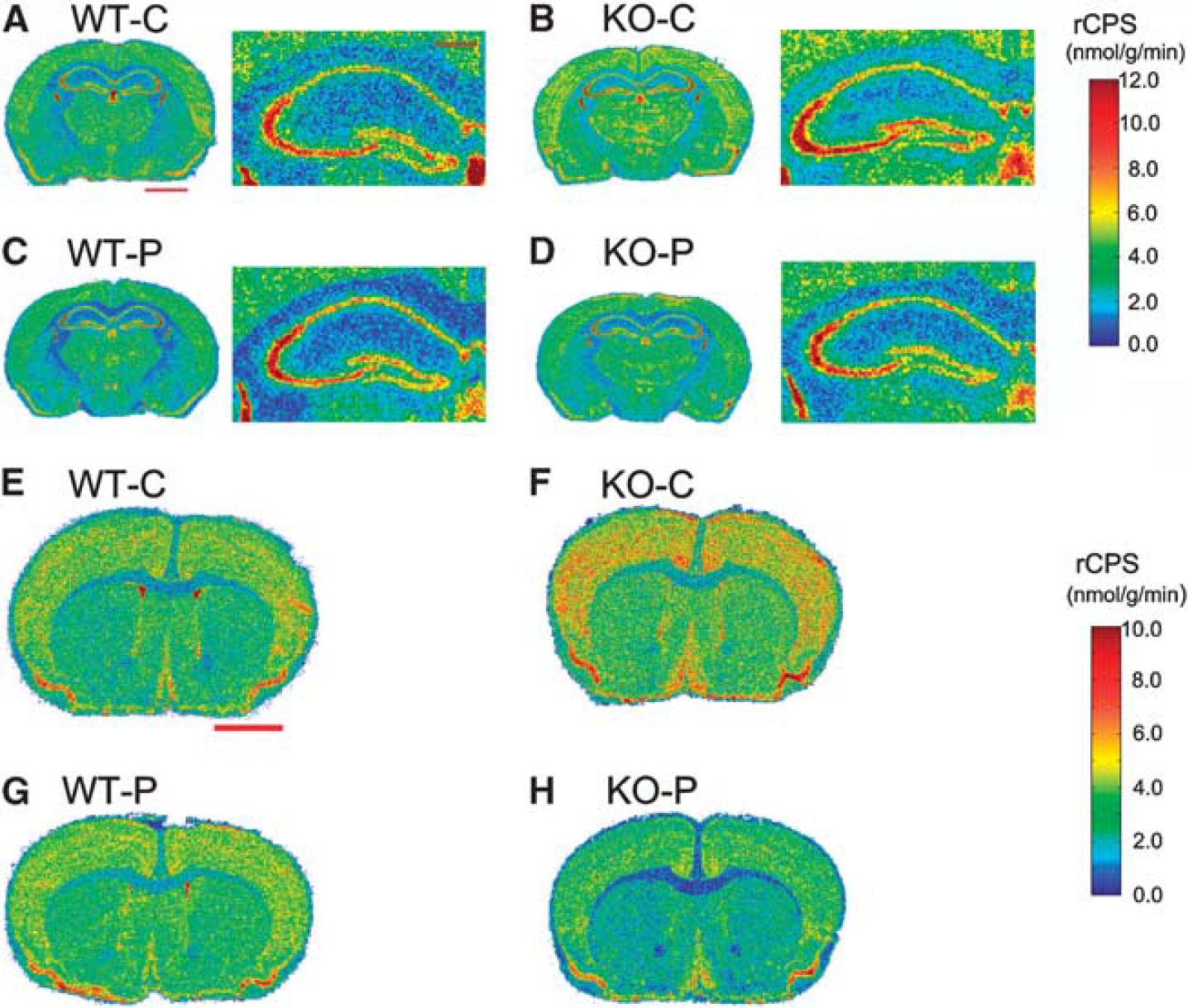
Effects of propofol sedation on rates of cerebral protein synthesis (rCPS) in wild-type (WT) and knockout (KO) mice. Representative digitized autoradiographic images from the four groups of mice at two levels of brain. Images are color coded for rCPS by the scales shown on the right. The upper colorbar pertains to (
DISCUSSION
The main finding of this study is that, during propofol sedation, subjects with FXS have significantly decreased rCPS in the brain as a whole, cerebellum, and parts of cortex compared with controls. Our studies in mice confirm our previous finding 10 that, in the absence of FMRP, rCPS is increased over WT in selective brain regions. Further, these studies show that, in rodents and humans without FMRP, propofol is associated with widespread decreases in rCPS whereas, in subjects with FMRP propofol has minimal effects. We do not know whether at baseline, FXS subjects have increased rCPS. The need to sedate FXS subjects added an unanticipated level of complexity to the study. Our results in the mouse model suggest that we would have found increased rCPS in FXS subjects had we been able to study them in the conscious state, assuming that the mouse model reflects the human disease. Moreover, our results suggest that drugs, such as propofol, that decrease neural activity and alter synaptic signaling may reverse the effects of altered rCPS in FXS and are highly therapeutically relevant.
We recognize that our sample was possibly biased toward higher functioning FXS participants as subjects taking psychotropic medication were excluded. However, all FXS subjects enrolled were intellectually impaired with full scale IQ scores between 47 and 63, and 6 of the 15 subjects met the criteria for autism. Moreover, we saw no relationship between rCPS and either IQ or autism scores in our small sample. We are aware, however, that the values of IQ that we measured with the WAIS III may have a ‘floor effect’ in this intellectually impaired population. If the phenotype of higher functioning subjects was closer to that of healthy volunteers, then we would have expected rCPS values to have been merely closer to control values, but we found decreased rCPS in these subjects. The direction of the change suggests that there may be an explanation that has more mechanistic implications. Among our control subjects, four had above average IQ scores. This was clearly a result of our successful recruitment efforts among fellows working at the NIH and at local college campuses. We have considered how our above average controls might have affected our results by looking for a relationship between IQ scores and rCPS. We find correlations which are statistically significant (
The primary methodology used in this study, the L-[1- 11 C]leucine PET method for measuring rCPS in human subjects, 12 was based on the autoradiographic L-[1- 14 C]leucine method for use in animals. 24 The PET method was validated in monkeys 13 and is characterized by low variability and good reproducibility in controls. 14 Whereas the PET method does not have the exquisite spatial resolution of the autoradiographic method, it has the distinct advantage that it can be used to repeatedly study this dynamic process in human subjects under different conditions. Our study shows that the PET method has the sensitivity to detect 8% to 10% changes in rCPS in relatively small numbers of subjects, provided that the subjects are well matched and the study well designed. The method, therefore, may provide a quantitative and objective means of assessing the presence of disease and the effect of proposed treatments.
An incidental finding of our study is the effect of FXS on regional brain volumes, the subject of several prior studies.27–29 Increased volume of the caudate nucleus (23% to 25%) is a common finding in two of the prior studies.28,29 We also found a 14% increase in caudate volume in FXS subjects although it did not quite reach statistical significance. Changes in the caudate nucleus are of interest in light of its involvement in executive function and reports that increased caudate volumes are associated with repetitive behaviors in autism. 30 There is general agreement that whole brain and cerebellum volumes are unaffected in FXS (present study; refs. 28 and 29), but differences in other regions analyzed makes comparability impossible. In our study, volumes of hippocampus, thalamus, and putamen were lower (14% to 21%) in the FXS subjects. These effects are of particular interest in view of the involvement of these three regions in many types of learning.
Based on our previous results in the
Propofol is widely used in clinical anesthesia and its primary mechanism of action is thought to be the enhancement of GABAA receptor function to produce hyperpolarization of neurons. Acting on GABAA receptors, propofol can also affect excitatory inputs by inhibiting glutamate release at the synapse. 31 Propofol can also block voltage-dependent sodium channels by binding to inactivated channel states, 32 and at higher concentrations propofol may directly affect GABAA receptor/chloride channel opening. 33 Further, propofol also decreases neural activity as indicated by decreases in cerebral blood flow and energy metabolism.34,35 Nevertheless, the mechanism by which propofol reduces rCPS in FXS is unknown and will be the subject of future investigation.
We think that the increased rCPS seen in the KO mouse model
10
is a core phenotype of FXS and is likely also pathogenic in the human disease. In
Herein, we show that an anesthetic, propofol, reduces rCPS in FXS mouse brain and that in FXS humans sedated with a continuous infusion of propofol, the levels of rCPS are reduced in whole brain compared with control subjects studied under the same conditions. Taken together and extrapolated clinically, our findings suggest that effecting changes in protein synthesis are a viable therapeutic strategy in FXS. This strategy can be accomplished with the use of agents that affect neuronal and synaptic activity, and the effect of strategies that alter protein synthesis can be monitored with the L-[1- 11 C]leucine PET method.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors thank Dr E Berry-Kravis, Dr Ben Oostra, Dr G Neri, and Dr R Hagerman who helped with patient recruitment; David L Nelson who gave us the
