Abstract
Neuronal endoplasmic reticulum (ER), continuous from soma to dendritic spines, undergoes rapid fragmentation in response to N-methyl-D-aspartate (NMDA) receptor stimulation in hippocampal slices and neuronal primary cultures. Here, we show that ER fragments in the mouse brain following cardiac arrest (CA) induced brain ischemia. The ER structure was assessed
Introduction
The endoplasmic reticulum (ER) is involved in biosynthesis, protein quality control, cellular stress responses, and Ca2+ homeostasis. Neuronal ER is a key regulator of intracellular Ca2+ signaling, acting as a sink and store of releasable Ca2+, thereby important for synaptic transmission and plasticity (Verkhratsky, 2005). Three-dimensional reconstructions depict the neuronal ER as a complex, continuous network of interconnected cisternae and tubules extending from soma to apical dendrites (Terasaki et al, 1994). The continuous ER lumen permits an efficient diffusion and equilibration of ions from the soma to the dendrites (Choi et al, 2006; Dayel et al, 1999) and ER is often referred to as neuron-within-a-neuron where ER Ca2+ channels transmit a signal along its membrane through different neuronal compartments (Berridge, 1998).
We have previously shown that extracellular Ca2+ influx triggers rapid and reversible loss of ER continuity (fragmentation) in hippocampal slices or cultured neurons when transiently stimulated by NMDA receptor agonists or high potassium concentrations (Kucharz et al, 2009; Kucharz et al, 2011 (in press)). It is well established that during stroke, brain trauma and epilepsy neurons are transiently or permanently overloaded with Ca2+ that could be detrimental (Lau and Tymianski, 2010). Therefore, we asked whether ER dynamics could be monitored in the living brain, and if rapid ER fragmentation occurs in a condition of neuronal Ca2+ overload.
We utilized
Materials and methods
Generation of Thy1-Enhanced Green Fluorescent Protein-Endoplasmic Reticulum Transgenic Mice
Transgenic C57BL/6 mice were generated to express ER lumen-targeted soluble EGFP (EGFP-ER hereafter called GreenER) under Thy1 promoter. The transgene construct was created using EGFP gene containing calreticulin ER-targeting sequence and KDEL ER-retention sequence (gift from Thomas Oertner; Friedrich Miescher Institute) cloned into pThy1 vector (gift from Joshua R Sanes; Washington University). We assessed the fluorescence signal and the cellular expression patterns among three generated transgenic lines and chose the Thy1-EGFP-ER-7b line for further studies.
Surgery
All animal procedures were approved by the Malmö/Lund ethical committee. The surgery was performed according to Holtmaat et al (2009). Thy1-EGFP-ER mice were anesthetized (3.5% isoflurane in N2O/O2 (30:70)). During all the steps of surgery and imaging, the temperature of animal was monitored by a rectal thermistor probe (Linton Instrumentation, Norfolk, UK) and maintained at 37°C. The head was mounted onto a stereotactic frame, the eyes lubricated with Viscotears (Novartis, Basel, Switzerland), and the animals were treated with Temgesic (0.1 mg/kg, Schering-Plough, Kenilworth, NJ, USA). Isoflurane was decreased to 1.8% to 2%, the scalp and periosteum were removed, and Marcaine (2.5 mg/mL, bupivacain; AstraZeneca, London, UK) was applied to the scar, then covered with tissue adhesive (Vetbond, 3M, St Paul, MN, USA, 3 mol/L). Animals were treated with Rimadyl (5 mg/kg carprofen; Pfizer, Kent, UK) and Dexafort (2 mg/kg, dexamethasone; Intervet, Sollentuna, Sweden). The craniotomy (4 mm diameter) was performed 2 mm posterior to bregma and 2 mm right lateral to midline. To prevent excessive heating, the skull was flushed every 5 minutes with cold saline. The bone flap was removed and the intact dura was covered with a wetted gelatin sponge (Spongostan, Ferrosan, Denmark). Low melting agarose (Promega, Madison, WI, USA) was applied to the brain surface and a sterile 5 mm diameter coverglass (Karl Hecht GmbH and Co. KG ‘Assistent’, Sondheim/Rho'n, Germany) was positioned on top of the cranial opening with dental cement. A custom-made titanium bar was attached to stabilize the head during the imaging procedures.
For induction of cardiac arrest (CA), a femoral vein catheter was inserted. Thereafter, the animal was mounted to the imaging stage insert by the titanium bar. The animal was spontaneously breathing 1.8% to 2% isoflurane in N2O/O2 (30:70) gas mixture during imaging. Cardiac arrest was induced by intravenous injection of 0.5 mL 2 mol/L KCl.
In Vivo Imaging
The
Fluorescence Recovery After Photobleaching Analysis
To assess the ER continuity, fluorescence recovery after photobleaching (FRAP) was used. From each mouse, 10 random dendrites with typical ER morphology were chosen. The GreenER signal was collected using Gallium–Arsenide–Phosphide detector from a 2 μm × 2 μm selected region of interest on a dendrite for 10 scanning cycles, bleached for 15 cycles with 900 nm Mai Tai laser, and the FRAP was recorded for 90 cycles. The parameters used for collecting FRAP were optimized for each animal used in the study. The data were normalized to the initial signal fluorescence intensity = 1.
Results
The Morphology of Endoplasmic Reticulum In Vivo Before Cardiac Arrest
The GreenER was expressed under the control of Thy1 promoter in a subset of cortical neurons. The morphology of ER defined by GreenER signal did not differ from that observed in our previous studies in primary neuronal cultures and hippocampal slice cultures (Kucharz et al, 2009). The ER appeared continuous with no abnormalities in morphology such as blebbing or vacuolization (Figure 1A). For our studies, we selected structures in dendrites with typical ER morphology expressing GreenER in molecular layer I or external granular layer II.
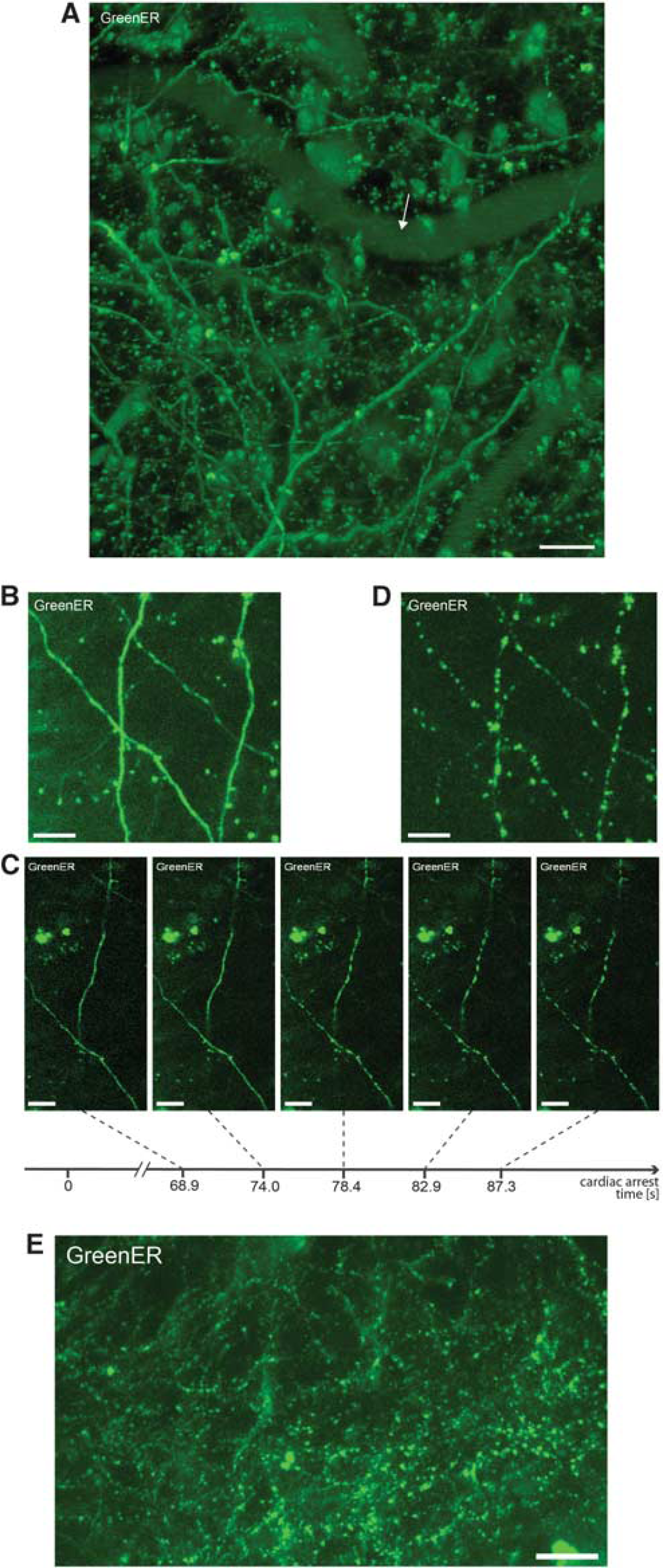
Rapid dendritc endoplasmic reticulum (ER) fragmentation in cortical neurons following the cardiac arrest. (
Endoplasmic Reticulum Fission in the Mouse Brain
The analysis of ER morphology showed that under basal conditions in the anesthetized mouse, the dendritic ER in cortical neurons is continuous. The continuity of ER was verified by
Assessing Endoplasmic Reticulum Fragmentation by Fluorescence Recovery After Photobleaching Analysis
To confirm the ER fragmentation and compartmentalization, we performed FRAP analysis on dendritic ER of cortical neurons. Because of the secondary effects related to phototoxic damage occurring during the high-power multiphoton bleaching step and the high frame rate of time lapse recording of signal recovery, the parameters for collecting the FRAP signal (i.e., the time interval between scans, bleaching laser power, and iterations) were adjusted separately for each mouse used in the study.
The dendritic signal from GreenER before CA displayed high degree of FRAP, reaching above 67% of initial signal strength, confirming the continuous structure of ER (Figure 2). Subsequently, CA was induced and 2 minutes thereafter the continuity of ER lumen was assessed by FRAP from neurons in the same brain area, as before CA. In all analyzed animals, the neurons exhibited loss of FRAP (Figure 2), confirming the reduction in ER lumen continuity. A similar loss of FRAP was seen in two additional investigated animals (data not shown).
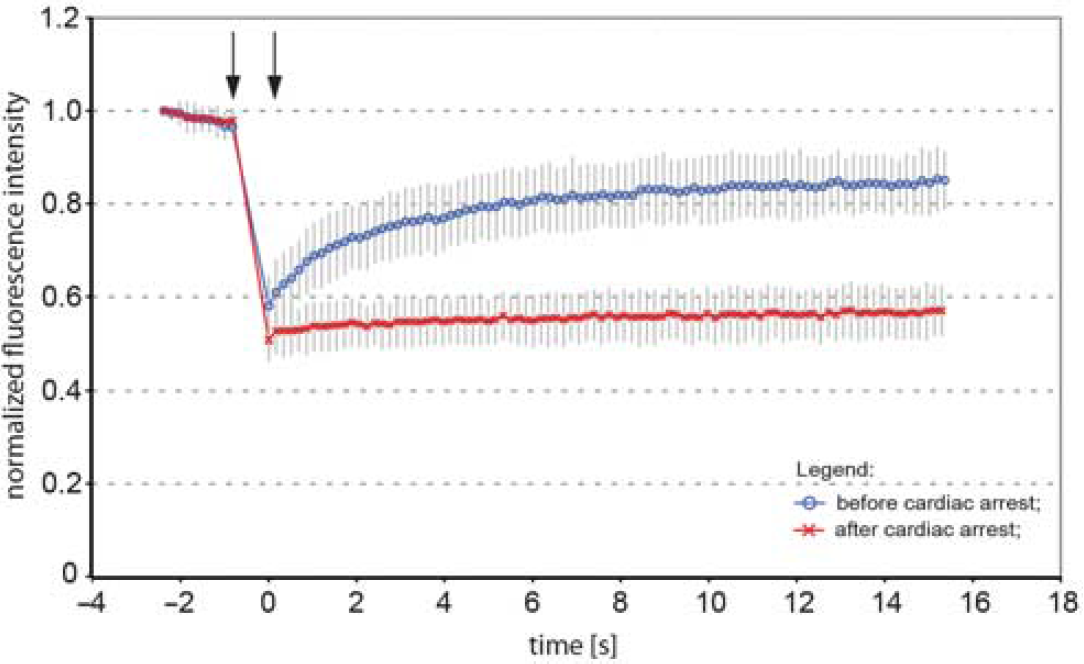
Fluorescence recovery after photobleaching (FRAP) analysis of dendritic endoplasmic reticulum (ER) fragmentation in cortical neurons following the cardiac arrest. Normalized average FRAP signal over time in dendrites of neurons in a mouse subjected to cardiac arrest. The graphs show FRAP signal recovery before (blue), and loss of FRAP (red) after the 2-minute of cardiac arrest. Photobleaching was performed in time between the arrows. Time = 0 was set to when photobleaching ends and fluorescence starts to recover. The recovery of FRAP signal from dendritic ER before the cardiac arrest indicates ER continuity, while loss of FRAP demonstrates a fragmented structure of ER. Error bars are s.e.m.,
Discussion
Multiphoton
The ER in primary cultures and organotypic slices undergoes rapid and reversible fragmentation upon NMDA receptor stimulation, while inhibition of NMDA receptors or removal of extracellular Ca2+ abolishes ER fragmentation (Kucharz et al, 2009; Kucharz et al, 2011 (in press)). Cardiac arrest results in an immediate loss of cerebral blood flow, followed by depletion of tissue glucose, ATP and PCr, and subsequently depolarization of neuronal plasma membranes (Hansen, 1985). The shortage of energy supply leads to inhibition of Ca2+ extrusion from neuronal cytosol and increase in intracellular Ca2+ levels (Silver and Erecinska, 1992). Noteworthy, the depolarization of cell membranes and intracellular Ca2+ increase is rapid and occurs 1 to 2 minutes after cessation of blood flow to the brain (Hansen, 1985; Xie et al, 1995). In our study, using potassium-induced CA we validated the occurrence of ER fragmentation
The CA experiments are terminal; however, in the transient middle cerebral artery occlusion experimental model of stroke, the infarct core with irreversibly injured tissue is surrounded by penumbra area with cells that have the potential to recover and exhibit trains of transient membrane depolarizations. A transient ER fragmentation in these conditions may contribute to neuroprotection by limiting the diffusion of Ca2+ within ER, as proposed previously (Kucharz et al, 2009; Kucharz et al, 2011 (in press)). Whether the reversible ER fission occurs in penumbra and if it contributes to cell survival are of subject of future research.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
