Abstract
Numerous studies of the effects of estrogens for stroke prevention have yielded conflicting results in human and animal studies alike. We present a systematical analysis of study design and methodological differences between 66 studies where estrogens' impact on ischemic brain damage in rat models has been investigated, providing evidence that the differences in results may be explained by high estrogen doses produced by slow-release pellets. These pellets have been used in all studies showing increased neurologic damage because of estrogens. Our data indicate that the increased neurologic damage is related to the pellets' plasma concentration profile with an early, prolonged, supraphysiological peak. Neither the method of inducing the ischemic brain lesions, the choice of variables for measuring outcome, the measured plasma concentrations of estrogens at the time of ischemia nor rat population attributes (sex, strain, age, and diseases) are factors contributing to the discrepancies in results. This suggests that the effects of estrogens for stroke prevention are concentration related with a complex dose-response curve, and underscores the importance of carefully validating the experimental methods used. Future studies of hormone-replacement therapy in women may have to take dosage and administration regimens into account.
Introduction
Men have a higher incidence of stroke compared with premenopausal women (Kolominsky-Rabas et al, 2001; Sacco et al, 1998; Stegmayr et al, 1997), indicating that estrogens may have neuroprotective properties. This in combination with the widespread use of estrogens as hormone-replacement therapy (HRT) for ameliorating postmenopausal symptoms has prompted numerous recent studies on the possible neuroprotective effects of estrogens on the brain in stroke. The earliest studies in women showed neuroprotective effects (Falkeborn et al, 1993; Finucane et al, 1993; Paganini-Hill et al, 1988), whereas more recent studies have seriously cast the beneficial effects of estrogens and HRT in doubt (Anderson et al, 2004; Grady et al, 2002; Hulley et al, 1998; Rossouw et al, 2002; Simon et al, 2001; Viscoli et al, 2001). The Women's Health Initiative trial (WHI), which included more than 16,000 women (Rossouw et al, 2002), has had the largest scientific impact among the general public. Its discontinuation because of a higher incidence of coronary heart disease, breast cancer, and stroke in the HRT group, for example, contributed to that almost half of Swedish HRT-treated women ceased their estrogen intake (Hoffmann et al, 2005), despite the fact that equine estrogens were used at one dosage level, the women included in the WHI study were on average 10 years older than the typical menopausal women and had substantially elevated body mass index.
Many of the issues concerning estrogens, for example, their mechanisms of action in the brain, can only be elucidated using animal models. Since Simpkins in 1997 pioneered in showing that female ovariectomized rats developed smaller stroke lesions if they received 17β-estradiol ahead of the lesion (Simpkins et al, 1997), numerous animal studies have researched the subject. Some claim that animal experiments have uniformly shown protective effects of estrogens in stroke models (Alonso de Lecinana and Egido, 2006; Hoffman et al, 2006; Pozzi et al, 2006; Suzuki et al, 2006), but even if the studies showing protective effects represent a solid majority, there are at least six studies from three different research groups (including ours), including data from more than 270 rats, showing the opposite—that administration of estrogens to estrogen-deficient rats results in increased ischemic neurologic damage. The six studies showing increased damage in response to estrogen treatment—which could be argued to agree better with the results of the most substantial human study in which negative effects of estrogens in cerebral ischemia were seen (WHI (Rossouw et al, 2002))—are contradicted by more than 50 studies clearly showing neuroprotective effects of estrogens.
In the studies investigating the effects of estrogens on ischemic brain damage the following experimental setup has generally been adopted:
Animals are ovariectomized (if female animals are used).
Some type of estrogen, most commonly 17β-estradiol, is administered to one or more of the groups (this step is in some experiments performed after the ischemic injury).
Ischemic brain injury is induced.
The effect of the brain injury is evaluated.
The fact that all included studies have followed these steps, though with substantial variation within each paragraph, makes this cluster of studies particularly suitable for analyzing specific methodological parameters. We attempted to explain the differences between rat studies reporting neuroprotection and those reporting neurodamage of interest, because this may provide clues to the parallel dichotomy in human studies. Therefore, the aim of the present review is to find an explanation for the dichotomy in study results by comparing the methods in the articles concerning the protective or damaging effects of estrogens in rat stroke models.
Methods
Article Search
Medline, Embase, and Scopus were searched using the keywords ‘rat’, ‘estrogens’ or ‘estradiol’, ‘cerebral’ and ‘ischemia’ in an attempt to find all original experimental studies published until July 2008, which have used the experimental setup described above. The reference lists of 30 review articles were searched for additional original studies (Alonso de Lecinana and Egido, 2006; Amantea et al, 2005; Behl, 2002; Bisagno et al, 2003; Dhandapani and Brann, 2002; Duckies and Krause, 2007; Garcia-Segura et al, 2001; Gibson et al, 2006; Green and Simpkins, 2000; Hoffman et al, 2006; Hum and Brass, 2003; Hum and Macrae, 2000; Lang and McCullough, 2008; Macrae and Carswell, 2006; McCullough and Hum, 2003; Merchenthaler et al, 2003; Pozzi et al, 2006; Roof and Hall, 2000; Scallet, 1999; Simpkins et al, 2005b; Suzuki et al, 2006; Weigl et al, 2005; Wise, 2003, 2000, 2002; Wise et al, 2005, 2001a, b ; Yang et al, 2005, 2003), and finally the reference lists of all original studies were searched. The earliest study included for analysis was from November 1997 (Simpkins et al, 1997).
Inclusion and Exclusion Criteria
For a study, or a particular set of experimental groups within a study, to be included in the current analysis the following was required (Figure 1):
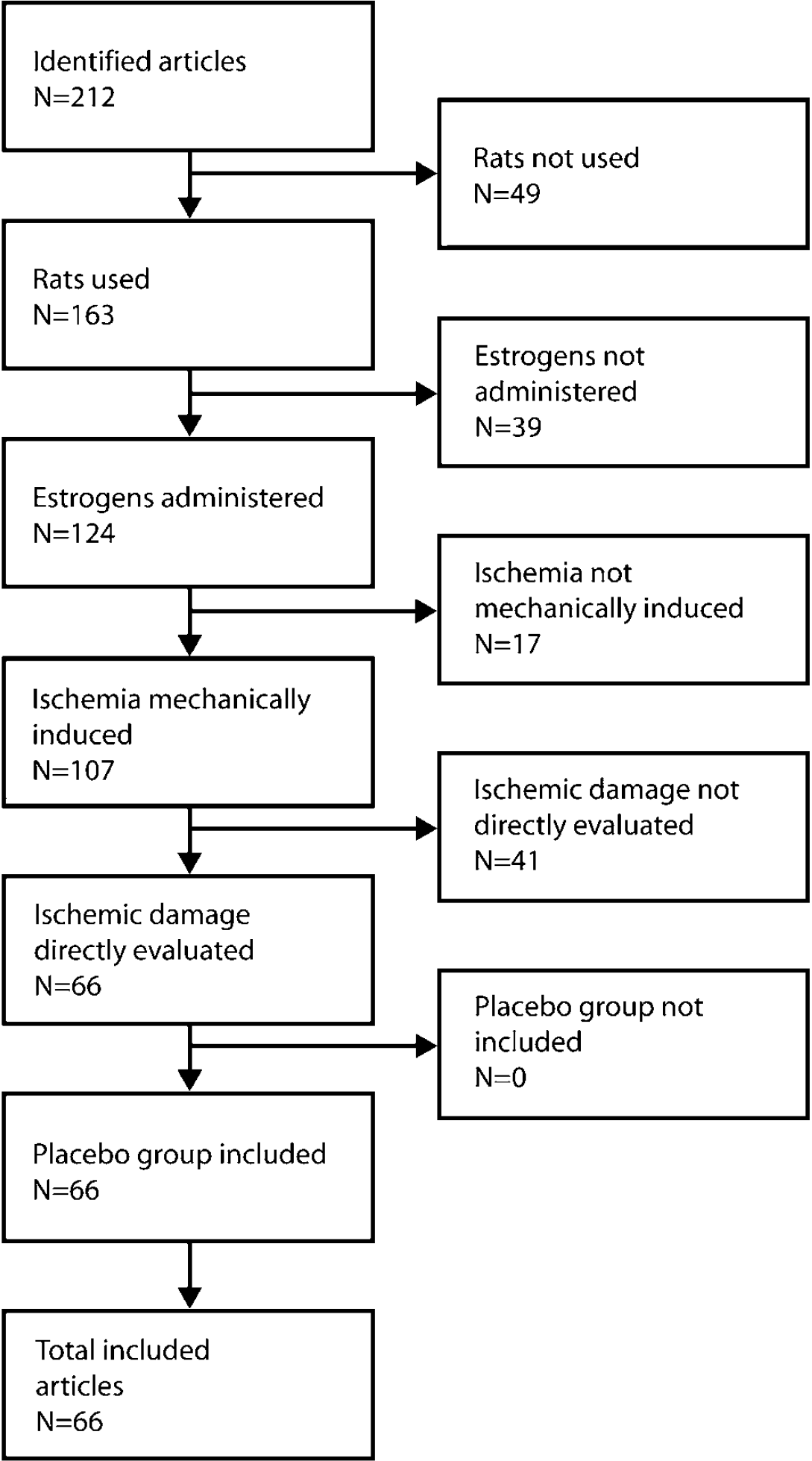
From 212 identified articles 49 were excluded because rats were not used, 39 because estrogens were not administered alone, 17 because ischemia was not mechanically induced, and 41 because ischemic damage was not directly evaluated. None of the remaining articles were excluded because of lack of a proper placebo group. After the exclusion process, 66 articles remained to be included in the review analysis.
The experiments must be conducted in rats.
Estrogen administration must be the variable differing between the experimental groups, with no additional drugs administered.
The injury must be mainly ischemic, and physically, not, for example, pharmacologically, induced.
The outcome measure must be directly related to the damage, for example, size of the cerebral ischemic lesion, neuronal cell loss in hippocampus, or neurologic deficits and not, for example, blood-brain barrier disruption or endothelial dysfunction. Also, the result must be quantitatively accounted for in the text, a table, or a graph.
Each estrogen treatment group must have a corresponding placebo group.
The study was restricted to one species—rats—because (1) they have been most extensively used for experimental stroke studies, (2) there are unexplained differences between research groups in the results and conclusions of these studies, and (3) because we have—both experimentally and by studying the results of others—reasons to believe we have found an explanation for the dichotomy of results in rat studies.
Finally a total of 66 studies were included, of which 56 concluded that estrogens exerted a protective effect (Alkayed et al, 2001, 2000; Amantea et al, 2007; Bagetta et al, 2004; Chiappetta et al, 2007; Choi et al, 2004; Dai et al, 2007; Dubai et al, 1998, 2006; Dubai and Wise, 2001; Dziennis et al, 2007; Fan et al, 2003; Feng et al, 2005; Fukuda et al, 2000; Green et al, 2001; Gulinello et al, 2006; Hawk et al, 1998; He et al, 2002; Jover-Mengual et al, 2007; Kii et al, 2005; Koh, 2007, 2008, 2006; Littleton-Kearney et al, 2005; Liu et al, 2007, 2002; McCullough et al, 2001; Miller et al, 2005; Nunez et al, 2007; O'Donnell et al, 2006; Pelligrino et al, 1998; Plamondon et al, 2006; Rau et al, 2003; Raval et al, 2007; Ritz et al, 2004; Rusa et al, 1999; Saleh et al, 2004, 2001a, b ; Sandstrom and Rowan, 2007; Schreihofer et al, 2005; Shi et al, 2001, 1997; Simpkins et al, 1997; Toung et al, 2004, 1998, 2000; Wang et al, 1999; Wen et al, 2004; Wise and Dubai, 2000; Won et al, 2005; Won et al, 2006a, b ; Yang et al, 2000; Yu et al, 2006; Zhang et al, 1998), whereas six, on the contrary, reported a damaging effect (Bingham et al, 2005; Carswell et al, 2004; Gordon et al, 2005; Harukuni et al, 2001; Theodorsson and Theodorsson, 2005; Yong et al, 2005). Four articles reported no difference between placebo and estrogen treatments (Farr et al, 2006; Goodrow et al, 2005; Santizo et al, 2002; Vergouwen et al, 2000).
Extraction of Information
Data from all included articles were transferred to a data sheet with a row for each single group of rats and columns for the different method parameters and for the results. If more than one of the included outcome measures were used in an article, they were prioritized according to how frequently they were used in the analysis as follows:
Size of ischemic lesion, most often evaluated with brain slicing and staining, but in some cases with MRI.
Histologic evaluation of neuronal cell loss or survival, most often in the hippocampus.
Neurologic deficit score.
Other outcome measures.
An exception from this order was used when extracting data from one article, where neurologic deficit score was chosen because of difficulties in assessing and summarizing the presented histologic data (Santizo et al, 2002). In the articles where the results were only given in graphical form, the numbers were extracted from carefully quantifying the information displayed in the graphs. In the case that more than one moment or location of damage evaluation was given for one or more groups of rats, and no summarized result were presented, the most representative numbers for the study's total result, or the most popularly evaluated site of injury, for example, the CA1 region in the hippocampus, were selected.
Investigation of the Effect of Method and Study Design Factors on the Effects of Estrogens on Ischemic Brain Damage
The aim of the present study was to find methodological or study design factors separating studies reporting diverse results on cerebral ischemia in response to estrogen treatment, and particularly to identify the factors contributing to neurodamage because a strong majority of the included studies reported neuroprotection. Four main categories of factors were found as detailed in Table 1.
Categories of factors studied

Nominal scale variables were analyzed as proportions, and presented in tables when relevant. Ratio scale variables (written in italic format in Table 1) were analyzed by Pearson's correlation analysis (r) using the proportional damage in estrogen- versus placebo-treated groups (abbreviated E/P ratio), weighted against the number of animals in each study (thus a comparison including 40 animals was given twice the weight of a comparison including 20 animals). If, for example, the area of ischemic lesion was halved by the estrogen treatment, the E/P ratio was 0.5, and if the area was exactly the same in the experimental groups, the E/P ratio was 1.0. For ‘positive’ outcome measures (e.g., cell count in hippocampus) the E/P ratio was inversed to enable comparisons to studies using ‘negative’ outcome measures (e.g., ischemic lesion area). A high r2 value shows the part of the overall variation in the data explained by a linear relation between the factors. The presented P-values were calculated according to Dunn—Sidak method.
Age was considered as an ordinal scale variable and no correlation analysis was performed, because in many articles it was merely stated that the used animals were ‘adult’ or of an adult weight.
In most of the studies where plasma estrogen concentrations were measured, the samples were obtained during or within 24 h of ischemic damage. To make a relevant analysis for correlation between plasma concentrations and E/P ratio, we included only measurements within this time frame.
Studies specifically investigating the effects of the methodological parameters themselves were found especially valuable as they provide a comparatively unbiased source of information. All of these were thoroughly analyzed and considered, but are for readability and convenience only presented when contributing to identifying the methodological parameters responsible for the estrogen-stroke dichotomy.
Estrogen administration regimen
Administration Mode
When comparing the administration methods used it was found that all studies reporting neurodamaging effects of estrogens used commercially manufactured slow-release pellets (Table 2; Bingham et al, 2005; Carswell et al, 2004; Gordon et al, 2005; Harukuni et al, 2001; Theodorsson and Theodorsson, 2005; Yong et al, 2005). Also, two unpublished studies by Wallace and co-workers, and Bingham and co-workers, referred to in one of the included articles (Carswell et al, 2004), both conducted in 2003 and both showing damaging effects of estrogens, used this type of pellets. This adds up to a total of eight studies (of which only six are included in the current review because the remaining two have not been published) showing detrimental effects of estrogens, all using commercial pellets, whereas none of the studies using other administration methods have shown neurodamaging effects of estrogens. As can be observed in Table 2, not all studies using pellets have found detrimental effects of estrogens, which is further discussed in the ‘Dosage regimen’ section.
Effect of exogenous estrogens depending on administration mode

The three most widely used methods for administering estrogens, commonly 17β-estradiol, were (1) injections (subcutaneous, intravenous, intraperitoneal or intracerebral), (2) commercially manufactured slow-release pellets (Innovative Research of America, Sarasota, FL, USA) and (3) silastic capsules filled with hormone in solution or in crystal form, used in 35, 21, and 16 of the included studies, respectively (some studies included more than one method). One study used oral administration of estrogens (Littleton-Kearney et al, 2005; Table 2).
Dosage Regimen
In the studies using commercial pellets, lower estrogen doses tended to decrease ischemic damage whereas higher doses tended to increase it. Supporting this pattern, some studies where other administration methods have been used have reported diminished protection with increased hormone levels or have failed to show a positive dose—response relation between estrogens and neuroprotection.
We will, in assessment of the current variable, first focus on the 21 pellet studies. The slow-release pellets are manufactured by the company Innovative Research of America, and each type of pellet has two main characteristics: dose and release time, from which a presumed daily dose can be calculated. Each of the studies using these pellets includes a comparison between at least one estrogen-treated and one placebo-treated rat group. Figure 2A depicts the damage in estrogen- and placebo-treated animals depending on the total estrogen dose of the pellets. As can be seen, lower pellet doses tend to decrease ischemic damage whereas higher doses tend to increase it, correlating with an r2 value of 0.24 (P < 0.001). In Figure 2B, a similar comparison is made, but using a calculated daily estrogen dose instead of total pellet dose, suggesting the same relation as in the previous figure, correlating with an r2 value of 0.29 (P < 0.001).
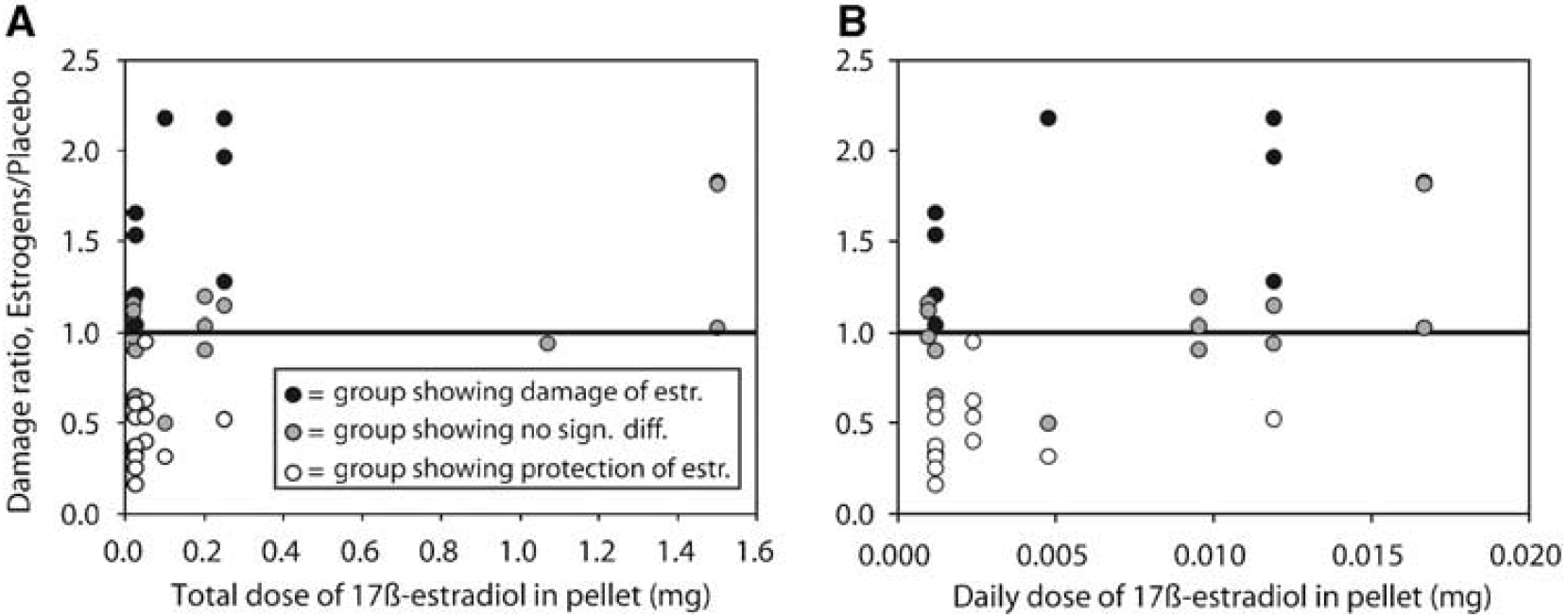
For each study group where pellets were used an ‘E/P ratio’ between damage in estrogen- and placebo-treated rats was calculated. An E/P ratio of 1 indicated that the estrogens had no impact on the ischemic damage. In the figures E/P ratio is plotted against the total dose of estrogens (
When considering the relation between dose and stroke outcome, it is of interest to note that some within-study comparisons of effects of different doses have been performed, which are presented in Table 3. As can be seen, the studies using pellets show a tendency of more cerebral damage with higher estrogen doses, whereas the studies using other administration methods have reported more variable effects of increasing the estrogen dose.
Studies investigating the effect of different doses of estrogens

Type of Estrogen
The overwhelming dominance of 17β-estradiol, used in 62 of the 66 studies, among these all the studies using slow-release pellets, excludes the possibility that type of estrogen is the sought confounder. Estradiol benzoate (Sandstrom and Rowan, 2007), estradiol valerate (Fukuda et al, 2000), and conjugated equine estrogens (Littleton-Kearney et al, 2005; McCullough et al, 2001; Rusa et al, 1999) were administered in altogether five studies, all showing protective effects.
Length of Time Between Ovariectomy and Estrogen Administration
The length of time between ovariectomy and estrogen administration, also referred to as ‘washout period’, does not influence the result of estrogens' effect on ischemic damage. When analyzing the correlation between washout period and E/P ratio, an r2 value of 0.016 was obtained. Including only the pellet studies resulted in an r2 value of 0.01.
Length of Time Between Estrogen Administration and Ischemic Damage
The length of time between administration of the estrogen and induction of ischemia could theoretically influence the impact of the hormone on the ischemic damage. Though, when analyzing the correlation between this time period and E/P ratio, an r2 value of 0.018 was obtained. Including only the studies using pellets resulted in an r2 value of 0.0003.
Measured Plasma Concentrations
Another way of addressing the complex relationship between administration method, dosage, and timing is to assess studies where the hormone concentrations have actually been measured during or close to the time of the ischemic episode. Though, when analyzing the correlation between plasma concentrations within 24 h of ischemic damage against E/P ratio no correlation was found (r2 = 0.012). There was also no correlation when only the pellet studies were included (r2 = 0.029).
Induction of ischemic brain lesions
Many different techniques for inducing ischemic lesions in the rat brain were used in the included studies, but neither neuroprotection nor neurodamage was confined to a specific method or method feature.
The methods for inducing lesions had two main characteristics: (1) the targeted vessel; and (2) whether the occlusion was transient, allowing reperfusion damage, or permanent. The most commonly occluded vessel was the middle cerebral artery (MCA), producing focal ischemia to mimic human ischemic strokes, in contrary to the four-vessel occlusion method that results in global ischemia, mimicking cardiac arrest. The MCA can be occluded transiently using the intraluminal filament method or by a microclip, or it can be occluded permanently, also using the intraluminal filament method, electrocoagulation, or irradiation. Other methods for induction of ischemic lesions include methods targeting the common carotid artery or internal carotid artery.
Neurodamage has been reported using both permanent (Bingham et al, 2005; Carswell et al, 2004; Gordon et al, 2005) and transient (Harukuni et al, 2001; Theodorsson and Theodorsson, 2005; Yong et al, 2005) occlusion targeting MCA (Bingham et al, 2005; Carswell et al, 2004; Gordon et al, 2005; Theodorsson and Theodorsson, 2005; Yong et al, 2005) and also using the four-vessel occlusion method of global ischemia (Harukuni et al, 2001). Neuroprotection has been shown using all described methods. Hence, the choice of method of inducing ischemic damage in the rat seems to have no impact on outcome of the trial (Table 4).
Effect of exogenous estrogens depending on ischemic damage method

MCA, middle cerebral artery; ICA, internal carotid artery; CC, common carotid.
Outcome measure
Type of Outcome Measure
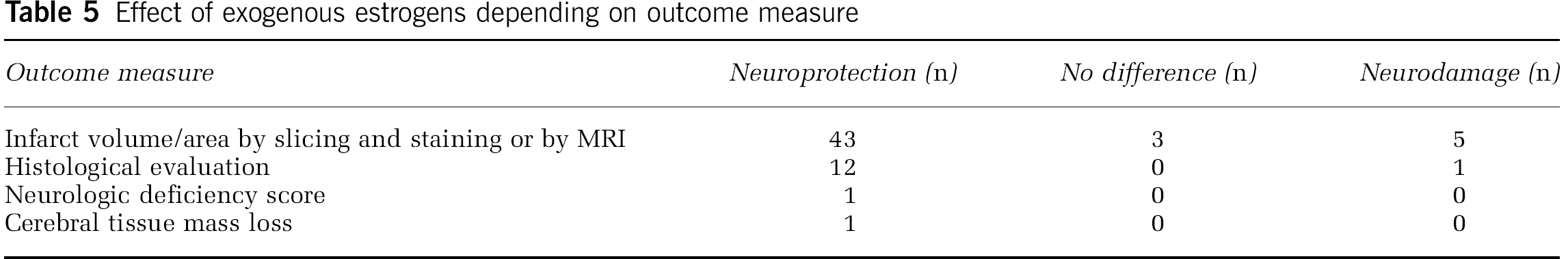
Nothing is added when assessing differences in outcome measure between studies, as the articles reporting neurodamage used similar outcome measures as the ones reporting neuroprotection (Table 5).
Effect of exogenous estrogens depending on outcome measure
Time of Damage Evaluation After Damage Infliction
The time of damage evaluation after ischemia is not a methodological factor explaining the differences between neurodamaging and neuroprotective results. When analyzing for correlation against E/P ratio an r2 value of 0.001 was obtained. Including pellet studies only slightly shifted the r2 value to 0.03.
Neurodamaging effects of estrogens have been reported when damage was evaluated from 24 h (Bingham et al, 2005; Carswell et al, 2004; Gordon et al, 2005) to 1 week (Harukuni et al, 2001) after ischemia. Neuroprotection has been reported in both shorter and longer times of expectation, ranging from 2 h (Shi et al, 2001) to 6 months (Plamondon et al, 2006).
Rat population attributes
Sex
There is no evidence that sex is a confounding factor differing between the studies showing neuroprotection and neurodamage in response to estrogens (Table 6). However, no studies have reported increased neurologic damage in response to estrogens in males. This could result from the fact that estrogens have been administered by slow-release pellets to male rats in only three studies. Of these, two reported protection (Toung et al, 1998, 2000) and one reported no significant difference between groups (Vergouwen et al, 2000).
Effect of exogenous estrogens depending on the sex of the rats used

Strain
The use of different rat strains is not the methodological parameter responsible for the differences in study results, because neither neuroprotective nor neurodamaging effects of estrogens are confined to a specific strain (Table 7).
Effect of exogenous estrogens depending on strain of rats

Age
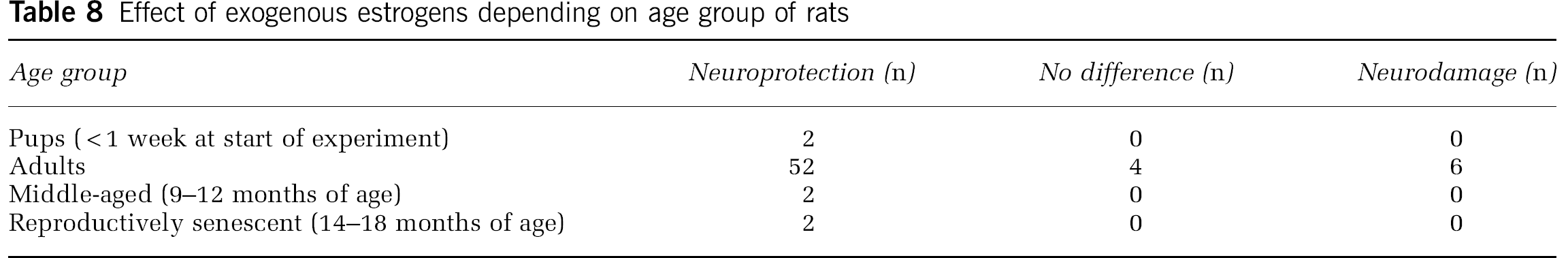
The studies conducted on rats of other ages than adults are at present too scarce to allow conclusions to be drawn, other than that they do not offer an explanation to the different effects of estrogens on brain damage (Table 8).
Effect of exogenous estrogens depending on age group of rats
Diseases
If all six studies showing damaging effects of estrogens were performed in, for example, diabetic rats, the question concerning the estrogen dichotomy would have been solved, though this is not the case (Table 9).
Effect of exogenous estrogens depending on disease of rats

Discussion
Analyzing the information in the 66 articles included in the current review, we found that neurodamaging effects of estrogens in rat models of ischemic brain damage are clearly related to the use of commercially manufactured slow-release pellets. This relation seems to be associated to the dose of estrogen delivered, because higher pellet doses correlate to increased neurodamage and less neuroprotection. No other methodological factors appear to contribute substantially to the differences in results.
Increased Neurologic Damage Related to Use of Slow-Release Pellets
The hormone release profile probably offers an explanation for the negative impact of the slow-release pellets. Pellets have been reported to produce early, extremely supraphysiological (300 to 550 pg/mL), plasma concentration peaks followed by a substantial decrease (Strom et al, 2008; Theodorsson et al, 2005; Figure 3). The early, high peak is also the main feature differing between the pellets and, for example, silastic capsules, producing plasma levels within the physiologic range (< 50 pg/mL) for several weeks (Strom et al, 2008; Figure 3). This suggests that the increased damage induced by the pellets is due to the early plasma peak. In contrast it could be argued that many of the injection regimens adopted in the included articles also have resulted in peaks that are even higher (Green et al, 2001; Liu et al, 2007) than reported for pellets (Strom et al, 2008), but here the peak's width/length of time must also be considered. Although it can take several days or even weeks to return to physiologic levels of 17β-estradiol after administration by slow-release pellets (Strom et al, 2008; Theodorsson et al, 2005), the peak induced by subcutaneous injections is to an end within 24 h (Green et al, 2001; Liu et al, 2007). If very high levels of estrogens contribute to neurodamage, it is reasonable that a long-time peak would exert a more substantial negative impact than a short-time peak. Also, the fact that the peak can last for weeks (Figure 3) makes it overlap the time for ischemic damage (occurring 1 to 2 weeks after hormone administration in the studies reporting neurodamaging effects of estrogens). In conclusion, it can be hypothesized that the drawn-out, very high plasma concentration peak produced by the pellets has a detrimental effect on the nervous system by itself or in combination with the subsequent ischemic damage.
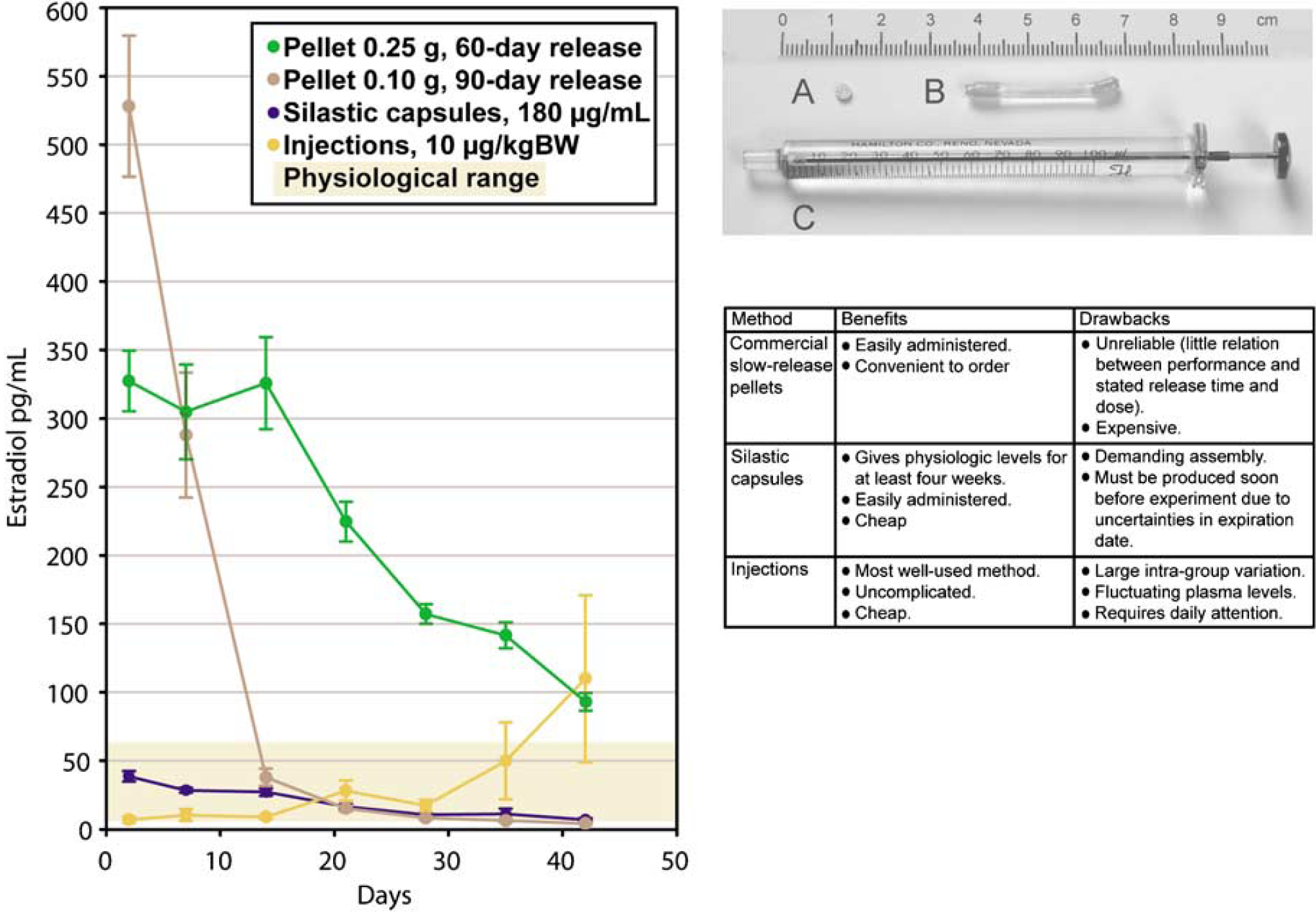
In a previous study (Strom et al, 2008), the three most well-used methods for administrating 17β-estradiol to ovariectomized rats were tested: slow-release pellets (0.25 mg 60-day release pellets, 0.10 mg 90-day release pellets), silastic capsules (silastic laboratory tubing, inner/outer diameter: 1.575/3.175 mm, filled with 20 mm columns of 180 μg 17β-estradiol per mL sesame oil) and daily subcutaneous injections (10 μg 17β-estradiol per kg bodyweight). Blood samples were obtained at days 2, 7, 14, 21, 28, 35, and 42 after administration, and hormone levels were analyzed. The resulting levels in comparison with physiologic levels of estrogen are presented in the graph to the left. In conclusion, silastic capsules are superior to pellets and injections in reliably producing long-term 17β-estradiol concentrations within the physiologic range. Innovative Research of America pellet (
As aforementioned, the theory that the estrogen plasma concentrations produced by the pellets lead to the deleterious effects is corroborated by the second finding of the current review; that higher pellet doses seem to contribute to increased neurodamage and less neuroprotection. Several studies using lower pellet estrogen doses have reported protective effects, whereas most of the studies using higher pellet estrogen doses have reported detrimental effects (Figure 2). However, the dose-response relation between pellet dose and neurodamage is not perfect (r2 = 0.24 and 0.29, respectively), which could be explained by the following:
There are probably other minor factors that contribute to the discrepancies in estrogens' effects in cerebral ischemia, for example, small differences in mean weight and age of the animals and so on, which because of their low impact have not been identified in the current review.
It has been shown that the pellets are unreliable considering what is stated about the dosage in relation to the actual plasma levels produced (Strom et al, 2008).
The hypothesis presented in the current paper, that estrogen is related to ischemic damage in a biphasic—or even more complex—manner does not presume that the dose-response curve in any specific interval along the dose axis is perfectly linear. The dose-response relation between the doses of the pellets (yielding relatively high levels of circulating estrogens) and the ischemic damage represents an interval on this presumably, for example, biphasic curve, where higher estrogen doses shift the effect from being protective to damaging.
The idea that the neuroprotective effects of estrogens may be reversed at higher doses/plasma concentrations is included in the concept of hormesis, which points out that many steroid hormones show a biphasic dose—response curve (Calabrese and Baldwin, 2003). Even if hormesis has mainly been associated to mechanisms mediated by intracellular receptors, it is crucial to realize that estrogens do not only exert their effects through ER-α and ER-β, but also through membrane receptors and by direct chemical interactions including antioxidant effects (Behl, 2002). The antioxidant effects of estrogens (phenolic scavenging of lipid peroxyl radicals) may prevail at physiologic concentrations whereas redox cycling at higher estrogen concentrations may lead to increased radical generation and damage (Prokai et al, 2003; Simpkins et al, 2005a). Estrogens may also protect against neurodamage by antiatherogenic actions through effects on vascular smooth muscles, endothelium and on hepatic expression of coagulation proteins, and also by direct effects on neurons by decreasing apoptotic gene expression, increasing antiapoptotic gene expression, counteracting release of cytochrome c from mitochondria, and by inhibiting inflammation through downregulation of tumor necrosis factor-α (reviewed in Alonso de Lecinana and Egido, 2006 and Amantea et al, 2005). Concerning the potential harmful effects, it has been reported that estrogens inflict damage by inducing the formation of free radicals (Desjardins et al, 1995). Also, mechanisms of estrogen neurodamage concerning increased excitotoxicity have been postulated (Macrae and Carswell, 2006), because estrogens has been reported to upregulate glutamate N-methyl-
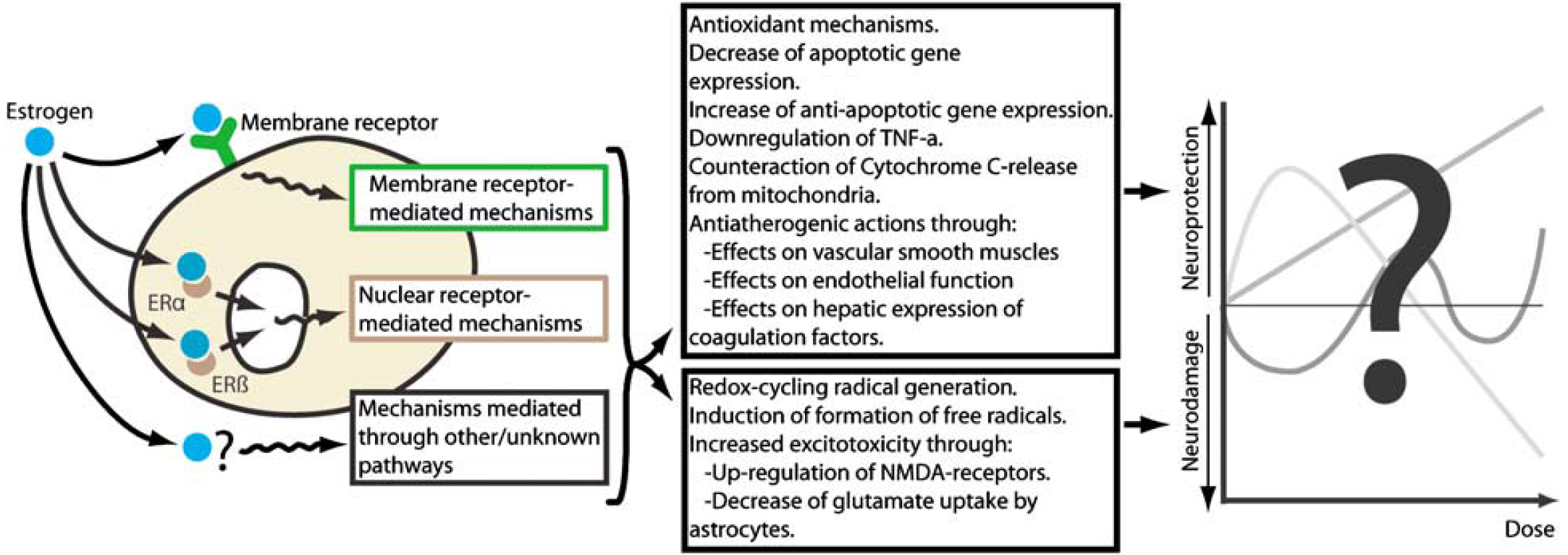
Estrogens have many pathways of inducing effects that are both potentially harmful and protective in the ischemic brain. The probable diversity of the pathways and effects in respect of their individual dose—response curves could produce a highly complex relation between the dose of estrogens and impact on ischemia. Thus, the question of whether estrogens are neuroprotective or neurodamaging is misleading—it is possible that, depending on the concentration, estrogens could be both.
The idea of a more complex, for example, biphasic, dose—response curve is supported by the observations from the studies investigating the effects of different doses, presented in Table 3. Considering that the pellets in general produce order of magnitude higher sustained levels of hormone than silastic capsules and injections do, the combined results in Table 3 could suggest that the estrogen neuroprotection is reversed at higher doses, because a significant dose—response relation between neuroprotection and estrogens is only seen in studies using injections (Choi et al, 2004) and silastic capsules (Dubai et al, 1998; Dubai and Wise, 2001), whereas studies testing different pellet doses reported dose—response relations in the damaging direction (Bingham et al, 2005; Carswell et al, 2004; Rusa et al, 1999).
Earlier Reviews and Theories
To the best of our knowledge, no systematic investigation attempting to map the methodological differences between studies showing neuroprotection versus neurodamage in rat models of ischemic damage has been previously published. Instead, earlier reviews in the area of estrogens and ischemic damage in rat models have to a large extent focused on potential biologic mechanisms (Alonso de Lecinana and Egido, 2006; Amantea et al, 2005; Behl, 2002; Dhandapani and Brann, 2002; Duckies and Krause, 2007; Garcia-Segura et al, 2001; Gibson et al, 2006; Green and Simpkins, 2000; Hoffman et al, 2006; Hum and Macrae, 2000; Lang and McCullough, 2008; McCullough and Hum, 2003; Merchenthaler et al, 2003; Pozzi et al, 2006; Roof and Hall, 2000; Scallet, 1999; Simpkins et al, 2005b; Suzuki et al, 2006; Weigl et al, 2005; Wise, 2003, 2000, 2002; Wise et al, 2005, 2001a, b ; Yang et al, 2005) or functional aspects (Bisagno et al, 2003), whereas others mainly have stated that estrogens may have a neuroprotective function (Hurn and Brass, 2003; Macrae and Carswell, 2006; Yang et al, 2003). Some, even though published after the studies showing neurodamage, neglect that there is a dichotomy concerning effects of estrogens in the rat brain (Alonso de Lecinana and Egido, 2006; Hoffman et al, 2006; Pozzi et al, 2006; Suzuki et al, 2006). One systematic analysis in the field of estrogens and stroke in animal models has been published previously, though differing fundamentally from the current review in that it did not focus on differences between neuroprotective and neurodamaging studies. The main finding of the previous review was that estrogens are neuroprotective in a positive dose-dependent manner, which could not be corroborated by the current review (Gibson et al, 2006).
The duration between ovariectomy and estrogen administration (washout period) has previously been proposed as the methodological factor that could switch the effects of estrogen from positive to negative. Suzuki et al (2006) reported that the neuroprotective effects of estrogens in a model of cerebral ischemia in mice were lost when prolonging the washout period, though no detrimental effect was found. Furthermore, by analogy, this factor might have contributed to the lack of neuroprotection found in the WHI trial, where the average participating women had been in menopause for almost a decade before estrogen treatment (Rossouw et al, 2002). However, this hypothesis is not corroborated by the current review because no relationship was found between the effects of estrogens and the washout period. Also, in a previous study in our laboratory, we found that the duration of the washout period did not influence the later induced plasma concentrations (Strom et al, 2008).
In 2006 MacRae and Carswell suggested that the influence of estrogens on ischemic damage is linked to the method of inducing cerebral ischemia, which is not corroborated by the current results (Macrae and Carswell, 2006).
Comparing Animal Experimental Studies to Human Population Studies
It requires emphasis that there are issues that make it difficult to directly extrapolate animal experimental data to clinical populations. For example, the human population studies investigate the reduction of stroke incidence with long-term (years) therapy whereas the animal experiments investigate a relatively acute (days to months) neuroprotective effect. Moreover, although estrogens are an obvious common denominator, HRT has other pharmacological components than the estrogen regimens adopted in many of the animal studies.
Conclusions
The discontinuation of the WHI trial because of HRT-related increases in risk of coronary heart disease, breast cancer, and stroke, despite a lack of effect on all-cause mortality, probably eliminates the possibility of performing further studies of the same kind. Regrettably, there are several problems when trying to extrapolate the WHI results to a broader population: first, the women included in WHI were on average 10 years older than the typical menopausal woman and, second, had substantially elevated body mass index. As a consequence, the importance of well-conducted animal studies is even greater than before.
In light of the presented hypothesis, that discrepancies in estrogen administration methods have yielded diametrically different effects on ischemic damage, it seems appropriate to underscore the importance of a thorough knowledge of the methods used in an experimental setup. In the current example of administrating estrogens, it is of utmost importance to make several consecutive measurements in the subject organism during the entire length of the experiment to acquire correct information about the method. If experimental methods (even commercially available and widely used) are not thoroughly validated, wrong conclusions can be drawn.
A final crucial aspect of the impact of estrogen administration methods in animal models of brain ischemia is what this may imply for the administration of HRT in relation to stroke risk. It could be hypothesized that the choice between different HRT administration regimens, each option rendering different plasma hormone profiles, could have an impact on HRT being neuroprotective or neurodamaging. Would it be possible to achieve the benefits of HRT at the same time as avoiding the risks by altering the dose and/or mode of estradiol administration?
Footnotes
Acknowledgements
The substantial contribution of four reviewers is gratefully acknowledged.
The authors declare no conflict of interest.
