Abstract
The effect of transplantation of adult bone marrow stromal cells (MSCs) into the freeze-lesioned left barrel field cortex in the rat was investigated by measurement of local cerebral glucose utilization (lCMRglc) in the anatomic structures of the whisker-to-barrel cortex sensory pathway. Bone marrow stromal cells or phosphate-buffered saline (PBS) were injected intracerebrally into the boundary zone 1 h after induction of the freezing cortical lesion. Three weeks after surgery, the 2-[14C]deoxyglucose method was used to measure lCMRglc during right whisker stimulation. The volume of the primary necrotic freezing lesion was significantly reduced (P<0.05), and secondary retrograde degeneration in the left ventral posteromedial (VPM) thalamic nucleus was diminished in the MSC-treated group. Local cerebral glucose utilization measurements showed that the freezing cortical lesion did not alter the metabolic responses to stimulation in the brain stem trigeminal nuclei, but eliminated the responses in the left VPM nucleus and periphery of the barrel cortex in the PBS-treated group. The left/right (stimulated/unstimulated) lCMRglc ratios were significantly improved in both the VPM nucleus and periphery of the barrel cortex in the MSC-treated group compared with the PBS-treated group (P<0.05). These results indicate that MSC transplantation in adults may stimulate metabolic and functional recovery in injured neuronal pathways.
Keywords
Introduction
Adult bone marrow contains multiple stem cells that can differentiate into hematopoietic cells as well as other cell lineages. Bone marrow nonhematopoietic mononuclear cells or bone marrow stromal cells (MSCs) are regarded as hematopoietic support cells (the ability to function as a feeder layer for the growth of hematopoietic stem cells) and can differentiate into various cell types, including fibroblasts, osteoblasts, and adipocytes (Prockop, 1997; Ferrari et al, 1998; Pereira et al, 1999). Bone marrow stromal cells were recently shown to differentiate into glial cells and neurons (Kopen et al, 1999; Munoz-Elias et al, 2003; Woodbury et al, 2000) and also to secrete various growth factors that may alleviate neurologic deficits after their transplantation into the injured brain (Chen J et al, 2001, 2003; Chen X et al, 2002; Li et al, 2002). Recently, animals with stroke or head injury were shown to recover from neurologic deficits after transplantation of MSCs (Mahmood et al, 2001; Chen et al, 2001; Lu et al, 2001a, b; Chopp and Li, 2002; Li et al, 2002; Lee et al, 2003), but these effects were evaluated only by behavioral tests, such as the rotarod test and neurologic scoring tests.
Electrophysiologic and metabolic mapping techniques have identified neural relay stations in the sensory pathway between the mystacial vibrissae (whiskers) and the sensory cortex in the rat. These include the trigeminal sensory neuronal network, the ventral posteromedial (VPM) thalamic nucleus, and the ‘whisker-barrel’ field in the sensory cortex (Hall and Lindholm, 1974; Gonzalez and Sharp, 1985; Sharp and Gonzalez, 1985). Unilateral whisker stimulation in the rat increases local cerebral glucose utilization (lCMRglc) in the ipsilateral brain stem trigeminal nuclei, that is, principal sensory trigeminal nucleus (PSTN) and spinal trigeminal nucleus (SPTN), and the contralateral VPM nucleus in the thalamus, barrel field of the primary somatosensory cortex (SI), and secondary somatosensory cortex (SII) (Gonzalez and Sharp, 1985; Sharp and Gonzalez, 1985).
Unilateral cortical ablation in rats results in decreased glucose utilization in the ipsilateral thalamus 3 to 4 days after surgery (Cooper and Thurlow, 1984). Ginsberg et al (1989) studied the effects of unilateral focal infarction in the rat cortical whisker-barrel field on functional activation of the whisker-barrel pathway 5 days after the infarction and found the stimulation of lCMRglc by whisker stimulation to be greatly depressed throughout the ipsilateral barrel cortex and ventrobasal thalamus. They considered the metabolic disturbance in the functionally related area remote from the primary lesion to be because of retrograde degeneration of the thalamic neurons after injury of the thalamocortical pathway.
Diaschisis, the remote effects of acute and chronic central nervous system (CNS) injury, may manifest itself as depression of neural function (electrical excitability), cerebral blood flow (CBF), and metabolism in the CNS at a distance (Yuan et al, 1986; Meyer et al, 1993). This phenomenon is thought to be the most likely mechanism for transneuronal metabolic depression because of either deafferentation or deefferentation in a transsynaptic neuronal network (Meyer et al, 1993; Kolb, 2003). The degree of clinical recovery is well correlated with restitution of local cerebral metabolism and perfusion, supporting the concept of functional neurologic correlates for diaschisis.
The present study was designed to determine whether transplantation of MSCs improves functional recovery, as assessed by the measurement of lCMRglc, in the rat whisker-to-barrel cortex pathway after freezing brain cortical injury.
Materials and methods
All procedures were performed out in accordance with the Japanese Association for Laboratory Animal Science Guidelines on the Care and Use of Animals and an animal study protocol approved by the Juntendo Casualty Center Animal Care and Use Committee.
Preparation of Bone Marrow Stromal Cells
Bone marrow stromal cells were prepared from normal adult male Wistar rats (300 to 400 g). Fresh bone marrow was displaced from the femurs via a 21G needle connected to a 1 mL syringe containing phosphate-buffered saline (PBS). The bone marrow was gently dissociated in a dish under aseptic conditions, and the dissociated bone marrow cells were seeded in plastic culture flasks in α-minimum essential medium Eagle (α-MEME) (Sigma, St Louis, MO, USA) supplemented with 10% fetal bovine serum, 2 mmol glutamine, 6 mg/100 mL kanamycin sulfate, and 100 ng/mL stem cell factor (Sigma), and incubated in a CO2 incubator for 72 h at 37°C. After 72 h, the cells (MSCs) adherent to the plastic were resuspended in new medium and grown for 4 days. Nerve growth factor (NGF 2.5S) (Invitrogen, Carlsbad, CA, USA) (100 ng/mL) was then added to the medium, and the MSCs were incubated for 1 week. Figures 1A and 1B show the freshly removed hematopoietic bone marrow cells and the cultured mononuclear MSCs.
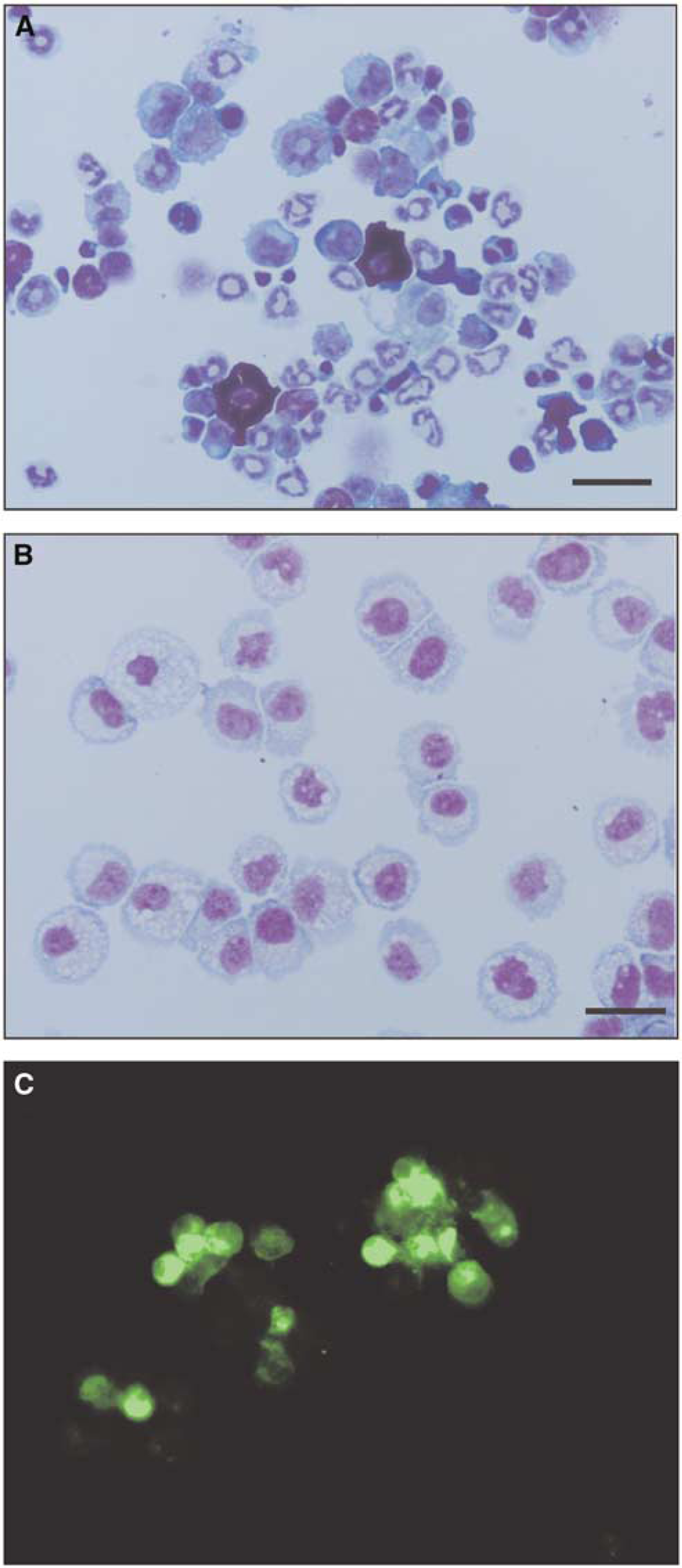
Photomicrographs showing hematopoietic cells and MSCs as follows: (
Induction of Freezing Lesions in the Primary Somatosensory Cortex Barrel Field
Under light halothane anesthesia (1.0% to 1.5% halothane in 70% N2O/30% O2) male Wistar rats (11 to 12 weeks old, 250 to 260 g) were fixed in a rat stereotaxic frame (NARISHIGE Scientific Instrument Lab., Tokyo, Japan), a 5-mm diameter hole was made in the left parietal bone with a dental drill, and the dura mater was exposed. A standardized freezing lesion was induced in the barrel field of the SI by applying an aluminum rod (4.5 mm in diameter), cooled by liquid nitrogen and positioned by means of a micromanipulator, to the exposed dura mater for 15 s. The center of the lesion was 2 mm posterior to the bregma and 7 mm lateral from the midline, corresponding to the center of the barrel field cortex. Body temperature was monitored by a rectal probe and maintained at 37°C during the procedure. The scalp incision was sutured, and the animals allowed 1 h for recovery before transplantation surgery was performed. Sham-operated controls received the same left parietal craniotomy but not the freezing lesion (N = 5).
Transplantation of Bone Marrow Stromal Cells into Cortex beneath Freezing Lesion
The cultured MSCs were resuspended in serum-free α-MEME at a density of 1 × 106 cells/mL, and 5 μL of DiO solution (Vybrant® DiO cell-labeling solution, Molecular Probes, Carlsbad, CA, USA) was added to the medium. The suspension was then incubated for 20 mins at 37°C and then centrifuged at 1500 rpm for 5 mins. The DiO-labeled MSCs in the sediment were washed three times with PBS and resuspended at a density of 4 × 105/10 μL in PBS (Figure 1C). The experimental rats with the freezing lesion were anesthetized with light halothane anesthesia as described above and fixed in the stereotaxic frame, and 5 μL of DiO-labeled MSCs (2 × 105 cells) in PBS was stereotaxically infused into the center and beneath the cortical freezing lesion via a Hamilton syringe with a 24G needle at a rate 1 μL/min. The skin was then sutured, the anesthesia discontinued, and the animals allowed to recover for various periods. Control rats were treated similarly with PBS alone. To observe the spatiotemporal profile of the transplanted DiO-labeled MSCs in the rat brain, the rats were killed with ether at 3 days, 1 week, 2, and 3 weeks after transplantation (N = 3 in each group), and the brains were removed and frozen in isopentane chilled by dry ice to −60°C. The frozen brains were cut into 10 μm-thick coronal sections in a cryostat at −20°C, and the sections were thaw-dried on glass slides at approximately 60°C and fixed with 4% paraformaldehyde in PBS for 20 mins at room temperature. The sections were then stained with Gills Hematoxylin Solution No. 3 (Wako Pure Chemical Industries Ltd, Osaka, Japan) for 10 s, washed with running water, and the transplanted DiO-labeled MSCs were examined by fluorescent microscopy.
Presence of neuronal or glial cells different from MSCs was assessed at 2 and 3 weeks after transplantation. Ten-micrometer-thick coronal frozen sections containing the DiO-labeled MSCs were dried, fixed with 4% paraformaldehyde in PBS for 20 mins at room temperature, and incubated in blocking buffer (1% bovine serum albumin, 0.2% Tween 20, 5% horse serum in PBS) for 1 h at room temperature, and then incubated with either mouse antineuronal nuclei (Neu N) monoclonal antibody (1:1,000, Chemicon, Temecula, CA, USA) or rabbit anti-glial fibrillary acidic protein (GFAP, 1:1,000, Chemicon) for 1 h at room temperature. The bound antibodies were visualized by binding with Alexa Fluor® 594 goat anti-mouse immunoglobulin G (IgG, 1:1,000) or Alexa Fluor® 594 goat anti-rabbit IgG (1:1,000) for 1 h at room temperature and fluorometric examination. Fluorescence was observed with excitation at 460-490 nm and emission at 510-550 nm for DiO and excitation of 520-550 nm and emission of more than 580 nm for Fluor® 594. The double-staining was evaluated with a fluorescence microscope (Model BX 51, Olympus, Tokyo, Japan). Images were processed with Meta View software (Nippon Roper Co., Chiba, Japan).
Determination of Local Cerebral Glucose Utilization During Unilateral Whisker Sensory Stimulation and Measurement of Necrotic Volume of Lesion in the Cortex
Transplantation of MSCs or injection of PBS in control rats at 1 h after induction of cortical freezing injury was performed as described above. Three weeks after MSCs transplantation (N = 6), PBS injection (N = 6), or sham operation (N = 5), the rats were anesthetized with light halothane anesthesia (1.0% to 1.5% halothane in 70% N2O/30% O2), and polyethylene catheters (PE50) were inserted into the left femoral artery and vein. Loose-fitting plaster casts were then applied to the pelvic area and taped to an iron brick to prevent locomotion. The whiskers (large vibrissae) on the left side of face were clipped and removed to avoid spurious stimulation on the control side. Body temperature was continuously monitored by a rectal probe and maintained at 37°C with a heating lamp. At least 4 h were then allowed for recovery from the anesthesia before starting measurement of lCMRglc. Arterial blood pressure, hematocrit, and blood gas tensions were measured, and animals with abnormal values were excluded from the studies.
Local cerebral glucose utilization was determined by the 2-[14C]deoxyglucose (2-[14C]DG) method as originally described (Sokoloff et al, 1977). The dose of 2-deoxy-
Arterial plasma 2-[14C]DG concentrations were assayed by liquid scintillation counting (Packard Instruments, Downers Grove, IL, USA) with external standardization. Arterial plasma glucose concentrations were measured with a Glucose Analyzer 2 (Beckman Instruments, Fullerton, CA, USA). The frozen brains were cut into 20-μm-thick coronal sections for quantitative autoradiography and 10-μm-thick coronal sections for hematoxylin-eosin staining in a cryostat maintained at −20°C. The sections to be autoradiographed were thaw-mounted on glass coverslips, dried on a hot plate at 60°C, mounted on cardboard, and autographed together with a set of calibrated 14C-labeled methylmethacrylate standards with Kodak BIOMAX MR film (Eastman Kodak Co., Rochester, NY, USA) in X-ray cassettes. The autoradiographs were digitized in an ES-2200 scanner (Seiko Epson Co., Nagano, Japan) and displayed on a computer monitor for densitometric analysis.
Local tissue 14C concentrations were determined from the local tissue optical densities in the autoradiographs and the calibration curve was derived from the 14C standards. Local cerebral glucose utilization was computed from the local tissue 14C concentrations and the time courses of the arterial plasma glucose and 2-[14C]DG concentrations by the operational equation of the method and the computer program developed by G Mies (Max Planck Institut fur Neurologische Forschung, Koln, Germany) for the NIH Image program (W Rasband, NIMH, Bethesda, MD, USA). The rate constants and lumped constant used in the calculations were those previously determined in the rat (Sokoloff et al, 1977). Local cerebral glucose utilization was determined in 26 brain structures, including those in the whisker-barrel sensory pathway (i.e., PSTN, VPM thalamic nucleus, barrel field of the SI, and the peripheral structures of the barrel field cortex corresponding to the SII). The brain structures visualized in the digitized images were identified according to the rat brain atlas of Paxinos and Watson (1997).
Regions of necrosis in the left barrel field caused by the freezing were excised during the cutting of the brain in the cryostat. These regions were visible in the digitized autoradiographs, and their volumes were determined by quantitative morphometric analysis by means of the NIH Image program.
Statistics
The data are presented as means ± standard deviations. Statistical significance of differences were analyzed by one-way analysis of variance (ANOVA) and Student's t-tests by means of SPSS 7.5.1 J for Windows (SPSS Japan Inc., Tokyo, Japan). P-values <0.05 were considered statistically significant.
Results
Migration of Transplanted Bone Marrow Stromal Cells beneath Cortical Freezing Lesion
Fluorescence microscopy showed that by 3 to 7 days after transplantation, MSCs transplanted into the center and beneath the freezing lesion had migrated along the periphery of the lesion (Figure 2A). The green fluorescent cells at 3 and 7 days after transplantation were uniformly stained in a similar manner to the DiO-labeled MSCs immediately before transplantation, suggesting that the DiO-labeled cells were transplanted MSCs at 3 and 7 days after transplantation. Such DiO-labeled MSCs were still recognizable at 2 weeks after transplantation despite the presence of extracellular deposits of DiO from autolysed MSCs. However, some of the fluorescent cells at 2 and 3 weeks after transplantation may be macrophages that had phagocytized autolysed MSCs.
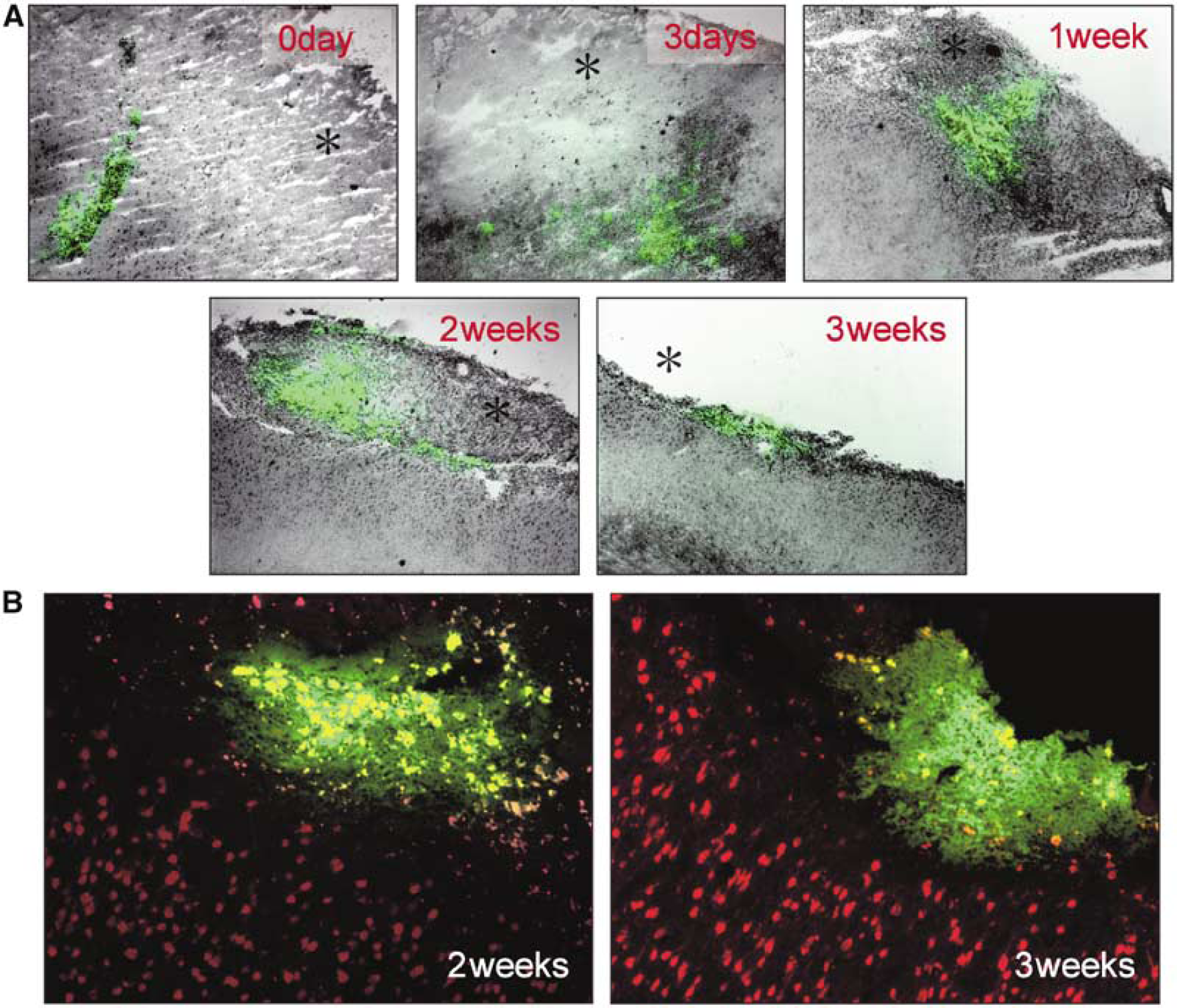
(
About half of the MSCs had migrated into the freezing lesion and a few into the corpus callosum. The distribution of the migrated MSCs appeared to correspond to the area of vasogenic edema around the cortical freezing lesion. Preliminary examination of the freezing lesion in the barrel field by intravenous Evans blue administration showed that the area of the vasogenic edema extended from the periphery of the lesion to the corpus callosum. There were indications that the transplanted MSCs had migrated with the edema fluid (data not shown). Bone marrow stromal cells that had migrated into the cortical lesion were lost when the necrotic tissue was removed during the brain cutting 3 weeks after transplantation. Bone marrow stromal cells were, however, still present in the bottom of the cortical lesion, but in lesser numbers than at 2 weeks.
Double-fluorescence labeling of MSCs with cell membranes labeled with DiO (green) and neocortical neurons with nuclei labeled with Neu N (red) showed no doubly labeled cells. DiO-labeled transplanted MSCs at 2 and 3 weeks expressed neither Neu N (Figure 2B) nor GFAP phenotypes (data not shown).
Physiologic Parameters in Rats Studied with 2-[14C]deoxyglucose Method
Body weight, mean arterial blood pressure, rectal temperature, hematocrit, and arterial PaO2, PaCO2, and pH at the time of measurement of lCMRglc were all within normal limits and not significantly different among the three experimental groups (Table 1).
Physiologic parameters in rat groups of the 2-[14C]DG experiment
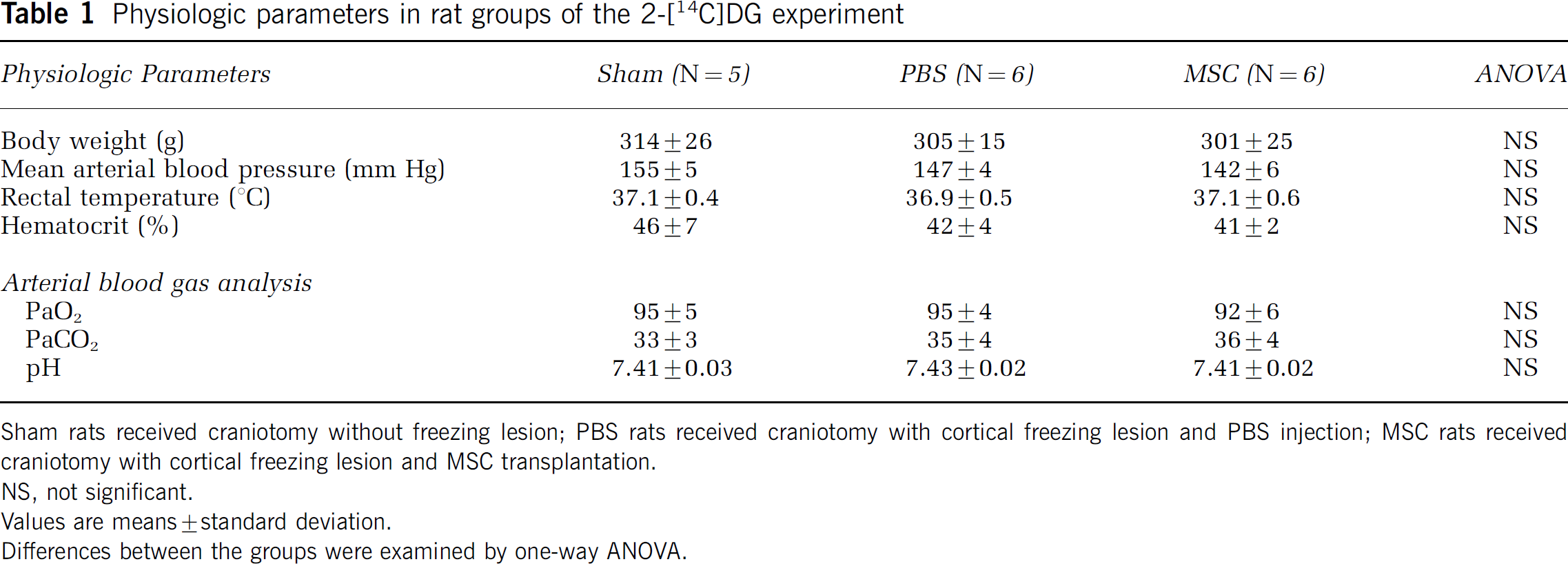
Sham rats received craniotomy without freezing lesion; PBS rats received craniotomy with cortical freezing lesion and PBS injection; MSC rats received craniotomy with cortical freezing lesion and MSC transplantation. NS, not significant.
Values are means±standard deviation.
Differences between the groups were examined by one-way ANOVA.
Effects of Bone Marrow Stromal Cell Transplantation on Histology and Necrotic Cortical Lesion Volume
Histologic examination of brain sections showed reactive gliosis in the periphery and bottom of the cortical freezing lesion (Figure 3). Ventral posteromedial thalamic neurons on the side of the lesion showed shrunken nuclei as well as slight neuronal loss associated with gliosis, considered to indicate retrograde degeneration (Figure 3). Such retrograde degenerative changes in the VPM in the thalamus were seen in both the MSC- and PBS-treated rats. It was difficult to identify any real differences in the incidence of shrunken nuclei and the degree of neuronal loss between these two groups, but the gliosis in the VPM was clearly less prominent in the MSC-treated than in the PBS-treated control rats (Figure 3). No osteoblasts nor adipocytes derived from MSCs were observed.
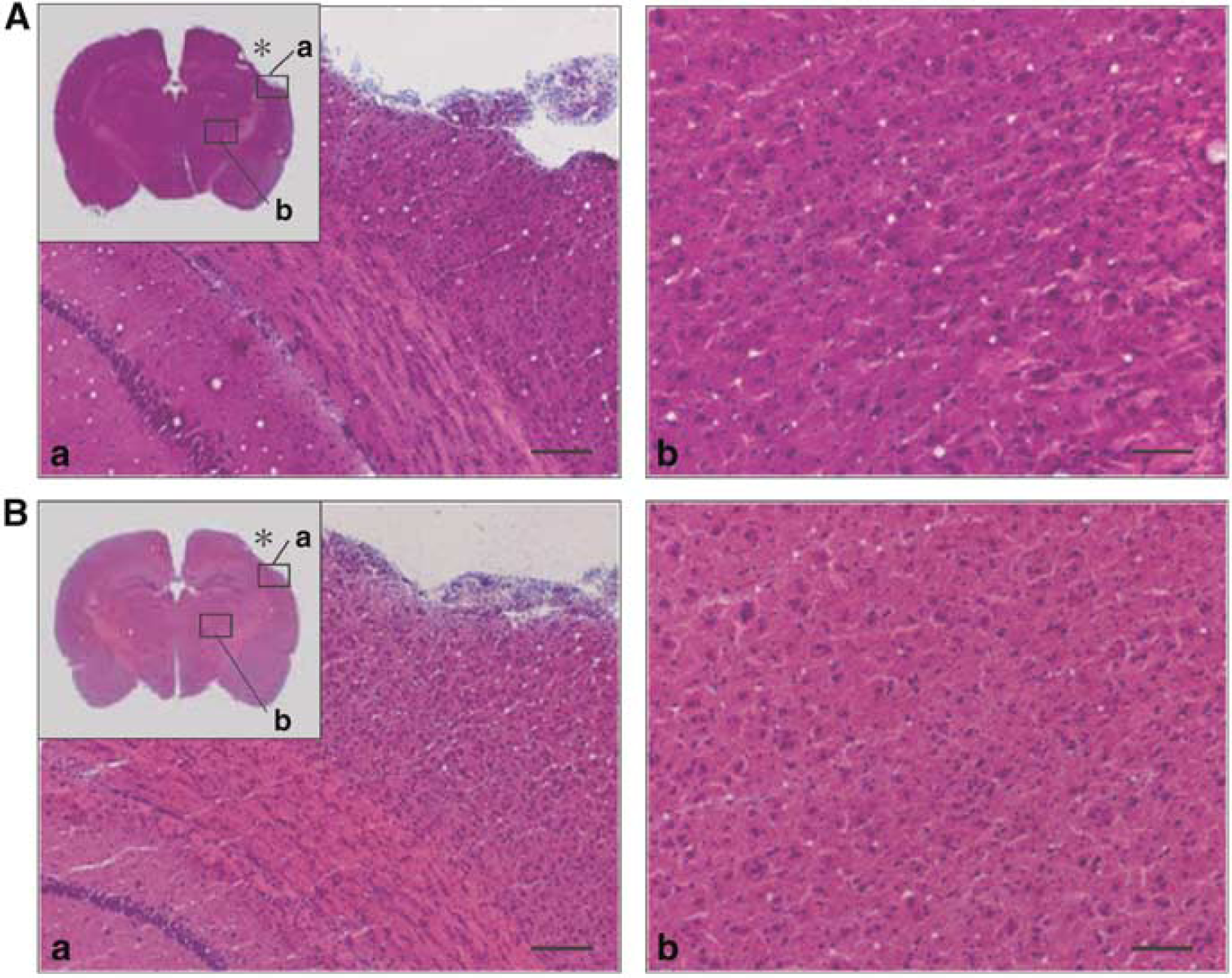
Photomicrographs of the freezing cortical lesion (a) and the ipsilateral VPM thalamic nucleus (b) after PBS injection (panel
The volume of the necrotic lesion in the cortex, determined from the digitized autoradiographs of the brain sections (Figure 4), was significantly smaller in the MSC-treated rats (3.77 ± 0.77 mm3) than in the PBS rats (5.26 ± 1.13 mm3) (P<0.05). The histology showed that MSC transplantation reduced the size of the primary cortical lesion and the degree of reactive gliosis, which may reflect retrograde degeneration in the ipsilateral thalamus.
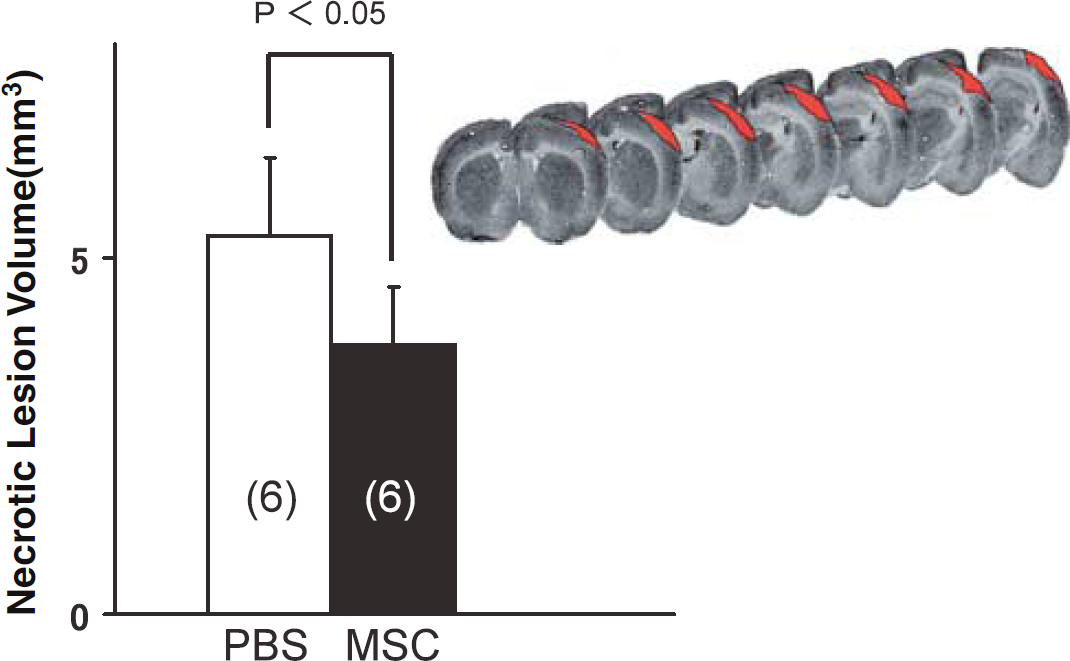
Volumes of cortical necrotic lesion in rats with PBS administration or MSC transplantation at 1 h after induction of the freezing cortical lesion. The red-colored area is the morphometrically measured necrotic area in digitized autoradiographic images of the brain coronal sections (top). Cortical necrotic volume was significantly decreased by MSC transplantation. Column heights and bars represent means ± standard deviations. The data were statistically analyzed by Student's t-test.
Effect of Bone Marrow Stromal Cell Transplantation on Local Cerebral Glucose Utilization
In the sham-operated rats, stimulation of the vibrissae on only the right side of the face resulted in statistically significantly higher rates of lCMRglc in the ipsilateral (right) PSTN (+49%) and SPTN (+94%) and contralateral (left) VPM thalamic nucleus (+29%), barrel cortex (SI, +67%), and periphery of the barrel cortex (SII, +33%) than those in the opposite unstimulated side (Table 2, Figure 5). In the MSC- and PBS-treated rats, lCMRglc was comparably stimulated in the PSTN and SPTN (P<0.01) ipsilateral to the right whisker stroking. The responses of lCMRglc to stimulation were, however, quite different in the thalamus and periphery of the cortical barrel field of the MSC- and PBS-treated rats. In the PBS-treated rats, lCMRglc was slightly lower in the left VPM (75 ± 8 μmol/100 g min) than in the right (79 ± 7 μmol/100 g min) and not statistically significantly higher in the periphery of the left than in the right barrel cortex, despite the fact that the structures on the left were in the stimulated side of the pathway. In contrast, in the MSC-treated rats, lCMRglc was significantly higher in the stimulated left VPM (85 ± 8 μmol/100 g min) than in the right unstimulated side (80 ± 6 μmol/100 g min) (P<0.01) and in the periphery of the stimulated left barrel cortex (107 ± 10 μmol/100 g min) than in the unstimulated right side (90 ± 10 μmol/100 g min) (P<0.01). Also, in the PBS but not MSC-treated rats, lCMRglc was significantly decreased in the left ventral anterior thalamic nucleus (P<0.05).
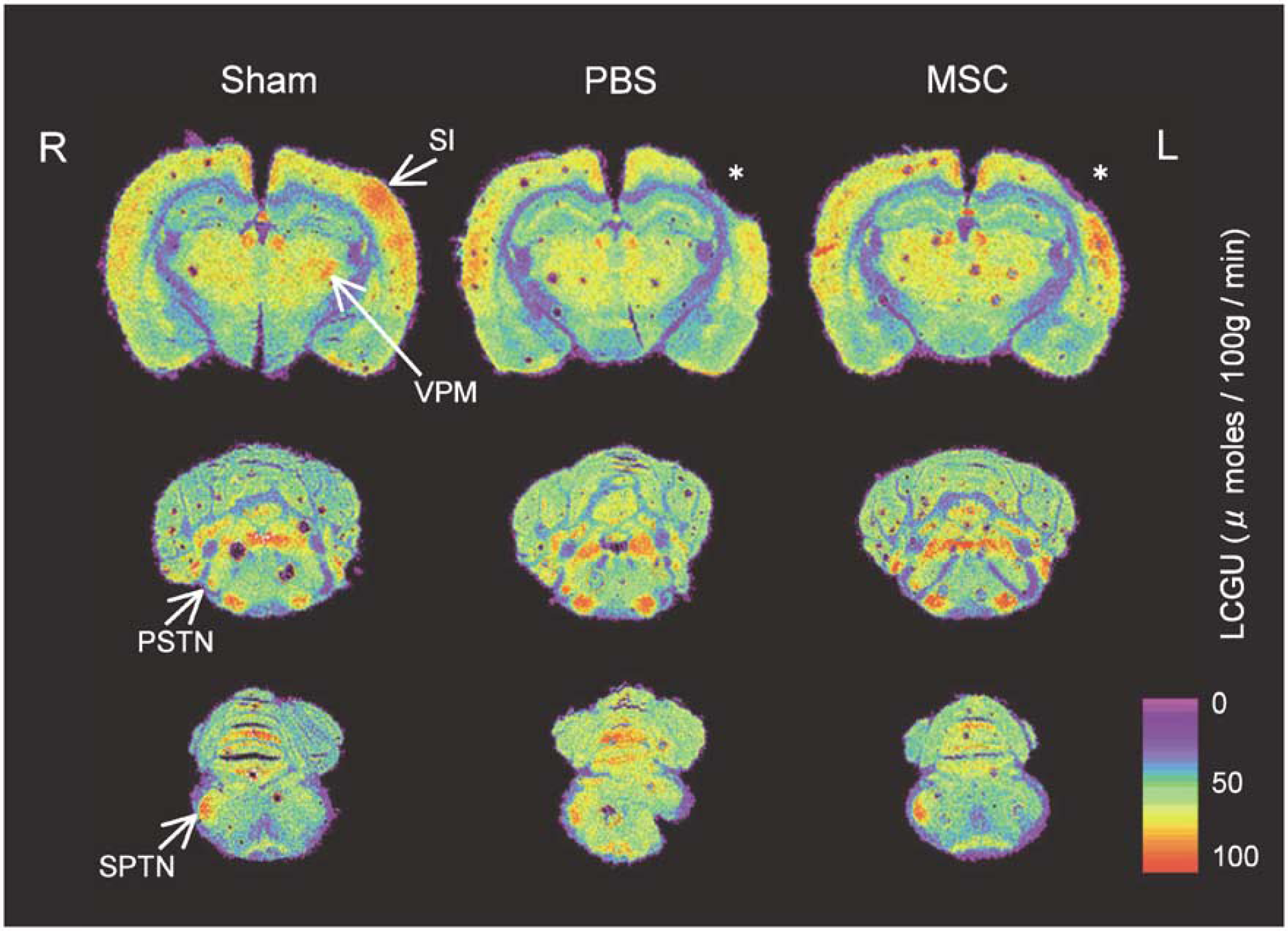
Digitized [14C]DG autoradiograms showing local rates of lCMRglc quantitatively encoded in color from rats with sham operation (left column); rats with freezing cortical lesion (asterisk); control rats with PBS administration (center column); and rats with MSC transplantation (right column). Unilateral whisker sensory stimulation markedly stimulated lCMRglc unilaterally in four structures of the functionally activated whisker-barrel sensory pathway, that is, SPTN, PSTN, VPM, and barrel field of the Sl, in the sham-operated rats. These increases in lCMRglc evoked by unilateral whisker stimulation were diminished in the left VPM and the periphery of the Si in the rats administered PBS but restored, at least partially, in the VPM and periphery of the Si in the rats with MSCs transplantation. R, right; L, left.
Local cerebral glucose utilization (μmol/100 g min)
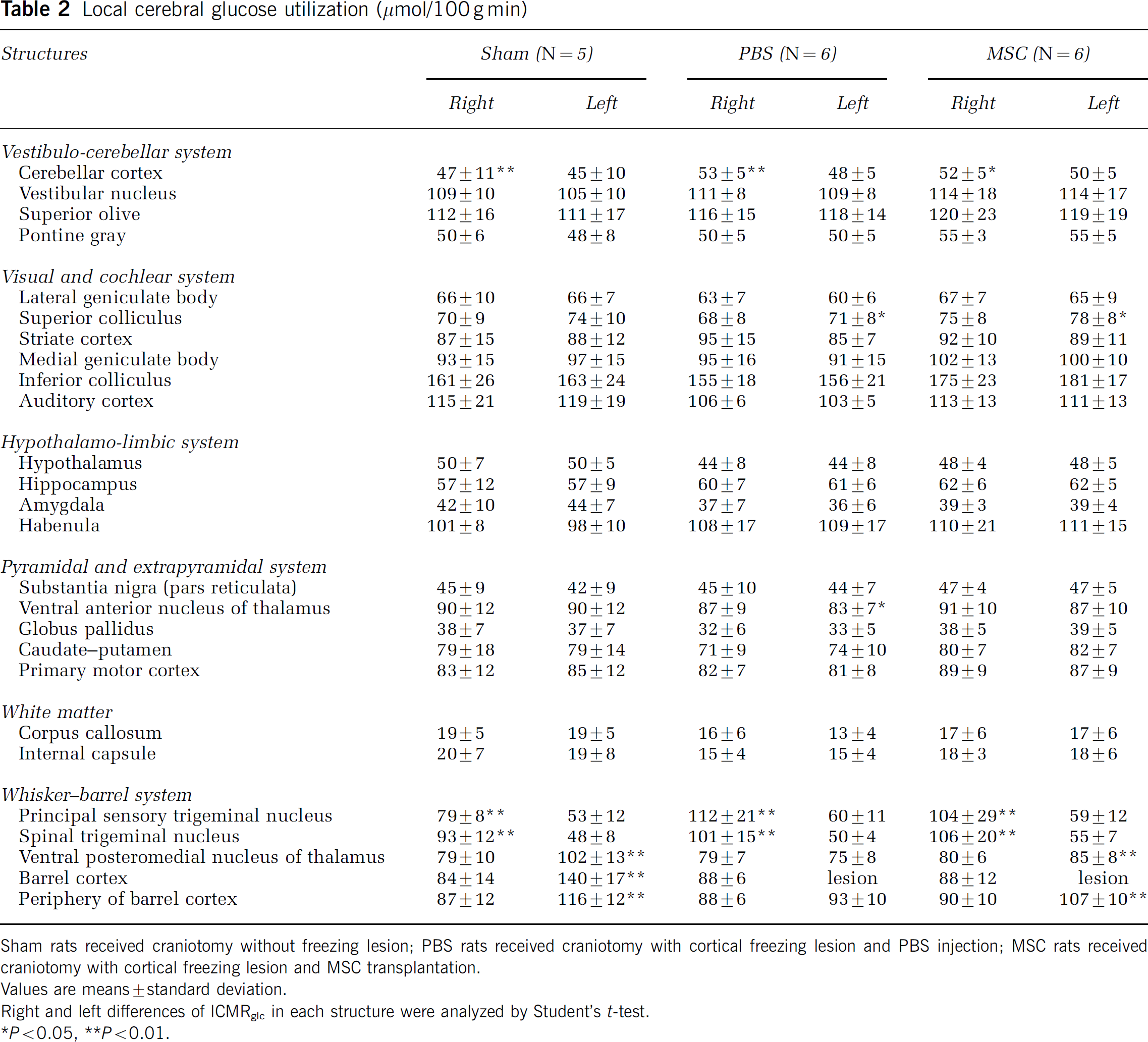
Sham rats received craniotomy without freezing lesion; PBS rats received craniotomy with cortical freezing lesion and PBS injection; MSC rats received craniotomy with cortical freezing lesion and MSC transplantation.
Values are means±standard deviation.
Right and left differences of lCMRglc in each structure were analyzed by Student's t-test.
Pμ0.05,
P<0.01.
The ratios of lCMRglc in the stimulated to unstimulated sides, right/left for SPTN and PSTN and left/right for VPM nucleus and barrel cortex, are shown in Figure 6. These ratios in the SPTN and PSTN did not differ statistically significantly among the three experimental groups (ANOVA), showing that neither PBS- or MSC-treatment affected functional metabolic activation in the primary synaptic regions of the pathway. In the VPM nucleus and the periphery of the barrel cortex, however, the ratios were markedly depressed by both treatments although even at 1 h after transplantation the ratios were significantly better in the MSC-treated than the PBS rats in the VPM nucleus (PBS, 0.94 ± 0.07 versus MSC, 1.05 ± 0.10) (P<0.05) and the periphery of the barrel cortex (PBS, 1.07 ± 0.03 versus MSC, 1.19 ± 0.06) (P<0.05) (Figures 5 and 6). These findings indicate that the MSC-treated rats had recovered some degree of metabolic activation in response to whisker stimulation in the VPM nucleus and periphery of the lesion in the barrel cortical field.
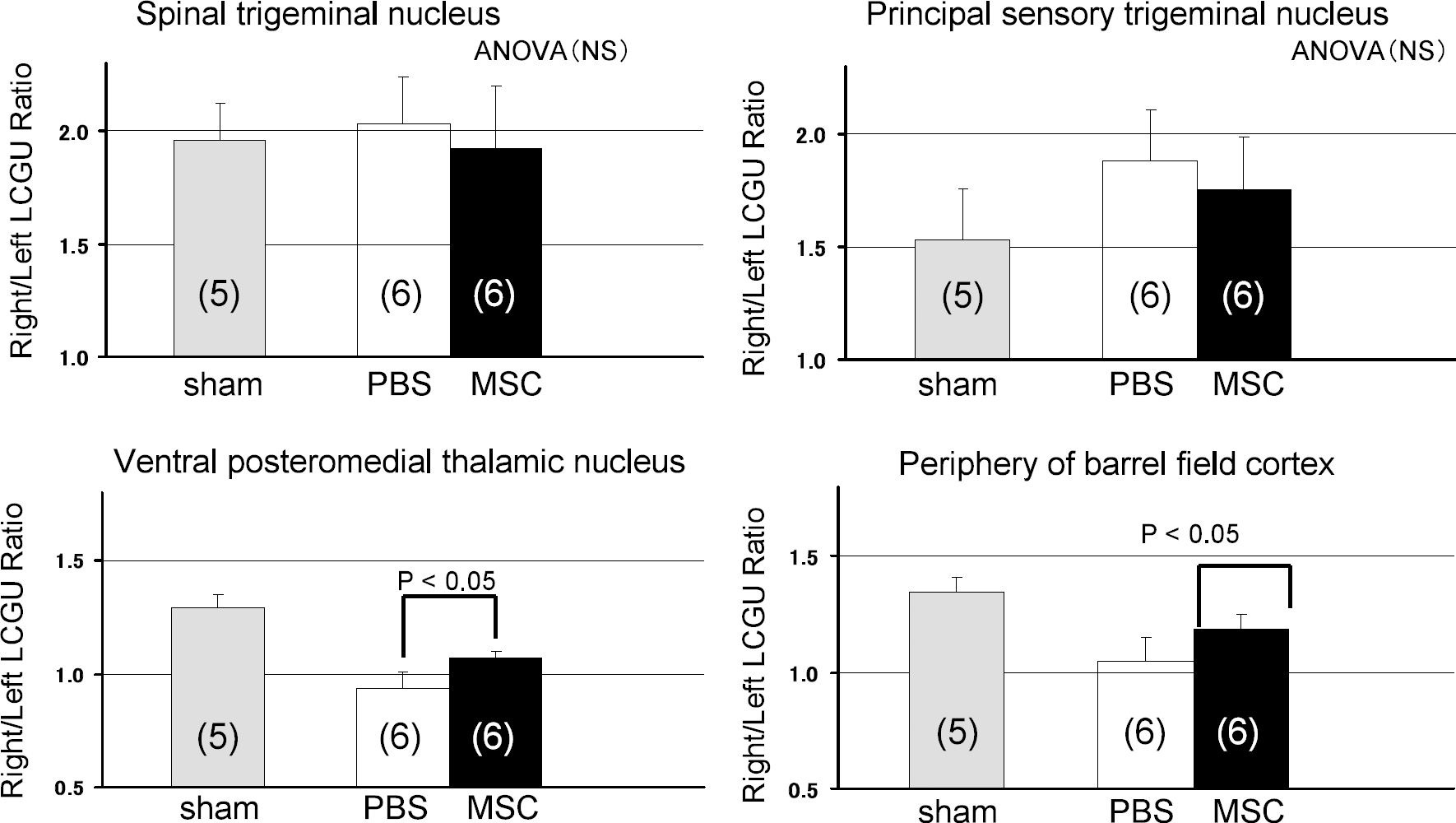
Effects of either MSC transplantation or control PBS administration after induction of freezing cortical lesion on the right/left or left/right (always stimulated/unstimulated sides) ratios of local cerebral glucose utilization (LCGU) in four structures of the whisker-barrel sensory pathway during unilateral whisker sensory stimulation. Column heights and bars represent mean ± standard deviation. Data were analyzed by one-way ANOVA and Student's t-tests. The stimulated/unstimulated lCMRglc ratios did not differ statistically significantly among the three experimental groups (ANOVA) in the spinal trigeminal nucleus and principal sensory trigeminal nucleus. The stimulated/unstimulated lCMRglc ratios were statistically significantly improved in both the ventral posteromedial thalamic nucleus and periphery of the barrel field cortex in the rats with MSC transplantation.
Discussion
The results of this study confirm previous observations that unilateral vibrissal stimulation increases lCMRglc in the ipsilateral brain stem trigeminal nuclei (PSTN and SPTN), the contralateral VPM nucleus in the thalamus, and the contralateral SI and SII in the neocortex as well as in other structures that receive inputs from the brain stem trigeminal nuclei, for example, the ipsilateral cerebellum and contralateral superior colliculus (Hall and Lindholm, 1974; Gonzalez and Sharp, 1985; Sharp and Gonzalez, 1985; Nakao et al, 2001). They also show that freezing lesions in the rat barrel cortical field suppress the functional metabolic responses in the VPM nucleus, while the brain stem trigeminal nuclei in the pathway retain normal metabolic activation to whisker stimulation.
As previously reported (Kataoka et al, 1989; Shiraishi et al, 1989; Ginsberg et al, 1989; Pappius, 1981; Hosokawa et al, 1985; Ciricillo et al, 1994), we also found attenuation of metabolic activation by whisker stimulation in VPM nuclei ipsilateral to unilateral cortical lesions. Five weeks after unilateral cortical barrel ablation, lCMRglc during whisker stimulation was 22% lower in the VPM nucleus ipsilateral to the lesion than in the contralateral side, whereas both resting and activated lCMRglc remained unaffected in the brain stem trigeminal nuclei (Ciricillo et al, 1994). Failure of whisker stimulation to activate axotomized VPM neurons despite metabolic activation in the trigeminal brain stem nuclei might be because of decreased excitability in the initial segment (axon hillock) of the axotomized neurons (Titmus and Faber, 1990; Tokuno et al, 1992; Yuan et al, 1986). Also, axotomy of cortical afferents to VPM neurons may deprive them of trophic factors released by cortical neurons that may maintain the excitability and metabolic activity in the neurons (Ciricillo et al, 1994; Eagleson et al, 1992; Sharp and Gonzalez, 1986; Sorensen et al, 1989).
Axotomized thalamic neurons show a number of structural and enzymatic changes, such as glucose hypometabolism, reduced succinate dehydrogenase activity, mitochondrial swelling, and synaptic degeneration, within a few days after induction of the cortical lesion (Okonkwo and Povlishock, 1999; Yamada et al, 1991; Kataoka et al, 1989), whereas microscopic changes within their somas are not seen within the first week after cortical damage. Retrograde degeneration, such as shrinkage and death of thalamic neurons, takes 1 to 3 weeks to appear after axonal injury, and these histologic changes are often associated with gliosis (Iizuka et al, 1990; Barron et al, 1973; Yamada et al, 1991; Kataoka et al, 1989; Hosokawa et al, 1985). We also found similar retrograde degenerative changes in the affected VPM thalamic nucleus 3 weeks after the induction of the freezing lesion in the barrel cortex. The mechanism of cell damage in neurons after axotomy is unclear. Axonal injury causes excessive Ca2+ ion influx, which induces calpain activation and mitochondrial swelling associated with cytochrome c release. Increased cytosolic cytochrome c activates caspase activity (Büki et al, 2000; Wolf et al, 2001; Okonkwo and Povlishock, 1999; Pettus and Povlishock, 1996), which is a key enzyme in the process of apotosis.
In the present study, the metabolic response to whisker stimulation in the VPM nucleus was significantly greater in the MSC-treated than in the PBS-treated rats, and this response was associated with metabolic activation in the periphery of the freezing lesion in the barrel field cortex. Morphometric examination showed that MSC transplantation also significantly reduced the volume of necrotic tissue in the freezing lesion in the barrel cortex as well as the degree of reactive gliosis, which may reflect the retrograde degenerative changes in the thalamus. The recovery of the glucose metabolic response in the VPM nucleus may be secondary to the reduced size of the cortical lesion after cell treatment.
Therapeutic MSC transplantation in rats subjected to various types of brain injury, such as traumatic brain injury and cerebral infarction, promotes neurologic functional recovery (Lu et al, 2001a, b; Mahmood et al, 2001; Chen J et al, 2001; Chopp and Li, 2002). Although the long-term strategy of replacement of injured tissue by cells differentiated from stem cells is to treat neuronal injury, the low level of MSC differentiation into neuronal and glial cells in the acute phase suggests that any functional recovery might be because of more production of trophic factors that protect the brain from the insult rather than because of differentiation of MSCs into cerebral tissue (Chen J et al, 2001; Lu et al, 2001a, b; Mahmood et al, 2001). Bone marrow stromal cells are known to secrete various cytokines and growth factors, and their secretive properties are influenced by the microenvironment (Takai et al, 1997; Labouyrie et al, 1999; Dormady et al, 2001; Chen X et al, 2002). Human MSCs cultured in the presence of extracts from traumatized rat brain tissue produce time-dependent increases in the levels of brain-derived neurotrophic factor, nerve growth factor, vascular endothelial growth factor, and hepatocyte-growth factor (Chen X et al, 2002), and these are believed to repair and protect injured brain tissue from various kinds of insults (Chopp and Li, 2002; Takami et al, 1992; Reilly and Kumari, 1996).
Intravenous MSC infusions promote expression of endogenous basic fibroblastic growth factor (bFGF) in the ischemic boundary zone in rat stroke models (Chen J et al, 2003). Several members of the fibroblastic growth factor family, particularly bFGF, are intimately involved in neuronal protection and repair after various types of brain injury (Alzheimer and Werner, 2002; Takami et al, 1992; Reilly and Kumari, 1996). Intracisternal administration of bFGF has also been found to prevent retrograde degeneration in thalamic neurons after ischemic axonal injury in the rat (Yamada et al, 1991). Transection of the fimbria-fornix caused retrograde degeneration in septal hippocampal cholinergic neurons, and intraventricular administration of nerve growth factor prevented the retrograde neuronal death (Hefti, 1986; Williams et al, 1986). These findings suggest that administration of trophic factors or transplantation of MSCs may serve to rescue axotomized neurons without retrograde degeneration. Presumably, the dendritic trees of the thalamic neurons are maintained by the transplanted cells in the cortex, thus facilitating synaptic input from the brain trigeminal nuclei to the VPM thalamic neurons, resulting in increased glucose metabolism in the axotomized thalamic neurons.
As previously observed (Azizi et al, 1998; Chen J et al, 2001), we also found that MSCs transplanted into the center and beneath a cortical freezing lesion migrated mainly into the periphery of the primary lesion; few cells migrated further into the corpus callosum. This migration pattern may reflect the vasogenic fluid flow from the freezing lesion (Klatzo et al, 1965), suggesting that extracellular edema fluid may facilitate migration of transplanted MSCs. Also, MSCs transplanted into the freezing lesion did not differentiate into neuronal, glial, or mesenchymal cells. The properties and facilitation of cellular differentiation might be influenced by the type of brain injury and surrounding microenvironment (Chen X et al, 2002). We assume that the transplanted MSCs in the freezing lesion were not a source of cellular replacement in the injured brain but served rather as a source of various growth factors that protected against cell death in the periphery of the primary lesion and tended to protect against subsequent retrograde degeneration of axotomized VPM neurons.
Activation of a neural pathway increases local ion-pumping, which then stimulates glucose utilization to provide the energy needed to restore the ionic gradients (Mata et al, 1980; Sokoloff, 1982). This coupling of functional activity and lCMRglc has been used to map local functional activity in neural pathways and to quantify relationships between stimulus intensity and magnitude of the metabolic response (Sokoloff et al, 1977; Sokoloff, 1982). In the present study, glucose metabolic mapping was used to show and quantify functional recovery in injured trigeminal neural pathways after intracerebral transplantation of MSCs. Transplantation of adult MSCs into cortical freezing lesions reduced the size of the primary lesions, and moreover, if performed early enough after induction of the freezing lesion, functional and metabolic responses of the VPM nucleus to activation of brain stem trigeminal nuclei by whisker stimulation could be restored by 3 weeks after the transplantation. These results suggest the potential usefulness of adult MSC transplantation as a treatment for injured neuronal networks.
