Abstract
Bone marrow stromal cells (BMSCs) facilitate functional recovery in rats after stroke when administered acutely (1 day) or subacutely (7 days). In this study, we postponed the time of cell transplantation to 1 month after stroke. Female retired breeder rats were subjected to 2 h of middle cerebral artery occlusion (MCAo). Male BMSCs (3×106) or phosphate-buffered saline were administered intravenously, and the animals were killed 3 months later. An additional population of nontreated rats was killed at 1 month after MCAo. Significant recovery of behavior was found in BMSC-treated rats beginning at 1 month after cell injection in the modified neurologic severity score test and the adhesive-removal test compared with control animals (P < 0.05). In situ hybridization showed that BMSCs survived and preferentially localized to the ipsilateral hemisphere. Double staining revealed that approximately 13% and 6% Y-chromosome-positive cells expressed the astrocyte marker, glial fibrillary acidic protein, and the neuronal marker, microtubule-associated protein-2, respectively. In addition, BMSC treatment reduced scar thickness, and increased the number of proliferating cells and oligodendrocyte precursor cells along the subventricular zone in the ipsilateral hemisphere. Expression of the chemokine stromal-cell-derived factor-1 (SDF-1) was significantly increased along the ischemic boundary zone compared with the corresponding areas in the contralateral hemisphere at 1 month and 4 months (P < 0.01) after stroke. The SDF-1 receptor, CXC-chemokine receptor-4 (CXCR4), was expressed in BMSCs both in vitro and in vivo. Our data show that the time window of BMSC therapy is at least 1 month after stroke; the interaction of SDF-1/CXCR4 may contribute to the trafficking of transplanted BMSCs.
Introduction
Bone marrow stromal cell (BMSC) transplantation has been employed in animal models of stroke (Chen et al, 2001b; Li et al, 2001; Rempe and Kent 2002; Song et al, 2004) as well as in human stroke patients (Bang et al, 2005). Promising data are accumulating regarding the effectiveness of this neurorestorative approach: BMSCs, when administered acutely 1 day or subacutely at 7 days after stroke in animal models, reduce neurologic deficits after stroke, and the beneficial effects persist for at least 4 months after middle cerebral artery occlusion (MCAo) (Chopp and Li 2002; Li et al, 2005). In an effort to investigate the time window for BMSC therapy, we postponed the time of cell transplantation to 1 month after stroke. We found that BMSCs, when administered to the animals 1 month after stroke, significantly improved functional outcome. We also investigated the signals guiding the BMSCs into the injured brain, and found significant upregulation of the chemokine, stromal-cell-derived factor-1 (SDF-1), within the boundary region of the infarct and robust expression of the SDF-1 receptor, CXC-chemokine receptor-4 (CXCR4), on the BMSCs.
Materials and methods
Experimental procedures were approved by the Care of Experimental Animals Committee of Henry Ford Hospital.
Middle Cerebral Artery Occlusion
Female retired breeder Wistar rats (n = 25, 10 to 12 months old) weighing 350 to 450 g were employed in our experiment. Transient MCAo was induced using a method of intraluminal vascular occlusion modified in our laboratory (Chen et al, 1992). Briefly, rats were initially anesthetized with 3.5% halothane and maintained with 1.0% to 2.0% halothane in 70% N2O and 30% O2 using a facemask. The rectal temperature was controlled at 37°C with a feedback-regulated water heating system. The right common carotid artery, external carotid artery (ECA), and internal carotid artery (ICA) were exposed. A length of 3-0 monofilament nylon suture (∼ 19.5 mm), determined by the animal weight, with its tip rounded by heating near a flame, was advanced from the ECA into the lumen of the ICA until it blocked the origin of the MCA. Two hours after MCAo, animals were reanesthetized with halothane, and reperfusion was performed by withdrawal of the suture until the tip cleared the lumen of the ECA.
Cell Transplantation Procedures and Experimental Groups
Primary cultures of BMSCs were obtained from donor young adult male rats and BMSCs were separated, as described previously (Shen et al, 2006). One month after ischemia, randomly selected rats received BMSC administration. Animals were anesthetized with 3.5% halothane and then maintained with 1.0% to 2.0% halothane in N2O:O2 (2:1). Approximately 3 × 106 BMSCs in 1 mL Plasma-Lyte (n = 8) or control fluid (1 mL Plasma-Lyte, n = 8) was slowly injected intravenously (i.v.) over a 5-min period into each rat. Immunosuppressants were not used in any animal in this study. These animals were killed 4 months after MCAo. A third group (n = 9) of rats not subjected to any treatment was killed 1 month after MCAo. This group was employed to analyze the expression of SDF-1.
Behavioral Tests
In the two experimental groups that contained rats killed 4 months after MCAo, two behavioral tests were performed before MCAo (baseline), at 1 and 28 days after MCAo, before BMSC administration, and at 1, 2, 4, 8, and 12 weeks after cell administration by an investigator who was blinded to the experimental groups.
Modified neurologic severity scores (Chen et al, 2001b): Modified neurologic severity score (mNSS) is a composite of motor, sensory, balance, and reflex tests. Neurologic function was graded on a scale of 0 to 18 (normal score 0; maximal deficit score 18). In the severity scores of injury, one point is awarded for the exhibition of a specific abnormal behavior or for the lack of a tested reflex; thus, the higher the score, the more severe is the injury.
Adhesive-removal somatosensory test (Schallert et al, 1997): Two small pieces of adhesive-backed paper dots (113.1 mm2 before and 56.6 mm2 after treatment) were used as bilateral tactile stimuli occupying the distal–radial region on the wrist of each forelimb. The rat was then returned to its home-cage. The time to remove each stimulus was recorded during three trials per day. Individual trials were separated by at least 5 mins. Before surgery, the animals were trained for 3 days. Once the rats were able to remove the dots within 10 secs, they were subjected to MCAo.
Histologic and Immunohistochemical Assessment
Among all the animals, rats (n = 9) subjected to MCAo alone were killed at 1 month after surgery (only for SDF-1 immunostaining), whereas the others (n = 16, eight BMSCs treated, eight Plasma-Lyte controls) were allowed to survive for three additional months. Rat brains were fixed by transcardial perfusion with saline, followed by perfusion and immersion in 4% paraformaldehyde, and then brain blocks were embedded in paraffin. One coronal paraffin slide (6 m thick) from each of the seven brain blocks (2 mm thick) of each rat was stained with hematoxylin and eosin (H&E). The seven coronal brain sections were traced using a Global Lab Image analysis system (Data Translation, Marlboro, MA, USA) for lesion volume evaluation. A series of 6-μm-thick slides were cut from a standard paraffin block obtained from the center of the lesion of the forebrain, corresponding to coronal coordinates for bregma ∼1 to 1 mm for immunostaining.
After deparaffinizing, brain sections were placed in boiled citrate buffer (pH 6) in a microwave oven (650 to 720 W). After blocking in normal serum, sections were incubated with antibodies against glial fibrillary acidic protein (GFAP; dilution, 1:5,000; Dako, Carpinteria, CA, USA), NG2 (dilution, 1:800; Chemicon, Temecula, CA, USA), Ki-67 (dilution, 1:300; Abcam, Cambridge, UK), or SDF-1 (dilution, 1:250; Santa Cruz Biotech Inc., Santa Cruz, CA, USA) at 4°C overnight. Then, the sections were incubated with avidin–biotin–horseradish peroxidase complex and developed in 3′3′-diaminobenzidine tetrahydrochloride (DAB). Double-immunohistochemical staining was employed to visualize the cellular colocalization of SDF-1 and cell-specific markers: microtubule-associated protein-2 (MAP-2; dilution 1:200; Chemicon, Temecula, CA, USA) for neurons; GFAP for astrocytes; or Von Willebrand factor (dilution, 1:400; Dako, Carpinteria, CA, USA) for endothelial cells. The fluorescein isothiocyanate-conjugated antibody (FITC; Jackson Immunoresearch, Baltimore, PA, USA) and CY3-conjugated antibody were employed for double immunoreactivity identification. Negative control sections from each animal received identical preparations for immunohistochemical staining, except that the primary antibodies were omitted.
In Situ Hybridization
Fluorescein isothiocyanate-conjugated in situ hybridization (FISH) was performed as described previously with some modifications (Chen et al, 2003). Briefly, the slides were digested with proteinase K (50 μg/mL) for 20 mins at 37°C. The prepared cDNA probe for Y-chromosome was labeled by a Random Primer DIG labeling and Detection Kit (Boehringer Mannheim, Indianapolis, IN, USA). Hybridizations were performed in a hybridization mixture (consisting of 50% deionized formamide, 10% salmon test DNA, 5% dextran sulfate, 1 × Denhardt's solution, 2 × standard saline citrate, 250 μg/mL dithiothreitol, 250 μg/mL poly-A, and 500 ng digoxigenin-labeled probe) at 50°C for 40 h. The digoxigenin-labeled Y-chromosome-positive BMSCs were visualized using the fluorescent antibody enhancer set (Boehringer Mannheim). The slides were then counterstained with 10 ng/mL of propidium iodide for nuclear staining and mounted with antifade solution and coverslips. Negative control slides from each animal received identical staining preparation, except that the probe or the antidigoxigen antibodies were omitted.
To visualize the cellular colocalization of the Y-chromosome and certain markers in the same cell, the FITC-conjugated antibody and DAB was employed for double labeling. Each coronal section was first treated with cell-type-specific antibodies: MAP-2 for neurons, GFAP for astrocytes, or CXCR4, and was visualized with DAB staining. Subsequently, FISH staining was employed for the identification of male BMSCs. Negative control sections from each animal received identical preparations for immunohistochemical staining, except that the primary antibody was omitted.
Quantification
Immunoreactive cells and in situ hybridization-positive cells were analyzed with NIH image software (Image J) based on the evaluation of an average of three histology slides (6 μm thick, 54 μm interval, every 10th slide) from the standard block of each animal.
The number of Y-chromosome-positive cells was counted in the ischemic boundary zone. In each animal, approximate 200 Y-chromosome cells were measured to obtain the percentage of Y-chromosome cell colocalized with cell-type-specific markers (MAP-2 and GFAP) by double staining.
The thickness of scar was measured and averaged from five fields of view along the ischemic boundary zone within the ipsilateral striatum. NG2-positive oligodendrocyte precursor cells and Ki-67-positive proliferating cells in the region of subventricular zone (SVZ) were counted in a 100-μm-thick band encircling the ependymal layer of the lateral ventricles on each slide. For SDF-1 immunostaining, 8 fields of view along the ischemic boundary zone and the corresponding areas in the contralateral hemisphere were digitalized under a × 20 objective and the percentage of immunoreactive-positive area was calculated and averaged.
In Vitro Assay
Reverse Transcription Polymerase Chain Reaction Analysis: Total RNA was isolated from cultured BMSCs using absolutely RNA Microprep Kit (Stratagene, La Jolla, CA, USA) according to the manufacturer's instructions. Ribogreen (Molecular Probes, Carlsbad, CA, USA) was used to determine RNA yield. RNA was subsequently reverse transcribed to cDNA with SuperScript First-strand Synthesis System (Invitrogen, Carlsbad, CA, USA). The primer for CXCR4 was designed using Primer Express software (ABI): 5′ to 3′: Fow, GGTCTGGAGACTATGACTC-CA; Rev, GTGCTGGAACTGGAACACCA. The PCR cycles consist of denaturation at 94°C for 1 min, annealing at 56°C for 40 secs, and extension at 72°C for 1 min for 40 cycles. A 10 μL PCR product was size-separated by electrophoresis on a 2% ethidium-bromide-containing agarose gel and photographed.
Immunocytochemistry: Bone marrow stromal cells cultured on coverslips were fixed with 4% paraformaldehyde for 30 mins. After blocking in normal serum, coverslips were incubated with CXCR4 antibody (dilution, 1:200; Chemicon, Temecula, CA, USA) at 4°C overnight, and then were visualized by CY3-conjugated second antibody. Negative control cells receive identical preparations for immunocytochemical staining, except that the primary antibody was omitted.
Statistical Analysis
Two behavior scores (the adhesive-removal test and mNSS test) were evaluated for normality. Repeated measures analysis was conducted to test the treatment effect on the behavior score. As a result, ranked data were considered because of a lack of normality. These functional outcome measures were incorporated into a global test, a sophisticated integrated outcome analysis, which considers correlation of outcomes (Tilley et al, 1996). The BMSC effect was significant, with a P-value for the global test < 0.05. The BMSC effect was further tested on each individual test score, if the global test was significant. Immunohistochemistry data were analyzed using Student's t-test. Data are presented as mean ± s.e.
Results
Neurologic Functional Test
Functional status was balanced between the BMSC-treated and the control retired breeder rats at baseline, and no significant difference on each behavioral score (mNSS and adhesive-removal test) was detected at day 1 or day 28 after MCAo. Figure 1 shows the temporal profile of functional recovery in control and BMSC-treated rats. Significant functional improvement was detected on the BMSC group at each time point starting 4 weeks after treatment on both tests.
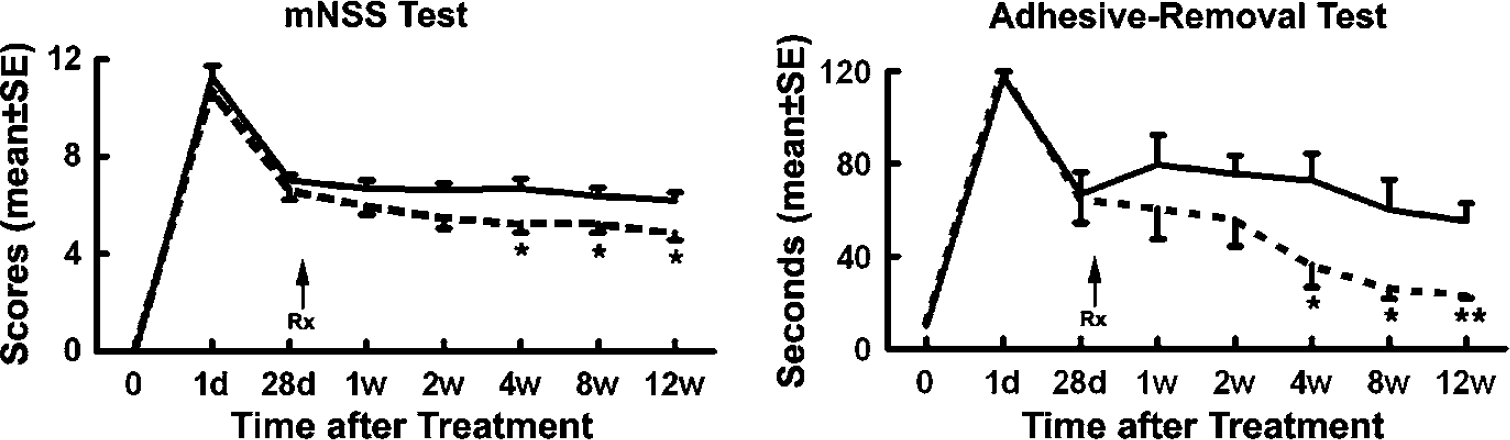
Line graphs showing the temporal profile of functional recovery in control and bone marrow stromal cell (BMSC)-treated rats. Significant BMSC effects are detected at each time point starting 4 weeks after treatment. *P < 0.05, **P < 0.01 versus control group.
Histology
Lesion volume: By H&E staining, reproducible neuronal damage was observed in the ischemic core of the striatum and cortex in all rats subjected to MCAo. At 4 months after stroke, ischemic core tissues transformed into various sizes of cysts in all rats treated without or with BMSCs. The damaged tissue consisted of a central cavitation surrounded by a scar wall (densely arranged GFAP-positive fibrils) and a boundary zone. The lesion volume showed a trend for reduction after BMSC treatment (39.0% ± 4.1% in control group versus 29.7%±4.5% in treated group, P>0.05). The reduction, however, was not statistically significant.
Donor Cells in the Brain
Within the brain tissue, cells derived from male BMSCs were identified by Y-chromosome staining. No Y-chromosome-positive cells were found without BMSC treatment. Bone marrow stromal cells identified by the Y-chromosomes localized solely to intact nuclei and were distributed throughout the damaged brain of recipient rats with the majority located in the IBZ (Figures 2A–2C). The number of donor-derived Y-chromosome-positive cells in the ipsilateral hemisphere was 59.5±10/section. Double-staining immunohistochemistry of brain sections revealed that some Y-chromosome-positive cells were reactive for the neuronal marker (MAP-2) (Figures 2D–2G) and for the astrocyte marker (GFAP) (Figures 2H–2K). The percentage of Y-chromo-some-positive cells that express MAP-2 and GFAP protein was 6.38%± 0.48% and 13.19%±1.3%, respectively.
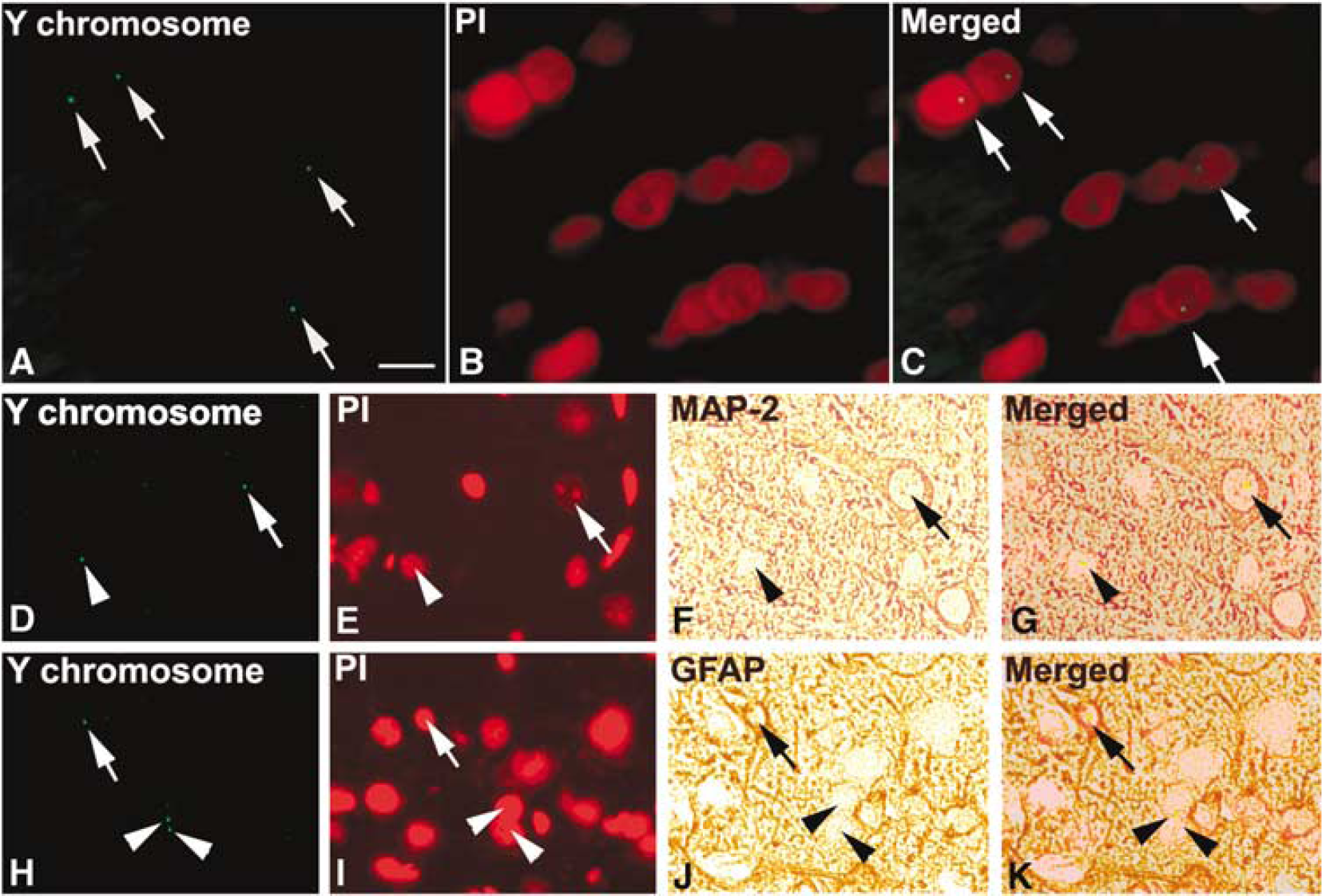
(
Changes along the Subventricular Zone
At 4 months after MCAo, high-density GFAP-positive fibrils were interwoven into a scar wall around the cystic core (Figures 3A and 3B). After BMSC treatment, a significantly (P < 0.05) thinner scar wall (Figures 3B and 3C) was observed compared with that of the control MCAo rats (Figures 3A and 3C).
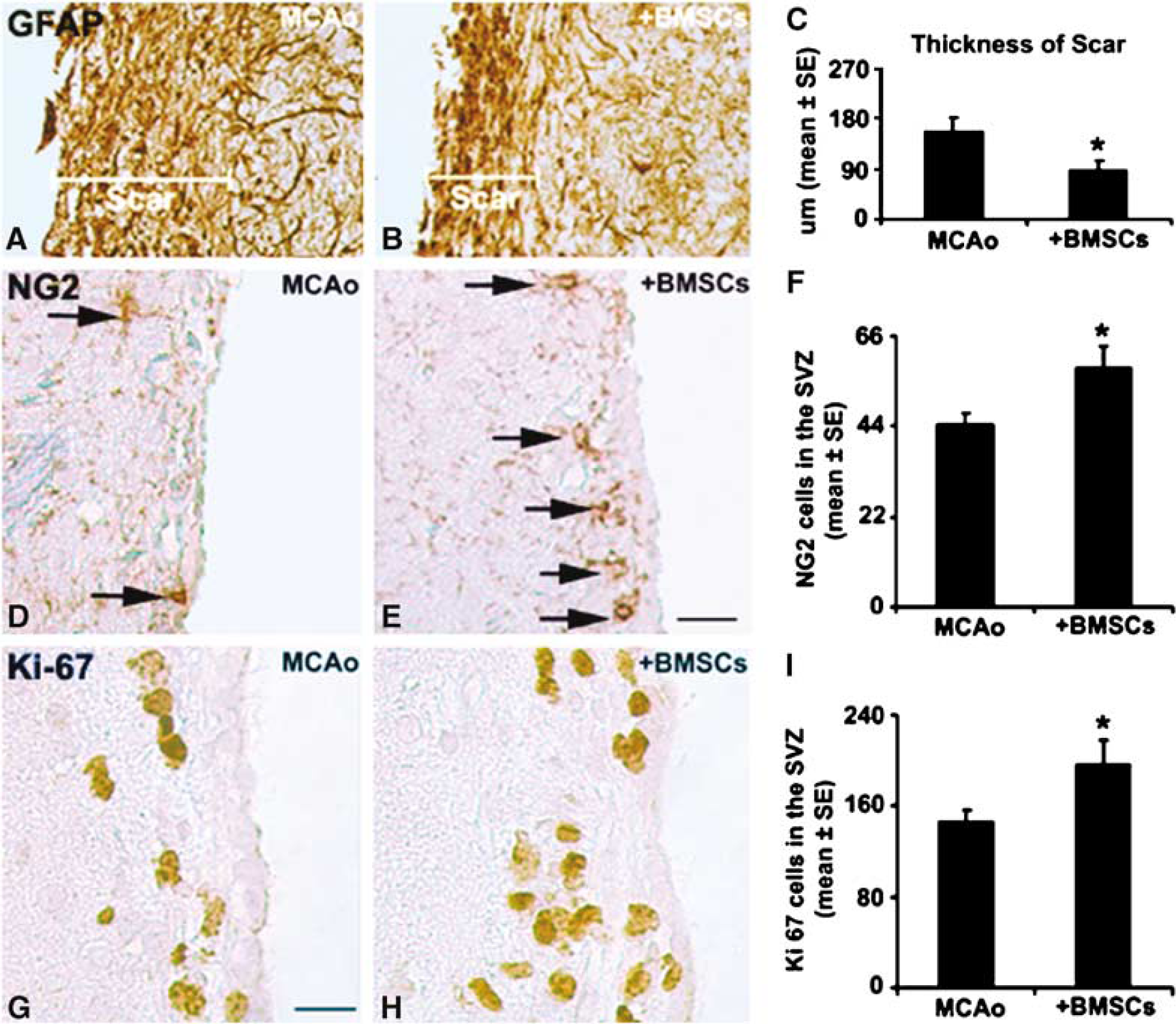
(
NG2 chondroitin sulfate proteoglycan is a specific marker of oligodendrocyte progenitor cells (OPCs) (Dawson et al, 2000). Bone marrow stromal cell administration significantly enhanced the number of NG2 cells in the SVZ compared with MCAo alone (P < 0.05; Figures 3D–3F).
The Ki-67 protein, which is present during all active phases of the cell cycle (G (1), S, G (2), and mitosis), but is absent from resting cells (G (0)), is a marker of cell proliferation (Scholzen and Gerdes, 2000). As shown in Figures 3G–3I, the majority of Ki-67-immunoreactive cells were located along the SVZ. Bone marrow stromal cell treatment also significantly increased the number of proliferating cells in the SVZ compared with control values (P < 0.05).
Stromal-Cell-Derived Factor-1-Related Immunohistochemistry
Stromal-cell-derived factor-1 protein expression, as detected by anti-SDF-1 antibody, was upregulated in the infarcted hemisphere at the time of cell administration (1 month after MCAo), and it persisted at least 4 months after occlusion (Figure 4B). The SDF-1 expression pattern was predominantly localized to the infarct boundary areas. Double-staining immunohistochemistry of brain sections revealed that SDF-1 labeling was mainly associated with blood vessels (Figures 4C–4E); however, it was also colocalized with neuronal (Figures 4F–4H) and astrocytic (Figures 4I–4K) markers. The intensity of SDF-1 immunoreactivity decreased over time between 1 and 4 months after MCAo, but remained significantly higher than that of the nonaffected contralateral side at 4 months after MCAo. Bone marrow stromal cell administration showed no effects on the pattern and density of SDF-1 expression on both hemispheres (data not shown).
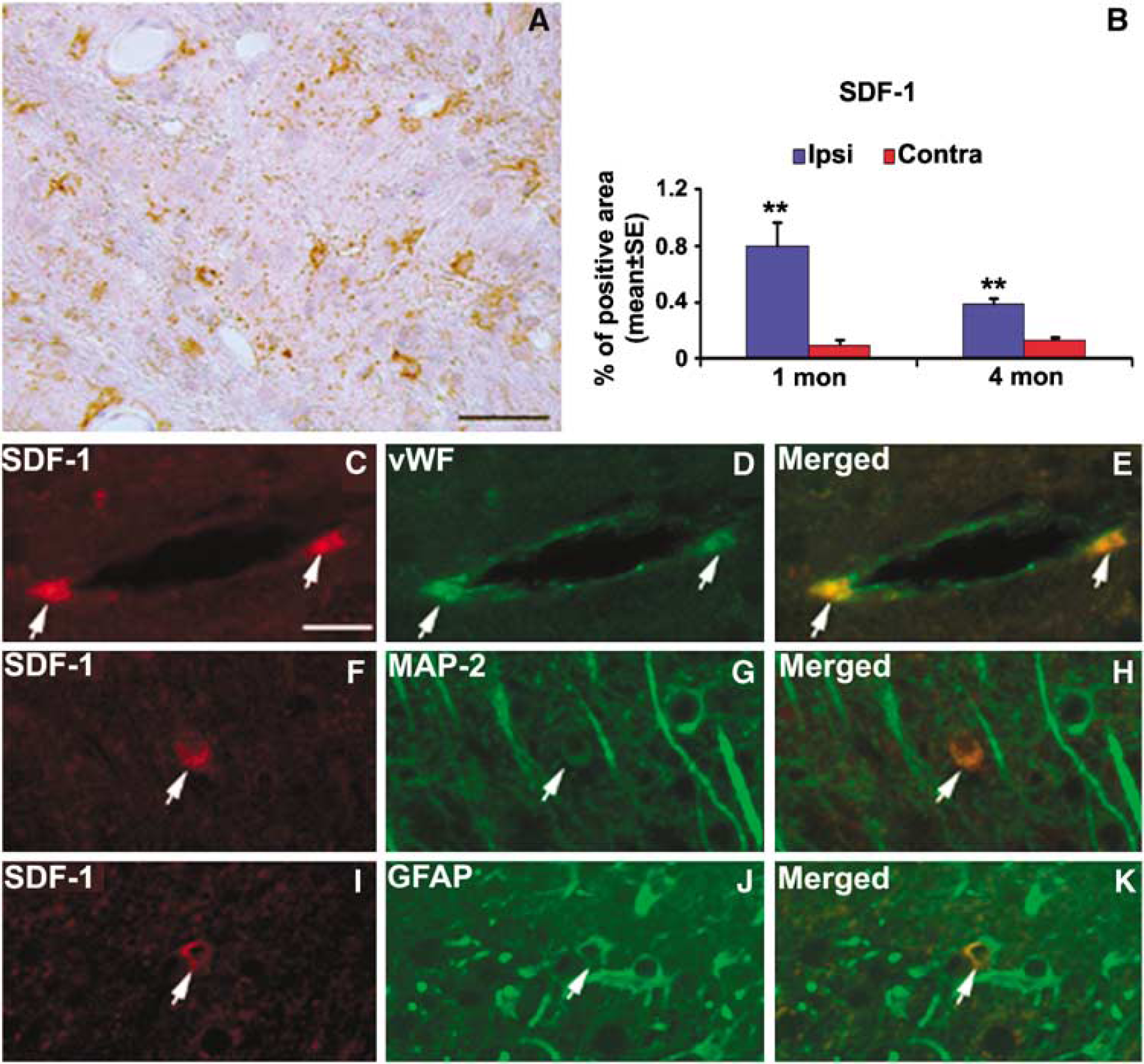
Photomicrographs showing the morphologic characteristics of stromal-cell-derived factor-1 (SDF-1)-positive cells in the brain. (
Expression of Stromal-Cell-Derived Factor-1 Receptor, CXC-chemokine receptor-4, in Bone Marrow Stromal Cells
We examined the expression of the SDF-1 receptor, CXCR4, in BMSCs by reverse transcription-polymerase chain reaction (PCR) and immunocytochemistry. Stromal-cell-derived factor-1 mRNA was present in BMSCs (Figure 5A). Immunocytochemical analysis showed CXCR4 protein localized to the cell membrane and in the cytoplasm of BMSCs (Figure 5B). In addition, double staining of ISH and CXCR4 immunohistochemistry showed that donor cells in the brain expressed CXCR4 too (Figure 5C).
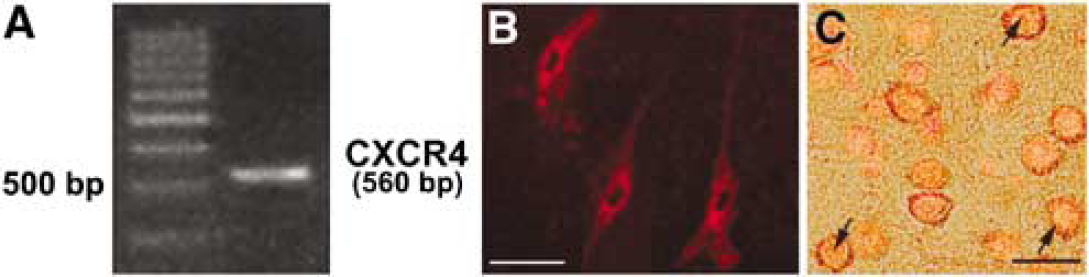
Expression of CXC-chemokine receptor-4 (CXCR4) by bone marrow stromal cell (BMSCs) at mRNA and protein levels. (
Discussion
We show for the first time that i.v. injection of male BMSCs at 1 month after stroke into older female rats significantly improves neurologic functional recovery. Concomitant with the therapeutic benefit is reduced scarring, an increase of proliferating cells, including OPCs, along the neurogenic zone in the ischemic hemisphere. Of interest is the finding that the chemokine SDF-1 is upregulated in the ischemic boundary zone at the time of cell transplantation (1 month after MCAo), and this increment persists until 4 months after the onset of focal brain ischemia. Since CXCR4, the specific receptor of SDF-1, is expressed in BMSCs at both mRNA and protein levels, our data suggest that the interaction of SDF-1 with its receptor, CXCR4, mediates the trafficking of BMSCs to the impaired site in the brain, especially in the chronic stage of stroke.
Severe behavioral deficits were evident in all animals 1 day after stroke, and all rats showed a progressive decrease in behavioral deficit over time. Therefore, in an effort to increase the sensitivity of the functional test, we employed half-sized (56.6 mm2 compared with 113.1 mm2) paper dots in the adhesive-removal test after treatment. Control rats took longer time to remove the half-sized dots than the BMSC-treated animals did at all the time points tested, and significant group differences were seen at 4 weeks after cell administration and persisted thereafter.
Although BMSC administration is a promising strategy to augment recovery from stroke, the optimal time to transplant cells is still unknown. To our knowledge, the longest time window tested for stroke cell therapy in animal models is 1 month after focal ischemia, in which NT2 cells were implanted directly into ischemic striatum (Borlongan et al, 1998). As for BMSCs, the latest time point of cell administration so far is 7 days after MCAo (Li et al, 2005; Zhao et al, 2002). Release of excitotoxic neurotransmitters, free radicals, and proinflammatory mediators in the acute setting could threaten cells introduced into the periinfarct region (Lo et al, 2003). However, it is reasonable to capitalize on local repair processes present during the early recovery phase to facilitate donor cell survival, differentiation, and integration. In the present study, we show that transplantation of BMSCs at 1 month after stroke evoked significant improvement in functional neurologic recovery. This finding is in line with the only available clinical trial, in which BMSC infusion was performed in all the patients not earlier than 1 month after the onset of stroke symptoms, and significant functional recovery was achieved in the treated patients (Bang et al, 2005).
Bone marrow stromal cells migrate selectively into damaged brain areas when administered at an early stage after the onset of ischemia (Chen et al, 2001a, b; Li et al, 2001; Zhao et al, 2002). Although initial in vitro studies suggest a role for chemokines such as monocyte chemoattractant protein-1, macrophage inflammatory protein-1alpha and interleukin-8, which are all related to the acute inflammatory reaction after ischemic injury (Wang et al, 2002a, b), it is of interest to identify the signals guiding the BMSC to sites of injury, when the inflammatory reaction is diminishing and the blood–brain barrier is intact. One candidate chemotaxic molecule is SDF-1, a member of the alpha CXC chemokine family. The interaction of SDF-1 with its specific receptor, CXCR4, mediates the homing of hematopoietic stem cells to the bone marrow (Peled et al, 1999). Stromal-cell-derived factor-1 induces migration of neural cells (Feil and Augustin 1998; Hesselgesser et al, 1997; Robin et al, 2005; Stumm et al, 2002). In this study, we showed that (1) SDF-1 expression was significantly upregulated in the ipsilateral hemisphere at 1 month after stroke at the time of cell administration; (2) although it diminished over time, the SDF-1 level in the ipsilateral hemisphere remained significantly higher than that in the contralateral hemisphere at 4 months after MCAo; (3) BMSCs expressed the specific receptor of SDF-1, CXCR4, both in vitro and in vivo. In light of the previous studies showing that SDF-1 is upregulated in the impaired site of the brain subsequent to ischemic stroke (Hill et al, 2004; Ji et al, 2004; Stumm et al, 2002) and after hypoglossal nerve injury (Ji et al, 2004), we suggest that the extended expression of the SDF-1 in the ischemic damaged region may serve as a chronic chemotactic signal, which guarantees the access of BMSCs administered at a later stage of stroke to the impaired sites in the brain.
Although the percentage of BMSCs expressing neural markers increased from 5% at 14 days after administration (Chen et al, 2003) to 20% at 4 months after MCAo in this study, the possibility that BMSCs benefit cerebral tissue by becoming brain cells is highly unlikely because of the small number of donor cells in the brain. Previous experimental data suggest that BMSCs likely exert their beneficial effects by activating endogenous restorative effects of the brain (Chopp and Li 2002). In agreement with this hypothesis, we found BMSC treatment increased the number of proliferating cells (indicative of neurogenesis), OPCs (indicative of oligogenesis and myelination), and decreased the thickness of glial scar along the neurogenic zone in the ipsilateral hemisphere. Since comparable changes in the brain have been evoked by BMSCs when treatment begins at earlier stages of stroke (Chen et al, 2003; Li et al, 2005), we propose that BMSC treatment induces similar restorative responses in ischemic brain at 1 month after MCAo similar to brain treated with BMSCs at acute or subacute stages after stroke.
In summary, our data indicate that the time window of BMSC therapy is at least 1 month after stroke, and the chemotactic molecule SDF-1 may partially mediate the homing of transplanted BMSCs to the injury sites in the brain. The beneficial effects evoked by BMSC treatment may be related to the induction of neurogenesis and reduction of glial scar formation.
Footnotes
Acknowledgements
We thank Cindi Roberts and Qing-e Lu for technical assistance.
