Abstract
Matrix metalloproteinases (MMPs) have been implicated in the pathophysiology of cerebral ischemia. In this study, we explored whether MMP activity can be visualized by noninvasive near-infrared fluorescence (NIRF) imaging using an MMP-activatable probe in a mouse model of stroke. C57BI6 mice were subjected to transient middle cerebral artery occlusion (MCAO) or sham operation. Noninvasive NIRF imaging was performed 24 h after probe injection, and target-to-background ratios (TBRs) between the two hemispheres were determined. TBRs were significantly higher in MCAO mice injected with the MMP-activatable probe than in sham-operated mice and in MCAO mice that were injected with the nonactivatable probe as controls. Treatment with an MMP inhibitor resulted in significantly lower TBRs and lesion volumes compared to injection of vehicle. To test the contribution of MMP-9 to the fluorescence signal, MMP9-deficient (MMP9−/-) mice and wild-type controls were subjected to MCAO of different durations to attain comparable lesion volumes. TBRs were significantly lower in MMP9−/- mice, suggesting a substantial contribution of MMP-9 activity to the signal. Our study shows that MMP activity after cerebral ischemia can be imaged noninvasively with NIRF using an MMP-activatable probe, which might be a useful tool to study MMP activity in the pathophysiology of the disease.
Keywords
Introduction
Matrix metalloproteinases (MMPs) comprise a family of zinc endopeptidases that are involved in the degradation and remodeling of the extracellular matrix, but seem to participate also in the etiology of several central nervous system pathologies (Rosenberg, 2002; Lee et al, 2004; Yong, 2005). During the inflammatory response after cerebral ischemia, gelatinase A (MMP-2) and gelatinase B (MMP-9) are highly upregulated in different cell types in the central nervous system, including endothelial cells, microglia, astrocytes, and neurons (Asahi et al, 2000; Rosenberg et al, 2001; Rivera et al, 2002; Magnoni et al, 2004). In addition, circulating neutrophils are also a source of MMPs (Justicia et al, 2003; Gidday et al, 2005). Matrix metalloproteinase activity after cerebral ischemia has been implicated in blood-brain barrier (BBB) impairment, edema formation, hemorrhagic transformation, neuronal cell death (Wang et al, 2004; Copin et al, 2005), as well as in repair and regeneration (Zhao et al, 2006). Emerging clinical data support the relevance of MMPs after stroke. Matrix metalloproteinase-9 is detected in human brain tissue sections after ischemic and hemorrhagic strokes (Rosell et al, 2006). Plasma levels of MMP-9 are increased in patients with acute stroke and correlate with the clinical outcome (Montaner et al, 2003a; Rosell et al, 2005). Patients with higher plasma MMP-9 levels are also more likely to undergo hemorrhagic transformation after tissue plasminogen activator treatment (Montaner et al, 2003b). In vivo visualization of MMP activity would therefore be desirable to study the role of these enzymes in the pathophysiology of the disease and to stratify and monitor the response to therapeutic intervention.
Noninvasive imaging of MMP activity in animal models of disease has been reported using near-infrared fluorescence (NIRF) imaging, magnetic resonance, and radionuclide imaging techniques (Weissleder et al, 1999; Schäfers et al, 2004; Su et al, 2005; Lancelot et al, 2008). In particular, NIRF imaging offers several advantages including high sensitivity and the use of nonionizing radiation, and it can be performed with comparatively simple and inexpensive instrumentation. Quenched NIRF probes were developed that become fluorescent through proteolytic cleavage by MMPs (Weissleder et al, 1999; Tung, 2004). Such MMP-activatable probes have the advantage of exhibiting low background fluorescence, and have been applied thus far in a variety of experimental disease models including cancer (Bremer et al, 2001) and cardiovascular diseases (Chen et al, 2005; Deguchi et al, 2006; Aikawa et al, 2007).
We have shown earlier that fluorochromes can be detected in the brains of mice with high sensitivity using noninvasive NIRF imaging (Klohs et al, 2006; Bourayou et al, 2008). We showed recently that the inflammatory receptor, CD40, can be specifically detected using NIRF imaging after cerebral ischemia (Klohs et al, 2008a). Here, we evaluated the use of an MMP-activatable probe for visualization of MMP activity using noninvasive NIRF imaging in a mouse model of focal cerebral ischemia.
Materials and methods
Animals
All experimental procedures conformed to institutional guidelines and were approved by an official committee (G0229/05, LaGeSo, Berlin, Germany). A total of 77 male C57Bl6/N mice (Charles River, Sulzfeld, Germany), six males and three females FVB.Cg-Mmp9tmtTvu/J (MMP9−/-), and four male and five female FVB/NJ wild-type (wt) littermates (University of Leuven, Belgium) weighing 18 to 26 g were housed under standard conditions with food and water available ad libitum.
Experimental Design
To assess MMP activity after cerebral ischemia, eight C57Bl6 mice were subjected either to 1 h of middle cerebral artery occlusion (MCAO; n = 6) or to sham operation (Sham n = 2). Gelatin zymography was performed 24 h after reperfusion.
A total of 31 C57B16 mice (MCAO n = 26; sham n = 5) were intravenously injected with either the MMP-activatable probe (MCAO n = 20; sham n = 5) or a nonactivatable probe (MCAO; n = 6) immediately after reperfusion. Near-infrared fluorescence imaging was performed 24 h after probe injection. In a subset of animals (MCAO, n = 10), multiparametric magnetic resonance imaging (MRI) was performed immediately after reperfusion and before injection of the MMP-activatable probe. Magnetic resonance imaging was repeated at 24 h after reperfusion with a coregistration of T2-weighted images, followed by NIRF imaging. Mice were sacrificed to assess MMP activity with gelatin zymography.
A total of 14 C57Bl6 mice were subjected to 1 h of MCAO. In two subgroups of animals, the MMP-activatable probe was injected immediately after reperfusion and NIRF imaging was performed either 4 h (n = 4) or 8 h (n = 8) after injection (i.e., 4 and 8 h after reperfusion). The other animals received the probe at either 24 h (n = 3) or 144 h (n = 3) after reperfusion, with NIRF being performed 24 h after injection (i.e., 48 and 168 h after reperfusion).
C57B16 mice were subjected to 1 h MCAO and were either treated with 100 mg/kg of the MMP inhibitor, GM6001, (n = 12) or with vehicle (n = 10). Surgery was performed with concealment of treatment allocation. Mice were randomly allocated to receive GM6001 or vehicle before MCAO and 12 h after reperfusion. The MMP inhibitor, GM6001, (Millipore, Bedford, MA, USA) was dissolved in hydroxypropy-β-cyclodextrin (45% (w/v); Sigma-Aldrich, Taufkirchen, Germany) as described (Gursoy-Ozdemir et al, 2004). All mice received the MMP-activatable probe directly after reperfusion and were imaged using NIRF 24h after injection.
MMP9−/- mice (n = 9) were subjected to 1 h MCAO, whereas wt littermates (n = 9) were subjected to 45 mins of MCAO. All mice received the MMP-activatable probe immediately after reperfusion and were imaged using NIRF 24h after injection.
Focal Cerebral Ischemia
Middle cerebral artery occlusion was performed as described (Meisel et al, 2004). Briefly, a monofilament was introduced into the common carotid artery under isoflurane anesthesia, advanced to the origin of the middle cerebral artery, and remained there for 45 mins or 1 h until reperfusion. Sham operation involved surgical procedures without occlusion of the middle cerebral artery. Brain slices were incubated in a 2% triphenyltetrazolium chloride (TTC) solution (Sigma-Aldrich, Hamburg, Germany) at 37°C for 30 mins. Direct cerebral lesion volumes were determined on 20 μm hematoxylin-stained cryostat sections using Image) (NIH, Bethesda, MD, USA).
Near-Infrared Fluorescence Imaging
For imaging MMP activity, we used an MMP-activatable probe (MMPSense, VisEn Medical, Woburn, MA, USA), which is activated by various MMPs, including MMP-2, −3, −7 −8, −9, −10, −12, −13, −14, −15, and −16 (Cortez-Retamozo et al, 2008). The probe fluoresces with the spectral characteristics of Cy5.5 after activation. A scrambled peptide sequence (VisEn Medical) served as nonactivatable probe. Compounds were injected slowly into the tail vein in a volume of 300 μL (equivalent to 4 nmol per mouse).
The planar NIRF imaging system was recently described in detail (Klohs et al, 2006, 2008a). For excitation of Cy5.5, an intensity-controlled laser diode emitting at 682 nm (40 mW) was used. Fluorescence emission was collected by a charge-coupled device (CCD) camera Vers Array 512, Roper Scientific, Duluth, GA, USA) equipped with two 710 nm interference filters to block the excitation light. Data acquisition times were 60 secs. For noninvasive NIRF imaging, mice were anesthetized by intraperitoneal injection of chloral hydrate solution (200 mg/kg; Merck, Darmstadt, Germany) and the skin overlying the parietal cortex was depilated. Brains were subsequently removed under deep anesthesia without prior perfusion of the animal and were placed in the NIRF system for ex vivo imaging. Coronal brain slices of 1-mm thickness were cut in a brain matrix using a razor blade.
Regions of interest (ROIs) were selected over the left and right hemispheres on noninvasive and NIRF images (outlined in Figure 2A) and over the left (ischemic) hemisphere on ex vivo NIRF images. Target-to-background ratios (TBRs) were calculated as described (Klohs et al, 2006).
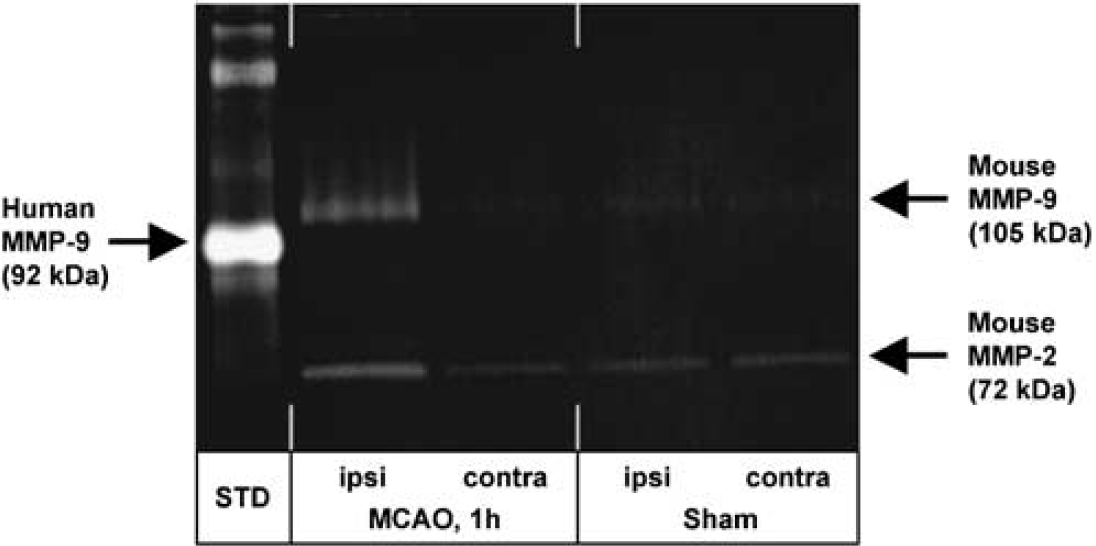
Gelatin zymography showed the induction of MMP-9 in the ischemic ipsilateral hemisphere of MCAO mice compared with the contralateral, nonischemic side at 24 h after reperfusion. Matrix metalloproteinase-2 was also detected in sham-operated animals.
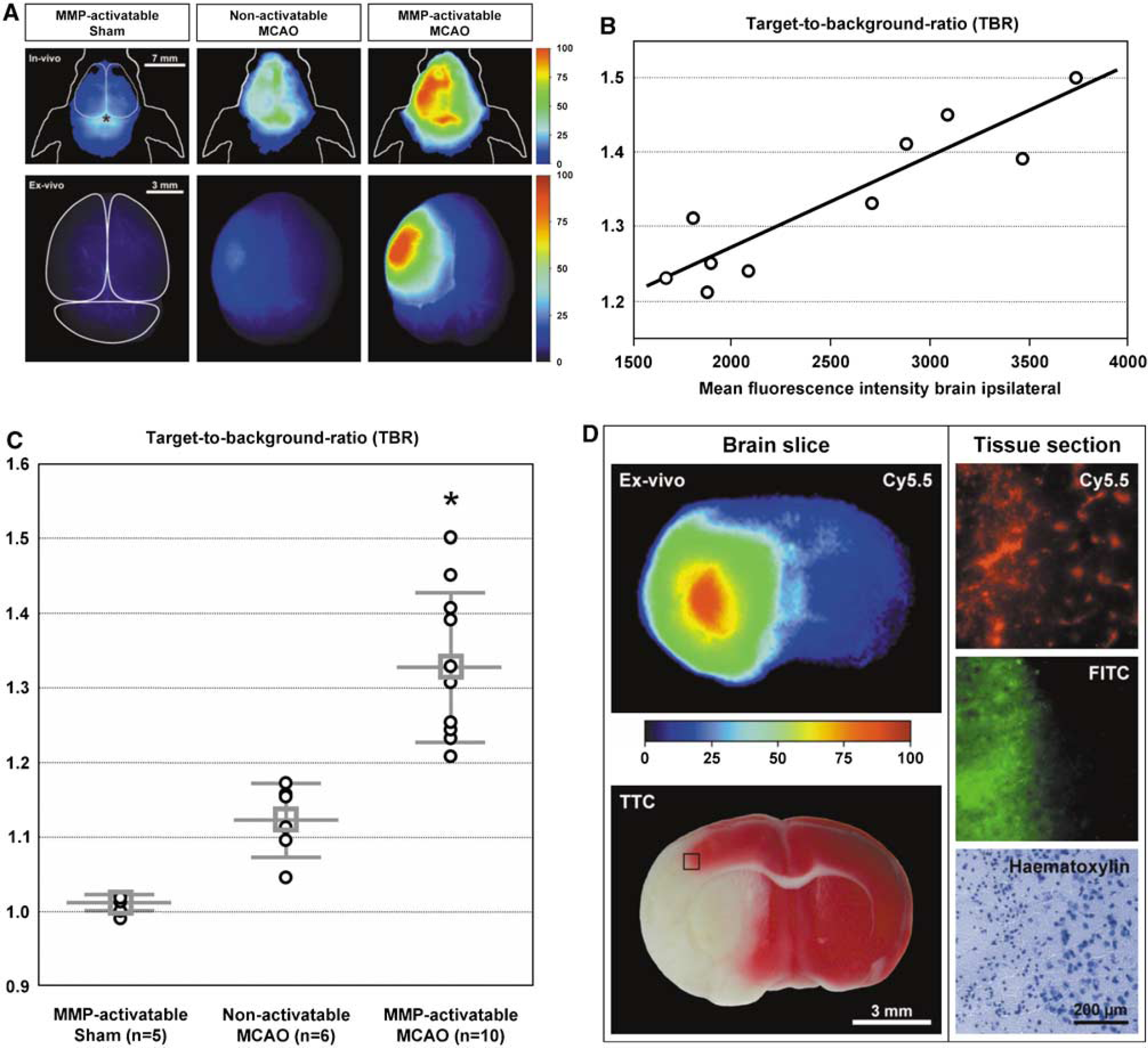
Representative in vivo (
Magnetic Resonance Imaging
Magnetic resonance imaging was performed on a dedicated animal scanner (7T Bruker PharmaScans 70/16 AS, Bruker Biospin, Ettlingen, Germany) equipped with a 98/38-mm RF Coil (Bruker). Data acquisition and image processing were performed using a Paravision software platform (Bruker). Mice were subjected to isoflurane anesthesia using a stereotactic frame and positioned in the magnet bore. Respiratory rate and body temperature were monitored using a physiology monitoring unit, and temperature was maintained within physiologic limits using a heated water jacket. Diffusion-weighted MRI (DWI-MRI) was performed with a multislice spin echo planar imaging sequence (imaging parameters: echo time = 66.5 msecs, D = 25 msecs, diffusion gradient duration = 5 msecs, 10 slices with slice thickness 1 mm, image matrix = 128 × 128, field of view = 20 × 20 mm, 15 averages, b = 1, 300 secs/mm2). Ten neighboring slices at 1 mm distance between the olfactory bulb and the cerebellum were selected. T2-weighted images were generated using a two-dimensional turbo spin-echo sequence (TR/TE = 4, 200/36 msecs, RARE factor 8, four averages). Twenty axial slices with a slice thickness of 0.5 mm, a field of view of 2.85 × 2.85 cm and a matrix of 256 × 256 were positioned over the brain excluding the olfactory bulb. The size of the lesion was determined on diffusion-weighted images using custom-written imaging processing tools programmed with MATLAB (The Math-works, Natick, MA, USA). The lesion, shown as hyperintensity in the T2-weighted image in the ischemic hemisphere, was defined by finding significant deviation from the signal distribution in the unaffected, contralateral hemisphere. The hyperintense ischemic areas in T2-weighted images were assigned using a ROI selection tool, and lesion volumes were calculated (Analyze 5.0, AnalyzeDirect, Lenexa, KA, USA).
In Vivo Blood-Brain Barrier Permeability Assay
Blood-brain barrier integrity was assessed in MCAO mice (n = 22) by measuring fluorescein isothiocyanate-labeled bovine serum albumin (FITC-BSA) extravasation. FITC-BSA (25 mg/kg; Invitrogen, Karlsruhe, Germany) was dissolved in saline (2 mg/mL) and injected intravenously 30 mins before euthanasia (i.e., 24 h after reperfusion). As a control for changes in the autofluorescence of brain tissue after MCAO, two mice received an intravenous injection of phosphate-buffered saline. Brains were cut into 20 μm coronal cryostat sections.
Gelatin Zymography
Whole ipsilateral ischemic and contralateral nonischemic brain hemispheres were homogenized in a lysis buffer (50 mmol/L Tris-HCl, pH 7.4, 150 mmol/L NaCl, 1% Nonidet P40, 0.1% sodium dodecyl sulfate (SDS), 0.1% deoxycholic acid, 2 mg/mL Aprotinin, and 0.5 mmol/L phenylmethylsulfonyl fluoride). After centrifugation (12,000 r.p.m. for 5 mins at 4°C), supernatants were affinity-precipitated with gelatin-Sepharose 4B as reported earlier (Justicia et al, 2003). Total protein content was measured using a BCA Protein Assay Kit (Pierce N23225, Pierce, Rockford, IL, USA). Gelatin zymography was performed as described (Gursoy-Ozdemir et al, 2004). In brief, SDS-denaturated MMP samples were separated on 10% gelatin-containing gels (Criterion zymogram gel, Bio-Rad, Hercules, CA, USA). Matrix metalloproteinase was renaturated by removing the SDS from the gel with a renaturating buffer. Gels were then incubated in a developing buffer for 40 h and were stained with 0.25% Coomassie blue R-250.
Fluorescence Microscopy
A fluorescence microscope equipped with a CoolSNAP CCD camera (Roper) was used for the detection of FITC (excitation = 470 nm and emission = 522 nm), and a Vers Array 512 CCD camera (Roper) for the detection of Cy5.5 (excitation = 682 nm and emission = 721 nm). Microscopic images were obtained at × 10 original magnification. Acquisition times for the detection of Cy5.5 were 60 secs. For assessing the distribution of FITC-BSA, single images were joined together using the technique of tiled-field mapping (MCID Elite, InterFocus, Mering, Germany). Regions of interest were specified using ImageJ with the technique of density slicing, including the setting of target acceptance criteria. For each brain, analysis included the cortex, striatum, and peri-ischemic territories in three coronal sections taken between 1.6 and −1.3 mm from the bregma.
Statistical Analysis
Data are presented as means ± s.d. Comparisons were made using analysis of variance on ranks followed by Dunn's test or Student's t-test, where applicable. Target-to-background ratios were plotted against mean fluorescence intensities, followed by a linear regression analysis to calculate R2 and to determine the regression equation. To test for differences in the mean fluorescence intensities measured over the left ischemic and right nonischemic hemispheres for animals assessed at each time point, a Wilcoxon rank test was carried out. A value of P < 0.05 was considered statistically significant.
Results
Noninvasive Near-Infrared Fluorescence Imaging of Matrix Metalloproteinase Activity after Cerebral Ischemia
Gelatin zymography was used to detect MMP activity in brain tissue homogenates. A representative gelatin zymogram shows the upregulation of MMP-9 (proform) in the ischemic (ipsilateral) hemisphere after 1 h of MCAO followed by 24 h of reperfusion (Figure 1). Basal MMP-2 activity was also observed in sham-operated animals.
Immediately after reperfusion, MCAO and sham-operated C57Bl6 mice were injected either with the MMP-activatable probe or with nonactivatable probe as control. Noninvasive NIRF imaging and ex vivo NIRF imaging of the brains removed from the skull were performed 24 h after probe injection (Figure 2A). No differences between the hemispheres were seen in vivo and ex vivo in sham-operated mice injected with the MMP-activatable probe. Slightly higher fluorescence intensities over the ischemic hemisphere compared to the contralateral side were observed in MCAO mice injected with the nonactivatable probe. Intense fluorescence was seen over the ischemic hemisphere of MCAO mice injected with the MMP-activatable probe.
Linear regression analysis showed linear correlation (R2 = 0.833) between the calculated TBRs and the mean fluorescence intensities measured over the ischemic hemisphere of ex vivo NIRF images of MCAO mice injected with the MMP-activatable probe (Figure 2B).
Sham-operated mice injected with the MMP-activatable probe and MCAO mice that were injected with the nonactivatable probe showed significantly lower TBR compared with those injected with the MMP-activatable probe (Figure 2C; 1.01 ± 0.01 and 1.12 ± 0.05 versus 1.33 ± 0.10, respectively, P < 0.001).
Ex vivo NIRF imaging of brain slices showed differences in fluorescence intensities in the brains of MCAO mice 24 h after injection of the MMP-activatable probe. Higher fluorescence intensities were observed in the ischemic lesion, corresponding to the pallor in TTC staining, compared to areas surrounding the lesion (Figure 2D, left panel). The highest fluorescence intensities were detected in the striatum of the ischemic hemisphere. Fluorescence microscopy was used to assess the distribution of the MMP-activatable probe with higher magnification. To detect BBB permeability, FITC-BSA was coadministered before sacrifice of the animals. Intense, diffuse Cy5.5 fluorescence was observed in addition with strong fluorescence of the extravasated FITC-BSA in the ischemic area of the cortex, whereas fluorescence in both channels is scarcely visible in the nonischemic area (Figure 2D, right panel). The lesion border is clearly delineated after hematoxylin staining. Two controls subjected to MCAO and injected with phosphate-buffered saline showed very low tissue autofluorescence in the ischemic cortex in the FITC and Cy5.5 channels (data not shown).
In a different set of MCAO mice, which received the MMP-activatable probe directly after reperfusion, assessment of the lesion with multiparametric MRI and investigation of MMP activity with gelatin zymography were carried out in conjunction with the NIRF imaging. Although no correlation was observed between the final lesion volume at 24 h obtained from T2-weighted images or the temporal evolution of the lesion (i.e., DWI volume increase) and NIRF signal intensities, NIRF signal intensities matched well with the MMP-9 bands in the gelatin zymogram (Supplementary Figure).
Time Course of Matrix Metalloproteinase Activity Measured Using Noninvasive Near-Infrared Fluorescence
The results from noninvasive NIRF images obtained at different time points after reperfusion are shown in Table 1. In mice imaged with NIRF at 4 or 8 h after reperfusion, the MMP-activatable probe circulated for 4 and 8h, respectively. In those mice, no significant differences were observed between the fluorescence intensities measured over the ipsilateral, ischemic hemisphere compared with the contralateral side (TBR: 1.05 ± 0.03, 1.04 ± 0.06). In contrast, mice imaged using NIRF at 24, 48, and 168 h after reperfusion significantly higher fluorescence intensities were detected over the ipsilateral hemisphere compared with the contralateral side (TBR: 1.33 ± 0.1, 1.35 ± 0.07, and 1.55 ± 0.08, respectively; P < 0.05). In those mice, the probe circulated for 24 h.
Noninvasive NIRF imaging was performed at different time points after 1 h of MCAO
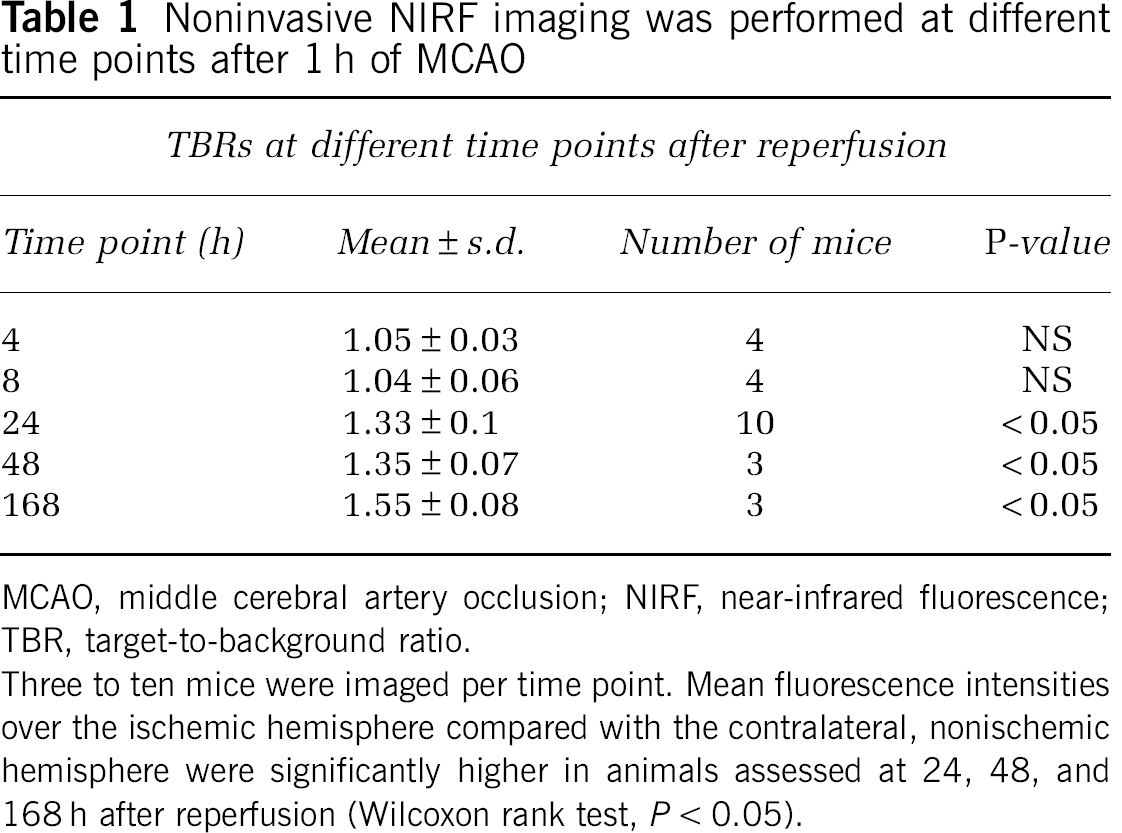
MCAO, middle cerebral artery occlusion; NIRF, near-infrared fluorescence; TBR, target-to-background ratio.
Three to ten mice were imaged per time point. Mean fluorescence intensities over the ischemic hemisphere compared with the contralateral, nonischemic hemisphere were significantly higher in animals assessed at 24, 48, and 168 h after reperfusion (Wilcoxon rank test, P < 0.05).
Matrix Metalloproteinase Inhibition Reduced Near-Infrared Fluorescence Signal, Lesion Volumes, and Blood-Brain Barrier Impairment
In an attempt to monitor the response of an MMP inhibitor with noninvasive NIRF imaging, MCAO mice that were injected with the MMP-activatable probe were either treated with the MMP inhibitor, GM6001, or with vehicle. MCAO mice injected with GM6001 (Figure 3A) showed significantly lower TBRs compared with mice that received vehicle only (vehicle: 1.33 ± 0.1, GM6001: 1.17 ± 0.18, P < 0.001). Infarct volumes were reduced by 21% compared with those that received vehicle only (Figure 3B; vehicle: 132.7 ± 16.2 mm3; GM6001: 104.9 ± 31.7mm3; P = 0.037). FITC-BSA was injected before sacrifice of the animals to show differences in the degree of BBB impairment. Mice treated with GM6001 tended to have smaller areas of FITC-BSA extravasation compared with vehicle-treated controls (Figure 3C), but this did not reach statistical significance (vehicle: 10.89 ± 4.0mm2; GM6001: 7.84 ± 2.89mm2; P = 0.073).
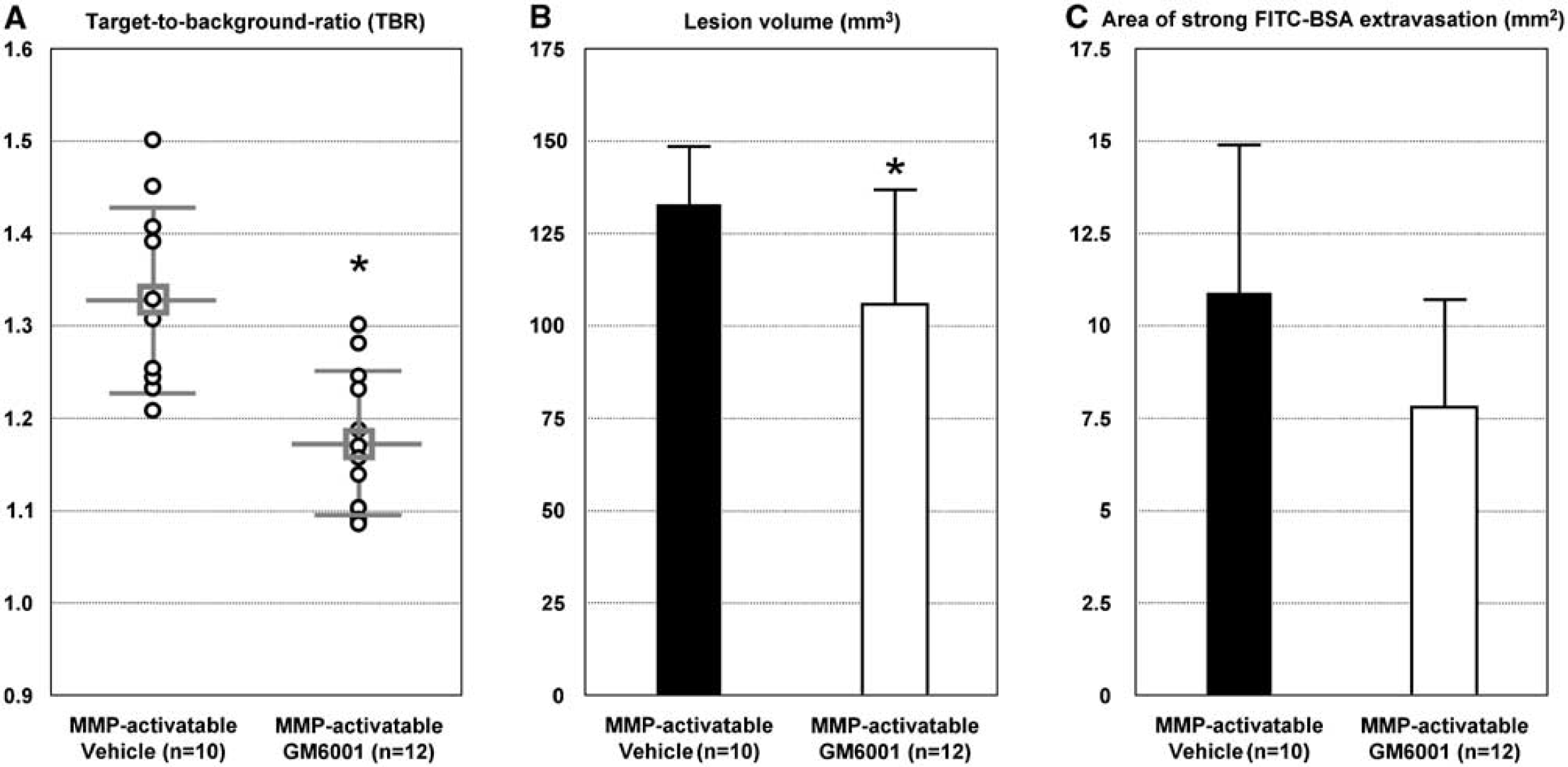
Treatment with MMP inhibitor significantly reduced infarct size and NIRF signal. (
Matrix metalloproteinase-9 Activity is a Major Contributor to the Near-Infrared Fluorescence Signal Detected In Vivo
MMP9−/- mice and wt littermates were used to assess the contribution of MMP-9 activity to the NIRF signal in vivo (Figure 4). Six of the transgenic animals were strongly pigmented and had to be excluded from the analysis of the NIRF images. Target-to-background ratios were significantly lower in MMP9−/- mice than in wt littermates (Figure 4A; wt: 1.24 ± 0.06; MM9−/- 1.11 ± 0.06; P < 0.004), even though cerebral lesion volumes were matched between the groups by titrating the MCAO intervals (Figure 4B; wt 45 mins MCAO: 78 ± 17.3 mm3; MMP9−/- 1 h MCAO: 75.9 ± 23.6 mm3; P = 0.796).
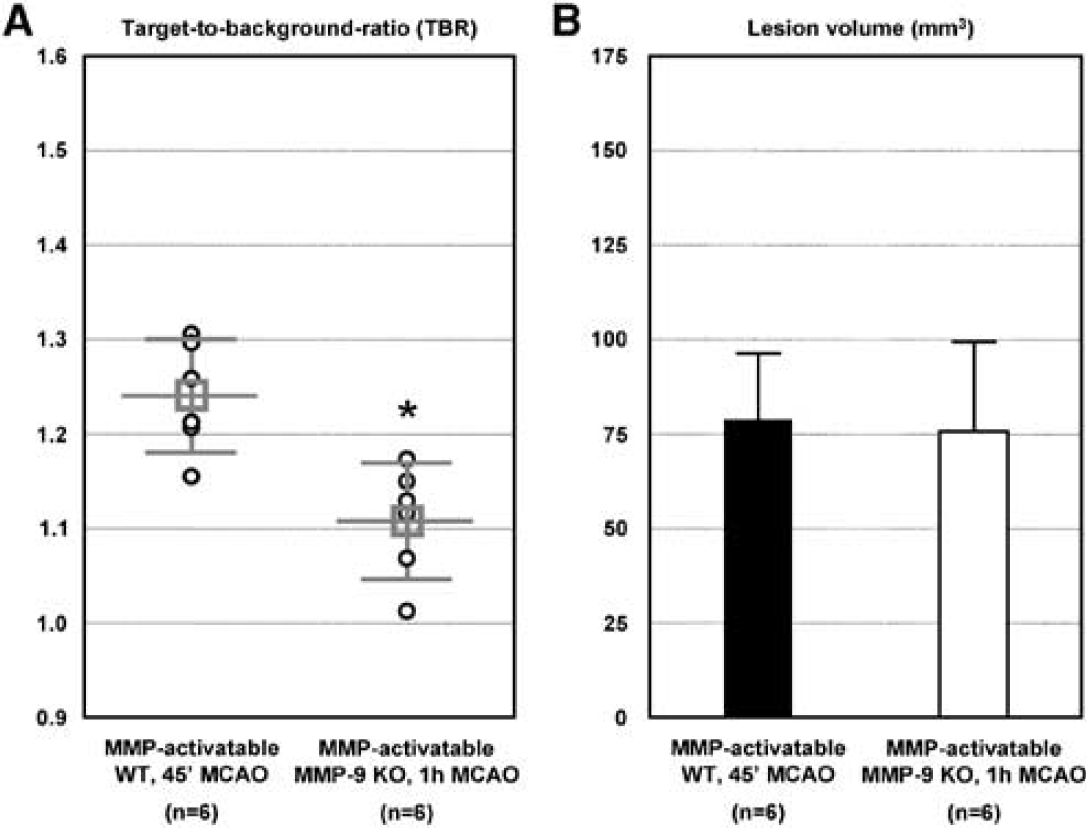
Near-infrared fluorescence imaging of MMP9-/- mice at 24 h after reperfusion. Middle cerebral artery occlusion duration was decreased to 45 mins in wt mice to attain lesion sizes similar to those after 60 mins MCAO in MMP9−/- mice. (
Discussion
Here, we report for the first time the detection of MMP activity by noninvasive NIRF imaging using an MMP-activatable probe in a mouse model of cerebral ischemia.
Activities of MMPs after cerebral ischemia are time-dependent and have multifaceted effects (Rivera et al, 2002; Rosenberg, 2002; Lee et al, 2004; Yong, 2005). Visualization of MMP activity, without the need to killing the animal under study, would therefore be highly desirable to study the contribution of these enzymes to the pathophysiology of the disease. Fluorescent probes that are activated by the proteolytic action of MMPs have been specifically developed for NIRF imaging (Weissleder et al, 1999; Tung, 2004). A primary aim of this study was to evaluate the use of an MMP-activatable probe in a mouse model of cerebral ischemia. We used gelatin zymography first to assess the induction of MMP activity in our model. We observed an upregulation of MMP-9 in the ischemic hemisphere of MCAO mice 24 h after reperfusion. Hence, we tested the MMP-activatable probe within this time frame in MCAO mice. We observed strong fluorescence intensities over the ischemic hemispheres compared with the nonischemic hemispheres on noninvasive and ex vivo NIRF images. To control for activation of the probe unrelated to cerebral ischemia (e.g., because of the depilation of mice), we injected the MMP-activatable probe in sham-operated mice. We found low fluorescence intensities equally distributed over both hemispheres on noninvasive NIRF images. To test for activation of the MMP-activatable probe in vivo, we compared MCAO mice injected with the MMP-activatable probe with those that were injected with the control probe, a scrambled peptide sequence that is not readily cleaved by MMPs. Noninvasive and ex vivo NIRF images showed slightly higher fluorescence intensities over the ischemic hemisphere compared with the nonischemic hemisphere in the mice that received the nonactivatable probe. The detected fluorescence might be either because of an accumulation of the nonactivated, quenched probe at the lesion or because of a slight activation of the probe in vivo. However, TBRs of MCAO mice injected with the MMP-activatable probe were significantly higher than those of mice that received the nonactivatable probe. In mice that received the MMP-activatable probe, bright fluorescence was observed in the ischemic hemispheres with ex vivo NIRF imaging, whereas fluorescence was scarcely visible in the ischemic hemisphere of mice injected with the nonactivatable probe. Taken together, this suggests that the signal after injection of the MMP-activatable probe is largely because of activation of the probe after cerebral ischemia.
Clinical studies have shown earlier a correlation between plasma levels of MMP-9 and MMP-13 measured within 3 h after the onset of ischemic stroke and the final extent of the lesions as assessed with MRI (Montaner et al, 2003a, b ; Rosell et al, 2005). However, the study by Rosell et al (2005) has shown no correlation between the plasma levels of MMP-1, −2, −3, −8, and −10 and the extent of the lesion. In a subset of MCAO mice injected with the MMP-activatable probe, multiparametric MRI was performed immediately and 24 h after reperfusion to assess the development of the ischemic lesion. We did not observe a correlation between the extent and growth of the lesion and the NIRF signal after MCAO. Although we have found a substantial contribution of MMP-9 to the detected NIRF signal (MMP-9 bands in the gelatin zymography matched well with the NIRF signal; TBRs were significantly reduced in MMP9−/- mice compared with those in wt mice), the MMP-activatable probe can be activated also by MMPs other than MMP-9 (Cortez-Retamozo et al, 2008). As the degree to which individual MMPs contribute to the expansion of the lesion after cerebral ischemia varies (Rosell et al, 2005), the overall detected pan-MMP activity might not be correlated with the extent or growth of the lesion. Alternatively, the MMP-activatable probe was injected immediately after reperfusion and was allowed to circulate for 24 h before NIRF imaging. Hence, the MMP activity of the detected NIRF signal most likely represents not the MMP activity of a single given time point but rather that of a time range over which the probe is activated. Therefore, the NIRF signal reflects also MMP activity at later time points after reperfusion, where a correlation between MMP activity and the extent of the lesion might no longer exist.
Matrix metalloproteinases are expressed and active after cerebral ischemia in a time-dependent manner (Rosenberg et al, 2001; Rivera et al, 2002). We have used NIRF imaging to detect MMP activity at different time points after reperfusion. Circulation times of the probe differed between the different time points. Target-to-background ratios calculated from noninvasive NIRF images acquired at 4 and 8 h after reperfusion (4 and 8 h after injection, respectively) were ~1 (i.e., no differences in fluorescence between hemispheres).
The absence of detectable NIRF signals at the earlier time points might be because of the fact that MMP expression or activation was too low. It is also possible that not sufficient MMP-activatable probe had been activated, or that both factors contributed to this dearth of NIRF signals. This precludes the use of the method for studying the effect of MMP activity on BBB impairment at the early time points after reperfusion. However, significantly increased TBRs were calculated from NIRF images of mice acquired at 24, 48, and 168h after reperfusion (i.e., 24h after injection). This enables the study of MMP activity during the stages of inflammation and regeneration after cerebral ischemia.
Matrix metalloproteinase inhibitors are promising neuroprotectants, as they can reduce brain damage in experimental stroke (Asahi et al, 2000; Zhao et al, 2006; Yang et al, 2007). In line with earlier studies, in our experiments the broad-spectrum MMP inhibitor, GM6001 (Gursoy-Ozdemir et al, 2004), significantly reduced lesion volumes. However, MMP inhibition was also shown to have detrimental effects, particularly through impeding MMP-dependent brain reorganization after stroke (Zhao et al, 2006). The focus of our study was to investigate whether in vivo noninvasive pathobiology-specific monitoring of treatment with an MMP inhibitor is feasible. Indeed, we found that the treatment effect of GM6001 was paralleled by significantly reduced TBRs. Pharmacological intervention of MMPs leads to a marked reduction in lesion size, which might also affect downstream pathophysiological processes that are only indirectly related to MMP activity (Asahi et al, 2000; Rivera et al, 2002; Justicia et al, 2003; Magnoni et al, 2004). Such downstream effects may alter the compartmental distribution of intravenously injected compounds, which complicates the use of genetic or pharmacological manipulation of MMPs as control for the specificity of imaging approaches (Klohs et al, 2008b).
Using gelatin zymography, we found strong upregulation of brain MMP-9 in our mouse model of stroke. Therefore, we used MMP9−/- to address the specific contribution of MMP-9 to the acquired NIRF signal. Earlier studies have shown that MMP9−/-mice develop smaller infarcts after MCAO compared with wt mice (Asahi et al, 2000). To circumvent the possible confounding effects of smaller cerebral lesion sizes in MMP9−/- mice on the NIRF signal, as an additional control we reduced MCAO intervals in wt mice to 45 mins in order to attain comparable lesion sizes. We were thus able to titrate occlusion intervals to comparable lesion volumes in MMP9−/-and wt mice. Nevertheless, TBRs were significantly reduced in MMP9−/- mice. Thus, the differences in TBR are unlikely to be caused by the differences in lesion size and distribution of the MMP-activatable probe, but results more probably from the differences in the activation of the MMP-activatable probe. Although MMPs other than MMP-9, such as MMP-2, −3, −7 −8, −9, −10, −12, −13, −14, −15, and −16, can activate the MMP-activatable probe in vivo (Cortez-Retamozo et al, 2008), the results suggest that MMP-9 is in our model a major contributor to the observed NIRF signal.
In conclusion, noninvasive visualization of MMP activity after cerebral ischemia using NIRF imaging is feasible and may provide an experimental tool to study the role of these enzymes in the pathophysiology of the disease.
Footnotes
Acknowledgements
We gratefully acknowledge Bart Hermans (University of Leuven, Belgium) for providing the MMP9−/- mice and Susanne Mueller (Center for Stroke Research Berlin, Germany) for performing MRI. The work was funded by the Bundesministerium für Bildung und Forschung, the Deutsche Forschungsgemeinschaft, the Herman and Lilly Schilling Stiftung, and the European Community's Seventh Framework Programme (FP7/2007-2013) under grant agreement nos. 201024 and 202213 (European Stroke Network).
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
