Abstract
The present study characterizes the effects of lysophosphatidic acid (LPA) on blood–brain barrier (BBB) permeability focusing specifically on the time of onset, duration, and magnitude of LPA-induced changes in cerebrovascular permeability in the mouse using both magnetic resonance imaging (MRI) and near infrared fluorescence imaging (NIFR). Furthermore, potential application of LPA for enhanced drug delivery to the brain was also examined by measuring the brain accumulation of radiolabeled methotrexate. Exposure of primary cultured brain microvessel endothelial cells (BMECs) to LPA produced concentration-dependent increases in permeability that were completely abolished by clostridium toxin B. Administration of LPA disrupted BBB integrity and enhanced the permeability of small molecular weight marker gadolinium diethylenetriaminepentaacetate (Gd-DTPA) contrast agent, the large molecular weight permeability marker, IRdye800cwPEG, and the P-glycoprotein efflux transporter probe, Rhodamine 800 (R800). The increase in BBB permeability occurred within 3 minutes after LPA injection and barrier integrity was restored within 20 minutes. A decreased response to LPA on large macromolecule BBB permeability was observed after repeated administration. The administration of LPA also resulted in 20-fold enhancement of radiolabeled methotrexate in the brain. These studies indicate that administration of LPA in combination with therapeutic agents may increase drug delivery to the brain.
Keywords
INTRODUCTION
Lysophosphatidic acid (LPA), also referred to as monoacyl-sn-glycerol-3-phosphate, is a phospholipid found in most cell types including neurons, Schwann cells, adipocytes, and fibroblasts. 1 It is produced in relatively high levels from activated platelets and can be detected in bodily fluids including serum, saliva, follicular fluid, and malignant effusions. 2 Although once considered an inert intermediate in lipid metabolism, the identification of LPA as a major mitogenic component in serum 3 provided the first initial evidence of the biologic activity of the phospholipid. It is now clearly established that LPA is a bioactive lipid capable of producing a variety of responses in mammalian cells. 2 Furthermore, as LPA is released locally in response to inflammation, elevations in LPA have been implicated in the pathogenesis of several conditions, including atherosclerosis and myocardial injury, 4 cancer, 5 and neurologic disorders. 6
The responses to LPA are initiated through the binding of the phospholipid to LPA receptors on the plasma membrane of the cell. 7 Currently, there are at least six different LPA receptors (LPAR1–LPAR6) that share the common G-protein coupled receptor structure.8, 9, 10Consistent with the diversity of G-protein coupled receptor signaling pathways, cellular responses to LPA vary, ranging from cell migration and survival to neurite retraction and gap junction closure. 2 An important cellular target of LPA is the vascular endothelium where LPA-induced changes in mitogenesis, cell migration, 11 and permeability12, 13 have been reported. The effects of LPA on vascular permeability are a topic of considerable debate with some studies reporting a decrease in endothelial cell permeability after LPA exposure,14, 15 whereas others support an increase in vascular leakage after LPA exposure.11, 12, 16 Studies of LPA on brain microvessel endothelial cell permeability support an enhancement of permeability with decrease in transcellular electrical resistance and increases in flux of vascular markers observed in primary cultured brain endothelial cell preparations.12, 16 More recent studies indicate increased permeability after localized application of LPA to the cerebral microvasculature. 16
While there are studies supporting LPA-induced increases in cerebrovascular permeability in vitro, the effects of LPA on BBB permeability at the whole animal or organ level are still poorly understood. The brain microvessel endothelial cells forming the blood–brain barrier (BBB) are structurally and functionally different from capillaries found in other organs. 17 The restrictive nature of the BBB is important for maintaining the proper extracellular fluid environment within the brain required for neuronal transmission. However, in terms of central nervous system therapy, an intact BBB presents a formidable obstacle to drug delivery to the brain. 18 Thus, depending on the magnitude and duration of response, LPA-mediated alterations in BBB permeability could have both pathophysiological and therapeutic implications. The present studies describe a series of experiments elucidating the effects of LPA on BBB permeability in vivo. Studies focused on characterization of the time to onset, and duration of action for LPA-induced changes in cerebrovascular permeability in the mouse. Furthermore, using both magnetic resonance imaging (MRI) and near infrared fluorescence (NIRF) contrast agents, alterations in BBB permeability to small and large molecular weight compounds as well as a P-glycoprotein substrate were evaluated. The results demonstrate that LPA-induced increases in BBB permeability occur within minutes of systemic administration. Equally important, BBB integrity was restored within 20 minutes. Furthermore, LPA-induced increases in BBB permeability were observed for both small and large molecular weight contrast agents and to a lesser extent, P-gp-dependent agents. The time frame for BBB disruption and the range of molecules affected suggest that LPA may be used to enhance the BBB permeability of a wide range of drug molecules.
MATERIALS AND METHODS
Lysophosphatidic acid was purchased from Sigma Chemical Company (Oakville, ON, Canada). Fluorescein labeled dextran with molecular weight of 3000 (FDX-3000) was purchased from Invitrogen Canada, Burlington, ON, Canada and Clostridium difficile toxin B was purchased from Sigma Chemical Company. The NIRF imaging agents, IRdye 800cw PEG and rhodamine 800 (R800), were obtained from Mandel Scientific (Guelph, ON, Canada) and Exciton (Dayton, OH, USA), respectively. Gadolinium diethylenetriaminepentaacetate (Gd-DTPA) contrast agent was obtained from Berlex, Lachine, QC, Canada. Ketamine hydrochloride and xylazine were purchased from Wyeth (Guelph, ON, Canada) and Bayer (Toronto, ON, Canada), respectively. All other reagents and chemicals were purchased from Sigma Chemical Company (St Louis, MO, USA).
Cell Culture Studies
In vitro studies were performed using primary cultured brain microvessel cells (BMECs). The bovine BMEC were isolated from the gray matter of fresh cow cerebral cortices using a combination of enzymatic digestion and centrifugal separation methods previously described. 19 Primary human BMECs were purchased from Cell Systems (Kirkland, WA, USA). The BMECs were seeded (50,000 cells/cm2) on collagen coated, fibronectin-treated, polycarbonate membrane inserts (0.4 μmol/L pore; 24 mm diameter) for permeability studies, or T-25 flasks for LPA receptor expression studies. The culture media consisted of the following: 45% minimum essential medium eagle, 45% Ham's F 12 nutrient mix, 10 mmol/L HEPES, 13 mmol/L sodium bicarbonate, 50 μg/mL gentamicin, 10% equine serum, 2.5 μg/mL amphotericin B, and 100 μg/mL heparin. The BMECs were grown in a humidified 37°C incubator with 5% CO2. Media was replaced every other day and were used for permeability studies after reaching the confluency.
Permeability studies were performed on confluent BMEC monolayers at 37°C as described previously. 20 Culture media was removed and replaced with assay buffer, and the cells were incubated at 37°C. After 30 minutes, LPA was added to the donor and receiver compartments to a final concentration of 0.1 to 10 μmol/L. Changes in monolayer permeability were assessed using 3.2 μmol/L FDX-3000 added to the donor compartment at the start of the permeability study. The concentration of FDX-3000 was measured spectrofluorometrically using a Shimadzu RFU5000 spectrofluorometer (Shimadzu Scientific Instruments, Columbia, MD, USA) with Ex (l) at 488 nm and Em (l) at 510 nm. Permeability was expressed as the percent transfer of the fluorescent marker across the monolayers. The percent transfer was determined by dividing the cumulative concentration of FDX-3000 in the receiver compartment by the concentration of FDX-3000 in the donor compartment at time 0.
Capillary Depletion
Capillary-enriched fractions from mouse brain homogenate were collected using the capillary depletion technique described by Triguero D et al.
21
A total of three mouse brains were weighed and pooled together. Brain buffer solution (10 mmol/L Herpes, 141 mmol/L NaCl, 4 mmol/L KCl, 2.8 mmol/L CaCl2, 1 mmol/L MgSO4, and 1 mmol/L NaH2PO4, 10 mmol/L
RT-PCR Analysis
Lysophosphatidic acid receptor expression was examined in both human BMEC as well as in mouse brain homogenates and capillary-enriched fractions. The RNA from human BMECs, brain homogenate, and brain capillary were isolated using TRIzol reagent (GIBCO, Burlington, ON, Canada) according to the manufacturer's instructions. Real-time PCR analysis of LPA receptors (LPAR1–LPAR5) and β-Actin gene expression were carried out using QuantiTect Probe RT-PCR SYBR Green Kit (Qiagen, Waltham, MA, USA). Gene expression was analyzed quantitatively using Mastercycler ep realplex system (Eppendorf, Mississauga, ON, Canada). Primers and the hybridization probes were synthesized by Invitrogen, and Qiagen with the following sequences:
LPAR1: forward: 5′-TGGTCATGGTGGCAATCTACGTCA-3′
Reverse: 5′-ACTAGGCAAATCCAGCCAAGAAGT-3′
LPAR2: forward: 5′-TGGCCTTGAACTCTTGATGGTCCT-3′
reverse: 5′-TCAGAGTGGAAAGGCTTGCTCTGT-3′
LPAR3: forward: 5′-TGAGAGTCCACAGCAACTTGACCA-3′
reverse: 5′-ATGTTGCAGAGGCAATTCCATCCC-3′
LPAR4: forward: 5′-ATGCTCTTCCTCACCTGCATCAGT-3′
reverse: 5′-CTTCAAAGCAAGTGGTGGTCGCAT-3′
LPAR5: forward: 5′-ACCTTGTTGTTCCCTACGATGCCT-3′
reverse: 5′-TGGAGCACAGAGCTGTTGGTAGAA-3′
LPA Response In Vivo
Adult female Balb/c mice were used to characterize the BBB-disruption profile produced by LPA. Mice were obtained from the University of Manitoba breeding colony and maintained in the Central Animal Care Facility under temperature-controlled environment with 12 hours dark–light cycle and unlimited access to food and water. All animal experiments were performed in accordance with the Canadian Council on Animal Care guidelines and were approved by the University of Manitoba Animal Care Committee (protocol number 09-049). Quantitative determination of BBB permeability was performed using three different imaging agents and MRI and near infrared fluorescence (NIRF) imaging modalities described below.
Magnet resonance imaging of BBB permeability
The onset and duration of LPA-induced alterations in BBB permeability were initially assessed using MRI with Gd-DTPA contrast agent. Mice were anesthetized and secured in a Bruker Biospect MR (7 tesla/21 cm spectrometer). A series of T1-weighted images (repetition time=852 milliseconds, averages=6, total image time=2.40 minutes) and T2-weighted images (repetition time=1640 ms, averages=12, total image time=10.50 minutes) of the mouse brain were obtained before administration of Gd-DTPA contrast agent to acquire background images. To determine the onset of LPA-induced BBB disruption, mice were given Gd-DTPA contrast agent (0.4 mmol/kg) together with LPA (0.1 to 10 mg/kg) or vehicle (0.25% bovine serum albumin in phosphate-buffered saline). A series of T1-weighted images were obtained immediately after Gd-DTPA injection and at 3-minute intervals throughout a 21-minute imaging session. After 21 minutes, a second dose of Gd-DTPA was administered and T1-weighted images obtained at 3-minute intervals for an additional 21-minute imaging session. To confirm the BBB-disruption period for LPA, a separate group of mice were administered LPA (1 mg/kg) or vehicle 20 minutes before the first Gd-DTPA (0.4 mmol/kg) injection. Immediately after administration of Gd-DTPA contrast agent, a series of T1-weighted scans were taken at an interval of 3 minutes for a period of 21 minutes. After the first 21 minutes of scanning, the mice received a second dose of Gd-DTPA and were imaged for an additional 21 minutes as described above. Quantitative assessment of Gd-DTPA enhancement in the brain was accomplished by manually outlining regions of interest within the coronal brain slices using Marevisi 3.5 software (Institute for BioDiagnostics, National Research Council, Winnipeg, MB, Canada). Changes in Gd-DTPA intensity in the brain were determined using a percent difference analysis of brain slice images within the Paravision 3.0 software package (Bruker, Milton, ON, Canada) according to the following formulae: 22
((Post-Gd-DTPA T1-weighted images−pre-Gd-DTPA T1-weighted images)÷pre-Gd-DTPA T1-weighted images) × 100.
Near infrared fluorescence imaging of BBB permeability
The LPA-induced changes in the BBB permeability of a large molecular weight compound as well as a P-gp-sensitive agent was also examined using NIRF. For these studies, mice received either IRdye800cwPEG (0.01 μmol/kg), a pegylated dye of approximately 25 kDa used for assessing macromolecule vascular leakage, 23 or R800 (0.032 μmol/kg), a NIRF dye with P-gp substrate properties. 24 The NIRF probes were administered either alone or in combination with LPA (1 mg/kg) via tail vein injection. The mice were killed at various times (10 to 40 minutes) after treatment via cardiac perfusion with 10% formaldehyde solution. The brain and other tissues were removed and the accumulation of NIRF dyes examined ex vivo using an Odyssey near infrared imaging system (Licor, Lincoln, NE, USA). Quantitative assessment of fluorescence was performed on regions of interest in 2-mm thick coronal tissue slices and normalized to fluorescence from blood samples taken at the time of tissue collection. Resulting values were presented as relative fluorescence units per mm3 of tissue divided by relative fluorescence units per μL blood.
Desensitization of lysophosphatidic acid -mediated blood–brain barrier disruption
Desensitization to BBB disruption produced by LPA was examined using both MRI and NIRF imaging agents. Mice were anesthetized and administered LPA via tail vein injection under three different dosing schedules (A–C). Those mice under dosing schedule A received LPA (1 mg/kg) as a single-bolus injection. Mice under dosing schedule B received a second bolus injection of LPA (1 mg/kg) 20 minutes after the first injection. Mice under dosing schedule C received a third bolus injection of LPA (1 mg/kg) 20 minutes after the second injection. All mice were imaged using either Gd-DTPA contrast agent or IRdye 800cw after the last LPA injection. Quantitative assessment of BBB disruption was determined as described above.
Lysophosphatidic acid-mediated blood–brain barrier permeability to radiolabeled methotrexate
To determine potential application of LPA for enhanced drug delivery to the brain, female Balb/c mice were administered a bolus injection (mL/kg) of 3H-methotrexate (4 μCi total activity) via the tail vein. The radiolabeled methotrexate was given in combination with either LPA (1 mg/kg) or vehicle (0.25% bovine serum albumin in phosphate-buffered saline solution). Fifteen minutes after the injection of methotrexate, mice were anesthetized using intraperitoneal injection of ketamine and xylazine and a sample of blood was removed by cardiac puncture, collected in heparinized tubes and centrifuged for 10 minutes at 1000 × g to obtain plasma aliquot (100 μL). After the blood sample, mice were killed and the brain was removed for determination of 3H-methotrexate. The brains were weighed and homogenized with double the volume of tissue solubilizing agent (Scintigest, Fisher Scientific, Fair Lawn, NJ, USA). A 200-μL aliquot of the tissue homogenate was used to analyze the tissue samples. The samples were then mixed with Ultima gold (Perkin Elmer) scintillation cocktail and analyzed by liquid scintillation counter (Perkin Elmer, Shelton, CT, USA) to determine the amount of 3H-methotrexate present in the plasma and tissue. The distribution of 3H-methotrexate was based on the ratio of radioactivity in the brain versus the radioactivity in the plasma sample.
RESULTS
Lysophosphatidic Acid Receptor Expression Profile and the In Vitro Response of Lysophosphatidic Acid
The messenger RNA expression profile of LPA receptors was examined in both human BMECs and mouse brain homogenate and capillary-enriched fractions. Both LPAR1, LPAR2, and LPAR3 were highly expressed in human BMECs (Figure 1A). All five LPARs were detected in mouse brain homogenates at the messenger RNA level (Figure 1B). The expression of LPAR1, LPAR4, and LPAR5 was enriched in the capillary fraction in comparison with that in the brain homogenate (Figure 1C). In contrast, expression levels of LPAR2 and LPAR3 were less in the capillary fraction compared with that observed in total brain homogenate (Figure 1C).
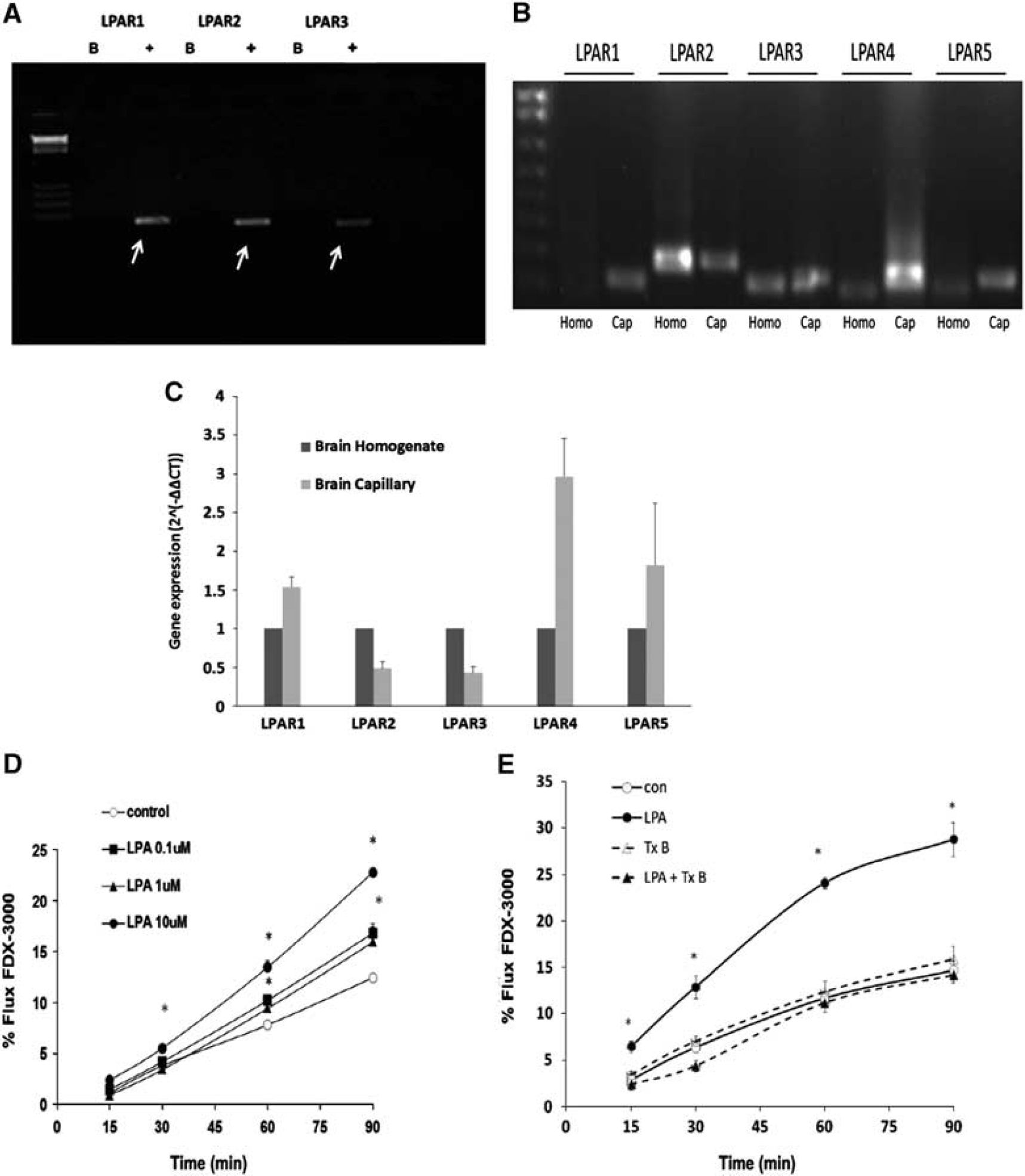
Lysophosphatidic acid (LPA) receptor expression profile and permeability responses in cultured brain microvessel endothelial cell (BMEC) after LPA exposure. Expression of various LPA receptors was examined using real-time polymerase chain reaction (RT-PCR) in (
LPA-Induced Changes in Brain Microvessel Endothelial Cell Monolayer Permeability
The effects of LPA on brain microvessel endothelial cell permeability were examined in primary cultured BMECs. Exposure of confluent bovine BMECs to LPA resulted in a rapid increase in permeability (Figure 1D). The effects of LPA were both rapid, occurring within 15 minutes, and concentration dependent (Figure 1D). LPA-induced increase in permeability was also observed in human BMEC monolayers (Figure 1E). The increases in permeability produced by LPA were completely abolished by pretreating the BMEC with the Rho kinase inhibitor, TcdB (Figure 1E).
Disruption of Blood–Brain Barrier Permeability After Lysophosphatidic Acid Exposure
The effects of LPA on BBB permeability was initially characterized using Gd-DTPA contrast-enhanced MRI. Figure 2 shows a representative series of T1-weighted MR images taken from the posterior, mid brain, and anterior brain regions of vehicle (2A and 2B) or LPA-treated (2C and 2D) mice. Under control conditions, the BBB is intact with negligible Gd-DTPA enhancement observed, as indicated by the similar Gd-DTPA intensity in the brain slices at time 0 (before the injection of vehicle solution) and time 6 (6 minutes after vehicle injection) (Figures 2A and 2B, respectively). Indeed, the only brain region showing appreciable accumulation of Gd-DTPA contrast agent under normal conditions was the ventricles (Figure 2B). In contrast, after the administration of 1 mg/kg LPA, the amount of Gd-DTPA contrast agent accumulating in brain after 6 minutes (Figure 2D) was substantially enhanced compared with images obtained before LPA injection at time 0 (Figure 2C). While there were regional differences in Gd-DTPA intensity observed in both the vehicle- and LPA-treated mice, increases in Gd-DTPA distribution to the brain after LPA administration was observed in all regions examined. Quantitative assessment of Gd-DTPA contrast enhancement in the various brain regions indicated an approximately fivefold increase in Gd-DTPA intensity in the LPA treatment group compared with control mice in all regions of the brain examined (Figure 2E–G). The increase in Gd-DTPA intensity was rapid, occurring within 3 to 6 minutes after systemic administration of LPA (Figure 2E–G). Furthermore, the disruption produced by LPA was dose dependent with both the 1 mg/kg and 10 mg/kg doses of LPA causing significant increases in Gd-DTPA accumulation in the brain (Figure 2H). In contrast, the lowest dose of LPA examined (0.1 mg/kg) had no effect on BBB permeability. Interestingly, the magnitude of BBB disruption produced by 10 mg/kg of LPA was less than that observed with the 1 mg/kg dosage (Figure 2H). The BBB disruption produced by bolus intravenous LPA injection was also short in duration with restoration of BBB permeability returning within a 20-minute period. This is demonstrated by the lack of further Gd-DTPA enhancement observed after a second dose of Gd-DTPA administered 21-minutes after LPA exposure (Figure 2E–G). Additional confirmation of transient nature of the BBB disruption is the observation that administration of Gd-DTPA contrast agent 20-minutes after LPA exposure resulted in no significant increases in Gd-DTPA accumulation in the brain (Figure 3).
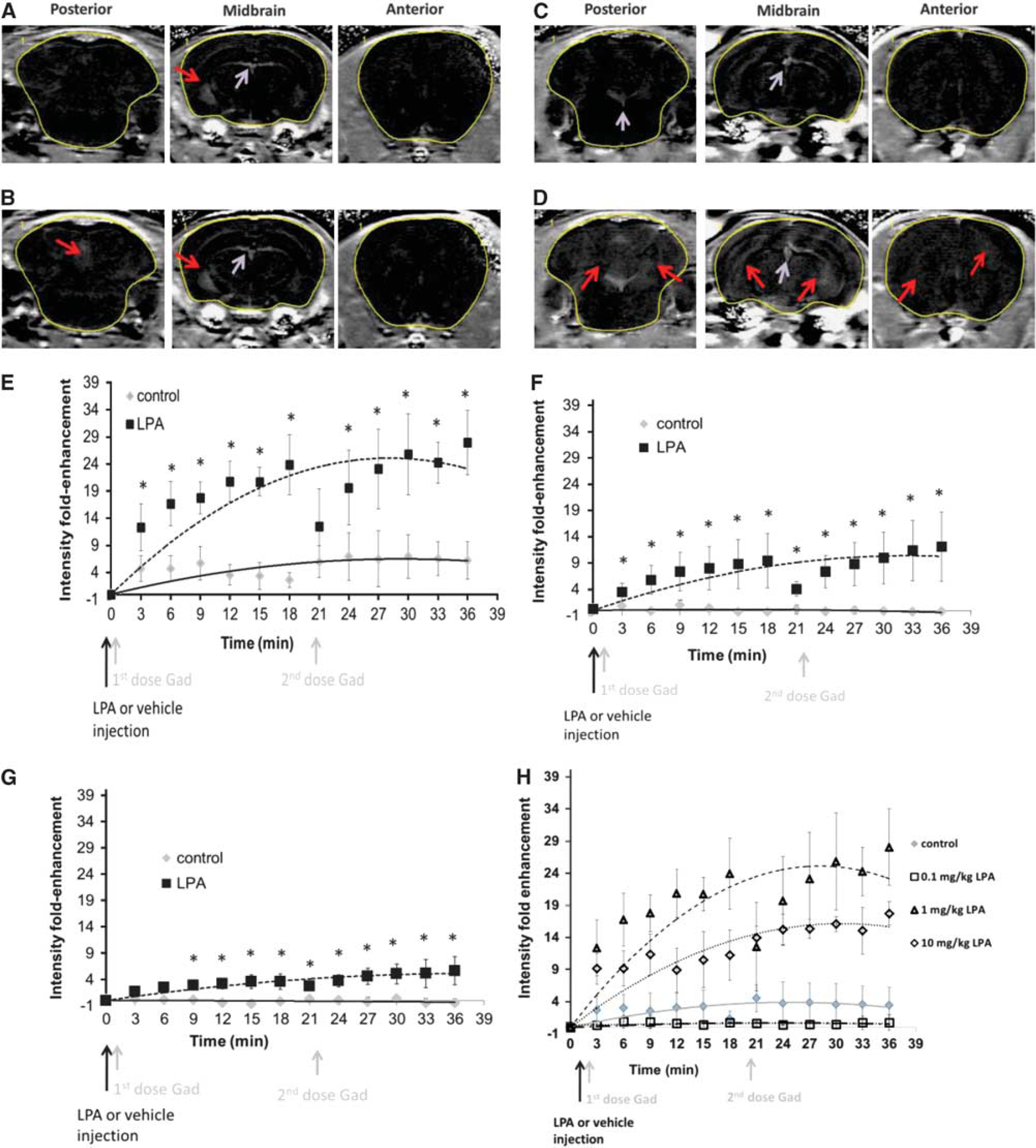
Assessment of lysophosphatidic acid (LPA)-mediated effects on blood–brain barrier (BBB) permeability using gadolinium diethylenetriaminepentaacetate (Gd-DTPA) contrast-enhanced magnetic resonance imaging. Representative T1-weighted images of mouse brain at time 0 (
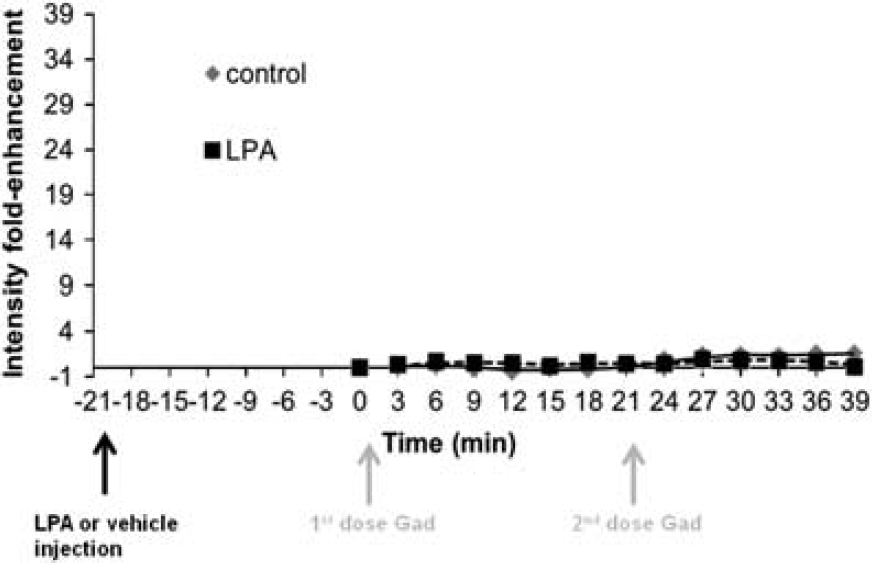
Quantitative analysis of pixel intensity for Gad enhancement normalized to the pixel intensity at time 0 after a 20-minute pretreatment with either vehicle (0.25% bovine serum albumin in phosphate-buffered saline) or 1 mg/kg lysophosphatidic acid (LPA). ∗P<0.05 compared with control mice at the same time point. Values represent the mean±s.e.m. for four mice per treatment group.
Effects of Lysophosphatidic Acid on Blood–Brain Barrier Permeability to Macromolecules
To determine the extent to which LPA exposure could alter macromolecule permeability in the BBB, studies were performed using IRdye800 cw PEG, a 25 kDa near infrared fluorescent permeability marker. Under control conditions, there was little if any appreciable dye accumulation in the brain, with most of the IRdye800 dye remaining within the vasculature of the brain as shown in Figures 4B and 4D. Indeed, the other brain regions were similar to the negative control animal receiving no IRdye800 cw PEG injection (Figure 4A). In contrast, exposure to LPA significantly enhanced the penetration of IRdye800 cw PEG in the brain tissue (Figures 4C and 4E) both at 20 and 40 minutes post injection of the dye. The time frame for disruption observed with IRdye800 was similar to that with Gd-DTPA. This was evident at the 20-minute time point, where intense fluorescence near and around the area of the cerebral blood vessels were observed, consistent with leakage of the dye from the cerebral vasculature, (Figure 4C). However, by 40 minutes post LPA exposure, fluorescence intensity in the area around the vasculature was reduced (Figure 4E). Quantitative assessment of IRdye800cw PEG penetration into the brain tissue indicated a significant increase in BBB permeability of IRdye800cw PEG after LPA (Figure 4F). Interestingly, systemic administration of LPA did not significantly impact the permeability of IRdye800cw PEG in other vascular beds including the lung, liver and kidney (Figure 4F).
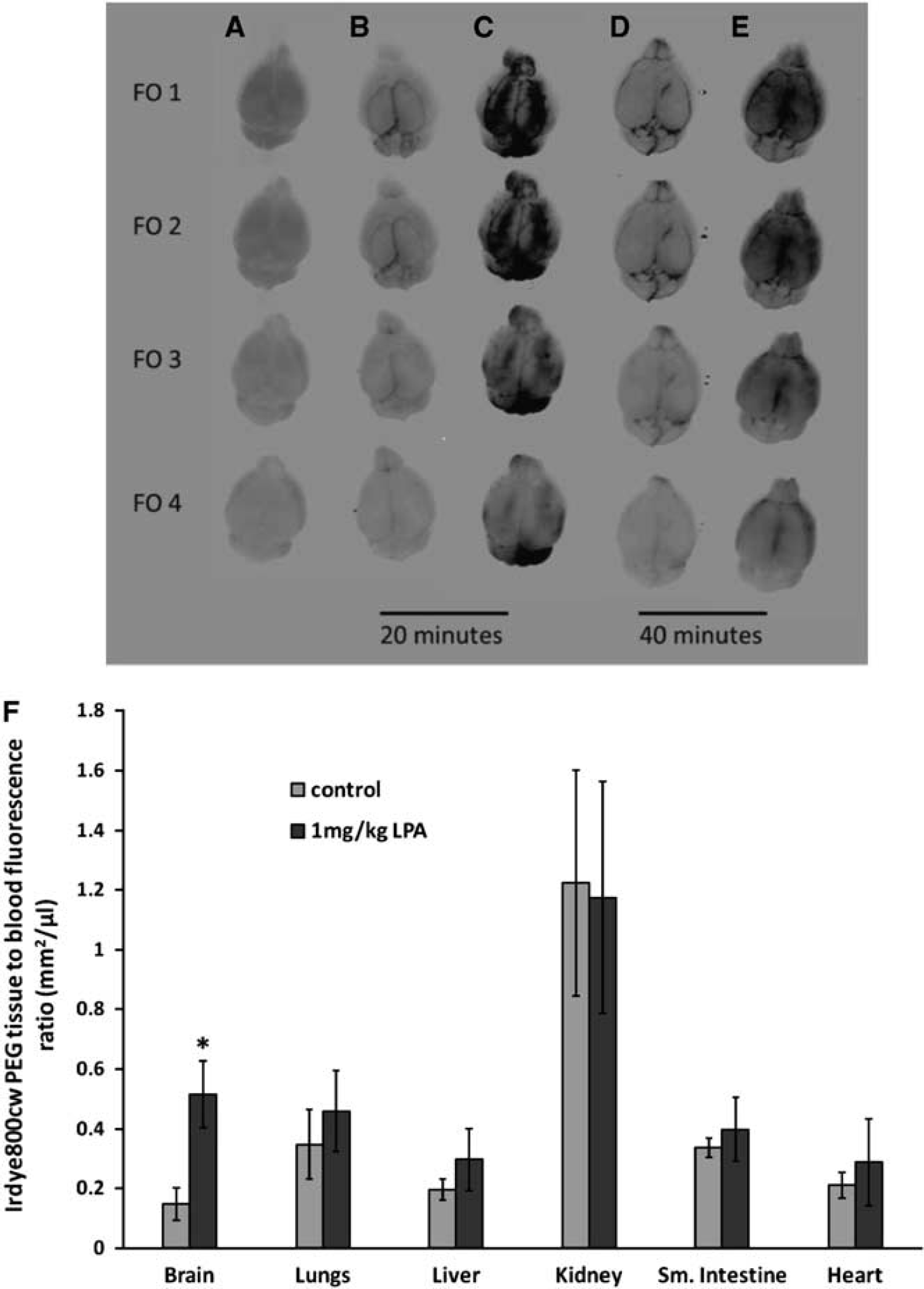
Effects of lysophosphatidic acid (LPA) on large macromolecule permeability in the blood–brain barrier. Qualitative accumulation of IRdye 800cw PEG in whole brains from vehicle (0.25% bovine serum albumin in phosphate-buffered saline) injected mice (
Tachphylaxis to Lysophosphatidic Acid-Induced Blood–Brain Barrier Disruption
Tachyphylaxis to LPA-induced BBB disruption was examined using both small (Gd-DTPA) and large (IRdye800 cw PEG) molecular weight permeability markers. In the case of IRdye800 cw PEG, quantitative analysis of the dye revealed significant brain enhancement in the LPA-treated mice at 20 minutes post injection of IRdye800 cw PEG when compared with their control counterparts (Figure 5B). However, when a second LPA injection was given to the mice, 20 minutes apart, the amount of IRdye800 cw PEG accumulating in the brain tissue was significantly less (60%) than observed after a single injection of the phospholipid (Figure 5A–B). By the third injection of LPA, the brain accumulation of IRdye800cw PEG contrast agent was similar to that of control mice receiving no LPA (Figure 5A–B). However, when the Gd-DTPA contrast agent was used to assess BBB disruption, there was no apparent loss of response to repeated injections of LPA (Figure 5C–D).
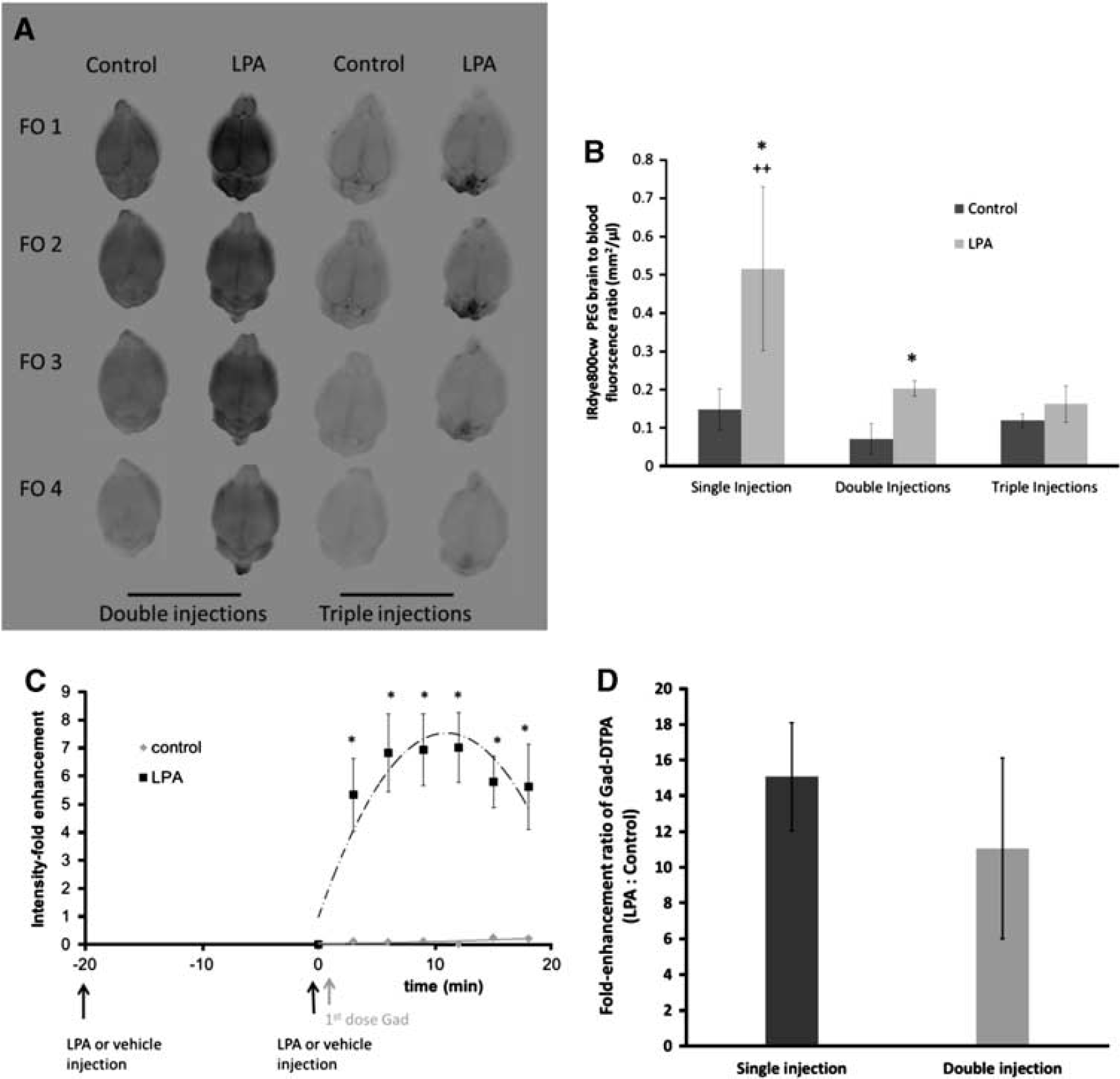
Permeability responses after repeated exposure to lysophosphatidic acid (LPA). Assessment of IRdye800cw PEG (
Effects of Lysophosphatidic Acid on R800 Permeability in the Blood–Brain Barrier
The accumulation of a P-gp substrate, Rhodamine 800 (R800), was also examined in mice brain under normal conditions and after BBB disruption with LPA. Representative ex vivo NIRF images of R800 in the brain are shown in Figure 6A. Initial assessment of BBB permeability, performed at 20 minutes after LPA, indicated no enhancement in R800 accumulation in the brain (Figure 6B). However, examination of R800 accumulation in the brain after a 10-minute exposure to LPA resulted in a nearly twofold enhancement in R800 fluorescence (Figure 6B).
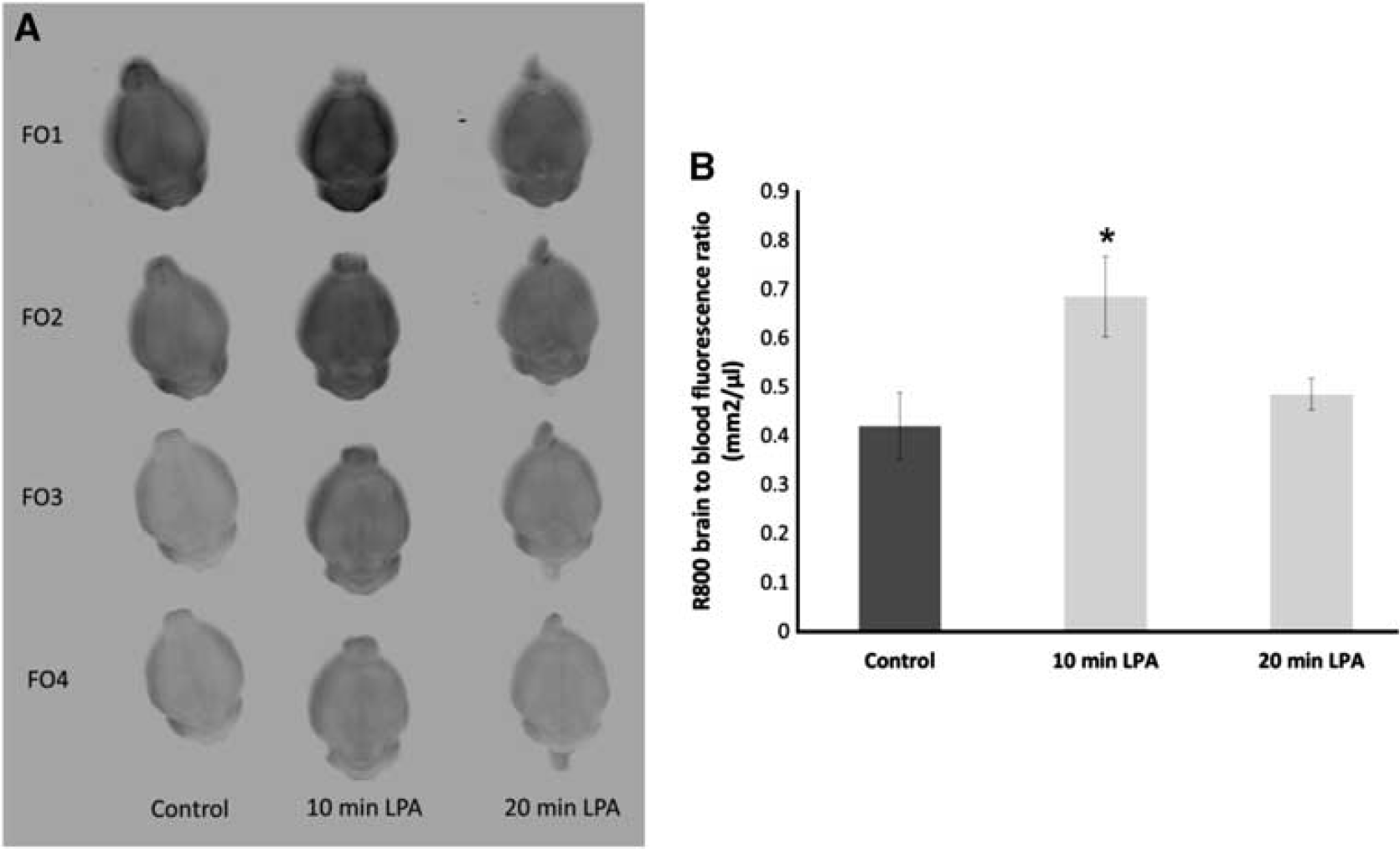
Effects of lysophosphatidic acid (LPA) on blood–brain barrier permeability of P-gp probe. Qualitative (
Lysophosphatidic Acid-Mediated Blood–Brain Barrier Permeability to Radiolabeled Methotrexate
As the first step in potential utilization of LPA to transiently disrupt BBB permeability for enhancing drug delivery to the brain, the study also examined the brain accumulation of radiolabeled methotrexate. Under control conditions, the brain penetration of methotrexate was minimal with a brain to plasma ratio of 1% to 2%. However, after LPA administration, radiolabeled methotrexate reached brain to plasma ratios of over 20%, an approximately 20-fold enhancement over that observed in control mice with intact BBB (Figure 7).
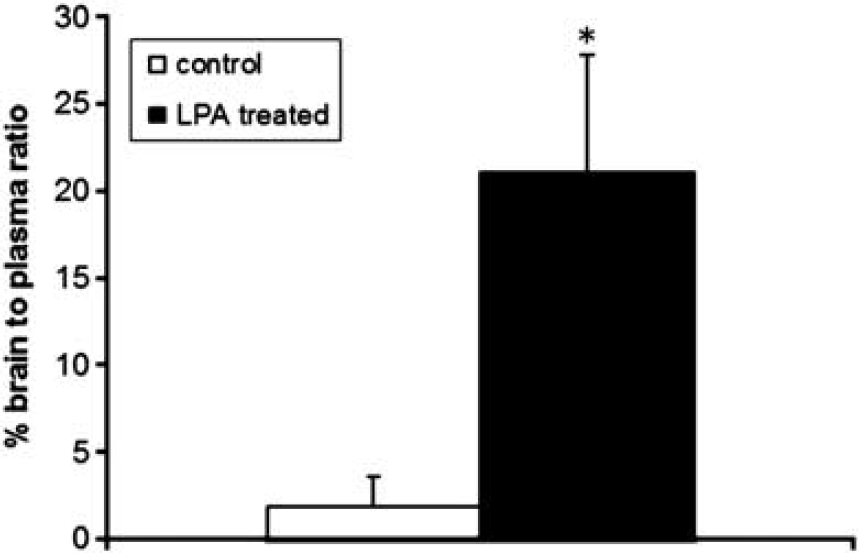
Quantitative assessment of radiolabeled methotrexate in the brain after single intravenous bolus injections of vehicle (0.25% bovine serum albumin in phosphate-buffered saline) or lysophosphatidic acid (LPA) (1 mg/kg). Values are expressed as the brain to plasma ratio of radioactivity and represent the mean±s.e.m. of six mice per treatment group. ∗P<0.05 compared with control mice receiving LPA vehicle injection.
DISCUSSION
Lysophosphatidic acid is one of the simplest glycerolphospholipids. While originally thought to be an intermediate in lipid metabolism, it is now known to mediate a variety of biologic responses depending on the cellular target. 2 Biologic responses to LPA are initiated through binding of the phospholipid to LPA receptors on the plasma membrane of the cell. Hecht JH et al 7 first identified LPA as a ligand for ventricular zone-1 receptor, which is a member of endothelial differentiation gene (EDG) family. There are currently at least six different types of LPA receptors (LPAR1–LPAR6).8, 10 Based on amino-acid sequence, LPAR1–LPAR3 share 50% homology and belong to the EDG subfamily of G-protein coupled receptors formally known as EDG2, EDG4, and EDG7, respectively. The remaining LPA receptors, including LPAR4 (GPR23), LPAR5 (GPR92), and LPAR6 (GPR87) are structurally distinct from the EDG family and share less than 40% amino-acid sequence homology with the conventional LPARs. 8
Within the brain, LPAR1 is the most highly expressed, although LPAR2 and LPAR3 are also present. 10 Considering the pleiotropic effects of LPA in the brain along with the upregulation of LPAR1-3 after injury, it has been proposed that LPA may regulate essential aspects of cellular organization and have a crucial role in reactive astrogliosis, neural regeneration, and axonal regrowth. 25 Less is known about the role of LPAR4 and LPAR5 within the brain. Studies by Yanagida K et al 26 reported a high expression of LPAR4 in neuronal cells and observed that activation of the LPAR4 receptor was associated with morphological changes including cell rounding and N-cadherin-associated cell aggregation. 26 LPAR5 is highly expressed in the brain, as well as genital organs, and skeletal muscle. 27 However, the role of LPAR5 within the brain tissue is largely unknown. In the context of the present study, all known LPARs were detected at the messenger RNA level in mouse brain homogenate. Compared with the expression pattern observed in brain homogenate, LPAR1, LPAR4, and LPAR5 had a much higher level of expression in the capillary fraction, suggesting potential enrichment in the brain capillaries. In contrast, the expression of LPAR2 and LPAR3 in the capillary fraction was substantially reduced compared with the brain homogenates suggesting non-capillary sources for these particular LPA receptors in the brain.
The enriched expression of several LPA receptors within the brain microvasculature is not surprising, given that the vascular endothelium is a major target for LPA with changes in proliferation, angiogenesis, and permeability reported after exposure to the phospholipid.12, 28 In the case of permeability, most studies including the present study support an increase in vascular leakage after LPA exposure.12, 16 In cultured BMECs, LPA exposure has been associated with increases in permeability.11, 12 However, there are contradictory reports of LPA-induced reductions in endothelial cell permeability (i.e., barrier-enhancing properties). 15 The conflicting reports regarding LPA-induced permeability could be dependent on a number of factors including differences in vascular bed, species, or the profile of LPA receptors expressed. In the case of the later, activation of both LPAR1 and LPAR3 has been linked to vascular permeability alterations.13, 28 The receptors appear to mediate divergent responses with LPAR1 enhancing permeability of the barrier through the activation of RhoA pathway and LPAR3 reducing permeability through the activation of Rac pathway.2, 12, 28, 29, 30 A pretreatment of human brain microvessel endothelial cell with either LPA or toxin B, an inactivator of the Rho family of small ATPases, 31 or combination of both LPA and toxin B confirmed that the permeability enhancing effect associated with LPA was most likely through the activation of Rho kinase pathway. Furthermore, an LPAR3 mechanism has been proposed to explain the ability of LPA to restore the barrier properties of primary small airway epithelial cells (HSAEpCs) after endotoxin-induced disruption. 29 Based on the apparent opposing effects that LPAR1 and LPAR3 activation have on permeability, it is tempting to suggest the dose-dependent effects of LPA observed in the present study may reflect differences in LPARs activated in the brain microvasculature. Further compounding this issue is the inhibitory cross-talk between Rac and RhoA that determines the balance of cellular response to LPA.2, 3 However, based on the expression levels of LPARs detected within the mouse capillary-enriched fractions, together with the role of LPAR1 in the vascular permeability increases produced with LPA, 13 this receptor is likely involved in the BBB permeability responses observed in the present study. Definitive determination of LPAR1 involvement in the BBB permeability alterations observed in the present study will require additional study with selective receptor antagonists or knockout mouse models.
The present study is the first to demonstrate enhanced BBB permeability after systemic administration of LPA in an in vivo model. In adult Balb/c mice, bolus injections of LPA produced dose-dependent disruption of BBB integrity as assessed using contrast agent. While the use of Gd-DTPA to identify localized cerebral microvascular leakage resulting from brain tumors or stroke is common, 22 the present study used contrast-enhanced MRI to characterize both the time course and magnitude of BBB disruption after LPA exposure. An advantage in using MR imaging technology is the fast acquisition time that allows monitoring of rapid changes in BBB permeability within the same animal. These studies demonstrated significant enhancement of BBB permeability to Gd-DTPA with the onset of effect observed within 3 minutes of LPA exposure. The rapid onset of LPA action is consistent with a G-protein coupled receptor response within the brain microvasculature.
In addition to its rapid onset, the LPA-induced modulation of BBB permeability was transient in nature with restoration of BBB integrity occurring within a 20-minute time frame. The rapid dissipation of LPA action is most likely due to the relatively short half life of LPA. Lipid phosphate phosphatases hydrolyze numerous lipid phosphates within the extracellular fluid compartment including LPA, phosphatidate, sphingosine-1-phosphate, ceremide-1-phosphate, and diacylglycerol pyrophosphatate. 32 Indeed, increased expression of lipid phosphate phosphatases resulted in the increased dephosphorylation of LPA, SIP, and CIP. 33 Furthermore, studies with LPP knockout mice (Ppap2atr/tr) showed a significant increase in plasma LPA concentration compared with wild-type mice. The studies with the Ppap2atr/tr mice reported a plasma half life for LPA of approximately 12 minutes as opposed to 3 minutes in wild-type mice. 34 As complete elimination of a bolus injected agent occurs within five elimination halflives, 35 there is likely to be negligible amounts of LPA left within 15 minutes of administration in the present study. Such a time frame matches BBB-disruption window observed with LPA administration in the present study.
Another advantage of using Gd-DTPA-based MRI to monitor changes in BBB permeability produced by LPA is the ability to capture regional differences in BBB permeability. Under control conditions, in the absence of LPA exposure, Gd-DTPA contrast enhancement was greatest in the posterior sections of the brain compared with mid brain and anterior regions. This suggests a slightly higher BBB permeability in the posterior brain regions. Such regional differences in BBB permeability have been reported previously; 36 however, identification is difficult with many of the techniques commonly used for assessing BBB permeability. Regional differences in the magnitude of Gd-DTPA contrast enhancement was also observed in the LPA treatment group. However, the net increase in BBB permeability to Gd-DTPA contrast agent after LPA exposure compared with vehicle-treated mice was approximately fivefold throughout all brain regions examined. These findings suggest that the magnitude of cerebrovascular permeability increases in response to LPA were similar throughout all regions of the brain examined.
To fully characterize the extent of BBB disruption produced by LPA, additional imaging studies were performed using the near infrared fluorescence (NIRF) contrast agent, IRdye 800cw PEG. Previous studies have used IRdye 800cw PEG to examine vascular leakage and lymphatic drainage. 23 With a molecular weight of 25 kDa, little dye penetration into the brain was expected after systemic administration. This was certainly the case in the present study, where, under control conditions, the IRdye800cw PEG dye was confined to the cerebrovascular space. However, when the dye was administered systemically, in combination with LPA, a substantial enhancement of IRdye 800cw PEG leakage was observed in the brain. These findings indicate that LPA-induced BBB disruption was not limited to small molecular weight compounds but was also present for larger macromolecules. Furthermore, the intense dye staining around the area of the vessels at 20 minutes compared with the more dispersed dye distribution in the brain observed at 40 minutes post LPA exposure further confirms the transient nature of the BBB disruption produced by the phospholipid. The enhanced BBB permeability observed with IRdye 800cw PEG was smaller in magnitude than that observed with Gd-DTPA contrast agent. This finding is consistent with LPA-induced disruption of tight junctions within the BMECs as such disruption would be expected to display a size dependency with small hydrophilic solutes having greater permeability changes compared with large macromolecule solutes.
To confirm that the mechanism of LPA-induced BBB disruption was primarily due to the enhanced paracellular diffusion through altered tight junction complexes, additional imaging studies were performed using R800. Rhodamine 800 is a small, lipophilic, NIRF molecule, which has been used to assess P-glycoprotein-mediated transport processes in the BBB. 24 Previous BBB permeability studies with R800 showed only minimal brain accumulation of the NIRF agent due to P-gp-mediated efflux transport at the BBB. 24 Therefore, R800 appeared to be an ideal agent to assess the effects of LPA-mediated BBB disruption on solutes that undergo transcellular routes of passage through the BBB. In contrast to both Gd-DTPA and IRdye 800cw PEG, the brain accumulation of R800 in LPA-treated mice was not significantly greater than vehicle-treated mice at 20 minutes after LPA. Examination of R800 fluorescence at 10 minutes post LPA treatment showed significantly greater brain penetration compared with control mice. However, it should be noted that even at this time point enhancement was not as pronounced as that observed with Gd-DTPA contrast agent. The reduced effects of LPA-mediated BBB disruption on R800 passage into the brain are most likely attributable to the significant transcellular exchange mechanisms that influence R800 permeability in the BBB. Thus, while LPA may increase the paracellular diffusion of R800 into the brain, this is off-set by transcellular diffusion and P-gp-mediated transport of the dye from brain tissue back to the blood. Based on these results, it appears that LPA-mediated disruption of the BBB will have the greatest impact on hydrophilic agents whose transcellular permeability in the BBB is limited.
Desensitization due to receptor internalization is a common feature of G-protein coupled receptors. 37 In the classic desensitization model, repeated exposure to the ligand initiates receptor desensitization by phosphorylating the receptor, thus preventing the coupling of G-protein and a loss of response (i.e., tachyphylaxis) to receptor activation. 38 Recent studies have reported tachyphylaxis in regards to the permeability response to LPA. 13 Using an in situ preparation in which LPA was topically applied to the cerebral microvasculature, continuous infusion of LPA over 10 minutes resulted in reduced permeability to subsequent LPA challenge. 13 The potential development of tachyphylaxis to LPA-induced BBB disruption in vivo was examined in the present study. While repeated exposure to bolus injections of LPA (at 20 minutes intervals) enhanced the BBB permeability of Gd-DTPA contrast agent to a similar extent, significant reductions in BBB disruption were observed when the IRdye 800cw PEG imaging agent was used. As Gd-DTPA contrast agent is a relatively small molecule (547 kDa) compared with IRdye 800cw PEG (approximately 25 kDa), the magnitude of disruption of tight junctions necessary to result in the loss of BBB integrity to small hydrophilic agents is substantially less than that required for larger macromolecules. Thus, desensitization of LPA receptor-mediated permeability responses is likely to be more apparent with large macromolecules in the BBB. The mechanism by which tachyphylaxis to LPA-mediated BBB disruption occurs remains to be determined, however, the decrease response of LPA is likely attributed to the uncoupling of the protein through the phosphorylation of the receptor and subsequent receptor internalization. In support of this, studies by Avendeno-Vazquez et al 9 reported rapid internalization of LPAR1 in hepatocytes continuously exposed to LPA.
While the current study showed LPA is capable of increasing BBB permeability in vivo, little is known about the potential pathophysiological role LPA may have in regulating BBB permeability. As the phospholipid is produced from activated platelets, 2 LPA may contribute to alterations in cerebrovascular permeability that occurs during ischemic stroke. In support of this, elevations in plasma LPA have been reported in patients with ischemic cerebrovascular disease. 39 Interestingly, the plasma levels of LPA in the ischemic cerebrovascular disease groups were approximately 3 μmol/L. This is comparable to the dose of LPA that produced maximal increases in BBB permeability in the current study (2.3 μmol/L/kg). Systemic exposure to this dose of LPA did not appear to influence permeability in other vascular beds known to be responsive to LPA such as the pulmonary vasculature, which requires a minimum dose of 5 to 10 μmol/L/kg, 40 suggesting the cerebral microvasculature may have a greater sensitivity to the barrier-disrupting properties of LPA.
The rapidity of the permeability response to systemically administered LPA observed in the present study, both in onset of effects and recovery to baseline, suggests LPA may also have applications for enhancing drug delivery to the brain. Transient disruption of BBB permeability through either hyperosmotic or chemical means has been used to enhance chemotherapeutic delivery in brain tumors. 41 As the first step in potential utilization of LPA to transiently disrupt BBB permeability for enhancing drug delivery to the brain, the present study examined the brain accumulation of radiolabeled methotrexate. Under control conditions, the brain penetration of methotrexate was minimal with a brain to plasma ratio of 1% to 2%. However, after LPA administration, radiolabeled methotrexate accumulation in the brain was enhanced by approximately 20-fold compared with that observed with an intact BBB. This is similar in magnitude to the responses reported previously with osmotic disruption and bradykinin receptor activation. 41 While more experiments are required, these results provide proof-of-principle for the potential use of LPA or LPA analogs for increasing drug delivery to the brain.
In summary, using various imaging modalities including MRI and NIRF, the present studies demonstrated that LPA was able to increase BBB permeability. Consistent with the disruption of tight junction complexes, increases in BBB permeability were most apparent for the small hydrophilic permeability marker, Gad. However, significant increases in BBB permeability were observed after LPA exposure for both the large hydrophilic permeability marker, IRdye800 cw PEG, and the P-glycoprotein-dependent permeability marker, R800. The LPA-induced enhancement of BBB permeability was transient with complete restoration of BBB integrity observed within a 20-minute time period. The ability of LPA to modulate BBB permeability has potential pathophysiological as well as therapeutic implications.
Footnotes
The authors declare no conflict of interest.
