Abstract
Persistent neurochemical abnormalities in frontal brain structures are believed to result from methamphetamine use. We developed a localized 13C magnetic resonance spectroscopy (MRS) assay on a conventional MR scanner, to quantify selectively glial metabolic flux rate in frontal brain of normal subjects and a cohort of recovering abstinent methamphetamine abusers. Steady-state bicarbonate concentrations were similar, between 11 and 15 mmol/L in mixed gray-white matter of frontal brain of normal volunteers and recovering methamphetamine-abusing subjects (
Introduction
The neuropathologic presentation of substance abuse and addiction, a serious health, social, and economic problem in the United States, involves abnormalities of the dopaminergic and glutamatergic systems leading to neuronal–glial dysfunction (Stephans and Yamamoto, 1994; Volkow et al, 2001, 2002). It has been commonly observed that objective neurochemical changes persist beyond the period of active drug use, inviting the hypothesis that persistent neurochemical abnormalities contribute to addictive behavior and relapse (Ernst et al, 2000; Sekine et al, 2002). The timing and possible role of glial injury in addiction and relapse is poorly understood but may have implications for the design of pharmaceuticals for future treatment of addiction and relapse prevention. Evolving methods of noninvasive brain analysis, in particular 13C magnetic resonance spectroscopy (MRS) with 13C glucose and 13C acetate, respectively (Bachelard, 1998; Bluml et al, 2001, 2002; Cerdan and Seelig, 1990; Gruetter et al, 1993; Lin et al, 2003; Mason et al, 1999), offer new opportunities to examine the impact of drugs of abuse on neurons and glia separately. This pilot study was undertaken to establish the feasibility of applying [1-13C]acetate MRS to quantify glial metabolic rate in frontal brain structures implicated in drug abuse, reward, and relapse (Gass and Olive, 2008). Future studies, in which the starting substrate would be [2-13C] glucose, could then be applied to probe the impact on neuronal metabolism directly (Gropman et al, 2009).
Glutamate (Glu) is an amino acid intimately involved in neurotransmission and a marker of mitochondrial oxidative status and tricarboxylic acid (TCA) cycle function. The glutamate–glutamine cycle links glia with neurons in the mechanism of glutamate neurotransmission, the neurotransmitter pathway overall believed to account for 80% of brain neurotransmitter function. The functions of glial cells in neurotransmission are often viewed as merely supportive; however, failure of glial reuptake of glutamate released at synapses can result in accumulation of extracellular glutamate, leading to neurotoxicity. Glutamate is crucial in regulating both the development and the expression of addictive behaviors, which requires glutamate receptor stimulation in the ventral tegmental area and is associated with enhanced glutamate release in the prefrontal cortex (Kalivas et al, 2009; Rebec and Sun, 2005). Using the single voxel proton MRS method, which determines steady-state concentration of brain metabolites, we found that preliminary data provide more direct evidence of elevated frontal brain glutamate concentration during the cerebral adaptation to methamphetamine abuse (Abulseoud et al, 2009). This is in addition to earlier reports of persistent neurochemical abnormalities; elevated myoinositol, a glial marker, and reduced
The aim of this pilot study was to determine the extent of glial metabolic abnormalities in abstinent methamphetamine-dependent (AMD) subjects using MRS. 13C MRS techniques, which permit TCA cycle rate determination directly and further allow the dissection of glial from neuronal pathways of glutamate metabolism based on the selective uptake and metabolism of acetate by glia (Muir et al, 1986) and glucose by neurons have recently been explored in intact brains of animals and humans (Bluml et al, 2002; Cerdan, 2003; Lebon et al, 2002; Ross et al, 2003; Rothman et al, 2003; Shen and Rothman, 2002). A limitation of previous studies in relation to the present purpose of drug abuse has been the need to confine human 13C MRS assays to posterior brain regions. This brain region is distant from heat-sensitive optical structures but purportedly of minimal relevance to the neuropsychopathology of drug abuse, which most investigators locate in frontal and limbic pathways (London et al, 2000). These technical limitations have recently been overcome (Sailasuta et al, 2008) with two distinct and robust
Materials and methods
Subjects
A total of 21
Carbon MRS
The techniques for quantification of glial metabolic rate of
To establish metabolic rates for [1-13C]acetate for the frontal lobe of human brain, not hitherto examined, we developed low rf power 13C MRS (Sailasuta et al, 2008). To minimize RF heating in a clinical magnetic resonance imaging system, all subjects underwent 13C MRS at 1.5 T GE clinical scanner (GE Healthcare, Milwaukee, WI, USA) equipped with broadband exciter, a stand-alone proton decoupler, and a vector signal generator (Agilent Technology E4433B, Santa Clara, CA, USA). A half-helmet head coil was used with the subject lying supine within the MR scanner. Its construction was a modification of the half-head coil used in the posterior brain 13C MRS study from this laboratory (Bluml et al, 2001). The coil consisted of a 4-inch proton loop and a 3.5-inch 13C loop for transmission and reception. Fast spin echo imaging scan was used to confirm the consistency of the observed volumes in all patients. For the frontal brain study, the half-helmet coil placement was such that the anterior portions of the frontal lobe were mainly included in the field of view and similarly for the posterior brain where the posterior region of the brain was included in the field of view (Gropman et al, 2009). The coil was tuned to best match both proton and carbon channels. Power deposition was continuously monitored and remained below Federal guidelines for Specific Absorption Rate.
Because the method used is new, to confirm that metabolic rates for [1-13C]acetate in human frontal lobe are comparable to posterior parietal brain, values previously reported using [1-13C]acetate by us, and also recently confirmed using [2-13C] acetate by Lebon et al (2002), assays of 13C enrichment in the posterior brain were performed in four normal subjects. To allow for complete clearance of 13C-enriched metabolites from the posterior brain for three of the four normal subjects, this examination was followed a minimum of 48 h later by examination of the frontal brain region in each of the three normal subjects.
Magnetic resonance image was acquired at the outset of each 13C MRS examination, indicating regions of interest of mixed white and gray matter of posterior parietal or frontal and prefrontal brain structures, respectively, for 13C MRS acquisition. Brain volumes, approximately 100 cm3 in each protocol, included similar proportions of white and gray matter, were comparable and have been previously published (Gropman et al, 2009; Sailasuta et al, 2008).
Determinations of Cerebral Bicarbonate Concentration from Natural Abundance 13C MRS and Maximum Rate of HCO3− (maxVHCO3) Production
Natural abundance 13C MRS was acquired for 20 mins before the start of the acetate infusion to show adjacent, well-resolved individual resonances for bicarbonate and for Cr + PCr (total creatine, tCr) as previously reported by Bluml et al (2001). Because tCr has been used successfully as an internal metabolite reference in several proton MRS studies in abstinent methamphetamine users (Nordahl et al, 2005, 2002; Salo et al, 2007) as well as other human studies (see review, Xu et al, 2008), and does not become enriched during the time scale of the present studies, we have used this as an internal reference to quantify HCO3− and its subsequent enrichment from [1-13C]acetate. To improve signal to noise (natural abundance 13C = only 1.1% of total) and thereby improve quantification, we acquired additional natural abundance 13C brain spectra during separate imaging sessions not associated with 13C infusions. The timed intravenous infusion of 99% 13C-enriched [1-13C]acetate infusion was started and brain spectra were acquired continuously thereafter for a minimum of 2 h. The 6½ min blocks of low-power noise nuclear overhauser effects (0.9 W for frontal brain and 2.5 W for the posterior brain) 13C MRS spectra acquired were stored and summed, as previously described (Sailasuta et al, 2008). 13C MRS examination times of 120 mins or more were well tolerated by all subjects with no observed adverse reactions. Maximum rate of HCO3− production (maxVHCO3−) was calculated from the natural abundance bicarbonate concentration at baseline and at the end of the infusion estimated from the ratio of the areas under the two resonances, HCO3− (161 p.p.m.) and tCr (158 p.p.m.) using a Marquardt fitting algorithm and assuming tCr concentration 11 μmol/g in the brain.
Infusion Protocol
The 99% 13C-enriched [1-13C]acetate solution was prepared under good manufacturing practice by the Cambridge Isotope Laboratories (Andover, MA, USA) and shipped in bulk to a licensed pharmacist in Minnesota, registered and approved by the US Food and Drug Administration to ship sterile, pyrogen-free reagents to California. The reagent was prepared on a named-subject basis and used within 48 h after preparation. This study was performed under the Food and Drug Administration Investigational New Drug Application no. 59690. To establish similar metabolic status, subjects were fasted for 6 h before the study. Intravenous administration (through an arm vein) of 120 to 300 ml of 0.4 mol/L sterile, pyrogen-free solution of [1-13C]acetate 99% enrichment was started (OA) at an infusion rate of 3 mg/kg per min for 60 mins.
Fractional Enrichment of Serum [1-13C]Acetate
Because earlier preclinical and clinical studies have established that 13C acetate consumption proceeds rapidly in the brain and the [1-13C]acetate resonance cannot readily be distinguished from its primary metabolic product, 5-13C glutamate on the basis of near-identical chemical shift, plasma 13C acetate enrichment was used as a surrogate marker of the fractional 13C enrichment (E) of stable isotope availability for glial oxidation. Serial blood samples were collected using heparinized syringe before infusion and at 15 min intervals until the end of the study (at 120 mins). Blood samples were centrifuged and the resulting protein-free plasmas were stored frozen until analysis. Plasma samples were deproteinized using 2:1 volume ratio of 99.9% ethanol to plasma and dried under vacuum. Samples were dissolved in 99.9% D2O before proton NMR measurements using a Varian 300 MHz spectrometer. Fractional enrichment of plasma acetate was determined under fully relaxed conditions (repetition time 18 s) from the ratio of the relative peak areas of the 13C satellites to the total area of the corresponding acetate H2 resonance at 1.9 p.p.m. (Moreno et al, 2001).
Data Processing and Fractional Enrichment Calculation of Cerebral Metabolites
The principal metabolic end point of this study was the quantification of intrinsic cerebral bicarbonate (HCO3−), together with the progressive increase in fractional 13C enrichment (%E) of the HCO3− pool as the result of complete oxidation of the sole 13C-enriched precursor of glial TCA cycle activity, [1-13C]acetate. Because 13C creatine and phosphocreatine do not become enriched from 13C acetate (Cerdan, 2003; Chapa et al, 1995) and resonate distinctly in a portion of the brain spectrum unencumbered by overlying resonances, we used 13C total signal intensity for [Cr + PCr] to quantify the initial brain bicarbonate concentration in nonenriched baseline 13C MR scans from each subject. We assume there are no significant differences in T1 or T2 relaxation times between the two. Fractional enrichment of HCO3− is then defined as the increase in intensity relative to that of [Cr + PCr] which is not enriched from the infused source of [1-13C]acetate. Progressively increasing intensity of 13C HCO3− resonance was recorded throughout and the relative rate of enrichment was expressed as percent over time, slope of initial increase, and final %E bicarbonate. The other anticipated cerebral metabolites of [1-13C]acetate, glutamine C1 and C5, and glutamate C1 and C5, were also readily observed. An observer-independent automatic IDL-based software developed in our laboratory (Shic and Ross, 2003) and SAGE (GE Healthcare, Milwaukee, WI, USA)/IDL (ITT Visual Information Solutions, Boulder, CO, USA), a commercially available software, were used for data processing (NS, KH). Data blocks (6½ min) were first zero filled to 16K data points, applied 7 Hz Lorenzian to Gaussian lineshape transformation, Fourier-transformed, and phase-corrected. Owing to the absence of overlapping resonances, relative contributions of 13C singlet of HCO3− (161 p.p.m.), C1-acetate and C5-Glu (182 p.p.m.), and C5-Gln (178 p.p.m.) were readily determined from peak amplitudes of the observed resonances. In the case of C1-acetate and C5-Glu, no separation of the resonances could be achieved under near-steady-state conditions. Based on earlier studies, the contribution of [1-13C]acetate cerebral flux, %E 13C brain was considered approximately %E acetate of blood.
13C fractional enrichments (
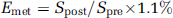
where
Determination of the Glial Acetate Oxidation Rate (V g ac)
For consistency with earlier studies, results are expressed as glial acetate oxidation rate (
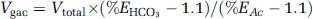
The total bicarbonate production,
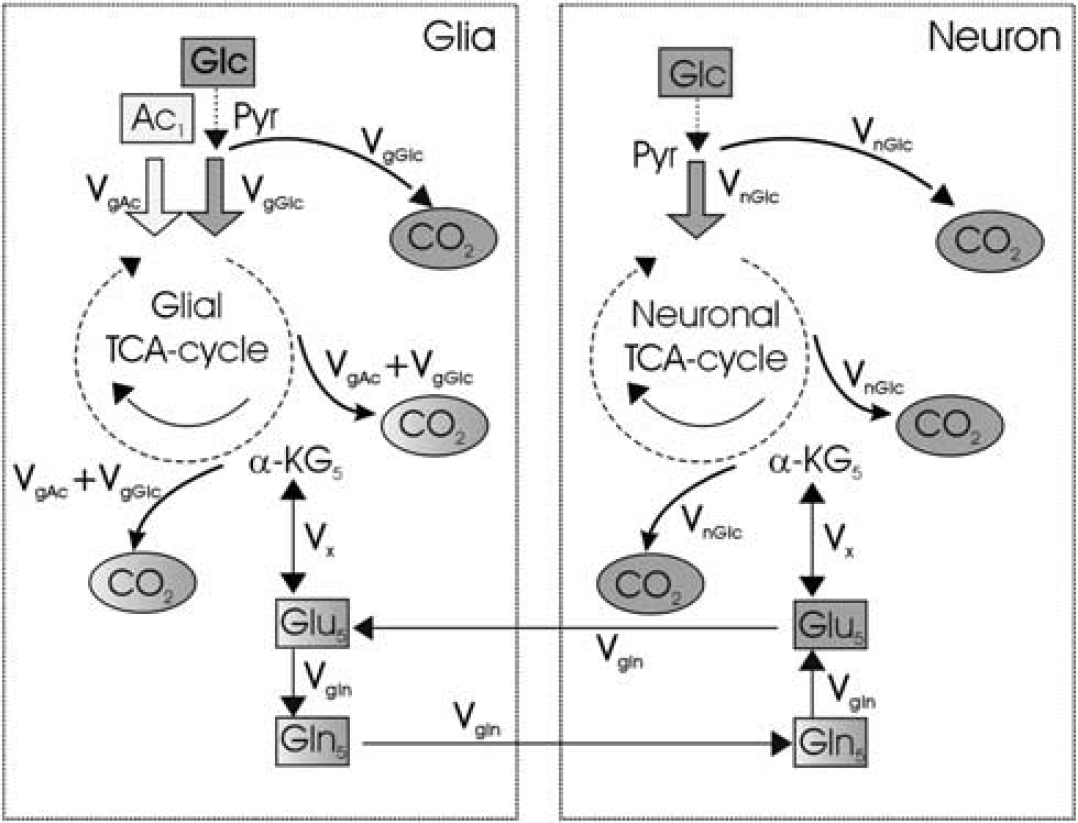
Glial versus neuronal metabolism: pathway of 13C label from [1-13C]acetate to tricarboxylic acid (TCA) cycle in human brain. Selective metabolism of acetate and glucose occurs in glia and neurons, respectively. Only the relevant intermediates are shown. Glia selectively transports acetate which is then metabolized through acetate thiokinase and reactions of the mitochondrial tricarboxylic acid (TCA) cycle forming 13C glutamate and glutamine, enriched in the C1 and C5 positions, before final oxidation and release as 13C bicarbonate (CO2 = HCO3−). Five of the relevant cerebral metabolic products are showed in sequential 13C brain spectra of Figure 3. The alternative metabolic pathways of 13C glucose metabolism, predominantly in neurons, but not contributing to this study in which only the single substrate, [1-13C]acetate is provided. Ac, acetate; Glc, glucose; Glu, glutamate; Gln, glutamine; CO2, carbon dioxide; Pyr, pyruvate; Vg glc, glial glucose oxidation; Vg ac, glial acetate oxidation; α-KG, α-ketoglutarate; Vglu, glutamate synthesis rate; Vgln, glutamine synthesis rate. Glucose is metabolized in neuron and glia whereas acetate is metabolized exclusively in glia.
An additional method of calculating maximum glial TCA cycle rate was applied, determining brain bicarbonate in natural abundance and 13C-enriched spectra with reference to the adjacent Cr/PCr resonance (see above).
Statistical Analysis
Statistical analyses were performed with StatView software (SAS Institute, Cary, NC, USA). For continuous variables, the means and standard deviations were calculated separately for the AMD and the normal subjects. An analysis was performed to show that variance in the two groups was the same using F test, followed by analysis with the two-sample
Results
Determination of Cerebral Bicarbonate Concentration from Natural Abundance 13C MRS
Cerebral [HCO3−] calculated in a contiguous volume of mixed white and gray matter of the human frontal lobe was found to be between 11 and 15 mmol/L (Figure 2), values consistent with literature data and comparable to the those obtained in earlier direct and indirect assays in simian and animal brains (Siesjo, 1964). Results obtained by direct 13C MRS assay in ‘frontal brain’ were also indistinguishable from those obtained in ‘posterior brain’ in the same group of control subjects (Table 1). Results for cerebral bicarbonate concentration in the cohort of recovering methamphetamine-abusing subjects (Table 2) fell within this ‘normal’ range indicating that metabolic pH homeostasis is broadly normal in the recently abstinent methamphetamine-abuse human brain.
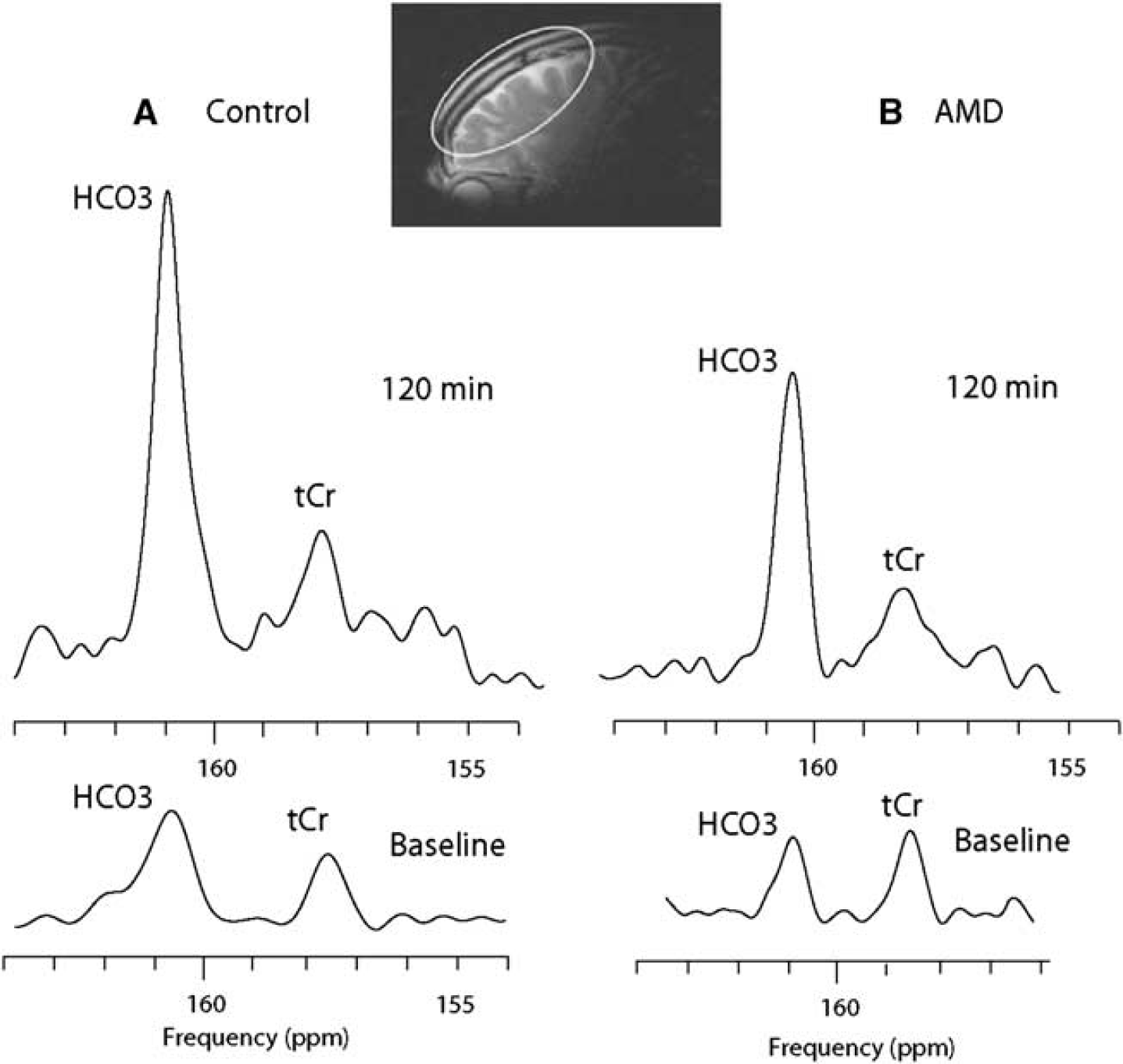
Human 13C MR frontal brain spectra acquired during [1-13C]acetate infusion: carbon-13 magnetic resonance spectroscopy (MRS) at baseline and 120 mins after the infusion (154 to 164 p.p.m.) in a control and an abstinent methamphetamine-dependent (AMD) subject. 13C MRS spectra show HCO3− and total Cr (tCr) chemical shift region (154 to 164 p.p.m.) at baseline and at the end of the infusion study in a normal control subject (
Fractional bicarbonate enrichment and glial metabolic rate for frontal and posterior brain in controls
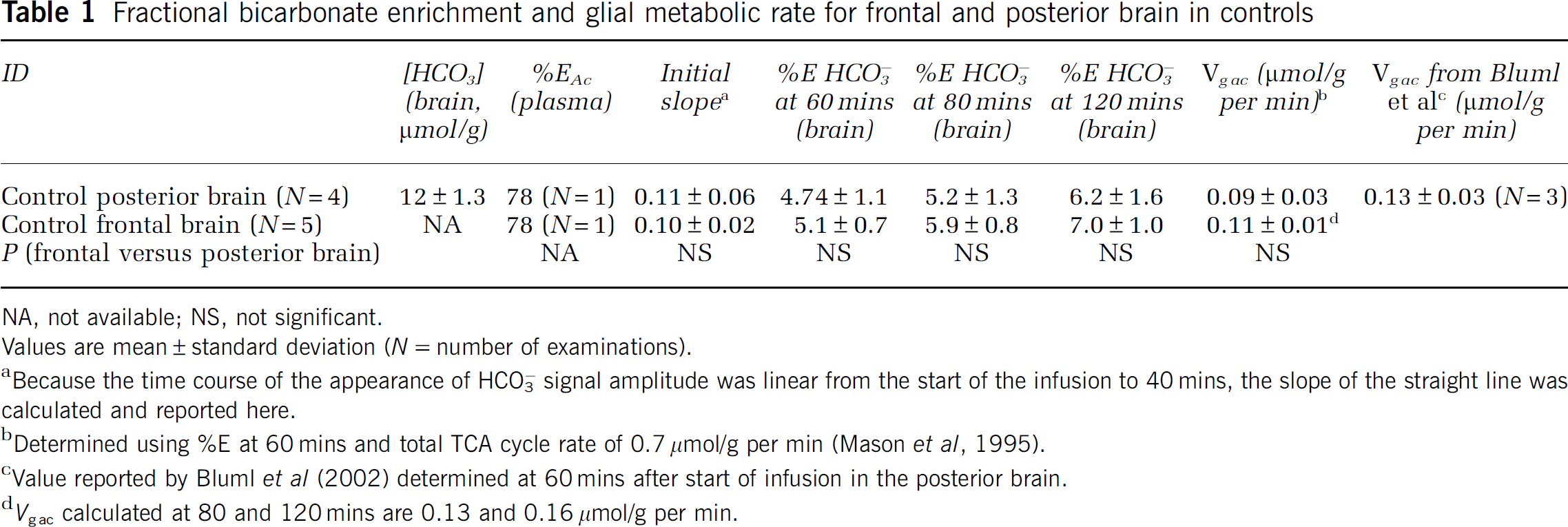
NA, not available; NS, not significant.
Values are mean ± standard deviation (
Because the time course of the appearance of HCO3− signal amplitude was linear from the start of the infusion to 40 mins, the slope of the straight line was calculated and reported here.
Determined using %E at 60 mins and total TCA cycle rate of 0.7 μmol/g per min (Mason et al, 1995).
Value reported by Bluml et al (2002) determined at 60 mins after start of infusion in the posterior brain.
Fractional bicarbonate enrichment and glial metabolic rate for frontal brain in control and AMD subjects
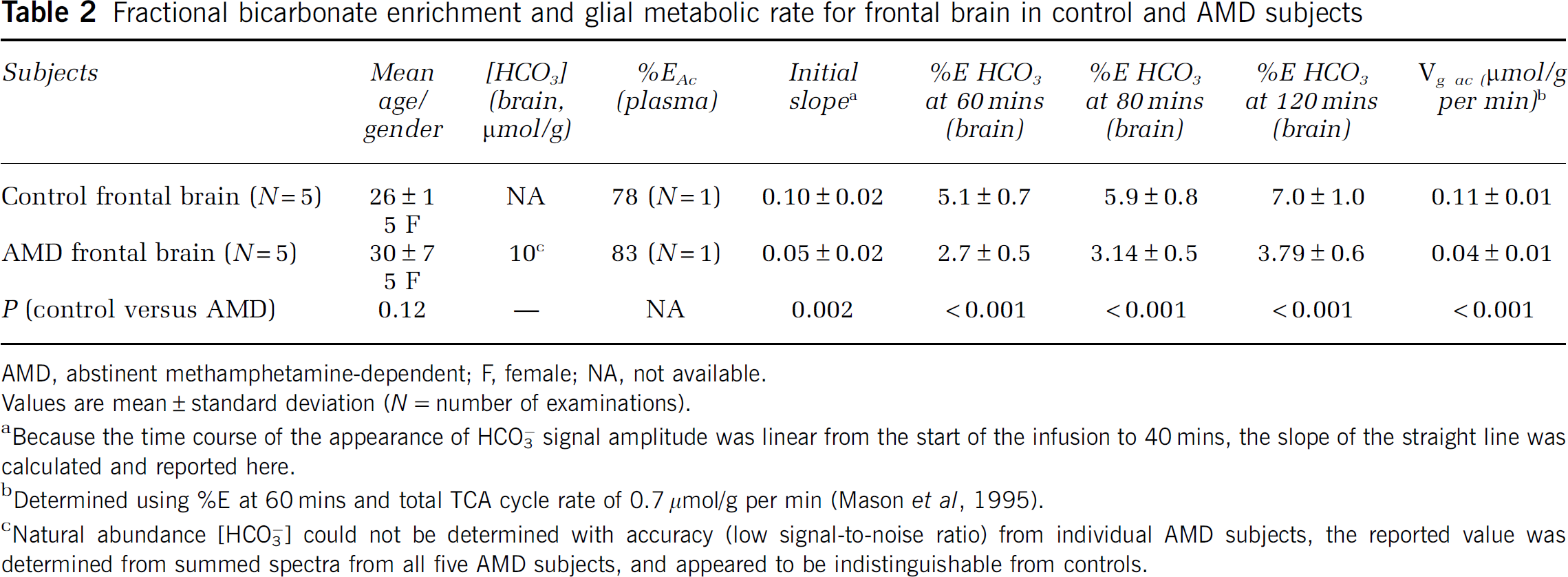
AMD, abstinent methamphetamine-dependent; F, female; NA, not available.
Values are mean ± standard deviation (
Because the time course of the appearance of HCO3− signal amplitude was linear from the start of the infusion to 40 mins, the slope of the straight line was calculated and reported here.
Determined using %E at 60 mins and total TCA cycle rate of 0.7 μmol/g per min (Mason et al, 1995).
Natural abundance [HCO3−] could not be determined with accuracy (low signal-to-noise ratio) from individual AMD subjects, the reported value was determined from summed spectra from all five AMD subjects, and appeared to be indistinguishable from controls.
Determination of the Maximum Rate of Bicarbonate Production (maxV HCO3) in Human Brain
In contrast, using time course data in the two groups of subjects, it was observed that the rate at which [1-13C]acetate was incorporated into the final oxidation product bicarbonate was markedly different between drug-abusing and normal subjects. Figure 2 compares 13C MRS spectra (154 to 164 p.p.m.) from a representative control subject (A) and a methamphetamine abstinent subject (B). From the start (natural abundance) to the end of the 2 h data acquisition, there was almost fivefold increase in 13C incorporation (fully 13C-enriched) in the control, whereas in the AMD subject, the full extent of 13C enrichment was closer to threefold. Natural abundance bicarbonate concentration at baseline and at the end of the infusion estimated was similar in the two subjects. In the control subject, the concentrations of bicarbonate determined in this way were 15 μmol/g at baseline and 49 μmol/g at the end of the 2 h infusion study (Cr does not become enriched) whereas in the AMD subject initial concentration of bicarbonate was similar, 10 μmol/g, at baseline but reached only 25 μmol/g after 2 h. This was less than 50% of the enrichment measured in the normal control. The baseline bicarbonate concentration is within the range of normal values calculated from Henderson–Hasselbach equation, pH = p
Comparison of Fractional Enrichment of Bicarbonate and Glial Acetate Oxidation Rate in the Posterior and Frontal Brain of Normal Subjects
Five control women were scanned using the frontal brain protocol and three of these women and one man were scanned using the posterior brain protocol. Summary of the results is shown in Table 1. Initial slope of H13CO3 appearance in brain was similar for frontal and posterior brain (
Appearance of 13C-Enriched Metabolites of [1-13C]Acetate in the Frontal Brain of Normal and AMD Subjects
Figure 3 is a time course of the appearances of 5-13C glutamate and [1-13C]acetate (A), 5-13C glutamine (B), and HCO3− (C) resonances in a control subject. Similar plots were obtained for AMD (not shown). Rapidly increased to peak amplitudes for the enriched acetate and 5-13C glutamate as well as 5-13C glutamine as were observed at 56 mins and slowly eliminated by approximately 120 mins. Bicarbonate signal amplitude reached a maximum and remained essentially constant after 60 mins. However, the pseudo-steady state for 5-13C glutamate could only be indirectly verified because of the co-resonance of the precursor [1-13C]acetate. Nevertheless, these observations confirmed that all of the 13C label entering the glial TCA cycle is being transferred to bicarbonate and is not sequestered in other metabolic pools.
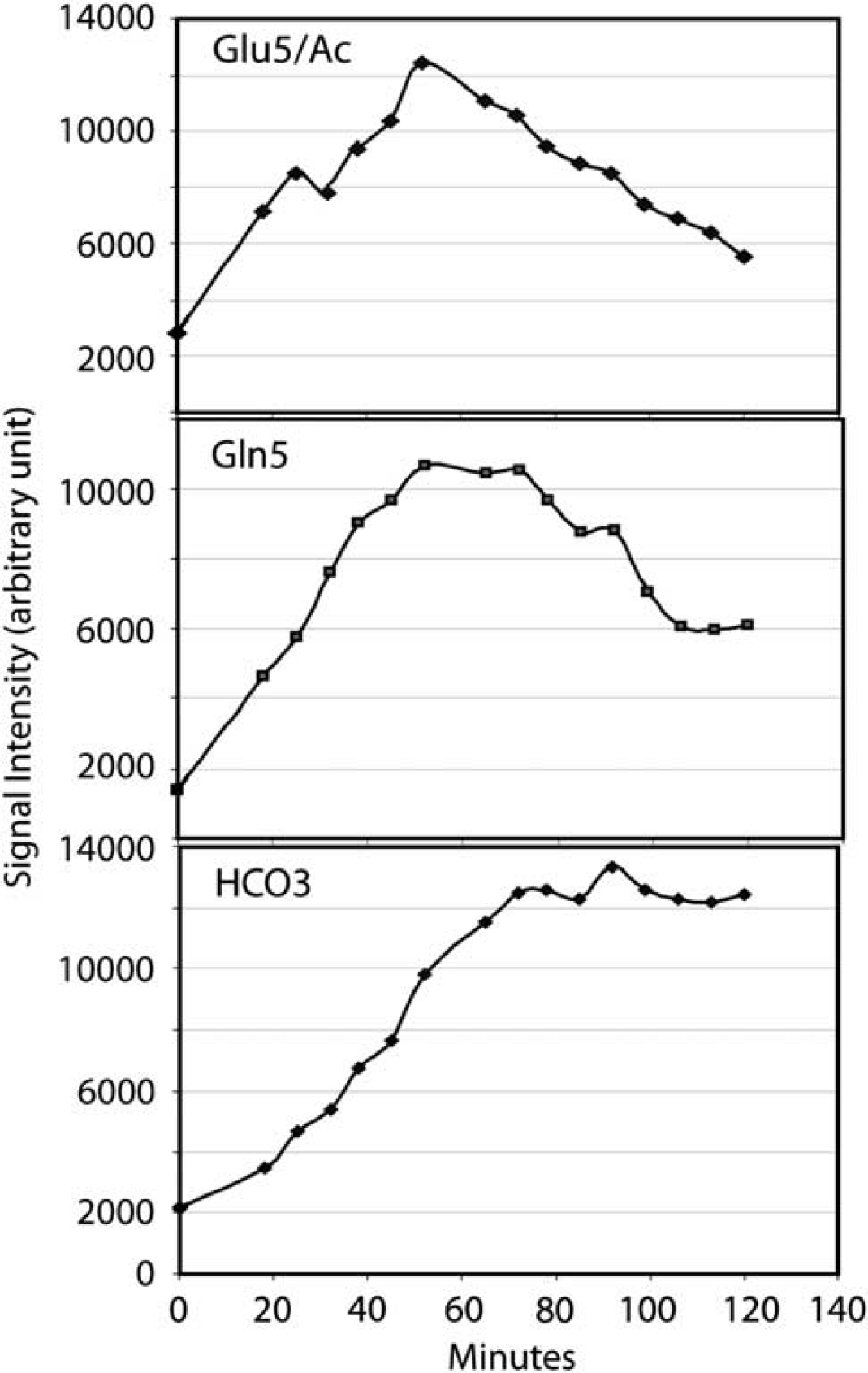
Time courses of glutamate C5 and acetate (
Figure 4 shows stack plots of labeled metabolites comparing results obtained in a normal control (A) with those from a representative AMD subject (C), over time and below, and the summed spectra of the total brain enrichment at the end of the infusion protocol for control (B) and AMD (D). C5 glutamate (and acetate), C5 glutamine, C1 glutamate–glutamine and bicarbonate resonances are clearly visible. The strikingly reduced appearance of 13C label in bicarbonate is evident in Figure 2D, in the AMD subject compared with control in Figure 2B. This result was confirmed in the entire group of AMD. The enrichment of HCO3− estimated from the time course of the fractional enrichment of HCO3− was significantly decreased in AMD (Figure 5) and the rate of acetate oxidation in glial,
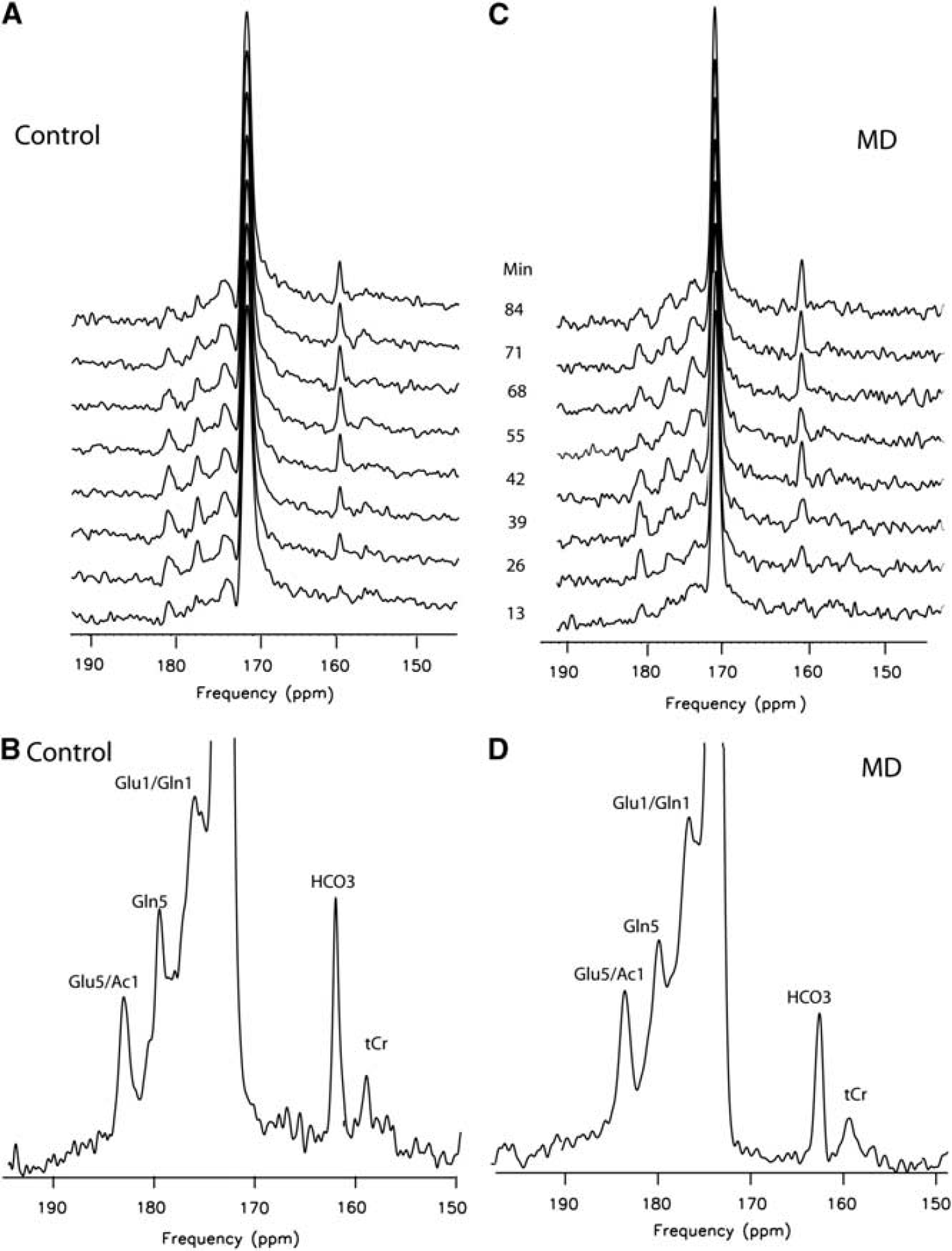
Effect of methamphetamine abuse on cerebral [1-13C]acetate metabolism. Frontal brain 13C metabolites observed during [1-13C]acetate infusion: figure shows carbon-13 MRS (150 to 195 p.p.m.) in a control and an abstinent methamphetamine-dependent subject after enrichment with [1-13C]acetate. Stacked sequential carbon-13 spectra and spectra summed for the entire infusion protocol (120 mins, bottom) are shown for a control (
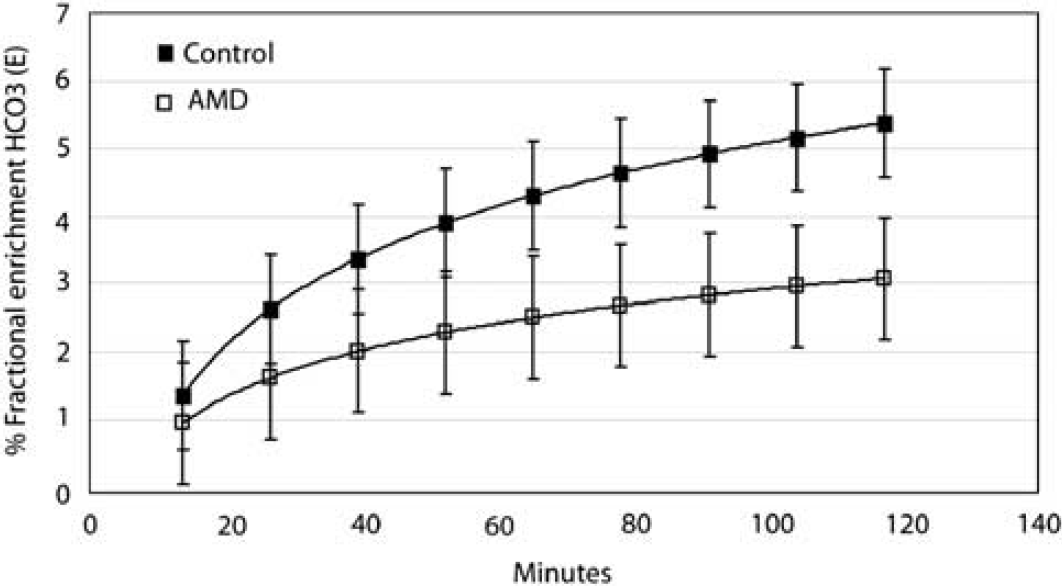
Effect of methamphetamine abuse on cerebral metabolism of 13C acetate. Time courses of appearance of the end product of [1-13C]acetate oxidation, HOC3− are compared. Low-power noise nuclear overhauser effects (NOE) spectra were acquired in 6.5 min blocks for up to 120 mins after enrichment by i.v. acetate (3 mg/kg body weight) from frontal brain in five abstinent methamphetamine-dependent (AMD) subjects and five control subjects. Logarithmic fits of the averaged percent fractional enrichment of bicarbonate (%E) were plotted as a function of time after start of the infusion. %E in AMD (lower trace) is shown to be lower than controls (upper trace). Standard deviations are also shown as cross-bars.
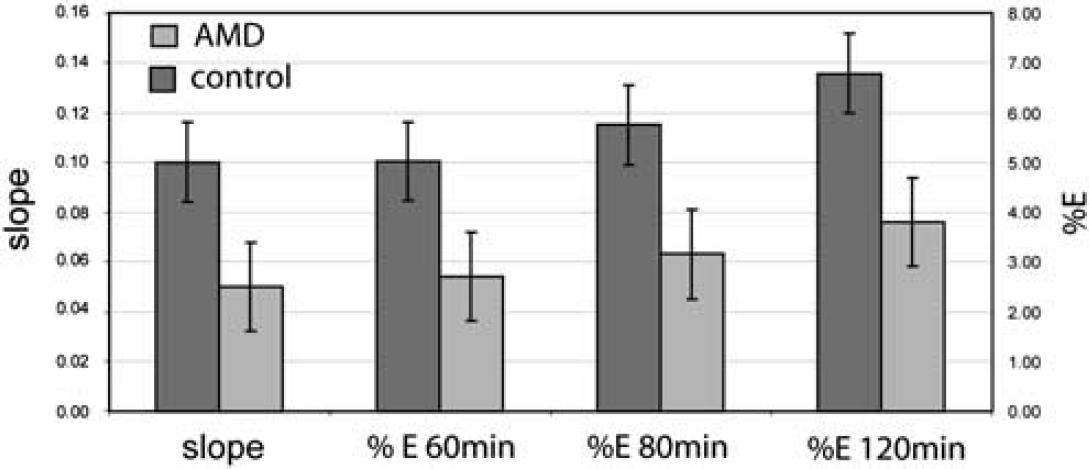
Comparison of methods to express effect of abstinent methamphetamine-dependent (AMD) in 13C HCO3− production from [1-13C]acetate in human frontal brain. Results shown in Figure 5 are quantified. Means and standard deviation of percent fractional bicarbonate enrichment calculated at three different time points (60, 80, and 120 mins) and the initial slope of bicarbonate production from control and AMD groups.
Discussion
In this pilot study we confirm the unique value of low-power 13C MRS applied for the first time to methamphetamine abusers in frontal regions of the human brain. We have discovered a hitherto unsuspected 50% reduction in oxidative rate in the recovering brain after methamphetamine abuse. Although it has been known for many years that acetate, which is a preferred glial respiratory fuel, can be applied to defining glial TCA cycle rate of the normal-functioning human brain
The glial bicarbonate production rate was subsequently calculated from exponential fitting of %E at 60 mins and therefore represents the glial TCA cycle rate. The glial TCA cycle rate determined this way represents approximately 10% of the total TCA cycle rate in the brain of fasted normal subjects. This rate is similar to the previously reported value by us in the parietal brain region using similar [1-13C]acetate protocol (Bluml et al, 2002).
Information regarding abnormalities of glial cells can typically be obtained with proton MRS. There are few MRS studies (nine studies from 2000 to 2008) on methamphetamine abuse that reveal abnormalities in metabolite concentrations in frontal cortex (Ernst et al, 2000), frontal white matter (Ernst et al, 2000; Nordahl et al, 2002), and basal ganglia (Sekine et al, 2002). Proton MRS studies show that methamphetamine-dependent subjects had decreased
Possible limitations to 13C MRS: as with all ‘isotope’ techniques, there is an unknown fraction of cerebral metabolism that will continue to consume (unenriched) endogenous respiratory fuels, in this case predominantly C-12 glucose. This may ‘dilute’ the resulting 13C bicarbonate pool derived from 13C-enriched acetate. One correction has been applied, as suggested by Bluml et al (2002) in expressing the 13C HCO3− as a fraction of the total bicarbonate produced in unit time. Although there is no way with the present technique to ensure that overall cerebral metabolic rate is normal in AMD (additional studies using 13C glucose will clarify the point further), nevertheless, it is difficult to envisage a more than 50% change in intrinsic glucose metabolic rate that would be necessary to explain the present findings. Earlier positron emission tomography studies of AMD argued against any significant increase in neuronal metabolic rate in frontal brain (Kim et al, 2005, 2009; London et al, 2005). Contrary results (small with reference to the present results) were obtained by Wang et al (2004). There are other limitations to our study. First, the study sample is small; though the effect is large and statistically robust. This is partly a consequence of the costs ([1-13C]acetate infusion) and technical difficulty of the present procedures. In the future, both aspects promise to be greatly simplified allowing large-scale replication of the present findings in this and other studies. Another possible limitation of the study is the assumption that fractional enrichment of [1-13C]acetate is similar between the brain and blood (Bluml et al, 2002). Note that the enrichments of Glu1, Gln1, as well as Gln5 were also observed but under our non-steady-state experimental condition, we did not generate kinetic data for metabolic rate calculations used in more complex theoretical models. Alternative, steady-state protocols, involving larger doses of [2-13C] acetate and longer protocols may lend themselves to resolving any remaining kinetic issues. Finally, the study of short-term abstinence may limit the generalizability of results to abuse versus craving and relapse in study of methamphetamine. We propose that this successful demonstration of a hitherto unrecognized but sizeable neurometabolic adaptation is worthy of further consideration and more sophisticated experimental design that may reveal novel mechanisms relevant to human drug abuse and many other conditions currently ascribed to glial dysfunction.
In this preliminary study, we used a novel method of frontal lobe 13C MRS to explore possible defects that may persist in the glia of drug-addicted subjects, using as the example, recently AMD subjects. Additional studies, using the neuronal substrate [2-13C] glucose, will be necessary to determine the cellular specificity and the frontal localization of these neurochemical injuries. The novel capability to dissect two major cell populations at risk in the intact brain and in patients at varying stages of the abuse–addiction–recovery cycle of methamphetamine abuse promises new insights into an intractable clinical problem.
Footnotes
Acknowledgements
We thank Mentors to the K Award (NS), Alan Stacy for advice and Thao Tran and Keiko Kanamori for the technical and scientific insights. OA was supported in part by research funds from Keck School of Medicine, Division of Psychiatry, University of Southern California.
The authors declare no conflict of interest.
