Abstract
The rates of glucose oxidized at glycolysis and pentose–phosphate pathway (PPP) in neurons are controversial. Using [3-3H]-, [1-14C]-, and [6-14C]glucose to estimate fluxes through these pathways in resting, intact rat cortical primary neurons, we found that the rate of glucose oxidized through PPP was, apparently, ∼14% of total glucose metabolized. However, inhibition of PPP rate-limiting step, glucose-6-phosphate (G6P) dehydrogenase, increased approximately twofold the glycolytic rate; and, knockdown of phosphoglucose isomerase increased ∼1.8-fold the PPP rate. Thus, in neurons, a considerable fraction of fructose-6-phosphate returning from the PPP contributes to the G6P pool that re-enters PPP, largely underestimating its flux.
INTRODUCTION
In neurons, pentose–phosphate pathway (PPP) is very active and contributes to the regeneration of glutathione redox status. 1 However, the actual value of the rate of glucose-6-phosphate (G6P) entering the PPP in neurons is yet elusive; moreover, it has recently been reported, on the basis of 13C isotopomer enrichments, 2 levels of PPP activity considerably lower than those previously estimated.1, 3, 4, 5 In view of this apparent controversy and the critical role of PPP in neuronal protection against oxidative stress, here we aimed to accurately determine the fluxes of glucose oxidation through PPP and glycolysis, both under normal and acutely inhibited key regulatory enzymes. We conclude that the estimated values for PPP activity in neurons are largely underestimated because of the fast equilibrium between PPP-derived fructose-6-phosphate (F6P) and G6P.
MATERIALS AND METHODS
Neurons and Astrocytes in Primary Culture
All protocols with animals were approved by the Bioethics Committee of the University of Salamanca following the Spanish (RD1201/2005) and European (609/CEE) directives on research animal protection and experimentation. Cortical neurons in primary culture were prepared from fetal Wistar rats (E16), seeded at 2.5 × 105 cells per cm2 in the bottom surface of 25-ml flasks, previously coated with poly-
Glycolysis and Pentose–Phosphate Pathway Determinations
To determine the glycolytic and the pentose–phosphate pathway fluxes, attached intact cells were washed with PBS and incubated in the presence of either 5
Cell Treatments
Transfections of primary neurons with small interfering RNA (100 nmol/L) against PGI (siPGI; 5′-CCTTACCAGACGTAGTGTT-3′; Dharmacon Research, Lafayette, Colorado, USA) or against luciferase (siControl; 5′- CTGACGCGGAATACTTCGA-3′; Dharmacon) were performed at day 3
Determination of Glucose-6-Phosphate Concentration
Neurons were lysed in 0.06 mol/L NaOH and the resulting extract was deproteinized and neutralized with the same volume of 1% w/v ZnSO4. After centrifugation of precipitated proteins (12,000
Statistical Analysis
Results are expressed as the mean±s.e.m. (standard error of the mean) values for three different culture preparations. Statistical analysis of the results was performed by the Student's
RESULTS AND DISCUSSION
Here, we determined the rate of glucose metabolized through glycolysis as a function of the rate of 3H2O production from [3-3H]glucose, a process that primarily takes place at aldolase and triose-phosphate isomerase, i.e. the glycolytic steps that immediately follow the rate-limiting, 6-phosphofructo-1-kinase-catalyzed reaction.
9
We found that, under resting conditions, the rate of glycolysis in neurons was ∼1.2 nmoL/minute × mg protein (Figure 1A), which is considerably lower than that found in attached astrocytes using identical protocol (3.14±0.27 nmoL/min × mg protein;
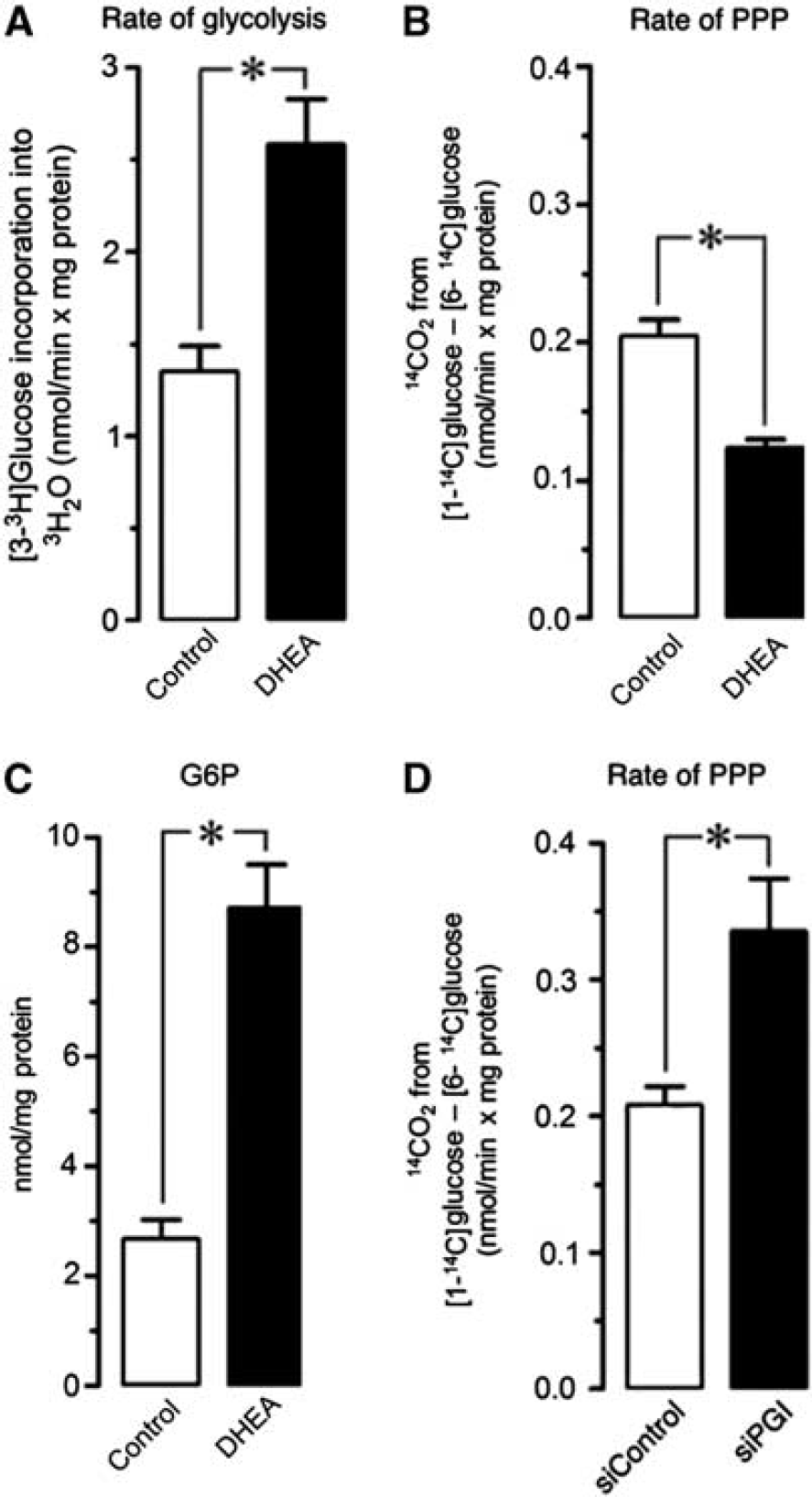
Glucose fluxes through glycolysis and pentose–phosphate pathway (PPP) in neurons. Incubation of rat cortical intact neurons in primary culture with dehydroepiandrosterone (DHEA) (1
Next, we aimed to determine the actual fraction of glucose oxidized through the PPP in intact primary neurons. To do so, cells were incubated in the presence of [1-14C]glucose or [6-14C]glucose, and the initial rates of 14CO2 released were quantified. 14CO2 is released from [1-14C]glucose at 6-phosphogluconate decarboxylation by 6-phosphogluconate dehydrogenase, plus at isocitrate and
6-Phosphofructo-1-kinase catalyzes a rate-limiting reaction–F6P to fructose-1,6-bisphosphate–that represents a glycolysis bottleneck in neurons (Figure 2).1, 12 In contrast, PGI is a near-equilibrium enzyme, very active at converting F6P into G6P in neurons.
13
As [1-14C]glucose yields unlabeled F6P, once converted into G6P, it will no longer contribute to 14CO2 detection. To test this possibility, we inhibited PGI activity using a previously validated RNA interference strategy.
1
The rate of PPP in PGI-knockdown neurons increased by 1.8-fold (Figure 1D), indicating enrichment of [1-14C]G6P-specific radioactivity likely responsible for the enhancement in apparent PPP activity. Given the high PGI activity in neurons,
13
the expected theoretical increase of 1.2 nmoL/min × mg protein in PPP activity by siPGI was not reached (Figure 1D), a fact that is likely due to the still high residual PGI activity after incomplete 70% PGI knockdown. Therefore, the estimated rates of PPP activity reported here may be largely underestimated because of the important contribution of PGI at equilibrating F6P and G6P. Furthermore, the underestimation is independent of the method used to estimate PPP. Thus, it has been reported, by measuring 13C-acetyl-CoA-derived 13C-glutamate and 13C-
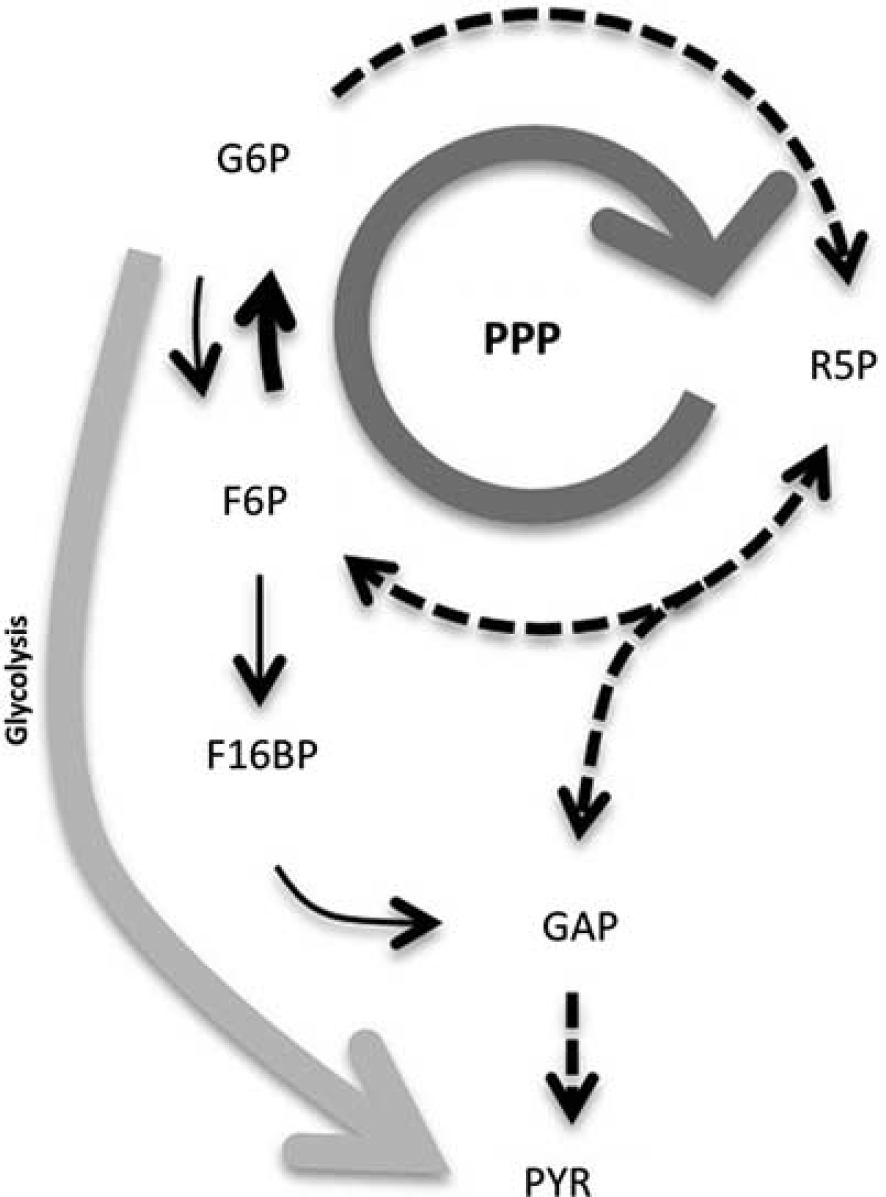
Pentose–phosphate pathway (PPP) is an important contributor to glucose consumption in neurons. Schematic representation of the main conclusion of this work, highlighting the critical contribution of phosphoglucose isomerase (PGI)–which converts PPP-derived F6P into glucose-6-phosphate (G6P)–to fueling PPP at the expense of G6P consumption through glycolysis. 6-Phosphofructo-1-kinase (PFK1), which converts fructose-6-phosphate (F6P) into fructose-1,6-bisphosphate (F16BP), is a ‘bottleneck’ in neurons due to the low levels of fructose-2,6-bisphosphate (a positive effector of PFK1); this facilitates F6P conversion into G6P. Stoichiometry has been omitted for clarity, and dotted lines represent multiple reactions. GAP, glyceraldehyde-3-phosphate; PYR, pyruvate; R5P, ribulose-5-phosphate.
We therefore conclude that an important proportion of glucose entering neurons is oxidized through the PPP (Figure 2). However, the extent of this metabolic route is yet unrecognized because of misinterpretation in isotopic labeling of metabolites, mostly related to the high activity of PGI in neurons. In contrast to astrocytes, neurons are deficient in
Footnotes
The authors declare no conflict of interest.
