Abstract
We tested the hypotheses that administration routes affect the migration and distribution of grafted neural progenitor cells (NPCs) in the ischemic brain and that the ischemic lesion plays a role in mediating the grafting process. Male Wistar rats (
Introduction
Transplantation of neural progenitor cells (NPCs) following experimental stroke has been shown to improve functional recovery (Ishibashi et al, 2004; Jiang et al, 2006; Mochizuki et al, 2008). The likely mechanisms associated with functional improvement have been suggested to result from the ability of the grafted cells to reduce apoptosis (Chen et al, 2003a) and brain atrophy (Chu et al, 2004; Mochizuki et al, 2008; Chen et al, 2008), enhance angiogenesis (Chen et al, 2003b), release neurotrophic factors (Li et al, 2002; Ishibashi et al, 2004; Garcia et al, 2004), differentiate into other cells (Zhao et al, 2002; Chen et al, 2008), and promote endogenous cell proliferation (Chen et al, 2003a) and axonal remodeling (Jiang et al, 2006; Li et al, 2006; Shen et al, 2006). Therapeutic benefits gained from cell-based therapy depend on migration and localization of grafted cells within the target tissue (Modo et al, 2002; Savitz et al, 2004; Mochizuki et al, 2008), which can be noninvasively and dynamically detected by magnetic resonance imaging (MRI) (Heohn et al, 2002; Zhang et al, 2004; Magnitsky et al, 2005; Arbab et al, 2006; Walczak et al, 2008). Being able to track the magnetically labeled cells
The cell delivery route chosen for the therapy may influence the migration and final destination of the transplanted cells. The cells can be precisely injected into the ischemic brain adjacent to the lesion area without loss (Chen et al, 2000; Zhao et al, 2002; Chen et al, 2008) or into the vascular system, for example, vein (Chen et al, 2001, 2003a,
Materials and methods
All experimental procedures were approved and conducted in accordance with the institutional guidelines for care and use of laboratory animals.
Neurosphere Culture and Cell Labeling
NPCs were dissociated from the subventricular zone of normal male Wistar rats (Jackson Laboratory, Bar Harbor, ME, USA) aged 3 to 4 months, as previously reported (Luskin et al, 1997). These subventricular zone cells have been shown to have characteristics of neural progenitor cells (Liu et al, 2007). The cells were suspended at a density of 10,000 to 20,000 cells per milliliter in serum-free growth medium containing Dulbecco's modified Eagle's-F-12 medium (DMEM; Invitrogen Corporation, Carlsbad, CA, USA), 20 ng/ml of epidermal growth factor (R&D Systems, Minneapolis, MN, USA), and 20 ng/ml of basic fibroblast growth factor (R&D Systems). The generated neurospheres (primary sphere) were passaged by mechanical dissociation and reseeded as single cells at a density of 20 cells per microliter in basic fibroblast growth factor- and epidermal growth factor-containing media. Passage-1 cells were used in this study.
Cells were labeled
The viability of the SPIO-labeled NPCs before transplantation was assessed by trypan blue exclusion assay (Amsalem et al, 2007) and the tests showed that more than 95% of the cells remained viable after labeling with SPIO.
Animal Model and Cell Transplantation Procedures
Male Wistar rats (300 to 350 g,
Rats were anesthetized with 3.5% halothane for induction and maintained by spontaneously respired 1.0% halothane in a 2:1 N2O:O2 mixture using a face mask. The rectal temperature was kept at 37°C ± 1°C throughout the surgical procedure using a feedback-regulated water-heating system. MCAo was induced by advancing a 4-0 surgical nylon suture, with its tip rounded by heating near a flame, from the external carotid artery into the lumen of internal carotid artery (ICA) to block the origin of right middle cerebral artery (MCA). After 2 h of MCAo, reperfusion was achieved by withdrawing the endovascular suture to the stump of the external carotid artery. The procedure provides a focal infarct in the unilateral striatum and cortex.
Rats subjected to transient MCAo were randomized to one of three treatment groups targeted for cell transplantation intra-arterially (carotid artery,
MR Imaging and Data Processing
MR imaging was performed using a 7 T, 20-cm-bore, superconducting magnet (Magnex Scientific, Abingdon, UK) interfaced to a Bruker console (Bruker, Boston, MA, USA). The animal was placed on a non-magnetic holder equipped with a nose cone for administration of anesthetic gases and stereotaxic ear bars to minimize movement of the head. A tri-pilot scan of imaging sequence was used for reproducible positioning of the animal in the magnet at each MRI session. During the imaging procedure, anesthesia was maintained with 1.0% halothane in 69% N2O and 30% O2, and rectal temperature was kept at 37°C ± 1.0°C using a feedback-controlled water bath.
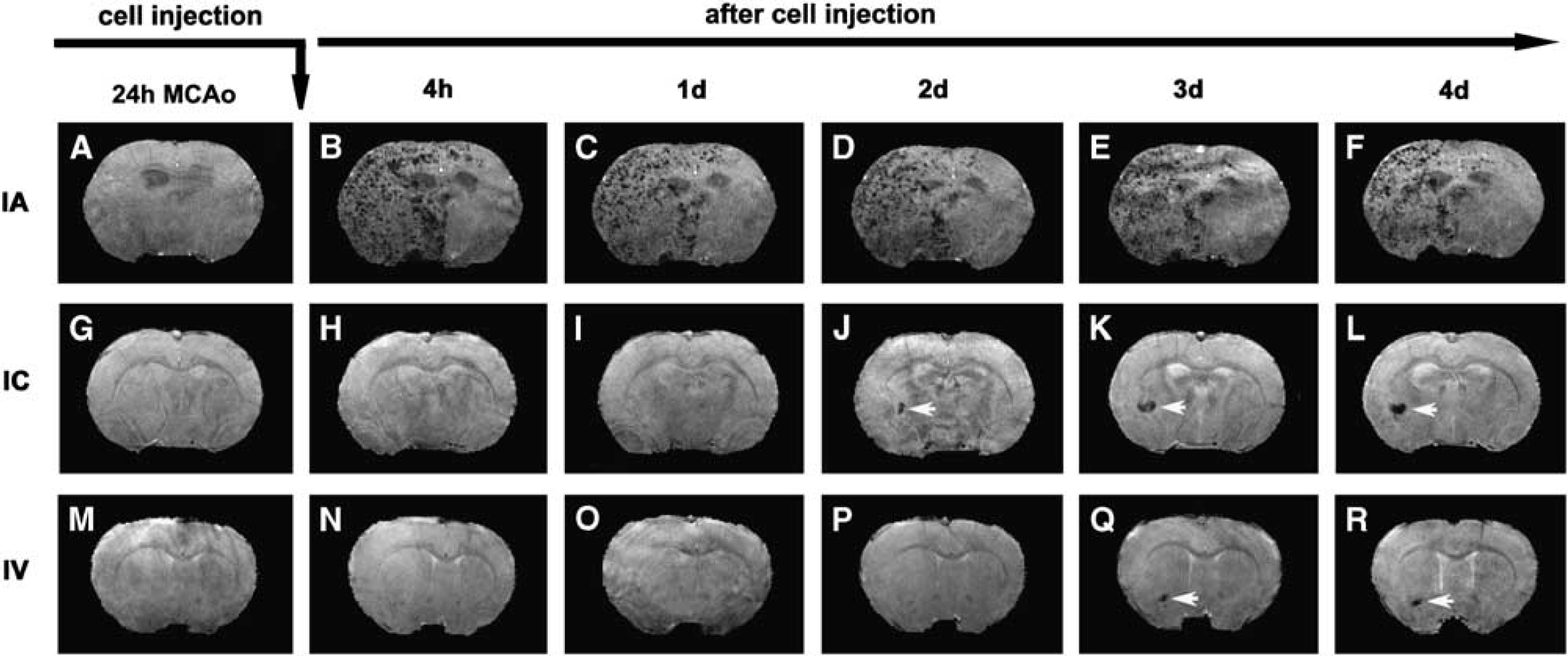
Consecutive 3D images of representative animals showing the dynamic evolution of migration, distribution, and amount of SPIO-labeled NPCs in the host brain after IA (top row), IC (middle row), and IV (bottom row) administration after transient MCAo. The ischemic lesions were located on the right side of the brain (the left side of the images). While the cells could be detected as soon as 4 h after IA injection (
In vivo MR Imaging
T2WIs were acquired using standard two-dimensional Fourier-transform, multi-slice (13 slices, 1 mm thick), multi-echo (six echoes) MRI. Six sets of images (13 slices per set) were obtained using echo times (TEs) of 15, 30, 45, 60, 75, and 90 ms, and a repetition time (TR) of 8 secs. Images were produced using a 32 × 32-mm2 field of view and a 128 × 64 image matrix. The total sequence time was approximately 9 min.
3D gradient echo images were acquired with TR of 40 ms, TE of 10 ms, flip angle of 30 degrees, and a 32 × 32 × 24-mm3 field of view. The 256 × 192 × 64 image matrix was interpolated to 256 × 256 × 64 for analysis.
Ex Vivo MR Imaging
Perfusion-fixed rat brains were removed and immersed in Fomblin (Thorofare, NJ, USA). High-resolution
MRI Data Processing
The ischemic lesion was identified on a T2 map (Figures 2A–2C). The lesion area on each slice of T2 map was specified by those pixels with a T2 value higher than the mean plus twice the standard deviation (mean + 2s.d.) provided by the normal tissue on the contralateral (nonischemic) side (Li et al, 2005). Lesion volume was then calculated on the basis of lesion areas on individual slices and slice thickness. Data are presented as percentage of brain volume. Lesion reduction after cell administration, the difference in lesion volume between the day of cell transplantation and 4 days after cell transplantation, was also calculated and presented as percentage of lesion volume on the day of cell injection.
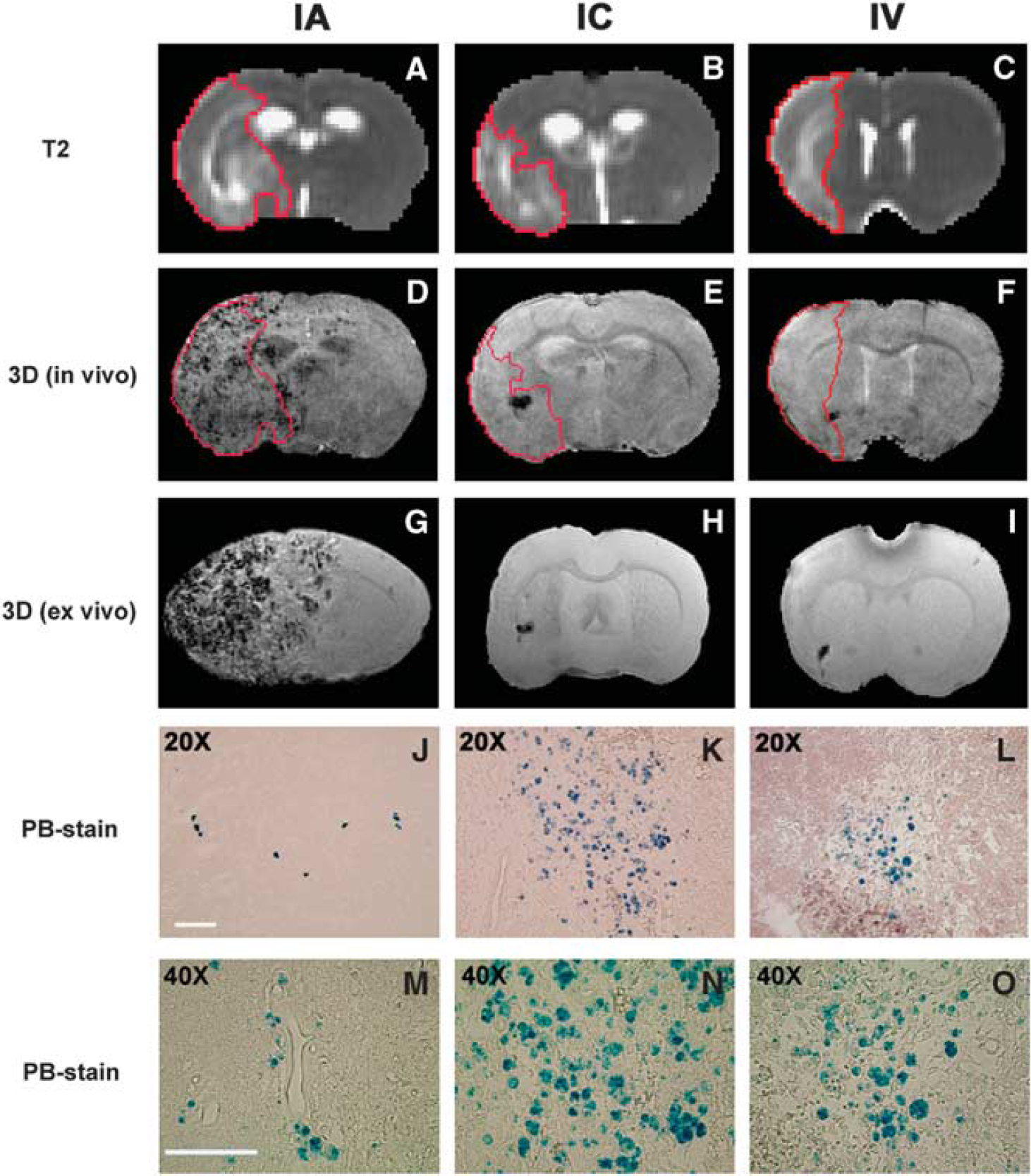
The distribution pattern of grafted NPCs in the host brain and the relationship between ischemic lesion and cell localization 4 days after IA (left column), IC (middle column), and IV (right column) administration. Lesion areas were identified on T2 maps
SPIO-labeled NPCs in the host brain resulted in dramatic signal reduction on 3D images, producing a strong contrast against the non-cell tissue background as shown in Figures 1 and 2. These hypo-intense areas on each slice of 3D images were specified by those pixels with signal intensity lower than the mean minus twice the standard deviation (mean–2s.d.) measured from non-cell tissue area on the contralateral side. Similarly, the volume of signal reduction for each animal was calculated from the hypo-intense areas identified on individual slices and slice thickness of 3D images. More labeled NPCs would induce a larger volume of signal loss on 3D images.
Tissue Preparation and Histology
Immediately after the final
To detect labeled NPCs in the host brain, tissue sections were stained for iron using Prussian blue (PB) reaction (Jiang et al, 2006). The coronal tissue sections were deparaffinized, rinsed in deionized water, and incubated for 30 min with 2% potassium ferrocyanide (Perls' reagent) in 6% HCl, and then washed well with distilled water and counterstained with nuclear fast red.
Microscopic observation and analysis were performed using a 3-CCD color video camera (Sony DXC-970MD; Ampronix Inc., Irvine, CA, USA) interfaced with a MicroComputer Imaging Device (MCID) system (Imaging Research Inc., St Catherines, ON, Canada).
Statistical Analysis
χ2-test/exact test was employed to compare the mortality due to cell delivery among treatment groups. The Kaplan–Meier approach was used to test the group difference in time point for grafted NPCs being detected in the host brain, using the log-rank test with the estimated median time (the time at which grafted cells were detected in 50% of subjects studied). One-way or two-way analysis of variance was used to compare the volume of SPIO-induced signal reduction, the lesion volume, and lesion reduction between treatment groups and between groups with more and fewer cells. Analysis began with testing the overall route effect, followed by pairwise comparisons if the effect was significant at 0.05 level. Correlative analysis was performed between lesion volume on the day of cell transplantation and volume of SPIO-induced signal reduction on 3D MRI 4 days after transplantation. The measurement results are summarized as mean ± s.e. Statistical significance was inferred for P ≤ 0.05.
Results
MRI Detection of Transplanted NPCs
Areas of signal intensity decrease on
Success of cell engraftment in target brain was examined by histology. Histological evaluation of PB-stained tissue sections showed that SPIO-labeled NPCs transplanted through IA, IC, and IV delivery routes following stroke arrived at the ischemia-injured brain. The cellular distribution in the host brain was represented by PB-positive material inside the engrafted NPCs (blue spots; Figures 2J–2L), indicative of the presence of iron that was used for labeling the cells. The PB-positive cells were found within the brain parenchyma 4 days after transplantation for three delivery routes, as shown by micrographs at higher magnification (Figures 2M–2O).
Comparison between 3D images and the corresponding PB-stained tissue sections showed that the hypo-intense site seen on 3D MRI coincided with the histological location of the iron-positive area for all animals (Figure 2). This spatial colocalization between cell identification on tissue slices and appearance of signal damping on MRI images indicates that SPIO-labeled NPCs can be visualized
Migration and Distribution of Transplanted NPCs
With all three delivery routes, both histological evaluation and MR imaging showed that most of the transplanted NPCs were located on the ischemic side of the brain, with very few on the contralateral side. On the ischemic side, cells were located in the lesion core and the border region between intact normal tissue and the area of infarction. Using T2 map to identify the ischemic lesion (Figures 2A–2C), the cells were detected within and surrounding the lesion area (Figures 2D–2F). As shown in Figure 2, the cell administration route remarkably affected the final pattern of cellular distribution in the host brain, even though the treated animals had a similar degree of ischemic injury (such as lesion volume) at the time of cell transplantation. Transplanted NPCs scattered discretely (as small clusters of cells; Figure 2J) throughout the entire lesion area on the ipsilateral side of the brain after IA administration (Figure 2D), whereas the typical cellular distribution pattern after IC and IV administration was characterized by the cell clusters with numerous NPCs (Figures 2K and 2L) congregating somewhere nearby the lesion boundary or adjacent to healthier tissue (Figures 2E and 2F). PB-stained tissue sections showed that after IC and IV administration, most NPCs entering the brain gathered in these cluster areas (with very few in the other site), which were readily detected by 3D MRI (Figures 1J–1L; Figures 1Q and 1R). Notably, while labeled NPCs could be detected as soon as 4 h after IA injection (Figure 1B), they were not seen until 1 to 2 days after IC (Figure 1J, white arrow), or 2 to 3 days after IV (Figure 1G, white arrow) injection. Statistical analysis showed that the time points for NPCs being detected in the brain were significantly different among the three treatment groups (overall route effect,
Lesion Volume and NPCs in the Host Brain
As described above, NPCs migrated into the ischemic brain after IA, IC, and IV administration. The amount of cells present in the host brain, as measured by hypo-intense pixels on 3D MRI, varied from group to group and from animal to animal even through the same administration route. However, we found two distinct clusters of pixels with hypo-intensity in all treatment groups, low and high, with no intermediate value. For the IA group, the total number of pixels with hypo-intensity on 3D images was either < 8 (
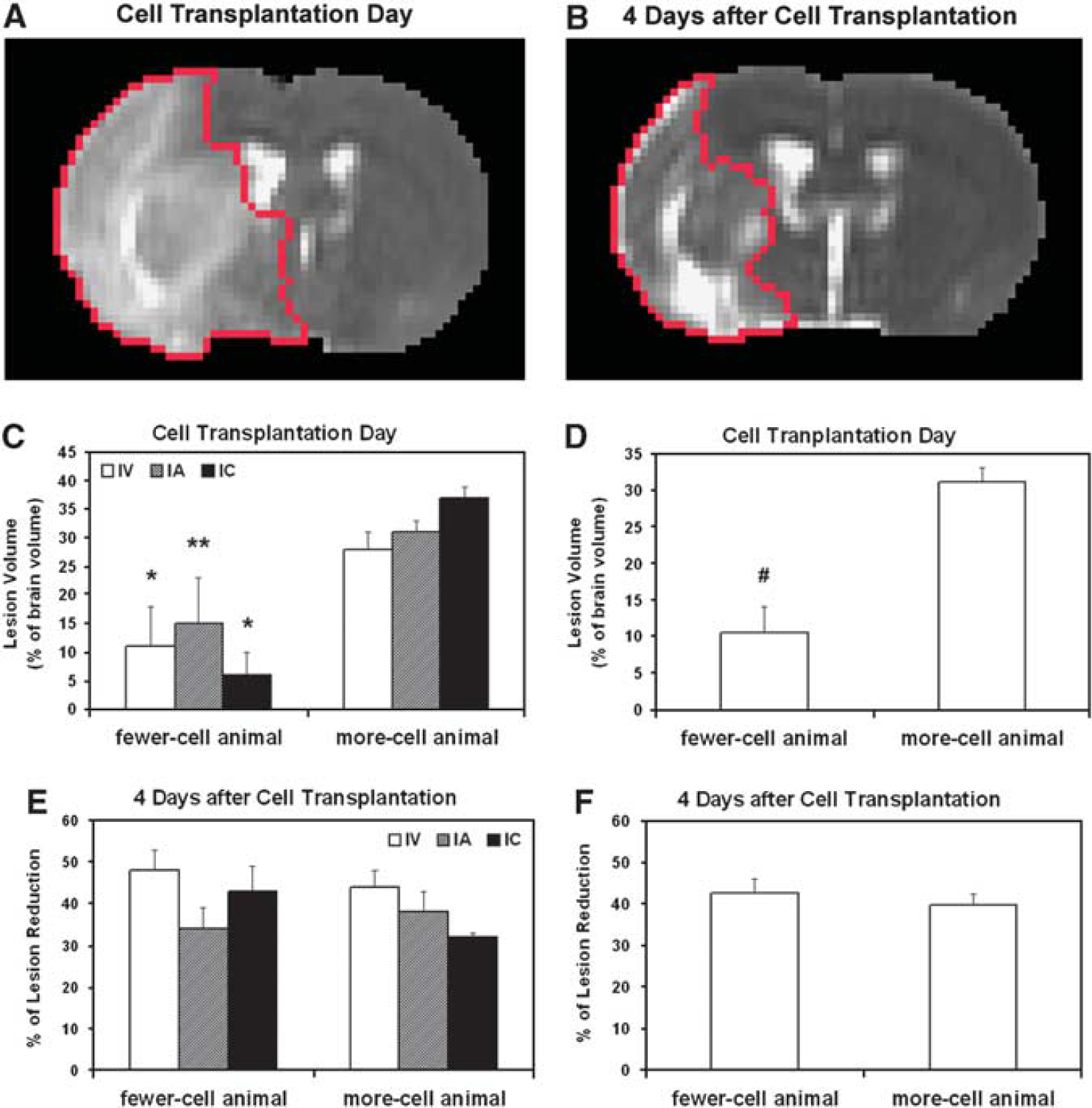
Lesion reduction after cell transplantation presented by the same slices of T2 maps at two time points (red tracks in panels
Correlation Analysis
The correlation between lesion volume on the day of cell transplantation and amount of cells detected in the brain (4 days after transplantation) was low for each treatment group (
Mortality during Transplantation of NPCs
Rat deaths, which occurred during or immediately after cell delivery, were considered as transplantation-associated death, and those rats were counted for each group. IA delivery resulted in a higher mortality than IC and IV delivery (
In addition to death resulting from cell injection, some rats died during the following night or during MRI scans after cell transplantation in each treatment group (
Discussion
The effects of delivery routes on the process of grafting NPCs into ischemic rat brain have been noninvasively and dynamically investigated using MRI. By monitoring engraftment
Cell-induced therapeutic benefits following stroke are evoked using various cellular delivery routes (Chopp and Li, 2002; Parr et al, 2007; Bacigaluppi et al, 2008). Among them, IV injection, a minimally invasive and clinically relevant technique, offers the most comfortable strategy for both subjects and operators. Some more invasive administration routes, such as IA and IC injection, are widely used in experimental studies and may be applied in future clinical practice (Li et al, 2001, Shen et al, 2006; Walczak et al, 2008; Zhang et al, 2004). We, therefore, assessed cell engraftment through these three delivery pathways.
Our data show that with all three delivery routes, transplanted NPCs can migrate to the ischemic brain and preferentially localize to the hemisphere ipsilateral to the ischemic lesion. While the cells appear in the lesion core area, numerous cells and cell clusters are usually detected nearby the lesion boundary, which provides a more supportive environment than the lesion core area for survival of transplanted NPCs (Savitz et al, 2004). In agreement with the previous finding that transplanted cells are attracted by and interact with tissue regions undergoing degeneration and reorganization (Modo et al, 2002), the injected cells are also found in the hemisphere contralateral to the ischemic lesion, although the amount is very small as compared with that in the ipsilateral hemisphere.
Our observations showed that delivery routes dramatically affected the migration and distribution of grafted cells. Despite an easy and safe pathway, IV administration led to delayed engraftment and lower numbers of grafted cells in the target brain than that by IC and IA administration (Figure 1), due to longdistance migration and a large proportion of cells trapped in the filtering organs (Kraitchman et al, 2005; Hauger et al, 2006; Parr et al, 2007). In contrast, IA administration could bring a large number of cells to the brain soon after transplantation (Figure 1) by bypassing the initial uptake by the systemic organs (Walczak et al, 2008). Since fewer cells were injected through the IC (1 × 105) than the IA and IV (1 × 106) routes in this study, this raises the possibility that the difference in the temporal profile of migration is due to the difference in the cell numbers injected. However, our data showed that even when more cells were infused through the tail vein, IV administration did not result in larger number and earlier arrival of grafted cells in the ischemic brain than IC administration (Figure 1). Therefore, delivery route is the determining factor for migration, not the cell numbers, at least in the range employed. It should be pointed out here that sensitivity of MRI to detect magnetically labeled cells depends on the MRI system and experimental setup (Magnitsky et al, 2005). Thus, very small cell clusters entering the ischemic brain may not be detected by MRI before a certain amount of these labeled cells has been accumulated, which could affect the estimate of the timing for grafted cells present in the brain. Our data suggest that under the current experimental conditions, initial accumulation of grafted cells within the target brain after IA administration was more rapid than IC and IV administration (overall route effect,
As shown previously, particular locations in the brain selected for cell injection after stroke are subsequently associated with different functional benefits (Modo et al, 2002; Mochizuki et al, 2008), suggesting that different sites of transplantation may have their own value, and the type of functional recovery is affected by the localization of grafted cells within the brain (Modo et al, 2002; Roitberg, 2004). In addition, functional outcome after cell-based therapy is dose-dependent and a certain amount of grafted cells is necessary to significantly attenuate the functional deficits induced by ischemic injury (Chen et al, 2001). After systemic administration, the majority of transplanted cells home into the hemisphere ipsilateral to the ischemic lesion and evoke antiapoptotic and restorative effects, such as angiogenesis and neurogenesis (Chopp and Li, 2002). However, the exact localization of exogenous cells within the target brain after systemic transplantation cannot be controlled even though homing tendency to the ischemic lesion is expected, unless an invasive implantation, such as intracerebral injection (Chen et al, 2000; Zhao et al, 2002; Chen et al, 2008), is used. Our data indicate that IA delivery provides a wider distribution and larger number of grafted cells in the target brain than that by IC and IV delivery (Figure 1). These characteristics with IA administration may contribute in part to the observation that similar functional improvement after stroke, as measured by adhesive-removal test and neurological severity scores, can be achieved by IA injection (Li et al, 2001) with a lower dose of bone marrow stromal cells (2 × 106) than IV injection (Li et al, 2002) (3 × 106). Although functional recovery after cell transplantation has been shown in the previous studies using various cellular delivery routes and treatment protocols (Parr et al, 2007; Bacigaluppi et al, 2008), outcomes associated with the distribution and numbers of grafted cells in the target brain require further investigation.
The precise mechanism by which the transplanted cells are guided to the site of damaged tissue remains unknown, but the brain expresses chemotactic signals in response to ischemic injury that attract the cells and direct their migration to the damaged areas (Chopp and Li, 2002; Magnitsky et al, 2005; Shyu et al, 2006; Bacigaluppi et al, 2008). At the molecular level, matrix metalloproteinases (Wang et al, 2006; Lee et al, 2006) and the SDF-1/CXCR4 system (Wang et al, 2008) are involved in the directed cell migration. Our MRI observations show that whichever route of cell administration is chosen (IA, IC, or IV), NPCs have the capacity to migrate to the brain lesion area, once again providing evidence to support the concept that chemoattractive gradient released in the site of brain lesion leads to selective and specific homing of transplanted cells in the brain area suffering ischemic injury. Therefore, the status of the ischemic brain may play a critical pathobiological role to mediate the cell grafting process. Our measurements showed that a smaller lesion on the day of cell transplantation was significantly associated with fewer cells detected in the brain (Figure 3D,
We are aware of limitations in our study. First, sensitivity of MRI to detect magnetically labeled cells is affected by a number of factors, including the hardware and software of MRI setup (Magnitsky et al, 2005). Therefore, our imaging measurements may not be sensitive enough to detect single cells or tiny cell clusters, which migrate to the host brain. Second, the degree of signal reduction in a local area relies on the concentration of SPIO or the size of the cell cluster. We used the same threshold to identify the volume of SPIO-induce signal reduction, which could underestimate the amount of grafted cells in the brain, especially for the IA group, since areas with signal alteration result from many small cell clusters, which might be excluded. Finally, hypo-intense areas on 3D MRI may include phagocytized PB-positive cells even though this effect is small on our study due to a short period after cell transplantation (4 days). However, our goal was to show the differences in migration and distribution following different cell delivery routes under the same experimental condition rather than to determine the minimum cell detection or quantify the grafted cells. The major characteristics for each route of cell engraftment seen on tissue slices were reflected on 3D MRI. As shown in Figure 2, more details depicted by
In summary, IA administration after transient MCAo shows significantly increased migration, a far more diffuse distribution pattern, and a larger number of transplanted NPCs in the target brain than IC or IV administration. However, high mortality with IA delivery poses a serious concern for using this route of administration. Further studies are clearly warranted before this strategy can be recommended for patients. Animals with smaller lesions (less than 10% of brain volume) on T2 map at the time of transplantation (24 h after MCAo) have fewer grafted cells into the parenchyma.
Footnotes
The authors declare no conflict of interest.
