Abstract
Recombinant human erythropoietin (rhEPO) induces neurogenesis and angiogenesis. Using a coculture system of mouse brain endothelial cells (MBECs) and neural progenitor cells derived from the subventricular zone of adult mouse, we investigated the hypothesis that neural progenitor cells treated with rhEPO promote angiogenesis. Treatment of neural progenitor cells with rhEPO significantly increased their expression and secretion of vascular endothelial growth factor (VEGF) and activated phosphatidylinositol 3-kinase/Akt (PI3K/Akt) and extracellular signal-regulated kinase (ERK1/2). Selective inhibition of the Akt and ERK1/2 signaling pathways significantly attenuated the rhEPO-induced VEGF expression in neural progenitor cells. The supernatant harvested from neural progenitor cells treated with rhEPO significantly increased the capillary-like tube formation of MBECs. SU1498, a specific VEGF type-2 receptor (VEGFR2) antagonist, abolished the supernatant-enhanced angiogenesis. In addition, coculture of MBECs with neural progenitor cells treated with rhEPO substantially increased VEGFR2 mRNA and protein levels in MBECs. These
Introduction
In the adult rodent brain, neural progenitor cells are localized adjacent to endothelial cells in the subventricular zone (SVZ) and the dentate gyrus (Palmer et al, 2000; Gotts and Chesselet, 2005). Cerebral ischemia induces neurogenesis and angiogenesis (Jin et al, 2001; Zhang et al, 2001; Arvidsson et al, 2002; Parent et al, 2002; Wang et al, 2004). Angiogenesis is coupled with neurogenesis (Palmer et al, 2000; Louissaint et al, 2002; Taguchi et al, 2004). Suppressing angiogenesis attenuates neuroblast migration toward the ischemic cortex (Ohab et al, 2006).
Erythropoietin (EPO), a hematopoietic cytokine, regulates neurogenesis and angiogenesis (Shingo et al, 2001; Wang et al, 2004). Treatment of stroke with recombinant human EPO (rhEPO) enhances neurogenesis and angiogenesis and improves neurologic outcome (Wang et al, 2004). Erythropoietin-enhanced angiogenesis promotes migration of new-born neurons toward the ischemic boundary region through MMP2 and MMP9 secreted by cerebral endothelial cells (Wang et al, 2006a, b ), suggesting that EPO-induced angiogenesis is coupled with neurogenesis in the ischemic brain.
Adult SVZ neural progenitor cells express many genes involved in angiogenesis (Liu et al, 2007). Ischemic stroke upregulates angiogenic gene expression in SVZ neural progenitor cells (Liu et al, 2007). Using a coculture system, we recently showed that neural progenitor cells derived from the ischemic SVZ promote
Materials and methods
All experimental procedures were approved by the Institutional Animal Care and Use Committee of Henry Ford Hospital. Wild-type male mice (C57BL6/J, 6 to 8 weeks) were purchased from The Jackson Laboratory (Bar Harbor, ME, USA). EPO receptor null male mice (ΔEPOR mice, C57BL6 background, 6 to 8 weeks) were provided by Dr Constance Tom Noguchi at NIDDK, NIH (Chen et al, 2007).
Neurosphere Culture
Subventricular zone neural progenitor cells were dissociated from wild-type male mice (
Mouse Brain Endothelial Cell Culture
Mouse brain endothelial cells (MBECs; ATCC, Manassas, VA, USA) were incubated in Dulbecco's modified Eagle's medium and 10% fetal bovine serum (GIBCO, Grand Island, NY, USA) and maintained at 37°C in 5% CO2/95% ambient mixed air. The culture media were changed every 48 h. Passage 10 to 12 MBECs were used in the experiments.
Mouse VEGFR2 siRNA and Transfection
Mouse VEGFR2 short interfering RNA (siRNA) was purchased from Dharmacon Inc. (Chicago, IL, USA). Mouse brain endothelial cells were transfected using FuGENE 6 Transfection Reagent (Roche Applied Science, Indianapolis, IN, USA) as per the manufacturer's instructions. The total amount of siRNA per transfection was kept constant to 1 μ
Preparation of Conditioned Media
Neural progenitor cells (1 times 106) were plated on six-well tissue culture plates containing reduced growth media (5 ng/mL bFGF). The cells were treated with rhEPO (0, 1, 5, or 10 U/mL of epoietin α; Amgen, Thousand Oaks, CA, USA) in the presence or absence of the PI3K/Akt inhibitors LY294002 (10 μmol/L; Calbiochem, San Diego, CA, USA) and wortmannin (2 μmol/L; Sigma-Aldrich, St Louis, MO, USA) (Wang et al, 2005) or the ERK1/2 inhibitors U0126 (10 μmol/L; Calbiochem) and PD98059 (10 μmol/L; Calbiochem) (Sengupta et al, 1998) for 24 h. After washing the cells with phosphate-buffered saline, 2 mL culture media (described above) was added to each well and the cells were incubated for another 48 h after which time the supernatant as conditioned medium was removed and immediately frozen at −80°C.
Enzyme-Linked Immunosorbent Assay
The concentration of VEGF in cultured neural progenitor cells supernatant was determined using a mouse VEGF ELISA kit according to the manufacturer's instructions (R&D Systems). Using known concentrations of VEGF (0 to 500 pg/mL), a standard curve was calculated for each assay.
Capillary-Like Tube Formation Assay
Mouse brain-derived endothelial cells (2 times 104 cells) were incubated in Matrigel (BD Biosciences, San Jose, CA, USA) for 5 h in conditioned medium collected from neural progenitor cells treated with rhEPO (0, 1, 5, and 10 U/mL) in the presence or absence of the PI3K/Akt inhibitor LY294002 (10 μmol/L) or the ERK1/2 inhibitor U0126 (10 μmol/L) or a specific VEGFR2 antagonist (SU1498, 5 μmol/L; LC Laboratories, Woburn, MA, USA) (Haralabopoulos et al, 1994). All assays were performed in n = 6/group. For quantitative measurements of capillary tube formation, Matrigel wells were digitized under a × 10 objective (Olympus BX40) for measurement of total length of capillary tubes using a video camera (Sony DXC-970MD) interfaced with the MCID image analysis system (Imaging Research, St Catharines, ON, Canada) at 5 h. Tracks of endothelial cells organized into networks of cellular cords (tubes) were counted and averaged in randomly selected five microscopic fields (Rikitake et al, 2002).
Coculture of Neural Progenitor Cells with Mouse Brain Endothelial Cells
Twenty-four hours before coculture, MBECs (2 times 105) were plated into the lower chamber of the six-well plate in medium with 10% FBS. Neural progenitor cells (1 times 106) were plated on six-well plates containing reduced growth media (5 ng/mL bFGF). The cells were treated with rhEPO (0, 1, 5, and 10 U/mL) with or without the PI3K/Akt inhibitors LY294002 (10 μmol/L) and wortmannin (2 μmol/L) or the ERK1/2 inhibitors U0126 (10 μmol/L) or PD98059 (10 μmol/L). After washing both the cells with phosphate-buffered saline, neural progenitor cells were seeded in the upper chamber of a Falcon 0.4 μm cell culture insert, in a total of 3 mL culture media (described above,
Immunocytochemistry and Quantification
Double immunofluorescent staining of cultured cells was performed, as described previously (Zhang et al, 2001, 2003). The following primary antibodies were used: rabbit anti-nestin (1:100; BD Biosciences) and rabbit anti-SOX2 (1:50; Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA). Cultured cells were fixed in 4% paraformaldehyde for 15 to 20 mins at room temperature. Nonspecific binding sites were blocked with 1% bovine serum albumin for 60 mins at room temperature. The cells were then incubated with the primary antibodies listed above and with CY3-conjugated or fluorescein isothiocyanate-conjugated secondary antibodies. Nuclei were counterstained with 4',6'-diamidino-2-phenylindole (Vector Laboratories, Burlingame, CA, USA).
Real-Time Reverse Transcription-PCR
Quantitative PCR was performed using SYBR Green real-time PCR method. Total RNA was isolated from neurosphere cultures using the Stratagene Absolutely RNA MicroRNA isolation kit (Stratagene, La Jolla, CA, USA). Quantitative reverse transcription-PCR (RT-PCR) was performed on an ABI 7000 PCR instrument (Applied Biosystems, Foster City, CA, USA) using three-stage program parameters provided by the manufacturer, as follows: 2 mins at 50°C, 10 mins at 95°C, and then 40 cycles of 15 secs at 95°C and 1 min at 60°C. Specificity of the amplification product produced was confirmed by examination of dissociation reaction plots. A distinct single peak indicated that a single DNA sequence was amplified during PCR. Each sample was tested in triplicate using quantitative RT-PCR, and samples obtained from three independent experiments were used for analysis of relative gene expression data using the 2−ΔΔ
Western Blot Analysis
Western blots were performed according to published methods (Wang et al, 2006a,
b
). Briefly, lysates from neural progenitor cells and MBECs were sonicated for 10 secs and centrifuged at 10,000
Statistical Analysis
A one-way analysis of variance was performed and statistical significance was set at
Results
Subventricular Zone Cells Exhibit Markers of Neural Progenitor Cells
Immunostaining analysis showed that the majority of SVZ cells cultured in the growth medium were nestin and SOX2 positive, markers of neural progenitor cells (Figure 1), which is consistent with published studies (Wang et al, 2007).
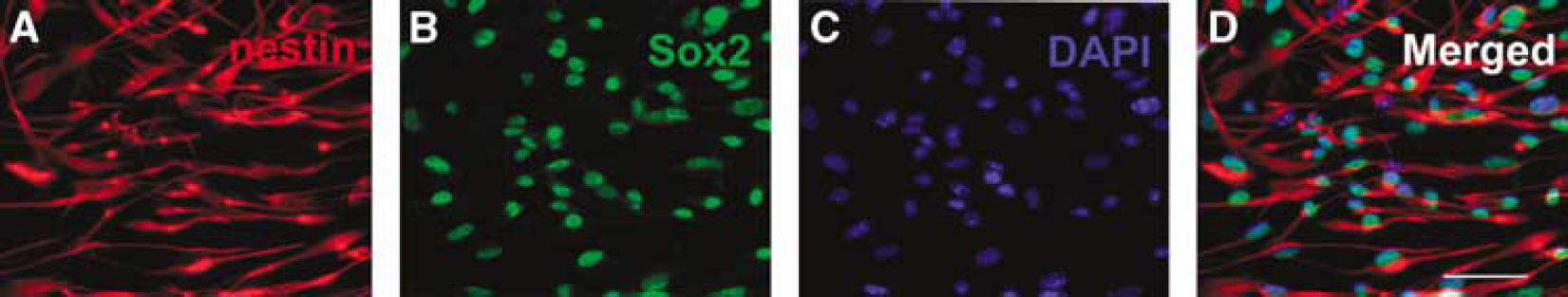
Adult SVZ cells are neural progenitor cells. Panels A to D show that the majority of SVZ cells are positive for markers of nestin (
Recombinant Human Erythropoietin Stimulates Neural Progenitor Cells to Secrete Vascular Endothelial Growth Factor
Adult neural progenitor cells express many angiogenic growth factors, including VEGF (Deleyrolle et al, 2006; Liu et al, 2007). To examine whether rhEPO upregulates VEGF expression, neural progenitor cells were incubated with rhEPO (0, 1, 5, and 10 U/mL) for 24 h. Real-time RT-PCR analysis showed that treatment with rhEPO dose-dependently upregulated VEGF expression in neural progenitor cells (Figure 2). To examine whether rhEPO increases VEGF proteins, VEGF levels were measured by means of an ELISA kit in the supernatant harvested from neural progenitor cells treated with rhEPO for 24 h. Incubation of neural progenitor cells in the presence of rhEPO (5 and 10 U/mL) significantly (
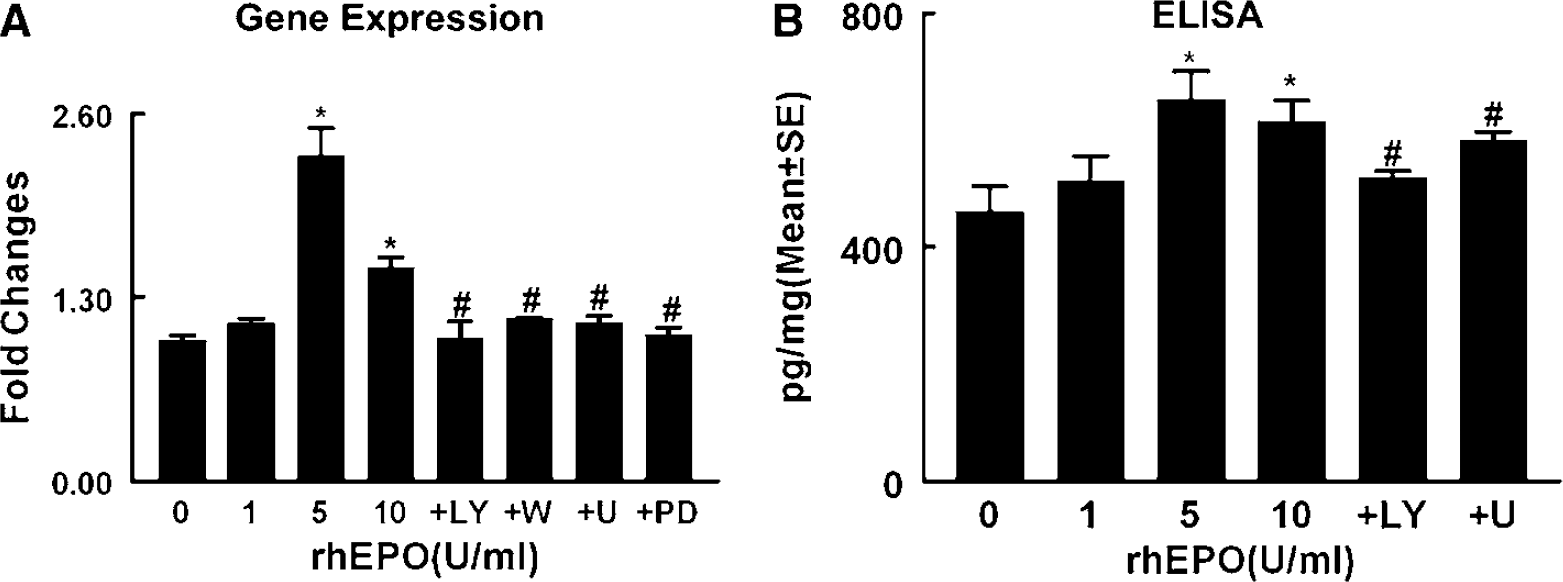
Effect of rhEPO on VEGF expression and secretion. Real-time RT-PCR (
In addition, western blot analysis showed that treatment of neural progenitor cells with rhEPO (5 U/mL) for 30 mins increased phosphorylated Akt and ERK1/2 but did not alter the levels of total Akt and ERK1/2 (Figure 3). To determine the ability of activated Akt to phosphorylate its downstream targets, phosphorylation of GSK3α/β, a well-characterized Akt substrate, was measured. Erythropoietin-activated Akt significantly increased serine phosphorylation of GSK3α/β (Figure 3). To evaluate whether activation of Akt and ERK1/2 affects VEGF expression, levels of VEGF mRNA and proteins were determined using real-time RT-PCR and enzyme-linked immunosorbent assay, respectively. Incubation of neural progenitor cells with rhEPO (5 U/mL) in the presence of PI3K/Akt inhibitors (LY29402 and wortmannin) or ERK inhibitors (U0126 and PD98059) (Fukazawa et al, 2002) substantially attenuated activation of Akt or ERK1/2, respectively, and abolished VEGF upregulation in neural progenitor cells (Figure 2) and VEGF proteins in the supernatant (Figure 2). Western blots showed that rhEPO phosphorylated Akt as early as 5 mins after incubation, whereas phosphorylation of ERK1/2 occurred at 10 mins (Figure 3). These data suggest that the PI3K/Akt and ERK1/2 signaling pathways mediate exogenous EPO-augmented VEGF and that the PI3K/Akt pathway may activate ERK1/2.
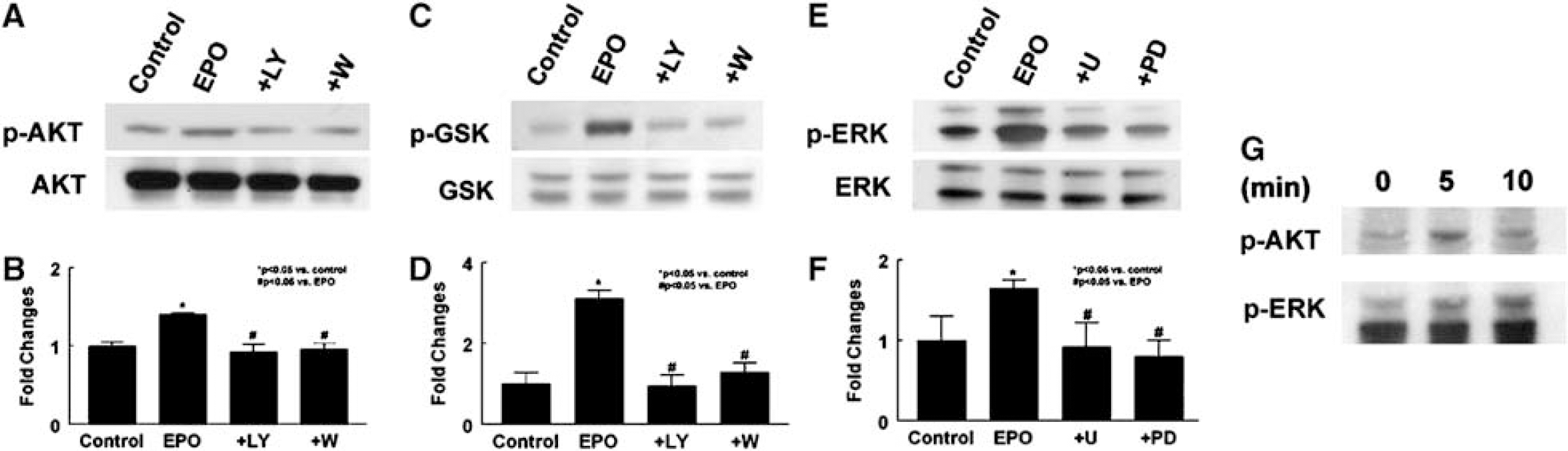
Effects of rhEPO on the PI3K/Akt and ERK1/2 signaling pathways. Western blot analysis shows that incubation of neural progenitor cells with rhEPO (EPO) significantly increased phosphorylated Akt (p-AKT;
Supernatant Harvested from Neural Progenitor Cells Treated with Recombinant Human Erythropoietin Enhances In Vitro Angiogenesis
To examine whether VEGF secreted by neural progenitor cells treated with rhEPO promotes angiogenesis, we performed a capillary tube formation assay that has been widely used for
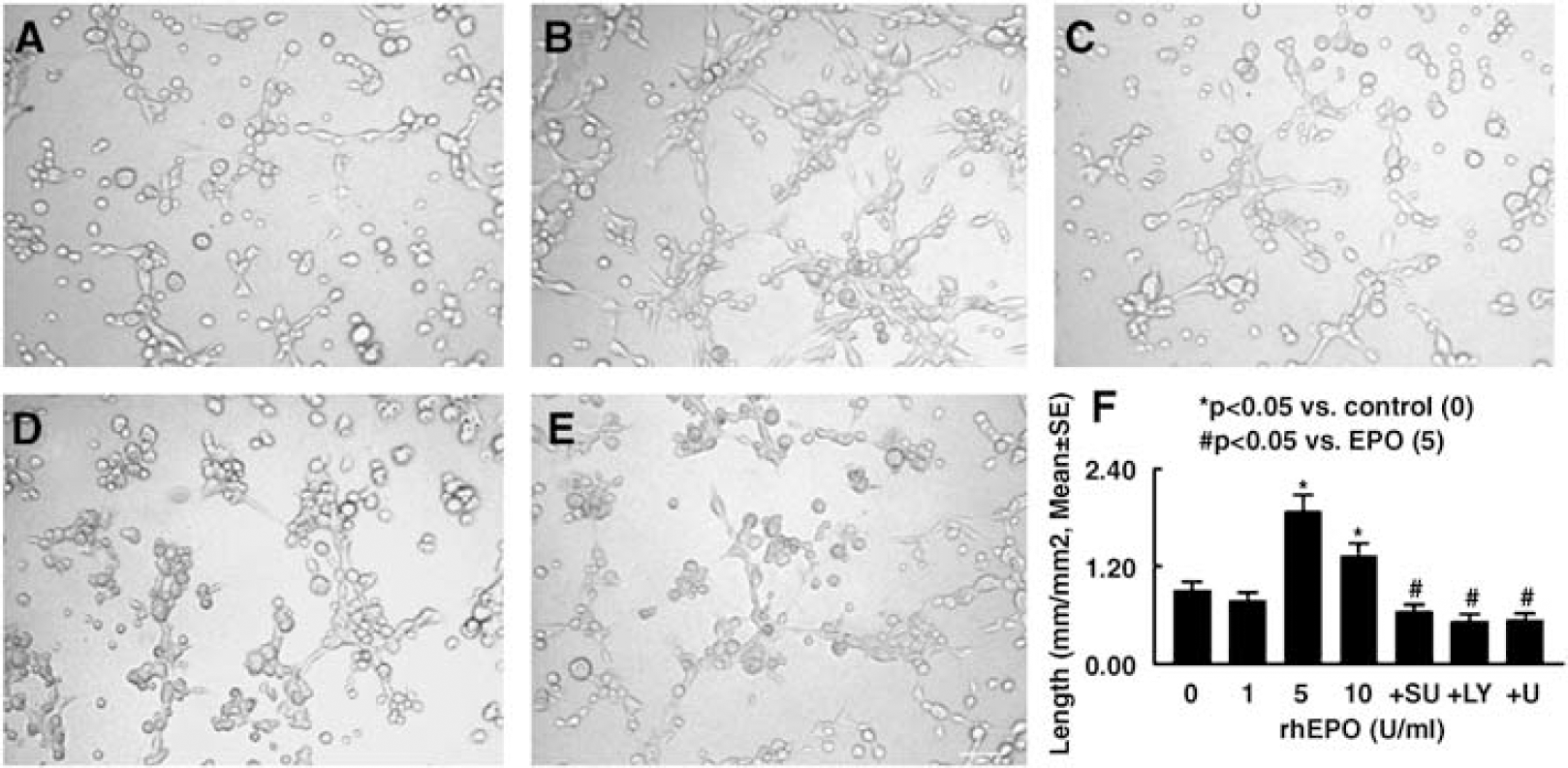
Neural progenitor cells treated with rhEPO promote capillary tube formation. Panels A to E show capillary-like tube formation in conditioned media from control (
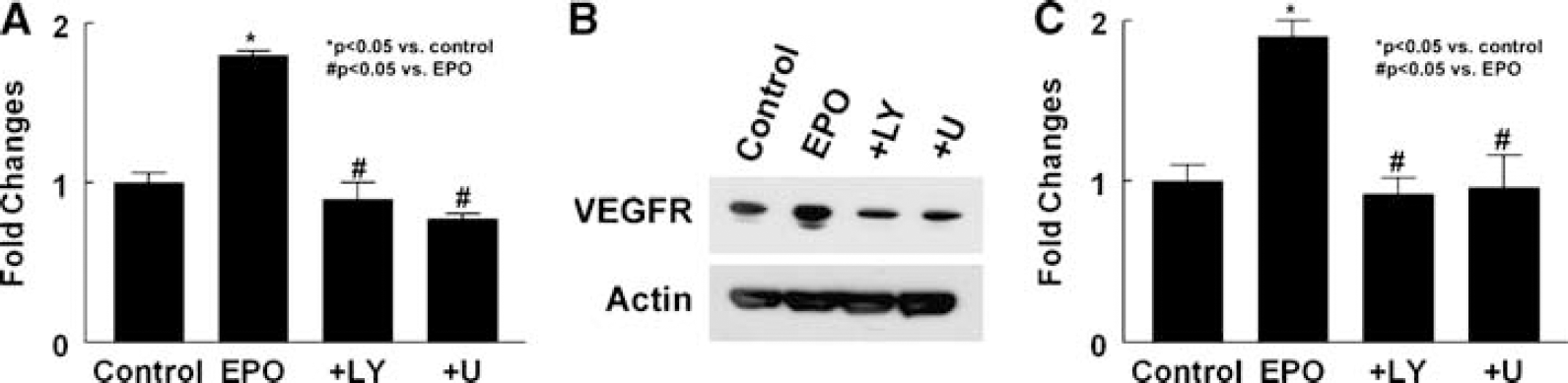
Effect of neural progenitor cells treated with rhEPO on expression of VEGFR2 in MBECs. Real-time RT-PCR (
To examine whether EPOR is required for upregulation of VEGFR2 in MBECs induced by the supernatant, rhEPO (5 U/mL) was incubated with neural progenitor cells derived from ΔEPOR mice. The supernatant harvested from this incubation did not elevate VEGFR2 mRNA levers in MBECs (1.43 ± 0.08,
Collectively, these results suggest that VEGF secreted by rhEPO-treated neural progenitor cells interacts with upregulated VEGFR2 in MBECs to stimulate
Discussion
Our
Adult SVZ neural progenitor cells express many angiogenic factors, including VEGF (Deleyrolle et al, 2006; Liu et al, 2007). This study shows that exogenous EPO stimulated the neural progenitor cells to secrete VEGF in a dose-dependent manner, suggesting a specific effect of exogenous EPO on neural progenitor cells. However, it remains to be determined whether rhEPO-increased VEGF protein results from increases of transcription and/or the stability of VEGF mRNA. The effect of EPO on VEGF requires EPOR in neural progenitor cells because increases in VEGF protein levels were not detected in the supernatant harvested from neural progenitor cells derived from ΔEPOR mice after incubation with rhEPO. These
Erythropoietin interacts with its receptor and activates many signaling pathways, including two important kinase cascades, the PI3K/Akt and ERK1/2 signaling pathways (Arcasoy and Jiang, 2005). Consistent with published studies, this study shows that treatment of neural progenitor cells with rhEPO activated Akt and its downstream target GSK3α/β, and ERK1/2 (Dong et al, 2001; Wang et al, 2006a, b ). Blockage of the PI3K/Akt and ERK1/2 pathways with pharmacological inhibitors abolished rhEPO-augmented VEGF. A time-course western blot analysis showed that activation of Akt by rhEPO occurs before phosphorylation of ERK1/2. These data suggest that exogenous EPO activates the PI3K/Akt and ERK1/2 signaling pathways that promote neural progenitor cells to secrete VEGF and the PI3K/Akt pathway may trigger activation of the ERK1/2 pathway.
Vascular endothelial growth factor mediates neurogenesis by augmenting proliferation and neuronal differentiation of neural progenitor cells (Jin et al, 2002; Meng et al, 2006). However, whether VEGF secreted by neural progenitor cells has any effects on angiogenesis has not been fully investigated (Teng et al, 2008). Using an
In summary, our results show that VEGF secreted by rhEPO-treated neural progenitor cells promotes angiogenesis and that the PI3K/Akt and ERK1/2 pathways mediate upregulation of VEGF in neural progenitor cells whereas the supernatant harvested from rhEPO-treated neural progenitor cells increases VEGFR2 levels in cerebral endothelial cells.
