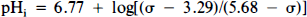Abstract
A new model of transient cerebral ischemia in 10-day-old rats is described. Under microscopic guidance, the right external and internal carotid arteries were electrically coagulated. A solid 0.47 mm diameter nylon thread was inserted into the right common carotid artery toward the ascending aorta up to 10–12 mm from the upper edge of the sternomastoid muscle (preischemic rats). A 60-min cerebral ischemia was induced by clamping the left external and internal carotid arteries (ischemic rats), followed by 3-h recirculation. 31P magnetic resonance (MRS) spectroscopic studies revealed that severe intracellular acidosis occurred and ATP disappeared completely for at least the last 20 min of ischemia. Cerebral blood flow (CBF), measured by the hydrogen clearance technique, decreased to ∼11% of the preischemic level in the frontal cortex soon after the induction of ischemia. On resuscitation, ATP recovered completely and the preischemic intracellular pH level was restored within 180 min. CBF had recovered to ∼30% of the preischemic level at 5 min after resuscitation. The CBF recovery was not complete even at 180 min after resuscitation. With this model, the effects of pure ischemia without hypoxia on the neonatal brain and the process of recovery from transient ischemia can be studied.
Hypoxic-ischemic encephalopathy (HIE) is one of the major and important causes of developmental disabilities such as mental retardation and cerebral palsy. It is the ultimate result of hypoxia and ischemia, which lead to metabolic deterioration. Although, in most cases, hypoxia and ischemia occur simultaneously on asphyxia, ischemia may have a greater biochemical effect on the central nervous system (CNS). It is known that periventricular leukomalacia in the preterm infant is primarily due to an ischemic lesion (DeReuck et al., 1972; Yoshioka et al., 1994). The pathogenesis of parasagittal cerebral injury, also called “watershed infarcts”, in full-term infants is principally related to perfusion impairment in the border zones between the end fields of the major cerebral arteries (Brierley, 1976). Therefore, it is said that hypoxia alone does not lead to brain damage, but, rather, its combination with ischemia, or isolated cerebral ischemia, is a necessary prerequisite for tissue injury to occur (Vannucci, 1992).
However, very little is known about the effects of pure ischemia without hypoxia on the perinatal brain since the only experimental method previously available for pure cerebral ischemia without hypoxia for the small laboratory animal involved decapitation. Thurston et al. (1969) found that cerebral ATP in the mature mouse had almost completely disappeared at 4 min after decapitation, while that in the neonatal mouse remained at 70% of the control level; thus, they concluded that the neonatal brain has a lower metabolic rate than does the mature one. Such data demonstrate specific aspects of the developing brain and suggest that data obtained in experiments on cerebral ischemia in mature animals cannot be simply extrapolated to immature ones. In addition, experiments involving decapitation cannot provide information on the process of recovery from cerebral ischemia. Therefore, it is indispensable to develop a model of transient cerebral ischemia in a neonatal animal for a more complete understanding of the pathogenesis of HIE.
In this article, we report a new model of transient cerebral ischemia in 10-day-old rats with no such complicating features as need for artificial ventilation, hypotension, or generalized seizures. Using this model, the effects of pure, transient ischemia without hypoxia on brain energy metabolism and CBF were studied by means of 31P-magnetic resonance spectroscopy (MRS) and the hydrogen clearance technique, respectively.
MATERIALS AND METHODS
Animal preparation and operation procedures
Figure 1-(1) illustrates the blood supply to the chest and the base of the brain in rats (Greene, 1955). The left common carotid and left subclavian arteries both arise from the aortic arch. The right common carotid and right subclavian arteries (the latter supplying the right vertebral artery) both arise from the innominate artery. The circle of Willis is well developed in rats, i.e., both the anterior and posterior communicating arteries are fairly large (Brown, 1966).
Blood supply to the chest and the base of the brain in rats. 1: Sham-operated rat. Arteries: 1, Anterior cerebral; 2, middle cerebral; 3, posterior cerebral; 4, right external; 5, right internal; 6, right common carotid; 7, right subclavian; 8, innominate; 9, anterior communicating; 10, posterior communicating; 11, basilar; 12, vertebral; 13, left common carotid; 14, left subclavian; 15, anterior spinal. 2: Preischemic rat. A, a coagulated site; B, a solid 0.47 mm diameter nylon thread; C, a nylon 0.09 mm in diameter thread; 3: An ischemic rat. D, A small clip.
Ten-day-old Wistar rats weighing 23–28 g were used in these experiments. Each rat was anesthetized with i.p. pentobarbital (25 mg/kg). A ventral transverse cervical incision, 20 mm in length, was made 5 mm distal from the sternum, and both common carotid arteries were identified. The bilateral omohyoid muscles were removed and the bifurcations of both carotid arteries were separated from contiguous structures (sham-operated rats). The right external and internal carotid arteries were electrically coagulated. A solid 0.47 mm diameter nylon thread was inserted into the right common carotid artery toward the ascending aorta up to 10–12 mm from the upper edge of the right sternomastoid muscle, followed by ligation with a 0.09 mm diameter nylon thread (preischemic rats) (Fig. 1-(2)). A 0.09 mm diameter nylon thread was placed loosely around the left carotid artery so that the bifurcation could be easily identified. All procedures were performed within 20 min.
Induction of ischemia
We induced ischemia and measured physiological variables in rats kept in the vertical position, since, as described below, we used a spectrometer with a vertical superconducting magnet for 31P-MRS measurements and we wanted to obtain physiological data under the same conditions. Eighteen rats were fixed in place on wooden tables and then the tables were set vertically. Their body temperatures were maintained at ∼37°C by means of a heating pad with circulating warm water. Ninety min after removal from the dams (−50 min after the operation), the bifurcation of the left carotid artery was exposed by pulling the thread around the artery, and cerebral ischemia was induced by clamping the left external and internal carotid arteries with a small clip weighing 0.04 g [Fig. 1-(3)]. Recirculation was started out by removing the clip after 60 min ischemia. Surviving animals were observed for 3 h after recirculation as to whether they exhibited seizures, dyspnea, or pale skin color.
Measurement of physiological variables
Measurement of arterial blood gases, pH, and glucose. Seven sham-operated rats and 14 preischemic rats were used. Their body temperatures were maintained ∼37°C by means of a heating pad with circulating warm water. Ninety minutes after removal from the dams, in seven sham-operated rats and seven preischemic rats, the left common carotid artery was cut and samples of arterial blood were immediately collected into heparinized capillary tubes. In the other seven preischemic rats, 60 min cerebral ischemia was induced, and samples of arterial blood from the surviving rats were collected before recirculation. Arterial blood gases and pH were measured with a 178 pH/Blood Gas Analyzer (Corning Medical and Scientific, Medfield, MA, U.S.A.), and glucose concentrations with a Glucoster II/Glucostix (Miles Inc., Elkhart, U.S.A.).
Monitoring of mean arterial blood pressure (MABP) and heart rate. A 0.3-mm diameter, 7-mm long, silicon-reinforced glass fiber was fixed at the end of PE-10 tubing (Intramedic). A part of the glass fiber was inserted, under microscopic guidance, into the left femoral artery, with cutdown procedures in five rats to monitor MABP and heart rate before, during, and after ischemia.
Perfusion with India ink
In three sham-operated rats, three preischemic rats, and five ischemic rats, the brains were perfused transcardially with a 1:1 mixture of gelatin and India ink [India ink: 12% gelatin (wt/vol)] at the flow rate of 260 ml/kg/min [cardiac output of an adult rat (Guyton et al., 1973)] for 5 min. After confirming that the tips of the tails were fully stained, the brains were removed and the gelatin solidified with ice.
Measurement of CBF by the hydrogen clearance technique
CBF was measured by the hydrogen clearance technique (PHG 201 UH meter, Sanei Ltd., Tokyo, Japan) in 10 sham-operated and 10 preischemic rats. Their body temperatures were maintained ∼37°C by means of a heating pad with circulating warm water. Two 0.2 mm diameter electrodes were inserted to a depth of 1 mm in the frontal cortex 1 mm rostral and 1 mm lateral from the bregma. It was later ascertained by microscopic examination that the tips of the electrodes were located in the frontal cortex. A reference electrode was placed subcutaneously in the back. CBF was calculated by the initial slope technique, using the first 2 min of the washout curve.
31P-MRS measurements
The experiments were performed on 25 rats. Each rat was positioned supine on the 31P-MRS probe with its forehead resting on a 10-mm, four-turn surface coil. 31P-MRS was performed using a SCM-200 spectrometer (JEOL Ltd., Tokyo, Japan) with a 9-cm wide bore vertical 4.7 Tesla superconducting magnet operating at 80.75 MHz. Field homogeneity was optimized by shimming on the water proton signal from a rat brain until the line width was 23.4375–39.1563 Hz. With a pulse width of 12 μs and an interpulse delay of 0.8 s, 750 transients were accumulated. The flip angle at the center of the coil was 90°. A phantom study revealed that in our system, the sensitive volume providing a signal extended to a depth of ∼3 mm (unpublished data). Free induction decays were processed by exponential line broadening at 30 Hz before Fourier transformation.
Raw spectra were used for measurement of the peak areas of PCr, Pi, and β-ATP using an analyzer (MOP/AMO3; Kontron Co., München, Germany) (Lorentzian curve-fitting software is not yet available in our laboratory). pH1 was calculated from the chemical shift of the Pi peak from the PCr peak using the standard equation:
where σ is the chemical shift of the Pi peak (Petroff et al., 1985).
The reduction of the resonance intensity due to saturation (the saturation factor) was determined by obtaining a fully relaxed spectrum (interpulse delay of 20 s) for six rats. The saturation factors of PCr, Pi, and β-ATP were 0.46 ± 0.08, 0.43 ± 0.03, and 0.63 ± 0.13, respectively.
There are three problems in the measurement of 31P-MRS. Firstly, rats must be in a vertical position, thus the possibility that such an artificial position may affect the cerebral energy metabolism. Secondly, the probe is removed from the magnet twice: once when ischemia is induced by clamping with a small clip and once when recirculation is started. Such manipulations may affect the shimming and, moreover, the results. Thirdly, insertion of a nylon thread into the right common carotid artery could affect the cerebral energy metabolism. Preliminary experiments were performed to examine these problems. PCr/Pi, PCr/ATP, and pH; were measured in six sham-operated and four preischemic rats. The PCr/Pi, PCr/ATP, and pH; values for sham-operated rats were 1.18 ± 0.10, 0.77 ± 0.09, and 7.19 ± 0.04, respectively; those for preischemic rats were 1.26 ± 0.06, 0.82 ± 0.03, and 7.20 ± 0.04, respectively. There were no significant differences between groups with respect to these parameters. Spectra were taken for preischemic rats every 10 min for 250 min, during which time the probe was removed from the magnet twice. Although these parameters fluctuated slightly, repeated analysis of variance (ANOVA) revealed no significant differences in them throughout the experiment. Results therefore indicated that these three problems do not affect energy metabolism.
Statistical analysis
Arterial blood gases, pH, glucose, and CBF data were analyzed by one-way ANOVA with repeated measures. A paired Mest was performed for the MABP and heart rate data. A paired Mest with Bonferroni correction was used to analyze the 31P-MRS data. All data are presented as means ± SD.
RESULTS
Survival rate
Of 18 rats, 13 (72.2%) were alive and five (27.8%) dead by the end of 60-min ischemia. All rats exhibited transient tachypnea during the first few minutes. Five rats exhibited dyspnea within 20 min, followed by cyanosis and incontinence, and died within 40 min. Regular respiration and good skin color were maintained in the survivors throughout the experiment. No rat exhibited seizures.
Physiological variables
Arterial blood gases, pH, and glucose. PaO2, PaCO2, pH, and glucose concentrations for sham-operated rats were 85.7 ± 6.8 mmHg, 42.2 ± 6.7 mmHg, and 7.41 ± 0.04, and 159 ± 27 mg/dl, respectively: those for preischemic rats were 88.4 ± 8.5 mmHg, 45.5 ± 3.1 mmHg, 7.41 ± 0.03, and 156 ± 24 mg/dl, respectively; and those for rats subjected to 60-min ischemia were 84.6 ± 9.5 mmHg, 47.0 ± 5.9 mmHg, 7.36 ± 0.05, and 165 ± 27 mg/dl, respectively. There were no significant differences in these variables between the three groups.
MABP and heart rate. MABP significantly rose to 42 ± 3 mmHg from the preischemic level of 33 ± 1 mmHg at the beginning of ischemia and remained at that level throughout the ischemic period. On recirculation, MABP recovered to the preischemic level. The heart rate increased significantly to 305 ± 28 beats/min from the preischemic level of 261 ± 31 beats/min immediately after ischemia and remained at that level even upon recirculation.
Perfusion with India ink
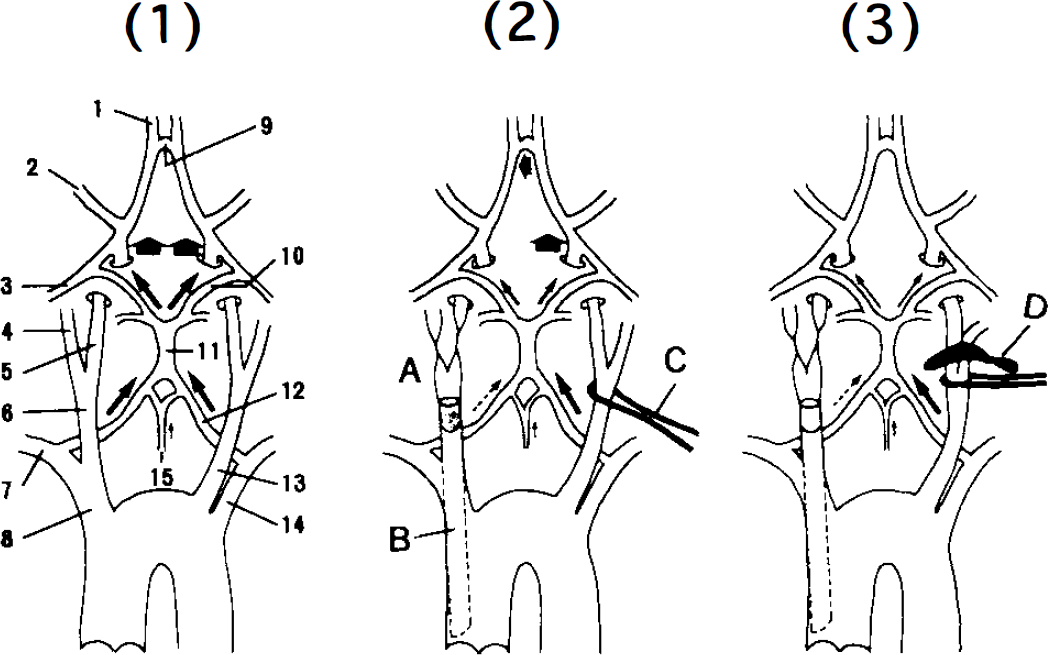
Figure 2 shows rat brains perfused with India ink. Perfusion of all brain structures was well maintained in preischemic rats as well as in sham-operated ones. Although perfusion of the cerebral hemispheres in ischemic rats was markedly decreased, that of the cerebellum and brain stem was preserved, as in preischemic rats.
Brains perfused with India ink. A, sham-operated rat; B, preischemic rat; C, ischemic rat.
CBF measurement
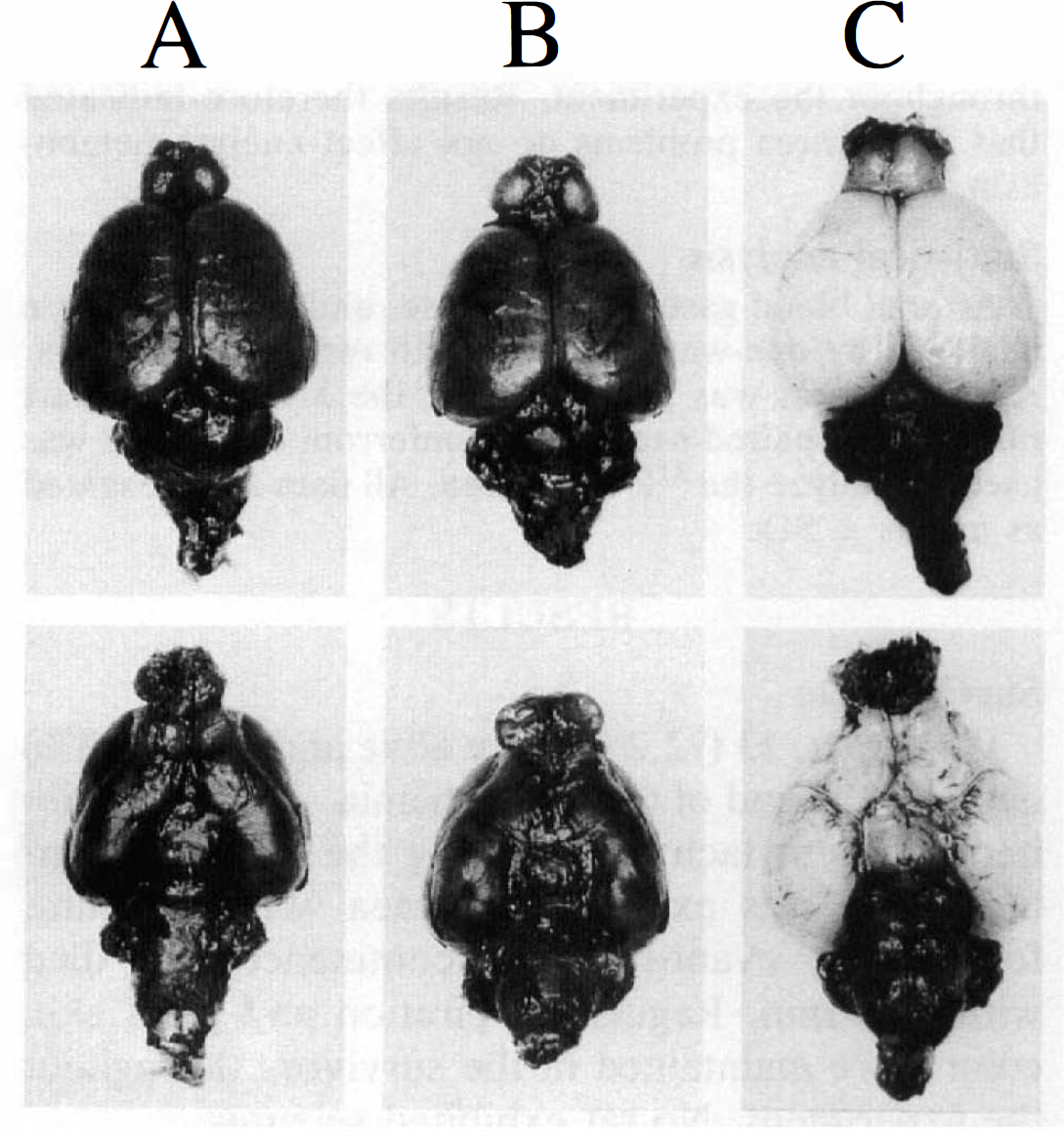
Figure 3 shows the changes in CBF during and after ischemia. CBF in sham-operated rats was 75.5 ± 9.6 ml 100 g−1 min−1 in the right frontal cortex and 76.8 ± 10.8 ml 100 g−1 min−1 in the left frontal cortex. In preischemic rats, CBF was 69.7 ± 7.7 ml 100 g−1 min−1 (92.3% of sham-operated rat level) in the right cortex and 71.8 ± 8.0 ml 100 g−1 min−1 (93.4% of sham-operated rat level) in the left cortex. No difference was detected between the two groups on either side. CBF decreased to 7.8 ± 5.7 ml 100 g−1 min−1 (11.2% of preischemic level) in the right frontal cortex and 8.1 ± 5.4 ml 100 g1 min−1 (11.3% of preischemic level) in the left frontal cortex at the beginning of ischemia. In the right frontal cortex, CBF had recovered to 23.5 ± 6.9 ml 100 g−1 min−1 (33.7% of preischemic level), 48.7 ± 8.4 ml 100 g−1 min−1 (69.9% of preischemic level), 54.4 ± 8.8 ml 100 g−1 min−1 (78% of preischemic level), 61.3 ± 12.7 ml 100 g−1 min−1 (87.9% of preischemic level), and 67.4 ± 8.8 ml 100 g−1 min−1 (96.7% of preischemic level) after 5, 30, 60, 120, and 180 min of recirculation, respectively. In the left frontal cortex, CBF had recovered to 25.9 ± 7.6 ml 100 g−1 min−1 (36.1% of preischemic level), 46.5 ± 6.3 ml 100 g−1 min−1 (64.8% of preischemic level), 59.2 ± 9.7 ml 100 g−1 min−1 (82.5% of preischemic level), 63.8 ± 11.3 ml 100 g−1 min−1 (88.9% of preischemic level), and 68.7 ± 7.5 ml 100 g−1 min−1 (95.7% of preischemic level) after 5, 30, 60, 120, and 180 min recirculation, respectively. Values after 180 min recirculation were not significantly different from those for preischemic rats. There was no significant difference between the two sides of the brain at any time of measurement.
Changes in CBF during and after ischemia (n = 10). Error bars represent standard deviation. S, sham-operated; P, preischemia; I, ischemia; R, recirculation. Statistically significant differences compared with preischemia: *, p < 0.05; **″, p < 0.01.
31P-MRS data
A series of representative 31P-spectra are shown in Fig. 4. With the onset of ischemia, PCr decreased faster than did β-ATP. At the end of ischemia, the spectrum showed the characteristic pattern of ischemia, i.e., the complete disappearance of PCr and β-ATP, an increase in Pi, and a decrease in pHi.
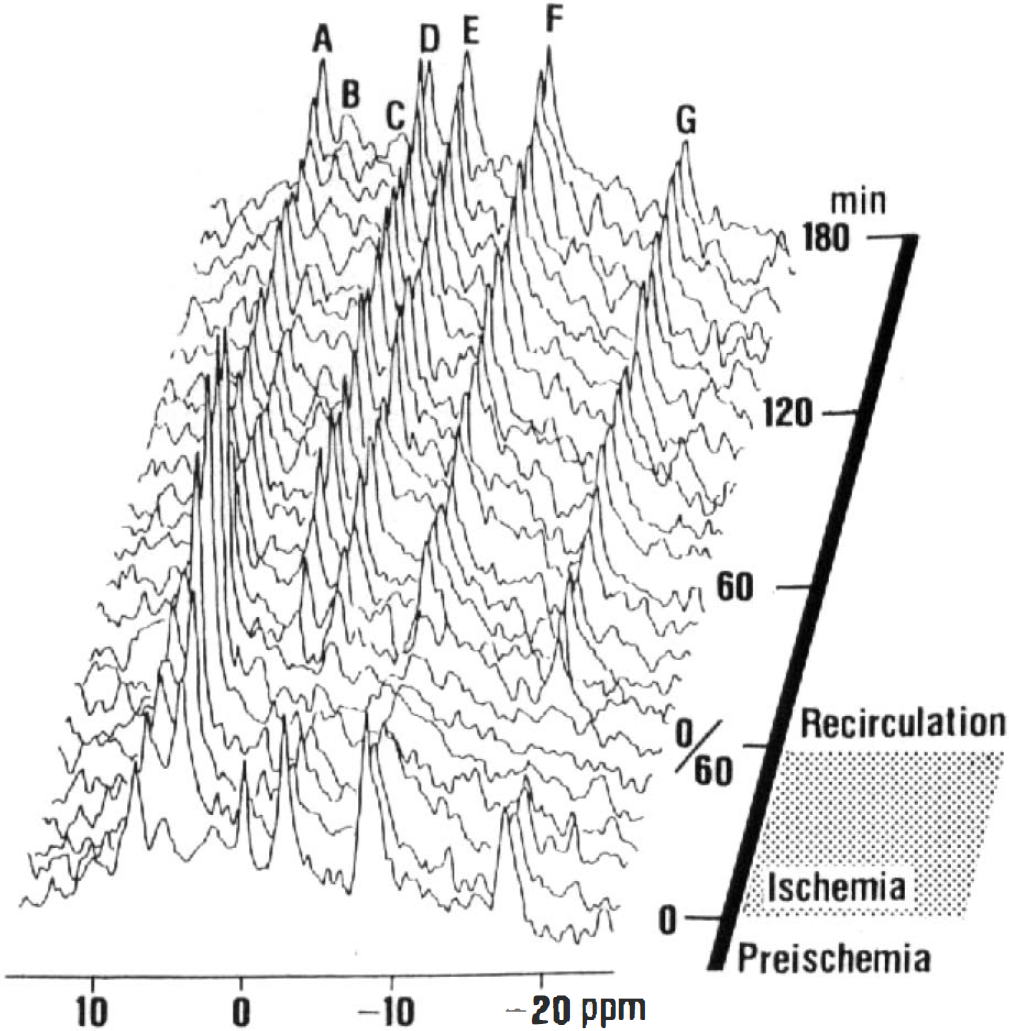
A series of representative 31P-spectra. Peak assignments; A, phosphomonoesters; B, inorganic phosphate; C, phosphodiesters; D, phosphocreatine; E, F, and G, γ-, α-, and β-phosphates of ATP, respectively.
Of 25 rats, 19 were alive and six dead at the end of ischemia. Data for 17 of the 19 live rats were included in the following analysis; MRS measurement in two live rats showed little changes in the spectra, i.e., slight decreases in PCr and pH; and no change in ATP. Figure 5 shows the sequential changes in PCr/Pi, %ATP (% of the preischemic level), and pH;. The preischemic PCr/Pi value was 1.71 ± 0.22, which decreased rapidly to 0 after 10–20 min of ischemia. On recirculation, PCr/Pi recovered almost linearly and reached the control level by 120 min recirculation. %ATP decreased slower than did PCr/Pi, but was 0 during the last 20 min of ischemia. %ATP recovered more rapidly than did PCr/Pi. pH; in preischemic rats was 7.19 ± 0.05, then decreased, reaching a value of 6.61 ± 0.10 at 20 min of ischemia. pH; recovered within 60 min recirculation.
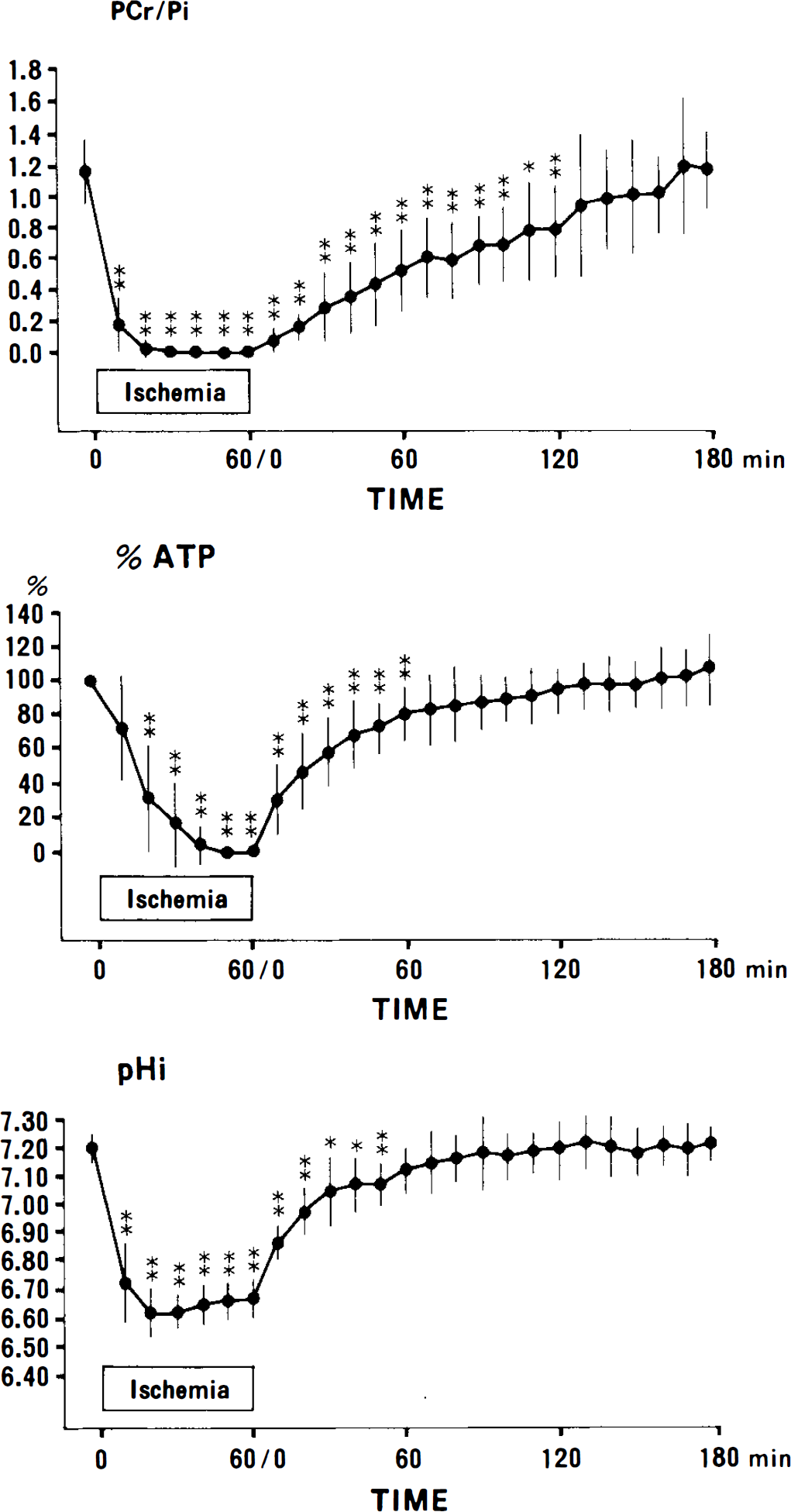
Time courses of changes in PCr/Pi, %ATP (% of preischemic level), and pH, (n = 17). Error bars represent standard deviation. Statistically significant differences compared with preischemia: *, p < 0.05; **, p < 0.01.
DISCUSSION
Our model is based on the following two characteristics of brain circulation in rats. First, the innominate artery gives rise to both the right common carotid and right subclavian arteries, the latter supplying the right vertebral artery. Insertion of a nylon thread into the right common carotid artery toward the ascending aorta interrupts the blood flow in the right common carotid artery completely and reduces that in the right vertebral artery. Secondly, CBF of the right hemisphere was maintained even when blood flow in the right common carotid artery was interrupted because the circle of Willis is well developed in the rat. This was confirmed by the results of a perfusion study: perfusion of all brain structures was well maintained in preischemic rats as well as in sham-operated ones.
In the survival rate study, 72.2% of the rats were alive and 27.8% dead, and in the 31P-MRS study, 76% were alive and 24% dead after 60 min of ischemia. A survival rate of >70% is sufficient for a neonatal rat model of transient cerebral ischemia. During 1 h of ischemia and 3 h-of recirculation, none of the surviving fats exhibited such complicating features as seizures, hypoxemia, or systemic hypotension. The perfusion study revealed that CBF in the cerebellum and brain stem was well preserved during ischemia. All of the dead cases had exhibited severe dyspnea within 20 min of ischemia. Pulsinelli and Brierley (1979) described a transient ischemic model in adult rats involving four-vessel occlusion. In their model, blood flow to the brain stem was considered to be maintained by a collateral via the anterior spinal artery. In our model, blood flow via the left vertebral artery was completely preserved, so the reason why there were cases that exhibited dyspnea is unclear. Since there are many variations in the circle of Willis in the rat (Brown, 1966), the ischemia may have been too severe for these animals and, therefore, may have caused a dysfunction of the respiratory center in the brain stem. The lack of significant increase in the serum glucose concentration during ischemia indicates the absence of epinephrine release from the adrenal glands. This implies that in this model, there is little effect on other organs; thus, the model is advantageous for investigating the effects of ischemia on the neonatal brain.
In the 31P-MRS study, PCr/Pi decreased from 1.17 ± 0.22 to 0.18 ± 0.17 at 10 min of ischemia. At this time, the ATP concentration was maintained at 75 ± 31% of control level. After disappearance of the PCr peak, ATP decreased rapidly to within noise level, at least for the last 20 min during ischemia in all rats. It has been reported that in complete ischemia in adult animals, electrical activity of the brain is suppressed (Hossmann and Sato, 1970) and that the first ATP concentration begins to decrease within a few minutes (Lowry et al., 1964). In the case of incomplete ischemia, as in this model, there are two critical thresholds of residual CBF: the upper threshold is for electrical failure and the lower one for energy and ion pump failures (Astrup et al., 1981). The upper threshold is considered to be 15–20 ml 100 g−1 min−1 (Heiss, 1983) and the lower one, 10 ml 100 g−1 min−1 in adult animals (Astrup et al., 1977; Branston et al., 1977). However, a higher flow than that of the lower threshold for a long period may cause energy failure and neuropathological damage. In this study, ATP began to decrease, at the latest, 40 min after the beginning of ischemia in all rats; thus, energy failure may occur in the neonatal brain with a CBF of 8 ml 100 g−1 min−1 for 40 min.
High energy metabolites and pH; were restored during recirculation in all animals. Figure 6 demonstrates the recovery processes of pHi, %ATP, and PCr/Pi during recirculation. pHi had recovered to 40% of preischemic level at 10 min of recirculation and to 91% at 70 min. ATP recovery was similar to or lagged behind pH; recovery. PCr/Pi recovery took much time. On the other hand, recent experiments on adult animals indicated that the pH; recovery lagged behind ATP recovery (Nishijima et al., 1989; Behar et al., 1989; Eleff et al., 1991). This phenomenon could be explained by persistent lactic acidosis during an early stage of recirculation (Behar et al., 1989). Permeability of the blood–brain barrier to lactate in 15-day-old rats is at least six times greater than that in adult animals (Cremer et al., 1976). The rapid recovery in the present model may be related to accelerated lactate washout in an early stage of recirculation.
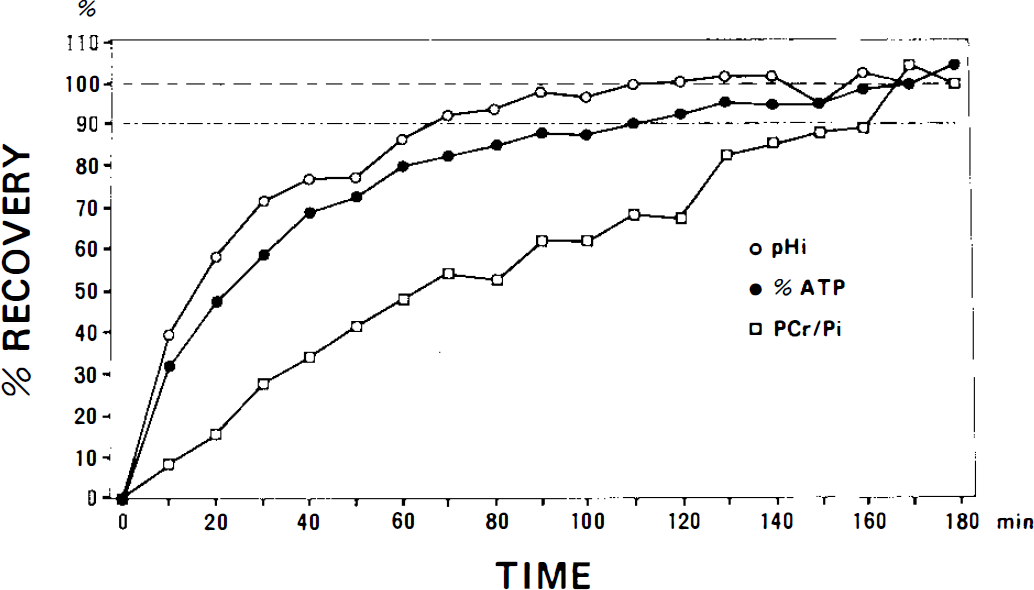
Processes of recovery of pHi, %ATP, and PCr/Pi during recirculation.
The rate of recovery of CBF was very slow; CBF had recovered to ∼70% of the preischemic level at 30 min of recirculation and to >90% at 180 min, despite adequate perfusion pressure. Although the mechanism underlying the delayed recovery of CBF remains unclear, the recirculation period is important because such perfusion impairment in the recirculation period may cause secondary damage to brain tissue. The fact that prolonged hypoperfusion is seen in transient ischemia of the immature brain is critical information for the development of therapeutic approaches to ischemic brain damage.
Footnotes
Acknowledgment:
The authors wish to thank Drs. Shoji Naruse and Kazuo Yoshizaki for their technical assistance. This study was supported in part by grants from the Ministry of Health and Welfare, and grants (nos. 03557046, 03670503, and 05670691) from the Ministry of Education, Japan.