Abstract
Peroxynitrite is a powerful oxidant capable of nitrating phenolic moieties, such as tyrosine or tyrosine residues in proteins and increases after traumatic brain injury (TBI). First, we tested the hypothesis that TBI increases nitrotyrosine (NT) immunoreactivity in the brain by measuring the number of NT-immunoreactive neurons in the cerebral cortex and hippocampus of rats subjected to parasagittal fluid-percussion TBI. Second, we tested the hypothesis that treatment with
Introduction
Traumatic brain injury (TBI) decreases cerebral blood flow (CBF) (Bouma et al, 1991; Marion et al, 1991). Although the exact mechanism of action is unknown, posttraumatic hypoperfusion is likely caused by a combination of trauma-induced release of vasoconstrictors and inhibition of cerebral vasodilators (DeWitt and Prough, 2003; Golding et al, 1999). Recent evidence suggests that oxygen free-radical-mediated inactivation of nitric oxide (NO), a powerful cerebral vasodilator (Furchgott and Zawadzki, 1980), plays an important role in this process (DeWitt et al, 1997; Cherian and Robertson, 2003). After head injury in humans and experimental animals (Cherian et al, 2000; Hlatky et al, 2002), brain tissue NO concentrations increase transiently, then significantly decrease. Traumatic brain injury produces superoxide anion radicals (Fabian et al, 1995) that can inactivate NO by converting it to peroxynitrite (ONOO−). Peroxynitrite is a powerful oxidant that, when protonated, can isomerize to trans-peroxynitrous acid, which in turn forms an excited state with reactive properties of the powerful oxidants nitrogen dioxide and the hydroxyl radical (Beckman et al, 1990).
Peroxynitrite exposure can cause systemic and cerebral vascular dysfunction. In rat spinal cord, ONOO− induced oxidation, tyrosine nitration (Bao et al, 2003), and lipid peroxidation (Liu et al, 2005). Thus, NO produced as a result of treatment with
Investigations of ONOO− are complicated as the ion is short-lived. ONOO− is capable of hydroxylation, nitration, and nitrosation reactions. Phenols react readily with ONOO− and the most common phenol in biologic systems is tyrosine. When ONOO− reacts with tyrosine residues in proteins, nitrotyrosine (NT) is produced; NT is therefore considered a biomarker of ONOO− production. Nitrotyrosine can be stained and localized using specific antibodies (Viera et al, 1999). Immunohistochemical staining revealed the presence of increased numbers of NT-positive neurons after spinal cord injury in rats (Liu et al, 2000), controlled cortical impact injury in mice (Whalen et al, 1999), fluid-percussion injury in rats (Besson et al, 2003), and head injury in humans (Forster et al, 1999). Although these reports indicate that central nervous system injury is associated with increases in NT immunoreactivity, the time course of protein nitration has not been established for TBI.
To measure the temporal course of protein nitration and determine whether
Materials and methods
Animals
Sixty-five adult (3 to 4 months) male Sprague—Dawley rats (350 to 400 g) were obtained from Harlan (Houston, TX, USA), housed 2 per cage with food and water ad libitum, and maintained at a constant temperature (21°C to 23°C) and humidity (45% to 50%) with lights on from 0700 to 1900 hours. After surgical manipulations, animals were housed singly. All experiments were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (National Research Council, 1996) and approved by the Institutional Animal Care and Use Committee of the University of Texas Medical Branch.
Surgical Procedures
Rats were anesthetized with 4% isoflurane, intubated and mechanically ventilated (NEMI Scientific; New England Medical Instruments, Medway, MA, USA) with 1.5% to 2.0% isoflurane in an oxygen-to-room-air ratio of 70:30, and prepared for parasagittal fluid-percussion TBI, as described previously (DeWitt et al, 1997). Briefly, the skin over the dorsal surface of the skull was incised in the midline and reflected, and a needle temperature probe (Yellow Springs Instruments, Yellow Springs, OH, USA) was placed under the reflected temporalis muscle. A 3 mm craniotomy was trephined 2.5 mm to the right of the midline, midway between lambda and bregma, and the dura was left intact. A modified plastic Luer-lok needle hub was cemented into the craniotomy using cyanoacrylic and dental cement. After surgery, rats were randomly assigned to receive sham TBI, moderate (2.0 atm) TBI, or moderate TBI +
Immunohistochemistry
Rats were reanesthetized with 4% isoflurane and ketamine (60 to 70 mg/kg intraperitoneally) plus xylazine (5 to 10 mg/kg intraperitoneally) either 12, 24, or 72 h after TBI or sham injury, and transcardially perfused with saline followed by 4% paraformaldehyde. The brains were removed, dehydrated, embedded in paraffin, and sectioned. Coronal sections (10 μm) were collected, deparaffinized with xylene and graded alcohols, placed in an antigen retrieval solution, and then immersed in 3% H2O2 in methanol (to block endogenous peroxidase activity) and goat-blocking serum (to block nonspecific binding).
The sections were then incubated with rabbit polyclonal anti-NT antibody (1:100 dilution; Upstate Biotechnology, Lake Placid, NY, USA) for 45 mins at 27°C and then overnight at 4°C, rinsed, and incubated with a biotinylated goat anti-rabbit IgG (1:200 dilution; Vectastain Elite ABC kit (Universal); Vector Laboratories, Burlingame, CA, USA) for 30 mins. The antigen—antibody complex was detected with a peroxidase-conjugated system (Vectastain Elite ABC kit (Universal); Vector Laboratories), developed with diaminobenzidine, and counterstained with hematoxylin. Additional sections were treated identically but were not incubated with the primary antibody (negative control).
Nitrotyrosine-immunoreactive neurons in sections on coded slides were counted in preselected regions of the hemispheres and hippocampal cell layers CA1 and CA3 and the polymorphic layer of the dentate gyrus (Figure 1) bilaterally by an investigator unaware of the treatment groups, and expressed as a percentage of total neurons in high-power microscopic fields. Neurons were identified by their large size and large, prominent euchromatic nuclei. Two slices were counted per animal, one each at two different levels of bregma (−3.3 and −5.2 mm). In the cortex, five zones were counted in each section. In the hippocampus, three zones were counted at bregma −3.3 (CA1, CA3, and dentate gyrus; see Figure 1).
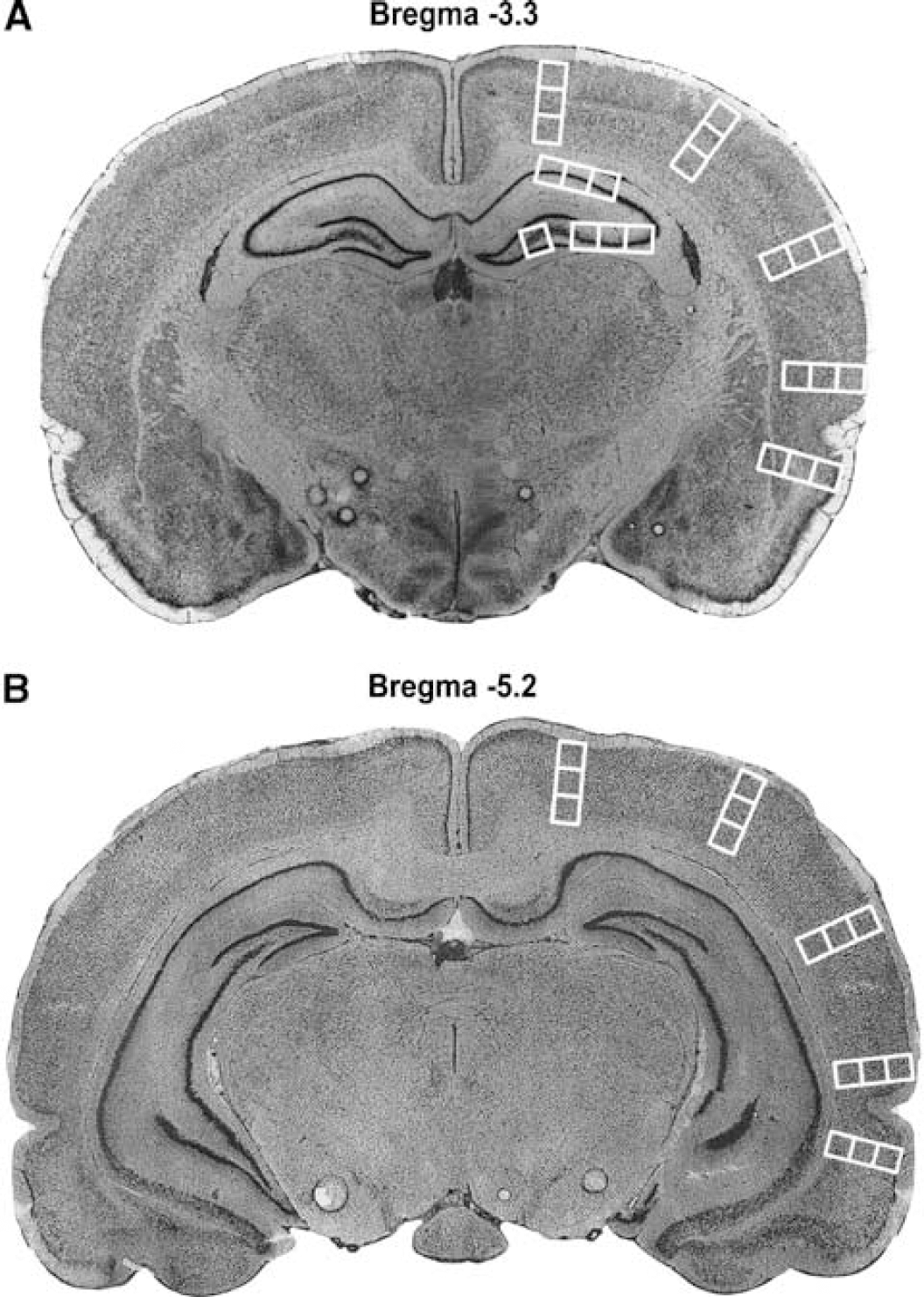
Coronal sections of rat brain are shown at −3.3 mm from bregma (
Statistical Analysis
Group descriptive statistics are expressed as mean ± s.e. of the mean and differences between groups were evaluated using analysis of variance (Table 1). The neuronal counting data were not normally distributed and are therefore expressed as quartiles. The effects of treatment group and time after injury were evaluated using a nonparametric two-way analysis of variance followed by Tukey's procedure using a two-sided alpha level of significance (P < 0.05). Comparisons between ipsilateral and contralateral hemispheres were evaluated using the Wilcoxon Signed Rank Test, with P < 0.05 considered significant. Neuronal counting statistical analyses were conducted using SAS (SAS Institute Inc (2004), SAS/STAT 9.1 User's Guide, Cary, NC, USA).
Group descriptive statistics
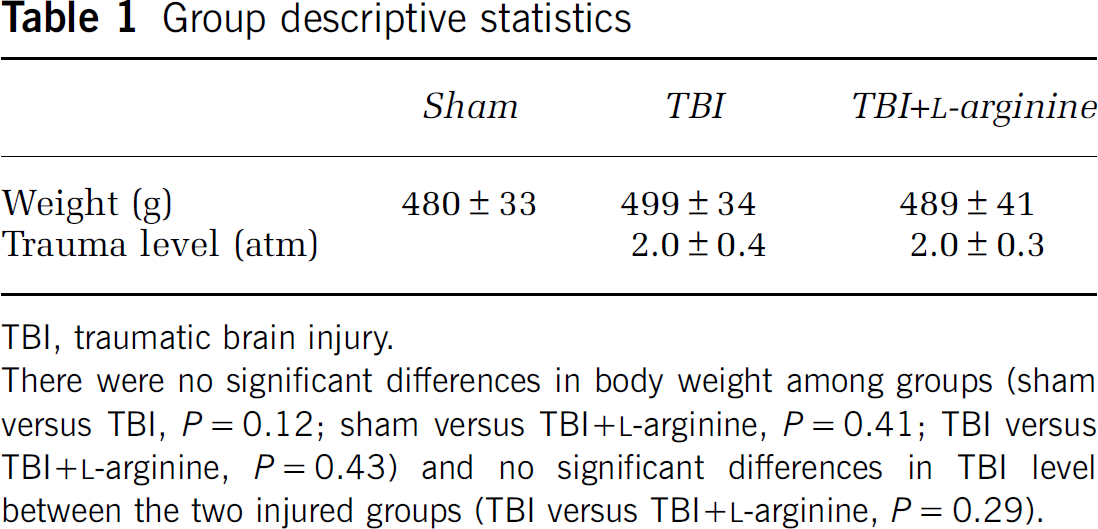
TBI, traumatic brain injury.
There were no significant differences in body weight among groups (sham versus TBI, P = 0.12; sham versus TBI +
Results
There were no significant differences in body weight among the three groups and no significant differences in TBI level between the two injured groups. Also, there were no deaths or unexpected complications because of surgery (Table 1). Nitrotyrosine staining was not observed in the negative control slides that were not incubated with the primary antibody. Nitrotyrosine immunoreactivity was observed in the cytoplasm (perikarya and processes) of neurons and occasionally in neuroglia. Only cells identified as neurons were counted.
Cerebral Cortex
Although there appeared to be fewer NT-positive neurons in the contralateral (relative to the injury site) compared with the ipsilateral cortex, there were no significant differences in the number of NT-positive cells between the ipsilateral and contralateral cortical hemispheres in the sham (P = 0.930), TBI (P = 0.221), or
In the ipsilateral cortex, a main effect of treatment (F2,63 = 12.06; P < 0.0001) was observed, whereas a main effect of time was not quite significant (F2,63 = 2.91; P = 0.062). Post hoc analysis revealed that the TBI group showed increased NT immuno-reactivity compared with the sham-operated group. Furthermore, animals that received
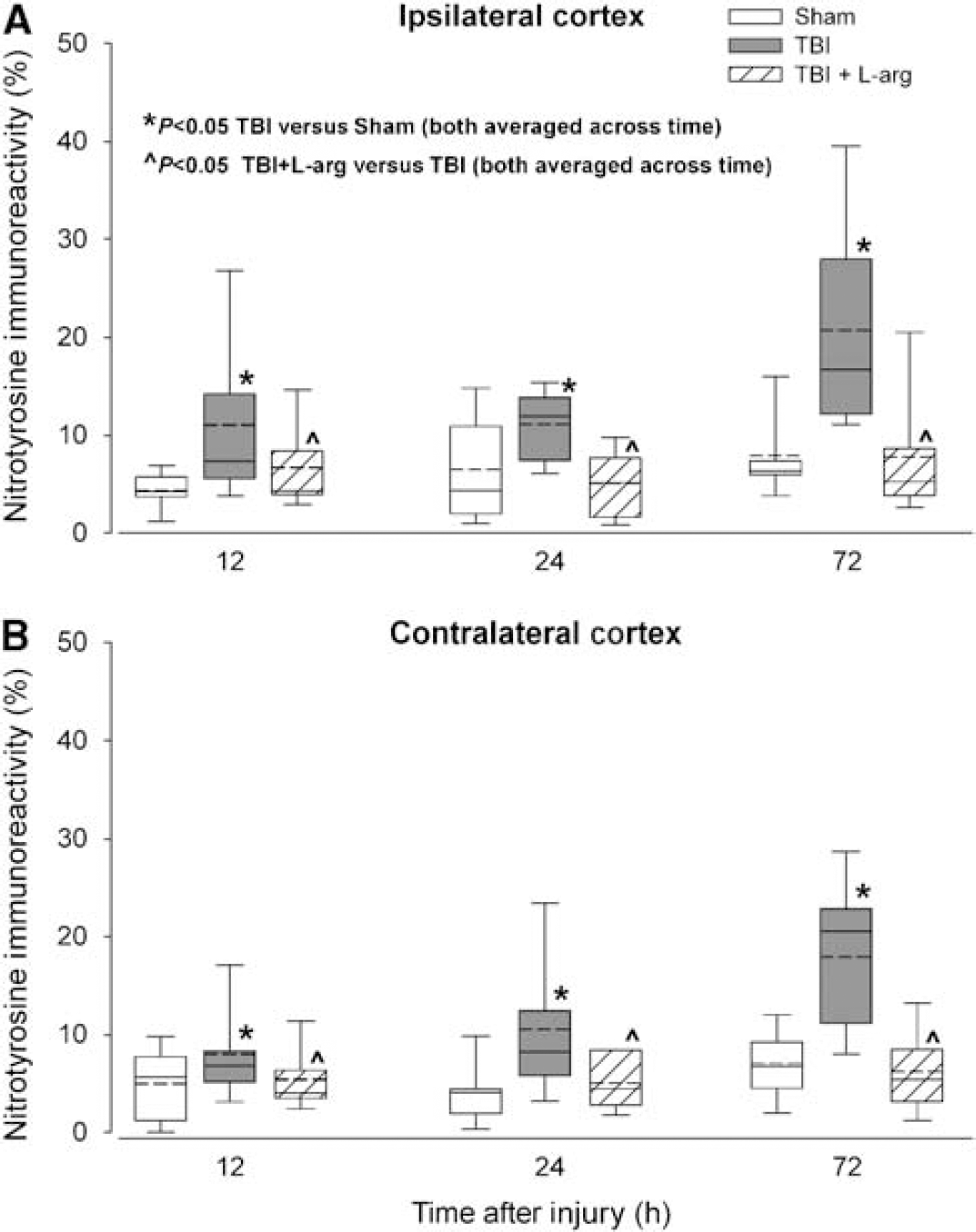
Percentages of NT-immunoreactive neurons in the ipsilateral (
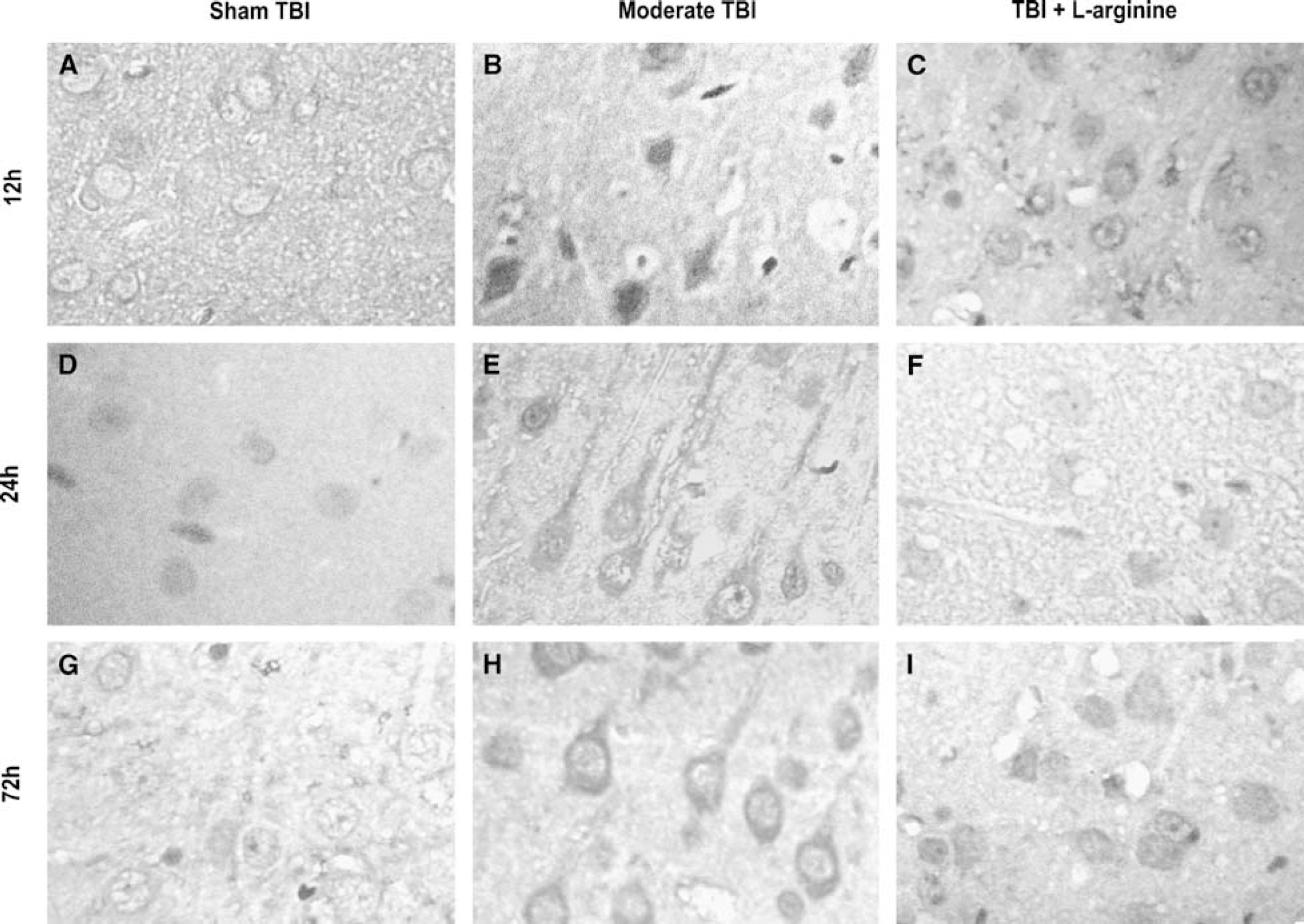
Examples of NT immunoreactivity taken from the ipsilateral cortex (relative to trauma site) are shown: sham TBI surgery (left column:
In the contralateral cortex, there was a main effect of treatment group (F2,63 = 6.51; P = 0.003) but not time (F2,63 = 2.34; P = 0.106). Post hoc analysis revealed effects similar to the ipsilateral cortex. The TBI group showed significantly higher percentages of NT-immunoreactive neurons compared with the sham group, and the
Hippocampus
Comparison between ipsilateral and contralateral regions showed no differences between the number of NT-positive cells in the sham (P = 0.051), vehicle-treated TBI (P = 0.332), or
In the ipsilateral hippocampus, there was no main effect of treatment (F2,64 = 1.85; P = 0.166) or time (F2,64 = 0.83; P = 0.441). However, in the contralateral hippocampus, a significant main effect of treatment (F2,64 = 3.82; P = 0.028) but not time (F2,64 = 1.16; P = 0.321) was observed. Post hoc analysis revealed that animals treated with
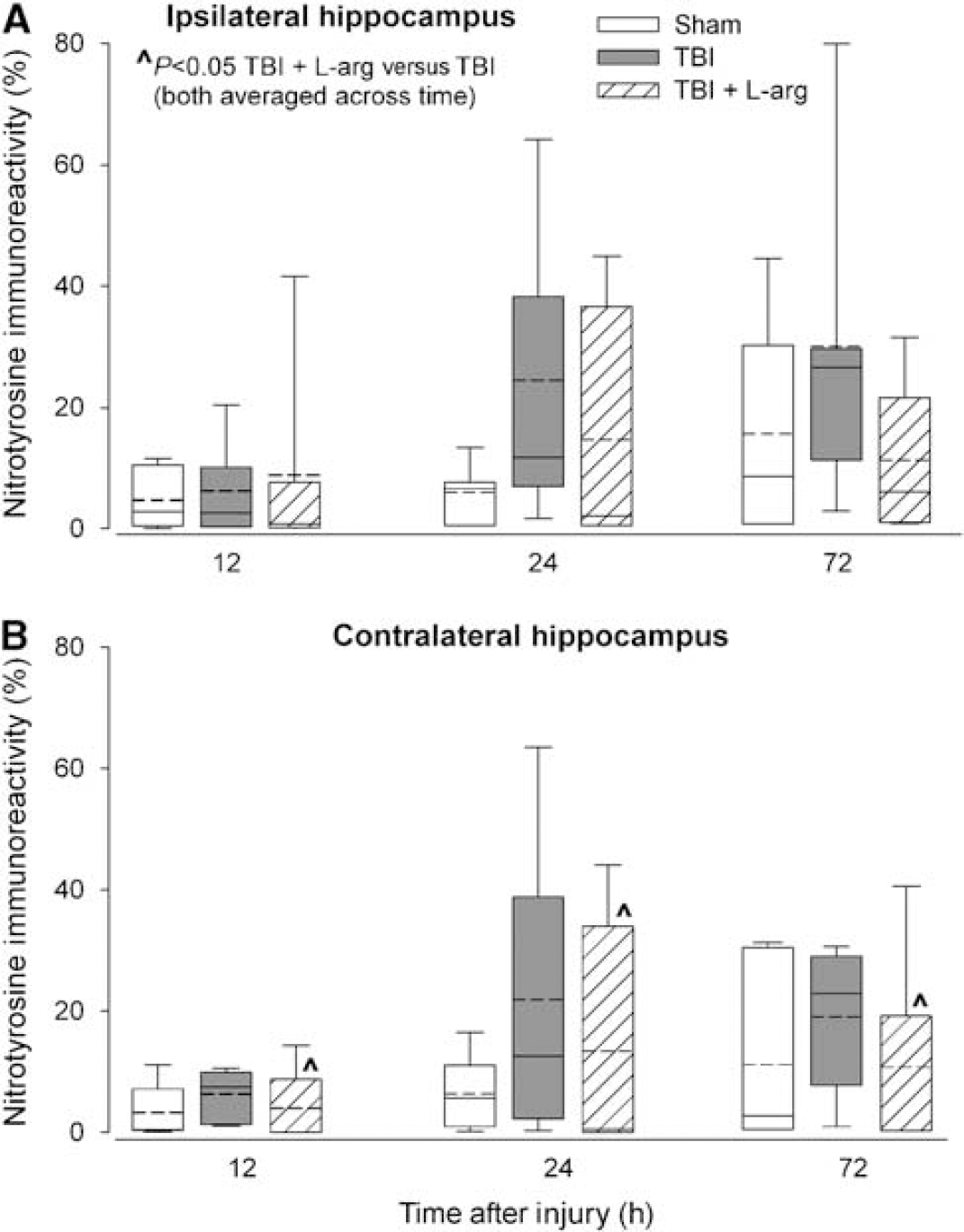
Percentages of NT-immunoreactive neurons in the ipsilateral (
Discussion
This is the first quantitative report of the time course of changes in NT immunoreactivity after fluid-percussion TBI in rats, and the first demonstration that treatment with
Our results are consistent with previous reports in regard to NT formation after traumatic injury in the central nervous system. In the brain, fluid-percussion injury in rats produced qualitative increases in NT immunoreactivity in the corpus callosum 30 mins and 2 h after injury, as well as in the temporoparietal cortex 4 h to 72 h after injury (Besson et al, 2003). Controlled cortical impact TBI followed by hyperoxic ventilation in rats also resulted in significant increases in the number of NT-positive neurons in hippocampal layers CA1 and CA3, 24 h after injury (Ahn et al, 2005). In mice, time-dependent increases in NT staining (Hall et al, 2004; Mesenge et al, 1998) or expression (Bayir et al, 2005) have also been observed after TBI. Furthermore, in human brains, autopsy revealed the presence of NT-immunoreactive neurons after TBI (Forster et al, 1999). Likewise, in the spinal cord, traumatic injury increased NT protein expression (Xu et al, 2001), the number of NT-immunoreactive neurons, and extracellular NT levels (Liu et al, 2000). Clearly, NT expression increases after different types of neuronal injury in rodents as well as humans.
After fluid-percussion TBI in rats, constitutive NOS activity increased significantly at the lesion site (Wada et al, 1998) and direct measurements of changes in brain tissue NO levels revealed significant, transient NO increases after controlled cortical impact (Cherian et al, 2000) or parasagittal fluid-percussion TBI in rats (Ahn et al, 2004). In a closed head injury model, the nonspecific NOS inhibitor
Alternatively, the reduction in the number of NT-positive neurons in the cortex and hippocampus with
Other explanations for
Although it may seem unlikely that a single treatment with
This shift in cerebral metabolic activity toward energy production through alternative pathways after TBI may be associated with changes in gene expression and/or influenced by antioxidant activity. For example, reduced levels of NAD+ and ATP after TBI may be because of the activation of the gene for poly(ADP-ribose) polymerase-1 (PARP-1) an enzyme that repairs DNA strand breaks. It has been shown that TBI results in the activation of PARP-1 (LaPlaca et al, 1999; Ha and Snyder, 2000). However, activation of PARP-1 may deplete NAD+, decrease the rate of electron transport and ATP formation, and eventually result in energy failure and cell death (Endres et al, 1997). Poly(ADP-ribose) polymerase-1 can be activated by superoxide anion radicals and ONOO− (Ha and Snyder, 2000), both of which are produced immediately after TBI (Fabian et al, 1995; Kontos and Wei, 1986). It is possible that NO produced by
The detection of NT-immunoreactive neurons in both ipsilateral and contralateral hemispheres (relative to the injury site) is because of the bilateral nature of parasagittal fluid-percussion TBI. Using parasagittal (injury site 2 to 3 mm from midline) fluid-percussion injury, others and we have shown bilateral consequences of injury. Neuronal counting at 15 days after injury showed bilateral decreases in neuronal survival, whereas treatment with hypertonic
The accuracy of our findings may be influenced by two general categories of limitations. First, limitations related to the accuracy of neuronal counting and second, limitations related to the specificity and accuracy of NT as a marker for ONOO−. Neuronal counting may be influenced by morphologic damage at the injury site, making the identification of neurons by morphologic criteria difficult and possibly resulting in an underestimation of the number of NT-positive neurons. Definitive differentiation of NT-positive neuronal from nonneuronal cells requires the use of double-labeling with specific neuronal antibodies (e.g., Neu-N), which was not performed in this study. Secondly, NT immunoreactivity may not be highly specific to the effects of ONOO−. Rather, NT immunoreactivity is likely a marker of ONOO−-related tyrosine nitration in vivo (Beckman et al, 1990). The formation of NT detected with myeloperoxidase (Eiserich et al, 1998) and eosinophil peroxidase (Duguet et al, 2001) suggests that tyrosine nitration is an indicator of reactive nitrogen species in general rather than ONOO− in particular.
The enzyme myeloperoxidase is secreted by activated neutrophils, and uses peroxide and chloride ions to produce hypochlorous acid (HOCl), an antibacterial agent. Under conditions when there is concomitant ONOO− and HOCl formation (e.g., sites of inflammation), HOCl may remove NT (Whiteman and Halliwell, 1999). Traumatic brain injury produces such sites of inflammation. In humans, TBI has been shown to induce a strong, locally restricted inflammatory response in which activated neutrophils infiltrate the site of tissue destruction (Bank et al, 1999). Therefore, the production of HOCl is possible in this scenario, making this mechanism another potential source of inaccuracy in our study. However, although multiple reactive pathways have been shown to effectively increase NT residues in vitro, only few have been replicated in vivo (Ischiropoulos, 1998). Interestingly, nitryl chloride, a potential nitration agent formed from the reaction between nitrite and neutrophil myeloperoxidase-derived HOCl, contributed minimally to the formation of intracellular NT in vivo (Whiteman et al, 2003), confirming that the NT immunoreactivity we detected originated from ONOO−.
In summary, we showed that parasagittal fluid-percussion TBI in rats resulted in time-dependent, significant increases in the number of NT-immunoreactive neurons bilaterally in the cerebral cortex and hippocampus. Post-injury treatment with
Footnotes
Acknowledgements
The research described in this article was supported in part through National Institutes of Health grant no. 5R01NS019355, Effects of O2 Radicals and Perivascular Nerves in Trauma, Douglas S DeWitt, PhD, principal investigator.
The authors state no conflict of interest.
