Abstract
Hypothermia is beneficial in adult models of traumatic brain injury (TBI), but it has not been evaluated in an immature animal model. We hypothesized that brief hypothermia applied after TBI would reduce cerebral edema and lesion volume in immature rats. Male Wistar rats (3–4 weeks of age, 90–140 g) were anesthetized, intubated, mechanically ventilated, and subjected to TBI by a weight drop onto the exposed right parietal cortex. Hypothermic rats were then cooled to a brain temperature of 32.0 ± 0.5°C for 4 h, and control rats were maintained at a brain temperature of 37.0 ± 0.5°C. Cerebral edema (wet — dry weight method) was assessed at 4 and 24 h, and lesion volume was assessed at 5 days. At 4 h, a reduction of percent brain water in the traumatized hemisphere was observed in hypothermic versus normothermic rats (81.75 ± 0.60 vs. 82.53 ± 0.67%; p < 0.05), but by 24 h posttrauma, the groups were similar (p = 0.82). Total lesion volume (47.2 ± 8.5 vs. 44.4 ± 10.0 mm3; p = 0.51) and necrotic volume (20.2 ± 6.3 vs. 20.0 ± 7.9 mm3; p = 0.95) were similar in the hypothermic and normothermic groups. We conclude that in this model, a transient (4-h) application of moderate (32°C) hypothermia reduces the cerebral edema characteristically seen in immature rats at 4 h, but this reduction is not sustained at 24 h. Attenuating or delaying the development of cerebral edema could have important therapeutic relevance after TBI. Transient hypothermia, however, did not reduce lesion volume at 5 days posttrauma.
Hypothermia after traumatic brain injury (TBI) has been studied in adult animal models. In a series of experiments in the 1950s and 1960s, hypothermia applied after brain injury (freeze lesion) reduced cerebral edema, mortality, and cerebrospinal fluid pressure (Rosomoff and Gilbert, 1955; Rosomoff, 1959; Shulman and Rosomoff, 1959; Clasen et al., 1968). More recently, adult rats treated with transient hypothermia (32°C for 1 h) beginning 5 min after TBI (fluid percussion) exhibited better functional outcome (beam walking and beam balance) than normothermic controls (Clifton et al., 1991). Further work with this model showed that hypothermia was effective if begun within 15 min, but not 30 min, posttrauma (Lyeth et al., 1993). In a controlled cortical impact model in adult rats with a moderate injury, transient (2-h) hypothermia (32°C) initiated before trauma reduced lesion volume at 2 weeks (Palmer et al., 1993). Similarly, with use of a fluid percussion model of TBI, posttraumatic application of moderate hypothermia (30°C for 3 h) led to less histologic damage at 3 days than in normothermic rats (Dietrich et al., 1994). These studies suggest that in adult models of TBI, a brief posttraumatic application of mild or moderate hypothermia attenuates secondary injury. The mechanism(s) for the beneficial effect of hypothermia is unknown, although Taft et al. (1993) reported that hypothermia after fluid percussion TBI in adult rats prevents the loss of microtubule-associated protein 2, an important component of the neuronal cytoskeleton. To our knowledge, however, no study has investigated the effect of hypothermia after TBI in an immature animal preparation.
Clinical studies support the concept that there are important age-related differences in the response to TBI. Diffuse brain swelling occurs more often in children than in adults after TBI (Bruce et al., 1981; Aldrich et al., 1992), and children with this finding have a threefold higher mortality rate than those without it (Aldrich et al., 1992). Obrist et al. (1984) reported that after severe head injury, the incidence of hyperemia was greater in young adults (70%) than in older adults (38%), as did Langfitt et al. (1977). Similarly, in a recent study, Muizelaar et al. (1989) reported that 88% of children had hyperemia at some point after severe TBI.
The response to TBI in experimental models also differs in immature versus mature animals. Recently, we reported that immature (3-week-old) rats develop cerebral edema more rapidly than do mature rats (3–4 months old) after weight drop TBI (Grundl et al., 1994). In addition, in the same model, immature rats exhibited more hyperemia than either mature or aged (14-month-old) rats at 24 h posttrauma (Biagas et al., 1992).
Although hypothermia has proven to be beneficial in several adult models of TBI, this therapy has not been tested in an experimental model of TBI in immature subjects. Given the differences between mature and immature subjects in their response to TBI, we felt that it would be important to evaluate this therapy in immature animals. Since children and immature animals seem to have more swelling and hyperemia after TBI, if hypothermia exerts its beneficial effects by affecting either of these deleterious posttraumatic processes, then it may be of particular benefit in these subjects. We hypothesized that a brief (4-h) period of mild to moderate brain hypothermia (32°C) would reduce brain edema and lesion volume in our established model of TBI in the immature rat.
MATERIALS AND METHODS
Animal preparation
Immature male Wistar rats, 3–4 weeks of age and weighing between 90 and 140 g, were anesthetized with halo thane (5% for induction and intubation, 1% for insertion of vascular catheters, then 0.4% for trauma and maintenance) in 70% N2O (balance O2). After intubation, rats were mechanically ventilated. Benzathine penicillin (36,000 U; Upjohn, Kalamazoo, MI, U.S.A.) and gentamicin (2 mg; Elkins-Sinn, Cherry Hill, NJ, U.S.A.) were administered intramuscularly to prevent iatrogenic infection. A femoral arterial catheter was surgically placed for withdrawal of blood samples, administration of medications, and continuous monitoring of MABP. Pancuronium bromide (0.4 mg; Elkins-Sinn) was administered hourly to achieve muscle paralysis to prevent accidental extubation and thermogenesis and to facilitate mechanical ventilation. With the torso wrapped in a plastic bag and the head affixed in a stereotaxic holder, the rat was placed prone in a small plastic bath.
Brain temperature and Paco2 were controlled, and other physiologic variables were monitored. A brain temperature probe (0.009-in diameter; Physi-Temp, Clifton, NJ, U.S.A.) was placed stereotaxically through a small burr hole in the parietal bone to a depth of 2 mm into the left (noninjured) hemisphere. Arterial blood gases were measured before trauma and at 1 and 3 h posttrauma. The target range for arterial Paco2 was between 35 and 45 mm Hg, and the target for arterial Pao2 was >80 mm Hg. Paco2 was controlled by adjusting tidal volume and/or ventilatory rate. Blood gases were not corrected for temperature [i.e., alpha-stat mode was used (Ream et al., 1982)]. Rectal temperature and MABP were continuously monitored. Hematocrit and plasma glucose were measured before trauma and at 3 h posttrauma.
Trauma
A small craniotomy (4 × 5 mm) was made over the right parietal cortex with a dental drill, and the bone flap was left in place. The rat was allowed to stabilize for 30 min at a brain temperature of 37.0 ± 0.5°C. The bone flap was then removed and the trauma was performed by the following method: A hollow glass cylinder was stereotaxically positioned above the exposed cortex, in a vertical orientation. A brass rod 3 mm in diameter and weighing 10 g was dropped through the glass cylinder from a height of 4.2 cm above the cortex. This technique has been used in immature rats in our laboratory (Grundl et al., 1994) and produces a consistent injury and reliable edema with long-term survival. The bone flap was replaced and sealed with dental cement, and the scalp was partially resutured, leaving a small opening for the brain temperature probe.
Hypothermia and normothermia treatment protocol
After trauma, rats were randomized to the hypothermia (treatment) or the normothermia (control) groups. Beginning at 10 min posttrauma, rats in the hypothermic group had ice packs placed on their head, and ice water was poured into the plastic container. The target brain temperature of 32.0 ± 0.5°C was achieved within 10 min (by 20 min posttrauma). The ice packs were then removed, and the ice water bath was drained. Brain temperature was then maintained at 32.0 ± 0.5°C for 4 h with a fan and by adjusting the temperature of the water in the bath as needed. Rectal temperature was generally between 32 and 34°C in the hypothermic group. The brain temperature of rats in the normothermic control group was maintained at 37.0 ± 0.5°C using warm water in the bath or a heat lamp as needed. In both groups, we attempted to maintain the desired brain temperature primarily through manipulation of the body temperature. However, if body temperature exceeded the predetermined acceptable range and brain temperature had not reached the target temperature, then direct brain cooling (fan or ice pack if necessary) or warming method (heat lamp) was used.
Two study protocols were used. In one protocol, cerebral edema was assessed at 4 or 24 h posttrauma. In the other protocol, lesion volume was assessed at 5 days posttrauma. Rats in which cerebral edema was measured at 4 h posttrauma (hypothermia, n = 8; normothermia, n = 7) were decapitated at the end of the 4-h hypothermic (without rewarming) or normothermic period. Rats in which either 24-h edema (hypothermia, n = 4; normothermia, n = 4) or 5-day lesion volume (hypothermia, n = 9; normothermia, n = 12) was measured were gradually rewarmed over ∼1.5 h at the end of the 4-h treatment interval. When rewarming the rat, we aimed for a gradual transition, because MABP decreased with rewarming. At the start of the rewarming period, we allowed the temperature to rise passively, with minimal intervention, although some intervention (warm bath water, heat lamp) was required in all cases. Rats in the normothermic group were also maintained on mechanical ventilation and anesthesia for an additional 1.5 h to mimic the total duration of mechanical ventilation and anesthesia used in the hypothermic rats. The femoral arterial catheter and temperature probes were then removed, the scalp was completely closed, and the rats were extubated, maintained on supplemental O2 for 30 min, and then returned to their cages.
Cerebral edema
Each rat was decapitated at 4 or 24 h posttrauma, and the brain was immediately removed. A 3-mm-thick coronal section was made through the brain (including all structures—cortex, thalamus, midbrain, etc.) at the level of the trauma using a rat brain sheer (Zivic Miller, Zelienople, PA, U.S.A.), and this section was divided into right (injured) and left (noninjured) halves. (This section, viewed en face, resembles Fig. 5, except it was 3 mm thick rather than 40 p.m). The percent brain water in each section was determined by the wet — dry weight method. Briefly, the 3-mm sections were weighed fresh, desiccated at 110°C for 48 h, then reweighed. Sections were kept in a capped glass vial to minimize evaporative loss. We previously demonstrated that edema does not develop in the left (noninjured) hemisphere in either mature (Schoettle et al., 1990) or immature (Grundl et al., 1994) rats after weight drop trauma of this severity.
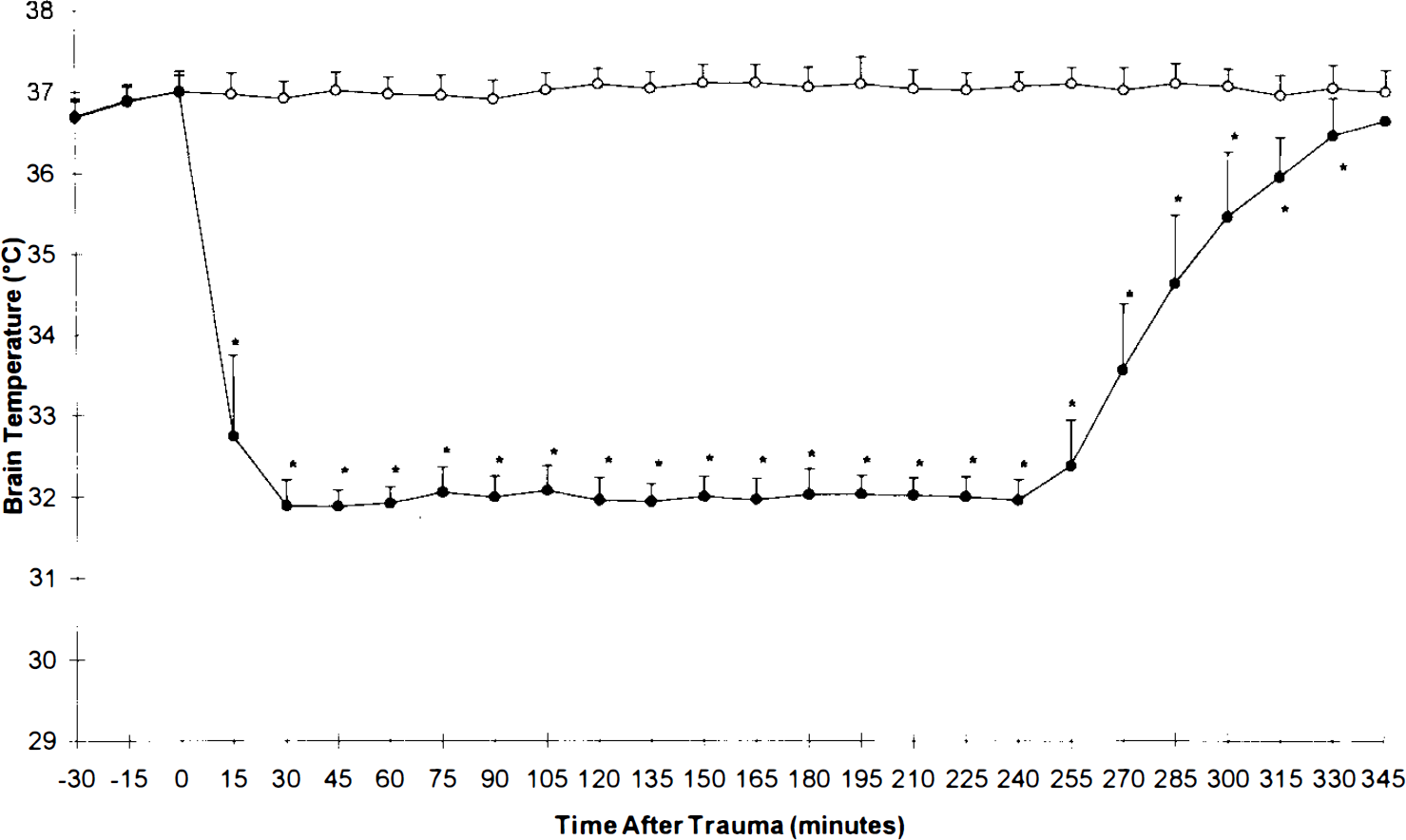
Graph depicting brain temperature versus time posttrauma in normothermic (open circles) and hypothermic (filled circles) groups. *p < 0.05 hypothermia versus respective normothermia value by two-way analysis of variance for repeated measures.
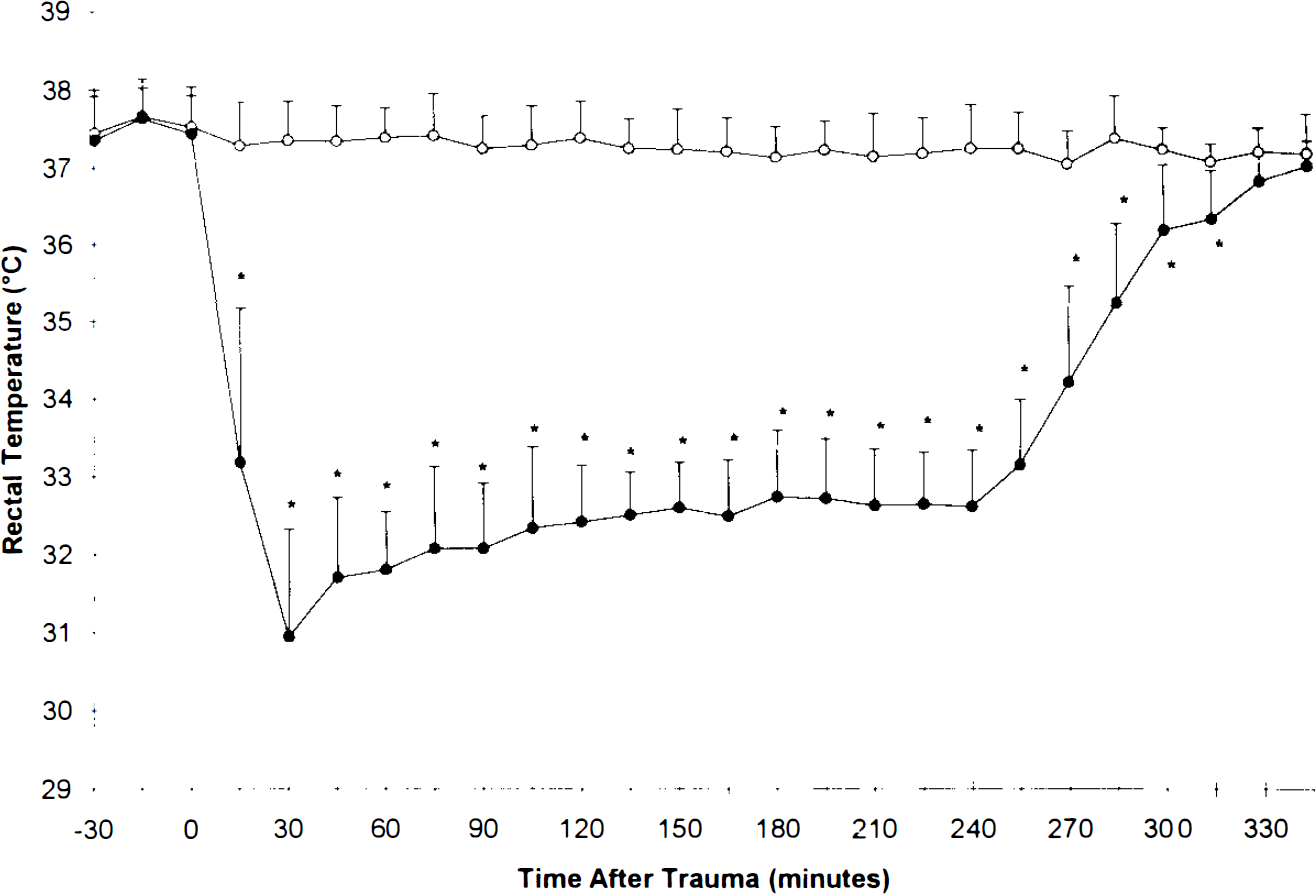
Graph depicting rectal temperature versus time posttrauma in normothermic (open circles) and hypothermic (filled circles) groups. *p < 0.05 hypothermia versus respective normothermia value by two-way analysis of variance for repeated measures.
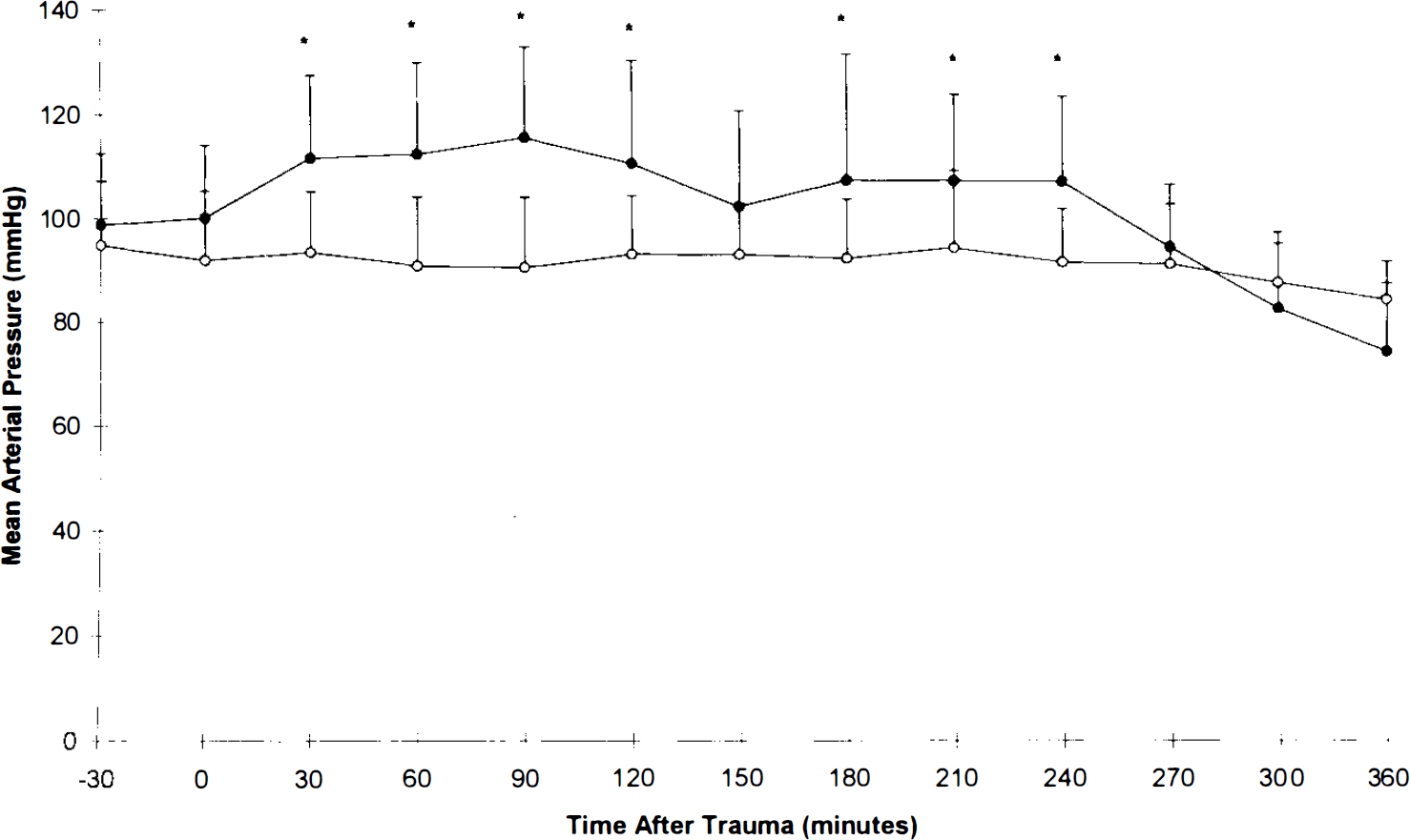
Graph depicting MABP versus time posttrauma in normothermic (open circles) and hypothermic (filled circles) groups. *p < 0.05 hypothermia versus respective normothermia value by two-way analysis of variance for repeated measures.
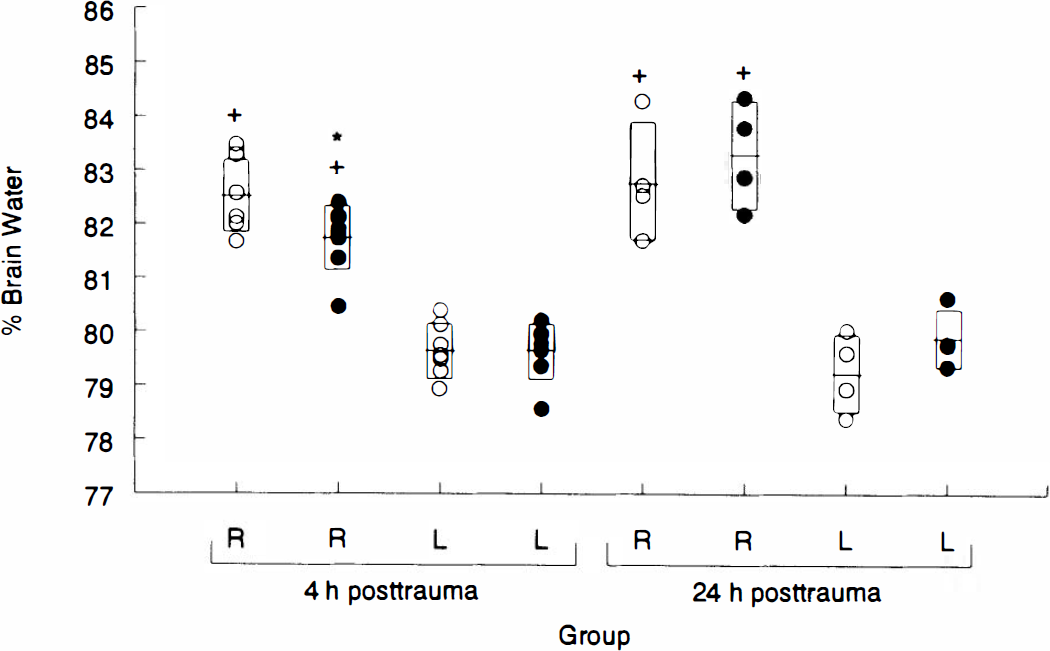
Graph depicting the effect of 4 h of hypothermia on percent brain water at 4 and 24 h after traumatic brain injury in immature rats. Percent brain water was increased in the injured right (R) hemisphere versus the noninjured left (L) hemisphere in both normothermic (open circles) and hypothermic (filled circles) groups at both 4 and 24 h posttrauma (+p < 0.05 for either R hemispheric value versus either L hemispheric value at a given time posttrauma, by one-way analysis of variance and Student-Newman-Keul test). At 4 h, percent brain water was reduced in the injured R hemisphere in hypothermic versus normothermic rats (*p < 0.05 by one-way analysis of variance and Student-Newman-Keul test). There was no difference in percent brain water in the injured R hemisphere between hypothermic and normothermic groups at 24 h. There was no difference between groups for percent brain water in the L hemisphere at any time posttrauma. Individual data points are shown. The horizontal bar and vertical rectangle represent mean ± SD, respectively.
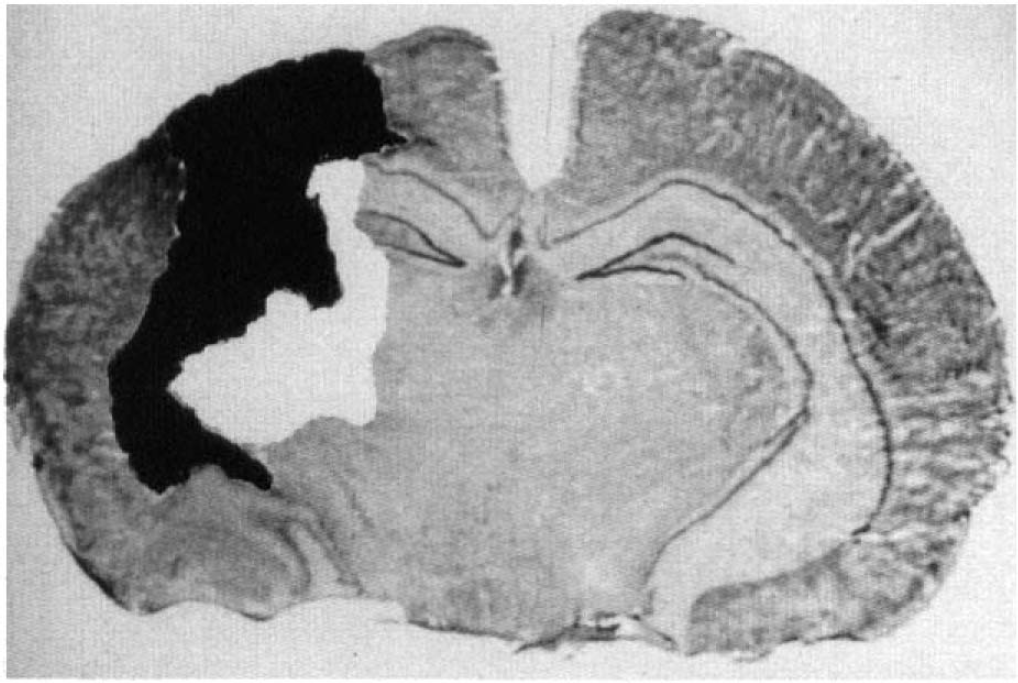
Representative image-processed coronal section through the center of the impact site from a rat at 5 days posttrauma. The necrotic area is outlined in black. Total lesion area is given by sum of the white (injured but not necrotic) and black outlined areas.
Lesion volume
Rats were decapitated at 5 days posttrauma, and the brains were immediately removed and frozen. Coronal brain sections (40-μm thickness) were sliced at 1-mm intervals from the frontal pole to the occiput and mounted on slides. (Each brain was thus represented by a set of 11 slides comprised of sections from each 1-mm interval.) Two sets of slides were prepared from each brain: One set was stained for Nissl substance with thionin (Carson, 1980) and the other with hematoxylin and eosin. For each slide (i.e., each 1-mm interval), both the necrotic area and the total lesion area were measured using computerized image analysis (Bioscience Research Software; Imaging Research, St. Catherines, Ontario, Canada) and a microscope. Necrotic area was defined as tissue that was either necrotic or absent, whereas total lesion area included necrotic areas as well as injured but intact areas containing cellular infiltrates or hemorrhage. Necrotic or total lesion volume of each brain was calculated by adding the respective lesion areas for all 11 sections from each brain. Slides were analyzed independently by two observers who were blinded to the treatment.
Statistics
All data (physiologic variables, edema, lesion volumes) are reported as means ± SD. Body weight, arterial blood gases, glucose, hematocrit, and lesion volume were compared using a two-tailed Student t test. Percent brain water was compared between and within groups at 4 and 24 h posttrauma using a one-way analysis of variance and the Student–Newman–Keul test. Continuous variables (brain and rectal temperatures and MABP) were compared using a two-way analysis of variance for repeated measures. Interobserver correlation and correlation between staining methods were assessed by linear regression analysis, p < 0.05 was regarded as significant.
RESULTS
The rats generally tolerated both the hypothermia (n = 23) and the normothermia (n = 25) conditions. There were two deaths in each group. Survivors resumed apparently normal activity (with a mild degree of right hindlimb paresis due to sacrifice of the artery) soon after extubation. Brain and rectal temperatures differed significantly between the two groups (Figs. 1 and 2) during both the entire hypothermia interval and most of the rewarming period. The hypothermic group had a higher MABP, but the normothermic group was not hypotensive (Fig. 3). The hypothermic group had a trend toward decreased MABP during rewarming, but this did not reach significance. The groups differed in pH, Paco2, and Pao2 at 1 h posttrauma (p < 0.01), but not before trauma or at 3 h posttrauma (Table 1). Differences between groups in blood glucose and hematocrit were observed at 3 h posttrauma (Table 1).
Physiologie data
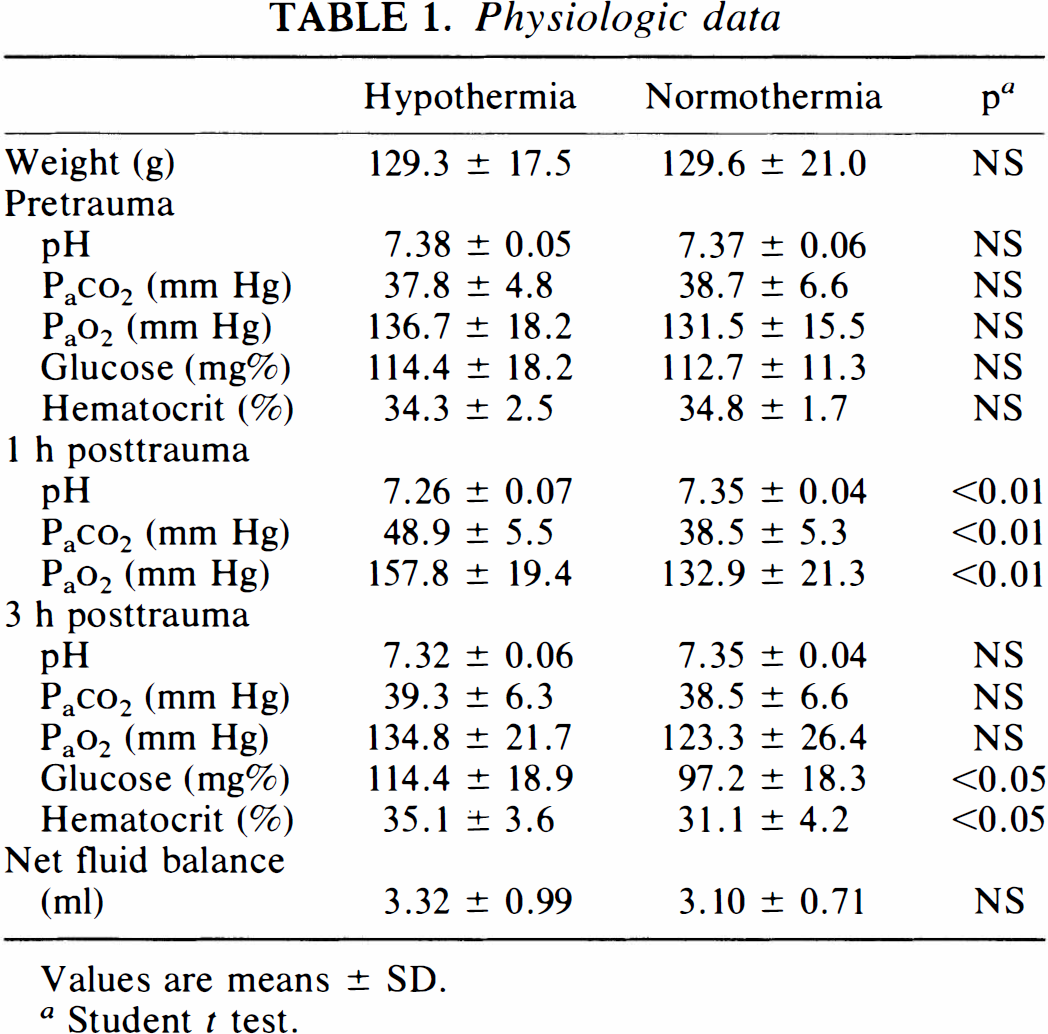
Values are means ± SD.
Student t test.
Cerebral edema
Percent brain water was increased in the injured right hemisphere in both normothermic and hypothermic groups at both 4 and 24 h posttrauma (all p < 0.05 vs. respective noninjured left hemisphere) (Fig. 4). Percent brain water in the injured hemisphere differed between normothermic and hypothermic groups at 4 h posttrauma (82.53 ± 0.67 vs. 81.75 ± 0.60%, respectively; p < 0.05) (Fig. 4). However, at 24 h posttrauma, no difference in percent brain water was observed between the normothermic and hypothermic groups.
Lesion volume
A 40-μm coronal section demonstrating necrotic and total lesion areas is shown in Fig. 5. The hypothermic and normothermic groups were similar in both necrotic volume (20.2 ± 6.3 vs. 20.0 ± 7.9 mm3, respectively; p = 0.95) (Fig. 6) and total lesion volume (47.2 ± 8.5 vs. 44.4 ± 10.0 mm3, respectively; p = 0.51) (Fig. 7) at 5 days posttrauma. Significant correlations were found for both necrotic and total lesion volumes (between observers) and for lesion volumes measured in thionin- and hematoxylin and eosin-stained sections (for same observer). All r2 values were between 0.94 and 0.97. Data reported represent lesion volumes for thionin-stained sections.
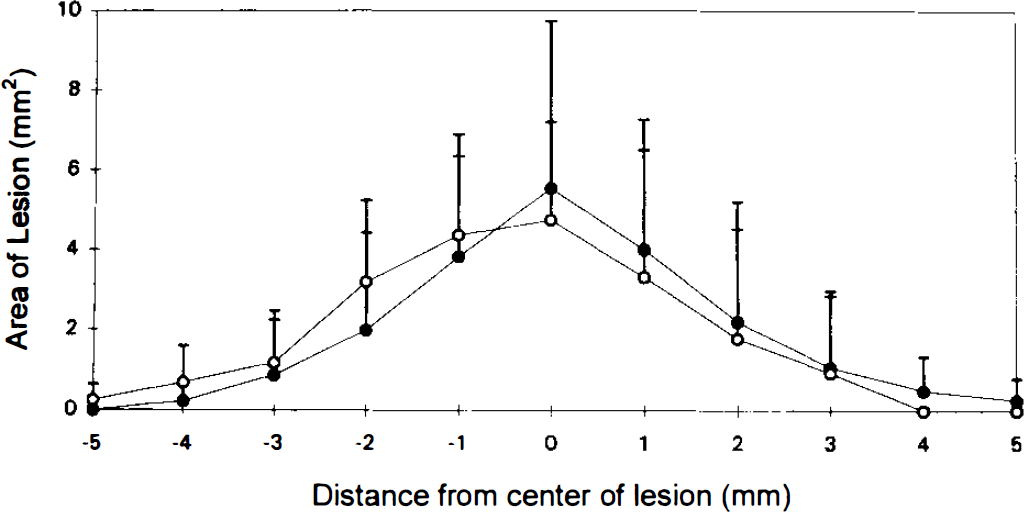
Graph depicting necrotic area of the lesion plotted as a function of distance from the lesion center measured in serial sections through the injured hemisphere from rats at 5 days posttrauma. Necrotic lesion volumes were calculated as the sum of the individual areas in the normothermia (open circles) and hypothermia (filled circles) groups (volume equals area under the curve). There was no difference between groups. Values are means ± SD.
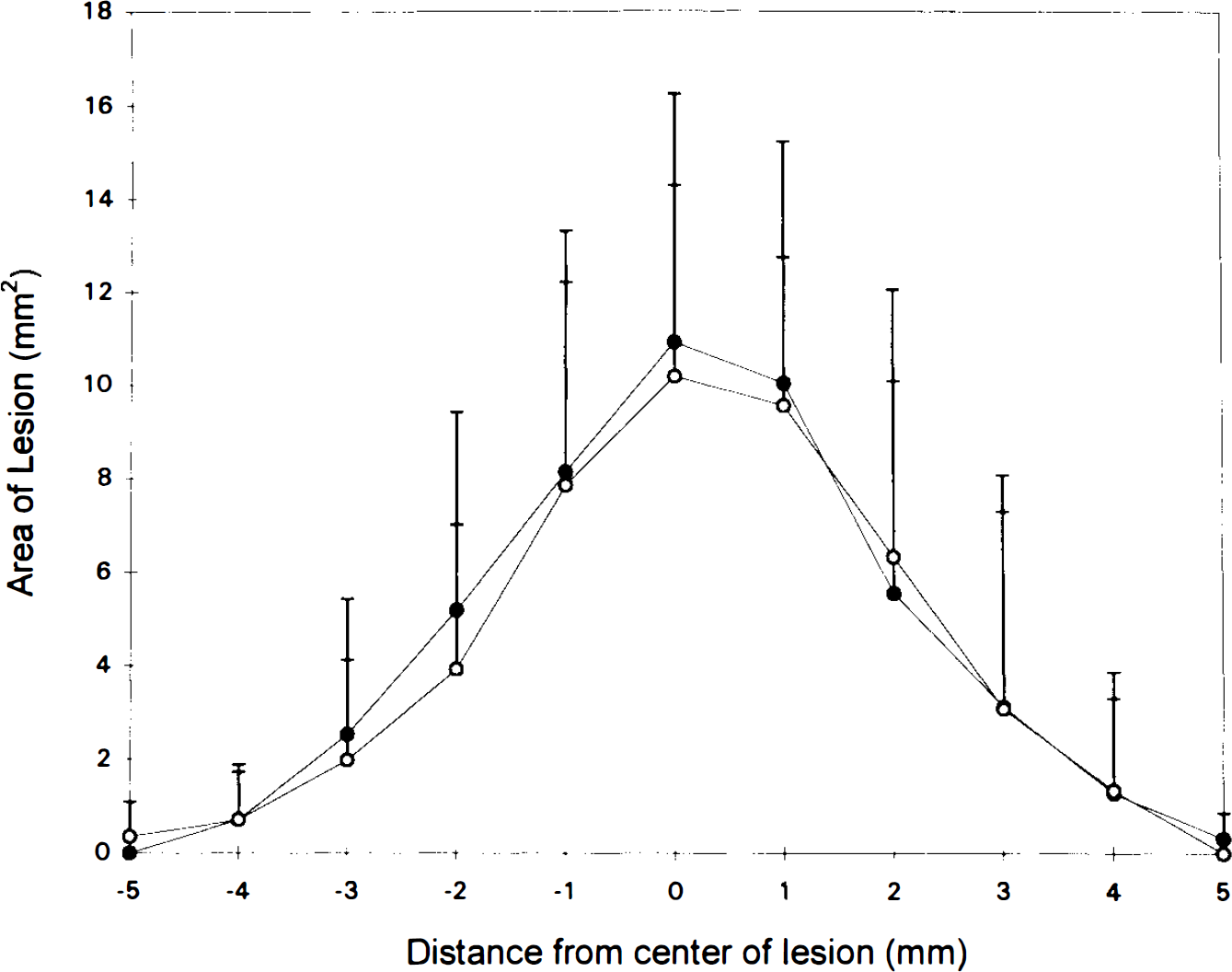
Graph depicting total lesion area plotted as a function of distance from the lesion center measured in serial sections through the injured hemisphere from rats at 5 days posttrauma. Total lesion volumes were calculated as the sum of the individual areas in the normothermia (open circles) and hypothermia (filled circles) groups (volume equals area under the curve). There was no difference between groups. Values are means ± SD.
DISCUSSION
In this model of TBI in immature rats, brief moderate brain hypothermia (32°C, applied posttrauma) reduced cerebral edema at 4 h. This reduction was not sustained, however, at 24 h. In addition, no difference was seen between the hypothermic and normothermic groups in lesion volume at 5 days. The posttraumatic application of hypothermia thus delays, rather than prevents, the development of cerebral edema in this model of TBI in immature rats. This finding is consistent with the results of several other studies on the effects of the transient application of mild or moderate hypothermia after cerebral ischemia or trauma in adult models. Postischemic hypothermia (30°C for 3 h) dramatically protected hippocampal neurons assessed at 3 days after transient incomplete global forebrain ischemia in rats. By 2 months postischemia, however, this protective effect was absent, suggesting that hypothermia had delayed, rather than prevented, ischemic neuronal injury in this selectively vulnerable brain region (Dietrich et al., 1993). Similar findings have been reported in some experimental models of TBI in mature animals. Clifton et al. (1991) reported that adult rats treated with 1 h of hypothermia (30 or 32°C) after TBI by fluid percussion had faster recovery of function during the first 2 days after injury, but the control group had the same degree of recovery by the third day posttrauma. Pomeranz et al. (1993) reported that after epidural compression in adult dogs, 5 h of moderate hypothermia (31°C) delayed the increase in intracranial pressure compared with that in normothermic controls. During a subsequent period of mild hypothermia (35°C), however, progressive increases in intracranial pressure were observed, and no overall beneficial effect on the incidence of herniation or death was afforded by hypothermia (although lesion volume was reduced). Taken together with our findings, these studies suggest that hypothermia delays, rather than prevents, the evolution of damage after TBI.
In contrast to our results and the aforementioned studies, transient hypothermia does cause a sustained reduction of histologic damage after TBI in some experimental models. Palmer et al. (1993) reported that 2 h of moderate hypothermia reduced lesion volume in adult rats measured at 2 weeks posttrauma. In reports by Palmer et al. (1993) and Dietrich et al. (1994), the beneficial effects of moderate hypothermia (applied posttrauma) on lesion volume were observed in models with less severe injuries than those produced in our model. For example, in the study by Palmer et al. (1993), the lesion volume observed after TBI was less than one-third of that observed in our model. The insult in our model is severe and may have exceeded the capacity of hypothermia (or any therapy) to influence either necrotic or total lesion volume. Nonetheless, the reduction in cerebral edema at 4 h suggests that an important outcome parameter can be therapeutically altered in our model.
Perhaps a longer duration of hypothermia would more permanently attenuate the development of cerebral edema or influence ultimate lesion volume. The optimal depth and duration of posttraumatic hypothermia have not been defined in clinical or experimental TBI. Pomeranz et al. (1993) reduced lesion volume by applying hypothermia for >60 h. Similarly, clinical studies suggest that moderate systemic hypothermia (32°C) is well tolerated for 24 or 48 h, reduces intracranial pressure (Marion et al., 1993; Shiozaki et al., 1993) and seizures (Clifton et al., 1993), and may improve survival (Shiozaki et al., 1993) after head injury. Most investigators working in adult models of experimental ischemic and TBI have used transient (1- to 4-h) hypothermia, and formidable technical difficulties might be encountered in applying moderate hypothermia for 24 or 48 h in a rodent model (particularly in immature rats weighing between 90 and 140 g). Therefore, we chose a 4-h treatment interval for this initial study in an immature model of TBI.
Immature response to TBI
There are important differences between mature and immature subjects in the response to TBI, and these age-related differences could contribute to the inability of hypothermia to influence lesion volume. Two clinical studies have demonstrated a greater diffuse brain swelling in response to TBI in children than in adults (Bruce et al., 1981; Aldrich et al., 1992). Although this phenomenon is not completely understood, it often develops rapidly and is frequently noted on the initial posttrauma computed tomographic scan (Aldrich et al., 1992). Several mechanisms may be involved in this process, including greater posttraumatic hyperemia in children than in adults (Obrist et al., 1984; Muizelaar et al., 1989; Biagas et al., 1992), a more rapid or greater diffusion of excitotoxins or other metabolites through the immature than through the mature brain parenchyma (Verheul et al., 1993), and/or more rapid development of cerebral edema in immatures (Grundl et al., 1994). We recognize that our model is one of focal brain injury and the mechanisms involved in the development of brain swelling may not completely mimic those involved in diffuse brain swelling. To our knowledge, a model of diffuse TBI in immature rats has not been published.
Certain biochemical features are also unique to the immature brain. Immature animals have a greater density of N-methyl-
We did not, however, anticipate (nor are we convinced) that hypothermia would be less effective after TBI in immatures than in adults. The most dramatic clinical examples of hypothermic protection of the brain from hypoxic–ischemic insults (i.e., cold water drowning) have been reported in children (Young et al., 1980). Intraischemic hypothermia also improves neuropathologic outcome (Yager et al., 1993), decreases mortality, decreases edema, and preserves high-energy phosphates in immature rats (Young et al., 1983). Hypothermia, therefore, is clearly effective in immature animals in certain settings. The ability of hypothermia to delay the development of edema, as we found in this study, is particularly relevant to the immature. Recent work by Lin et al. (1994) suggests that transient mild hypothermia expands the therapeutic time window for the use of other pharmacologic agents such as N-methyl-
Physiologic derangements
Differences in MABP were observed between groups. Hypothermic rats had a higher MABP than normothermic rats until rewarming, although the normothermic group was not hypotensive. It could be argued that this may have favored vasogenic edema formation in the hypothermic group and attenuated some portion of the beneficial effect on posttraumatic edema. Alternatively, hypertension may have improved CBF in injured regions with impaired autoregulation, thereby reducing ischemia in the early posttrauma period (Grundl et al., 1994). We suspect that peripheral vasoconstriction secondary to hypothermia increased MABP. However, the impact of this unavoidable effect of hypothermia on either edema or lesion volume is unclear.
Steen et al. (1980) noted cardiovascular collapse with severe tissue hypoxia and acidosis during rewarming of dogs after 48 h of hypothermia. We did not observe a significant difference in MABP between treatment groups during rewarming; however, we did not monitor MABP for an extended period after rewarming was complete. It is possible that rewarming may have produced deleterious effects in a more protracted fashion. We removed arterial catheters and weaned the rats from mechanical ventilation to avoid the risks of complications from very prolonged mechanical ventilation, anesthesia, and vascular catheterization, particularly in immature rats. Even without extending the monitoring period, the rats were already intubated and monitored for a period of nearly 8 h. In defense of this approach, only two rats died in each group. In addition, the two rats that died in the hypothermic group died during hypothermia, not during or after rewarming.
There were significant differences in the arterial blood gas values at 1 h posttrauma. The hypothermic group had better oxygenation, but worse ventilation (i.e., higher Paco2) than the normothermic group. We did not correct for temperature effect on blood gases [we used alpha-stat mode (Ream et al., 1982)]. We could postulate that the water surrounding the rat exerted a restrictive effect on pulmonary function and impaired ventilation. Whatever the reason for the differences, they were transient. The ventilator was adjusted immediately after obtaining the blood gases at 1 h posttrauma, and the gases were similar at 3 h. We cannot rule out a possible deleterious effect of mild hypercarbia in the early posttrauma period.
There were also differences in the hematocrit and glucose at 3 h posttrauma. The difference in blood glucose between groups at 3 h was significant, but was very modest (97.2 vs. 114.4 mg% in normothermic and hypothermic rats, respectively). Increased blood glucose in the hypothermic group may have resulted from a stress response to the hypothermia. The higher hematocrit in the hypothermic group may be the result of hemoconcentration, due to either the higher MABP increasing urine output or cold-induced diuresis. We carefully recorded fluid removed and administered through the arterial line (not different between groups; Table 1); we did not record urine output. We cannot rule out an influence of these mechanisms on our findings.
We recognize that hypothermia and rewarming may have adversely altered electrolytes. We did not measure electrolytes because of concerns about extensive phlebotomy. Each of these tests requires a quantity of blood that is a substantial fraction of the total blood volume in the immature rat.
CONCLUSIONS
A 4-h period of mild hypothermia applied after TBI delayed the characteristically rapid development of posttraumatic cerebral edema in the immature rat; however, lesion volume was not reduced at 5 days. This transient effect of hypothermia in the immature rat is consistent with some previous studies of hypothermia in models of both ischemic and TBI in mature animals. Further studies of the effect of hypothermia on the response of the immature rat to TBI are warranted.
Footnotes
Acknowledgment:
The authors thank Dr. Steven DeKosky and Scot Styren for their helpful input. We thank Drs. Peter Safar, Steven DeKosky, and Donald Marion for reviewing, Linda Ryan, Marci Provins, and Arlene Toledo for preparing, and Francie Siegfried for editing this manuscript. This study was supported by a Seed Grant from the Department of Anesthesiology and Critical Care Medicine, University of Pittsburgh. Dr. Kochanekis supported in part by an Established Investigator Grant from the Society of Critical Care Medicine.
