Abstract
Poly-ADP-ribosylation (PAR) of proteins by poly(ADP-ribose) polymerases (PARP) occurs after experimental traumatic brain injury (TBI) and modulates neurologic outcome. Several promising pharmacological PARP inhibitors have been developed for use in humans, but there is currently no clinically relevant means of monitoring treatment effects. We therefore used an enzyme-linked immunosorbent assay to measure PAR-modified proteins in cerebrospinal fluid (CSF). Cerebrospinal fluid samples from 17 pediatric TBI patients and 15 controls were plated overnight and then incubated with polyclonal antibody against PAR. Histone-1, a PARP substrate, was incubated with active PARP, NAD, and nicked DNA, and served as the standard. Both peak and mean CSF PAR-modified proteins were increased in TBI patients versus controls. Peak CSF PAR-modified protein levels occurred on day 1 and levels remained increased on day 2 after TBI. Increases in peak CSF PAR-modified protein concentrations were independently associated with age and male sex, but not initial Glasgow Coma Scale score, Glasgow outcome score, or mechanism of injury. The increase in PAR-modified proteins in CSF after TBI may be because of increased PARP activation, decreased PAR degradation, or both. As PAR-modified protein concentration correlated with age and male sex, developmental and sex-dependent roles for PARP after TBI are implicated.
Keywords
Introduction
Poly-ADP-ribosylation (PAR) of proteins is a post-translational modification catalyzed by nuclear, cytosolic, and mitochondrial poly(ADP-ribose) polymerases (PARP) (Ueda and Hayaishi, 1985; Virag and Szabo, 2002; Du et al, 2003; Lai et al, 2008). During PARP activation, more than 200 ADP-ribose moieties can be added to target proteins using one molecule of NAD+ for each ADP-ribose moiety (Ueda and Hayaishi, 1985). Polymerization of ADP-ribose moieties to target proteins confers a negative charge and alters protein function (Ueda and Hayaishi, 1985; Ha et al, 2002; Virag and Szabo, 2002; Lai et al, 2008), and has been used as a relatively quantitative biomarker of PARP activation (Yu et al, 2002; Du et al, 2003; Satchell et al, 2003). Poly(ADP-ribose) polymerase serves important homeostatic roles in DNA repair, transcription, cell cycle regulation, and memory (de la Lastra et al, 2007). However, during periods of severe cellular stress and energy imbalance, such as after traumatic brain injury (TBI) or stroke, overactivation of PARP can result in depletion of NAD+, energy failure, and cell death and dysfunction (Eliasson et al, 1997; Endres et al, 1997; Whalen et al, 1999; Satchell et al, 2003; Clark et al, 2007). In addition to consumption of NAD+, PARP activation can also directly inhibit electron transport, thereby reducing ATP production and energy repletion, compounding energy failure (Halmosi et al, 2001; Ha et al, 2002; Lai et al, 2008). Poly(ADP-ribose) polymerase can also mediate apoptotic cell death through the release of apoptosis-inducing factor from mitochondria (Yu et al, 2002; Du et al, 2003), a process that occurs after TBI (Zhang et al, 2002). It follows that PARP inhibition has been targeted in the prevention of cellular injury and death after TBI and stroke, as well as other diseases where energy failure contributes to pathology.
Worldwide, TBI is a major cause of mortality and morbidity, and is the leading cause of death in children in the United States (Adekoya et al, 2002). Both pharmacological and genetic PARP inhibitions have been shown to be remarkably protective after experimental TBI (Cosi et al, 1996; Klaidman et al, 1996; Eliasson et al, 1997; Endres et al, 1997; Whalen et al, 1999; Satchell et al, 2003; Clark et al, 2007). Novel PARP inhibitors are being developed for clinical use both alone and as chemosensitizers in combination with chemotherapy (de la Lastra et al, 2007). However, there is currently no efficient means for monitoring the pharmacological action of PARP inhibitors clinically. Here, we report the use of an enzyme-linked immunosorbent assay-based method for the quantification of PAR-modified proteins—a surrogate biomarker of PARP activation—in cerebrospinal fluid (CSF) from pediatric patients after severe TBI.
Materials and methods
Cerebrospinal Fluid Samples
This study was approved by the Institutional Review Board at the University of Pittsburgh Medical Center. Cerebrospinal fluid was obtained from a series of 17 infants and children after severe TBI obtained over a period of up to 4 days after injury, randomly selected from a TBI CSF tissue bank (Satchell et al, 2005). All the patients were admitted to the Pediatric Intensive Care Unit at the Children's Hospital of Pittsburgh and had an extraventricular drain placed for intracranial pressure monitoring and CSF drainage as standard-of-care for our institution. Cerebrospinal fluid was collected under sterile conditions from the graduated drip chamber of the closed extraventricular drain apparatus. Cerebrospinal fluid is measured in the drip chamber then drained hourly; thus, the collected CSF is at room temperature for a maximum period of 1 h. Samples were centrifuged at 3,000 r.p.m. for 10 mins to remove cellular debris and supernatants were decanted and stored at −70°C until batch analysis. Control CSF represented excess CSF obtained via lumbar puncture from 15 infants and children who were found not to have meningitis based on cell count, culture, and Gram stain. Protein concentrations for each CSF sample were measured using the bicinchoninic acid method.
Clinical data collected included demographic information, mechanism of injury, Glasgow Coma Scale (GCS) score on admission, and Glasgow outcome score (GOS) determined at 6 months after injury. Inflicted TBI from child abuse was diagnosed by the Children's Hospital of Pittsburgh Child Protection Team, and was independent of enrollment into the study and intensive care unit management.
Poly(ADP-ribose)-Modified Protein Standards
To quantify PAR-modified proteins, a standard was produced using the known PARP substrate histone-1. A reaction mixture was prepared consisting of 16.6 μmol/L histone-1 (Trevigen, Gaithersburg, MD, USA), 2 μL active PARP enzyme (> 2 U/μL; Trevigen), 100 μmol/L NAD + (Sigma, St Louis, MO, USA), and 50 μg nicked DNA (Trevigen), and was incubated for 1 h at 37°C. Serial dilutions of the PAR-modified histone mixture were used to generate standard curves, based on the known concentration of histone-1 and assuming close to 100% efficiency of the reaction. There was close agreement between duplicate samples, suggesting that the assay is reliable (
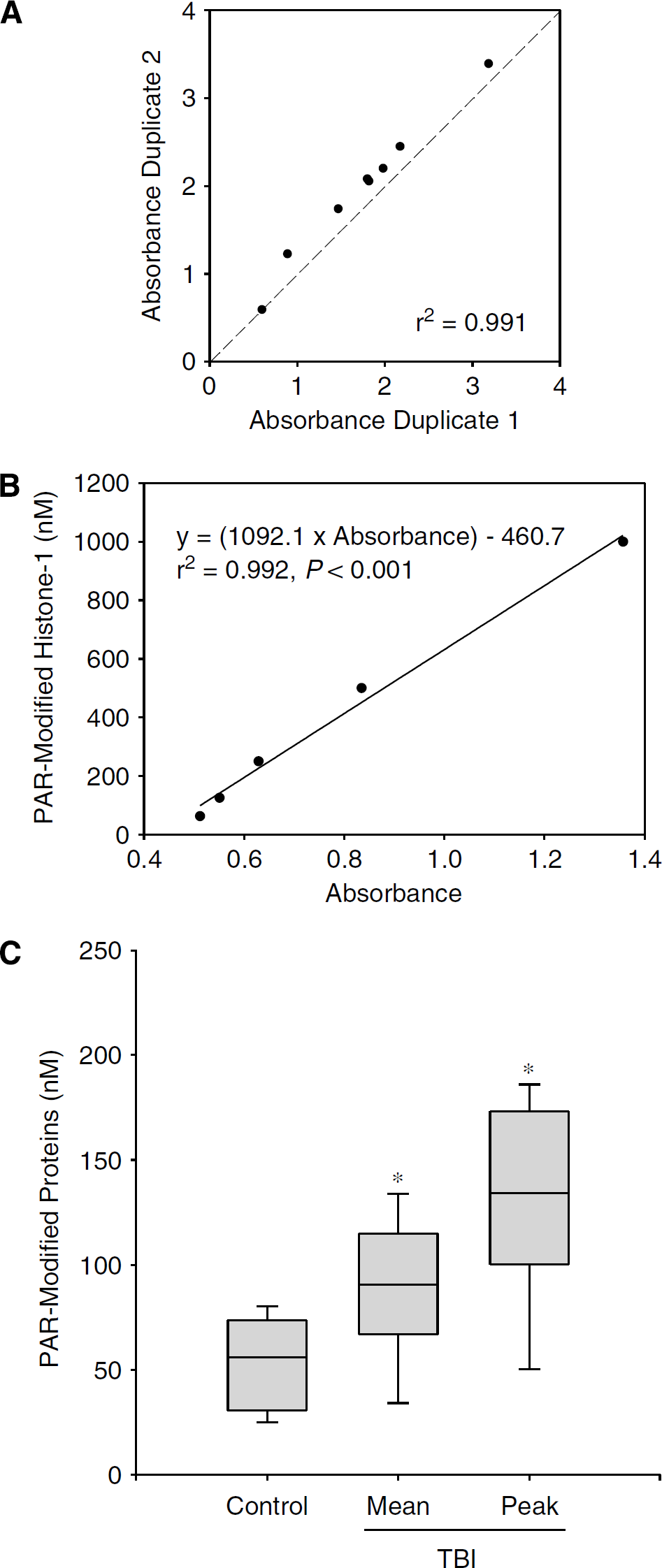
(
Poly(ADP-ribose)-Modified Protein Enzyme-Linked Immunosorbent Assay
Poly-ADP-ribosylation-modified histone-1 standards and samples of CSF from TBI patients and controls (100 μL) were diluted 1:2 with phosphate-buffered saline and then placed into 96-well polystyrene microtiter plates with high protein binding properties (catalogue no. 3590, Corning Inc., Corning, NY, USA) in duplicate. Samples were incubated overnight at 4°C to allow for protein binding to the microtiter plate. A commercially made enzyme-linked immunosorbent assay blocking solution (catalogue no. 80160, Alpha Diagnostic, San Antonio, TX, USA) was then added to each well for 2 h at room temperature. To identify PAR-modified proteins captured on the microtiter plate, a 1:1,000 dilution of rabbit polyclonal antibody against PAR (catalogue no. 4336-BPC-100, Trevigen) was added to each well and incubated at room temperature for 60 mins, followed by incubation in a 1:2,000 dilution of secondary anti-rabbit antibody conjugated to horseradish peroxidase for 30 mins at room temperature. A colorimetric detection system was used and absorbance was determined at 450 nm. Cerebrospinal fluid PAR-modified protein concentrations were calculated against the PAR-modified histone-1 standards.
Statistical Analysis
Data are presented as mean ± s.e.m. or median [range] where appropriate. The mean and peak CSF PAR-modified protein levels were determined for each patient. Comparisons between TBI and control groups were made using a Mann—Whitney rank sum test, as the data were not normally distributed. Comparisons between CSF PAR-modified protein levels for each day after injury were made using ANOVA on ranks. Comparisons between peak CSF PAR-modified protein levels and the clinical variables age, sex, admission GCS, 6-month COS, and accidental versus inflicted TBI were made using linear regression or Spearman rank tests for parametric and nonparametric data, respectively. A multivariate linear regression model was used that included age, sex, admission GCS, 6-month COS, and accidental versus inflicted TBI mechanism, to determine independent predictors of CSF PAR-modified protein levels. Accidental versus inflicted TBI was analyzed because inflicted TBI as a mechanism of injury is known to have a unique biomarker profile and unfavorable outcome compared with accidental injury (Clark et al, 2000; Berger et al, 2002; Satchell et al, 2005). Multivariate analysis was also performed using only independent variables with a
Results
Patient demographic data are shown in Table 1. The age range for TBI patients was 2 months to 11 years. Ten of the 17 TBI patients were male. The median GCS assigned on admission to the emergency department was 7. The majority of patients had TBI as a consequence of motor vehicle-related accidents (collisions, pedestrian or bicycle struck by motor vehicle), followed by inflicted injury because of child abuse. The survival in this cohort of TBI patients was 94%. Controls were younger (range 1 month to 7 years) than TBI patients (
Patient demographics
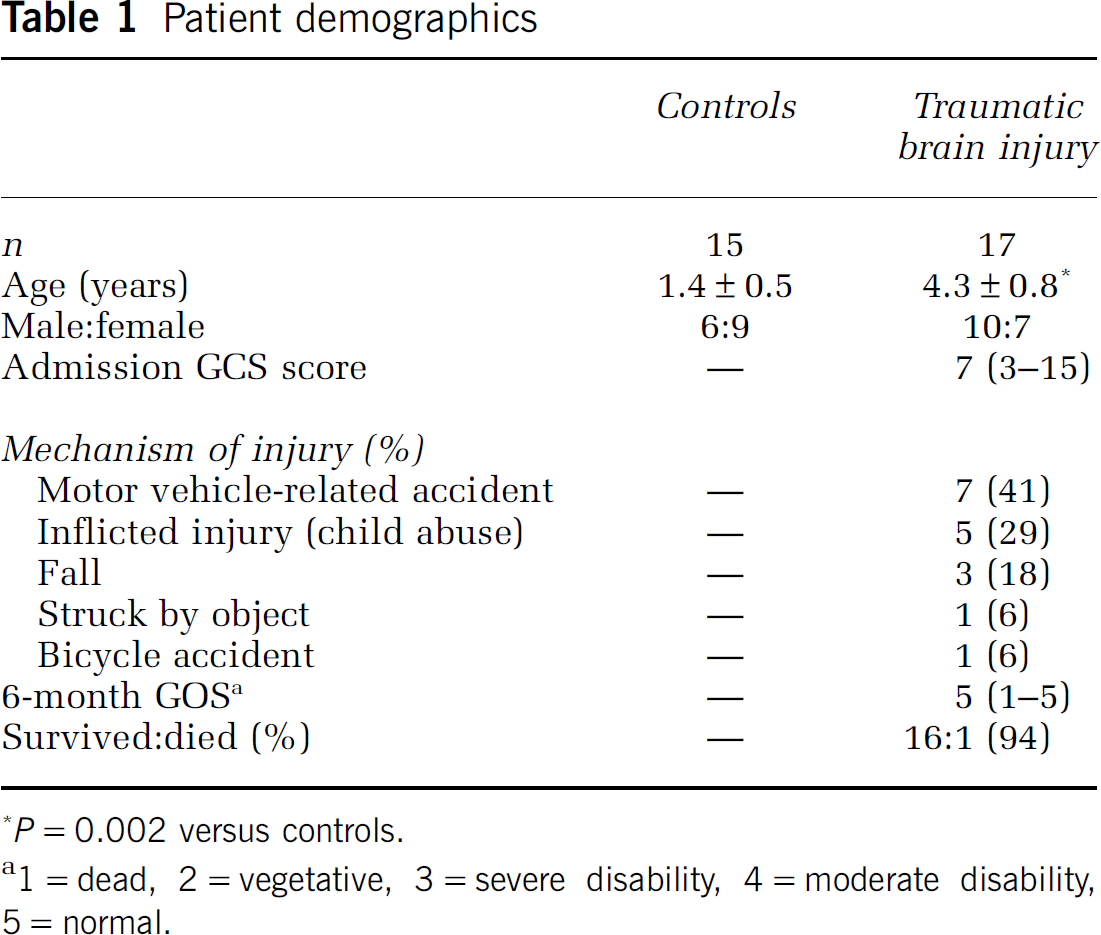
1 = dead, 2 = vegetative, 3 = severe disability, 4 = moderate disability, 5 = normal.
Figure 1C shows the mean and peak CSF PAR-modified protein levels for TBI patients versus the CSF PAR-modified protein levels for controls, presented as median, 25th to 75th, and 5th to 95th percentiles. The mean and peak CSF PAR-modified protein levels were increased in TBI patients compared with controls (88.1 ± 8.1, 132.5 ± 11.5, and 53.6 ± 5.2 nmol/L, respectively; mean ± s.e.m.,
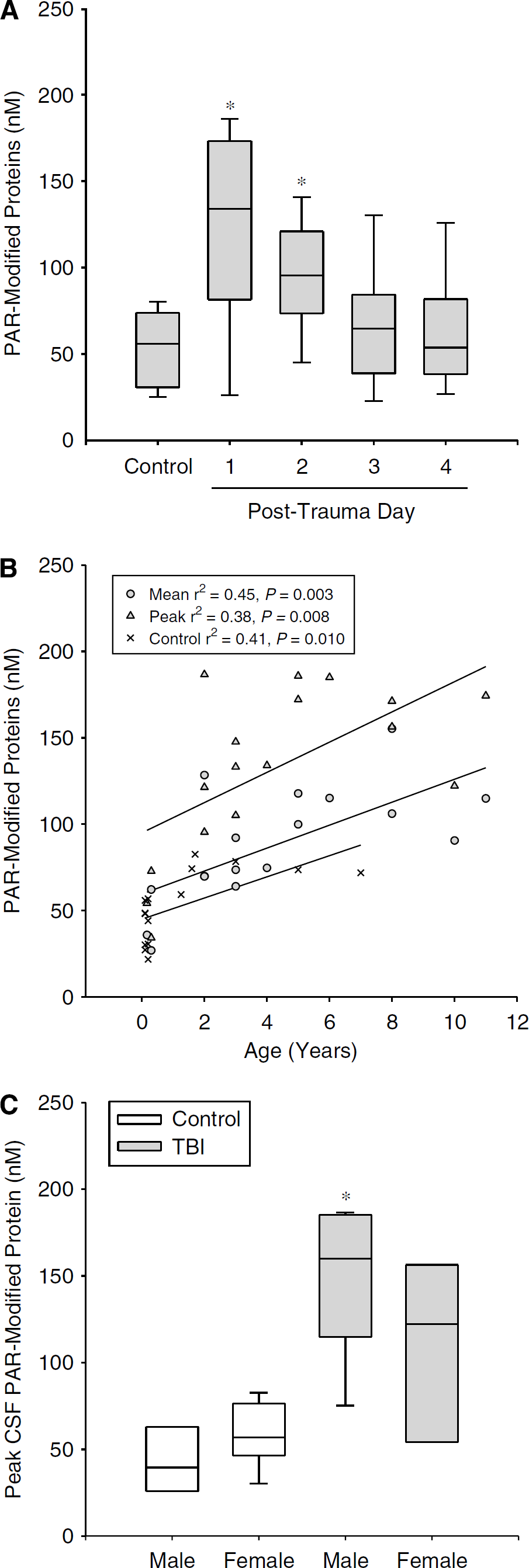
Cerebrospinal fluid PAR-modified protein levels and clinical variables. (
Univariate and multivariate analysis of peak CBF poly(ADP-ribose) modified protein levels an clinical variables
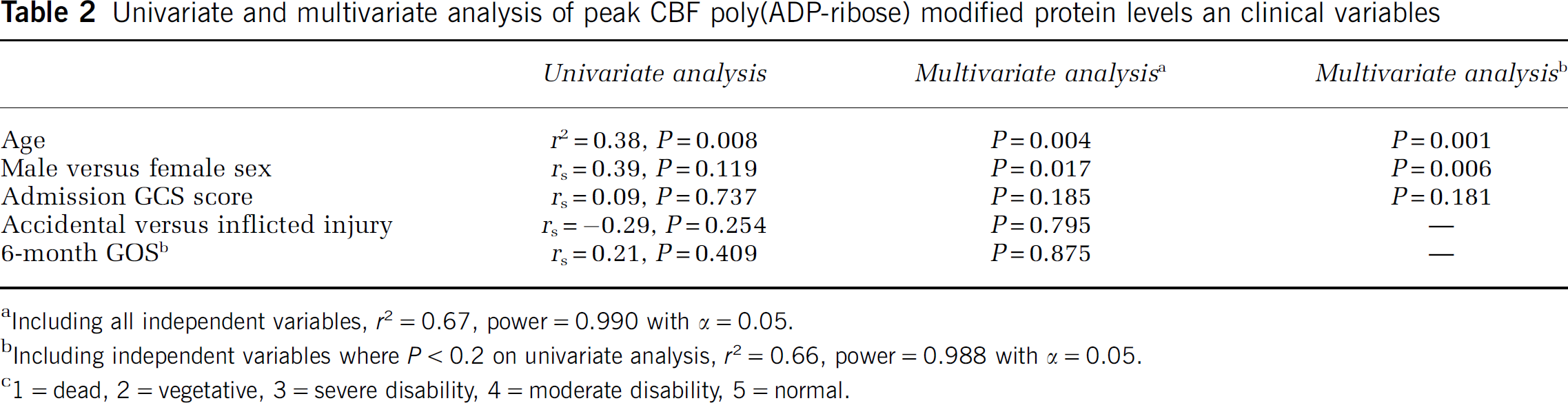
Including all independent variables,
Including independent variables where
1 = dead, 2 = vegetative, 3 = severe disability, 4 = moderate disability, 5 = normal.
Discussion
We report a reliable method for detection and quantification of PAR-modified proteins in CSF. In so much as PAR-modified proteins reflect PARP activity (Yu et al, 2002; Du et al, 2003; Satchell et al, 2003), this assay may prove useful for the evaluation of the role of PARP in diseases such as TBI, as well as for therapeutic drug monitoring of pharmacological PARP inhibitors. Using this assay, we were able to show an increase in CSF PAR-modified proteins and associations between male sex and increasing patient age.
Increased PARP activation has been shown in multiple experimental models of TBI, including controlled cortical impact in mice and rats, and fluid percussion injury in rats (LaPlaca et al, 1998; Satchell et al, 2003; Lai et al, 2008). Importantly, a detrimental role for PARP overactivation after TBI has been shown using both PARP-1 knockout mice (Whalen et al, 1999) and pharmacological PARP-1 inhibitors (LaPlaca et al, 2001; Satchell et al, 2003; Clark et al, 2007). Although, to the best of our knowledge, there have been no clinical studies implicating a role for PARP activation after TBI, there is a report showing increased PAR in human brain after stroke (Love et al, 1999). Based on preclinical studies, testing the effect of PARP inhibitors in patients with severe TBI appears justified. It logically follows that use of this (or a similar) assay to verify therapeutic effectiveness of PARP inhibition and perhaps tailor therapy would improve the scientific rigor of such a study.
The association between PAR-modified protein levels and male sex in TBI patients but not controls is in agreement with experimental
The basis for the association between PAR-modified protein levels and increasing age is less clear. Possible explanations for this finding include increased or more mature PARP enzymes, or decreased PAR hydrolysis by poly(ADP-ribose) glycohydrolase, with age. As one popular theory for aging is that DNA damage increases and accumulates with time, one could speculate that PARP enzymes are also increasing in amount or activity with age (Messripour et al, 1994). It has been reported that the amount of PARP-1 activity correlates with species-specific lifespan (Grube and Burkle, 1992). However, there does appear to be an upper limit, as elderly humans have less constitutive PARP-1 and PARP-2 compared with young humans, although in healthy centenarians amounts of PARP-1 and PARP-2 appear to be preserved (Chevanne et al, 2007). In whole-brain homogenates of late fetal rats, PARP activity was found to be 2.5 times that of postnatal day 4 rats, implicating a role for PARP in CNS cell differentiation, synaptogenesis, and development (Shambaugh et al, 1988). Although the effect of PARP activation on outcome after brain injuries sustained during development is less clear, the degree of PARP activation in neonatal rats after cerebral hypoxia—ischemia correlates with the degree of cerebral injury (Martin et al, 2005).
The finding that CSF PAR-modified protein levels in both TBI patients and controls positively correlated with age raises a limitation of this study, as age differed between groups, with the controls being much younger. As such, the difference in PAR-modified protein levels between TBI and control groups could potentially be explained by age alone and not TBI. However, peak CSF PAR-modified protein levels in 14 of the 17 TBI patients were above the highest control value (82.6 nmol/L, 1.7 years old; Figure 2B). Certainly, increasing the number of control CSF samples from older patients would be ideal, but these were not available. We also recognize that with a sample size of 17 patients, our multivariate analysis is exploratory. The sample size of 17 reported here is above the minimum number generally accepted for multivariate analysis that includes 3 independent variables (at least 5 per variable); however, studies in a larger sample are needed to confirm these findings and determine whether GCS, GOS, and the mechanism of injury are also predictive variables. There are also caveats related to the assay; for example, we did not control for hydrolysis of PAR. In tissues with active poly(ADP-ribose) glycohydrolase, the primary enzyme responsible for metabolizing PAR, or other hydrolytic enzymes, this assay would not reflect the degree of PARP activity. Nonetheless, this enzyme-linked immunosorbent assay appears to be capable of quantifying PAR-modified proteins in CSF, and testing in other tissues and diseases appears warranted.
In conclusion, age- and sex-dependent increases in CSF PAR-modified proteins are seen during the first 2 days after TBI in infants and children. The increase in PAR-modified proteins may be because of increased PARP activation, decreased PAR degradation, or both. This assay may represent a useful method for evaluating the role of PARP enzymes, and the potency and bioavailability of PARP inhibitors, in TBI and other disorders.
