Abstract
Cytochrome
Introduction
In the last 10 years, we have demonstrated that epoxyeicosatrienoic acids (EETs) are made within astrocytes in the brain; promote blood vessel formation, and angiogenesis and vasodilation in the rat brain. Epoxyeicosatrienoic acids are formed by the catalytic conversion of arachidonic acid (AA) via cytochrome P450 (CYP)2C11 enzymes in rat astrocytes (Alkayed et al, 1996) and by CYP4X1 in rat brain (Bylund et al, 2002). Four regioisomers of EETs are formed from AA, namely, 5,6-, 8,9-, 11,12-, and 14,15-EET. All regioisoforms are capable of supporting angiogenesis; however, our previous findings suggest that 8,9- and 11,12-EET are the most potent (Zhang and Harder, 2002). The ability of EETs to induce angiogenesis is not limited to the brain. Epoxyeicosatrienoic acids have been shown to be angiogenic in human lung microvascular endothelial cells and other vascular beds (Medhora et al, 2003; Michaelis et al, 2003; Zhang and Harder, 2002). We have previously described that formation of EETs in astrocytes are induced by glutamate or neural activation (Alkayed et al, 1997; Peng et al, 2002, 2004). Inhibition of EETs with mechanistically different cytochrome P450 2C11 epoxygenase (CYP epoxygenase) inhibitors blocks formation of capillaries or endothelial tube-like structures
It is estimated that there are over 350,000 persons in the United States diagnosed with a brain tumor of which gliomas account for approximately 50%. Of the individuals with central nervous system tumors, approximately 10% to 15% are malignant. Brain tumors are the leading cause of death from childhood cancer up to the age of 19 years and is the second leading cause of cancer-related deaths in men 20 to 39 years old (statistics obtained from: http://www.abc2.org/statistics.shtml). Malignant gliomas are very aggressive tumors of the central nervous system and are resistant to conventional therapies like radiation and chemotherapy (Rich and Bigner, 2004).
Rat glioma 2 (RG2) cells are nondifferentiated astrocytes, which form aggressive tumors when injected into the forebrain of Fischer rats. In this study, glioblastomas were formed experimentally in the forebrain of male rats via direct injection of RG2 cells. Two mechanistically different CYP enzyme inhibitors were used in the study: 17-octadecynoic acid (17-ODYA) and miconazole, both of which have been shown in previous publications to block formation of EETs in brain tissue (Alkayed et al, 1996; Bhardwaj et al, 2000; Peng et al, 2002).
Materials and methods
Materials
Male 8- to 10-week-old Fischer rats were purchased from Taconic Inc., Hudson, NY. All the animal studies were approved by the Institutional Animal Care Committee and were performed according to the guidelines of the NIH. Rat glioma 2 cells were obtained from ATCC (Manassa, VA, USA); 17-ODYA from Biomol (Plymouth Meeting, PA, USA); miconazole and all other chemicals and reagents were purchased from Sigma-Aldrich Co. (St Louis, MO, USA). Culture media, fetal bovine serum, and trypsin-EDTA (ethylenediaminetetraacetic acid) and penicillin-streptomycin were purchased from Invitrogen (Carlsbad, CA, USA). Polyclonal CYP2C11 antibody was raised in rabbits in our laboratory against the synthetic peptides derived from both the amino and carboxy terminals of CYP2C11 (Alkayed et al, 1996). Horseradish peroxidase-conjugated goat anti-rabbit IgG and goat anti-mouse IgG were purchased from Bio-Rad (Hercules, CA, USA). β-Actin antibody was purchased from Sigma-Aldrich. ECL plus reagent was obtained from GE healthcare (Buckinghamshire, UK).
Methods
Visualization of tumor vasculature and comparison of capillary density
Results
Formation and Characterization of Tumor
Intracranial tumors formed in all subject animals in about 11 days. Furthermore, all animals died from massive intracranial tumors suggesting that RG2 cells are a good model for induction of glioblastoma multiforme. No infection of surgical sites was noted. We observed that onset of symptoms such as body weight loss and neurological deficits were delayed in the groups treated with CYP inhibitors.
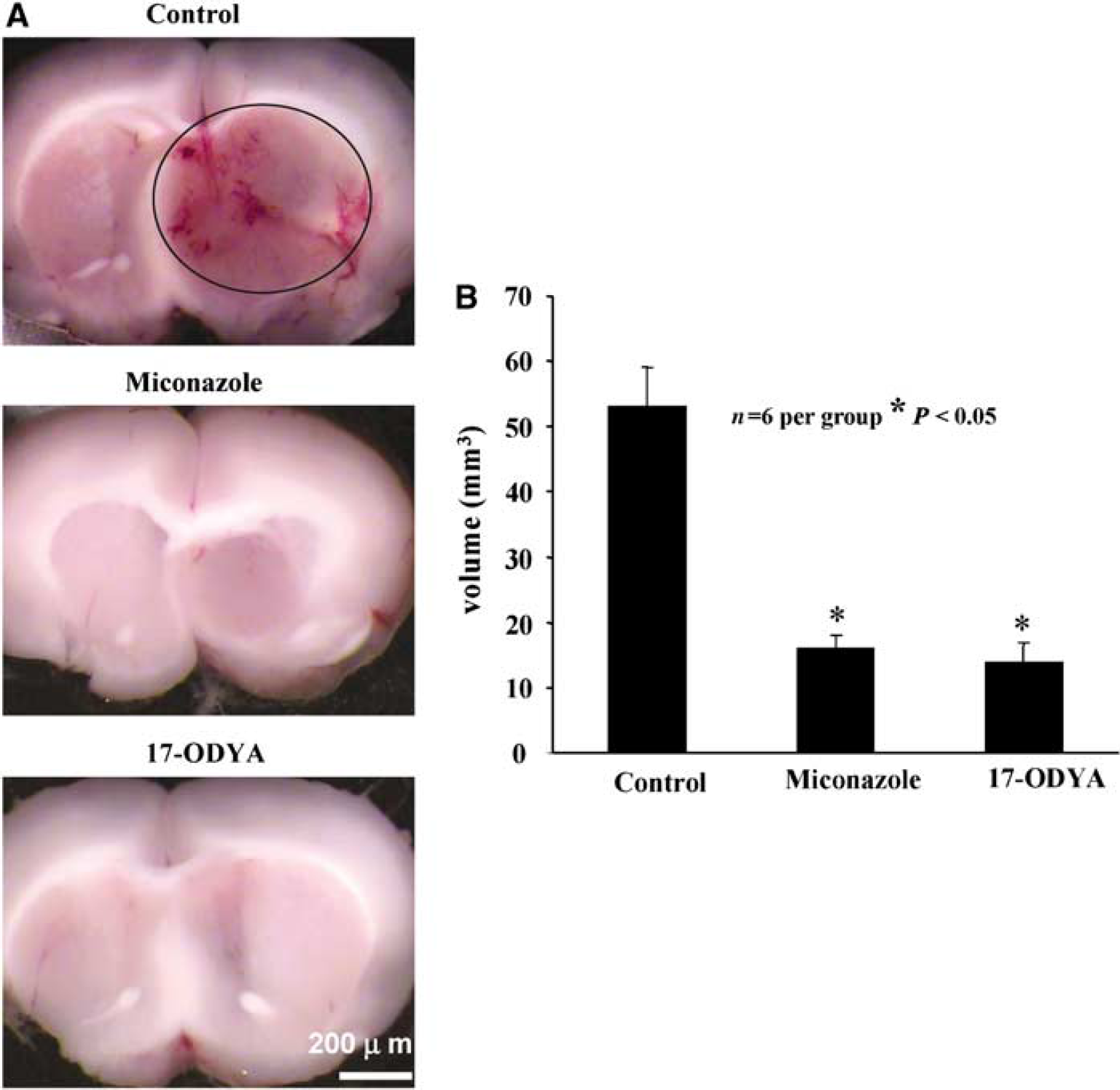
To determine invasiveness of developed tumors, fixed coronal sections were stained with cresyl violet, which allowed us to differentiate the boundaries between normal neuronal tissue from the glioma under a light microscope (Roth et al, 1999; Stojiljkovic et al, 2003). Coronal sections of brain (Figure 1A to 1C) showed massive, highly vascularized intracranial tumors with multiple areas of hemorrhage. Occasionally, large feeding vessels were found on the periphery of tumors suggesting that even at this stage the tumors had developed their own vascular supply. Furthermore, significant edema of surrounding tissue was also noted with classical pathological signs including shifting of midline (butterfly effect). Results from cresyl violet staining are summarized in Figure 2A (control) clearly demonstrating invasive growth pattern of developing tumors.
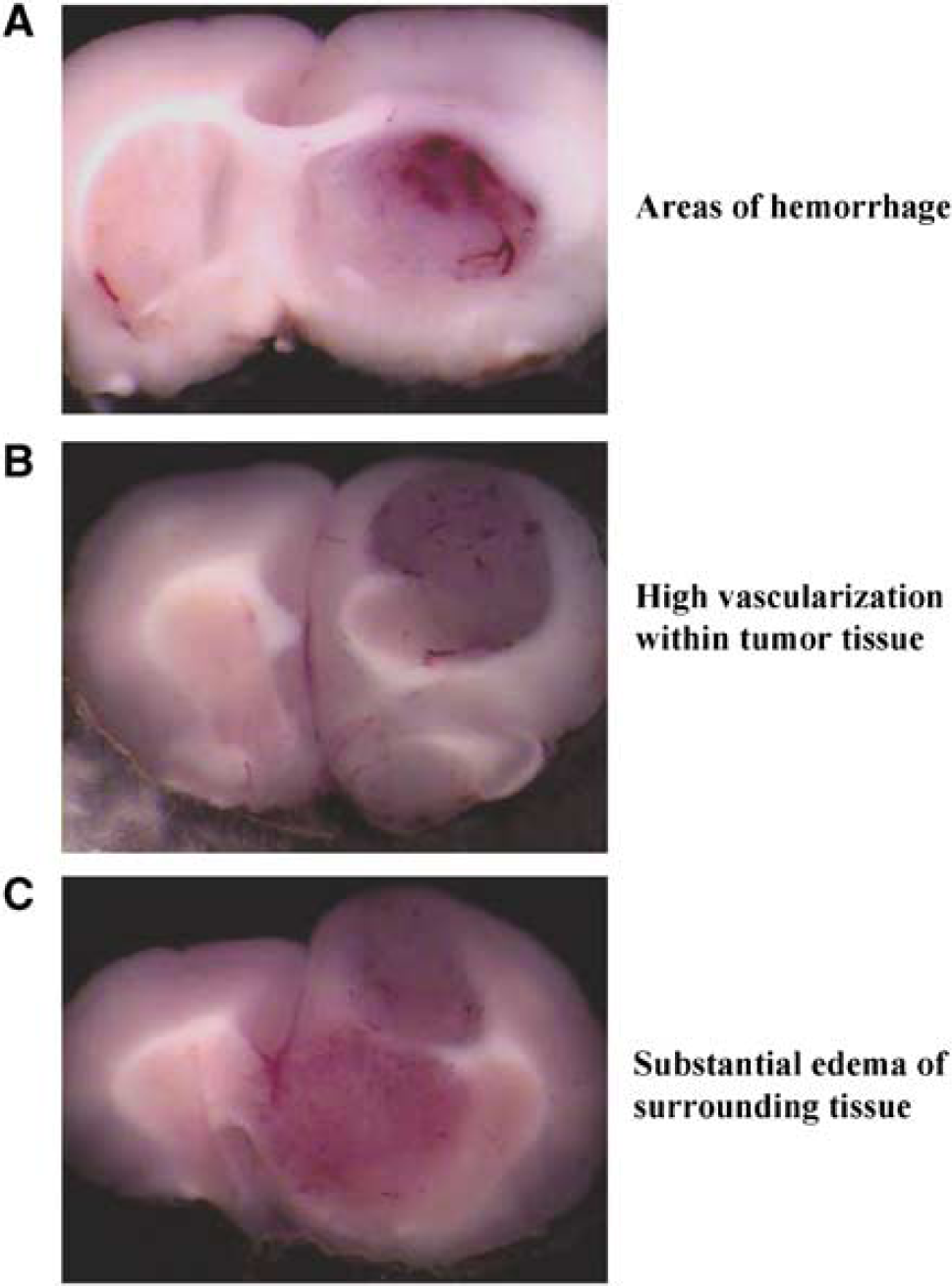
Pathological characteristics of tumors derived from RG2 cells (n = 6). (
(
Tumor Volume
Comparison of tumor volume revealed significant reduction of tumor size in miconazole- and 17-ODYA-treated rats. Average tumor volume for control group (Figure 2A) was 53.2 ± 6 mm3 (
Assessment of Tumor Vasculature
Numerous studies have shown that the tumor vasculature is highly heterogeneous and that a number of vessels are nonfunctional (Rich and Bigner, 2004; Cavallaro and Christofori, 2000; Carmeliet and Jain, 2000). To visualize the state of tumor vasculature, we perfused the brain with dextran-conjugated FITC. Using this technique, only patent and functional vessels were visualized. Figure 3 depicts the tumor vasculature after FITC-conjugated dextran staining. Permeability of the vasculature in the experimental groups is summarized in Figure 3A, which indicates that tumor capillaries are more permeable to FITC-conjugated dextran than normal vessels. Further analysis of tumor microcirculation suggests that density of capillaries is highly heterogeneous within tumor. Peripheral parts of tumor have significantly higher number of capillaries per 200 mm3 than central parts. A significant reduction in the number of vessels was observed in the miconazole- and 17-ODYA-treated groups compared with the control (Figure 3A)
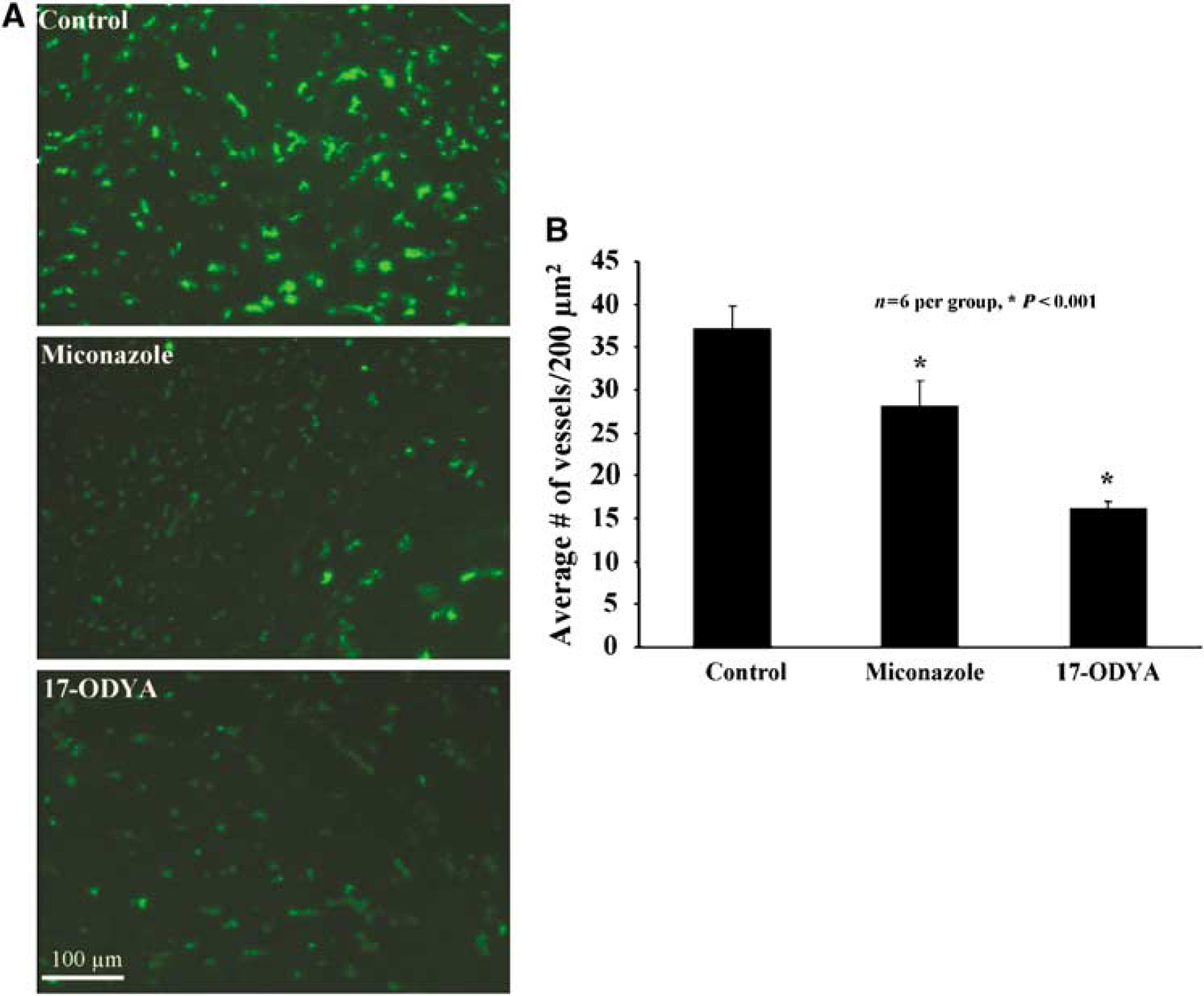
(
Survival Data
Median survival time for vehicle-treated tumor-developed animals (aCSF treatment,
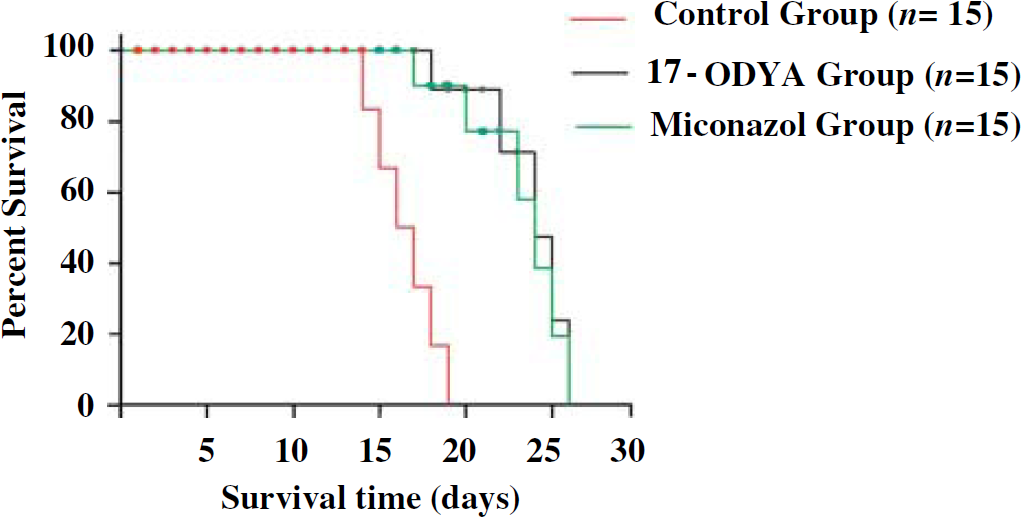
Shows the survival time of rats in days of different treatment groups with glioblastoma multiforme. Red line denotes the control group with the least survival time compared with green and black lines that are miconazole- and 17-ODYA-treated groups, respectively. The statistical significance of survival analysis were computed using multiple comparison procedures of Holm—Sidak method,
CYP2C11 Expression in Tumor
Examination of the expression level of CYP2C11 epoxygenase using antibody against CYP2C11 and western blot analysis revealed increased expression of the protein for CYP2C11 epoxygenase in tumor brain tissues as compared with the control (Figure 5).
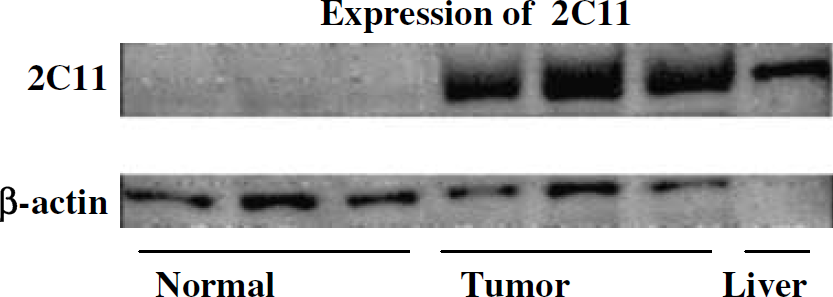
Differential expression of CYP2C11 epoxygenase protein in normal and tumor brain samples as determined by western blot analysis using male rat liver microsomes as positive control.
Effect of 17-ODYA and Miconazole on CYP Activity in Tumor Tissue
The fact that both tumor and normal brain tissues produce EETs (Figure 6) is suggestive of the contribution of the angiogenic EETs in promoting increased vascularity of the tumor tissue. We have also observed that both normal and tumor tissue metabolize AA to 5-, 8-, 12-, 15-, and 20-hydroxyeicosatetraenoic acids (HETEs) (Figure 6). However, the observed inability of the two mechanistically different CYP enzyme inhibitors to reduce endogenous level of EETs and HETEs (Figure 7) appears to suggest the prevalence of preformed EETs and HETEs that are stored in esterified form in cell membrane phospholipid pools (Carroll et al, 1997; Fang et al, 1995; VanRollins et al, 1996) that could account for the observed unaltered level of EETs after CYP epoxygenase inhibition. This lack of effect of the CYP inhibitors may further suggest that the reduction in tumor size after the administration of the CYP inhibitors could be the result of the pharmacological actions of the CYP inhibitors themselves. The potential mechanisms of such actions of these CYP inhibitors are open for further investigation.
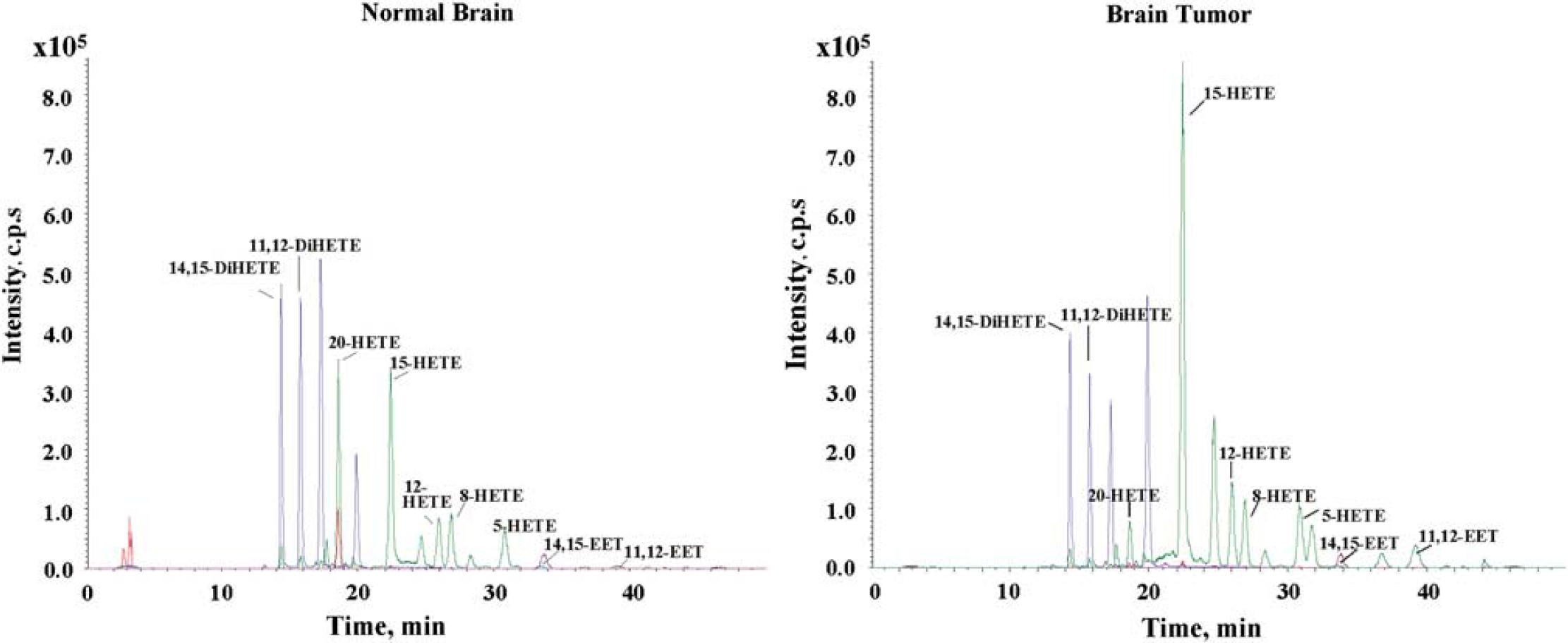
Representative LC-MS profile of CYPAA metabolites in normal and tumor rat brain tissue samples. Normal brain and tumor core tissue were removed from rat brain and frozen in liquid nitrogen. The samples were homogenized and assayed for CYP activity as described in the Methods section. Epoxyeicosatrienoic acids and Di-HETEs (8,9-, 11,12-, and 14,15-EETs) and HETEs(5-, 8-, 12-, 15-, and 20-HETE) were detected in both normal and tumor tissues.
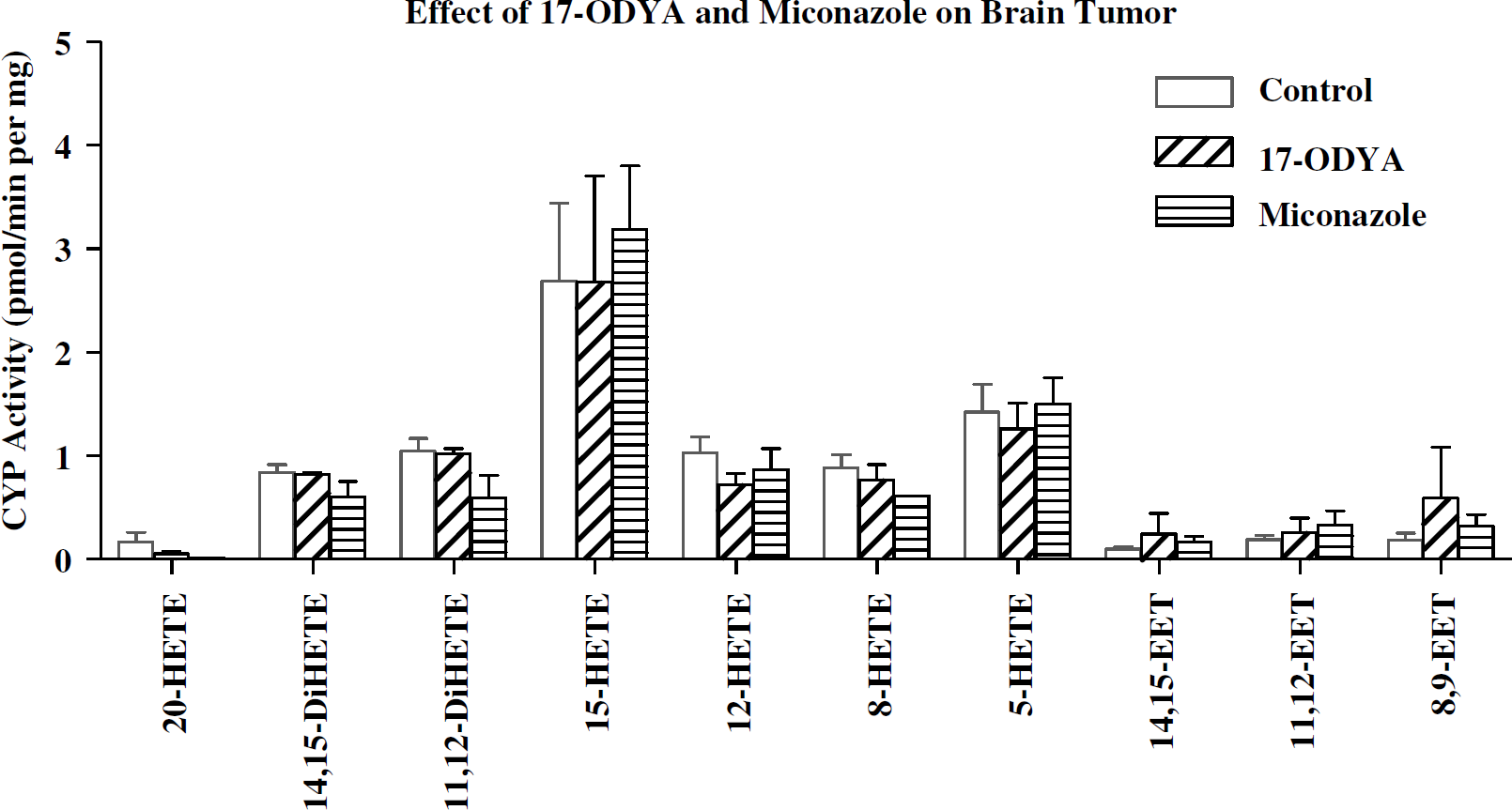
Effect of 17-ODYA and miconazole on CYP activity in normal and tumor samples. The brain tissue samples from the rats infused by implanted minipumps containing vehicle control, 17-ODYA (0.3 mmol/L), or miconazole (0.3 mmol/L) were removed and assayed for CYP activity by LC-MS as described in the Methods section. There was no significant difference between the levels of CYP metabolites in control and treated tumor brain tissues.
Discussion
Glioblastoma-like tumors were induced with Fischer rat RG2 cells, which are one of the most potent gliosarcoma cell lines currently available to researchers. Tumors developed from RG2 cells closely mimic human tumors in growth pattern and resistance to therapy (Rich and Bigner, 2004). To avoid one of the critical issues in cancer research involving immunological response from host, we used Fischer rats to match the RG2 cell host. Earlier findings from our laboratory and by others have shown that EETs generated by the action of CYP epoxygenases on AA are highly angiogenic in function (Munzenmaier and Harder, 2000; Medhora et al, 2003; Zhang and Harder, 2002). It is likely that these EETs could contribute to the increased vascularity in tumor tissue and thus promote tumor growth.
In the present study, both 17-ODYA and miconazole were found to be equally effective in reducing tumor size and prolonging survival time. Our choice of two mechanistically different inhibitors of CYP enzyme—17-ODYA, an irreversible and nonselective suicide substrate inhibitor of CYP enzymes, and miconazole with strong selective inhibition of CYP2C epoxygenase family—is based on previously published studies by us and those of other investigators in this field (Alkayed et al, 1997, 1996; Campbell et al, 1996; Gebremedhin et al, 1992; Munzenmaier and Harder, 2000; Roman, 2002). We chose to deliver these two inhibitors directly into the developing tumor by the use of osmotic minipumps for the following reasons: (1) to achieve continuous and proper concentration of inhibitors in tumor, because of unknown pharmacodynamic properties of these drugs in the rat (e.g., binding to plasma proteins, rate of metabolism and excretion, crossing blood—brain barrier and so on). (2) Recent studies (Rich and Bigner, 2004) also showed significant advantage of continuous-versus-intermittent treatment if target of therapy are endothelial cells, especially undergoing proliferation. Intermittent schedules revealed that rapidly dividing cells (such as endothelial or cancer cells) can escape intended inhibition. (3) We used a concentration of 0.3 mmol/L even though the reported IC50 for these compounds is around 0.005 mmol/L
In the present study, we also found that expression of the CYP2C11 epoxygenase protein is markedly upregulated in tumor tissue compared with the normal tissue, which is consistent with a previous finding that CYP2J2 epoxygenase promotes the neoplastic phenotype of carcinoma cells and is upregulated in human tumors (Jiang et al, 2005). However, in our studies 17-ODYA and miconazole have no effect on EET and DHET (dihydroxy eicosatrienoic acid) levels, unlike Jiang's study, where 17-ODYA inhibited the formation of 14,15-DHET (Jiang et al, 2005). This could be because of the different carcinoma cell type used and the overexpression of CYP2J2 epoxygenase in these cells. Similar studies reported by Guo et al (2005) further strengthen our observation that CYP inhibitors are antitumorogenic. In their study, the authors have shown that human U251 glioma cells do not synthesize 20-HETE but produce other HETEs and EETs that were not inhibited by HET0016 (Guo et al, 2005). Our results are in agreement with these studies in that the 17-ODYA reduces tumor formation without affecting the EETs synthesis (Chen et al, 2005; Guo et al, 2006). Guo et al (2005) have reported that brain tumors developed by the injection of 9L gliosarcoma cells in Fischer rats were reduced in size by 80% by treating the rats with HET0016, a selective CYP4A inhibitor, when administered chronically twice daily by intraperitoneal injections at a concentration of 10 mg/kg per day in 10% lecithin in water for 2 weeks. It is interesting to note that the 9L rat gliosarcoma cells do not synthesize 20-HETE but synthesize 5-, 12- and 15-HETEs. Yet HET0016, a 20-HETE specific CYP4A inhibitor mitigates the tumor (Guo et al, 2006). One reason may be that these inhibitors are acting on other fatty acid substrates that may promote angiogenesis. Also, it is possible that the inhibitors are targeting the EETs in endothelial cells in the tumor vasculature, thus limiting angiogenesis. This is difficult to follow up given the absence of effective techniques for measurement of EET levels in exclusively endothelial cells derived from tumor vasculature. Further studies are required to understand the mechanism of action of these compounds in attenuating the tumor growth.
The concept of targeting blood vessels that feed tumors as an approach to limit and/or mitigate tumor formation dates back to the early 1970s (Folkman, 1971). However, it has only been within the last decade that major developments have been made to understand the molecular mechanisms involved in tumor-induced angiogenesis (Carmeliet and Jain, 2000; Cavallaro and Christofori, 2000). To this end, several antiangiogenic agents are in development with some in Phase III clinical trials for aggressive tumors like small cell lung carcinoma (Blackhall and Shepherd, 2004). Examples of these agents include matrix metalloproteinase inhibitors, angiogenic promoter antagonists, endogenous inhibitors (i.e., endostatin and angiostatin), and select novel agents (e.g., celecoxib, integrin antagonists, and so on). In fact, given the significant number of pathways and steps involved in new blood vessel formation, it is likely that tumor angiogenesis will continue to be an important target for cancer therapeutics.
Implementation of sophisticated brain imaging three decades ago significantly improved early diagnosis, however, prognosis has not changed, suggesting that new treatment approaches and/or modalities are desperately needed for this devastating disease. The focus of most current therapies are geared toward cancer cells. However, all solid tumors are comprised not only of cancer cells but also contain vascular components and interstitial tissue. The vascular component of glioblastoma is extremely important because these cells also show some malignant features. More recently, microvascular proliferation was added as an independent prognostic factor for these tumors, suggesting that tumors with increased microvascular proliferation are more aggressive and resistant to therapy and survival (Rich and Bigner, 2004). Because glioblastoma is resistant to many therapies, the current available treatment approach is radical or debulking surgery followed by chemotherapy. Even after an initial good response to therapy and clinical improvement, these tumors usually recur after a few months and show accelerated growth. In such circumstances, inclusion of 17-ODYA or miconazole, which in the present study was found to reduce tumor growth, as chemotherapeutic drugs after resection of the tumor may be beneficial in preventing reoccurrence.
In the present study, miconazole- or 17-ODYA -treated rats survived approximately 5 to 7 days longer than control, respectively. Similar survival rates have been observed with 9 L gliosarcoma cell-induced brain tumors in Fischer rats treated with the CYP4A ω-hydroxylase inhibitor HET0016 (Guo et al, 2006). This survival time frame in rat is equivalent to 3 to 4 months life extension in humans. This could be a significant treatment option if the survival could be further extended with combinatorial treatment regimes such as partial resection followed by treatment with either 17-ODYA, miconazole, or combination of CYP inhibitors along with other antiangiogenic agents. In summary, our present observation that the CYP inhibitors miconazole and 17-ODYA reduced tumor size supports the possible use of these compounds in cancer therapy for the treatment of devastating disease such as glioblastoma multiforme.
