Abstract
Vascular cells provide a neural stem/progenitor cell (NSPC) niche that regulates expansion and differentiation of NSPCs within the germinal zones of the embryonic and adult brain under both physiologic and pathologic conditions. Here, we examined the NSPC—endothelial cell (NSPC/EC) interaction under conditions of ischemia, both
Introduction
The adult mammalian brain harbors two germinal centers that continuously give rise to new neurons throughout adulthood. These include the subgranular zone of the dentate gyrus, which gives rise to new dentate granule neurons, and the subventricular zone (SVZ) surrounding the lateral ventricles, which gives rise to new neurons within the adult olfactory bulb. Neural stem/progenitor cells (NSPCs) that reside within these germinal centers are self-renewing, mitotically active, and multipotent cells with the potential to become neurons, astrocytes, or oligodendrocytes (Gottlieb, 2002; Lim et al, 2007; Temple and Alvarez-Buylla, 1999).
Neural stem/progenitor cells within the adult brain germinal centers reside in a specialized microenvironmental niche, closely associated with blood vessels throughout life (Alvarez-Buylla and Lim, 2004; Doetsch, 2003; Palmer et al, 2000; Wurmser et al, 2004b). Reciprocal signaling between NSPCs and endothelial cells (ECs) within the microenvironmental niche is thought to regulate both neurogenic and angiogenic processes. Louissaint et al (2002) showed a causal interaction between angiogenesis and neurogenesis in the adult songbird brain, involving reciprocal vascular endothelial growth factor (VEGF) and brain-derived neurotrophic factor (BDNF) signaling. Palmer et al (2000) also provided compelling evidence that neurogenesis is associated with active vascular recruitment and remodeling in the adult mammalian brain, and that adult neurogenesis occurs within an angiogenic niche.
Much research effort has recently been focused on understanding the neurogenic response to ischemic brain injury, which may play a role in regeneration and repair processes. Focal cerebral ischemia induced by middle cerebral artery occlusion (MCAO) stimulates increased proliferation of SVZ progenitors and massive migration SVZ-derived neuroblasts into the lesioned striatum in rodent. The neurogenic response is delayed and of long duration, such that the migration of neuroblasts does not peak until 1 to 2 weeks after injury and continues for several months (Arvidsson et al, 2002; Kokaia et al, 2006). Interestingly, the onset of the neurogenic response occurs concomitant with the angiogenic response to stroke, and is correlated with the onset of spontaneous improvements in behavioral deficits and cognitive function, even though the percentage of neuroblasts that survive to give rise to postmitotic neurons represents only an estimated 0.2% of lost neurons (Thored et al, 2006). Evidence suggests a functional association between neurogenic and angiogenic responses to stroke (Liu et al, 2007; Ohab et al, 2006; Yamashita et al, 2006). Blood vessels provide a physical substrate for neuroblast migration (Ohab et al, 2006; Yamashita et al, 2006), and both the neurogenic and angiogenic responses to stroke are governed by common growth factors and migratory cues (Ward and Lamanna, 2004). Several lines of evidence have led to the notion that angiogenesis stimulates the migration of neuroblasts after ischemic injury, but the converse may also be true, that is, that the neurogenic response is vasculotrophic, and thereby critical for the stabilization of new vasculature and successful revascularization after stroke.
In this study, we explore the NSPC/EC interactions in the context of ischemia. Neural stem/progenitor cells isolated from the embryonic mouse brain were studied for their ability to provide trophic support for ECs in culture, using a well-characterized
Materials and methods
Cell Culture
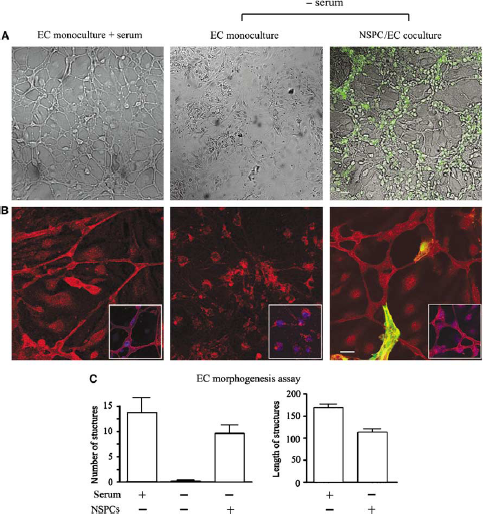
Neural stem/progenitor cells support ECs morphogenesis and survival in serum-free conditions. Micrographs show ECs and NSPC/EC cocultures plated on Matrigel matrix. GFP-positive NSPCs are shown using the green channel. (
This coculture system allowed NSPCs and ECs to share the same growth medium, and allowed substances to freely diffuse through cell culture insert or cell strainer pores without physical contact between the two cell types.
Oxygen and Glucose Deprivation
Cell cultures were subjected to OGD injury as described previously (Wetzel et al, 2008). Cultures were placed in an anaerobic chamber (Coy Laboratories, Grass lake, MI, USA) containing a gas mixture of 5% H2, 5% CO2, and 85% N2 to obtain < 0.2% O2. Normal culture medium was replaced with deoxygenated, glucose-free Earle's Balanced Salt Solution (EBSS), and cells were exposed to glucose-free anaerobic conditions for 3 or 5 h at 37°C. Control cell cultures were placed in EBSS containing 25 mmol/L glucose and incubated under normal tissue culture conditions for the same period.
Endothelial Cell Morphogenesis Assay
Endothelial cells were trypsinized, and after centrifugation, resuspended in EC medium, containing 10% fetal bovine serum and plated at the density of 2 times 104 cells per 12 mm diameter glass coverslips precoated with Matrigel matrix. Six hours after plating, the ECs were either cocultured with NSPCs (see direct NSPC/EC cocultures above) or incubated with a growth factor-free NSPC-conditioned medium (NSPC-CM) (medium collected from NSPCs cultured for 3 days). When plated on Matrigel or collagen, ECs form cellular networks (mesh-like structures) and capillary tubes. In addition, for better EC visualization, immunofluorescence staining for endothelial marker CD31 was performed before analyses.
Endothelial cell morphogenesis was assessed under an inverted confocal microscope, and images were taken using LSM-META confocal imaging system. A total of 10 to 15 images per coverslip were taken systematically across the coverslips. The number and diameter of capillary-like structures were counted using LSM Image Browser. Statistical analysis was performed using GraphPad Prism software.
Immunocytochemistry
Cells were fixed with 4% paraformaldehyde solution and quenched with 50 mmol/L ammonium chloride. After permeabilization with 0.1% (v/v) Triton X-100 and blocking with 1% horse serum, cells were incubated with primary antibodies for 1 h followed by fluorophore-conjugated secondary antibodies for 40 to 60 mins at room temperature. The following antibodies were used: mouse monoclonal anti-nestin (1:1,000; BD Pharmingen, San Diego, CA, USA), mouse monoclonal (1:1,000) and rabbit polyclonal (1:500) anti-GFAP (Accurate Chemical & Scientific Corp., Westbury, NY, USA), goat polyclonal anti-doublecortin (1:300; Santa Cruz Biotechnology, Santa Cruz, CA, USA), mouse monoclonal anti-βIII tubulin/Tuj1 (1:300; Promega, Madison, WI, USA), Alexa Fluor-conjugated rat monoclonal anti-mouse CD106/VE-cadherin (1:300; BioLegend, San Diego, CA, USA), rat monoclonal anti-mouse CD31/PECAM-1 (platelet—endothelial cell adhesion molecule-1) (1:300; BD Biosciences), mouse monoclonal anti-VEGF (1:200; Abcam, Cambridge, MA, USA), and rabbit polyclonal anti-HIF-1α (1:300; Chemicon, Temecula, CA, USA). Secondary antibodies were used at a common dilution of 1:200, and included Cy3-conjugated goat anti-rabbit, FITC-conjugated goat anti-mouse, Cy3-conjugated donkey anti-goat, and Cy3-conjugated donkey anti-mouse (Jackson ImmunoResearch Laboratories, West Grove, PA, USA). 4',6-Diamidino-2-phenylindole (DAPI) staining was used for detection of nuclei.
Western Blot Analyses
The cells were scraped from the cell culture dishes and filter inserts either directly with 2 × SDS sample buffer, or in lysis buffer (250 μL per filter) containing 1% (vol/vol) Triton X-100, 150 mmol/L NaCl, 10 mmol/L Tris-HCl, pH 7.4, and a protease inhibitor cocktail. To ensure equal protein loading, the relative protein concentration in the samples (lysed with 1% Triton X-100 lysis buffer) was quantified according to absorbance at 280 nm. Protein loading was additionally confirmed by comparing actin immunoreactivity across lanes. The proteins were separated on 4 to 20% gradient Criterion precast gels (Bio-Rad, Hercules, CA, USA). A broad-range molecular weight calibration marker from 10,000 to 250,000 MW (Bio-Rad) was used as a standard. The proteins were transferred onto nitrocellulose membrane and identified using mouse monoclonal anti-VEGF (1:100; Abcam) and rabbit polyclonal anti-actin (1:1,000, Sigma, St Louis, MO, USA). Horseradish peroxidase-labeled secondary antibodies were obtained from Amersham Biosciences (Piscataway, NJ, USA) and used in dilution of 1:3,000. For HIF-1α detection, we used a biotinylated anti-HIF-1α antibody (200 ng/mL) provided in the Surveyor™ IC Intracellular HIF-1α immunoassay kit (R&D Systems, Minneapolis, MN, USA), followed by horseradish peroxidase-conjugated streptavidin (1:200). Incubation with both primary and secondary antibodies was performed for 1 h at room temperature.
MTT Colorimetric Cell Viability Assay
Cells were incubated with 0.5 mg/mL methyltetrazolium (MTT, Sigma) for 4 h at 37°C, and treated with 1:1 ethanol/DMSO (dimethyl sulfoxide) for 20 mins at room temperature. The ability of cells to convert MTT into purple formazan provides an indication of the mitochondrial integrity and activity, interpreted as a degree of cell viability. The optical density was measured at 570 nm (with background subtraction at 630 nm) using a microplate reader (Dynex Technologies, Chantilly, VA, USA). Absorbance at 570 nm is directly proportional to the number of viable cells. SU1498 was purchased from Calbiochem (San Diego, CA, USA) and used at concentrations of 5 to 20 μmol/L; Flt-1-Fc (Sigma) was used at a concentration of 30 ng/mL.
Vascular Endothelial Growth Factor and Hypoxia-Inducible Factor-1α Immunoassay
For VEGF immunoassay, conditioned medium was collected, filtered to remove cells and debris, and concentrated eightfold using iCON™ concentrator with 9K molecular weight cut-off (Pierce, Rockford, IL, USA) and centrifugation at 3,000
Cell Transplantation and Middle Cerebral Artery Occlusion
All surgical procedures were approved by the University of New Mexico Animal Care Committee and conformed to the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals. Eight-week-old male C57BL/6 mice 20 to 25 g (The Jackson Laboratory) were used. Mice received buprenorphine (0.05 mg/kg, s.c.) at the onset of all surgery as analgesic. Anesthesia was induced with 1.5% isoflurane and maintained with 1.0% isoflurane in 1 L O2, using a vaporizer (Summit Medical Equipment, Bend, OR, USA). Enhanced green fluorescent protein NSPCs were implanted into the corpus striatum of 8-week-old C57BL/6 mouse recipients. Mice received stereotaxic injections using a Hamilton microsyringe with a 26-gauge blunt needle. Each animal received an injection of 2.5 μL of cell suspension (5 times 104 cells per μL in phosphate-buffered saline; PBS) at the rate of 1 μL per min at the following stereotactic coordinates (from bregma: A + 1.0 mm, L + 2.0 mm, V −2.6 mm). Sham-transplanted mice were injected with the same volume of PBS at the same stereotactic coordinates. At 3 days after transplantation, mice were either killed or subjected to MCAO as described below (
Mild transient focal ischemia was induced by right MCAO using the intraluminal thread method as previously described (Kokovay et al, 2006). After the induction of anesthesia as described above, the right common carotid artery was exposed and ligated with 6-0 silk suture. A 6-0 round tip nylon suture was introduced into the common carotid artery to occlude the middle cerebral artery. Successful MCAO resulted in 80% decrease of blood flow to the right cerebral hemisphere as assessed by laser Doppler analysis. After 30 mins, the intraluminal thread was withdrawn and the common carotid artery was ligated with a silk suture above the point of thread insertion. Reperfusion occurred via the Circle of Willis for 3 days before mice were killed for histologic analysis.
Histologic Procedures and Image Analysis
Mice were overdosed with sodium pentobarbital administered intraperitoneally (150 mg/kg) and transcardially perfused with PBS containing 0.1% procaine and 2 U/mL heparin, followed by 4% paraformaldehyde containing 0.075 M lysine and 0.01 M sodium periodate. Brains were postfixed overnight, cryoprotected in 30% sucrose, and sectioned at 30 μm thickness in the coronal plane using a sliding knife freezing microtome. Floating sections were subjected to immunofluorescence staining as previously described (Kokovay et al, 2006). Sections were permeabilized with 0.1% (v/v) Triton X-100, blocked with 5% donkey serum, and incubated with primary antibodies overnight at 4°C. Primary antibodies and dilutions were as follows: rabbit anti-HIF-1α (1:150; Chemicon), biotinylated anti-HIF-1α from the Surveyor IC Intracellular HIF-1α immunoassay kit (200 ng/mL; R&D Systems), mouse anti-VEGF (1:200; Abcam), and rabbit anti-GLUT-1 (1:200; Chemicon). Cy3- and FITC-conjugated secondary antibodies, and Cy3-conjugated streptavidin were used at a 1:250 dilution (Jackson ImmunoResearch Laboratories). All samples were imaged on a Zeiss LSM510 or Zeiss LSM510-META confocal imaging system.
Results
Neural Stem/Progenitor Cells Promote Endothelial Cell Morphogenesis and Prevent Endothelial Cell Death after Serum Starvation and Oxygen and Glucose Deprivation
The vasculotrophic effects of NSPCs were initially tested
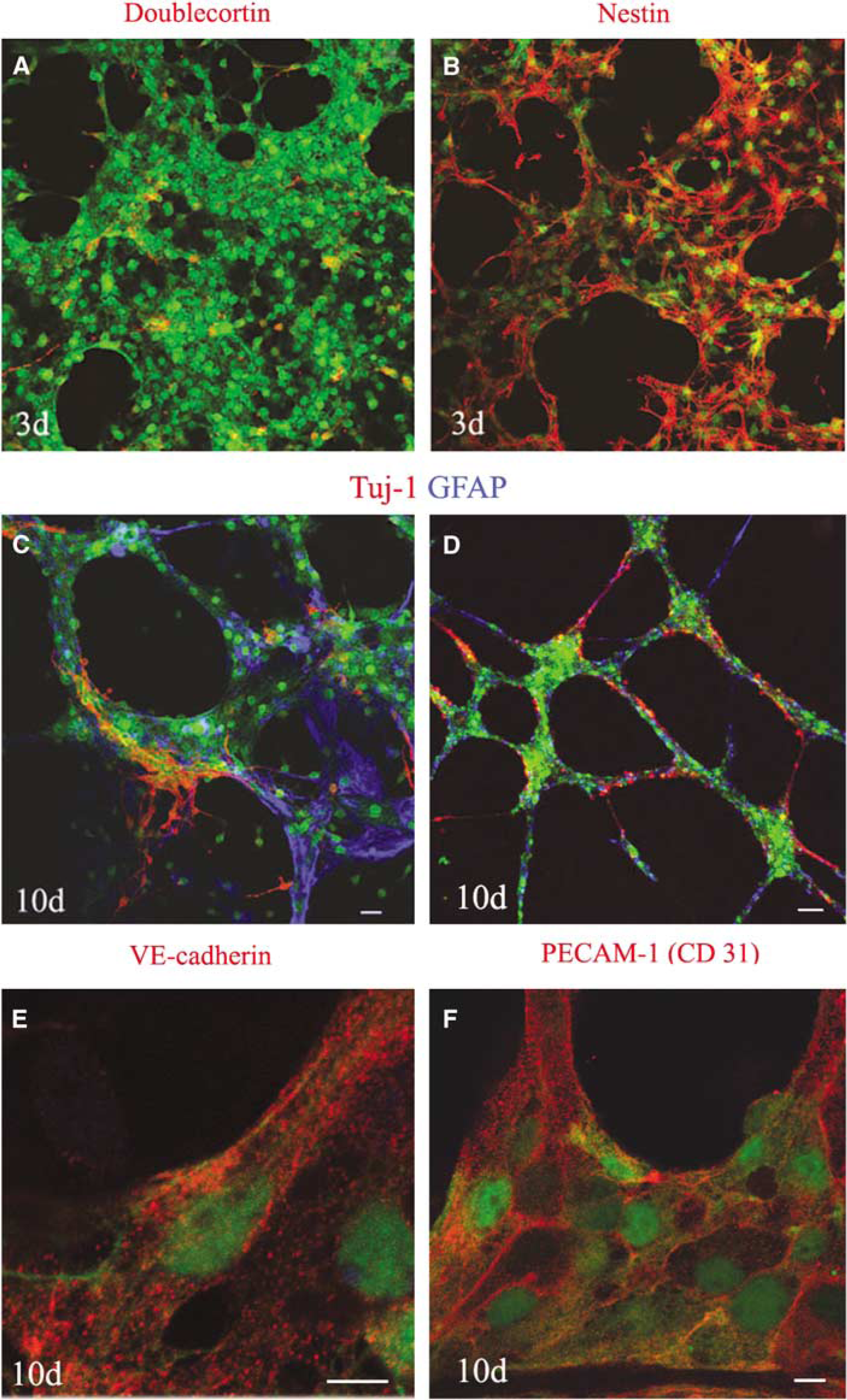
Phenotypic analysis of EGFP/NSPCs in coculture with ECs. Neural (doublecortin for neuroblasts; Tuj-1 for neurons, and GFAP for astrocytes), stem cell (nestin), and endothelial cell (VE-cadherin, CD31) markers were used to characterize the phenotype of NSPCs cocultured with ECs. Enhanced green fluorescent protein-neural stem/progenitor cells (green) were cocultured with ECs for 3 days (
To determine whether NSPCs provide trophic support to ECs under conditions of
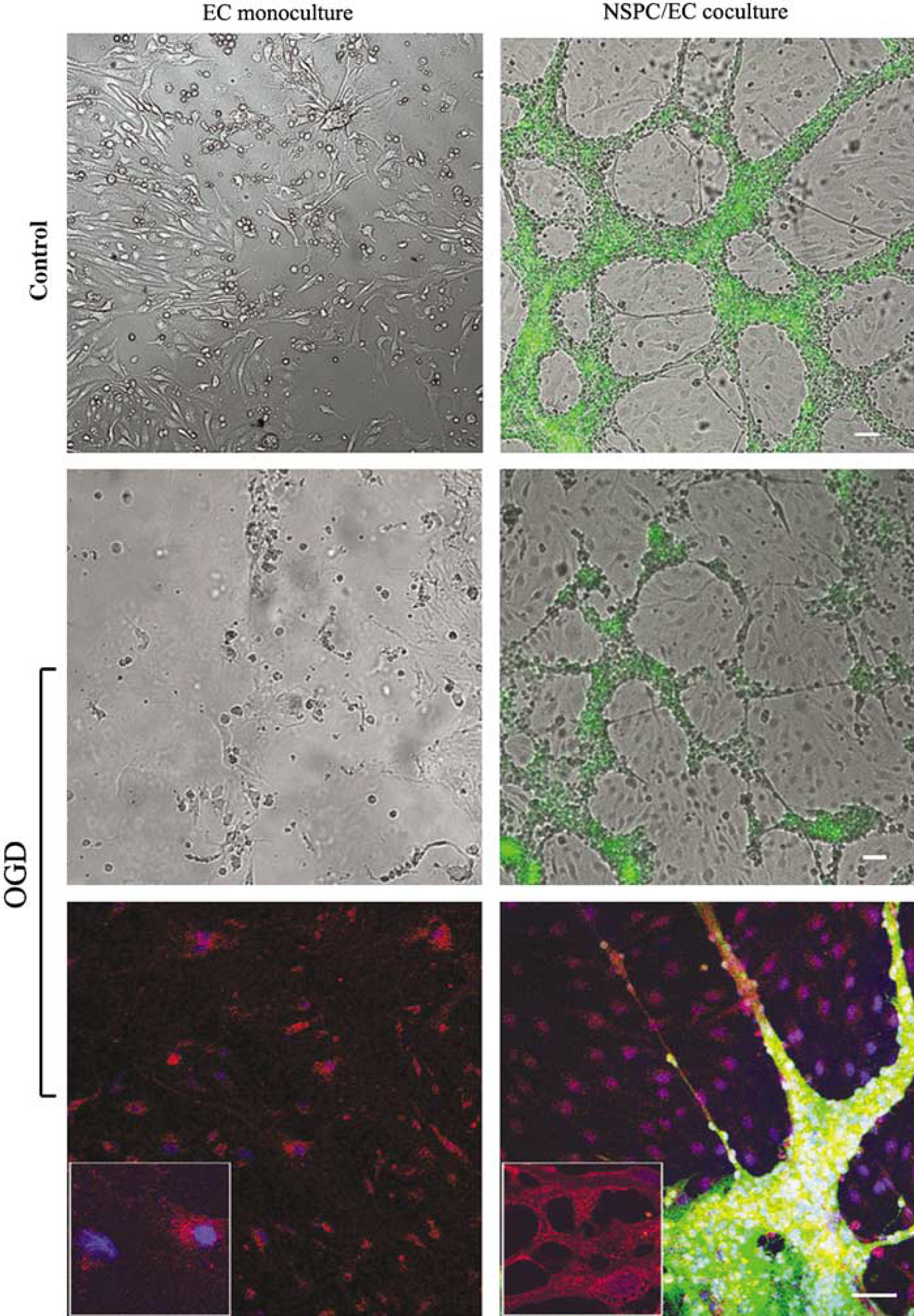
Neural stem/progenitor cells support morphology and survival of ECs after oxygen-glucose deprivation (OGD). Direct NSPC/EC cocultures and EC cultures (both grown in serum-free and growth factor-free medium) were subjected to 3-hour OGD and analyzed at 7 days after OGD (middle and bottom panels). Control cultures (top panel), grown in the same experimental conditions, were not subjected to OGD. Endothelial cell morphology was assessed using DIC confocal microscopy imaging and immunostaining with the antibody against CD31/PECAM-1 (bottom panels). Enhanced green fluorescent protein-neural stem/progenitor cells are shown on the green channel. Bars = 50 μm.
Hypoxia-Inducible Factor-1α and Vascular Endothelial Growth Factor are Constitutively Expressed by Cultured Neural Stem/Progenitor Cells and Become Upregulated after Oxygen and Glucose Deprivation
To determine whether NSPCs respond to OGD via increased expression of the transcription factor, HIF-1α, and its downstream target, VEGF, we used western blot analysis, enzyme-linked immunoassay, and immunofluorescence (Figure 4). Neural stem/progenitor cells constitutively expressed of both HIF-1α and VEGF under normoxic conditions, as shown by western blot and immunoassay (Figures 4A and 4B). Furthermore, both HIF-1α expression and VEGF release were increased approximately twofold in response to 3 h exposure to OGD (Figure 4B). Immunostaining of NSPC cultures under normoxic conditions also showed prominent expression of VEGF and HIF-1α (Figures 4C and 4D). The pattern of VEGF immunofluorescence appeared vesicular within the cytoplasm, whereas HIF-1α immunofluorescence was present in both the cytoplasm and nucleus of many cells. The nuclear localization of HIF-1α, confirmed by colocalization with DAPI nuclear staining (Figure 4D, arrow), suggests transcriptional activity. Western blotting showed no detectable HIF-1α or VEGF in EC monocultures (Figure 4A). These results indicate that cultured NSPCs constitutively express both HIF-1α and VEGF, and respond to ischemic conditions
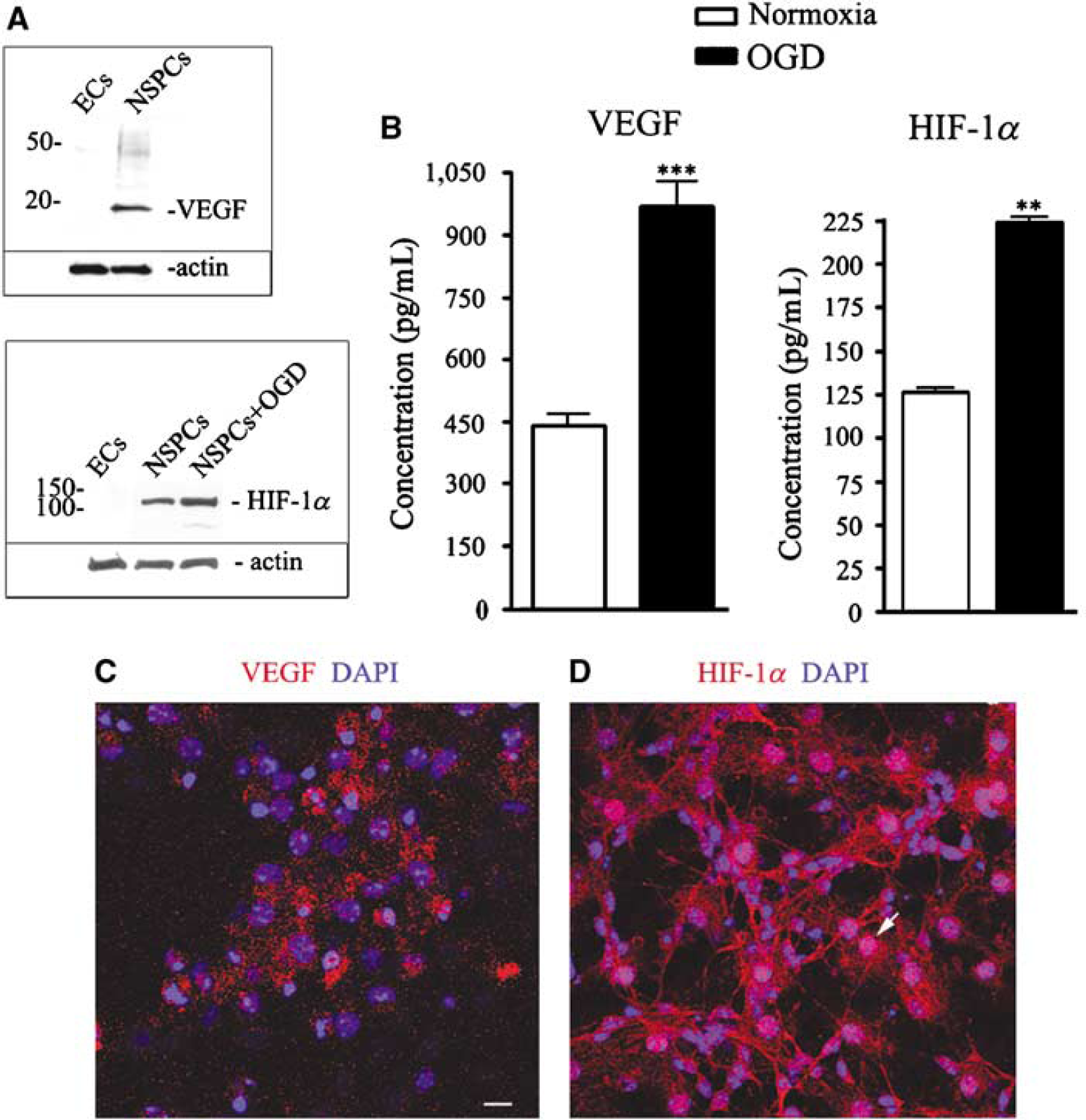
Neural stem/progenitor cells constitutively express both HIF-1α and VEGF in culture, and upregulate expression after exposure to OGD. (
These results also indicate that NSPCs show increased resistance to OGD, compared with ECs, as evidenced by their robust morphologic appearance and upregulated expression of HIF1-α and VEGF at 3 days after the 3 h OGD exposure. Unlike ECs, NSPCs grown in monoculture survive 7 days after a 3-h OGD exposure, and maintain their ability to differentiate (Supplementary Figure 1D).
Inhibition of Vascular Endothelial Growth Factor Signaling Impairs Neural Stem/Progenitor Cell-Mediated Survival and Morphogenesis of Endothelial Cells after Serum Deprivation
To determine whether diffusible VEGF mediates the protective effects of NSPCs against serum withdrawal, ECs were incubated for 3 days in serum-free medium conditioned by NSPCs in the presence or absence of the VEGFR2 tyrosine kinase inhibitor SU1498 (5 to 20 μmol/L) or Flt-1-Fc (30 ng/mL). Flt-1-Fc is a fusion protein comprising human IgG-Fc fragment fused to the high-affinity ligand-binding ectodomain of VEGFR1, and acts as a decoy receptor to inhibit VEGF signaling. As shown in Figure 5 (open bars), NSPC conditioned media (NSPC-CM) protected ECs against serum withdrawal, and SU1498 potently inhibited this protection in a dose-dependent manner with 55% inhibition beginning at the lowest dose of 5 μmol/L. Exposure of ECs to 20 μmol/L SU1498 for the same duration had no effect on EC survival, indicating that SU1498 is not directly toxic at the concentrations used in this experiment (data not shown). Similarly, incubation with Flt-1-Fc significantly reduced the protective effect of NSPC-CM by approximately 50%.
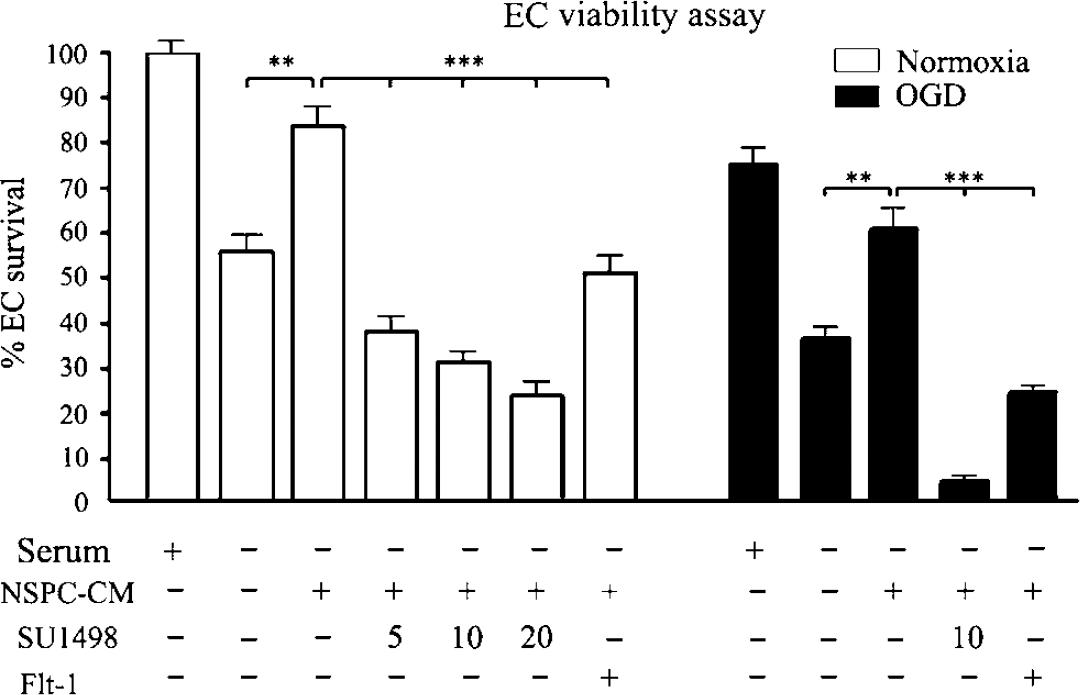
Inhibition of VEGF signaling impairs NSPC-mediated survival and morphogenesis of ECs after serum deprivation and OGD. (open bars, left) Viability of ECs after 3 days of culture in the presence or in the absence of serum, NSPC-conditioned medium (NSPC-CM), or the VEGF inhibitors, SU1498 or FIt-1-Fc. (filled bars, right) Viability of ECs exposed to 3 h of OGD, followed by 3 days of incubation with or without serum, NSPC-CM, or VEGF inhibitors. Percent viability in each culture condition was compared with the viability of EC cultures grown under normoxic conditions in the presence of serum, which was set to 100% (far left open bar = control). Neural stem/progenitor cell-CM represents media conditioned for 3 days by monocultures of NSPCs grown in serum-free and growth factor-free media. Data were acquired using the MTT colorimetric viability assay as described under Materials and methods section, **
To determine whether VEGF signaling is obligatory for NSPC-mediated protection against OGD, ECs were exposed to 3 h of OGD followed by growth in NSPC-CM with or without SU1498 or Flt-1-Fc for 3 days. Both SU1498 (10 μmol/L) and Flt-1-Fc (30 ng/mL) potently inhibited the protective effects of NSPC-CM under conditions of OGD (Figure 5, filled bars). The vasculotrophic effect of NSPC-CM against OGD was not influenced by incubation with control IgG-Fc. Taken together, these data suggest that VEGF signaling mediates the protective effects of NSPC-CM against both serum starvation and OGD conditions
Neural Stem/Progenitor Cells Provide Vasculotrophic Support against Mild Focal Ischemia after Intracerebral Transplantation
To determine whether NSPCs are vasculotrophic
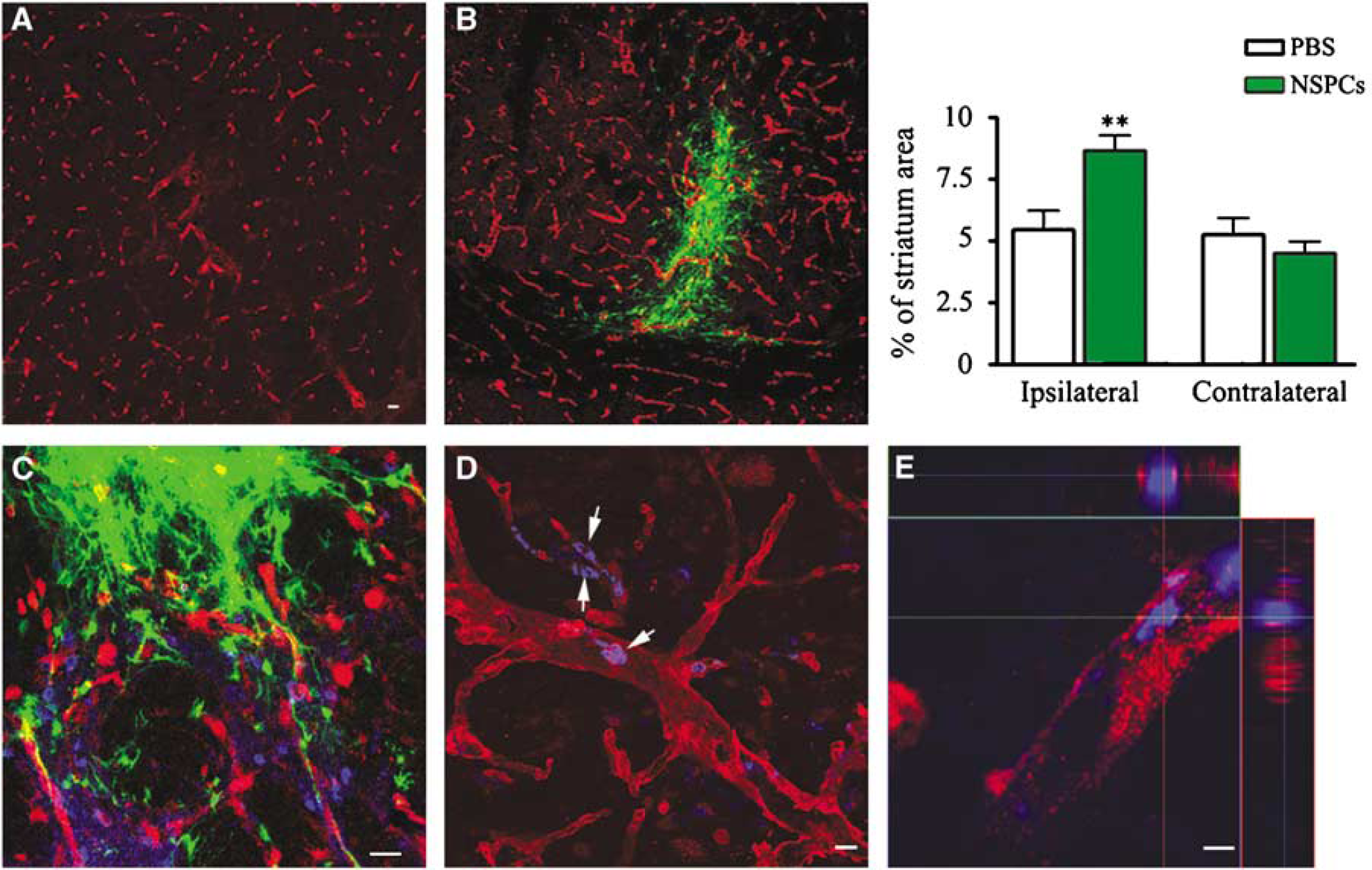
Neural stem/progenitor cells support brain revascularization after ischemia. (
Discussion
In this study, we explored the vasculotrophic effects of embryonic NSPCs under conditions of ischemia in culture and after intracerebral transplantation in a mouse model of focal cerebral ischemia. We found that NSPCs are vasculotrophic under both
Although several
Our initial observations that NSPCs release a diffusible substance that promotes capillary tube formation and provides robust protection of ECs against OGD naturally led us to investigate the expression and function of VEGF in our
Our observation that NSPCs appear relatively resistant to OGD
Much evidence suggests a functional link between neurogenesis and angiogenesis in rodent models of stroke. In rodent models of focal and global ischemia, reactive astrocytes and blood vessels appear to provide the physical substrate for neuroblast migration (Ohab et al, 2006; Thored et al, 2007; Yamashita et al, 2006), and microarray analysis has shown concomitant upregulation of neurogenic and angiogenic genes within adult SVZ after focal ischemia in mice (Liu et al, 2007). These studies and others have lead to the notion that angiogenesis provides the necessary cues for the neurogenic response to stroke. Our studies suggest that activated NSPCs may provide a source of angiogenic factors that enhance the growth and/or stabilization of newly formed vasculature after stroke. Our studies showing increased vascularization after intracerebral transplantation of NSPCs into the adult mouse brain support this. Other studies have also shown increased vascularization after intracerebral transplantation of adult rat NSPCs after embolic stroke (Jiang et al, 2005) and increased vascularization after transplantation of human NSPCs genetically modified to overexpress VEGF in a mouse model of hemorrhagic stroke (Lee et al, 2007). Interestingly, our studies also suggest that NSPCs can adopt an endothelial lineage after exposure to ECs in coculture. That direct contact with ECs can convert NSPCs into the endothelial lineage through cell fusion-independent differentiation was previously indicated by Wurmser et al (2004a), raising the possibility that NSPCs may also contribute to angiogenesis after stroke by adopting an EC fate and direct incorporation into nascent vasculature. Because neurogenic and angiogenic processes are governed by common growth factors, including VEGF, fibroblast growth factor-2 (FGF-2), stromal derived growth factor 1 (SDF1), and angiopoietin 1 (Ang1) (for review, see Ward and Lamanna, 2004), deciphering the selective role of NSPC-derived factors in promoting angiogenesis after stroke using pharmacological inhibitors
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
