Abstract
Inflammation plays an important role in the pathogenesis of stroke. The differential expression of inflammatory and angiogenic factors in thrombi and plasma remain undefined. In this observational cohort study, we evaluated angiogenic factors and inflammatory cytokines, in cerebral thrombi, local cerebral plasma (CP), and peripheral plasma (PP) in patients with acute ischemic stroke. Protein analysis of thrombi, CP and PP were used to measure angiogenic and inflammatory proteins using electrochemiluminescence. Our data indicate that VEGF-A, VEGF-C, bFGF, IL-4, IL-13, IL-1β, IL-2, IL-8, IL-16, IL-6 and IL-12p70 were higher in the thrombi of acute ischemic stroke patients than in the CP and PP of stroke patients. Moreover, the protein levels of GM-CSF were lower in the PP than in the CP and the clot. Moreover, VEGF-D, Flt-1, PIGF, TIE-2, IL-5, TNF-β, IL-15, IL-12/IL-23p40, IFN-γ and IL-17A were higher in PP and CP than in thrombi. Our results show that cytokines mediating the inflammatory response and proteins involved in angiogenesis are differentially expressed in thrombi within the cerebral and peripheral circulations. These data highlight the importance of identifying new biomarkers in different compartments of the circulatory system and in thrombi that may be used for the diagnosis and treatment of stroke patients.
Introduction
Acute ischemic stroke (AIS) is a leading cause of morbidity and mortality worldwide. 1 However, current management of AIS is limited to intravenous thrombolysis within 4.5 hours of symptom onset and/or endovascular thrombectomy. 2 While early reperfusion is associated with a significant improvement in outcomes, many patients are ineligible, lack access, or remain significantly disabled or deceased despite treatment. Thus, it remains critical to identify biomarkers that may be used to predict outcomes and the effectiveness of treatment modalities as well as to provide a greater understanding of the pathophysiology underlying AIS.
The inflammatory response is a well-known contributor to stroke pathology,3,4 and inflammatory cytokines have been described in both serum and plasma. 5 In addition, the oxygen depletion associated with AIS activates endogenous mechanisms aimed at restoring the lower oxygen availability to the infarcted area in the brain. These compensatory mechanisms are mediated in part by angiogenic proteins that contribute to improved outcomes on AIS patients.6,7 However, a detailed evaluation of the expression levels of inflammatory cytokine and angiogenic factors after AIS has not been performed.
The expression of inflammatory proteins in body fluids during the acute phase of brain injury appears to influence long term outcomes in stroke and traumatic brain injury.8–11 However, in order to develop new therapies and to identify biomarkers that may be used to monitor the health of these patient population, it is imperative to perform a thorough characterization of the inflammatory protein profile associated with large vessel occlusion stroke (LVOS). 12 In the present study, we determined the neuroinflammatory-vascular protein profile of cytokines and angiogenic proteins in the cerebral plasma (CP), thrombi and peripheral plasma (PP) of patients to characterize the inflammatory and angiogenic responses that contribute to the pathology of AIS in the brain at the level of the clot, and in the cerebral and systemic circulation.
Materials & methods
Participants & thrombectomy
Plasma and clot samples were obtained by written informed consent from a total of 40 patients with AIS with a median age of 75.5 years old (Range: 34 to 92 years old) admitted to Jackson Memorial Hospital/University of Miami Hospital between November 2018 and November 2019 (Table 1). All patients with age ≥18 years old who presented with acute stroke and underwent thrombectomy with retrieval of thrombus material were eligible for the study. Ethics approval was approved by the Institutional Review Board at the University of Miami (IRB 20160699), according to the ethical standards of the responsible committee on human experimentation at the University of Miami and in compliance with the Declaration of Helsinki. Informed consent was obtained from all patients included in this study. Patients were excluded if adequate thrombus material could not be obtained, or the patient/legal representative refused to participate in the study. Patient demographics, clinical presentation, neurological exam (National Institutes of Health Stroke Scale (NIHSS)), pre-procedural imaging results, intravenous tissue plasminogen activator (IV-tPA) administration, procedural details including number of passes, thrombectomy technique used, recanalization results (Thrombolysis in Cerebral Infarction (TICI) scale, and follow-up data were collected. Clots were collected by mechanical thrombectomy performed or supervised by board-certified neurointerventional experts under biplane neuroangiography (Artis Q, Siemens Healthcare, Erlangen, Germany) as described in. 13 PP was taken from a sheath in the femoral or radial artery, and CP from a catheter in the cervical internal carotid artery proximal to the site of occlusion. To address potential sources of bias, samples used in this study were collected from patients regardless of sex and ethnicity as long as they met the inclusion criteria as described. Groups in this study correspond to thrombi of patients with AIS, plasma taken from the peripheral circulation (PP) and from the cerebral circulation (CP). PP and CP analyzed in this study were collected at the conclusion of the thrombectomy procedure.
Characteristics of patients used in this study.

tPA: tissue plasminogen activator; mRS: modified ranking score; LKN: last known normal; TICI: thrombolysis in cerebral infarction.
Protein array
Investigators were blinded to the experimental groups in this study. A multiplex assay using the V-Plex Neuroinflammation Panel 1 Human Kit (Cat # K15210D, MSD) was carried out to analyze the protein levels in pg/mL of VEGF-A, VEGF-C, VEGF-D, Flt-1, PIGF, TIE-2, bFGF, IL-4, IL-5, IL-13, IL-10, GM-CSF, IL-1α, IL-1β, TNF-α, TNF-β, IL-8, IL-2, IL-7, IL-15, IL-16, IL-12/IL-23p40, IFN-γ, IL-6, IL-17A and IL-12p70 according to manufacturer instructions (MSD) using the MESO-QuickPlex SQ-120 as described in. 14 Briefly, for the angiogenesis panel, 150 µL of Blocker A solution were added to each well followed by incubation for 1 hr and 3 washing steps in Wash Buffer prior to sample incubation. For all assays (angiogenesis and cytokine panels), 50 µL of either diluted sample or calibrator were added to each well followed by incubation for 2 hrs and 3 washing steps. Samples were then incubated in detection antibody for 2 hrs followed by 3 washings steps and addition of 2X Read Buffer to each well. The plate was then analyzed in the MESO-QuickPlex SQ-120 (MSD). Prior to analysis, protein lysates of clots were obtained by lysing the clots in 150 µL of lysis buffer containing protease inhibitor cocktail (Sigma) as described in. 15 Plasma samples collected in EDTA tubes were used for the CP and PP groups.
Statistical analyses
Statistical Analyses were carried out with Prism 9.0 software (GraphPad Prism). For statistical computations, first outliers were identified by the ROUT method (Q = 1%) and then removed from further analyses. Normality was tested by the Shapiro-Wilk Test. Since data in this study were not normally distributed, data were analyzed by the Kruskal-Wallis test. Furthermore, to decrease the false-positive results, the two-stage linear step-up procedure of Benjamini, Krieger and Yekutieli was used to adjust the P values into false discovery rates (FDR). A FDR corrected p < 0.05 (5%) was set as the significance threshold. Power analysis was carried out using G*Power 3.1.9.6 with alpha set to 0.5 and a power to 0.8 for 3 groups. The following multiple comparisons were carried out in this study: Clot vs peripheral plasma (PP), Clot vs cerebral plasma (CP) and PP vs CP. Statistical significance was set at p < 0.05. Data in this study are presented as box plots representing the 5th and 95th percentile. Simple linear regression analysis to predict the protein levels in the CP using the protein levels in the PP for each analyte was fit using R studio software (version 1.2.5033). Furthermore, those linear regression models that were statistically significant were used to identify analytes that then underwent machine learning approaches by using supervised learning to fit predictive models using the CARET package in R setting the seed at 40, with an 80% split with a trainControl method of repeated cross validation (repeatedcv) with 5 repeats performed 5 times.
Results
Vascular endothelial growth factor (VEGF) expression patterns in thrombi and plasma from patients with AIS
VEGF has been implicated in stroke pathology associated with atherosclerosis. 16 To determine the expression patterns of this key angiogenic factor, we analyzed the expression levels of VEGF-A (Figure 1(a)), VEGF-C (Figure 1(b)) and VEGF-D (Figure 1(c)) in the plasma and clots of AIS patients. Accordingly, the levels of VEGF-A were lower in the CP (95% confidence interval: 0.05203–0.1057) than in the clot (0.2463–0.4775), whereas the levels of VEGF-A in PP (0.1047–0.4658) were as elevated as in the clots of AIS patients (Figure 1(a)). In addition, the levels of VEGF-C were lower in CP (0.08803–0.1972) when compared to clots (13.90–34.08) (Figure 1(b)), whereas the levels of VEGF-C in PP (1.246–7.920) were not significantly different to the CP and the clot. Interestingly, the levels of VEGF-D were higher in the PP (36.82–152.1) and the CP (15.42–42.24) when compared to the clots (0.06998–0.1394) of AIS patients (Figure 1(c)). These findings indicate a differential role in the angiogenic response by VEGF following AIS.
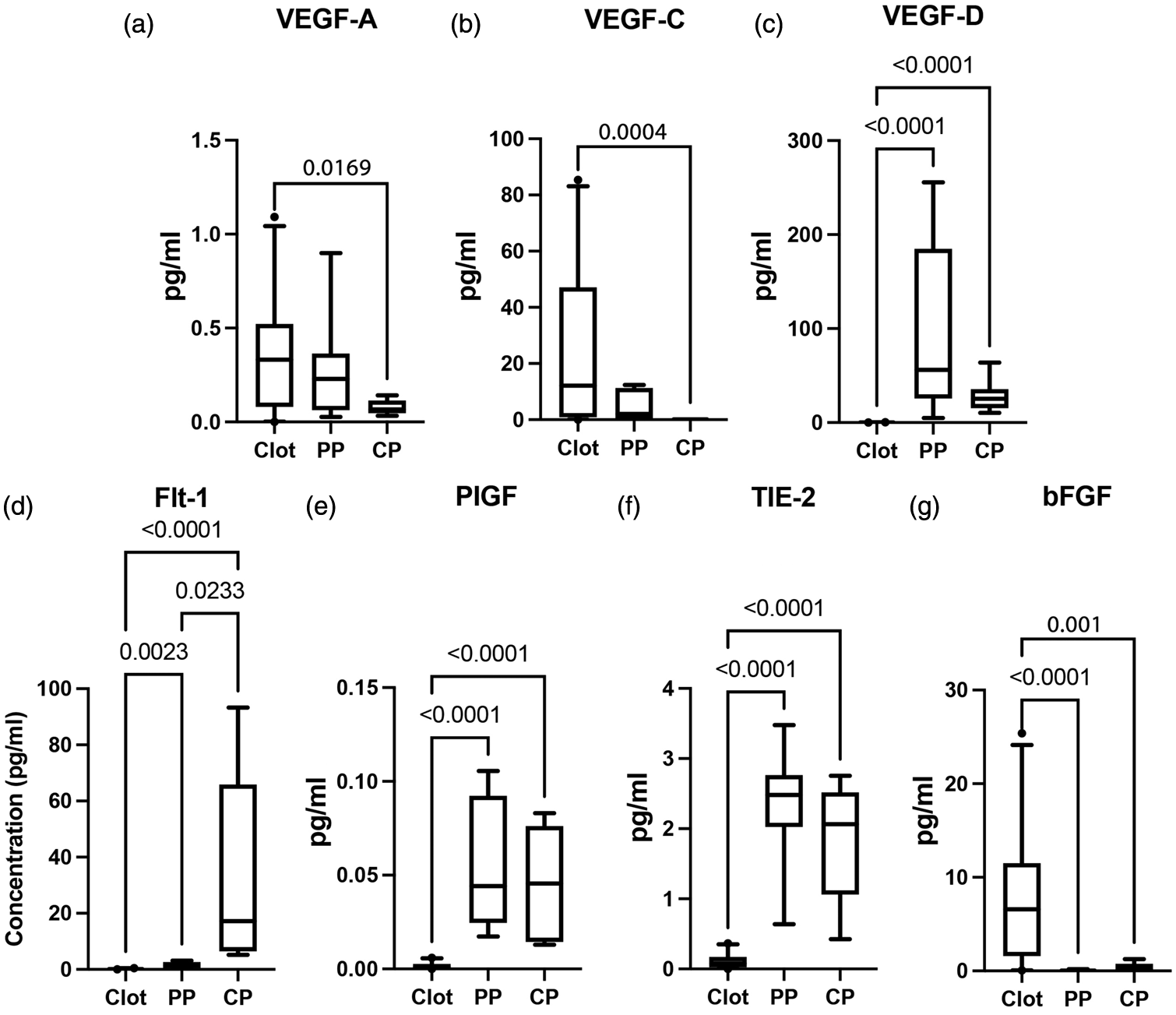
VEGF profile, Flt-1, PlGF, TIE-2 and bFGF in clots and plasma of AIS patients: Protein expression levels of (a) VEGF-A: Clot: 29, PP: 11, CP: 10; (b) VEGF-C: Clot: 31, PP: 12, CP: 8; (c) VEGF-D: Clot: 34, PP: 12, CP: 10. (d) Flt-1: Clot: 25, PP: 8, CP: 12, (e) PIGF: Clot: 27, PP: 12, CP: 12, (f) TIE-2: Clot: 25, PP: 12, CP: 12 and (g) bFGF: Clot: 30, PP: 8, CP: 10. Box and whiskers are shown for the 5th and 95th percentile.
Flt-1, PlGF TIE-2 and bFGF present distinctly different patterns of protein expression in thrombi and plasma of AIS patients
FMS-like tyrosine kinase (Flt-1) is the receptor of VEGF 17 and Placental Growth Factor (PlGF) interacts with Flt-1, 16 and high levels of the angiopoietin receptor TIE-2 have been implicated in reducing blood brain barrier (BBB) leakage and promoting vascular stabilization, 18 and basic fibroblast growth factor (bFGF) is a potent angiogenic stimulator that has been shown to be neuroprotective in rodent models of stroke. 19 To determine the expression patterns of these angiogenic proteins in AIS, the clots and plasma of AIS patients were analyzed. Accordingly, the protein levels of Flt-1 (Figure 1(d)) and PIGF (Figure 1(e)) were higher in PP (Flt-1: (0.6005–2.407), PIGF (0.03240–0.07666)) and CP (Flt-1: (11.83–56.13), PIGF (0.02837–0.06477)) than in clots (Flt-1: (0.07126–0.1963), PIGF (0.001052–0.002393)). In addition, the protein levels of Flt-1 were higher in the CP (11.83–56.13) than in the PP (0.6005–2.407). Interestingly, the expression pattern of TIE-2 and bFGF was inversely proportional such that TIE-2 was more elevated in the PP (1.781–2.800) and CP (1.265–2.335) than in clots (0.06657–0.1504) (Figure 1(f)), and bFGF was elevated in the clots (4.922–10.47) while the protein levels in the PP (0.003052–0.1100) and CP (0.09145–0.7669) were very low (Figure 1(g)). These findings suggest a differential role of these angiogenic proteins in clot than in circulation.
TH2 response proteins in thrombi and plasma of patients with AIS
Interleukin (IL)-4, IL-5. IL-10 and IL-13 have been recently shown to be elevated in the serum of severe stroke patients when compared to mild cases. 5 Here we determined the protein levels of the TH2 cytokines IL-4 IL-5, and IL-10 as well as IL-13. 20 The protein levels of IL-4 were higher in the clot (8.097e-005–0.0002304) than in the PP (7.691e-009–1.479e-007) and CP (2.154e-008–6.499e-007) (Figure 2(a)), whereas the levels of IL-5 were higher in the PP (3.424e-005–0.0001188) and CP (3.371e-005–0.0001230) of AIS patients when compared to the protein levels in clots (1.097e-005–1.753e-005) (Figure 2(b)). In addition, similarly to IL-4, IL-13 levels were significantly higher in the clots (0.02092–0.04193) of patients with AIS when compared to the cerebral (4.082e-005–0.0001218) and PP (4.115e-005–0.0001359) (Figure 2(c)). There was also a trend of higher expression levels of IL-10 (Figure 2(d)) in the clots (9.052e-005–0.0001989) of AIS patents when compared to CP (2.754e-005–6.303e-005) and PP (2.648e-005–9.505e-005); however, there was no statistical significance for this protein. These findings indicate that of these TH2 cytokines, IL-4 and IL-13 play a role in clot formation, probably associated with the fibrin that is characteristic of clots and IL-5 in the anti-inflammatory response in the CP and PP. In addition, the cytokine granulocyte-macrophage colony-stimulating factor (GM-CSF) has been shown to have neuroprotective and anti-inflammatory properties after stroke, 21 and our data indicate that GM-CSF is expressed at lower levels in the PP (84.77–136.0) than in the CP (134.2–142.8) and the clot (130.5–135.1) of patients with AIS. Moreover, the protein levels of GM-CSF were higher in the CP(134.2–142.8) than in the clot (130.5–135.1) of AIS patients (Figure 2(e)).
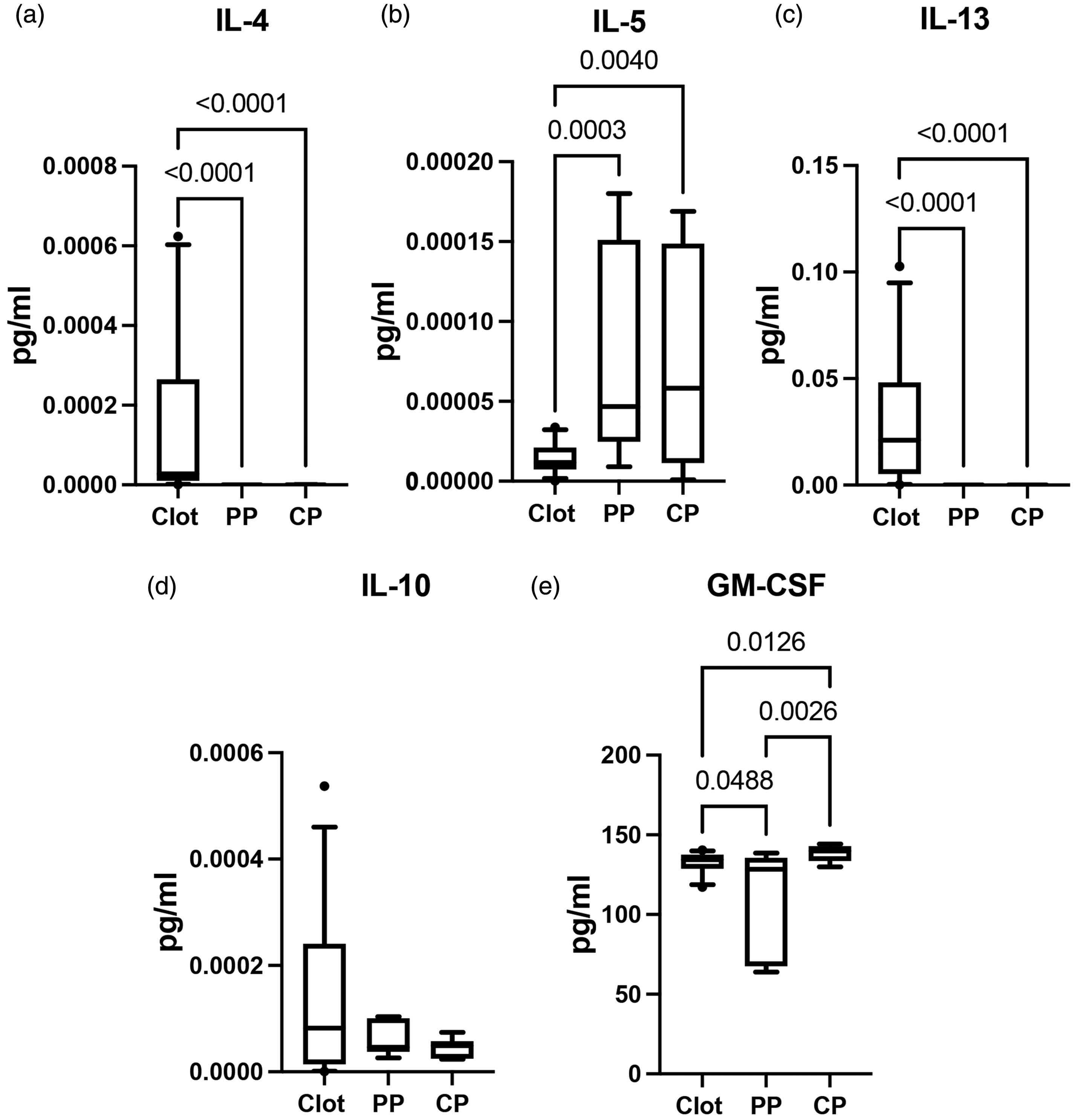
TH2 profile in clots and plasma of AIS patients: Protein expression levels of (a) IL-4: Clot: 28, PP: 6, CP: 7; (b) IL-5: Clot: 33, PP: 12, CP: 12; (c) IL-13: Clot: 34, PP: 11, CP: 9. (d) IL-10: Clot: 32, PP: 6, CP: 7 and (e) GM-CSF: Clot:28, PP:9, CP:8. Box and whiskers are shown for the 5th and 95th percentile.
TH1 and TH17 response in thrombi and plasma of AIS patients
IL-1 and tumor necrosis factor (TNF) cytokines are two of the most common study family of cytokines, both of which have been implicated in stroke.22,23 In this study we determined the role of the IL-1 cytokines IL-1α (Figure 3(a)) and IL-1β (Figure 3(b)) as well as the TNF cytokines TNF-α (Figure 3(c)) and TNF-β (Figure 3(d)). Of the cytokines analyzed, we found that IL-1β was higher in the clot (0.01224–0.03937) than in the cerebral (−1.436e-007–5.152e-006) and PP (1.417e-007–1.806e-006) of AIS patients (Figure 3(b)), whereas the levels of TNF-β were statistically significantly higher in the PP (2.082e-006–7.016e-006) and the CP (3.663e-006–1.394e-005) than in the clots (2.448e-007–7.586e-007) (Figure 3(d)). However, no statistical difference was detected for IL-1α (Figure 3(a)) and TNF-α (Figure 3(c)) between clots (IL-1α: (0.001231–0.002962), TNF-α (0.004689–0.01052)) and the cerebral (IL-1α: (0.0004170–0.003472), TNF-α (0.002617–0.01216)) and PP (IL-1α: (0.001104–0.004322) TNF-α (0.004938–0.009222)), indicating a role for IL-1β in the inflammatory response present in clots.
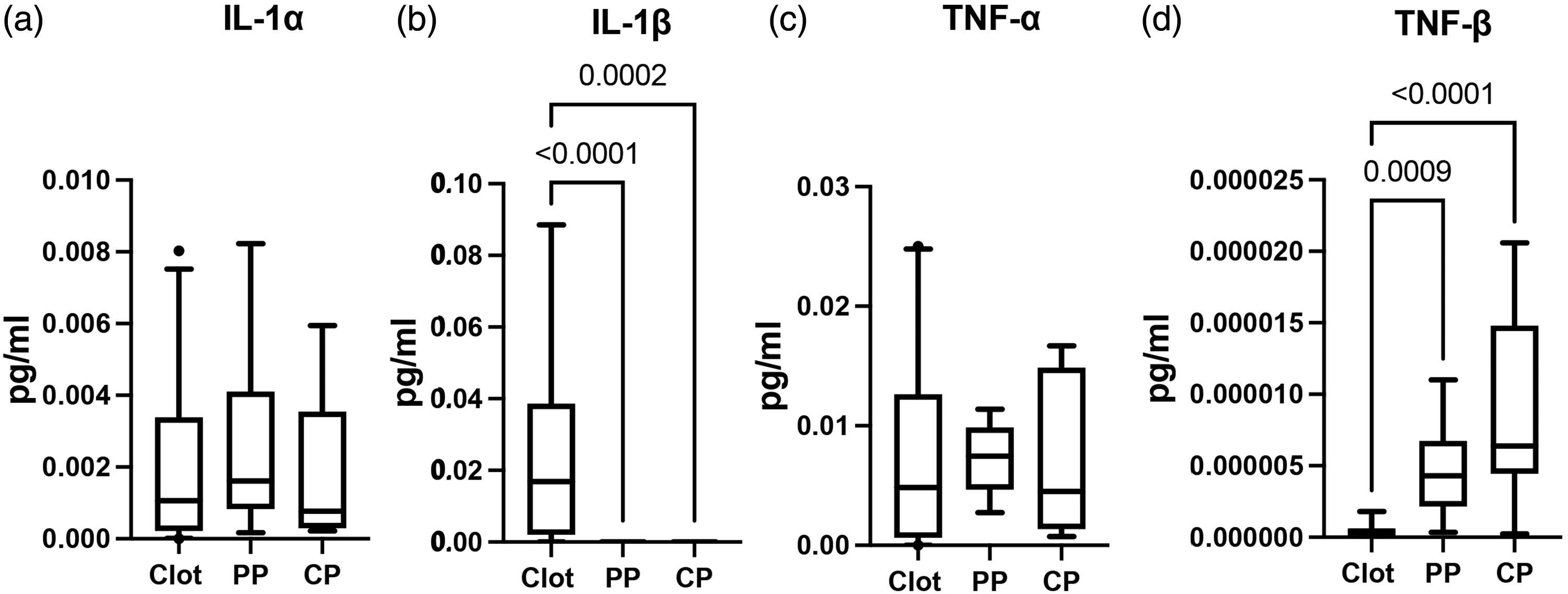
IL-1 and TNF cytokines in AIS patients: Protein expression levels of (a) IL-1α: Clot: 30, PP: 12, CP: 10; (b) IL-1β: Clot: 17, PP: 7, CP: 7; (c) TNF-α: Clot: 31, PP: 10, CP: 10 and (d) TNF-β: Clot: 17, PP: 9, CP: 9. Box and whiskers are shown for the 5th and 95th percentile.
Serum cytokines show a prognostic value in AIS. 5 Thus, we further analyzed the levels of IL-2, IL-7, IL-8, IL-15 and IL-16 in the clots and plasma of AIS patients and found that IL-2 (Figure 4(a)), IL-8 (Figure 4(c)) and IL-16 (Figure 4(e)) were higher in the clots (IL-2: (0.0002443–0.0006013), IL-8: (2284–3573), IL-16: (451.0–1021)) than in the cerebral (IL-2: (3.430e-006–1.018e-005), IL-8: (0.01152–0.01342), IL-16: (2.029–6.275)) and PP (IL-2: (1.753e-006–4.750e-006), IL-8: (0.002021–0.09296), IL-16: (3.026–8.679)). Similarly, although not statistically significant, IL-7 (Figure 4(b)) showed a higher trend of expression in clots (0.02457–0.05152) when compared to PP (0.005444–0.02735) and CP (0.006941–0.04112). In contrast, IL-15 (Figure 4(d)) was higher in the PP (0.001980–0.004511) and CP (0.001373–0.004364) than in clots (0.0003654–0.001040).
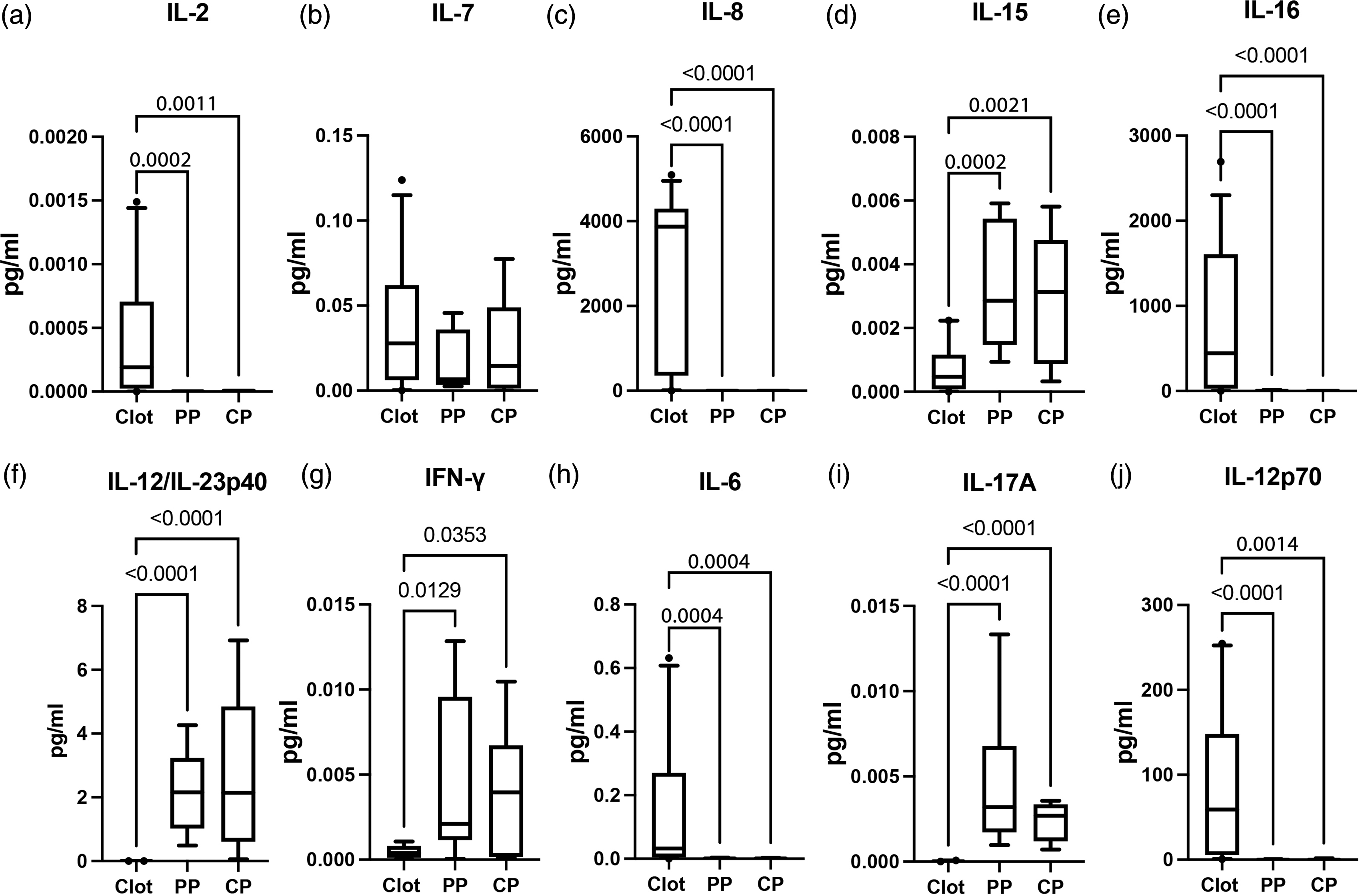
Inflammatory cytokines in AIS patients: Protein expression levels of (a) IL-2: Clot: 32, PP: 8, CP: 8; (b) IL-7 Clot: 31, PP: 12, CP: 12; (c) IL-8: Clot: 35, PP: 8, CP: 6. (d) IL-15: Clot: 21, PP: 12, CP: 10. (e) IL-16: Clot: 33, PP: 12, CP: 10. (f) IL-12/IL-23p40: Clot: 24, PP: 10, CP: 10; (g) IFN-γ: Clot: 18, PP: 10, CP: 10; (h) IL-6: Clot: 24, PP: 10, CP: 10. (i) IL-17A: Clot: 27, PP: 10, CP: 10 and (j) IL-12p70: Clot: 29, PP: 7, CP: 5. Box and whiskers are shown for the 5th and 95th percentile.
Moreover, IL-12 stimulates TH1 cells to produce IFN-γ, and IL-6 and IL-12/IL-23p40 stimulate TH17 cells to produce IL-17. Here we analyzed the levels of these proteins in the clots and plasma of these patients to identify the TH1 and TH17 response at distinct sites after AIS. Here we found that the protein levels of IL-12/IL-23p40 (Figure 4(f)), IFN-γ (Figure 4(g)) and IL-17A were elevated (Figure 4(i)) in the CP (IL-17: (0.001562–0.003110), IFN-γ: (0.001183–0.006873), IL-12/IL-23p40: (0.8944–4.559)) and PP (IL-17: (0.001834–0.007485), IFN-γ: (0.001322–0.008537), IL-12/IL-23p40: (1.249–3.112)) of AIS patients when compared to the clot (IL- 17: (1.507e-005–3.098e-005), IFN-γ: (0.0002627–0.0006305), IL-12/IL-23p40: (8.747e-005–0.0002035)). In contrast, we found that the levels of IL-6 (Figure 4(h)) and IL-12p70 (Figure 4(j)) were elevated in clots (IL-6: (0.07290–0.2460), IL-12p70: (47.02–111.2)) when compared to CP (IL-6: (0.0007618–0.001950), IL-12p70: (0.2186–1.341)) and PP (IL-6: (0.0005976–0.002383), IL-12p70: (0.04312–0.3419)). Taken together, these findings indicate the presence of TH1 and TH17 responses in the plasma of AIS patients.
Linear regression model to predict inflammation and angiogenesis in cerebral plasma from peripheral plasma
To determine the approximate contribution of each protein in the peripheral plasma to the inflammatory and angiogenic response by the same protein in the cerebral plasma, we fit a simple linear regression model (Table 2). Accordingly, we found that the protein levels of bFGF, IL-5, TNF-α, IL-7, IL-15, IL-12/IL-23p40, IFN-γ and IL-6 in the peripheral circulation significantly contribute to explain the levels of those same proteins in the cerebral circulation with an adjusted R2 of approximately 0.63, 0.69, 0.60, 0.81, 0.34, 0.59, 0.89 and 0.96, respectively (Table 2). However, we did not find a statistically significant contribution in the linear regression models fit between CP and PP for the other proteins analyzed, suggesting that bFGF, IL-5, TNF-α, IL-7, IL-15, IL-12/IL-23p40, IFN-γ and IL-6 in the PP can be used to predict the cerebral inflammatory and angiogenic profile. Accordingly, we then carried out a supervised approach of machine learning by fitting a linear model with a repeated CV of 5 repeated 5 times for bFGF, IL-5, TNF-α, IL-7, IL-15, IL-12/IL-23p40, IFN-γ and IL-6 (Table 2), which resulted in models with high Adjusted R2 (above 0.9) when comparing the testing with the training sets.
Simple linear regression results of CP ∼ PP and results of predictive linear models of CP ∼ PP with a repeated CV = 5.
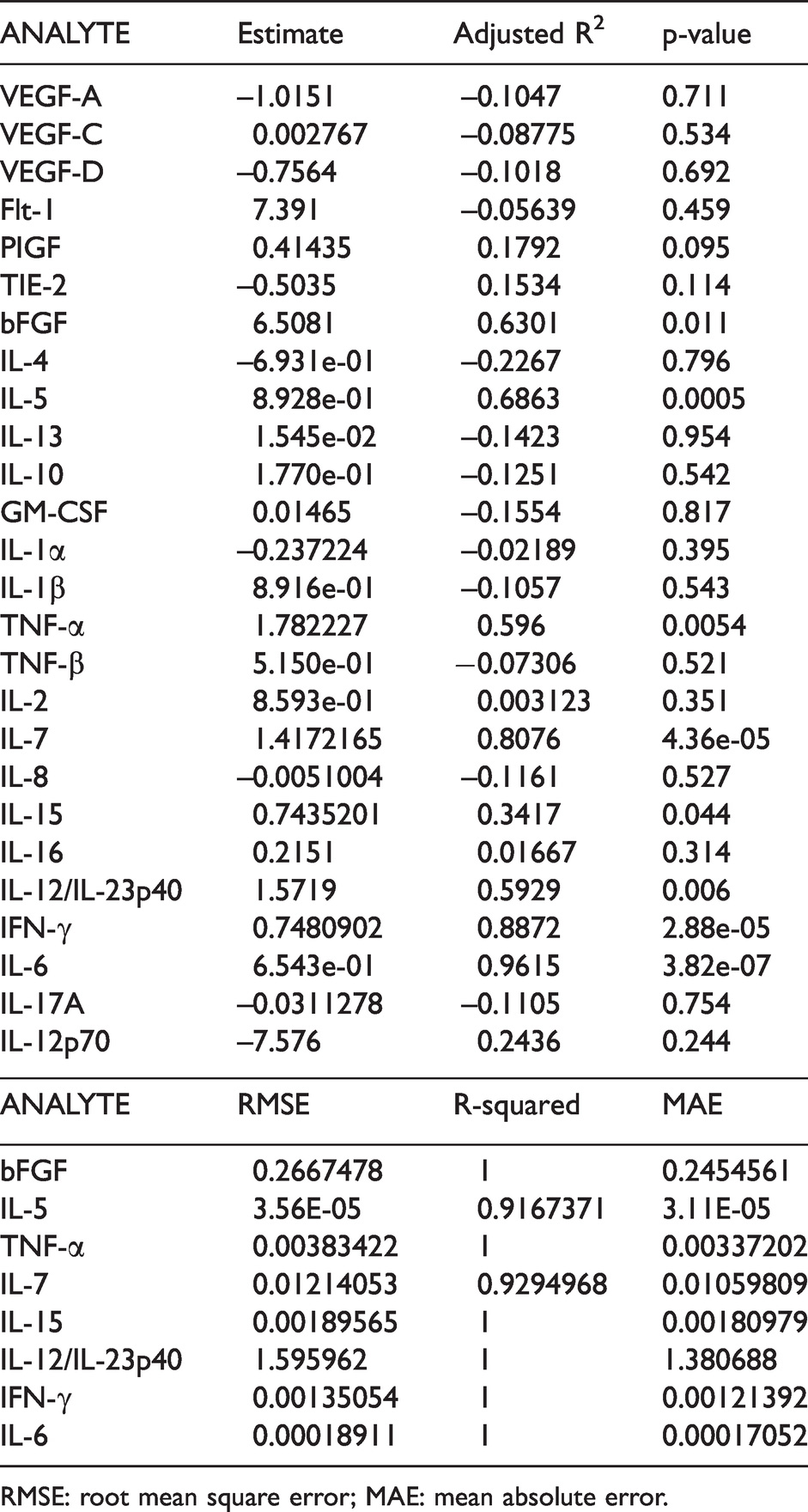
RMSE: root mean square error; MAE: mean absolute error.
Discussion
Several factors contribute to the pathology of AIS including inflammation 11 and angiogenesis. 16 Previous studies have analyzed the protein profile of a variety of proteins that contribute to stroke pathology with the goal of identifying biomarkers that may be used in the care of patients with AIS.12,24 To the best of our knowledge, this study is the first report to evaluate the expression levels of angiogenic and inflammatory proteins in thrombi, CP and PP in patients with AIS (Figure 5). This study presents new clinical data indicating the generation and release of inflammatory and angiogenic mediators in the thrombi as well as the cerebral and PP. The clinical significance of the findings implicates an intravascular mechanism by which an acute thrombotic event leading to focal cerebral ischemia can potentially alter the inflammatory status of downstream and remote circulatory beds, thereby altering vascular and organ function. In addition, the observed differential expression patterns suggest the potential for regional blood biomarker assessments to help guide personalized therapeutic interventions following clot removal to minimize recruiting cerebrovascular events and to maximize cerebral blood flow normalization and function.
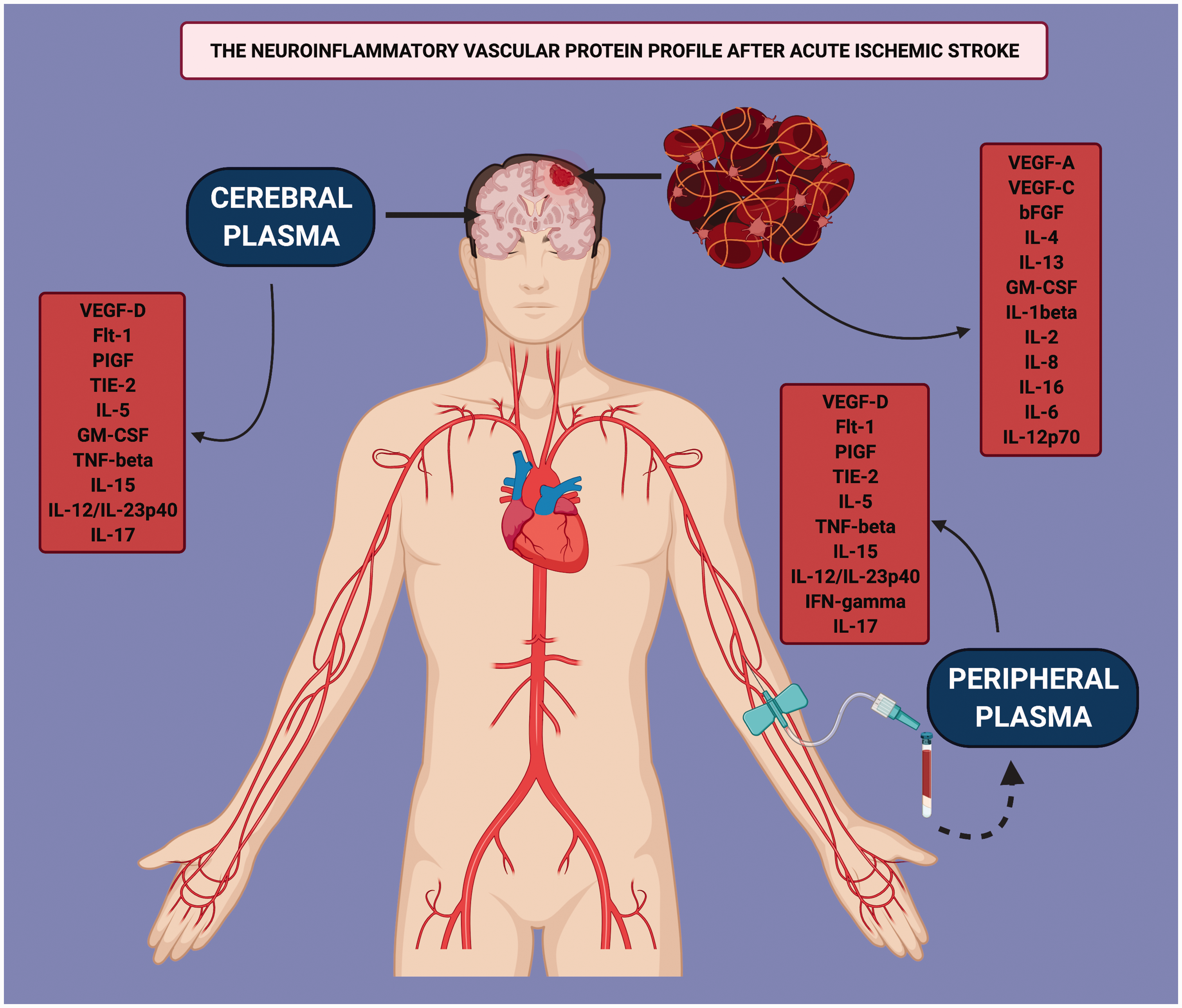
The Neuroinflammatory Vascular Protein Profile in AIS: Differential protein expression in the cerebral plasma, peripheral plasma and the thrombus that characterizes the expression of inflammatory cytokines and angiogenesis-associated proteins in patients with AIS.
In the current study, levels of inflammatory mediators and angiogenic factors were measured in AIS patients undergoing endovascular thrombectomy. The findings of increased levels of these proteins in the thrombus and blood samples implicate a role in the early pathophysiology of the evolving ischemic injury during the early reperfusion phase after mechanical thrombectomy.13,25 The effects of inflammation in stroke has been well characterized.13,22,26 Following cerebral ischemia, there are a variety of sources contributing to the inflammatory response which play a significant role in the extent of the ischemic penumbra. These sources include stagnant blood flow before reperfusion, release of pro- and anti-inflammatory proteins originating in endothelial cells and the brain parenchyma as well as extravasation of leukocytes. 26 In the brain, altered rheology from stagnant blood flow and the shear stress undergone by vascular endothelial cells contribute to leukocyte activation that results in the production of pro- and anti-inflammatory cytokines. 27 In addition, disruption of the blood brain barrier facilitates the transport of cytokines between the peripheral circulation and the cerebral circulation. 26 Acute changes (within 7 days) in inflammatory proteins following AIS have been shown to contribute significantly to long term outcomes in both humans and animal models.8,10 Thus, highlighting the importance of the acute inflammatory response on long term outcomes following injury to the CNS.
We have previously demonstrated that inflammasome activation occurs in cerebral thrombi of patients. 13 Inflammasome-mediated activation of IL-1β may initiate a cascade of proinflammatory cytokines that recruits myeloid cells to the vascular endothelium to initiate remodeling and perpetuate inflammation. 28 IL-1β down-regulates thrombomodulin and impairs protein C activity, thus acting as a procoagulant. In addition, platelets express IL-1-R1 receptor and the presence of its ligand, IL1β, results in platelet hyperactivation and clumping. 29 IL-1β also induces transcription of IL-6. IL-6 regulates the immune response, hemopoiesis, and the acute phase response. 30 IL-6 acts as a procoagulant by activating membrane-bound β-receptor glycoprotein 130 (gp130) in resting platelets, increasing platelet thrombogenicity. In this study, we find that IL-6 is elevated in clots when compared to CP and PP. IL-1 also induces the transcription of TNFα. High levels of TNFα have been shown to be detrimental following cerebral ischemia in rodents 31 and elevated levels have been observed in the plasma of patients after stroke. 32 Here, we found IL-1β to be elevated in the clots when compared to PP and CP, consistent with the presence of inflammasomes and NETs in thrombi, 13 while TNF-β was elevated in the PP and CP when compared to the clot, whereas no difference in IL-1α and TNFα was detected between the three samples.
Recent findings suggest that IL-17, IL-21, and IL-12/IL-23p40 may be important mediators in the post-infarct inflammatory cascade. 33 IL-17 has been reported to be elevated in the serum, plasma and brain of cadavers who presented with stroke and associated with worse outcomes. 34 Here we report that the protein levels of the TH17 inflammatory cytokines IFN-γ and IL-17A were elevated in the plasma compared to clots, whereas IL-6 and IL-12p70 were higher in the clots. Signaling through the IL-23 receptor increases the production of T cells that secrete IL-17. In mice, IL-17 expression increased in the peripheral blood and in the brain in the days following middle cerebral artery occlusion, and it was preceded by IL-23 in a process that is in part mediated by the inflammasome. 35 Thus, it is possible that IL-12/IL-23p40 in plasma initiates a cascade that leads to systemic effects via the increased production of IL-17.
IL-1 receptor signaling also induces angiogenesis. 36 Following AIS, angiogenesis may act to restore blood flow to the infarcted region in order to preserve or restore neurovascular function. 6 Proteins regulating angiogenesis include VEGF, platelet-derived growth factor (PDGF), transforming growth factor (TGF) and bFGF, among others.37–39 Delayed treatment with VEGF activate angiogenesis that supports tissue repair in the penumbra or increase neuronal survival. However, excessive VEGF levels, particularly early after infarction, adversely affect outcomes by increasing vascular permeability, resulting in increased interstitial edema and intracranial hypertension. 40 Furthermore, increased vascular permeability at the blood brain barrier can damage neurons directly via disturbance of ion hemostasis and influx of inflammatory cells. Here we measured the protein levels of three isoforms of VEGF (-A, -C and -D). VEGF-A is involved in promoting angiogenesis associated with atherosclerotic arteries, 41 and was significantly elevated in the clot when compared to the CP. VEGF-C has been shown to be expressed in the subventricular zone, resulting in increased blood vessel diameter 42 and may provide hippocampal tolerance after ischemia. 43 We found that VEGF-C was elevated in thrombi when compared to CP. However, VEGF-D expression, which is involved in dendrite arborization, 44 was higher in the PP and CP when compared to the thrombus. These findings indicate that VEGF-A and VEGF-C are elevated in thrombi, and VEGF-D is elevated in plasma, suggesting a central role in the brain for VEGF-A and -C after ischemia, and a potential role for VEGF-D in the systemic effects associated with AIS.
bFGF is another key protein involved in the upregulation of VEGF in vascular endothelial cells. 45 bFGF appears to have a synergistic effect with VEGF on angiogenesis in that the combination of VEGF and bFGF induces more angiogenesis than VEGF alone as determined by an increase in collateral circulation upon the combined administration of these two proteins. 46 In addition, bFGF has also been shown to enhance functional recovery associated with increased neuronal sprouting in rodents following cerebral ischemia. 47 Here we detected higher levels of bFGF in the clot, consistent with the expression of VEGF-A and VEGF-C, suggesting a synergistic role for these proteins at the site of the thrombus. VEGF also induces increased expression of the angiopoietin receptor Tie-2 in endothelial cells after stroke. 48 Tie-2 binds angiopoietin-1 and -2 and enhances angiogenesis by signaling endothelial cell migration 49 and survival. 50 In addition, VEGF is capable of activating Tie-2, independently of Tie ligands. 48 Here we found that Tie-2 was elevated in the CP and the PP when compared to the clot, suggesting a potential downstream effect of VEGF on cerebral and peripheral endothelial cells after stroke.
Flt-1, also known as VEGF receptor-1, is most strongly expressed in vascular endothelial cells and binds VEGFA, VEGF-B, and PIGF (a VEGF family member). It has been shown to be upregulated in neurons and vascular cells in peri-infarct regions. Flt-1 and PIGF have a potential role in angiogenesis and inflammation. PIGF is associated with atherosclerosis and plaque rupture, while an anti-Flt-1 antibody suppressed neovascularization in tumors and reduced atherosclerotic plaque growth. In this study we found that Flt-1 and PIGF were elevated in the CP.
Outcomes after stroke are heavily influenced by the balance between pro-inflammatory and anti-inflammatory cytokines. In this study, we also analyzed anti-inflammatory cytokines. We detected an increase in the anti-inflammatory cytokine IL-5 in the cerebral and PP when compared to the clot. Higher levels of GM-CSF were detected in the peripheral circulation when compared to the CP, while the clot presented higher levels of IL-4 and IL-13. Although not statistically significant, a trend of higher levels of IL-10 were also detected in the clots. Low plasma levels of IL-10 in peripheral circulation have been associated with subsequent stroke risk and increased mortality due to stroke. 51 Another study has shown that lower IL-10 expression correlates with neurological deterioration in subcortical and lacunar infarcts. 52 IL-4 has been shown to be neuroprotective in animal models of stroke, 53 and IL-10, IL-4, IL-5 and GM-CSF are elevated in the serum of patients with favorable outcomes following AIS. 5 Our data show the anti-inflammatory cytokine profile for these cytokines in different compartments indicating a differential expression of the anti-inflammatory cytokine environment in the clots, PP and CP of AIS patients.
It has been previously shown that the physiological state of the cerebral tissue in patients presenting small to moderate ischemic core, reperfusion has the potential to preserve penumbral tissue by reducing edema. 54 Thus, it is possible that a decrease in the inflammatory response in the cerebral circulation, perhaps following reperfusion, offers a potential explanation as to the neuroprotective role associated with reperfusion. Moreover, previous studies indicate that favorable tissue level collateral profile on hypoperfusion imaging correlate with a decrease in ischemic lesion net water uptake, resulting in improved functional outcomes. 55
Furthermore, we have recently shown that IL-1β levels in clots contribute to the number of passes to achieve complete recanalization; 13 suggesting that the presence of an elevated inflammatory response affects recanalization. Similarly, it has been shown that there is an association between lower hypoperfusion intensity ratio and early post-thrombolysis recanalization in which early recanalization is also determined by ischemia severity. 56 Thus, it is likely that the presence of an exacerbated inflammatory response in the cerebral circulation contributes to the severity of the ischemic core, which is not necessarily reflected by the cytokine and angiogenic profile in the peripheral circulation given the regional differences among the proteins presented in this study.
The majority of studies of retrieved clots have reported the structural variability of clots and evaluated the concentration of fibrin, red blood cells, neutrophils and platelets. 57 Our results show that cytokines mediating the inflammatory response and factors regulating the angiogenic response are differentially expressed in thrombi and within the cerebral and peripheral circulation. Therefore, the inflammatory and angiogenic response following AIS varies in plasma obtained from different locations of the circulation and largely varies from that found in thrombi. However, the mechanisms that govern this differential protein expression profile remains to be determined. In addition, it is important to highlight that the inflammatory and angiogenic proteins analyzed in this study are not specific to stroke. However, they are an indicator of the inflammatory and angiogenic profile that is present in the CP and PP after AIS. Proteins analyzed in this study have the potential to represent a signature panel of the inflammatory and angiogenic response after stroke. Furthermore, although all samples were obtained by thrombectomy within a few hours after AIS, it is possible that the protein levels analyzed in this study may differ from patient to patient due to slight variation in the time course at which thrombectomy was performed between patients. However, given the nature that all these patients presented AIS, then major changes in protein levels is not expected; however, those changes may explain the variabilities (SEM) present for some of the analytes. Furthermore, since sex differences have been described in stroke outcomes58–60 and inflammation has been shown to differ between male and females, 15 in this study we analyzed samples from 17 males and 23 females. Correlation analyses between sex and the protein levels of the different analytes did not show a significant effect. However, similar studies powered for the study of sexual dimorphism should be considered in the future.
The linear regression models fit in this study indicate that bFGF, IL-5, TNF-α, IL-7, IL-15, IL-12/IL-23p40, IFN-γ and IL-6 in the peripheral circulation (PP) can be used to predict protein levels in the cerebral circulation (CP). Thus, identification of associations between peripheral and cerebral circulation could allow for the development of nomograms that may be used to estimate the cerebral inflammatory and angiogenic profile by measurement of these proteins in the peripheral circulation. However, in this study, in addition to fitting linear models, we similarly fit random forest models for regression (data not shown). However, care must be taken when interpreting these predictive models since they were fit using a small sample size. Thus, future studies should examine the use of machine learning approaches to determine the inflammatory and angiogenic composition of the cerebral circulation using markers in the peripheral circulation as surrogates with a larger sample size and with the use of multiple analytes as explanatory variables of cerebral inflammation and angiogenesis.
Moreover, in this study, it is possible that the difference between CP and PP may be due to poor vascular clearance of proteins released from the cerebral vasculature rather than inherent differences in the synthesis and release of these proteins after thrombotic occlusion, resulting in protein build up at the site of occlusion. In addition, since placement and manipulation of the thrombectomy device damages the cerebral endothelium, thus causing an acute damage, it is possible that this damage may contribute to the protein profile detected in the PP, instead of this protein expression profile being due to the thrombotic ischemic event itself. Future studies are necessary to determine if the inflammatory protein expression is the cause for the formation of emboli or a subsequent result of an emboli lodging within the vasculature causing ischemia. Taken together, our data indicate that a careful characterization in different compartments of the circulatory system and the protein composition of the clot is needed for the development of biomarkers for AIS. 61 Moreover, since it remains a challenge to obtain CP from healthy individuals, which can then be used to compare the levels of inflammatory and angiogenic proteins between CP and PP, future studies in animal models of cerebral ischemia and naïve mice have the potential to identify regional differences in protein expression patterns in the CP and the PP. In addition, these studies have the potential of providing insights regarding the mechanism by which these inflammatory and angiogenic proteins interact with each other to contribute to the ischemic core and the ischemic penumbra.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received grant funding from the Robert J. Dempsey Cerebrovascular Research Award to SHC and JPdRV, an R01 grant from the NIH/NINDS to RWK and JPdRV (R01NS113969-01) and a James and Ester King Biomedical Research Program grant from the State of Florida (7JK03) to WDD. RMS research is supported by the NREF, Joe Niekro Foundation, Brain Aneurysm Foundation, Bee Foundation, and by the NIH (R01NS111119-01A1) and (UL1TR002736, KL2TR002737) through the Miami Clinical and Translational Science Institute, from the National Center for Advancing Translational Sciences and the National Institute on Minority Health and Health Disparities.
Acknowledgements
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JPdRV, RWK and WDD are co-founders and managing members of InflamaCORE, LLC and have patents on inflammasome proteins as biomarkers of injury and disease as well as on targeting inflammasome proteins for therapeutic purposes. JPdRV, RWK and WDD are scientific advisory board members of ZyVersa Therapeutics. ECP is a consultant for Stryker Neurovascular, Penumbra, Medtronic Neurovascular and Cerenovus, as well as a stockholder for RIST Neurovascular. YD is a consultant for Medtronic Neurovascular, Cerenovus, Rapid Medical and Neuralanalytics. RMS has consulting and teaching agreements with Penumbra, Abbott, Medtronic, InNeuroCo and Cerenovus.
Authors’ contributions
SHC, DY, ECP, and RMS were involved in the collection of the samples. XOS, SHC, RH and JPdRV were involved in the analysis of the samples. XOS, SHC, RH, DY, ECP, RMS, WDD, RWK and JPdRV were involved in the interpretation of the data, and XOS, SHC, RH, DY, ECP, RMS, WDD, RWK and JPdRV were involved in the preparation of the manuscript. OS, SHC, RH, DY, ECP, RMS, WDD, RWK and JPdRV approved the final version of this manuscript.
